Submitted:
11 October 2025
Posted:
13 October 2025
You are already at the latest version
Abstract
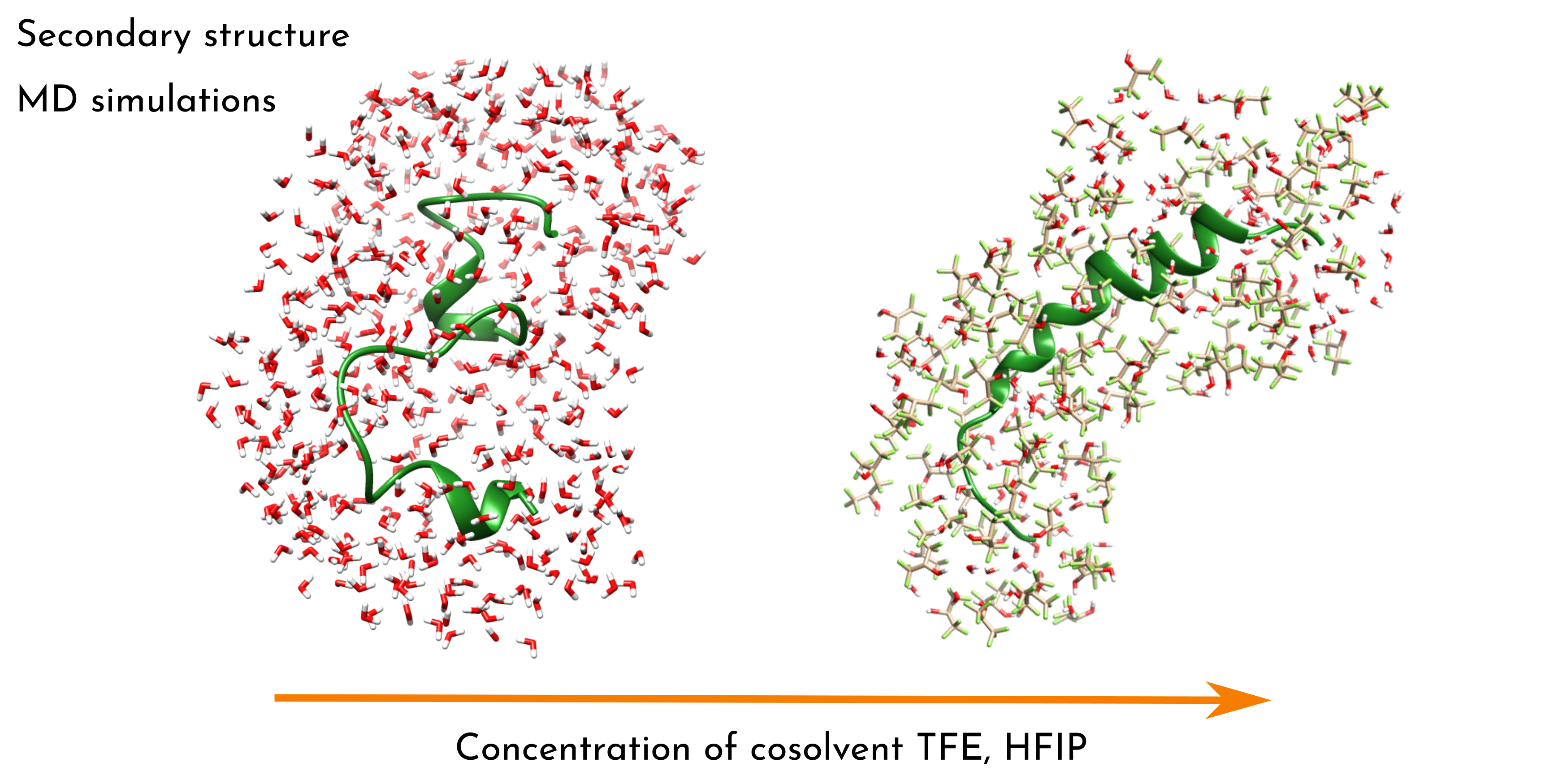
Keywords:
1. Introduction
2. Methods
2.1. Molecular Dynamics Simulations
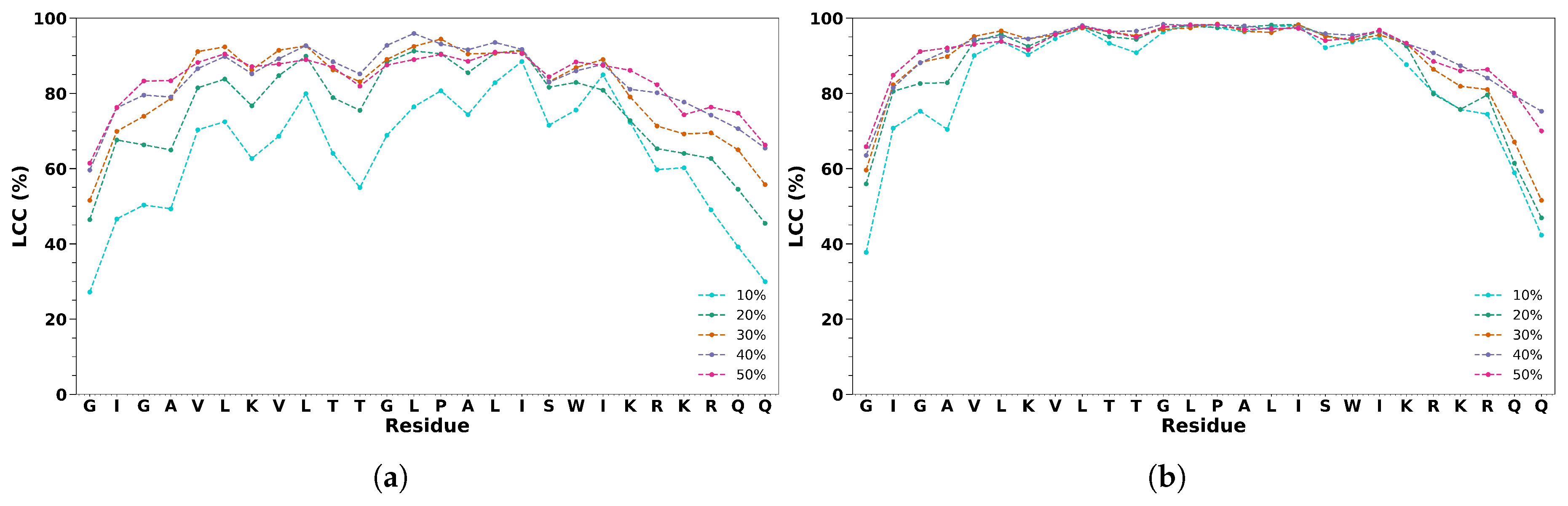
3. Results
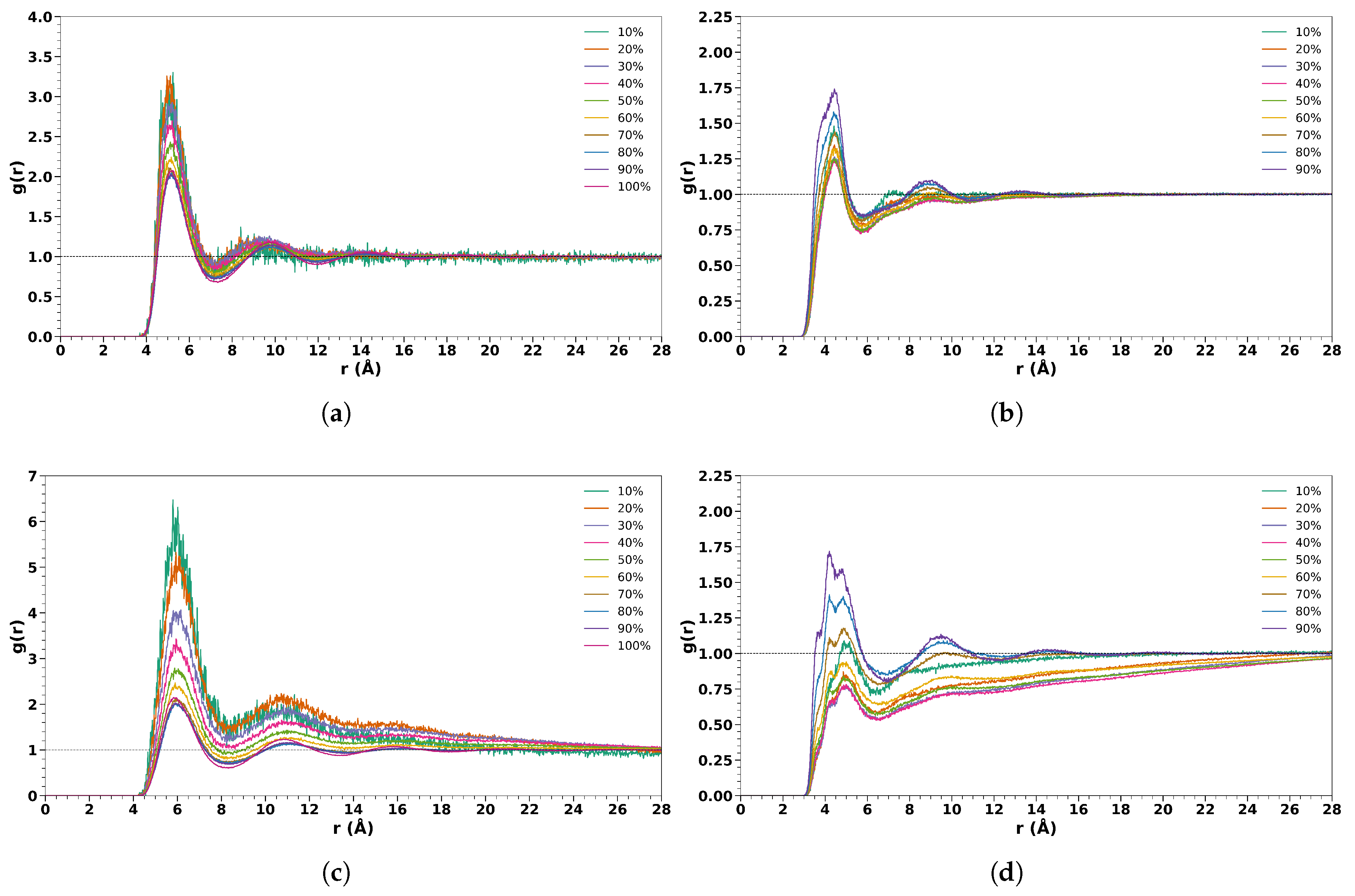
3.1. Cosolvent/Water Solutions
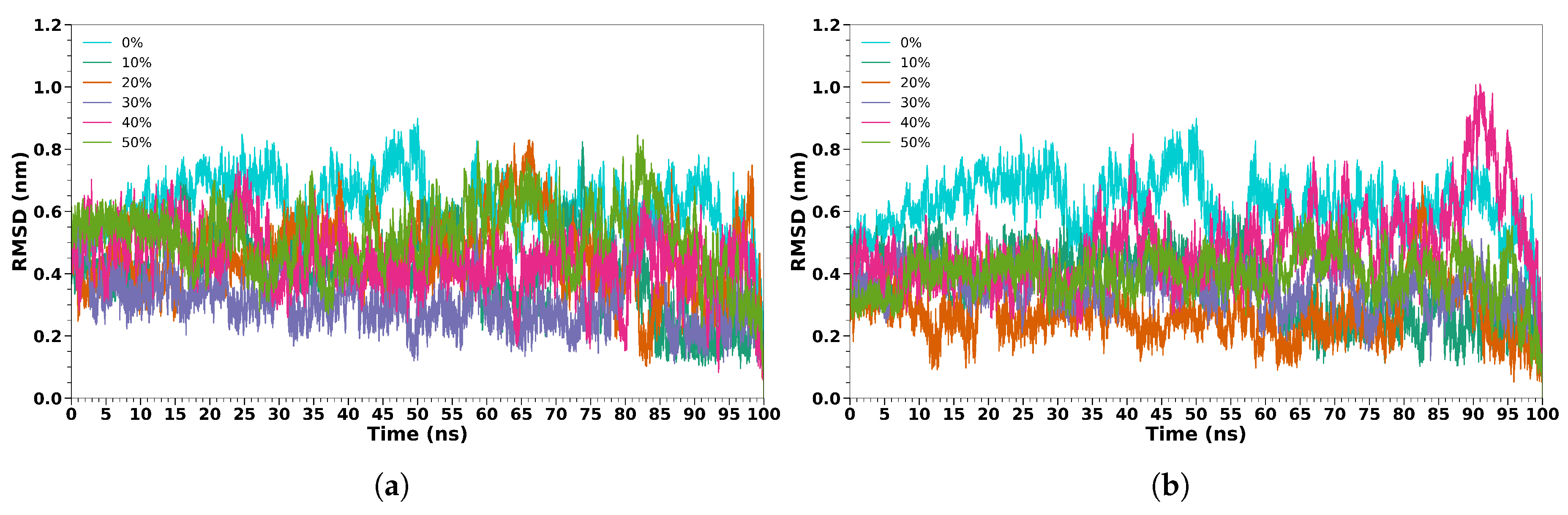
3.2. Melittin in Cosolvent/Water Solutions
4. Conclusions
Supplementary Materials
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Searle, M.S.; Zerella, R.; Williams, D.H.; Packman, L.C. Native-like β-hairpin structure in an isolated fragment from ferredoxin: NMR and CD studies of solvent effects on the N-terminal 20 residues. Protein Eng. Des. Sel. 1996, 9, 559–565. [Google Scholar] [CrossRef]
- Buck, M. Trifluoroethanol and colleagues: cosolvents come of age. Recent studies with peptides and proteins. Q. Rev. Biophys. 1998, 31, 297–355. [Google Scholar] [CrossRef]
- Hong, D.P.; Hoshino, M.; Kuboi, R.; Goto, Y. Clustering of fluorine-substituted alcohols as a factor responsible for their marked effects on proteins and peptides. J. Am. Chem. Soc. 1999, 121, 8427–8433. [Google Scholar] [CrossRef]
- Roccatano, D.; Colombo, G.; Fioroni, M.; Mark, A.E. Mechanism by which 2,2,2-trifluoroethanol/water mixtures stabilize secondary-structure formation in peptides: a molecular dynamics study. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 12179–12184. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, L.S.; Pande, J.; Shekhtman, A. Helical Structure of Recombinant Melittin. J. Phys. Chem. B 2019, 123, 356–368. [Google Scholar]
- Fioroni, M.; Burger, K.; Mark, A.E.; Roccatano, D. Model of 1,1,1,3,3,3-hexafluoro-propan-2-ol for molecular dynamics simulations. J. Phys. Chem. B 2001, 105, 10967–10975. [Google Scholar] [CrossRef]
- Fioroni, M.; Burger, K.; Mark, A.E.; Roccatano, D. A New 2,2,2-trifluoroethanol Model for Molecular Dynamics Simulations. J. Phys. Chem. B 2000, 104, 12347–12354. [Google Scholar] [CrossRef]
- Macchiagodena, M.; Mancini, G.; Pagliai, M.; Del Frate, G.; Barone, V. Fine-tuning of atomic point charges: Classical simulations of pyridine in different environments. Chem. Phys. Lett. 2017, 677, 120–126. [Google Scholar] [CrossRef]
- Macchiagodena, M.; Mancini, G.; Pagliai, M.; Cardini, G.; Barone, V. New atomistic model of pyrrole with improved liquid state properties and structure. Int. J. Quantum Chem. 2018, 118, e25554. [Google Scholar] [CrossRef]
- Macchiagodena, M.; Pagliai, M.; Andreini, C.; Rosato, A.; Procacci, P. Upgraded AMBER Force Field for Zinc-Binding Residues and Ligands for Predicting Structural Properties and Binding Affinities in Zinc-Proteins. ACS Omega 2020, 5, 15301–15310. [Google Scholar] [CrossRef]
- Macchiagodena, M.; Pagliai, M.; Andreini, C.; Rosato, A.; Procacci, P. Upgrading and Validation of the AMBER Force Field for Histidine and Cysteine Zinc(II)-Binding Residues in Sites with Four Protein Ligands. J. Chem. Inf. Model. 2019, 59, 3803–3816. [Google Scholar] [CrossRef]
- Casoria, M.; Macchiagodena, M.; Rovero, P.; Andreini, C.; Papini, A.M.; Cardini, G.; Pagliai, M. Upgrading of the general AMBER force field 2 for fluorinated alcohol biosolvents: A validation for water solutions and melittin solvation. J. Pept. Sci. 2024, 30, e3543. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and testing of a general amber force field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef] [PubMed]
- GAFF and GAFF2 are public domain force fields and are part of the AmberTools23 distribution, available for download at https://ambermd.org/ internet address (accessed 23). According to the AMBER development team, the improved version of GAFF, GAFF2, is an ongoing project aimed at “reproducing both the high-quality interaction energies and key liquid properties such as density, the heat of vaporization and hydration free energy”. GAFF2 is expected “to be an even more successful general purpose force field and that GAFF2-based scoring functions will significantly improve the success rate of virtual screenings". 20 May.
- Kemple, M.D.; Buckley, P.; Yuan, P.; Prendergast, F.G. Main chain and side chain dynamics of peptides in liquid solution from 13C NMR: melittin as a model peptide. Biochemistry 1997, 36, 1678–1688. [Google Scholar] [CrossRef]
- Hirota, N.; Mizuno, K.; Goto, Y. Group additive contributions to the alcohol-induced α-helix formation of melittin: implication for the mechanism of the alcohol effects on proteins. J. Mol. Biol. 1998, 275, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 2015, 1-2, 19–25. [Google Scholar] [CrossRef]
- Lindorff-Larsen, K.; Piana, S.; Palmo, K.; Maragakis, P.; Klepeis, J.L.; Dror, R.O.; Shaw, D.E. Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins 2010, 78, 1950–1958. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Pagliai, M.; Macchiagodena, M.; Procacci, P.; Cardini, G. Evidence of a Low–High Density Turning Point in Liquid Water at Ordinary Temperature under Pressure: A Molecular Dynamics Study. J. Phys. Chem. Lett. 2019, 10, 6414–6418. [Google Scholar] [CrossRef]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef] [PubMed]
- Hess, B.; Bekker, H.; Berendsen, H.J.; Fraaije, J.G. LINCS: a linear constraint solver for molecular simulations. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle mesh Ewald: An N·log (N) method for Ewald sums in large systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef]
- Allen, M.P.; Tildesley, D.J. Computer Simulation of Liquids; 1987.
- Kabsch, W.; Sander, C. Dictionary of Protein Secondary Structure: Pattern Recognition of Hydrogen-bonded and Geometrical Features. Biopolymers 1983, 22, 2577–2637. [Google Scholar] [CrossRef] [PubMed]
- Roccatano, D.; Fioroni, M.; Zacharias, M.; Colombo, G. Effect of hexafluoroisopropanol alcohol on the structure of melittin: A molecular dynamics simulation study. Protein Science 2005, 14, 2582–2589. [Google Scholar] [CrossRef]
- Madelung, O. Static Dielectric Constants of Pure Liquids and Binary Liquid Mixtures. Numerical Data and Functional Relationships in Science and Technology 1991, 6, 459. [Google Scholar] [CrossRef]
- Rochester, C.; Symonds, J. Densities of solutions of four fluoroalcohols in water. J. Fluor. Chem. 1974, 4, 141–148. [Google Scholar] [CrossRef]
- Cabani, S.; Gianni, P.; Mollica, V.; Lepori, L. Group contributions to the thermodynamic properties of non-ionic organic solutes in dilute aqueous solution. J. Solut. Chem. 1981, 10, 563–595. [Google Scholar] [CrossRef]
- Marenich, A.V.; Kelly, C.P.; Thompson, J.D.; Hawkins, G.D.; Chambers, C.C.; Giesen, D.J.; Winget, P.; Cramer, C.J.; Truhlar, D.G. Minnesota solvation database (MNSOL) version 2012 2020. [CrossRef]
- Caleman, C.; van Maaren, P.J.; Hong, M.; Hub, J.S.; Costa, L.T.; van der Spoel, D. Force Field Benchmark of Organic Liquids: Density, Enthalpy of Vaporization, Heat Capacities, Surface Tension, Isothermal Compressibility, Volumetric Expansion Coefficient, and Dielectric Constant. J. Chem. Theory Comput. 2012, 8, 61–74. [Google Scholar] [CrossRef]
- Macchiagodena, M.; Mancini, G.; Pagliai, M.; Barone, V. Accurate prediction of bulk properties in hydrogen bonded liquids: amides as case studies. Phys. Chem. Chem. Phys. 2016, 18, 25342–25354. [Google Scholar] [CrossRef]
- R. Harris, K.; J. Newitt, P.; J. Derlacki, Z. Alcohol tracer diffusion, density, NMR and FTIR studies of aqueous ethanol and 2,2,2-trifluoroethanol solutions at 25°C. J. Chem. Soc., Faraday Trans. 1998, 94, 1963–1970. [Google Scholar] [CrossRef]
 Random Coil
Random Coil  -Helix
-Helix  Bent
Bent  Turn
Turn  5-Helix
5-Helix  3-Helix.
3-Helix.
 Random Coil
Random Coil  -Helix
-Helix  Bent
Bent  Turn
Turn  5-Helix
5-Helix  3-Helix.
3-Helix.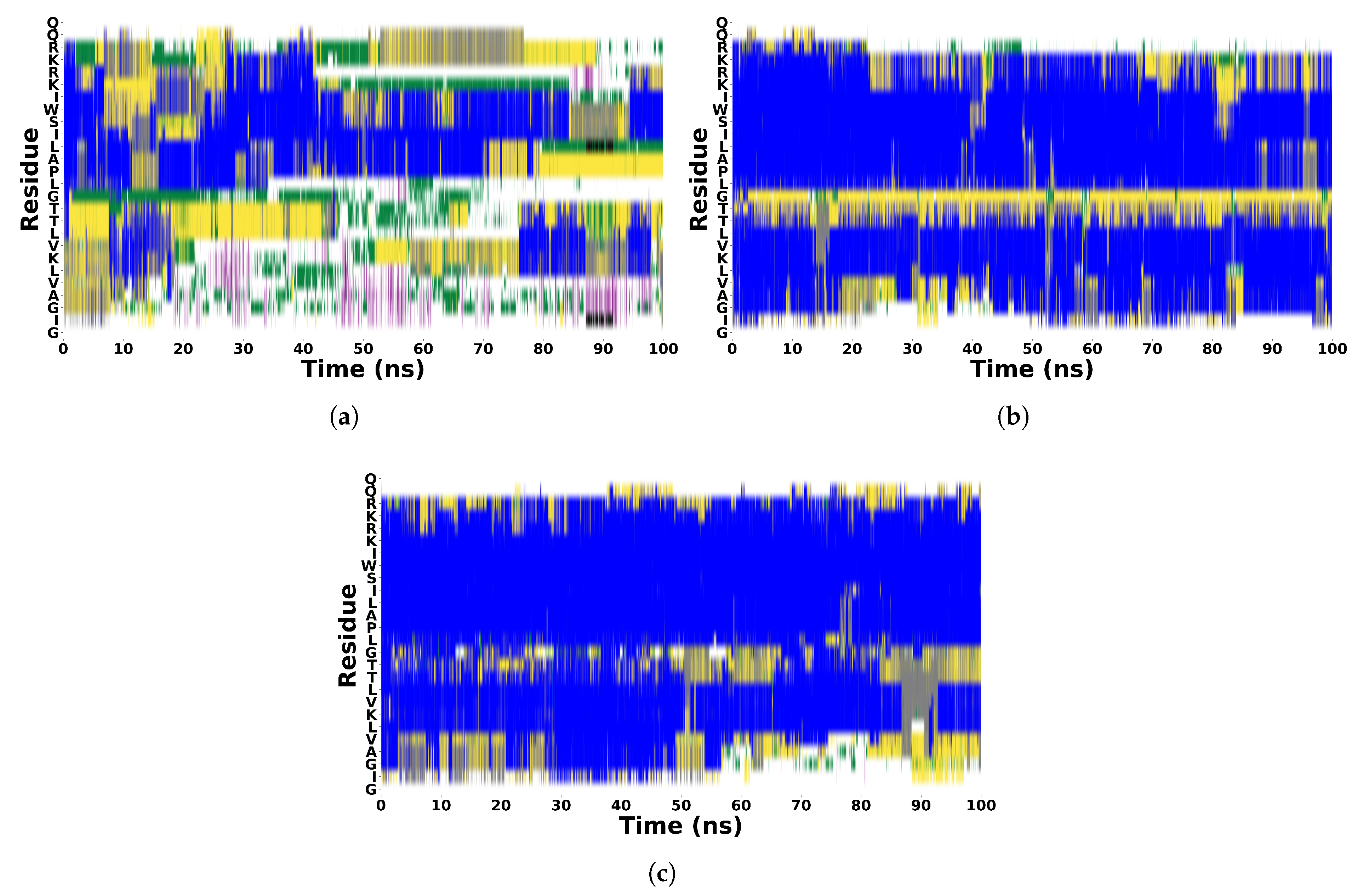
| % (v/v) | TFE mixture | HFIP mixture | ||
| N. water | N. cosolvent | N. water | N. cosolvent | |
| 10 | 5838 | 162 | 5886 | 114 |
| 20 | 5646 | 354 | 5754 | 246 |
| 30 | 5418 | 582 | 5592 | 408 |
| 40 | 5142 | 858 | 5382 | 618 |
| 50 | 4800 | 1200 | 5124 | 876 |
| 60 | 4362 | 1638 | 4770 | 1230 |
| 70 | 3786 | 2214 | 4284 | 1716 |
| 80 | 3000 | 3000 | 3558 | 2442 |
| 90 | 1842 | 4158 | 2358 | 3642 |
| 100 | 0 | 6000 | 0 | 6000 |
| % (v/v) | TFE mixture | HFIP mixture | ||
|---|---|---|---|---|
| Dielectric constant | Density (kg/m) | Dielectric constant | Density (kg/m) | |
| 10 | 87.78 ± 0.85 | 1014.12 ± 0.05 | 81.43 ± 1.90 | 1036.68 ± 0.04 |
| (75.5 ± 0.3) | (71.3±0.2) | |||
| 20 | 77.01 ± 0.57 | 1050.81 ± 0.04 | 78.16 ± 0.46 | 1093.70 ± 0.03 |
| (70.5 ± 0.2) | (65.6±0.2) | |||
| 30 | 65.11 ± 1.74 | 1086.17 ± 0.03 | 59.69 ± 0.49 | 1151.01 ± 0.04 |
| (64.8 ± 0.3) | (60.2±0.2) | |||
| 40 | 58.16 ± 0.68 | 1121.20 ± 0.07 | 46.61 ± 0.54 | 1210.52 ± 0.02 |
| (57.5 ± 0.1) | (53.4±0.2) | |||
| 50 | 50.96 ± 1.03 | 1156.00 ± 0.04 | 38.01 ± 0.62 | 1267.36 ± 0.03 |
| (53.0 ± 0.4) | (49.0±0.3) | |||
| 60 | 39.62 ± 0.34 | 1191.00 ± 0.04 | 32.36 ± 0.35 | 1326.01 ± 0.05 |
| (46.7 ± 0.3) | (43.2±0.5) | |||
| 70 | 35.93 ± 0.49 | 1226.51 ± 0.04 | 21.99 ± 0.24 | 1383.52 ± 0.07 |
| (41.5 ± 0.2) | (37.4±0.2) | |||
| 80 | 25.41 ± 0.41 | 1262.94 ± 0.06 | 16.30 ± 0.09 | 1442.22 ± 0.05 |
| (35.3 ± 0.3) | (30.3±0.3) | |||
| 90 | 20.76 ± 0.19 | 1302.29 ± 0.05 | 9.67 ± 0.21 | 1503.59 ± 0.04 |
| (31.5 ± 0.2) | (24.3±0.4) | |||
| 100 | 15.05 ± 0.22 | 1343.33 ± 0.07 | 4.25 ± 0.03 | 1560.10 ± 0.04 |
| (27.1 ± 0.1) | (17.8±0.1) | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




