Submitted:
20 October 2025
Posted:
24 October 2025
You are already at the latest version
Abstract
Tropical fruit waste composed of peels, pulp, and discards, presents a growing disposal challenge in high and rising fruit production regions. This review explores transforming this waste into bioethanol which can also be defined as a clean-burning biofuel. It examines pre-treatment techniques like enzymatic and acidic hydrolysis that explains how complex carbohydrates is broken down into fermentable sugar efficiently. These techniques are very much required for a complete and efficient production of bioethanol. Additionally, the study focuses on optimizing fermentation conditions, including temperature, yeast strain selection, and nutrient supplementation, to maximize bioethanol yield. The impact of fruit ripeness on bioethanol yield is discussed, noting how sugar content changes during ripening affecting the ethanol output. Saccharomyces cerevisiae , a robust fermenting agent, is highlighted for its potential in bioethanol production. The feasibility of bioethanol production from various fruit substrates using a simulation model is highlighted. The model incorporates key factors such as substrate concentration of glucose, yeast cell density where various parameters of Saccharomyces cerevisiae is considered, and ethanol production. While the simulation results exhibit similar trends for different fruits, factors like model simplifications and parameter sensitivity can influence the outcomes. By integrating findings from various studies and other sources, this review aims to develop a cost-effective and sustainable bioethanol production process using tropical fruit waste.
Keywords:
1. Introduction
2. Fruit Profiles for Bioethanol Production
2.1. Rambutan (Nephelium lappaceum)
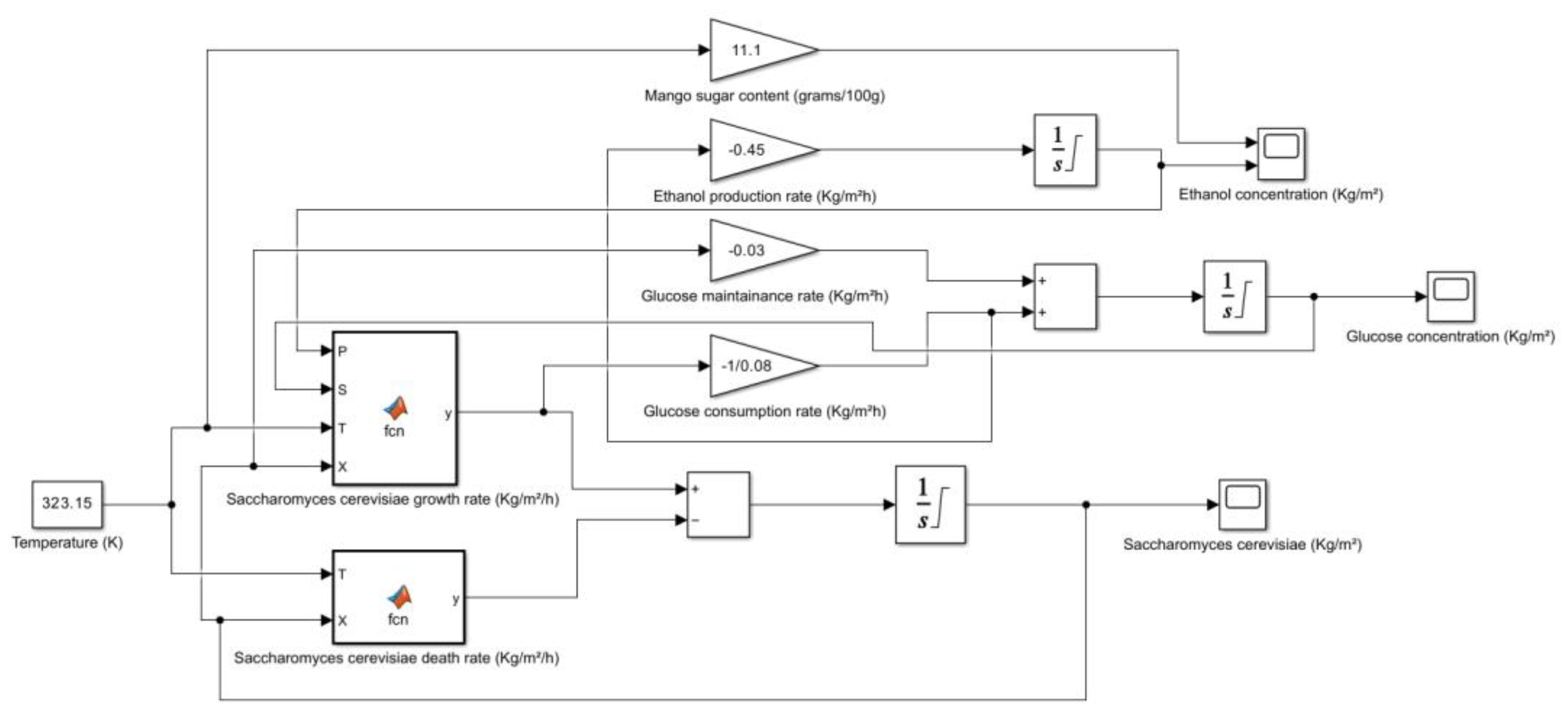
2.2. Mango (Mangifera indica)
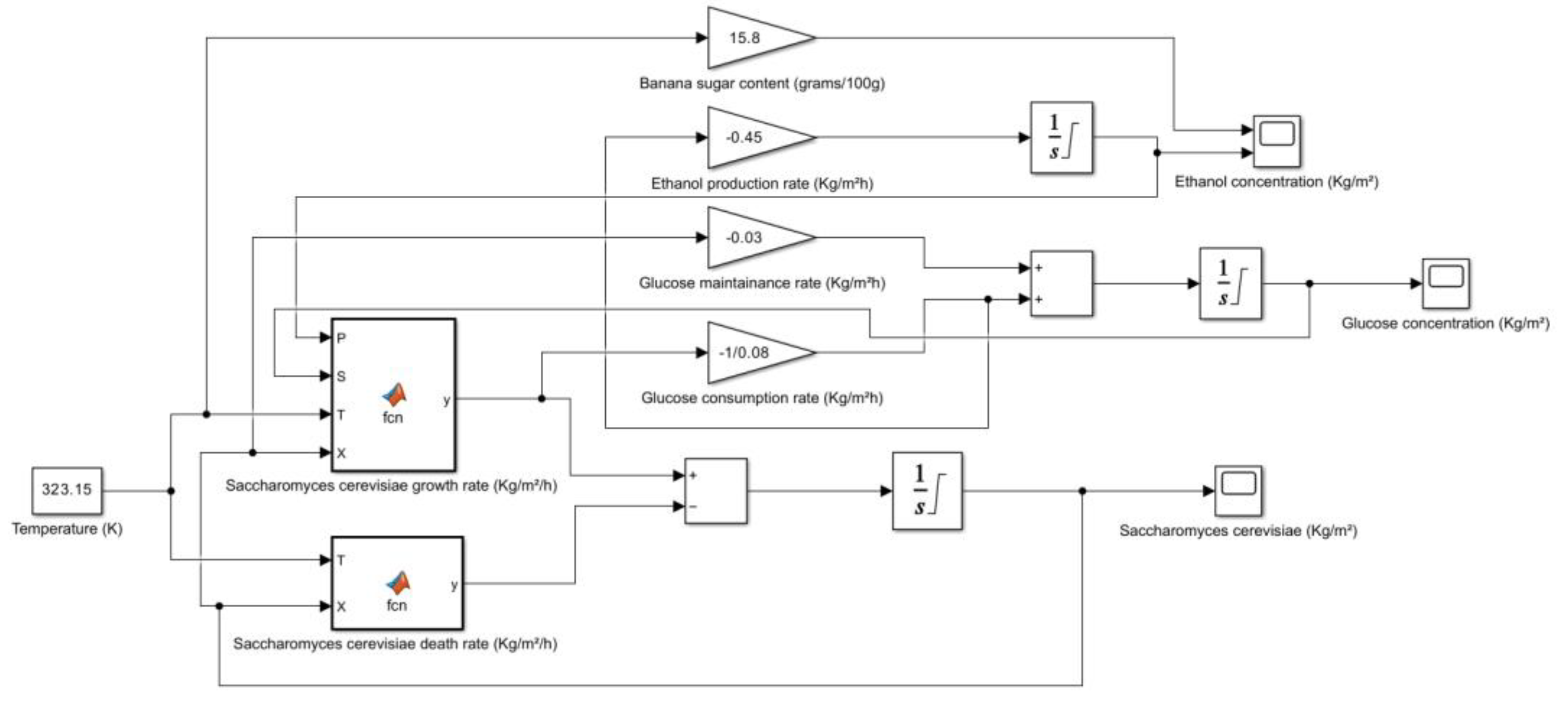
2.3. Banana (Musa paradisiaca Linn)
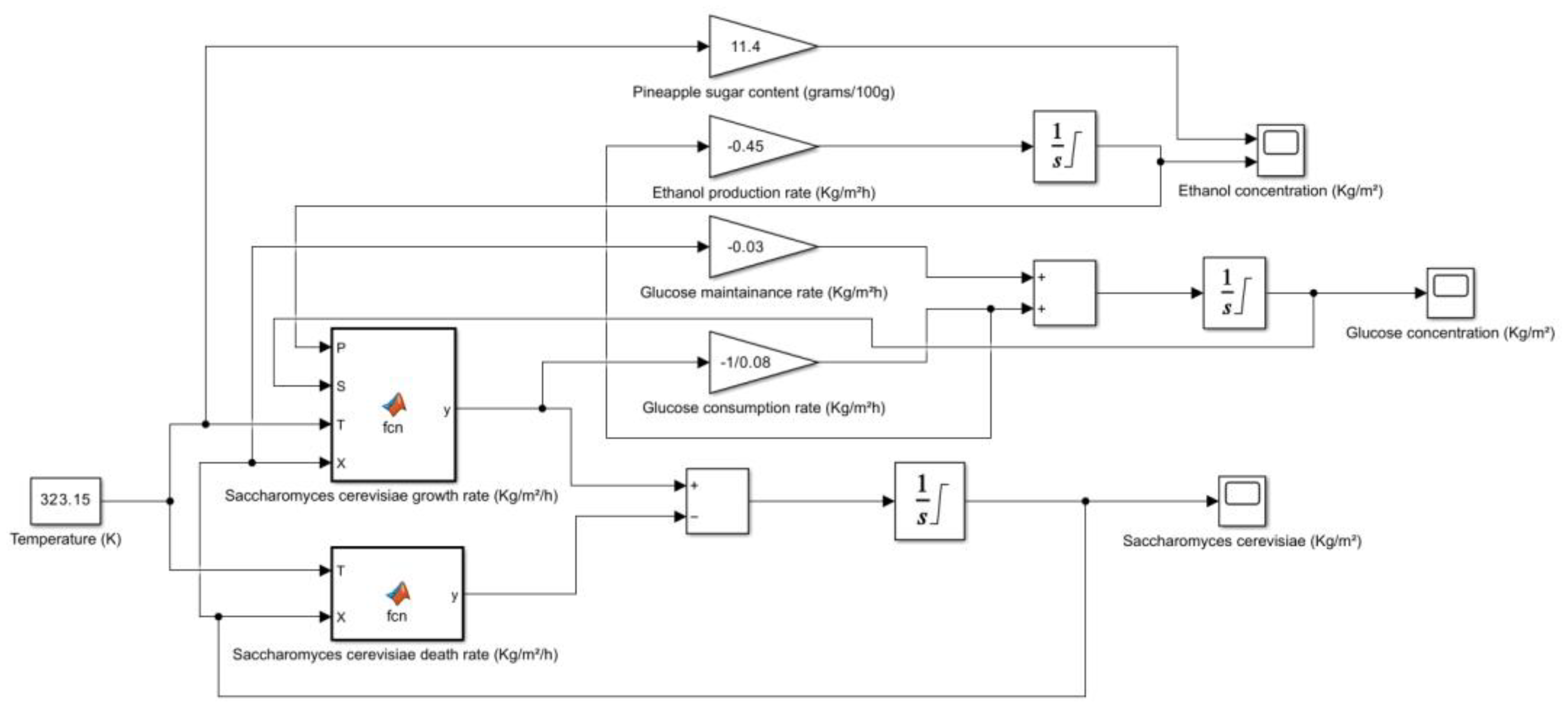
2.4. Pineapple (Ananas comosus)
2.5. Jackfruit Rind (Artocarpus heterophyllus)
2.6. Durian (Durio zibethinus L.)
2.7. Guava (Psidium guajava L.)
2.8. Dragon Fruit (Selenicereus undatus)
2.9. Muskmelon (Cucumis melo)
2.10. Mangosteen (Garcinia mangostana)
2.11. Lychee (Litchi chinensis)
3. Systematic Approaches for the Production of Bioethanol
3.1. Pre-Treatment Methods and Substrate Preparation
3.2. Acid Hydrolysis Technique
- i.)
- Cellulose (main polysaccharide in peels):
- ii.)
- Hemicellulose (another polysaccharide in peels):
3.3. pH Modulation Parameters
3.4. Fermentation Medium and Yeast Microorganism
3.5. Fermentation Process
3.6. Distillation Method
3.7. Quantification of Bioethanol Production
3.7.1. Dichromate Test
3.7.2. Gas Chromatography
3.7.3. High-Performance Liquid Chromatography (HPLC)
3.7.4. Enzymatic Assays
3.7.5. Near-Infrared (NIR) Spectroscopy
3.8. Analysis of Ethanol Content
4. Comparative Studies of Different Strains of Saccharomyces cerevisiae
4.1. Characterization and Isolation
4.2. Thermotolerant Properties
4.3. Bioethanol Production Potential
4.4. Thermotolerant Property Detection by Molecular Screening
4.4.1. Cell Lysis and Centrifugation
4.4.2. Heat and Freeze-Thaw Cycles
4.4.3. Protein Removal and Centrifugation
4.4.4. Isopropanol Precipitation
4.5. Isolating a Stress-Resistant Yeast Strain for high-gravity (HG) fermentation
4.6. Comparison of SC-GR Strain and RK1 Strain
4.6.1. Substrate Specificity and Sugar Utilization
4.6.2. Ethanol Yield
4.6.3. Stress Tolerance
5. Kinetic and Simulation model of Saccharomyces cerevisiae for the production of Bioethanol
| Parameter | Expression | Description |
|---|---|---|
| Microbial Growth Rate | Rate of change of biomass concentration | |
| Substrate Consumption Rate | Rate of change of substrate concentration | |
| Product Formation Rate | Rate of change of product concentration |
| Parameter | Value |
|---|---|
| 1.7 kg/m³ | |
| 93 kg/m³ | |
| n | 0.52 |
| 12.108 exp | |
| m | 0.03 h−1 |
| 0.08 | |
| 0.45 | |
| Initial concentration of Saccharomyces cerevisiae | 1.5 kg/m³ |
| Initial concentration of glucose | 220 kg/m³ |
| Fermentation temperature | 30 ℃ |
| Fruit | Sugar Content (grams/100g) | Method of Measurement | Other Notable Components | **Potential Sources |
|---|---|---|---|---|
| Mango (Ataulfo, Peeled, Raw) | 11.1 | HPLC | Vitamin A, C, B6, E, potassium, magnesium, copper, folate, antioxidants, fiber | USDA FoodData Central, Healthline, WebMD |
| Banana (Overripe, Raw) | 15.8 | HPLC | Potassium, Vitamin B6, Vitamin C, Fiber | USDA FoodData Central, Healthline, WebMD |
| Pineapple (raw) | 11.4 | HPLC | Vitamin C, B6, thiamin, manganese, copper, potassium, bromelain | USDA FoodData Central, Healthline, WebMD |
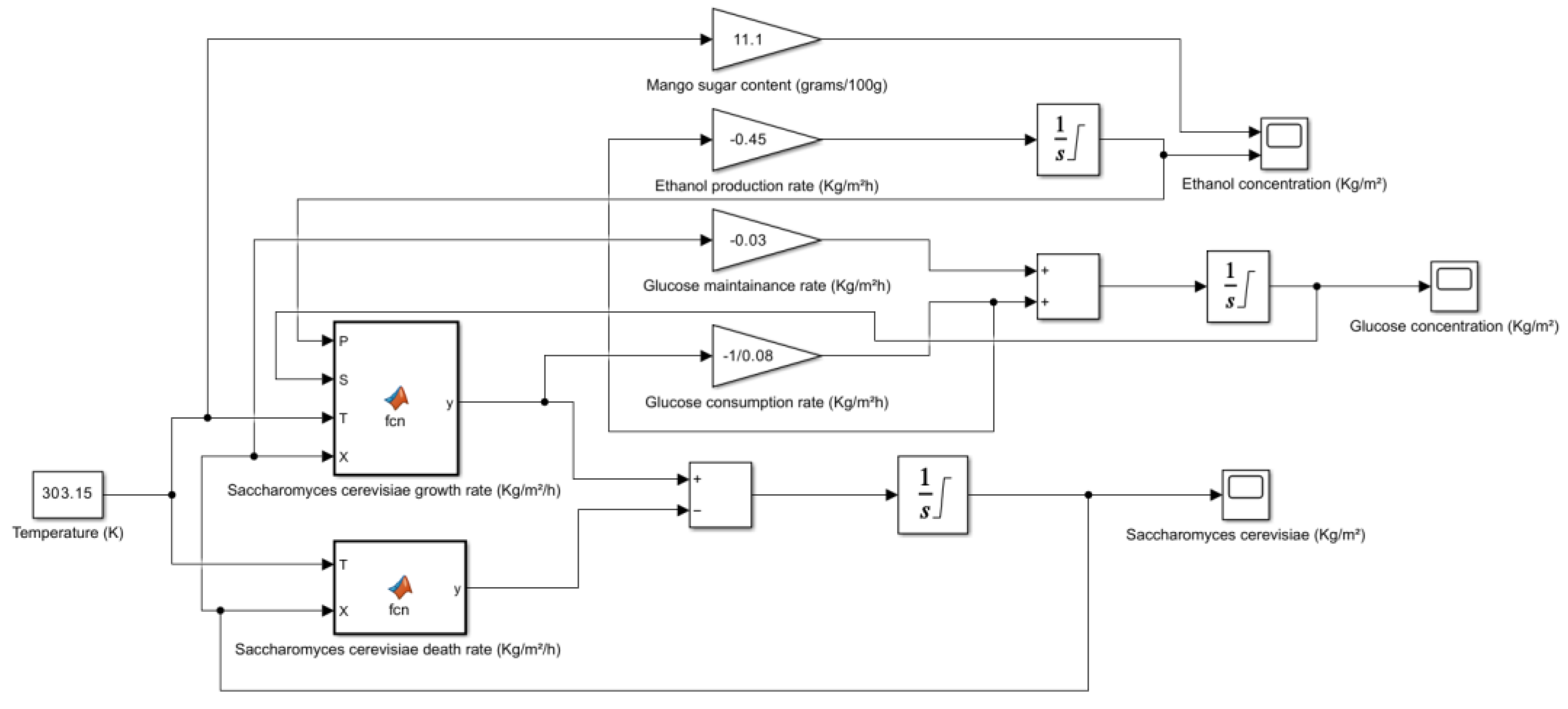
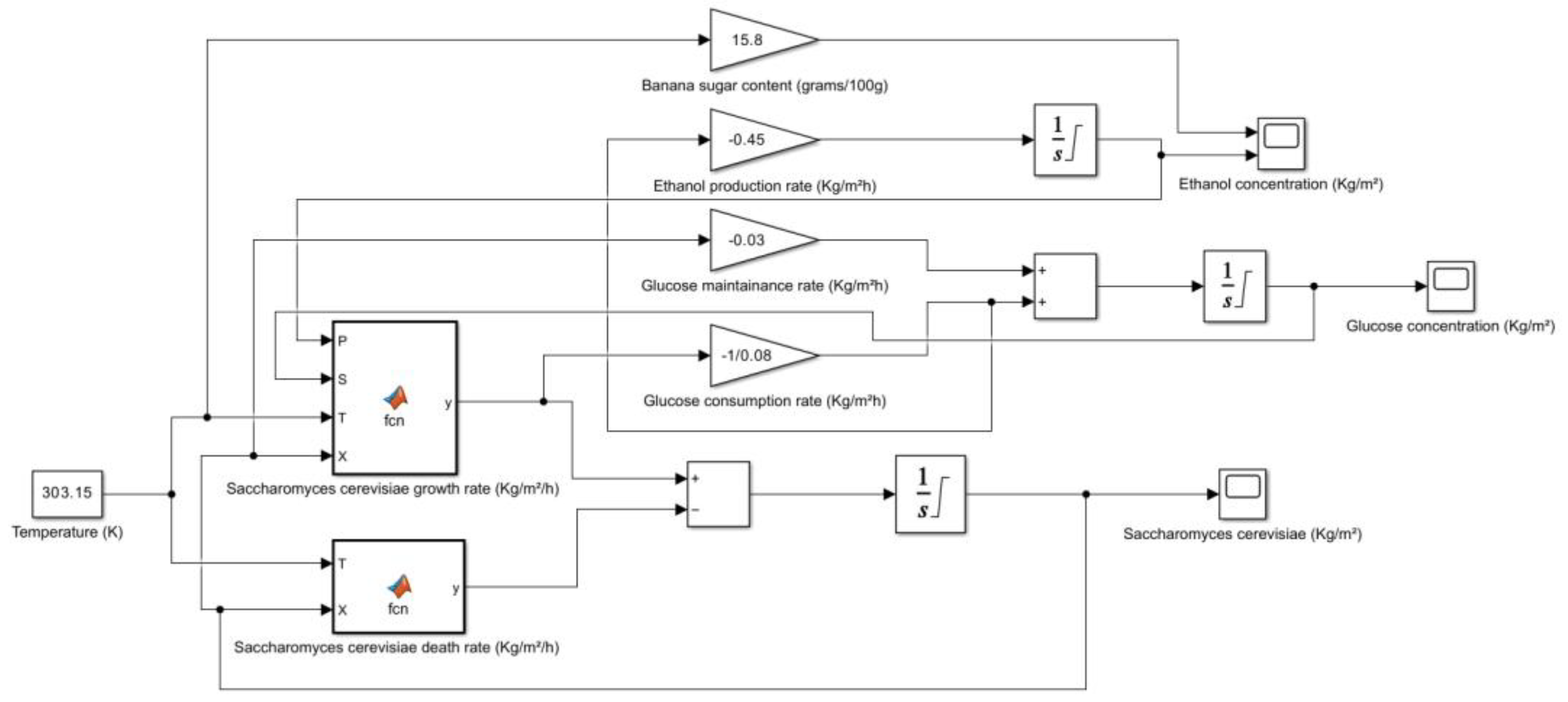
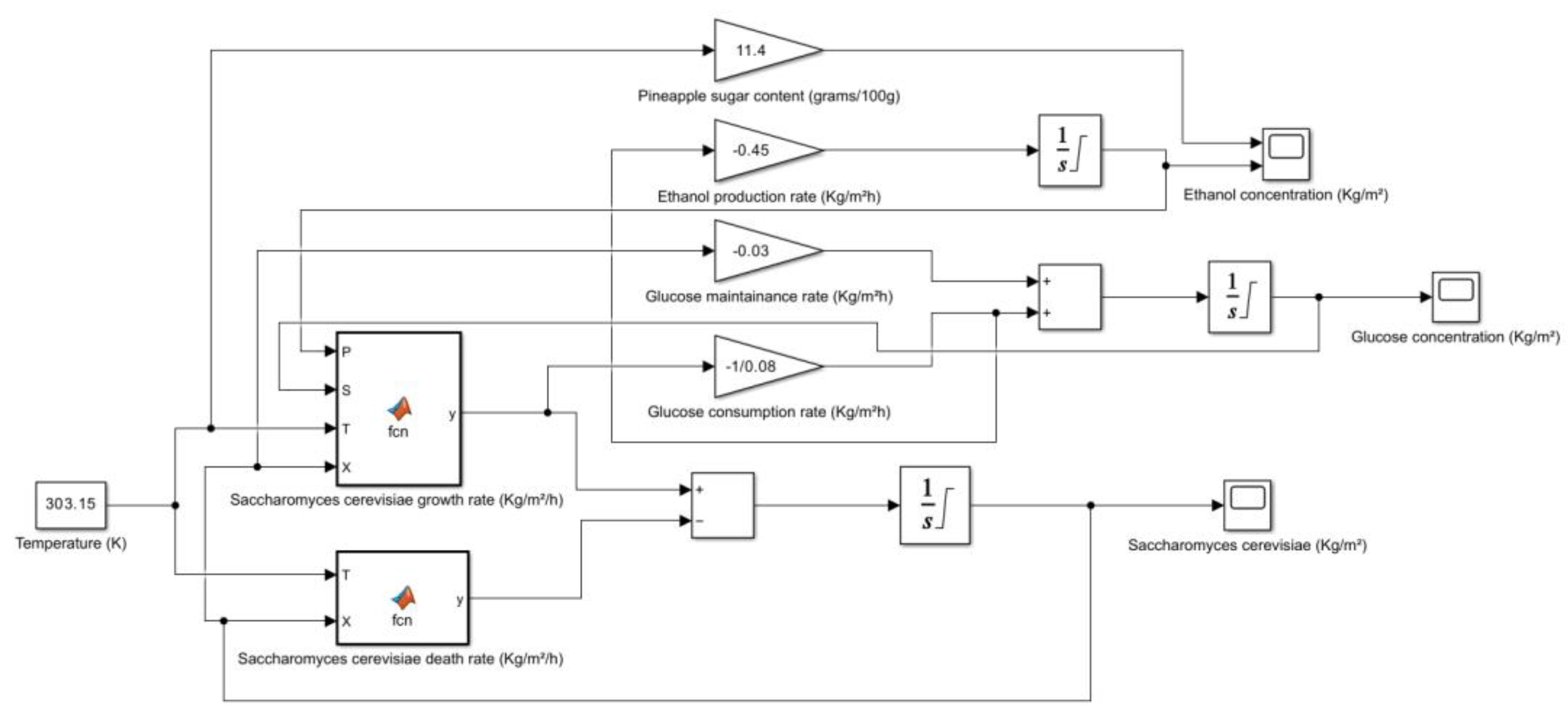
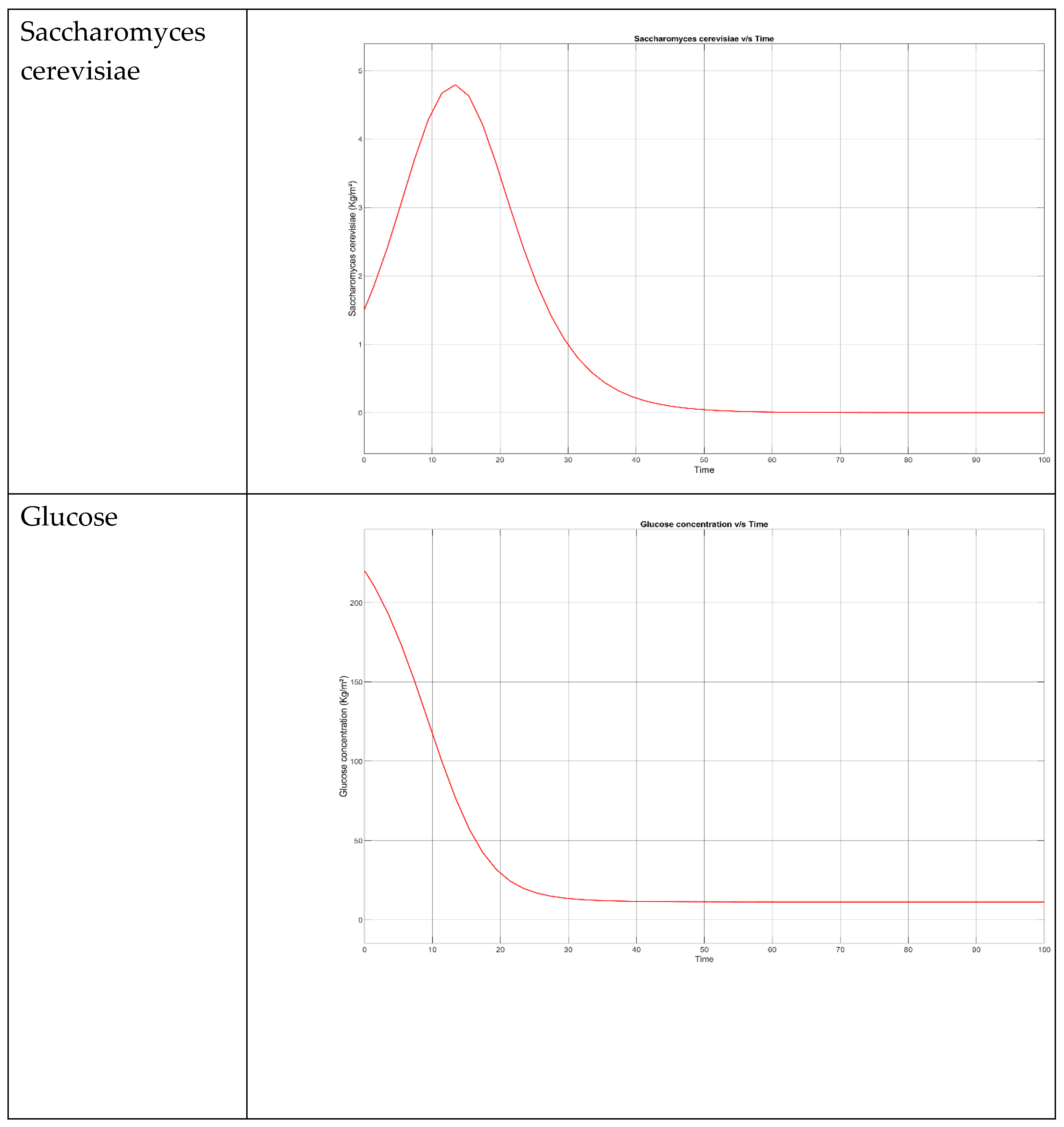
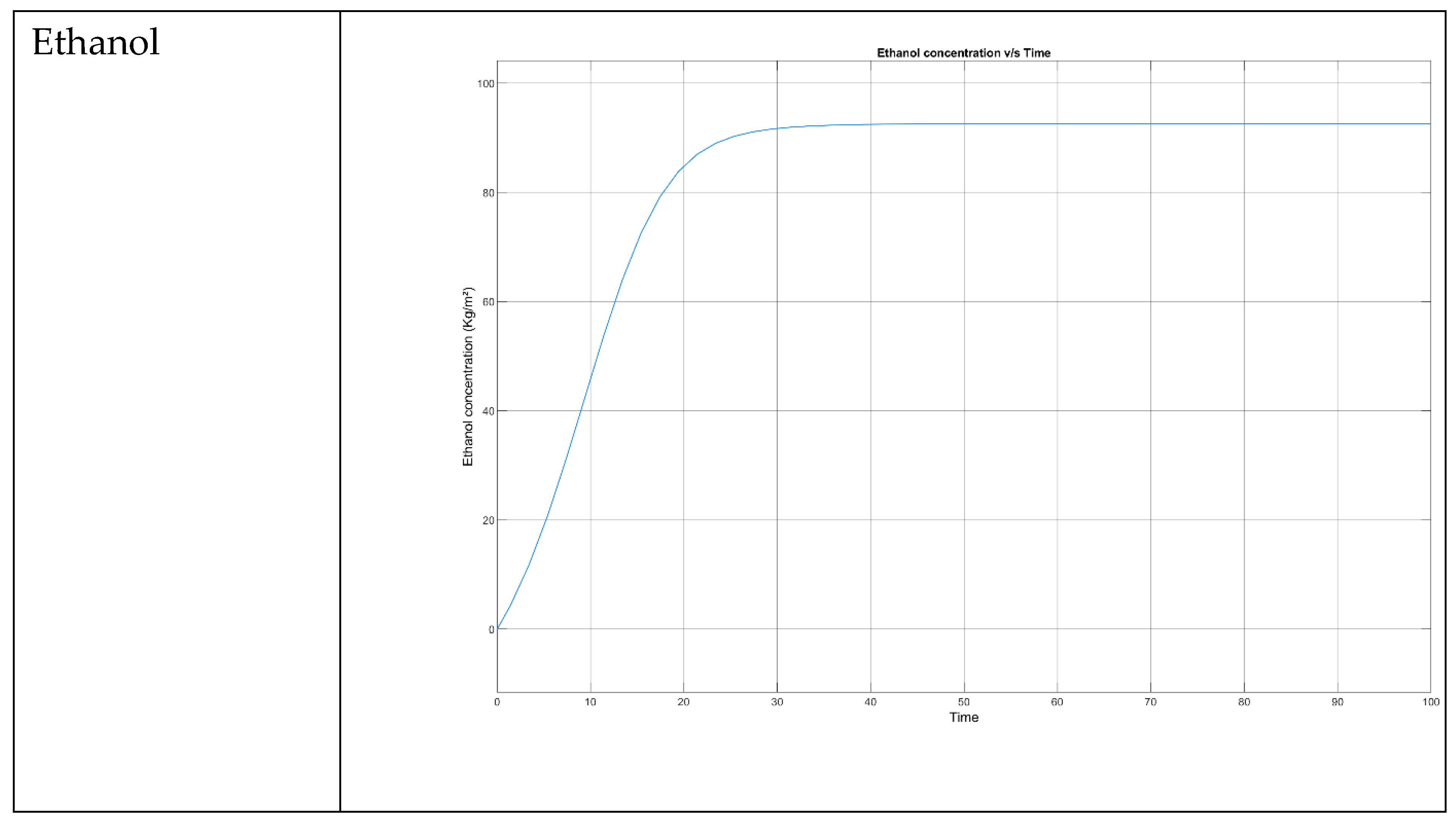
- → No microbial growth takes place as the death rate is dominant.

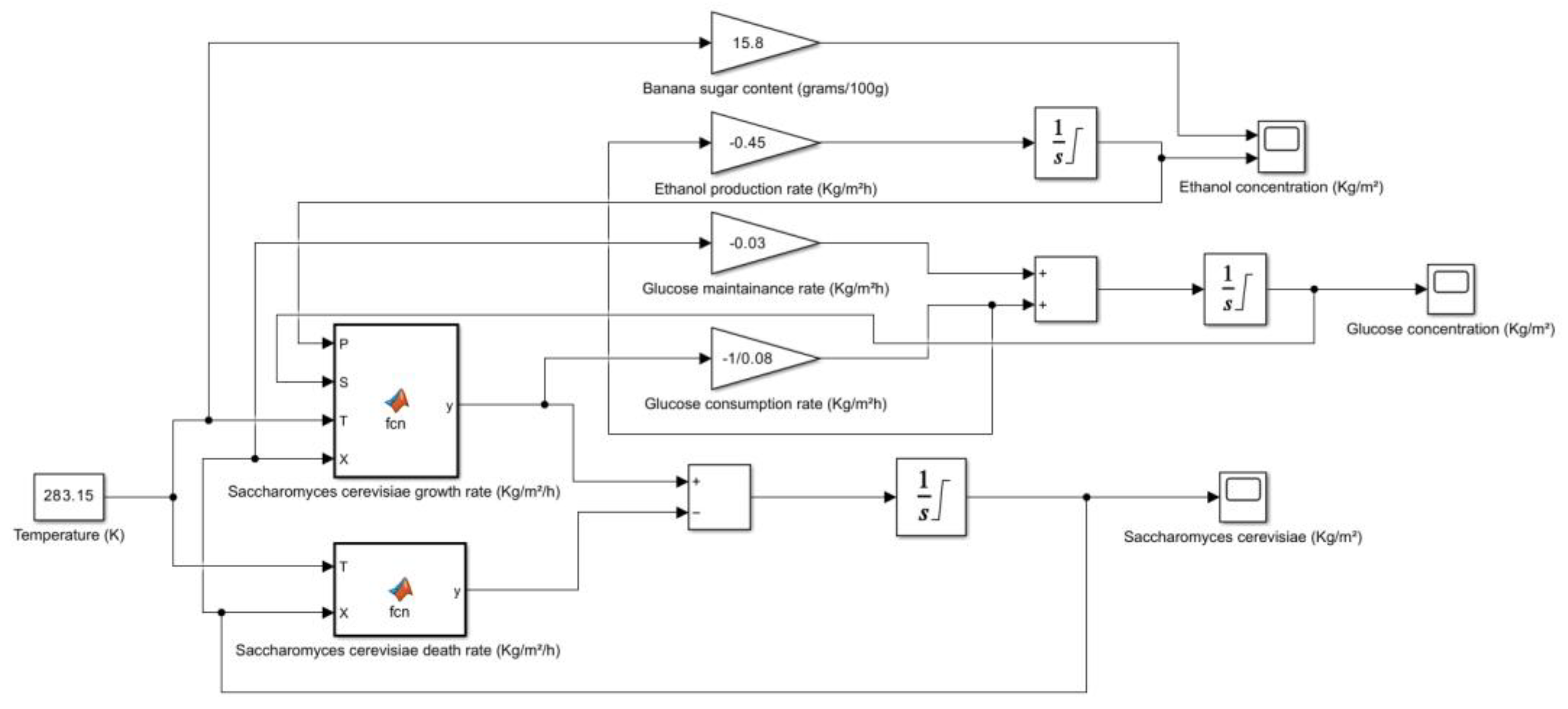
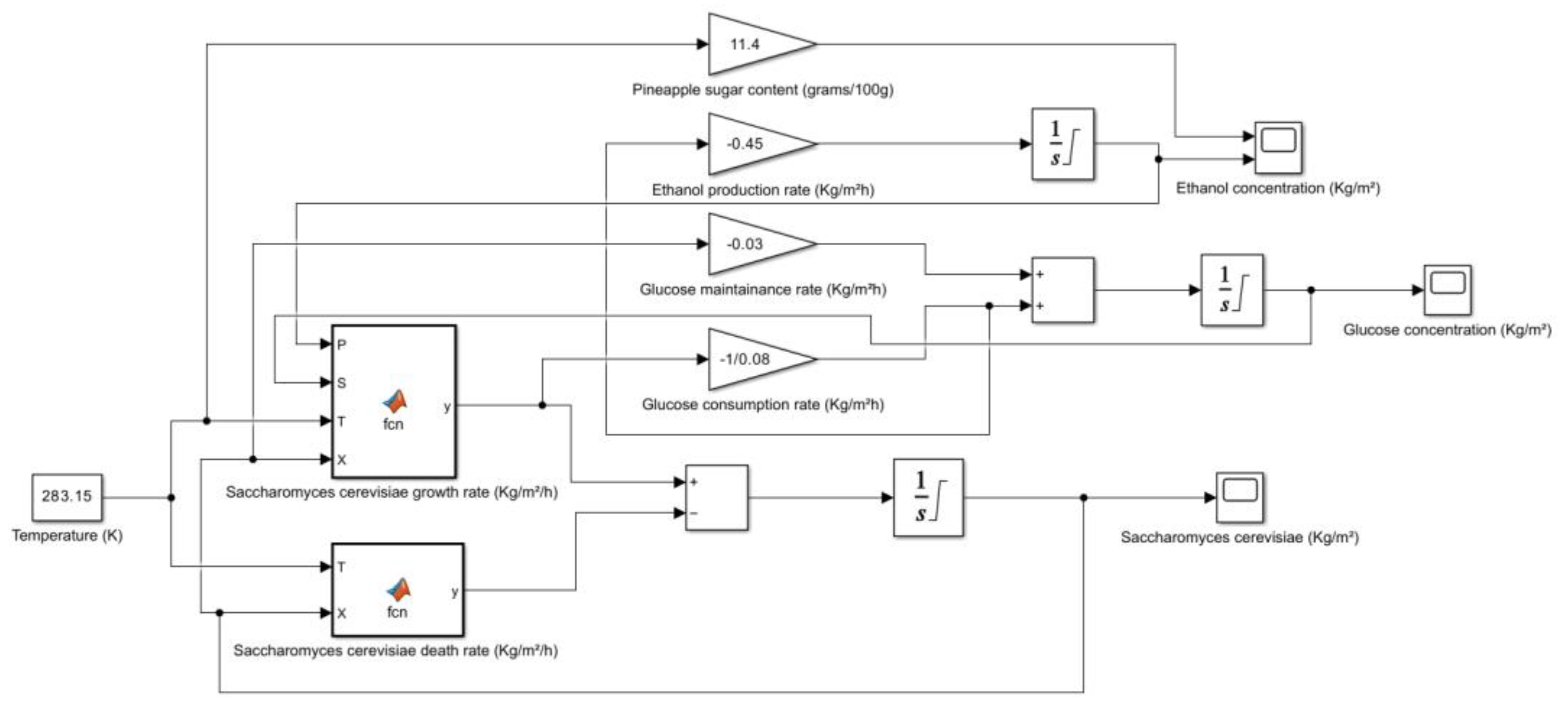
6. Conclusions
Supplementary Materials
Acknowledgments
References
- Ingale, S.; Joshi, S.J.; Gupte, A. Production of bioethanol using agricultural waste: banana pseudo stem. Braz. J. Microbiol. 2014, 45, 885–892. [Google Scholar] [CrossRef]
- A.B.M.S.Hossain1, A Hadeel, K. Mseddi, Nasir, A. Ibrahim, and V. N. V, “Comparative Studies of Bio-ethanol Production from Different Fruits Biomasses,” Global Journal of Life Sciences and Biological Research, vol. 1, no. 2, pp. 1–6, 2024.
- A, H.; A, B.M.S.H.; Khayyat, L.; Haitham, A.; Jama, A.; AlHewiti, N. Bioethanol fuel production from rambutan fruit biomass as reducing agent of global warming and greenhouse gases. Afr. J. Biotechnol. 2011, 10, 10157–10165. [Google Scholar] [CrossRef]
- Alam Imteaz, M.; Hossain, A.B.M.S.; Ahsan, A. Data-driven modelling of bioethanol fuel production from rambutan fruit waste. Proc. Inst. Civ. Eng. - Waste Resour. Manag. 2023, 176, 70–76. [Google Scholar] [CrossRef]
- H. Tanambell, “Application of semi-dried rambutan (nephelium lappaceum l.) peel and ethyl-acetic peel extract to coconut (cocos nucifera l.) sap to delay alcoholic fermentation for alcohol production - Universitas Pelita Harapan Institutional Repository,” Uph.edu, Aug. 2015.
- R. Arumugam and M. Manikanda, “Fermentation of Pretreated Hydrolyzates of Banana and Mango Fruit Wastes for Ethanol Production,” Readkong.com, 2014. https://www.readkong. 6039.
- Awodi, P.; Ogbonna, J.; Nwagu, T. Bioconversion of mango (Mangifera indica) seed kernel starch into bioethanol using various fermentation techniques. Heliyon 2022, 8, e09707. [Google Scholar] [CrossRef]
- L. Veeranjaneya Reddy and O. Vijaya Sarathi Reddy, “Production of Ethanol from Mango (Mangifera indica L.) Fruit Juice Fermentation,” scialert.net, Jul. 03, 2012. https://scialert.net/abstract/?doi=jm.2007.763.
- Palafox-Carlos, H.; Yahia, E.; González-Aguilar, G. Identification and quantification of major phenolic compounds from mango (Mangifera indica, cv. Ataulfo) fruit by HPLC–DAD–MS/MS-ESI and their individual contribution to the antioxidant activity during ripening. Food Chem. 2012, 135, 105–111. [Google Scholar] [CrossRef]
- M. Jahid, A. M. Jahid, A. Gupta, and D. K. Sharma, “Production of Bioethanol from Fruit Wastes (Banana, Papaya, Pineapple and Mango Peels) Under Milder Conditions,” Journal of Bioprocessing & Biotechniques, vol. 08, no. 03, 2018. [CrossRef]
- Waghmare, A.G.; Arya, S.S. Utilization of unripe banana peel waste as feedstock for ethanol production. Bioethanol 2016, 2, 146–156. [Google Scholar] [CrossRef]
- M. Ariff Amir Hamzah, A. Bahari Alias, and N. Elina Ahmad, “A3024109119 - International Journal of Engineering and Advanced Technology (IJEAT),” International Journal of Engineering and Advanced Technology (IJEAT), Dec. 20, 2021. https://www.ijeat.org/portfolio-item/a3024109119/.
- Prakash, H.; Chauhan, P.S.; General, T.; Sharma, A.K. Development of eco-friendly process for the production of bioethanol from banana peel using inhouse developed cocktail of thermo-alkali-stable depolymerizing enzymes. Bioprocess Biosyst. Eng. 2018, 41, 1003–1016. [Google Scholar] [CrossRef]
- E. Bardone et al., “Bioethanol from Fresh and Dried Banana Plant Pseudostem,” Chemical Engineering Transactions, vol. 38, Sep. 2014. [CrossRef]
- Casabar, J.T.; Unpaprom, Y.; Ramaraj, R. Fermentation of pineapple fruit peel wastes for bioethanol production. Biomass- Convers. Biorefinery 2019, 9, 761–765. [Google Scholar] [CrossRef]
- Tropea, A.; Wilson, D.; La Torre, L.G.; Curto, R.B.L.; Saugman, P.; Troy-Davies, P.; Dugo, G.; Waldron, K.W. Bioethanol Production From Pineapple Wastes. J. Food Res. 2014, 3, p60. [Google Scholar] [CrossRef]
- C. Gazey, “Recurring lime applications to fix acidity in the whole soil profile - Grains Research and Development Corporation,” Grains Research and Development Corporation, 2019. https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2019/02/recurring-lime-applications-to-fix-acidity-in-the-whole-soil-profile.
- Brahma, R.; Ray, S. Finding out various potentials and possibilities of jackfruit seed and its usage in the industry: a review. Food Prod. Process. Nutr. 2023, 5, 1–17. [Google Scholar] [CrossRef]
- Sarangi, P.K.; Srivastava, R.K.; Singh, A.K.; Sahoo, U.K.; Prus, P.; Dziekański, P. The Utilization of Jackfruit (Artocarpus heterophyllus L.) Waste towards Sustainable Energy and Biochemicals: The Attainment of Zero-Waste Technologies. Sustainability 2023, 15, 12520. [Google Scholar] [CrossRef]
- Q. Liu, F. Lou, J. Geng, Y. Li, and F. Gao, “Review of research on anaerobic fermentation of food waste,” IOP Conference Series: Earth and Environmental Science, vol. 330, no. 3, p. 032041, Oct. 2019. [CrossRef]
- R. Arif, H. Natsir, H. Rohani, and A. Karim, “Effect of pH fermentation on production bioethanol from jackfruit seeds (Artocarpus heterophyllus) through separate fermentation hydrolysis method,” Journal of Physics: Conference Series, vol. 979, no. 1, p. 012015, Mar. 2018. [CrossRef]
- Nuriana, W.; Wuryantoro Ethanol Synthesis from Jackfruit (Artocarpus Heterophyllus Lam. ) Stone Waste as Renewable Energy Source. Energy Procedia 2015, 65, 372–377. [Google Scholar] [CrossRef]
- Widyaningrum, T.; Utami, L.B.; Prastowo, I.; Meylani, V.; Permadi, A. Comparison of Bioethanol Production Using Saccharomyces cerevisiae and Zymomonas mobilis in Fermented Jackfruit Peel Treated with Blend Crude Cellulose Enzymes. Int. J. Des. Nat. Ecodynamics 2022, 17, 633–637. [Google Scholar] [CrossRef]
- N. Raju and Y. N. Sidabutar, “Effect of durian and jackfruit seed starch combination variation on bioethanol content using Aspergillus niger and Saccharomyces cerevisiae,” IOP Conference Series Earth and Environmental Science, vol. 1302, no. 1, pp. 012118–012118, Feb 2024. [CrossRef]
- Nurhayati, Iis Aisyah, Citra Deliana Dewi Sundari, Risa Rahmawati Sunarya, and Siti Suryaningsih, “Optimization of bioethanol production from jackfruit straw waste through the addition of a starter and fermentation duration,” AIP Conference Proceedings, Jan 2023. [CrossRef]
- Z. Ash et al., “Immobilization of Saccharomyces cerevisiae in Jackfruit (Artocarpus heterophyllus) Seed Fiber for Bioethanol Production,” ASEAN Journal of Chemical Engineering, vol. 22, no. 1, pp. 141–155, 2022.
- Chriswardana, T.H.; Mulyaningsih, Y.; Bahar, A.H.; Riayatsyah, T.M.I. Optimization of sugar production from Durian seeds via alkaline hydrolysis for second-generation bioethanol production. Clean Energy 2021, 5, 375–386. [Google Scholar] [CrossRef]
- K. A. Ghazali, S. F. Salleh, T. M. I. Riayatsyah, H. B. Aditiya, and T. M. I. Mahlia, “The effect of dilute acid pre-treatment process in bioethanol production from durian (Durio zibethinus) seeds waste,” IOP Conference Series: Earth and Environmental Science, vol. 32, p. 012058, Mar 2016. [CrossRef]
- Purnomo, A.; Yudiantoro, Y.A.W.; Putro, J.N.; Nugraha, A.T.; Irawaty, W.; Ismadji, S. Subcritical water hydrolysis of durian seeds waste for bioethanol production. Int. J. Ind. Chem. 2015, 7, 29–37. [Google Scholar] [CrossRef]
- Q. H. Seer, J. Nandong, and T. Shanon, “Experimental study of bioethanol production using mixed cassava and durian seed,” IOP Conference Series: Materials Science and Engineering, vol. 206, p. 012020, Jun 2017. [CrossRef]
- Hermansyah, T. Xayasene, N. Huu Tho, Miksusanti, Fatma, and A. T. Panagan, “Bioethanol Production from Cassava (Manihot esculenta) Peel Using Yeast Isolated from Durian (Durio zhibetinus),” Journal of Physics: Conference Series, vol. 1095, p. 012016, Sep 2018. [CrossRef]
- G. DUSSAP, “Biofuels: Alternative Feedstocks and Conversion Processes for the Production of Liquid and Gaseous Biofuels,” Biofuels: Alternative Feedstocks and Conversion Processes for the Production of Liquid and Gaseous Biofuels, 2019. https://www.academia.edu/111415130/Biofuels_Alternative_Feedstocks_and_Conversion_Processes_for_the_Production_of_Liquid_and_Gaseous_Biofuels.
- Contreras, J.M.R.; Berasain, M.D.M.; Martínez, D.L.P.; Velarde, E.D.A.; Otero, A.M.R. Ethanol and aqueous extracts characterization from guava (Psidium guajava L.) and avocado (Persea americana Mill.) residues. Food Sci. Technol. 2021, 41, 596–602. [Google Scholar] [CrossRef]
- Srivastava, S.; Modi, D.; Garg, S. Production of ethanol from guava pulp by yeast strains. Bioresour. Technol. 1997, 60, 263–265. [Google Scholar] [CrossRef]
- Silveira-Junior, E.G.; Perez, V.H.; Justo, O.R.; David, G.F.; Simionatto, E.; de Oliveira, L.C.S. Valorization of guava (Psidium guajava L.) seeds for levoglucosan production by fast pyrolysis. Cellulose 2020, 28, 71–79. [Google Scholar] [CrossRef]
- Naik, Angothu; et al., “Production Of Renewable Future Fuel (Bioethanol) From Various Sources Through Simultaneous Saccharification And Fermentation,” 2023.
- Widyaningrum, T.; Parahadi, M. Bioethanol Levels of Dragon Fruit (Hylocereus polyrhizus) Peel with the Addition of Blend Crude Cellulase Enzyme from Trichoderma reesei and Aspergillus niger. J. Trop. Biodivers. Biotechnol. 2020, 5, 1–5. [Google Scholar] [CrossRef]
- S. Sarungu et al., “Production of Bioethanol from Dragon Fruit Wastes by Using Aspergillus niger and Saccharomyces cerevisae,” Unmul.ac.id, vol. 16, 2021.
- M. N. Ali and M. Khan, “Ethanol Fuel Production Through Microbial Extracellular Enzymatic Hydrolysis And Fermentation From Renewable Agrobased Cellulosic Wastes,” Ijpbs.net, vol. Volume 2, no. Issue 2, pp. 321–331, 2015.
- Widmer, W.; Zhou, W.; Grohmann, K. Pretreatment effects on orange processing waste for making ethanol by simultaneous saccharification and fermentation. Bioresour. Technol. 2010, 101, 5242–5249. [Google Scholar] [CrossRef]
- Anak Agung Istri Ratnadewi, Indras Dwi Anggita, R. Safitri, Firda Marta Safitri, and Boy Arief Fachri, “Cellulose hydrolysis process of red dragon fruit pell (Hylocereus costaricensis) as candidate for bioethanol production,” AIP conference proceedings, vol. 2911, pp. 030006–030006, Jan 2023. [CrossRef]
- Omar, S.; Rudi, N.N.; Muhamad, M.S.; Hamidon, N.; Hamid, N.H.A. Bioethanol made from Muskmelon (Cucumis Melo) Peels as an Alternative Fuel. Int. J. Recent Technol. Eng. 2019, 8, 321–324. [Google Scholar] [CrossRef]
- Rico, X.; Yáñez, R.; Gullón, B. Evaluation of strategies for enhanced bioethanol production from melon peel waste. Fuel 2022, 334. [Google Scholar] [CrossRef]
- Carrillo-Nieves, D.; Ruiz, H.A.; Aguilar, C.N.; Ilyina, A.; Parra-Saldivar, R.; Torres, J.A.; Hernández, J.L.M. Process alternatives for bioethanol production from mango stem bark residues. Bioresour. Technol. 2017, 239, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Ünal, M.Ü.; Chowdhury, G.; Şener, A. Effect of temperature and nitrogen supplementation on bioethanol production from waste bread, watermelon and muskmelon by Saccharomyces cerevisiae. Biofuels 2020, 13, 395–399. [Google Scholar] [CrossRef]
- G. Chowdhury and M. Ümit, “A STUDY ON BIOETHANOL PRODUCTION FROM WASTE BREAD, WATERMELON AND MUSKMELON BY Saccharomyces cerevisiae ATIK EKMEK, KAVUN VE KARPUZDAN Saccharomyces cerevisiae.
- MC, M.; R, C.; NA, A.; G, R.; Gl, B.; L, R.; G, T.; S, L. Studies on efficient utilization of fruit peel waste for Bio-Ethanol production. J. Pharmacogn. Phytochem. 2023, 12, 141–145. [Google Scholar] [CrossRef]
- Shringala Thimappa Girisha, “GREEN CONVERSION OF WASTE FRUIT RINDS IN TO BIOETHANOL,” Journal of Advanced Scientific Research, vol. 10, no. 02, pp. 23–29, 2019.
- Cho, E.J.; Park, C.S.; Bae, H. Transformation of cheaper mangosteen pericarp waste into bioethanol and chemicals. J. Chem. Technol. Biotechnol. 2018, 95, 348–355. [Google Scholar] [CrossRef]
- Eun Jin Cho, Chan Song Park, Gahui Gwak, and Hyeun-Jong Bae, “IJASEAT Production of Bioethanol and Biomaterials from Mangosteen Peelwaste by Popping Pretreatment,” Iraj.in, 2019.
- Zamila Mohd Senu et al., “Production of Glucose, Galactose and Mannose from the Skins of Durian and Mangosteen,” Springer eBooks, pp. 123–130, Jan. 2015. [CrossRef]
- Ahmad, M.; Yamin, B.M.; Lazim, A.M. A study on dispersion and characterisation of α-mangostin loaded pH sensitive microgel systems. BMC Chem. 2013, 7, 85–85. [Google Scholar] [CrossRef]
- Gutierrez-Orozco, F.; Failla, M.L. Biological Activities and Bioavailability of Mangosteen Xanthones: A Critical Review of the Current Evidence. Nutrients 2013, 5, 3163–3183. [Google Scholar] [CrossRef]
- Yodhnu, S.; Sirikatitham, A.; Wattanapiromsakul, C. Validation of LC for the Determination of -Mangostin in Mangosteen Peel Extract: A Tool for Quality Assessment of Garcinia mangostana L. J. Chromatogr. Sci. 2009, 47, 185–189. [Google Scholar] [CrossRef]
- Hermansyah; Novia; Sugiyama, M.; Harashima, S. Candida tropicalis Isolated from Tuak, a North Sumatera-Indonesian Traditional Beverage, for Bioethanol Production. Microbiol. Biotechnol. Lett. 2015, 43, 241–248. [CrossRef]
- Van Nguyen, P.; Nguyen, K.H.V.; Nguyen, N.L.; Ho, X.T.T.; Truong, P.H.; Nguyen, K.C.T. Lychee-Derived, Thermotolerant Yeasts for Second-Generation Bioethanol Production. Fermentation 2022, 8, 515. [Google Scholar] [CrossRef]
- Maldonado-Celis, M.E.; Yahia, E.M.; Bedoya, R.; Landázuri, P.; Loango, N.; Aguillón, J.; Restrepo, B.; Ospina, J.C.G. Chemical Composition of Mango (Mangifera indica L.) Fruit: Nutritional and Phytochemical Compounds. Front. Plant Sci. 2019, 10, 1073. [Google Scholar] [CrossRef]
- Hu, K.; Dars, A.G.; Liu, Q.; Xie, B.; Sun, Z. Phytochemical profiling of the ripening of Chinese mango (Mangifera indica L.) cultivars by real-time monitoring using UPLC-ESI-QTOF-MS and its potential benefits as prebiotic ingredients. Food Chem. 2018, 256, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Bangar, S.P.; Kumar, M.; Whiteside, W.S.; Tomar, M.; Kennedy, J.F. Litchi (Litchi chinensis) seed starch: Structure, properties, and applications - A review. Carbohydr. Polym. Technol. Appl. 2021, 2. [Google Scholar] [CrossRef]
- Unpaprom, Y.; Saetang, N.; Tipnee, S. Evaluation of mango, longan and lychee trees pruning leaves for the production of biogas via anaerobic fermentation. Maejo Int. J. Energy Environ. Commun. 2019, 1, 20–26. [Google Scholar] [CrossRef]
- Singh, Y.; Singh, D.; Singh, N.K.; Sharma, A.; Rahim, E.A.; Ranganathan, A.; Palanichamy, P.; Palamanit, A.; Kumar, S. Production of bio-oil from lychee-based biomass through pyrolysis and maximization of bio-oil yield with statistical and machine learning techniques. J. Clean. Prod. 2023, 413. [Google Scholar] [CrossRef]
- Sai-Ut, S.; Kingwascharapong, P.; Mazumder, A.R.; Rawdkuen, S. Optimization of Ethanolic Extraction of Phenolic Antioxidants from Lychee and Longan Seeds Using Response Surface Methodology. Foods 2023, 12, 2827. [Google Scholar] [CrossRef]
- Abambagade Abera Mitiku and Tanje Mada Hatsa, “Bioethanol Production from Decaying Fruits Peel Using Saccharomyces cerevisiae,” International Journal of Current Research and Academic Review, vol. 8, no. 5, pp. 50–59, May 2020. [CrossRef]
- Alves, J.A.; Lima, L.C.d.O.; Nunes, C.A.; Dias, D.R.; Schwan, R.F. Chemical, Physical–Chemical, and Sensory Characteristics of Lychee (Litchi chinensis Sonn) Wines. J. Food Sci. 2011, 76, S330–S336. [Google Scholar] [CrossRef]
- Azhar, S.H.M.; Abdulla, R.; Jambo, S.A.; Marbawi, H.; Gansau, J.A.; Faik, A.A.M.; Rodrigues, K.F. Yeasts in sustainable bioethanol production: A review. Biochem. Biophys. Rep. 2017, 10, 52–61. [Google Scholar] [CrossRef]
- Zimmermann, A.; Kaltschmitt, M. Quantification of fructans in cereal-based bioethanol stillage based on a simplified analytical method. Cereal Chem. 2022, 99, 737–750. [Google Scholar] [CrossRef]
- Mark, R. Wilkins, Wilbur W. Widmer, and Karel Grohmann, “Enzymatic Hydrolysis of Grapefruit Peel to Produce Ethanol and Other Products,” 2005 Tampa, FL July 17-20, 2005, 2005. [Google Scholar] [CrossRef]
- M. Jayakumar, S. M. Jayakumar, S. Kuppusamy Vaithilingam, N. Karmegam, K. B. Gebeyehu, M. S. Boobalan, and B. Gurunathan, “Chapter 6 - Fermentation technology for ethanol production: current trends and challenges,” ScienceDirect, Jan. 01, 2022. https://www.sciencedirect.com/science/article/abs/pii/B9780323900409000151.
- O. P. Ward and A. K. Singh, “Bioethanol Technology: Developments and Perspectives,” Advances in Applied Microbiology, pp. 53–80, Jan 2002. [CrossRef]
- Saini, R.; Chen, C.-W.; Patel, A.K.; Saini, J.K.; Dong, C.-D.; Singhania, R.R. Valorization of Pineapple Leaves Waste for the Production of Bioethanol. Bioengineering 2022, 9, 557. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, “Production and Investigation of Parametric Effect on Bio-ethanol by Sapota Using Separation Technique,” Korean Chemical Engineering Research, vol. 61, no. 2, pp. 234–239, 2023. [CrossRef]
- Fernandes, F.d.S.; de Souza, É.S.; Carneiro, L.M.; Silva, J.P.A.; de Souza, J.V.B.; Batista, J.d.S. Current Ethanol Production Requirements for the Yeast Saccharomyces cerevisiae. Int. J. Microbiol. 2022, 2022, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ruchala, J.; Kurylenko, O.O.; Dmytruk, K.V.; Sibirny, A.A. Construction of advanced producers of first- and second-generation ethanol in Saccharomyces cerevisiae and selected species of non-conventional yeasts (Scheffersomyces stipitis, Ogataea polymorpha). J. Ind. Microbiol. Biotechnol. 2020, 47, 109–132. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, P.; Wu, T.; Ruan, H. Bioethanol Production Based on Saccharomyces cerevisiae: Opportunities and Challenges. Fermentation 2023, 9, 709. [Google Scholar] [CrossRef]
- Tse, T.J.; Wiens, D.J.; Reaney, M.J.T. Production of Bioethanol—A Review of Factors Affecting Ethanol Yield. Fermentation 2021, 7, 268. [Google Scholar] [CrossRef]
- Khatun, F.; Islam, Z.; Habib, A.; Saha, S.; Yasmin, S. SUSTAINABLE UTILIZATION OF FRUIT WASTES FOR PRODUCTION OF BIOETHANOL USING THERMOTOLERANT SACCHAROMYCES CEREVISIAE YEAST ISOLATED FROM COMMON FRUITS OF BANGLADESH. J. Anim. Plant Sci. 2022, 33, 440–452. [Google Scholar] [CrossRef]
- Gronchi, N.; De Bernardini, N.; Cripwell, R.A.; Treu, L.; Campanaro, S.; Basaglia, M.; Foulquié-Moreno, M.R.; Thevelein, J.M.; Van Zyl, W.H.; Favaro, L.; et al. Natural Saccharomyces cerevisiae Strain Reveals Peculiar Genomic Traits for Starch-to-Bioethanol Production: the Design of an Amylolytic Consolidated Bioprocessing Yeast. Front. Microbiol. 2022, 12, 768562. [Google Scholar] [CrossRef]
- Gonçalves, F.A.; Ruiz, H.A.; dos Santos, E.S.; Teixeira, J.A.; de Macedo, G.R. Bioethanol production by Saccharomyces cerevisiae, Pichia stipitis and Zymomonas mobilis from delignified coconut fibre mature and lignin extraction according to biorefinery concept. Renew. Energy 2016, 94, 353–365. [Google Scholar] [CrossRef]
- Shah, K.R.; Vyas, R.; Patel, G. Bioethanol production from pulp of fruits. Biosci. Biotechnol. Res. Commun. 2019, 12, 464–471. [Google Scholar] [CrossRef]
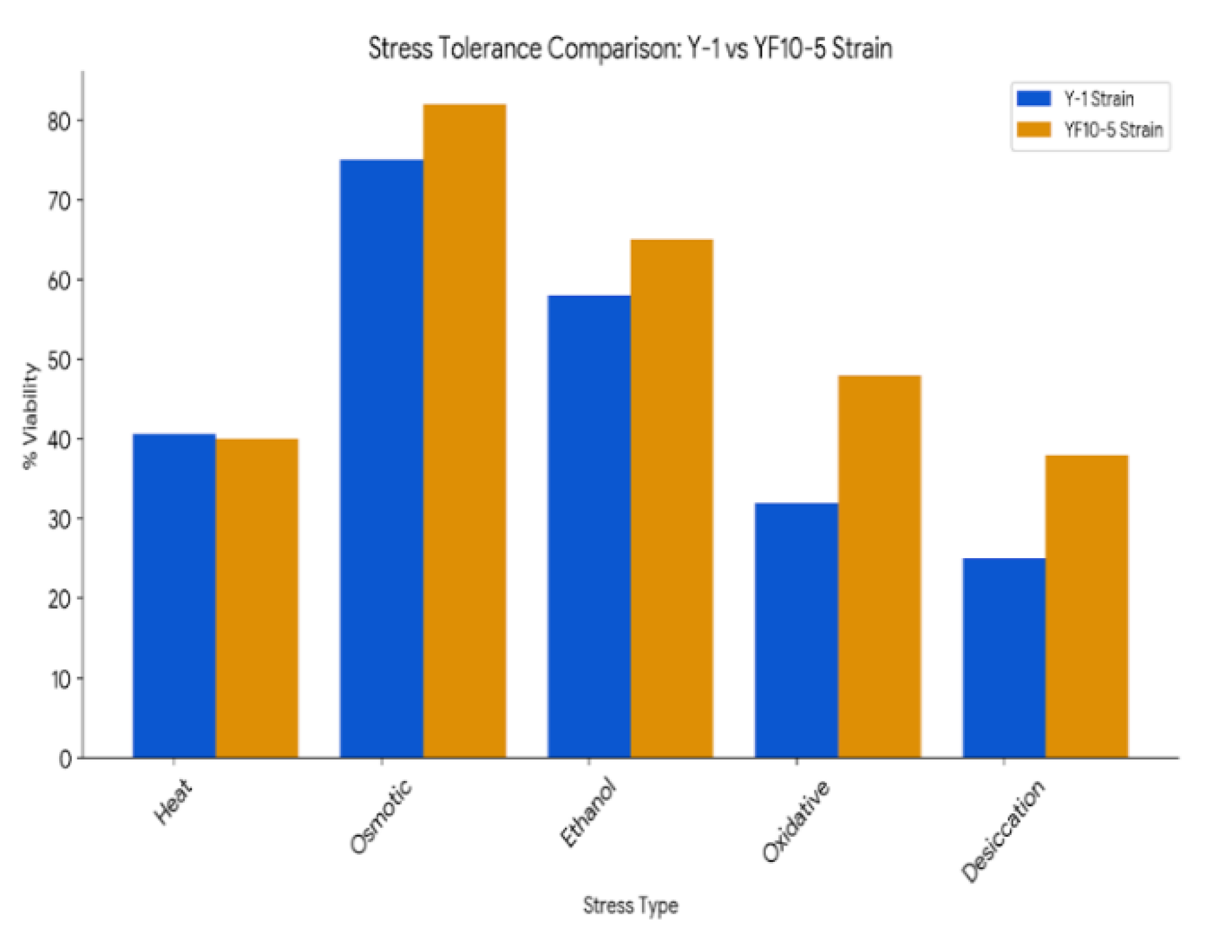
- Zhang, Q.; Jin, Y.-L.; Fang, Y.; Zhao, H. Adaptive evolution and selection of stress-resistant Saccharomyces cerevisiae for very high-gravity bioethanol fermentation. Electron. J. Biotechnol. 2019, 41, 88–94. [Google Scholar] [CrossRef]
- Tesfaw, A.; Assefa, F. Current Trends in Bioethanol Production by Saccharomyces cerevisiae: Substrate, Inhibitor Reduction, Growth Variables, Coculture, and Immobilization. Int. Sch. Res. Not. 2014, 2014, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Jiang, S.; Lu, S.; Jiang, S.; Jiang, S.; Deng, Y.; Lu, J.; Wang, H.; Zhou, Y. Ethanol yield improvement in Saccharomyces cerevisiae GPD2 Delta FPS1 Delta ADH2 Delta DLD3 Delta mutant and molecular mechanism exploration based on the metabolic flux and transcriptomics approaches. Microb. Cell Factories 2022, 21, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Varize, C.S.; Bücker, A.; Lopes, L.D.; Christofoleti-Furlan, R.M.; Raposo, M.S.; Basso, L.C.; Stambuk, B.U. Increasing Ethanol Tolerance and Ethanol Production in an Industrial Fuel Ethanol Saccharomyces cerevisiae Strain. Fermentation 2022, 8, 470. [Google Scholar] [CrossRef]
- Fentahun, M.; Andualem, B. Optimization of bioethanol production using stress-tolerant yeast strains isolated from household alcoholic beverages (Tella, Tej, and Areke) and molasses (as substrate). F1000Research 2024, 13, 286. [Google Scholar] [CrossRef]
- John, I.; Pola, J.; Thanabalan, M.; Appusamy, A. Bioethanol Production from Musambi Peel by Acid Catalyzed Steam Pretreatment and Enzymatic Saccharification: Optimization of Delignification Using Taguchi Design. Waste Biomass- Valorization 2019, 11, 2631–2643. [Google Scholar] [CrossRef]
- Aleman-Ramirez, J.; Pérez-Sariñana, B.; Torres-Arellano, S.; Saldaña-Trinidad, S.; Longoria, A.; Sebastian, P. Bioethanol production from Ataulfo mango supplemented with vermicompost leachate. Catal. Today 2019, 353, 173–179. [Google Scholar] [CrossRef]
- Aminu, B.; Bukar, A.; Ado, A.; Bello, M. Bioethanol production from two varieties of mango kernel using co-culture of Bacillus subtilis, Aspergillus niger and Saccharomyces cerevisiea. Bayero J. Pure Appl. Sci. 2019, 11, 349. [Google Scholar] [CrossRef]
- M. Khandaker, M. Saifuddin, A. Hossain, N. Mat, and A. Boyce, “Bioethanol Production from Mango Waste (Mangifera indica L. cv chokanan): Biomass as Renewable Energy,” Australian Journal of Basic and Applied Sciences, vol. 8, no. 9, pp. 229–237, 2014, Accessed: Oct. 16, 2024.
- Yesmin, M.N.; Azad, M.A.K.; Uddin, M.N.; Khatun, J.A.; Hossain, M.A. Bioethanol Production from Different Varieties of Over-ripen Mango (Mangifera indica L.) of Bangladesh. Mod. Chem. 2021, 9, 25. [Google Scholar] [CrossRef]
- M. Muhammad and T. and E. Arid Zone Journal of Engineering, “COMPARISM OF THE PROPERTIES AND YIELD OF BIOETHANOL FROM MANGO AND ORANGE WASTE,” www.academia.edu, vol. 13, Dec. 2017.
- Patel, V.; Tripathi, A.D.; Adhikari, K.S.; Srivastava, A. Screening of physicochemical and functional attributes of fermented beverage (wine) produced from local mango (Mangifera indica) varieties of Uttar Pradesh using novel saccharomyces strain. J. Food Sci. Technol. 2020, 58, 2206–2215. [Google Scholar] [CrossRef]
- Hoang, V.L.T.; Pierson, J.-T.; Curry, M.C.; Shaw, P.N.; Dietzgen, R.G.; Gidley, M.J.; Roberts-Thomson, S.J.; Monteith, G.R. Polyphenolic contents and the effects of methanol extracts from mango varieties on breast cancer cells. Food Sci. Biotechnol. 2015, 24, 265–271. [Google Scholar] [CrossRef]
- Lebaka, V.R.; Wee, Y.-J.; Ye, W.; Korivi, M. Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit. Int. J. Environ. Res. Public Health 2021, 18, 741. [Google Scholar] [CrossRef]
- Vijayanand, P.; Deepu, E.; Kulkarni, S.G. Physico chemical characterization and the effect of processing on the quality characteristics of Sindura, Mallika and Totapuri mango cultivars. J. Food Sci. Technol. 2013, 52, 1047–1053. [Google Scholar] [CrossRef]
- Dars, A.G.; Hu, K.; Liu, Q.; Abbas, A.; Xie, B.; Sun, Z. Effect of Thermo-Sonication and Ultra-High Pressure on the Quality and Phenolic Profile of Mango Juice. Foods 2019, 8, 298. [Google Scholar] [CrossRef] [PubMed]
- Varakumar, S.; Kumar, Y.S.; Reddy, O.V.S. CAROTENOID COMPOSITION OF MANGO (MANGIFERA INDICA L.) WINE AND ITS ANTIOXIDANT ACTIVITY. J. Food Biochem. 2011, 35, 1538–1547. [Google Scholar] [CrossRef]
- Chew, Kit Wayne, Wen Yi Chia, and Dina Maged Salah Mohammed Tawfik, “ Utilization of agricultural production waste for potential bioethanol generation,” 2022. https://www.abrinternationaljournal.org/articles/utilization-of-agricultural-production-waste-for-potential-bioethanol-generation.
- Hossain, A.S.; Uddin, M.M. Fruit Bio-Waste Derived Bio-Ethanol Production and Bioelectricity Generation as Renewable Energy. Am. J. Environ. Sci. 2021, 17, 82–91. [Google Scholar] [CrossRef]
- Palanisamy, U.; Cheng, H.M.; Masilamani, T.; Subramaniam, T.; Ling, L.T.; Radhakrishnan, A.K. Rind of the rambutan, Nephelium lappaceum, a potential source of natural antioxidants. Food Chem. 2008, 109, 54–63. [Google Scholar] [CrossRef]
- Albuquerque, B.R.; Pinela, J.; Dias, M.I.; Pereira, C.; Petrović, J.; Soković, M.; Calhelha, R.C.; Oliveira, M.B.P.; Ferreira, I.C.; Barros, L. Valorization of rambutan (Nephelium lappaceum L.) peel: Chemical composition, biological activity, and optimized recovery of anthocyanins. Food Res. Int. 2023, 165, 112574. [Google Scholar] [CrossRef]
- Akin-Osana, B.; Nzelibe, H.; Agbaji, A. Ethanol Production from Carica papaya (Pawpaw) Fruit Waste. Asian J. Biochem. 2008, 3, 188–193. [Google Scholar] [CrossRef]
- Fagundes, V.D.; Machado, Ê.L.; Schneider, R.d.C.d.S.; Colla, L.M. Life cycle assessment of bioethanol production from banana, potato, and papaya waste. Int. J. Life Cycle Assess. 2024, 29, 1846–1862. [Google Scholar] [CrossRef]
- Jayaprakashvel, M.; Akila, S.; Venkatramani, M.; Vinothini, S.; S, S.; Bhagat, J.; Hussain, A.J. Production of Bioethanol from Papaya and Pineapple Wastes using Marine Associated Microorganisms. Biosci. Biotechnol. Res. Asia 2014, 11, 193–199. [Google Scholar] [CrossRef]
- Oiwoh, O.; Ayodele, B.; Amenaghawon, N.; Okieimen, C. Optimization of bioethanol production from simultaneous saccharification and fermentation of pineapple peels using Saccharomyces cerevisiae. J. Appl. Sci. Environ. Manag. 2018, 22, 54. [Google Scholar] [CrossRef]
- M. H. S. Ginting, Irvan, E. Misran, and S. Maulina, “Potential of durian, avocado and jackfruit seed as raw material of bioethanol: a review,” IOP Conference Series: Materials Science and Engineering, vol. 801, no. 1, p. 012045, May 2020. [CrossRef]
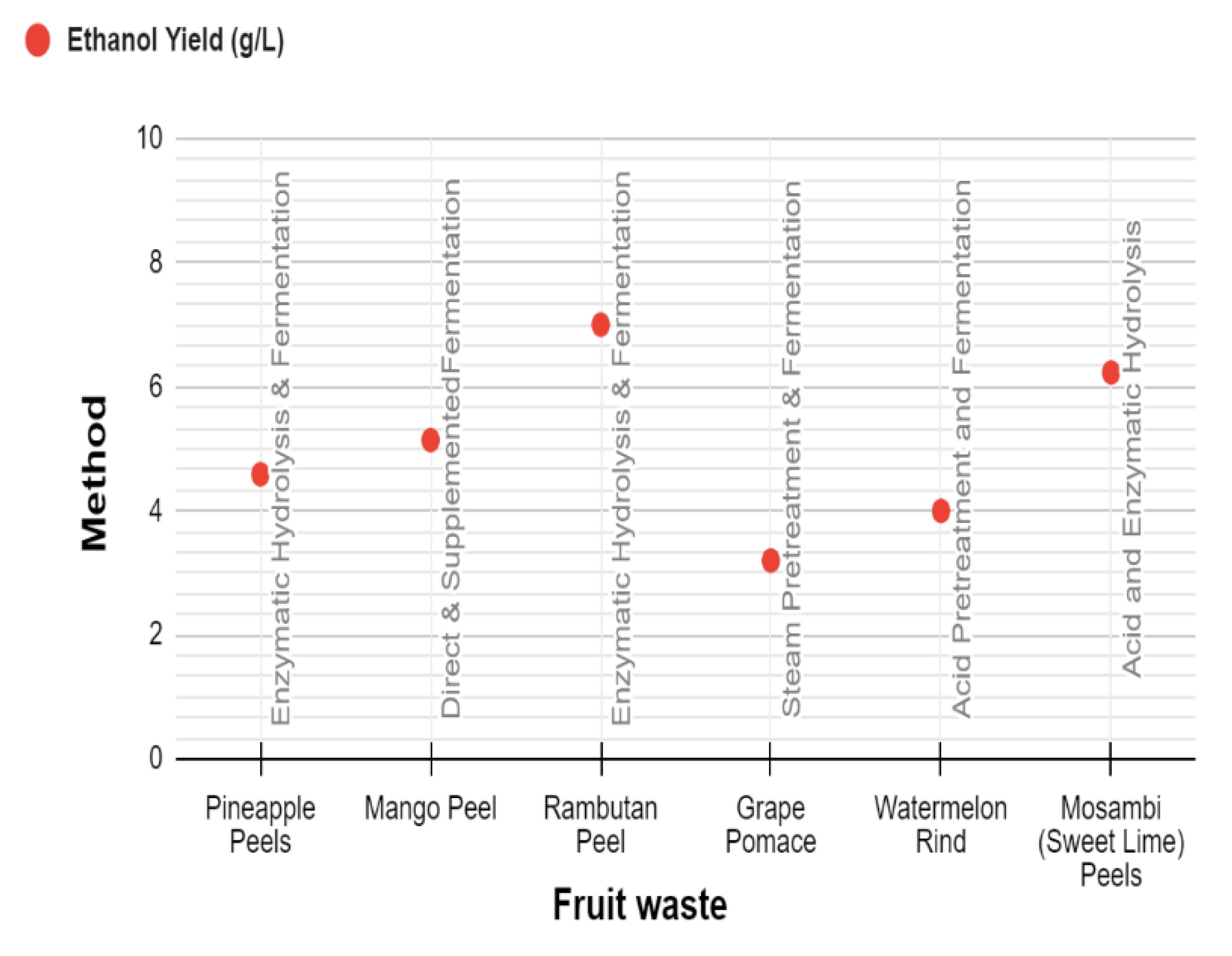
- Mgeni, S.T.; Mero, H.R.; Mtashobya, L.A.; Emmanuel, J.K. The prospect of fruit wastes in bioethanol production: A review. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Triwahyuni, E.; Muryanto; Sudiyani, Y.; Abimanyu, H The Effect of Substrate Loading On Simultaneous Saccharification And Fermentation Process For Bioethanol Production from Oil Palm Empty Fruit Bunches. Energy Procedia 2015, 68, 138–146. [CrossRef]
- Favaretto, D.P.C.; Rempel, A.; Lanzini, J.R.; Silva, A.C.M.; Lazzari, T.; Barbizan, L.D.; Brião, V.B.; Colla, L.M.; Treichel, H. Fruit residues as biomass for bioethanol production using enzymatic hydrolysis as pretreatment. World J. Microbiol. Biotechnol. 2023, 39, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ucuncu, C.; Tari, C.; Demir, H.; Buyukkileci, A.O.; Ozen, B. Dilute-Acid Hydrolysis of Apple, Orange, Apricot and Peach Pomaces as Potential Candidates for Bioethanol Production. J. Biobased Mater. Bioenergy 2013, 7, 376–389. [Google Scholar] [CrossRef]
- Mushimiyimana and, P. Tallapragada, “Bioethanol Production from Agro Wastes by Acid Hydrolysis and Fermentation Process,” Journal of Scientific & Industrial Research, vol. 75, pp. 383–388, 2016.
- Ahmed, B.; Mabrouk, K.; Cherif, K.; Boudjemaa, B. Bioethanol production from date palm fruit waste fermentation using solar energy. Afr. J. Biotechnol. 2016, 15, 1621–1627. [Google Scholar] [CrossRef]
- Kularathne, I.W.; Gunathilaka, C.A.; Ratnaweera, A.C.; Kalpage, C.S.; Rajapakse, S.; Gamage, P. Optimization of Fermentation Process Parameters for Bioethanol Production from Sri Lankan Overripe Fruits. Eng. J. Inst. Eng. Sri Lanka 2021, 54, 77–86. [Google Scholar] [CrossRef]
- Choi, I.S.; Lee, Y.G.; Khanal, S.K.; Park, B.J.; Bae, H.-J. A low-energy, cost-effective approach to fruit and citrus peel waste processing for bioethanol production. Appl. Energy 2015, 140, 65–74. [Google Scholar] [CrossRef]
- Cutzu, R.; Bardi, L. Production of Bioethanol from Agricultural Wastes Using Residual Thermal Energy of a Cogeneration Plant in the Distillation Phase. Fermentation 2017, 3, 24. [Google Scholar] [CrossRef]
| Mango Variety | Pre-treatment Method | Temp (°C) | Time (minutes) | pH | Enzyme | Comments |
References |
|---|---|---|---|---|---|---|---|
| Baganpalli | Steam explosion | 140 | 8 | 4 | Cellulase | Moderate severity Pre-treatment |
(John et al., 2019; Alema-Ramirez et al., 2020; Khandekar et al., 2024) |
| Langra | Microwave-assisted extraction followed by enzymatic hydrolysis | 120 | 15 | 5 | Pectinase & Cellulase | Sequential Pre-treatment |
(Khandekar et at., 2014; Nilufa et al., 2021) |
| Dashehari | Steam explosion | 150 | 12 | 4 | Cellulase | High glucose content | (Muhammad et al., 2017; Patel et al., 2021) |
| Alphonso | Microwave-assisted extraction | 120 | 15 | 5 | Pectinase | Suitable for preserving flavour | (Le et al., 2015; Vijayanand et al., 2013) |
| Totapuri | Dilute acid hydrolysis | 110 | 45 | 2.5 | Hemicellulase & Cellulase | Requires shorter fermentation time | (Dars et al., 2019; Varakumar et al., 2011) |
| Feedstock | Pre-treatment Method | Temperature (°C) | Time (hours) | pH | Enzymes | Comments | References |
|---|---|---|---|---|---|---|---|
| Rambutan | Ultrasound | 40 | 60 | 4 | Pectinase | Ripe rambutans. Pulp has lower cellulose content and fermentable sugars than skin | (Chew et al., 2022; Hossain et al., 2021; Palanisamy et al., 2008; Albuquerque et al., 2023)) |
| Banana, Mango, Papaya | Acid Hydrolysis (H2SO4) | 60 - 110 | 3 - 48 | 1.0 - 2.0 | Cellulase, β-glucosidase | Highly acidic pH. Minimize sugar degradation and by-product formation (furfural, hydroxymethylfurfural). | (Akin-Osanaiye et al., 2008; Fagundes et al., 2024; Jayprakashvel et al., 2016) |
| Pineapple Peel | Microwave-assisted Alkali Pre-treatment | 80 - 120 | 10 - 20 | 11.0 - 12.0 | Cellulase, β-glucosidase | Saccharomyces cerevisiae WLP300 is used. Microwave pre-treatment can accelerate the process but requires further optimization. Similar to orange peels, alkali pre-treatment might necessitate detoxification steps. |
(Casabar et al., 2019; Tropea et al., 2014; Saini et al., 2022 Oiwoh et al., 2018) |
| Jackfruit Rind | Steam Explosion or Two-stage Acid Hydrolysis | 160 - 190 (Steam) | 1 - 10 (Steam) | Not Applicable (Steam) | 90 - 110 (Stage 1 Acid) | 1.0 - 2.0 (Stage 1 Acid). In fermentation process urea and NPK was used in fermentation | (Trianik et al., 2022; Nurhayati et al., 2023; Ash et al., 2022; Ginting et al., 2020) |
| Durian | Steam explosion | 121 | 60 | 4.5 | Cellulase | Ripened fruit. Alkaline solution like NaOH is used for the process of chemical hydrolysis. | (Purnomo et al., 2015; Seer et al., 2017; Hermansyah et al., 2018) |
| Guava | Acid pretreatment | 50 | 120 | 4.8 | Pectinase, Cellulase | Pink guava variety. Saccharomyces cerevisiae MTCC 1972, Isolate-1, and Isolate-2 is used | (Euripedes et al., 2020; Naik et al., 2023; Srivastava et al., 1997) |
| Dragon Fruit | Enzymatic hydrolysis | 40-50 | 60 | 4.8-5.0 | Pectinase, Cellulase | Red flesh variety. Fungus like Aspergillus niger or Trichoderma reesei to break down complex sugars in the waste | (Widyaningrum et al., 2017; Widmer et al., 2010; Anak et al., 2023; Sarungu et al., 2021) |
| Muskmelon | Steam explosion | 121 | 45 | 4.5-5.0 | Cellulase | Ripe muskmelon. Glucose is used in the first 6 hours. | (Ünal et al ., 2020; Shringala et al., 2019) |
| Mangosteen | Acid and enzymatic | 30 | 120 | 5 | Pectinase , Cellulase | Ripe mangosteen. Popping pre-treatment method is used | (Yodhnu et al., 2009; Ahmad et al., 2013) |
| Lychee | Ultrasound | 50 | 30 | 3.5-4.0 | Pectinase | Fresh lychee. Yeast strains like H1, H19, and H23 showed the max growth | (Nguyen et al., 2022; Bangar et al., 2021; Yuwalee et al., 2019) |
| Stage | Description | Parameters Investigated | Observations made |
References |
|---|---|---|---|---|
| Substrate Preparation | Washing, outer coat removal (optional), size reduction (1-2cm pieces), drying (room temp. & oven), grinding, storage | Break down the complex carbohydrates like cellulose and hemicellulose in the substrate into simpler sugars (glucose) that can be fermented by microorganisms | Grinding enhances the accessibility of cellulose fibres for enzymatic breakdown during hydrolysis. |
(Shedrack et al., 2024; Triwahyuni et al., 2015; Favaretto et al., 2023) |
| Acid Hydrolysis | Break down polysaccharides (cellulose, hemicellulose) into fermentable sugars (glucose, xylose) using H2SO4 as a catalyst. | - Substrate concentration (500g peels) - Acid concentration (0% to 3% v/v) - Temperature (60°C to 110°C) - Reaction time (3 to 48 hours) |
Optimal conditions minimize sugar degradation and by-product formation (furfural, hydroxymethylfurfural). |
(Ucuncu et al., 2013) |
| pH Adjustment | Adjust the pH of the pre-treated and hydrolyzed mixture to optimize the enzyme for fermentation | - Initial pH measured with a digital pH meter (5.0 to 5.5) for fermentation enzymes. | Optimal pH range of 5.0-5.5 for enzyme activity during fermentation. |
(Shah et al., 2019) |
| Fermentation | Conversion of sugars to bioethanol using S. cerevisiae | - Temperature (22°C to 43°C) - pH (4.5 to 6) - Yeast inoculum (0.5 to 3.5 g/L) - Fermentation time (24 to 96 hours) |
Batch fermentation was used for initial studies due to simplicity. The shaking incubator provides a controlled environment. | (Ahmed et al., 2016; Asanga et al., 2021) |
| Yeast Activation | Enhance yeast viability and performance (for active dry yeast) | -Temperature (30 to 35°C) -pH (4.5 to 5.5). -Media composition (e.g., 5% glucose solution, peptone, urea, magnesium sulphate). |
Pre-activation of starting materials can improve ethanol production compared to non-activated yeast. |
(Favaretto et al., 2023) |
| Monitoring & Analysis | Tracks progress and identifies optimal conditions | Daily ethanol concentration measurement by a shake flask method. | Yeast requires essential nutrients like nitrogen which includes ammonium sulphate, urea; phosphorus which includes diammonium phosphate; and vitamins for growth and ethanol production. |
(Seong et al., 2015) |
| Distillation | Purification of bioethanol that is produced during fermentation by separating it from the fermentation broth. | Temperature is around 78.2°C in industries. | The composition of the fermented broth, particularly the ethanol concentration, affects the efficiency of the entire distillation process. |
(Raffaela et al., 2017) |
| Feature | SC-GR Strain | RK1 Strain |
|---|---|---|
| Substrate Specificity | Wide range from glucose and xylose to arabinose is employed | Primarily only glucose is employed |
| Ethanol Yield | Potentially high | Consistent but gives a moderate yield |
| Stress Tolerance | Acidic or Alkaline condition | Tolerance to high temperatures, low pH, and inhibitors |
| Advantages | Versatile sugar utilization, potentially high yields | Reliable, robust, well-characterized |
| Disadvantages | Limited stress tolerance data | Less versatile sugar utilization |
| Fermentation Efficiency | Exhibit higher efficiency up to 80-85% |
Relatively high up to 85-90% |
| Environmental tolerance | Higher tolerance than the RK1 strain | Mild Tolerant of certain factors |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




