Submitted:
17 October 2025
Posted:
27 October 2025
You are already at the latest version
Abstract
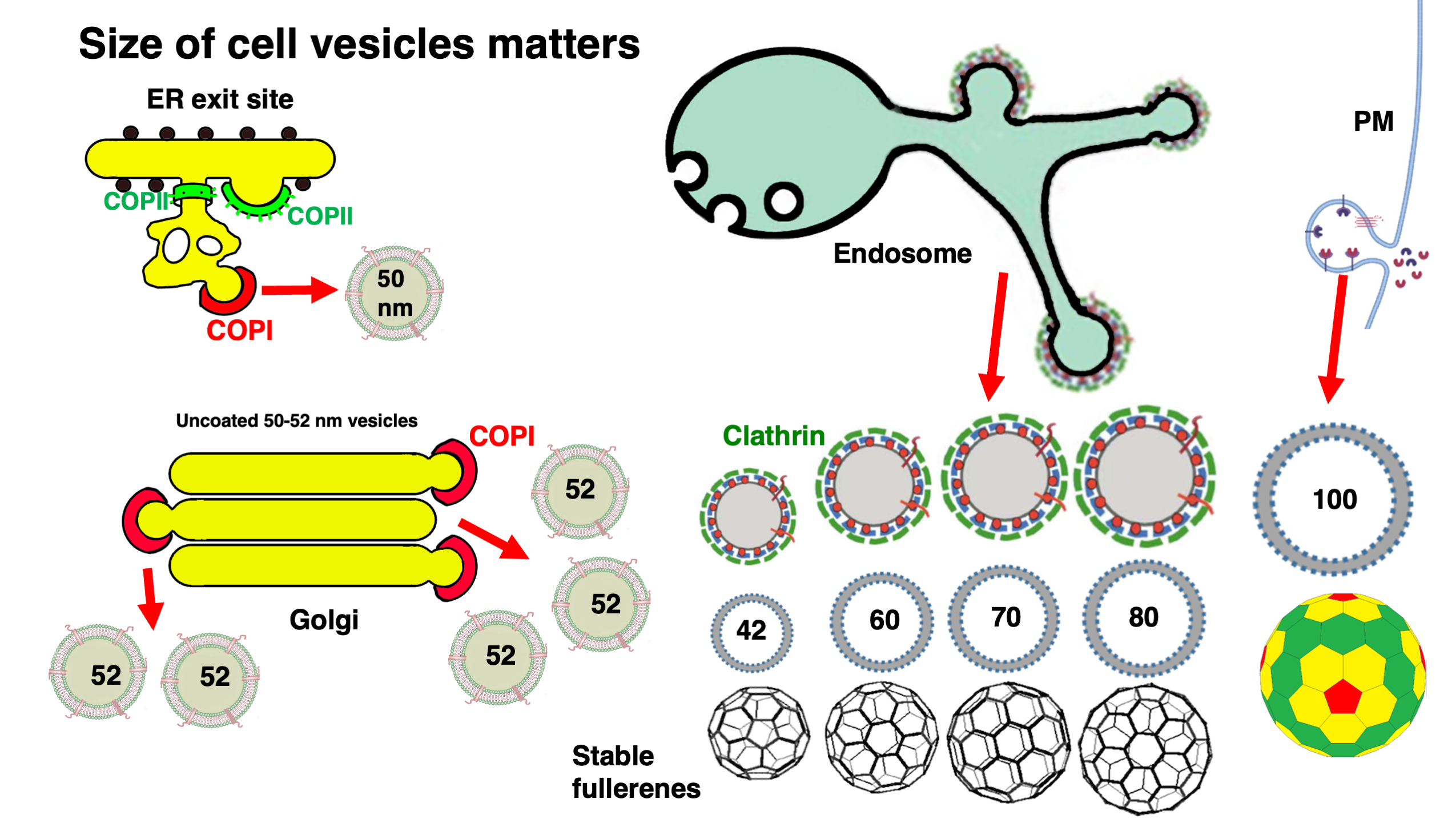
Keywords:
1. Introduction
2. Material and Methods
2.1. Cells
2.2. Antibodies
2.3. Constructs
2.4. Cell Culture and Their Permeabilization
2.5. Isolation of Golgi Membranes
2.6. Isolation of Vesicles
2.8. Microscopies
2.9. Stereology
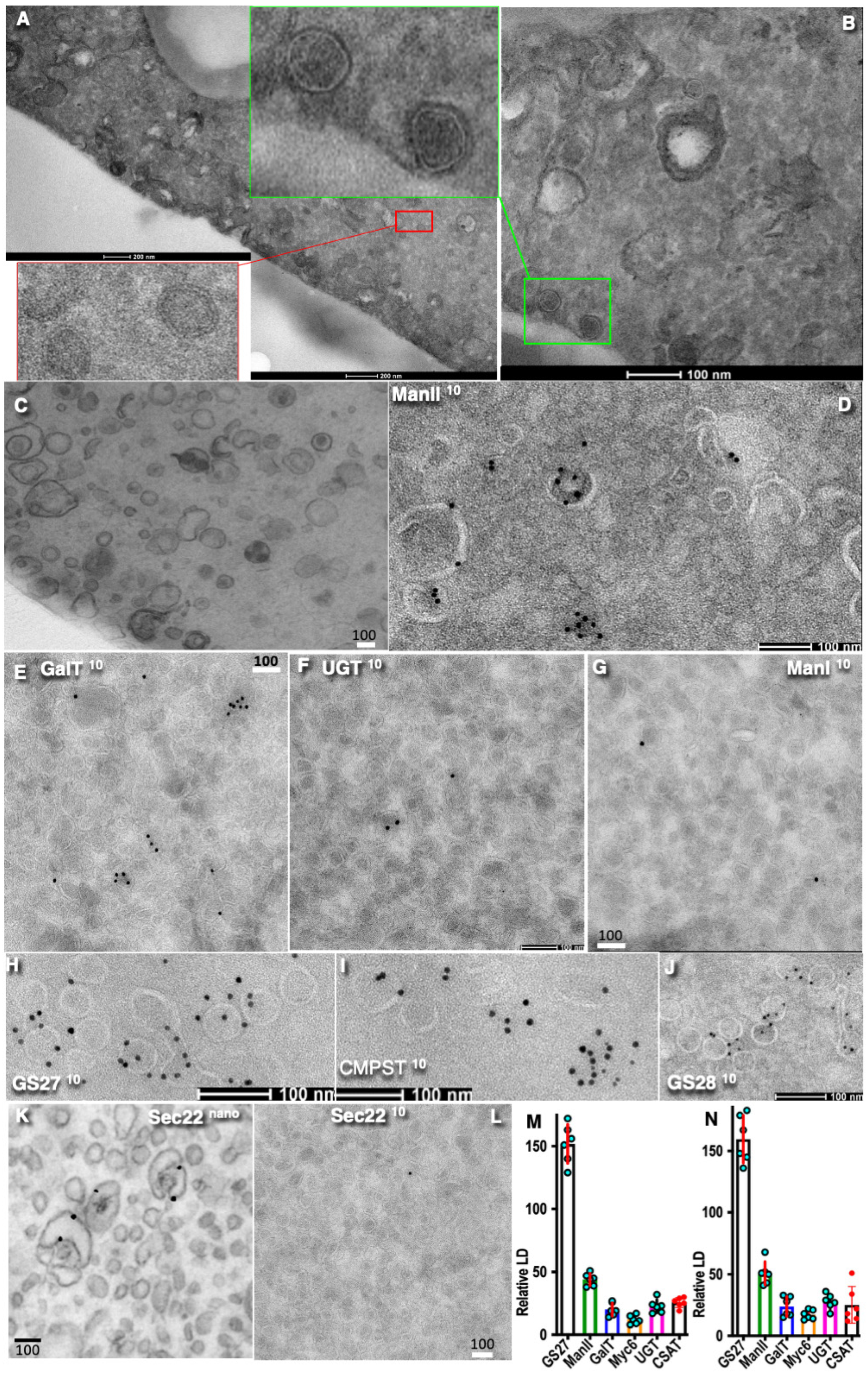
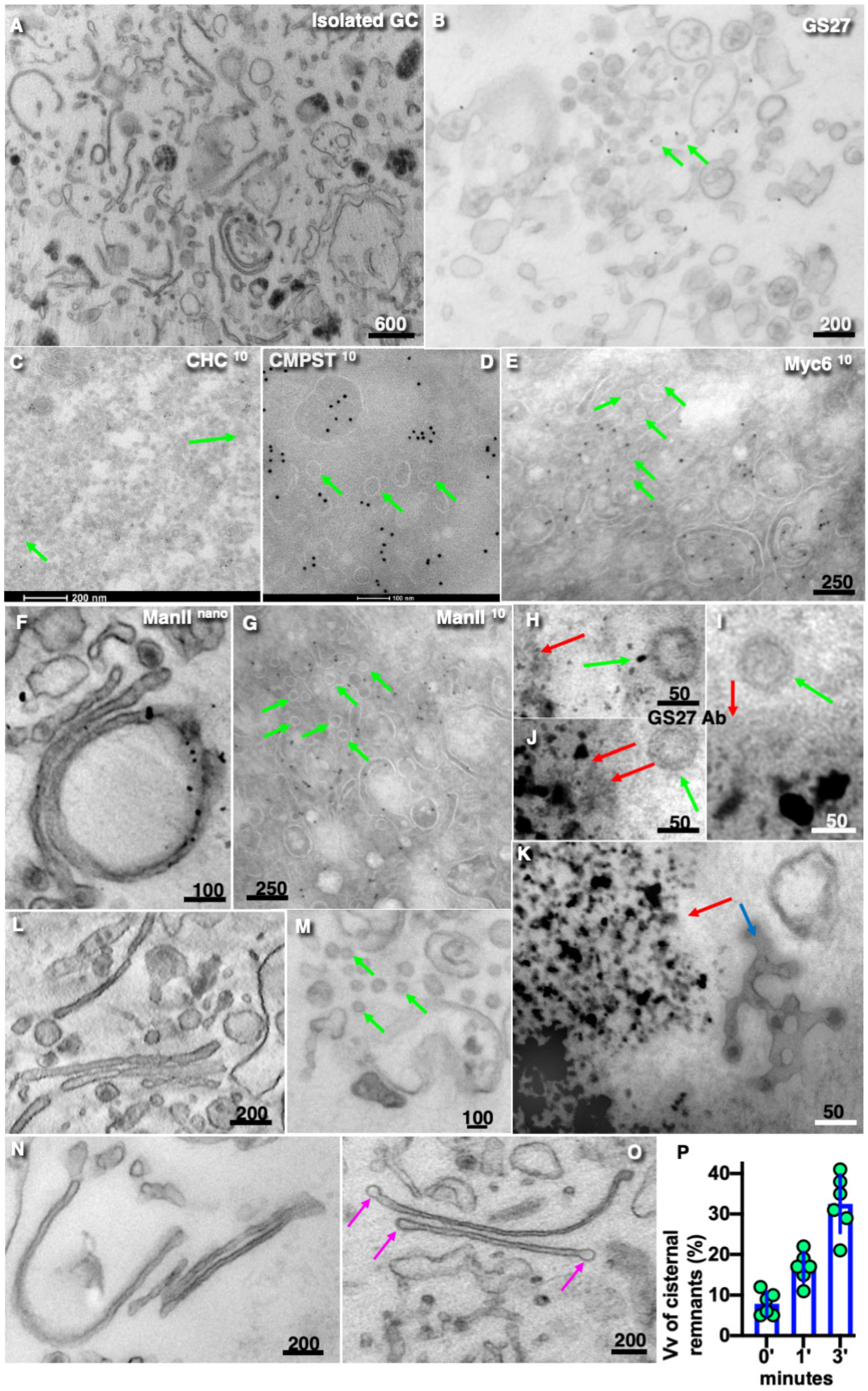
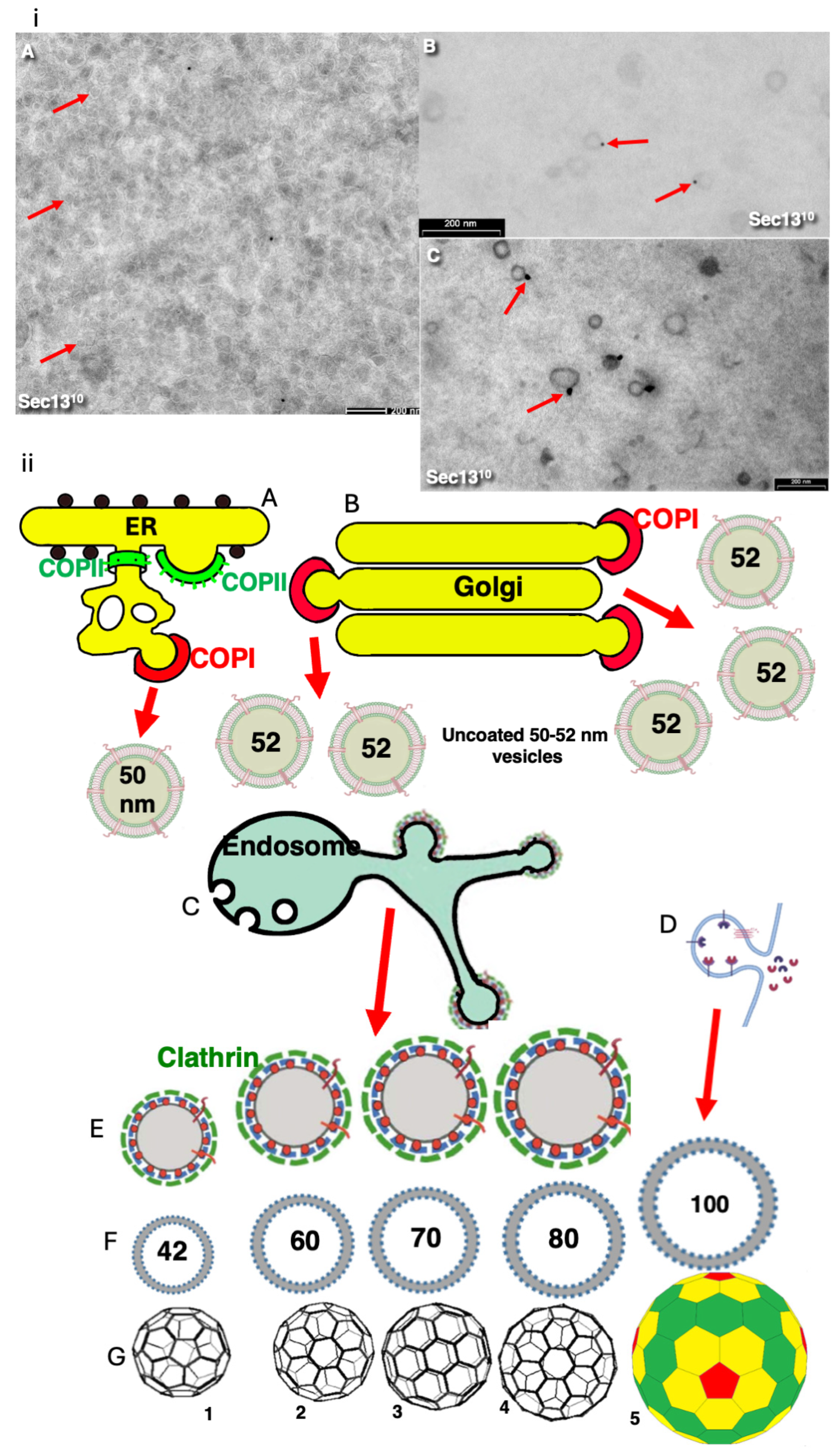
3. Results
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aridor M, Bannykh SI, Rowe T, Balch WE. Sequential coupling between COPII and COPI vesicle coats in endoplasmic reticulum to Golgi transport. J Cell Biol. 1995 Nov;131(4):875-893. [CrossRef]
- Aridor M, Bannykh SI, Rowe T, Balch WE. 1999. Cargo can modulate COPII vesicle formation from the endoplasmic reticulum. J Biol Chem 274:4389-4399. [CrossRef]
- Allan, B.B., Moyer, B.D., and Balch W.E. 2000. Rab1 recruitment of p115 into a cis-SNARE complex: programming budding COPII vesicles for fusion. Science. 289:444–448.
- Aridor, M., and Balch, W. E. (2000). Kinase signaling initiates coat complex II (COPII) recruitment and export from the mammalian endoplasmic reticulum. J. Biol. Chem. 275, 35673–35676. [CrossRef]
- Aridor M, Fish KN, Bannykh S, Weissman J, Roberts TH, Lippincott-Schwartz J, and Balch WE. 2001. The Sar1 GTPase coordinates biosynthetic cargo selection with endoplasmic reticulum export site assembly. J Cell Biol. 152(1):213-229.
- Ahluwalia, J.P., Topp, J.D., Weirather, K., Zimmerman, M., and Stamnes M. 2001. A role for calcium in stabilizing transport vesicle coats. J. Biol. Chem. 276(36):34148-34155.
- McMahon HT, Mills IG. COP and clathrin-coated vesicle budding: different pathways, common approaches. Curr Opin Cell Biol. 2004 Aug;16(4):379-91. [CrossRef]
- Adolf F, Herrmann A, Hellwig A, Beck R, Brügger B, Wieland FT. Scission of COPI and COPII vesicles is independent of GTP hydrolysis. Traffic. 2013 Aug;14(8):922-932. [CrossRef]
- Andika IB, Zheng S, Tan Z, Sun L, Kondo H, Zhou X, Chen Endoplasmic reticulum export and vesicle formation of the movement protein of Chinese wheat mosaic virus are regulated by two transmembrane domains and depend on the secretory pathway. J. Virology. 2013 Jan 20;435(2):493-503. [CrossRef]
- Gomez-Navarro N, Miller EA. COP-coated vesicles. Curr Biol. 2016 Jan 25;26(2):R54-R57. [CrossRef]
- Arakel EC, Schwappach B. Formation of COPI-coated vesicles at a glance. J Cell Sci. 2018 Mar 13;131(5):jcs209890. [CrossRef]
- Taylor RJ, Tagiltsev G, Briggs JAG. FEBS Lett. 2023 Mar;597(6):819-835. [CrossRef]
- Arab M, Chen T, Lowe M. Mechanisms governing vesicle traffic at the Golgi apparatus. Curr Opin Cell Biol. 2024 May 4;88:102365. [CrossRef]
- Johnson A. Mechanistic divergences of endocytic clathrin-coated vesicle formation in mammals, yeasts and plants. J Cell Sci. 2024 Aug 15;137(16):jcs261847. [CrossRef]
- Viotti C. ER to Golgi-Dependent Protein Secretion: The Conventional Pathway. Methods Mol Biol. 2016;1459:3-29. [CrossRef]
- Bacia K, Futai E, Prinz S, Meister A, Daum S, Glatte D, Briggs JA, Schekman R. Multibudded tubules formed by COPII on artificial liposomes. Sci Rep. 2011, 1:17. [CrossRef]
- Bykov YS, Schaffer M, Dodonova SO, Albert S, Plitzko JM, Baumeister W, Engel BD, Briggs JA. 2017. The structure of the COPI coat determined within the cell. Elife. 6. pii: e32493. [CrossRef]
- Pyle E, Miller EA, Zanetti G. Cryo-electron tomography reveals how COPII assembles on cargo-containing membranes. Nat Struct Mol Biol. 2025 Mar;32(3):513-519. [CrossRef]
- Chung KP, Frieboese D, Waltz F, Engel BD, Bock R. Identification and characterization of the COPII vesicle-forming GTPase Sar1 in Chlamydomonas. Plant Direct. 2024 Jun 16;8(6):e614. [CrossRef]
- Mironov AA, Beznoussenko GV. Models of intracellular transport: Pros and cons. Front Cell Dev Biol. 2019, 7, 146. [CrossRef]
- Mironov, A. A.; Fusella, A.; Beznoussenko, G. V. The 50-nm Free Vesicles Visible in Saccharomyces cerevisiae Are Not COPII-Dependent. Current Issues in Molecular Biology, 2025, 47, 336. [CrossRef]
- Holzman, E. Cytochemical studies of protein transport in the nervous system. Philos. Trans. R. Soc. Lond. 1971, 261, 407–421. [CrossRef]
- Friend DS, Farquhar MG. Functions of coated vesicles during protein absorption in the rat vas deferens. J Cell Biol. 1967 Nov;35(2):357-76. [CrossRef]
- Griffiths G, Fuller SD, Back R, Hollinshead M, Pfeiffer S, Simons K. 1989. The dynamic nature of the Golgi complex. J Cell Biol 108:277-297.
- Klumperman, J., Hille, A., Veenendaal, T., Oorschot, V., Stoorvogel, W., von Figura, K., and Geuze, H.J. (1993). Differences in the endosomal distributions of the two mannose 6-phosphate receptors. J Cell Biol 121, 997-1010.
- Le Borgne, R., Griffiths, G., and Hoflack, B. (1996). Mannose 6-phosphate receptors and ADP-ribosylation factors cooperate for high affinity interaction of the AP-1 Golgi assembly proteins with membranes. J Biol Chem 271, 2162-2170.
- Bonfanti L, Mironov AA Jr, Martínez-Menárguez JA, Martella O, Fusella A, Baldassarre M, Buccione R, Geuze HJ, Mironov AA, Luini A. 1998. Procollagen traverses the Golgi stack without leaving the lumen of cisternae: evidence for cisternal maturation. Cell. 95(7):993-1003. [CrossRef]
- Martínez-Menárguez, J. A., Geuze, H. J.; Ballesta, J. Identification of two types of beta-COP vesicles in the Golgi complex of rat spermatids. Eur. J. Cell. Biol. 1996, 71, 137–143.
- Stoorvogel, W., Oorschot, V., and Geuze, H.J. 1996. A novel class of clathrin-coated vesicles budding from endosomes. J. Cell Biol. 132:21-33.
- Ladinsky, M. S., Mastronarde, D. N., McIntosh, J. R., Howell, K. E., and Staehelin, L. A. Golgi structure in three dimensions, functional insights from the normal rat kidney cell. J. Cell. Biol. 1999, 144, 1135–1149. [CrossRef]
- Puri C, Tosoni D, Comai R, Rabellino A, Segat D, Caneva F, Luzzi P, Di Fiore PP, Tacchetti C. Relationships between EGFR signaling-competent and endocytosis-competent membrane microdomains. Mol Biol Cell. 2005 Jun;16(6):2704-18. [CrossRef]
- Beznoussenko GV, Ragnini-Wilson A, Wilson C, Mironov AA. 2016. Three-dimensional and immune electron microscopic analysis of the secretory pathway in Saccharomyces cerevisiae. Histochem Cell Biol. 146(5):515-527. [CrossRef]
- Mironov AA, Sesorova IS, Seliverstova EV, Beznoussenko GV. 2017. Different Golgi ultrastructure across species and tissues: Implications under functional and pathological conditions, and an attempt at classification. Tissue Cell 2017, 49 (2 Pt A):186-201. [CrossRef]
- Mironov AA, Beznoussenko GV. The Regulated Secretion and Models of Intracellular Transport: The Goblet Cell as an Example. Int J Mol Sci. 2023 May 31;24(11):9560. [CrossRef]
- Fusella, A., Micaroni, M., Di Giandomenico, D., Mironov, A. A., and Beznoussenko, G. V. Segregation of the Qb-SNAREs GS27 and GS28 into Golgi vesicles regulates intra-Golgi transport. Traffic 2013, 14, 568–584. [CrossRef]
- Bomsel M, de Paillerets C, Weintraub H, Alfsen A. Lipid bilayer dynamics in plasma and coated vesicle membranes from bovine adrenal cortex. Evidence of two types of coated vesicle involved in the LDL receptor traffic. Biochim Biophys Acta. 1986 Jul 10;859(1):15-25. [CrossRef]
- Avinoam O, Schorb M, Beese CJ, Briggs JA, Kaksonen M. ENDOCYTOSIS. Endocytic sites mature by continuous bending and remodelling of the clathrin coat. Science. 2015 Jun 19;348(6241):1369-72. [CrossRef]
- Mettlen M, Chen PH, Srinivasan S, Danuser G, Schmid SL. Regulation of Clathrin-Mediated Endocytosis. Annu Rev Biochem. 2018 Jun 20;87:871-896. [CrossRef]
- Shevchuk AI, Hobson P, Lab MJ, Klenerman D, Krauzewicz N, Korchev YE. Endocytic pathways: combined scanning ion conductance and surface confocal microscopy study. Pflugers Arch. 2008 Apr;456(1):227-35. [CrossRef]
- Diao A, Rahman D, Pappin DJ, Lucocq J, Lowe M. The coiled-coil membrane protein golgin-84 is a novel rab effector required for Golgi ribbon formation. J Cell Biol. 2003 Jan 20;160(2):201-212. [CrossRef]
- Peters A, Palay SL, Webster H. Fine Structure of the Nervous System: Neurons and Their Supporting Cells. Oxford, New York Toronto. Oxford University Press. 3 edition. (1991) 528 pages.
- Beznoussenko GV, Kweon HS, Sesorova IS, Mironov AA. Comparison of the Cisterna Maturation-Progression Model with the Kiss-and-Run Model of Intra-Golgi Transport: Role of Cisternal Pores and Cargo Domains. Int. J. Mol. Sci. 2022, 23, 3590. [CrossRef]
- Sumya FT, Pokrovskaya ID, D’Souza Z, Lupashin VV. Acute COG complex inactivation unveiled its immediate impact on Golgi and illuminated the nature of intra-Golgi recycling vesicles. Traffic. 2023 Feb;24(2):52-75. [CrossRef]
- Waters, M.G., Clary, D.O., and Rothman, J.E. 1992. A novel 115-kD peripheral membrane protein is required for intercisternal transport in the Golgi stack, J. Cell Biol. 118:1015–1026.
- Ostermann J, Orci L, Tani K, Amherdt M, Ravazzola M, Elazar Z, Rothman JE. Stepwise assembly of functionally active transport vesicles. Cell. 1993 Dec 3;75(5):1015-1025. [CrossRef]
- Williger BT, Ostermann J, Exton JH. Arfaptin 1, an ARF-binding protein, inhibits phospholipase D and endoplasmic reticulum/Golgi protein transport. FEBS Lett. 1999 Jan 25;443(2):197-200. [CrossRef]
- Love HD, Lin CC, Short CS, Ostermann J. Isolation of functional Golgi-derived vesicles with a possible role in retrograde transport. J Cell Biol. 1998;140(3):541-551. [CrossRef]
- Lanoix J, Ouwendijk J, Lin CC, Stark A, Love HD, Ostermann J, Nilsson T. GTP hydrolysis by arf-1 mediates sorting and concentration of Golgi resident enzymes into functional COP I vesicles. EMBO J. 1999 Sep 15;18(18):4935-48. [CrossRef]
- Gilchrist, A., Au, C. E., Hiding, J., Bell, A. W., Fernandez-Rodriguez, J., Lesimple, S., et al. (2006). Quantitative proteomics analysis of the secretory pathway. Cell 127, 1265–1281. [CrossRef]
- Malsam J, Gommel D, Wieland FT, Nickel W. 1999. A role for ADP ribosylation factor in the control of cargo uptake during COPI-coated vesicle biogenesis. FEBS Lett. 462(3):267-272.
- Adolf F, Rhiel M, Hessling B, Gao Q, Hellwig A, Béthune J, Wieland FT. Proteomic Profiling of Mammalian COPII and COPI Vesicles. Cell Rep. 2019 Jan 2;26(1):250-265.e5. [CrossRef]
- Adolf F, Rhiel M, Reckmann I, Wieland FT. Sec24C/D-isoform-specific sorting of the preassembled ER-Golgi Q-SNARE complex. Mol Biol Cell. 2016 Sep 1;27(17):2697-707. [CrossRef]
- Adolf F, Wieland FT. Analysis of Golgi complex functions: in vitro reconstitution systems. Methods Cell Biol. 2013;118:3-14. [CrossRef]
- Adolf F, Rhiel M, Hessling B, Gao Q, Hellwig A, Béthune J, Wieland FT. Proteomic Profiling of Mammalian COPII and COPI Vesicles. Cell Rep. 2019 Jan 2;26(1):250-265.e5. [CrossRef]
- Sumya FT, Aragon-Ramirez WS, Lupashin VV. Mol Deep proteomic profiling of the intra-Golgi trafficking intermediates. Biol Cell. 2025 Jul 1;36(7):ar87. [CrossRef]
- Kweon HS, Beznoussenko GV, Micaroni M, Polishchuk RS, Trucco A, Martella O, Di Giandomenico D, Marra P, Fusella A, Di Pentima A, Berger EG, Geerts WJ, Koster AJ, Burger KN, Luini A. and Mironov AA. 2004. Golgi enzymes are enriched in perforated zones of golgi cisternae but are depleted in COPI vesicles. Mol Biol Cell. 15:4710-4724. [CrossRef]
- Beznoussenko GV, Bejan AI, Parashuraman S, Luini A, Kweon H.-S.; Mironov AA. The Diffusion Model of Intra-Golgi Transport Has Limited Power. Int J Mol Sci. 2023 Jan 10;24(2):1375. [CrossRef]
- Mironov A.A., Colanzi A., Polishchuk R.S., Beznoussenko G.V., Mironov A.A. Jr., Fusella A., Di Tullio G., Silletta M.G., Corda D., De Matteis M.A. and Luini A. 2004. Dicumarol, an inhibitor of ADP-ribosylation of CtBP3/BARS, fragments Golgi non-compact tubular zones and inhibits intra-Golgi transport. Eur. J. Cell Biol. 83:263-279.
- Ha VL, Thomas GM, Stauffer S, Randazzo PA. Preparation of myristoylated Arf1 and Arf6. Methods Enzymol. 2005;404:164-74. [CrossRef]
- Godi A, Santone I, Pertile P, Devarajan P, Stabach PR, Morrow JS, Di Tullio G, Polishchuk R, Petrucci TC, Luini A, De Matteis MA. ADP ribosylation factor regulates spectrin binding to the Golgi complex. Proc Natl Acad Sci U S A. 1998 Jul 21;95(15):8607-12. [CrossRef]
- Elkin SR, Oswald NW, Reed DK, Mettlen M, MacMillan JB, Schmid SL. Ikarugamycin: A Natural Product Inhibitor of Clathrin-Meiated Endocytosis. Traffic. 2016 Oct;17(10):1139-49. [CrossRef]
- Barnard RJ, Morgan A. and Burgoyne RD. 1996. Domains of alpha-SNAP required for the stimulation of exocytosis and for N-ethylmalemide-sensitive fusion protein (NSF) binding and activation. Mol Biol Cell. 7:693-701.
- German CL, Howe CL. Preparation of biologically active subcellular fractions using the Balch homogenizer. Anal Biochem. 2009 Nov 1;394(1):117-24. [CrossRef]
- Beznoussenko GV, Pilyugin SS, Geerts WJ, Kozlov MM, Burger KN, Luini A, Derganc J, Mironov AA. 2015. Trans-membrane area asymmetry controls the shape of cellular organelles. Int J Mol Sci. 16(3):5299-5333. [CrossRef]
- Yang JS, Lee SY, Gao M, Bourgoin S, Randazzo PA, Premont RT, Hsu VW. 2002. ARFGAP1 promotes the formation of COPI vesicles, suggesting function as a component of the coat. J Cell Biol. 159(1):69-78.
- Marsh, B. J., Mastronarde, D. N., Buttle, K. F., Howell, K. E., and McIntosh, J. R. (2001). Organellar relationships in the Golgi region of pancreatic beta cell line, HIT-T15, visualized by high resolution electron tomography. Proc. Natl. Acad. Sci. U.S.A. 98, 2399–2406. [CrossRef]
- 1Beznoussenko, G. V., Parashuraman, S., Rizzo, R., Polishchuk, R., Martella, O., Di Giandomenico, D., et al. (2014). Transport of soluble proteins through the Golgi occurs by diffusion via continuities across cisternae. Elife 3:e02009. [CrossRef]
- Mironov, A. A. Jr.; Mironov, A. A. Estimation of subcellular organelle volume from ultrathin sections through centrioles with a discretized version of vertical rotator. J. Microsc. 1998, 192, 29-36. [CrossRef]
- Vetterlein, M., Ellinger, A., Neumuller, J.; Pavelka, M. Golgi apparatus and TGN during endocytosis. Histochem Cell Biol. 2002, 117:143-150.
- Beznoussenko GV, Pilyugin SS, Geerts WJ, Kozlov MM, Burger KN, Luini A, Derganc J, Mironov AA. 2015. Trans-membrane area asymmetry controls the shape of cellular organelles. Int J Mol Sci. 16(3):5299-5333. [CrossRef]
- Kaiser, C. A.; Schekman, R. Distinct sets of SEC genes govern transport vesicle formation and fusion early in the secretory pathway. Cell 1990, 61, 723–733. [CrossRef]
- Barlowe, C., Orci, L., Yeung, T., Hosobuchi, M., Hamamoto, S., Salama, N., Rexach, M.F., Ravazzola, M.; Amherdt, M.; Schekman, R. COPII: a membrane coat formed by Sec proteins that drive vesicle budding from the endoplasmic reticulum. Cell 1994, 77, 895–907. [CrossRef]
- Bednarek, S. Y., Ravazzola, M., Hosobuchi, M., Amherdt, M., Perrelet, A., Schekman, R., et al. COPI- and COPII-coated vesicles bud directly from the endoplasmic reticulum in yeast. Cell 1995, 83, 1183–1196. [CrossRef]
- Sesorova IS, Karelina NR, Kazakova TE, Parashuraman S, Zdorikova MA, Dimov ID, Seliverstova EV, Beznoussenko GV, Mironov AA. 2020. Structure of the enterocyte transcytosis compartments during lipid absorption. Histochem Cell Biol. 2020 Jun;153(6):413-429. [CrossRef]
- Peters A, Palay SL, Webster H. Fine Structure of the Nervous System: Neurons and Their Supporting Cells. Oxford, New York Toronto. Oxford University Press. 3 edition. (1991) 528 pages.
- Mironov AA, Sesorova IS, Vavilov PS, Longoni R, Briata P, Gherzi R, Beznoussenko GV. Structure of the Secretory Compartments in Goblet Cells in the Colon and Small Intestine. Cells. 2025 Jul 31;14(15):1185. [CrossRef]
- Mironov AA, Sesorova IS, Vavilov PS, Longoni R, Briata P, Gherzi R, Beznoussenko GV. Structure of the Secretory Compartments in Goblet Cells in the Colon and Small Intestine. Cells. 2025 Jul 31;14(15):1185. [CrossRef]
- Orci L, Malhotra V, Amherdt M, Serafini T, Rothman JE. Dissection of a single round of vesicular transport: sequential intermediates for intercisternal movement in the Golgi stack. Cell. 1989 Feb 10;56(3):357-68. [CrossRef]
- Rexach MF, Latterich M, Schekman RW. Characteristics of endoplasmic reticulum-derived transport vesicles. J Cell Biol. 1994 Sep;126(5):1133-48. [CrossRef]
- Cosson, P.; Letourneur, F. 1994. Coatomer interaction with di-lysine endoplasmic reticulum retention motif. Science 263:1629-1631.
- Bannykh, S. I., Rowe, T.; Balch, W. E. The organization of endoplasmic reticulum export complexes. J. Cell. Biol. 1996, 135, 19–35. [CrossRef]
- Cosson P, Amherdt M, Rothman JE, Orci L. A resident Golgi protein is excluded from peri-Golgi vesicles in NRK cells. Proc Natl Acad Sci U S A. 2002 Oct 1;99(20):12831-4. [CrossRef]
- German CL, Howe CL. Preparation of biologically active subcellular fractions using the Balch homogenizer. Anal Biochem. 2009 Nov 1;394(1):117-24. [CrossRef]
- Orci, L.; Amherdt, M., Ravazzola, M., Perrelet, A., and Rothman, J. E. (2000a). Exclusion of Golgi residents from transport vesicles budding from Golgi cisternae in intact cells. J. Cell. Biol. 150, 1263–1270. [CrossRef]
- Volchuk A, Ravazzola M, Perrelet A, Eng WS, Di Liberto M, Varlamov O, Fukasawa M, Engel T, Söllner TH, Rothman JE, Orci L. Countercurrent distribution of two distinct SNARE complexes mediating transport within the Golgi stack. Mol Biol Cell. 2004 Apr;15(4):1506-18. [CrossRef]
- Jokitalo, E., Cabrera-Poch, N., Warren, G., and Shima, D.T. 2001. Golgi clusters and vesicles mediate mitotic inheritance independently of the endoplasmic reticulum. J. Cell Biol. 154(2):317-330.
- Gaietta GM, Giepmans BN, Deerinck TJ, Smith WB, Ngan L, Llopis J, Adams SR, Tsien RY, Ellisman MH. Golgi twins in late mitosis revealed by genetically encoded tags for live cell imaging and correlated electron microscopy. Proc Natl Acad Sci U S A. 2006 Nov 21;103(47):17777-82. [CrossRef]
- Martinez-Menárguez, J. A., Prekeris, R., Oorschot, V. M., Scheller, R., Slot, J. W., Geuze, H. J., et al. Peri-Golgi vesicles contain retrograde but not anterograde proteins consistent with the cisternal progression model of intra-Golgi transport. J. Cell. Biol. 2001, 155, 1213–1224. [CrossRef]
- Mandon EC, Milla ME, Kempner E, Hirschberg CB: Purification of the Golgi adenosine 30-phosphate 50-phosphosulfate transporter, a homodimer within the membrane. Proc Natl Acad Sci U S A 1994, 91:10707-10711.
- Puglielli L, Hirschberg C: Reconstitution, identification, and purification of the rat liver Golgi membrane GDP-fucose transporter. J Biol Chem 1999, 274:35596-35600.
- Puglielli, L., Mandon, E. C., Rancour, D. M., Menon, A. K., and Hirschberg, C. B. (1999) Identification and purification of the rat liver Golgi membrane UDP-N-acetylgalactosamine transporter. J Biol Chem. 274: 4474-4479.
- Parker JL, Newstead S. 2019. Gateway to the Golgi: molecular mechanisms of nucleotide sugar transporters. Curr Opin Struct Biol. 2019 Aug;57:127-134. [CrossRef]
- Maszczak-Seneczko D, Wiktor M, Skurska E, Wiertelak W, Olczak M. Delivery of Nucleotide Sugars to the Mammalian Golgi: A Very Well (un)Explained Story. Int J Mol Sci. 2022 Aug 3;23(15):8648. [CrossRef]
- Weidman P, Roth R, Heuser J. Golgi membrane dynamics imaged by freeze-etch electron microscopy: views of different membrane coatings involved in tubulation versus vesiculation. Cell 1993, 75(1), 123-133. 80089-80093.
- Mossessova, E., Bickford, L.C., and Goldberg, J. SNARE selectivity of the COPII coat. Cell. 2003, 114:483–495. [CrossRef]
- Khamatgalimov AR, Kovalenko VI. Substructural Approach for Assessing the Stability of Higher Fullerenes. Int J Mol Sci. 2021 Apr 4;22(7):3760. [CrossRef]
- Khamatgalimov AR, Idrisov RI, Kamaletdinov II, Kovalenko VI. Open-shell nature of non-IPR fullerene С40: isomers 29 (C2) and 40 (Td). J Mol Model. 2021 Jan 7;27(2):22. [CrossRef]
- den Otter WK, Briels WJ. The generation of curved clathrin coats from flat plaques. Traffic. 2011 Oct;12(10):1407-16. [CrossRef]
- Fotin A, Cheng Y, Grigorieff N, Walz T, Harrison SC, Kirchhausen T. Structure of an auxilin-bound clathrin coat and its implications for the mechanism of uncoating. Nature. 2004 Dec 2;432(7017):649-53. [CrossRef]
- Fotin A, Cheng Y, Sliz P, Grigorieff N, Harrison SC, Kirchhausen T, Walz T. Molecular model for a complete clathrin lattice from electron cryomicroscopy. Nature. 2004 Dec 2;432(7017):573-9. [CrossRef]
- Kukulski W, Schorb M, Kaksonen M, Briggs JA: 2012. Plasma membrane reshaping during endocytosis is revealed by time-resolved electron tomography. Cell. 150:508-520.
- Kukulski W, Schorb M, Welsch S, Picco A, Kaksonen M, Briggs JA. 2011. Correlated fluorescence and 3D electron microscopy with high sensitivity and spatial precision. J Cell Biol. 192(1):111-119.
- Ostermann J. 2001. Stoichiometry and kinetics of transport vesicle fusion with Golgi membranes. EMBO Rep. 2(4):324-329.
- Sonnichsen, B., Lowe, M., Levine, T., Jamsa, E., Dirac-Svejstrup, A.B., Warren, G. 1998. A role for Giantin in docking COPI vesicles to Golgi membranes. J. Cell Biol. 140:1013–1021.
- Malhotra V.; Serafini T, Orci L, Shepherd JC, Rothman JE. Purification of a novel class of coated vesicles mediating biosynthetic protein transport through the Golgi stack. Cell. 1989 Jul 28;58(2):329-36. [CrossRef]
- Adolf F, Rhiel M, Hessling B, Gao Q, Hellwig A, Béthune J, Wieland FT. Proteomic Profiling of Mammalian COPII and COPI Vesicles. Cell Rep. 2019 Jan 2;26(1):250-265.e5. [CrossRef]
- Vasile E, Oka T, Ericsson M, Nakamura N, Krieger M. IntraGolgi distribution of the Conserved Oligomeric Golgi (COG) complex. Exp Cell Res. 2006 Oct 1;312(16):3132-3141. [CrossRef]
- Eckert ES, Reckmann I, Hellwig A, Röhling S, El-Battari A, Wieland FT, Popoff V. Golgi phosphoprotein 3 triggers signal-mediated incorporation of glycosyltransferases into coatomer-coated (COPI) vesicles. J. Biol. Chem. 2014, 289, 31319-31329. [CrossRef]
- Oka T, Vasile E, Penman M, Novina CD, Dykxhoorn DM, Ungar D, Hughson FM, Krieger M. Genetic analysis of the subunit organization and function of the conserved oligomeric golgi (COG) complex: studies of COG5- and COG7-deficient mammalian cells. J Biol Chem. 2005 Sep 23;280(38):32736-45. [CrossRef]
- Bailey Blackburn J, Pokrovskaya I, Fisher P, Ungar D, Lupashin VV. 2016. COG Complex Complexities: Detailed Characterization of a Complete Set of HEK293T Cells Lacking Individual COG Subunits. Front Cell Dev Biol. 2016 Mar 30;4:23. [CrossRef]
- Shin JJH, Crook OM, Borgeaud AC, Cattin-Ortolá J, Peak-Chew SY, Breckels LM, Gillingham AK, Chadwick J, Lilley KS, Munro S. Spatial proteomics defines the content of trafficking vesicles captured by golgin tethers. Nat Commun. 2020 Nov 25;11(1):5987. [CrossRef]
- Wong M, Munro S. Membrane trafficking. The specificity of vesicle traffic to the Golgi is encoded in the golgin coiled-coil proteins. Science. 2014 Oct 31;346(6209):1256898. [CrossRef]
- Welch LG, Peak-Chew SY, Begum F, Stevens TJ, Munro S. GOLPH3 and GOLPH3L are broad-spectrum COPI adaptors for sorting into intra-Golgi transport vesicles. J Cell Biol. 2021 Oct 4;220(10):e202106115. [CrossRef]
- Rexach MF, Schekman RW. Distinct biochemical requirements for the budding, targeting, and fusion of ER-derived transport vesicles. J Cell Biol. 1991 Jul;114(2):219-29. [CrossRef]
- Ktistakis NT, Brown HA, Waters MG, Sternweis PC, Roth MG. Evidence that phospholipase D mediates ADP ribosylation factor-dependent formation of Golgi coated vesicles. J Cell Biol. 1996 Jul;134(2):295-306. [CrossRef]
- Stamnes M, Schiavo G, Stenbeck G, Söllner TH, Rothman JE. ADP-ribosylation factor and phosphatidic acid levels in Golgi membranes during budding of coatomer-coated vesicles. Proc Natl Acad Sci U S A. 1998 Nov 10;95(23):13676-80. [CrossRef]
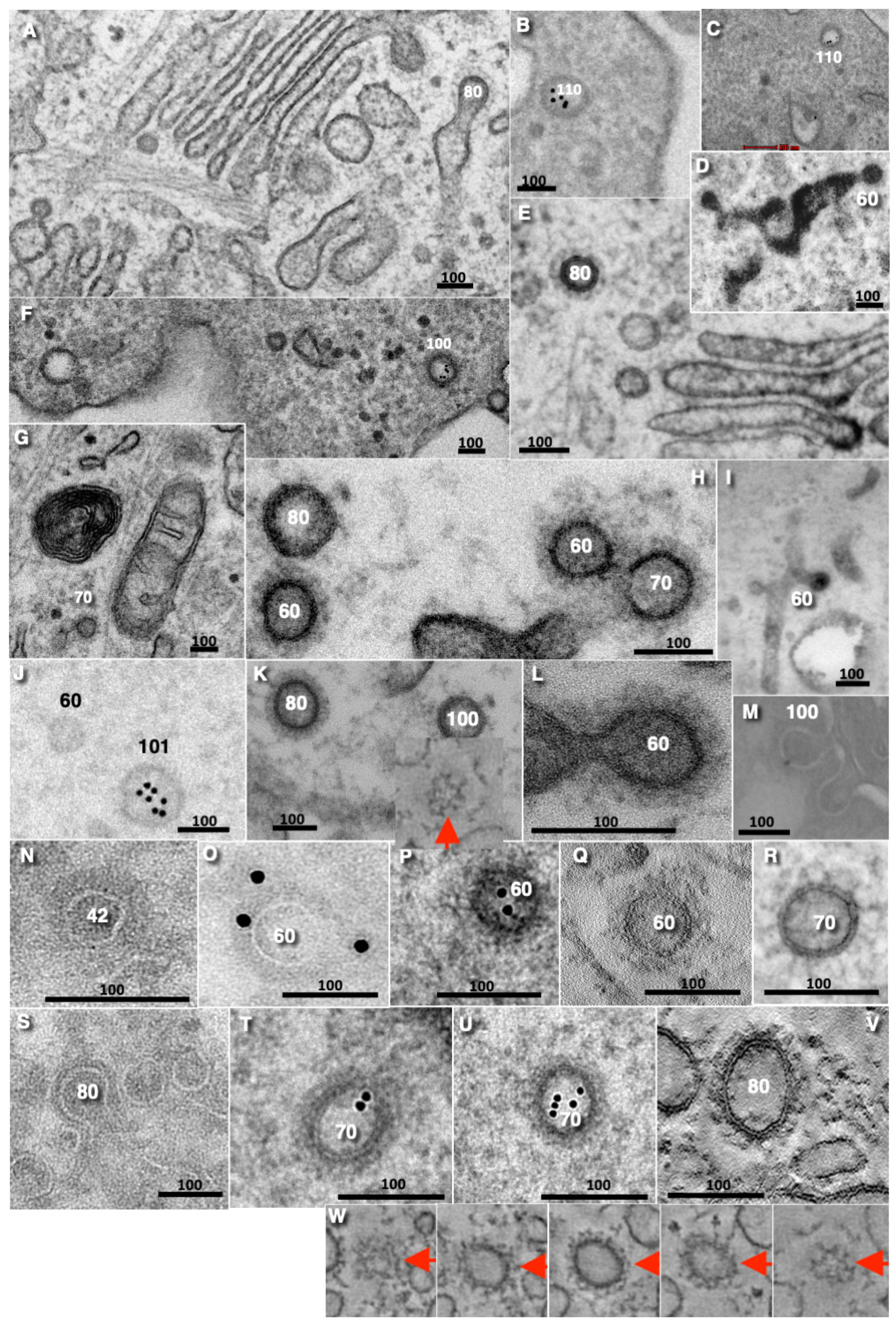
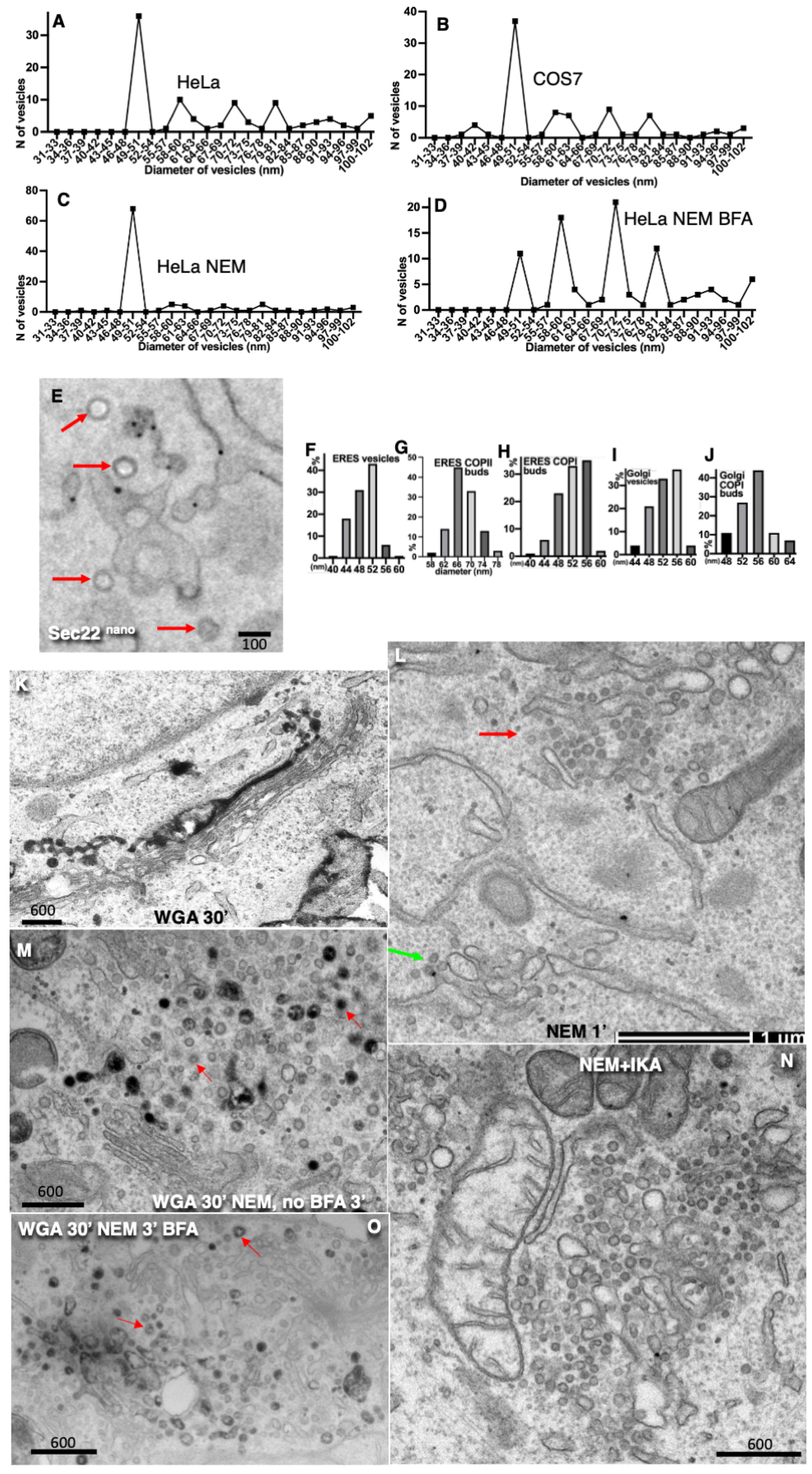
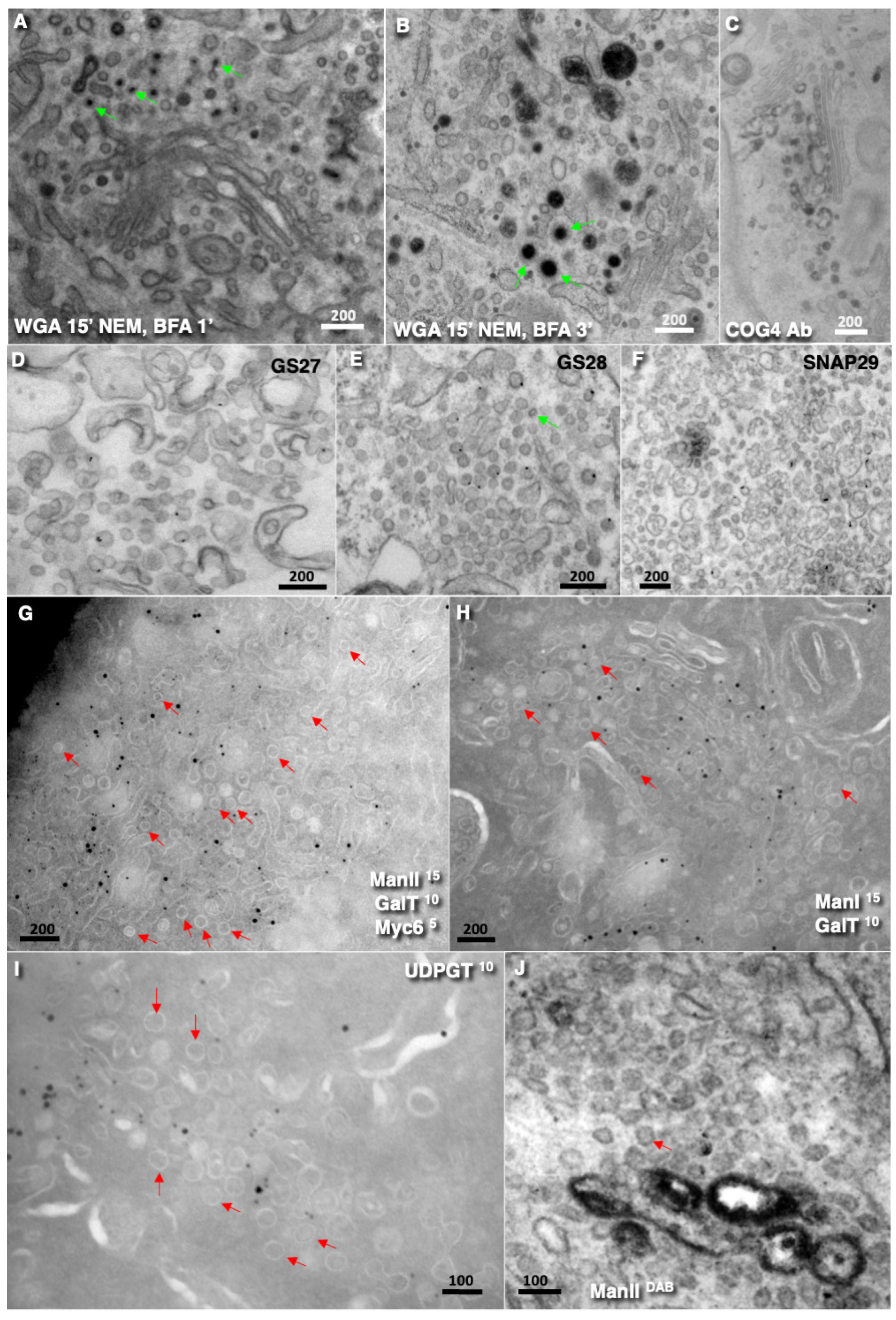
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



