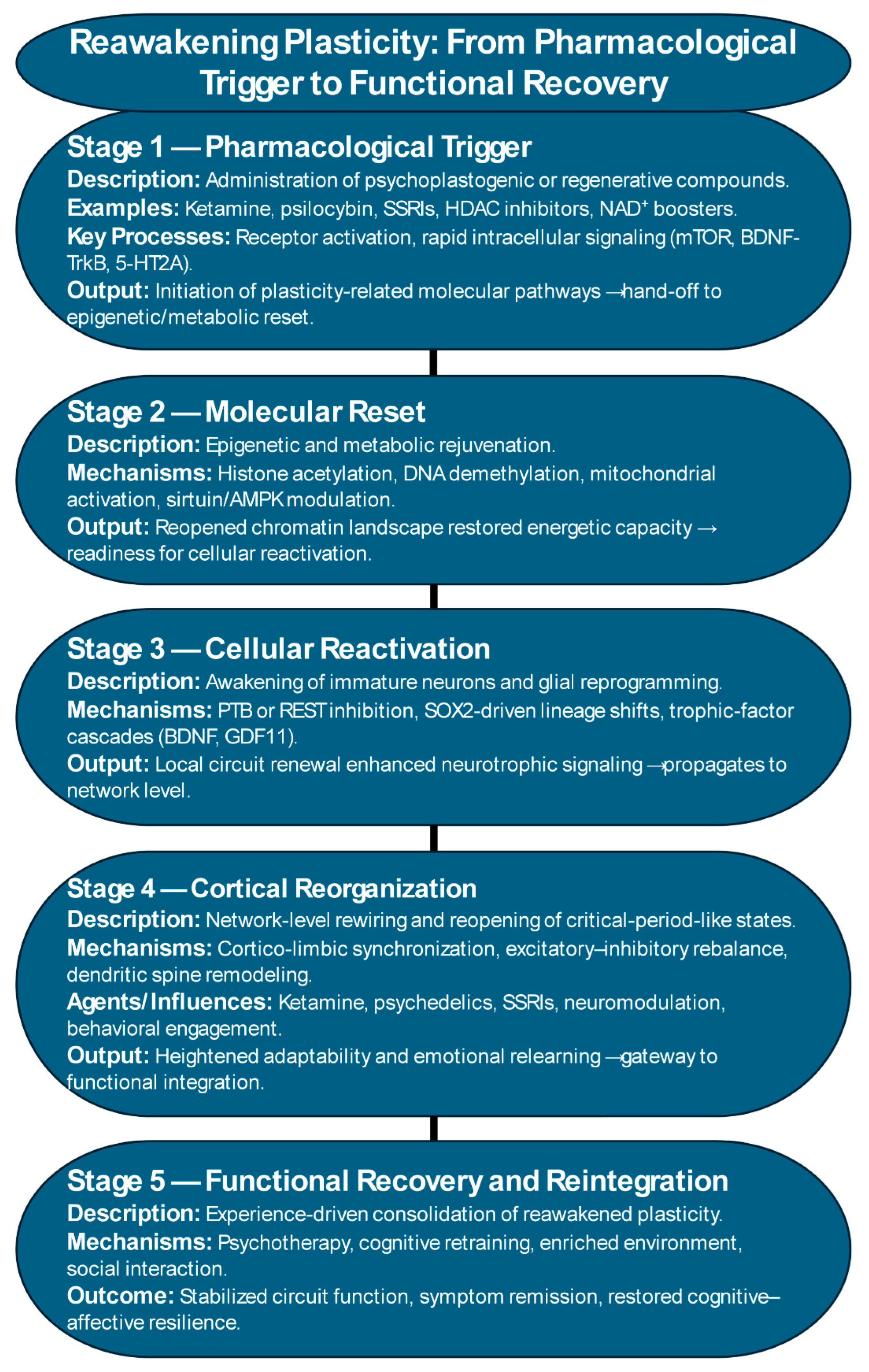
1. Reawakening the Regenerative Brain
For most of modern psychopharmacology, progress has been defined by control rather than repair. Antipsychotics dampened dopaminergic hyperactivity, antidepressants prolonged serotonergic tone, and anxiolytics suppressed excitation. These agents stabilized malfunctioning circuits, yet few restored the cellular vitality and adaptive flexibility that constitute true mental health [
1,
2]. Emerging evidence now reframes this limitation as an opportunity beneath the adult brain’s apparent rigidity lies a dormant capacity for rejuvenation and structural renewal [
3,
4].
Advances in stem-cell biology, cortical network mapping, and epigenetic modulation have converged on a provocative idea that pharmacology can do more than modulate neurotransmitters, it can reawaken intrinsic programs of plasticity. Drugs may one day reopen developmental-like windows, reactivate immature neurons, reprogram glia, and metabolically rejuvenate exhausted circuits. This shift marks the rise of regenerative pharmacology, a discipline aiming not merely to alleviate symptoms but to teach the brain how to heal itself. In the context of neuropsychiatric disorders, conditions rooted in maladaptive connectivity and impaired plasticity, this perspective offers a unifying goal to transform pharmacological intervention from chemical compensation to biological reactivation.
2. Latent Developmental Programs: Immature Neurons and Glial Reprogramming
The discovery that the adult mammalian brain retains limited neurogenic capacity in the subventricular zone (SVZ) and hippocampal dentate gyrus inspired decades of hope for cell-based regeneration [
5,
6]. In rodents, these germinal niches remain functionally active throughout life, supporting repair and behavioral adaptability. In contrast, in humans, this regenerative substrate appears markedly restricted, as SVZ neurogenesis declines sharply within the first few years of life. Nevertheless, some studies have reported evidence that hippocampal neurogenesis may persist in adults, albeit likely at very low levels—sparse, context-dependent, or perhaps even vestigial in nature [
7,
8]. This evolutionary divergence suggests that human brain plasticity must rely on alternative, more subtle cellular reserves.
Recent work points to a population of immature, prenatally generated neurons that persist in adulthood, particularly within associative cortices, the amygdala, and the claustrum [
9,
10]. These “neotenic” cells maintain a molecular phenotype of youth—expressing doublecortin, PSA-NCAM, and other developmental markers—yet remain functionally quiescent. They may serve as a latent reservoir of adaptability, capable of integrating into existing circuits under appropriate physiological or pharmacological cues. Reawakening these cells could represent a more feasible regenerative route than inducing de novo neuron formation.
Parallel advances in glial reprogramming further expand this landscape. Astrocytes, once considered passive support cells, can be converted into neurons through genetic, epigenetic, or small-molecule interventions [
11,
12,
13]. Modulating transcriptional regulators such as PTB, SOX2, or REST, or influencing metabolic and inflammatory states, has been shown to shift astrocytic identity toward neuronal lineages (
Figure 1). Such reprogramming could, in principle, restore circuit elements lost to degeneration or trauma without exogenous transplantation.
Together, these findings redefine the architecture of regeneration in the human brain. Instead of generating new cells, regenerative pharmacology aims to awaken dormant developmental programs; reactivating immature neurons, redirecting glial trajectories, and restoring the cellular flexibility that underlies true neuropsychiatric recovery.
3. Cortical Modulation and Experience-Driven Plasticity
If dormant developmental programs provide the cellular foundation for regeneration, then cortical networks constitute its command architecture. The cortex is not merely the target of pharmacological intervention but the
regulator of plasticity throughout the brain. Activity-dependent modulation of subcortical niches—especially the ventral SVZ and hippocampus—demonstrates that cortical excitability, oscillatory states, and neuromodulatory tone can directly influence stem-cell proliferation, glial differentiation, and neurotrophic signaling [
14,
15,
16,
17,
18]. In this view, the cortex acts as a top-down driver of regeneration, capable of orchestrating cellular and molecular renewal when the appropriate physiological or pharmacological conditions are met [
19].
Psychiatric pharmacology provides clear evidence for this principle. The delayed onset of antidepressant efficacy—despite rapid synaptic effects on monoaminergic transmission—has long hinted that symptom recovery involves slower, structural reorganization. Chronic SSRI treatment enhances neurogenesis in animal models and alters dendritic morphology in cortical and limbic circuits, suggesting that sustained cortical remodeling underlies therapeutic response [
20,
21]. Rapid-acting antidepressants such as ketamine amplify this process by engaging glutamatergic burst activity, BDNF-TrkB signaling, and mTOR-dependent synaptogenesis within hours (
Figure 1) [
22]. Similarly, psychedelics acting through 5-HT2A receptors reopen a transient window of developmental-like plasticity, promoting dendritic growth, synaptic spine formation, and emotional relearning (
Figure 2) [
23]. Collectively, these “psychoplastogenic” agents demonstrate that cortical networks can be pharmacologically returned to a youthful, plastic state in which maladaptive connections may be rewritten.
However, cortical reactivation alone is insufficient. Experience and environment determine whether reopened plasticity leads to repair or dysregulation. Behavioral engagement, psychotherapy, and enriched experience provide instructive
signals that stabilize beneficial circuit changes, a concept paralleling rehabilitation in motor recovery [
24,
25]. Hence, pharmacological and experiential interventions must be coupled, allowing drugs to unlock plasticity and experience to direct it.
This convergence of cortical modulation, pharmacological reactivation, and guided experience represents the translational frontier of regenerative pharmacology. By reopening critical-period-like states in targeted circuits, drugs may enable the adult brain not merely to adapt, but to relearn and restore—transforming therapy from neurotransmitter tuning into network re-education.
4. Epigenetic and Metabolic Rejuvenation — Resetting the Cellular Clock
If cortical modulation provides the system-level switch for plasticity, epigenetic and metabolic reprogramming defines the intracellular machinery that makes it possible. Every act of regeneration—whether the activation of an immature neuron or the conversion of an astrocyte—requires a permissive chromatin landscape and an energetic state capable of sustaining biosynthetic renewal. The adult brain’s relative resistance to change reflects not only circuit rigidity but also a progressive closure of its molecular potential, gene expression patterns become canalized, mitochondrial dynamics slow, and chromatin marks of development are replaced by those of stability [
26,
27,
28].
Pharmacological interventions can, in principle, reverse these molecular signatures of aging and constraint. Epigenetic modulators such as histone-deacetylase (HDAC) inhibitors, DNA-methyltransferase antagonists, and histone-acetylation enhancers reopen transcriptional access to developmental and plasticity-related genes (
Figure 2) [
29,
30]. Agents like valproate or sodium butyrate have been shown to enhance learning, promote neurotrophins expression, and facilitate reprogramming when combined with environmental enrichment. Similarly, small molecules influencing NAD⁺ metabolism, sirtuin activity, and AMPK signaling restore mitochondrial flexibility and redox balance—features essential for the anabolic demands of neurite outgrowth and synaptogenesis (
Figure 1) [
31,
32,
33,
34].
This bioenergetic rejuvenation is not merely supportive but instructive. Metabolic flux determines epigenetic state through cofactors such as acetyl-CoA, α-ketoglutarate, and NAD⁺ that directly regulate chromatin-modifying enzymes [
35]. Thus, pharmacological restoration of mitochondrial health can reactivate gene networks associated with youthful plasticity. The emerging field of mitochondrial pharmacology is beginning to intersect with psychiatry, linking metabolic normalization to cognitive resilience and antidepressant response [
36].
Taken together, these findings outline a molecular infrastructure of regeneration. Epigenetic flexibility reopens the genome’s capacity for change, while metabolic rejuvenation supplies the energy to execute it. When integrated with cortical and cellular reactivation, these processes form a coherent hierarchy from chromatin to circuit through which regenerative pharmacology can transform therapeutic design. Drugs that reset the cellular clock may one day serve as catalysts for enduring recovery, allowing the adult brain to regain a measure of its developmental vitality without losing the stability that defines maturity.
5. Towards Regenerative Pharmacology — From Modulation to Reprogramming
The emerging convergence of cellular, cortical, and molecular insights heralds the emergence of a new therapeutic paradigm in regenerative pharmacology. This framework diverges from the traditional psychopharmacological model of chemical compensation and instead aims to reprogram the brain’s intrinsic mechanisms of repair and renewal (
Table 1). Its central premise is that recovery from neuropsychiatric illness depends not merely on modulating neurotransmission but on reactivating dormant plasticity programs that operate across multiple biological scales, encompassing gene regulatory networks, cellular phenotypes, circuit connectivity, and systems integration (
Figure 2) [
37,
38,
39].
At the cellular level, immature neurons and glia constitute the biological reserve through which structural renewal may be achieved. At the network level, cortical modulation provides the control interface capable of reopening critical periods and guiding adaptive rewiring. At the molecular level, epigenetic and metabolic rejuvenation supply the enabling conditions that determine whether reprogramming can occur. Together, these domains form a multi-scale hierarchy of intervention from chromatin to cognition through which pharmacology can restore not just chemical balance, but biological plasticity itself.
Translating this framework into practice will require new methodologies for measuring regeneration
in vivo. Multi-modal imaging of synaptic density, transcriptomic profiling of peripheral biomarkers, and circuit-level electrophysiology could serve as proxies for cellular renewal and network reorganization [
40,
41,
42]. The design of next-generation compounds will need to integrate systems pharmacology, stem-cell biology, and computational modeling, enabling drugs to act not as single-target ligands but as orchestrators of adaptive cascades.
Crucially, regenerative pharmacology will also demand ethical and clinical reorientation. Drugs that reopen developmental programs must be paired with structured behavioral and environmental scaffolds to ensure that reactivated plasticity leads to recovery rather than maladaptation. Psychotherapy, cognitive training, and social engagement may thus become essential co-therapies, transforming pharmacological treatment into a biopsychosocial process of guided regeneration.
The next revolution in neuroscience will not hinge on faster receptor kinetics or novel ligands, but on our ability to pharmacologically unlock the brain’s latent potential for self-repair. In doing so, regenerative pharmacology offers a unifying vision of medicine that restores, rebalances, and ultimately reawakens the human mind.
6. Future Perspectives
Realizing the promise of regenerative pharmacology will require a new scientific and clinical ecosystem one that bridges molecular, cellular, and behavioral scales. Pharmacologists can identify the compounds that reopen plasticity; neuroscientists can map the circuits they transform; psychiatrists and psychologists must define how reactivated plasticity translates into meaningful recovery. This integrative vision demands cross-disciplinary consortia, combining single-cell transcriptomics, advanced neuroimaging, and computational modeling to capture the multiscale signatures of regeneration in the living brain (
Figure 3) [
43,
44].
Equally critical is the ethical design of interventions that manipulate developmental programs. Reawakening plasticity without proper guidance may cause instability, so it is essential to pair pharmacological reprogramming with structured experiential frameworks such as rehabilitation, psychotherapy, and enriched environments. The outcome depends on this alignment between molecular and experiential modulation, which determines whether renewal results in genuine recovery. Regulatory frameworks and clinical trials must therefore evolve to evaluate not only symptom reduction but also restorative function, adaptability, and resilience as central therapeutic goals.
As neuroscience advances beyond receptor modulation toward circuit and cellular rejuvenation, a new therapeutic philosophy is emerging. The next generation of drugs will not aim to suppress dysfunction within the brain but to enable it to rebuild its own networks. In this process of reawakening, psychiatry and pharmacology may finally converge on a shared goal that centers on restoring the brain’s intrinsic capacity for change, adaptation, and healing.
Funding
There are no funds to declare for this work.
Author Information
Moawiah M Naffaa ‘Corresponding Author’, Telephone: (+1) 3143194274. E-mail: Moawiahn@hotmail.com
Conflicts of Interest
The authors declare no conflicts of interest.
Availability of data and materials
Not applicable.
Authors' contributions
M.N. designed and wrote the main manuscript text.
References
- Correll, C.U.; Detraux, J.; De Lepeleire, J.; De Hert, M. Effects of antipsychotics, antidepressants and mood stabilizers on risk for physical diseases in people with schizophrenia, depression and bipolar disorder. World Psychiatry 2015, 14, 119-136. [CrossRef]
- Baldessarini, R.J. The impact of psychopharmacology on contemporary psychiatry. Can J Psychiatry 2014, 59, 401-405. [CrossRef]
- Bouchard, J.; Villeda, S.A. Aging and brain rejuvenation as systemic events. J Neurochem 2015, 132, 5-19. [CrossRef]
- Naffaa, M.M. Disruptions in Adult Neurogenesis: Mechanisms, Pathways, and Therapeutic Strategies for Cognitive Decline and Neurodegenerative Diseases in Aging. Nature Cell and Science 2025, 3(1), 27–53, doi:doi: 10.61474/ncs.2024.00055.
- Alvarez-Buylla, A.; Garcia-Verdugo, J.M. Neurogenesis in adult subventricular zone. J Neurosci 2002, 22, 629-634. [CrossRef]
- Bond, A.M.; Ming, G.L.; Song, H. Adult Mammalian Neural Stem Cells and Neurogenesis: Five Decades Later. Cell Stem Cell 2015, 17, 385-395. [CrossRef]
- Ming, G.L.; Song, H. Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 2011, 70, 687-702. [CrossRef]
- Kumar, A.; Pareek, V.; Faiq, M.A.; Ghosh, S.K.; Kumari, C. ADULT NEUROGENESIS IN HUMANS: A Review of Basic Concepts, History, Current Research, and Clinical Implications. Innov Clin Neurosci 2019, 16, 30-37.
- Bonfanti, L.; La Rosa, C.; Ghibaudi, M.; Sherwood, C.C. Adult neurogenesis and "immature" neurons in mammals: an evolutionary trade-off in plasticity? Brain Struct Funct 2024, 229, 1775-1793. [CrossRef]
- Sorrells, S.F.; Paredes, M.F.; Velmeshev, D.; Herranz-Perez, V.; Sandoval, K.; Mayer, S.; Chang, E.F.; Insausti, R.; Kriegstein, A.R.; Rubenstein, J.L.; et al. Immature excitatory neurons develop during adolescence in the human amygdala. Nat Commun 2019, 10, 2748. [CrossRef]
- Tan, Z.; Qin, S.; Liu, H.; Huang, X.; Pu, Y.; He, C.; Yuan, Y.; Su, Z. Small molecules reprogram reactive astrocytes into neuronal cells in the injured adult spinal cord. J Adv Res 2024, 59, 111-127. [CrossRef]
- Wang, F.; Cheng, L.; Zhang, X. Reprogramming Glial Cells into Functional Neurons for Neuro-regeneration: Challenges and Promise. Neurosci Bull 2021, 37, 1625-1636. [CrossRef]
- Wu, Z.; Parry, M.; Hou, X.Y.; Liu, M.H.; Wang, H.; Cain, R.; Pei, Z.F.; Chen, Y.C.; Guo, Z.Y.; Abhijeet, S.; et al. Gene therapy conversion of striatal astrocytes into GABAergic neurons in mouse models of Huntington's disease. Nat Commun 2020, 11, 1105. [CrossRef]
- Ruggiero, R.N.; Rossignoli, M.T.; Marques, D.B.; de Sousa, B.M.; Romcy-Pereira, R.N.; Lopes-Aguiar, C.; Leite, J.P. Neuromodulation of Hippocampal-Prefrontal Cortical Synaptic Plasticity and Functional Connectivity: Implications for Neuropsychiatric Disorders. Front Cell Neurosci 2021, 15, 732360. [CrossRef]
- Naffaa, M.M. Significance of the anterior cingulate cortex in neurogenesis plasticity: Connections, functions, and disorders across postnatal and adult stages. Bioessays 2024, 46, e2300160. [CrossRef]
- Naffaa, M.M.; Khan, R.R.; Kuo, C.T.; Yin, H.H. Cortical regulation of neurogenesis and cell proliferation in the ventral subventricular zone. Cell Rep 2023, 42, 112783. [CrossRef]
- Naffaa, M.M.; Yin, H.H. A cholinergic signaling pathway underlying cortical circuit activation of quiescent neural stem cells in the lateral ventricle. Sci Signal 2024, 17, eadk8810. [CrossRef]
- Toda, T.; Parylak, S.L.; Linker, S.B.; Gage, F.H. The role of adult hippocampal neurogenesis in brain health and disease. Mol Psychiatry 2019, 24, 67-87. [CrossRef]
- Naffaa, M.M. Neural circuit regulation of postnatal and adult subventricular zone neurogenesis: Mechanistic insights, functional models, and circuit-based neurological disorders. Archives of Stem Cell and Therapy 2024, 5 (1), doi:DOI: . [CrossRef]
- Segi-Nishida, E. The Effect of Serotonin-Targeting Antidepressants on Neurogenesis and Neuronal Maturation of the Hippocampus Mediated via 5-HT1A and 5-HT4 Receptors. Front Cell Neurosci 2017, 11, 142. [CrossRef]
- Rosas-Sanchez, G.U.; German-Ponciano, L.J.; Guillen-Ruiz, G.; Cueto-Escobedo, J.; Limon-Vazquez, A.K.; Rodriguez-Landa, J.F.; Soria-Fregozo, C. Neuroplasticity and Mechanisms of Action of Acute and Chronic Treatment with Antidepressants in Preclinical Studies. Biomedicines 2024, 12. [CrossRef]
- Kang, M.J.Y.; Hawken, E.; Vazquez, G.H. The Mechanisms Behind Rapid Antidepressant Effects of Ketamine: A Systematic Review With a Focus on Molecular Neuroplasticity. Front Psychiatry 2022, 13, 860882. [CrossRef]
- Vargas, M.V.; Dunlap, L.E.; Dong, C.; Carter, S.J.; Tombari, R.J.; Jami, S.A.; Cameron, L.P.; Patel, S.D.; Hennessey, J.J.; Saeger, H.N.; et al. Psychedelics promote neuroplasticity through the activation of intracellular 5-HT2A receptors. Science 2023, 379, 700-706. [CrossRef]
- Jeffers, M.S.; Corbett, D. Synergistic Effects of Enriched Environment and Task-Specific Reach Training on Poststroke Recovery of Motor Function. Stroke 2018, 49, 1496-1503. [CrossRef]
- Walker-Batson, D.; Smith, P.; Curtis, S.; Unwin, D.H. Neuromodulation paired with learning dependent practice to enhance post stroke recovery? Restor Neurol Neurosci 2004, 22, 387-392.
- Liu, H.; Song, N. Molecular Mechanism of Adult Neurogenesis and its Association with Human Brain Diseases. J Cent Nerv Syst Dis 2016, 8, 5-11. [CrossRef]
- Navarro Negredo, P.; Yeo, R.W.; Brunet, A. Aging and Rejuvenation of Neural Stem Cells and Their Niches. Cell Stem Cell 2020, 27, 202-223. [CrossRef]
- Seng, C.; Luo, W.; Foldy, C. Circuit formation in the adult brain. Eur J Neurosci 2022, 56, 4187-4213. [CrossRef]
- Bae, W.; Ra, E.A.; Lee, M.H. Epigenetic regulation of reprogramming and pluripotency: insights from histone modifications and their implications for cancer stem cell therapies. Front Cell Dev Biol 2025, 13, 1559183. [CrossRef]
- Benatti, B.M.; Adiletta, A.; Sgado, P.; Malgaroli, A.; Ferro, M.; Lamanna, J. Epigenetic Modifications and Neuroplasticity in the Pathogenesis of Depression: A Focus on Early Life Stress. Behav Sci (Basel) 2024, 14. [CrossRef]
- Intlekofer, K.A.; Berchtold, N.C.; Malvaez, M.; Carlos, A.J.; McQuown, S.C.; Cunningham, M.J.; Wood, M.A.; Cotman, C.W. Exercise and sodium butyrate transform a subthreshold learning event into long-term memory via a brain-derived neurotrophic factor-dependent mechanism. Neuropsychopharmacology 2013, 38, 2027-2034. [CrossRef]
- Perisic, T.; Zimmermann, N.; Kirmeier, T.; Asmus, M.; Tuorto, F.; Uhr, M.; Holsboer, F.; Rein, T.; Zschocke, J. Valproate and amitriptyline exert common and divergent influences on global and gene promoter-specific chromatin modifications in rat primary astrocytes. Neuropsychopharmacology 2010, 35, 792-805. [CrossRef]
- Hyun, D.H.; Lee, J. A New Insight into an Alternative Therapeutic Approach to Restore Redox Homeostasis and Functional Mitochondria in Neurodegenerative Diseases. Antioxidants (Basel) 2021, 11. [CrossRef]
- Mormone, E.; Iorio, E.L.; Abate, L.; Rodolfo, C. Sirtuins and redox signaling interplay in neurogenesis, neurodegenerative diseases, and neural cell reprogramming. Front Neurosci 2023, 17, 1073689. [CrossRef]
- Yu, X.; Ma, R.; Wu, Y.; Zhai, Y.; Li, S. Reciprocal Regulation of Metabolic Reprogramming and Epigenetic Modifications in Cancer. Front Genet 2018, 9, 394. [CrossRef]
- Kim, Y.; Vadodaria, K.C.; Lenkei, Z.; Kato, T.; Gage, F.H.; Marchetto, M.C.; Santos, R. Mitochondria, Metabolism, and Redox Mechanisms in Psychiatric Disorders. Antioxid Redox Signal 2019, 31, 275-317. [CrossRef]
- Marei, H.E. Neural Circuit Mapping and Neurotherapy-Based Strategies. Cell Mol Neurobiol 2025, 45, 75. [CrossRef]
- Diniz, C.; Crestani, A.P. The times they are a-changin': a proposal on how brain flexibility goes beyond the obvious to include the concepts of "upward" and "downward" to neuroplasticity. Mol Psychiatry 2023, 28, 977-992. [CrossRef]
- Gazerani, P. The neuroplastic brain: current breakthroughs and emerging frontiers. Brain Res 2025, 1858, 149643. [CrossRef]
- Cadwell, C.R.; Scala, F.; Li, S.; Livrizzi, G.; Shen, S.; Sandberg, R.; Jiang, X.; Tolias, A.S. Multimodal profiling of single-cell morphology, electrophysiology, and gene expression using Patch-seq. Nat Protoc 2017, 12, 2531-2553. [CrossRef]
- Khan, A.F.; Iturria-Medina, Y. Beyond the usual suspects: multi-factorial computational models in the search for neurodegenerative disease mechanisms. Transl Psychiatry 2024, 14, 386. [CrossRef]
- Ramezani, M.; Ren, Y.; Cubukcu, E.; Kuzum, D. Innovating beyond electrophysiology through multimodal neural interfaces. Nat Rev Electr Eng 2025, 2, 42-57. [CrossRef]
- Krejcar, O.; Namazi, H. Multiscale brain modeling: bridging microscopic and macroscopic brain dynamics for clinical and technological applications. Front Cell Neurosci 2025, 19, 1537462. [CrossRef]
- Camunas-Soler, J. Integrating single-cell transcriptomics with cellular phenotypes: cell morphology, Ca(2+) imaging and electrophysiology. Biophys Rev 2024, 16, 89-107. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).