Submitted:
21 October 2025
Posted:
22 October 2025
You are already at the latest version
Abstract
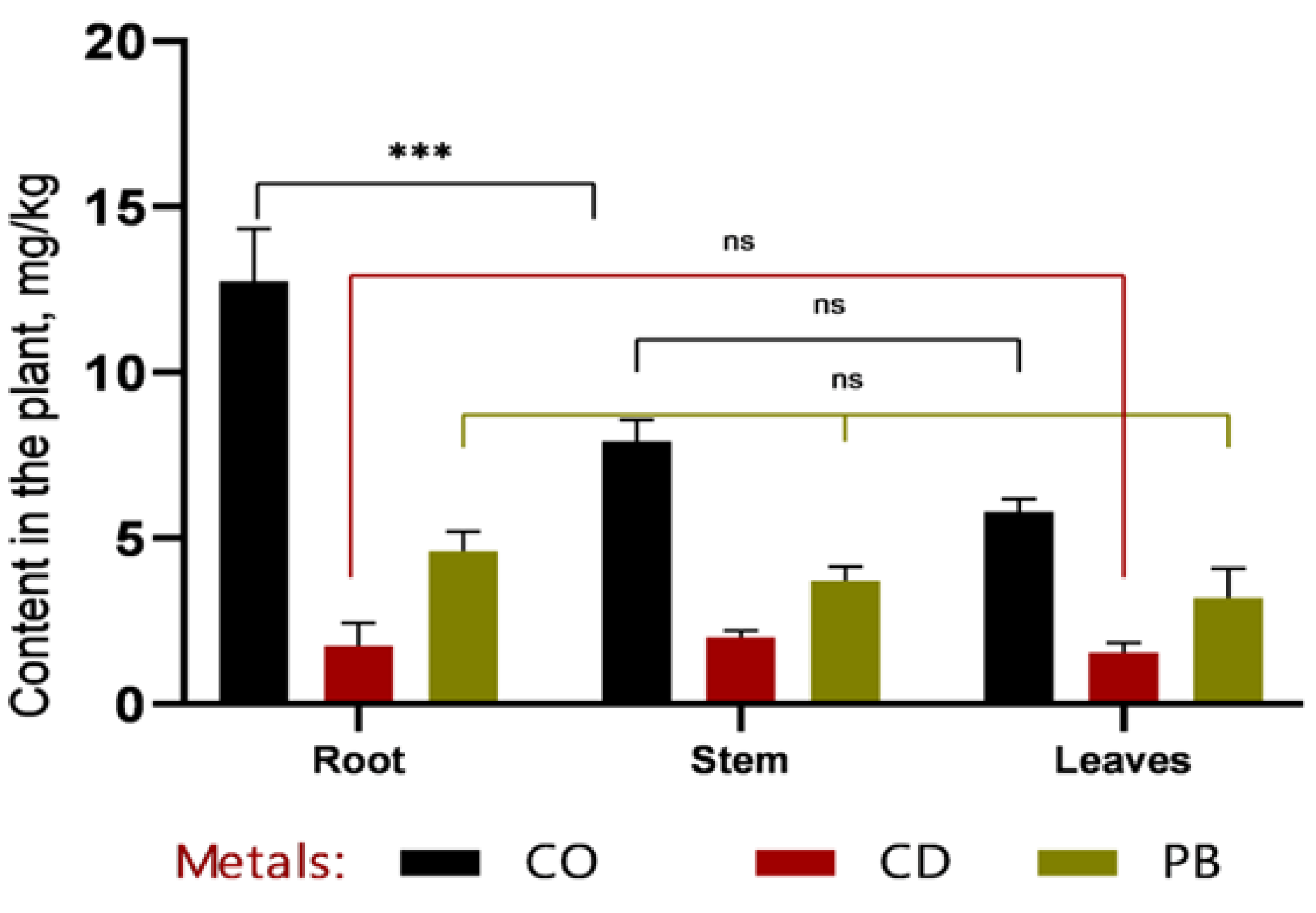
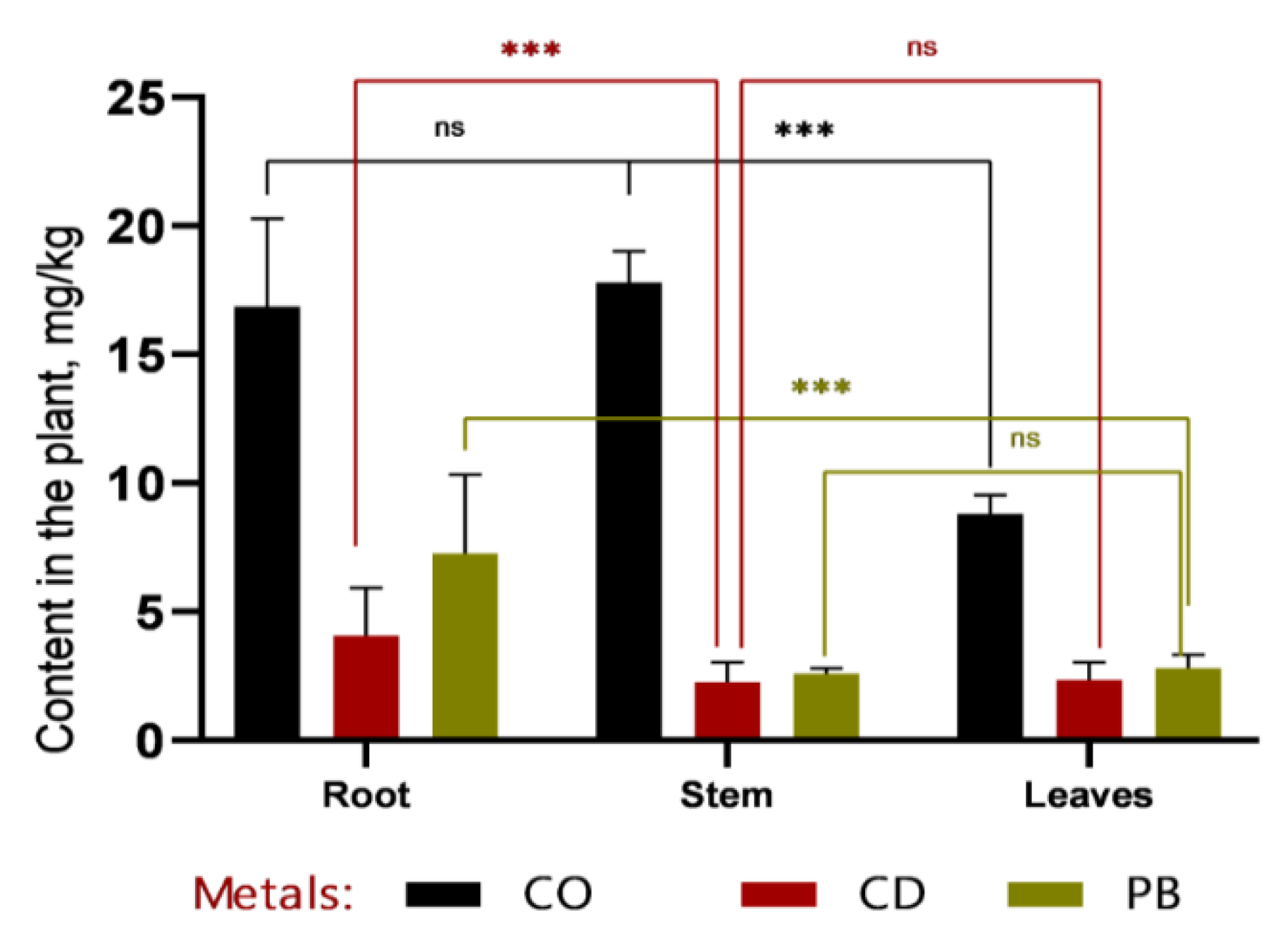
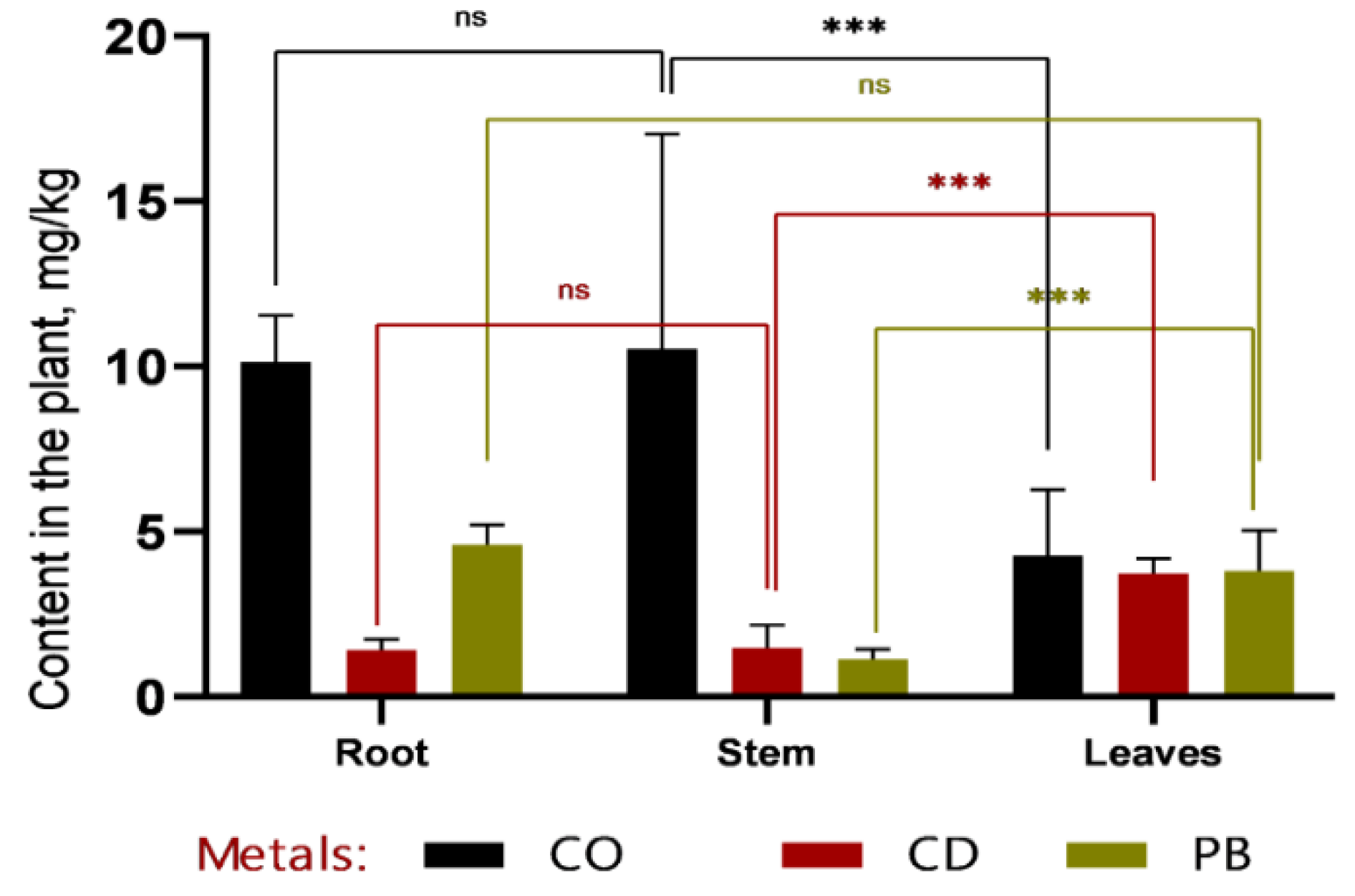
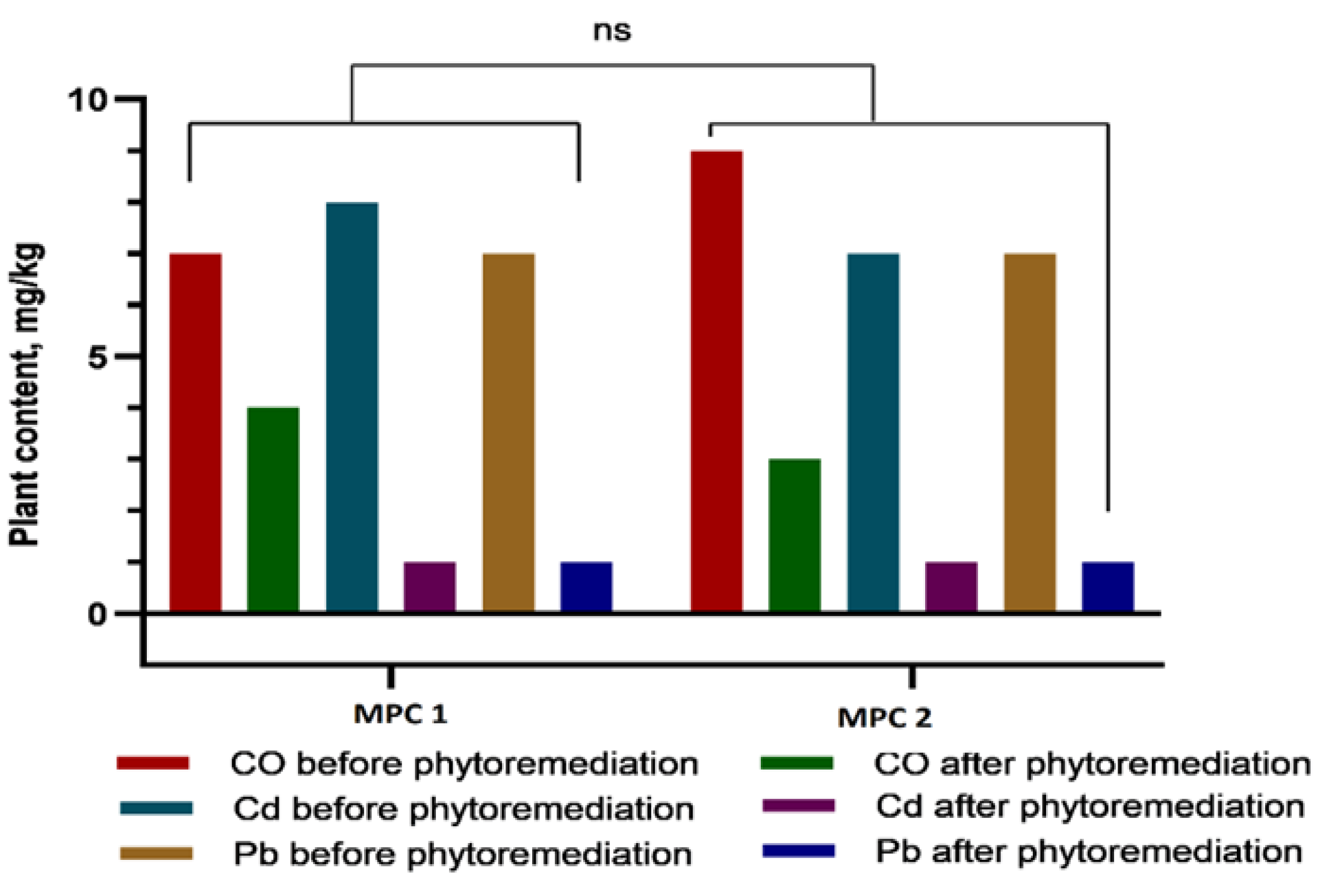
In recent years, soil contamination with heavy metals—such as plumbum, cadmium, and cobalt—has become an increasingly pressing environmental issue due to the rapid expansion of industry, transportation, and urbanization. These elements are non-biodegradable, gradually accumulate along trophic chains, and can ultimately exert serious adverse effects on human health. In this study, we focused on sweet sorghum (Sorghum bicolor L.) as a promising candidate for phytoremediation of heavy metal–contaminated soils. Both field and laboratory approaches were employed: in vitro culture of somatic cells was used to select stress-tolerant and high-yielding genotypes, while atomic absorption spectroscopy (AAS) was applied to quantify heavy metal concentrations in soil and plant tissues. Experimental results demonstrated that callus induction frequency was strongly genotype-dependent. The highest callus formation rates were observed in Hybrid-2 (69.11%), SAB-3 (43.83%), SABB-1 (42.31%), and SAB-10 (40.32%). Among these, Hybrid-2 (27.94%) and SAB-3 (23.28%) also exhibited the highest frequencies of morphogenic callus formation, confirming their suitability for subsequent micropropagation and cell-based selection. Particular attention was paid to the distribution of toxicants among plant organs. The rate and direction of metal translocation from roots to shoots were found to critically influence the efficiency of subsequent metal removal, as stems constitute the major component of easily harvestable biomass. Nevertheless, a substantial proportion of heavy metals remained sequestered in the roots: cobalt concentrations reached 12.7 ± 1.32 mg/kg at 1 MPC (Maximum Permissible Concentration) and 16.87 ± 2.78 mg/kg at 2 MPC, accounting for more than 50% of the total accumulated amount. Similar trends were observed for cadmium (~49%) and plumbum (up to 53%). Thus, our findings underscore the pivotal role of genotype in in vitro callus formation and morphogenesis, and further highlight the potential of sweet sorghum as an effective phytoremediation agent for reclaiming heavy metal–contaminated lands, particularly in arid regions.
Keywords:
1. Introduction
2. Results
2.1. Selection of High-Performing Sweet Sorghum Genotypes for Phytoremediation
3.2. Comparative Analysis of Heavy Metal Accumulation in Sorghum Tissues and Soil
3. Discussion
4. Materials and Methods
4.1. Development of a High-Productivity Genotype of Sweet Sorghum (Sorghum bicolor L.)
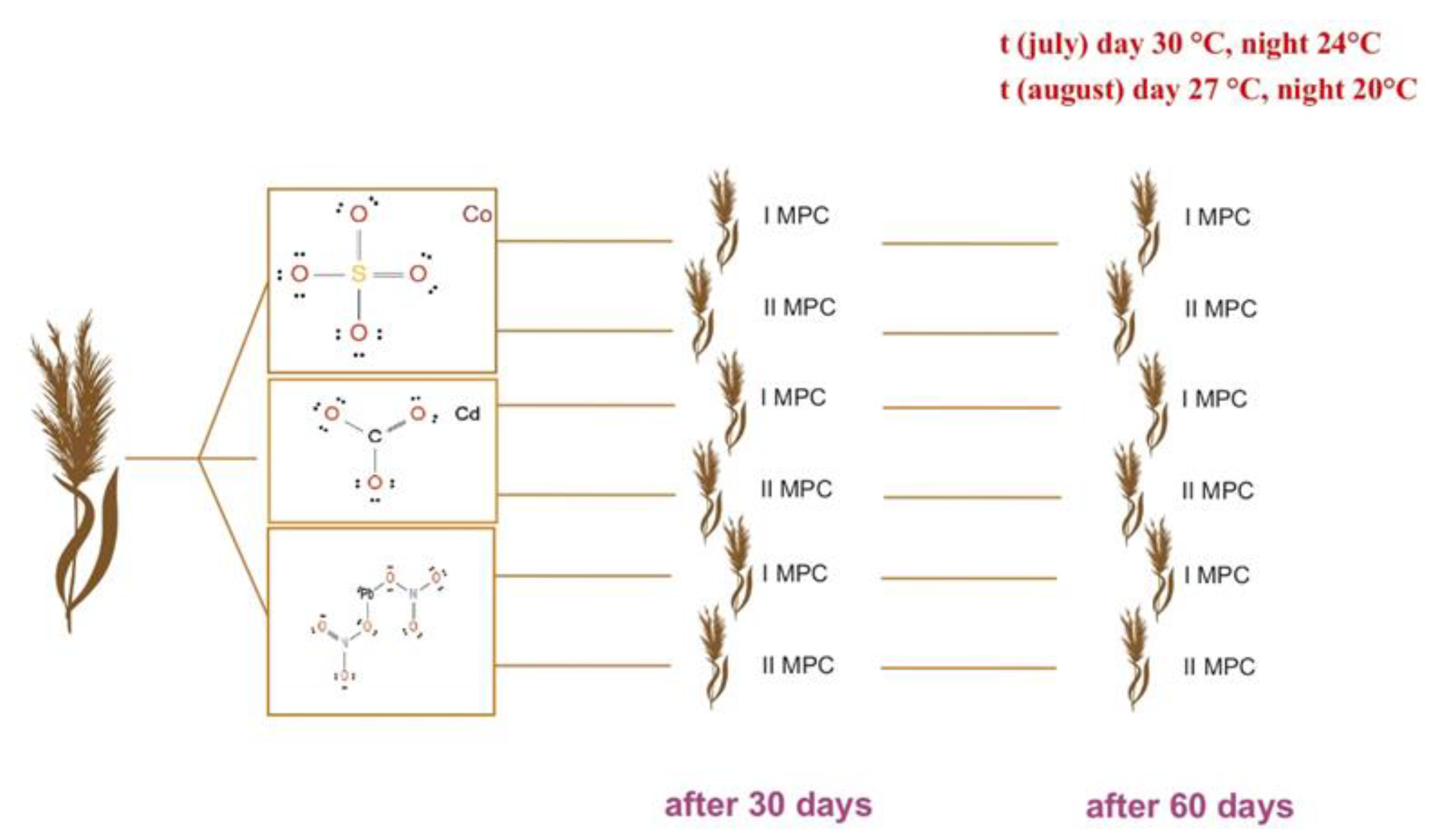
4.2. Laboratory-Scale Model Experiment on Phytoremediation of Heavy Metal–Contaminated Soils
4.3. Determination of Heavy Metal Content in Soil and Plant Tissues
4.4. Statistical Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AAS | Atomic absorption spectroscopy |
| MPC | Maximum Permissible Concentration |
| As | Arsenic |
| Cd | Cadmium |
| Cr | Chromium |
| Hg | Mercury |
| Cu | Copper |
| Pb | Рlumbum |
| Ni | Nickel |
| Zn | Zinc |
| MAC | Maximum allowable concentration |
| MS | Murashige and Skoog |
References
- Bhunia, P. Environmental toxicants and hazardous contaminants: Recent advances in technologies for sustainable development. Journal of Hazardous, Toxic, and Radioactive Waste 2017, 21, 02017001. [Google Scholar] [CrossRef]
- Priya, A. K. , & Nagan, S. (2015). Bioremediation of dye effluent and metal contaminated soil: Low-cost method for environmental clean up by microbes. Journal of Environmental Science and Engineering 2015, 57, 109–119. [Google Scholar]
- Alengebawy, A. , Abdelkhalek, S. T., Qureshi, S. R., & Wang, M.-Q. (2021). Heavy metals and pesticides toxicity in agricultural soil and plants: Ecological risks and human health implications. Toxics 2021, 9, 42. [Google Scholar] [PubMed]
- Kapoor, D. , & Singh, M. P. (2021). Heavy metal contamination in water and its possible sources. In Heavy metals in the environment 2021, (pp. 179–189). Elsevier.
- Jabeen, R. , Ahmad, A., & Iqbal, M. (2009). Phytoremediation of heavy metals: Physiological and molecular mechanisms. Botanical Review 2009, 75, 339–364. [Google Scholar]
- Berglund, M. , Larsson, K., Grandér, M., Casteleyn, L., Kolossa-Gehring, M., Schwedler, G., et al. Exposure determinants of cadmium in European mothers and their children. Environmental Research 2015, 141, 69–76. [Google Scholar] [CrossRef]
- Dada, E. O. , Njoku, K. I., Osuntoki, A. A., & Akinola, M. O. A review of current techniques of physico-chemical and biological remediation of heavy metals polluted soil. Ethiopian Journal of Environmental Studies and Management 2015, 8, 606–615. [Google Scholar] [CrossRef]
- Dixit, R. , Wasiullah, M. D., Pandiyan, K., Singh, U. B., Sahu, A., Shukla, R., Singh, B. P., Rai, J. P., Sharma, P. K., Lade, H., & Paul, D. Bioremediation of heavy metals from soil and aquatic environment: An overview of principles and criteria of fundamental processes. Sustainability 2015, 7, 2189–2212. [Google Scholar]
- Sarwar, N. , Imran, M., Shaheen, M. R., Ishaq, W., Kamran, A., Matloob, A., Rehim, A., & Hussain, S. Phytoremediation strategies for soils contaminated with heavy metals: Modifications and future perspectives. Chemosphere 2017, 171, 710–721. [Google Scholar]
- Maszenan, A. M. , Liu, Y., & Ng, W. J. Bioremediation of wastewaters with r. ecalcitrant organic compounds and metals by aerobic granules. Biotechnology Advances 2011, 29, 111–123. [Google Scholar]
- Stearns, J. C. , Shah, S., & Glick, B. R. Increasing plant tolerance to metals in the environment. In N. Willey (Ed.), Phytoremediation: Methods and Reviews 2006, (pp. 15–26). Humana Press.
- Verma, R.K. , Awasthi, K., Sankhla, M.S., Jadhav, E.B. and Parihar, K. Phytoremediation of Heavy Metals Extracted from Soil and Aquatic Environments: Current Advances as Well as Emerging Trends. Biointerface Research in Applied Chemistry 2022, 12, 5486–5509. [Google Scholar]
- Sharma, J. K.; Kumar, N.; Singh, N. P.; Santal, A. R. Phytoremediation Technologies and Their Mechanism for Removal of Heavy Metal from Contaminated Soil: An Approach for a Sustainable Environment. Front. Plant Sci. 2023, 14, 1076876. [Google Scholar] [CrossRef] [PubMed]
- Baker, A. J. M. , & Brooks, R. R. Terrestrial higher plants which hyperaccumulate metal elements—A review of their distribution, ecology and phytochemistry. Biorecovery 1989, 1, 81–126. [Google Scholar]
- Ali, H. , Khan, E., & Sajad, M. A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar]
- Chandra, R. , Saxena, G., & Kumar, V. Phytoremediation of environmental pollutants: An eco-sustainable green technology to environmental management. In R. Chandra (Ed.), Advances in biodegradation and bioremediation of industrial waste 2015, pp. 1–30. CRC Press.
- Mahar, A. , Wang, P., Ali, A., Awasthi, M. K., Lahori, A. H., Wang, Q., Li, R., & Zhang, Z. Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: A review. Ecotoxicology and Environmental Safety 2016, 126, 111–121. [Google Scholar]
- Muthusaravanan, S. , Sivarajasekar, N., Vivek, J. S., Paramasivan, T., Naushad, M., Prakashmaran, J., Gayathri, V., & Al-Duaij, O. K. Phytoremediation of heavy metals: Mechanisms, methods and enhancements. Environmental Chemistry Letters 2018, 16, 1339–1359. [Google Scholar]
- Oh, K. , Li, T., Cheng, H., Xie, Y., & Yonemochi, S. Study on tolerance and accumulation potential of bio-fuel crops for phytoremediation of heavy metals. International Journal of Environmental Science and Development 2013, 4, 152–156. [Google Scholar]
- Oh, K. , Cao, T., Li, T., & Cheng, H. Study on application of phytoremediation technology in management and remediation of contaminated soils. Journal of Clean Energy Technologies 2014, 2, 216–220. [Google Scholar]
- Zaynutdinova, E. M. , Yagafarova, G. G., Shamsudinova, E. A., & Mazitova, A. K. Phytoremediation of Technogenically Disturbed Areas. Bulletin of Kazan Technological University, 2017; 157–159. [Google Scholar]
- Elekes, C. C. Eco-technological solutions for the remediation of polluted soil and heavy metal recovery. In M. C. Hernández-Soriano (Ed.), Environmental risk assessment of soil contamination 2014, (pp. 309–335). InTech.
- Lin, H. , Wang, Z., Liu, C., & Dong, Y. (2022). Technologies for removing heavy metal from contaminated soils on farmland: A review. Chemosphere 2022, 305, 135457. [Google Scholar]
- Noor, I. , Sohail, H., Sun, J., Nawaz, M. A., Li, G., Hasanuzzaman, M., & Liu, J. Heavy metal and metalloid toxicity in horticultural plants: Tolerance mechanism and remediation strategies. Chemosphere 2022, 303, 135196. [Google Scholar] [PubMed]
- Chauhan, P. , & Mathur, J. Phytoremediation efficiency of Helianthus annuus L. for reclamation of heavy metals-contaminated industrial soil. Environmental Science and Pollution Research 2020, 27, 29954–29966. [Google Scholar]
- Zou, J. , et al. Phytoremediation potential of wheat intercropped with different densities of Sedum plumbizincicola in soil contaminated with cadmium and zinc. Chemosphere 2021, 276, 130223. [Google Scholar] [CrossRef] [PubMed]
- Yang, W. , et al. Strengthening role and the mechanism of optimum nitrogen addition in relation to Solanum nigrum L. Cd hyperaccumulation in soil. Ecotoxicology and Environmental Safety 2019, 182, 109444. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X. , Xiong, T., Yao, S., Liu, C., Yin, Y., Li, H., & Li, N. A real field phytoremediation of multi-metals contaminated soils by selected hybrid sweet sorghum with high biomass and high accumulation ability. Chemosphere 2019, 237, 124536. [Google Scholar]
- Pandey, J. , Verma, R. K., & Singh, S. Suitability of aromatic plants for phytoremediation of heavy metal contaminated areas: A review. International Journal of Phytoremediation 2019, 21, 405–418. [Google Scholar]
- Sheoran, V. , Sheoran, A. S., & Poonia, P. Factors affecting phytoextraction: A review. Pedosphere 2016, 26, 148–166. [Google Scholar]
- Salifu, M. , John, M., Abubakar, M., Bankole, I., Ajayi, N., & Amusan, O. Phytoremediation strategies for heavy metal contamination: A review on sustainable approach for environmental restoration. Journal of Environmental Protection 2024, 15, 450–474. [Google Scholar]
- Anapiyaev, B. B. , Iskakova, K. M., Beisenbek, E. B., Kapalova, S. K., Sagimbayeva, A. M., & Omarova, A. Sh. Features of Sorghum bicolor L. Resistance to Biotic Stress Factors under Arid Conditions of Southeastern Kazakhstan. Agrarian Science 2019, (S1), 166–170. [Google Scholar]
- Sagimbayeva, A. M. Selection of Highly Productive Genotypes of Sweet Sorghum (Sorghum bicolor L.) for Further Use in Phytoremediation of Soils Contaminated with Heavy Metals. Biosafety and Biotechnology 2022, (11), 30–38. [Google Scholar]
- Soudek, P. , Petrová, Š. , Vaňková, R., Song, J., & Vaněk, T. Accumulation of heavy metals using Sorghum sp. Chemosphere 2014, 104, 15–24. [Google Scholar]
- Wang, X. , Chen, C., & Wang, J. Phytoremediation of strontium contaminated soil by Sorghum bicolor (L.) Moench and soil microbial community-level physiological profiles (CLPPs). Environmental Science and Pollution Research 2017, 24, 7668–7678. [Google Scholar] [PubMed]
- Di Giuseppe, D. , Melchiorre, M., Bianchini, G., & Vaněk, T. Assessment of heavy metal bioaccumulation in sorghum from neutral saline soils in the Po River Delta Plain (Northern Italy). Environmental Earth Sciences 2017, 76, 519. [Google Scholar]
- Wang, S.; Luo, Y.; Zhou, Q.; Zhang, Y.; Xu, Y.; Chen, W. Energy Sorghum Removal of Soil Cadmium in Chinese Subtropical Farmland: Effects of Variety and Cropping System. Agronomy 2023, 13, 2487. [Google Scholar] [CrossRef]
- Osman, H. E. , Fadhlallah, R. S., Alamoudi, W. M., Eid, E. M., & Abdelhafez, A. A. Phytoremediation potential of sorghum as a bioenergy crop in Pb-amendment soil. Sustainability 2023, 15, 2178. [Google Scholar]
- Murashige, T. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiology 1962, 15, 473–497. [Google Scholar] [CrossRef]
- George, E.F.; Hall, M.A.; de Klerk, G.J. Plant Propagation by Tissue Culture. Springer: Dordrecht, The Netherlands 2008, p. 401.
- ISO 11466:1995; International Organization for Standardization. ISO 11466:1995—Soil Quality—Extraction of Trace Elements Soluble in Aqua Regia; ISO: Geneva, Switzerland, 1995. [Google Scholar]
- ISO 11047:1998; International Organization for Standardization. ISO 11047:1998—Soil Quality—Determination of Cadmium, Chromium, Cobalt, Copper, Lead, Manganese, Nickel and Zinc—Flame and Electrothermal Atomic Absorption Spectrometric Methods; ISO: Geneva, Switzerland, 1998. [Google Scholar]

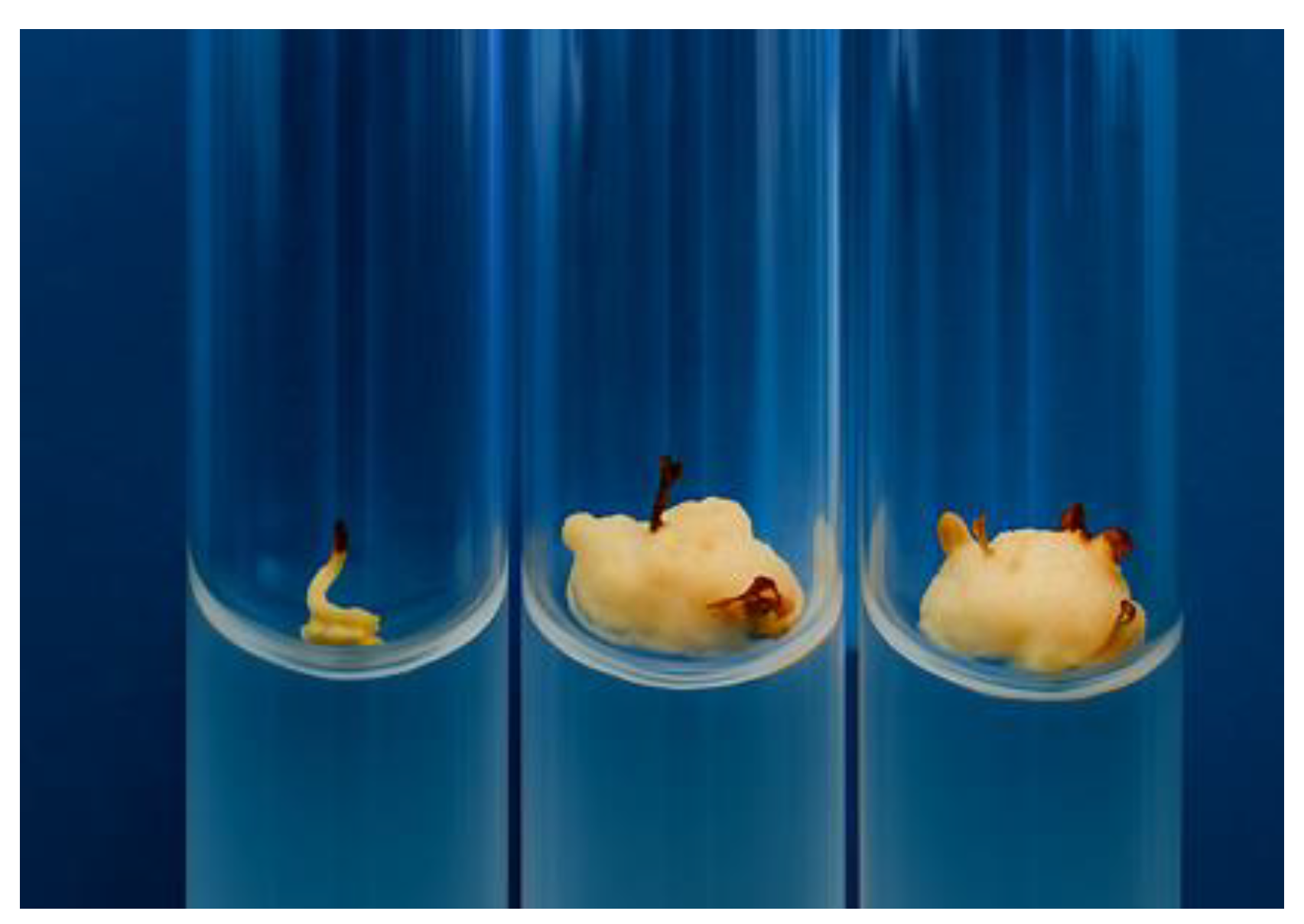

| Genotype | Number of isolated embryos | Number of calli formed | Callus induction rate (%) |
| SABB-1 | 52 | 22 | 42,31 |
| SAB-2 | 67 | 3 | 4,47 |
| SAB-3 | 73 | 32 | 43,83 |
| SAB-10 | 62 | 25 | 40,32 |
| SAB-11 | 79 | 29 | 36,70 |
| Hybrid-1 | 75 | 19 | 25,33 |
| Hybrid-2 | 68 | 47 | 69,11 |
| Genotype | Number of isolated embryos | Number of calli formed | Callus induction rate (%) |
| SABB-1 | 52 | 9 | 2,99 |
| SAB-2 | 67 | 0 | 0 |
| SAB-3 | 73 | 17 | 23,28 |
| SAB-10 | 62 | 8 | 12,90 |
| SAB-11 | 79 | 6 | 7,59 |
| Hybrid-1 | 75 | 12 | 16,0 |
| Hybrid-2 | 68 | 19 | 27,94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




