Submitted:
09 October 2025
Posted:
10 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
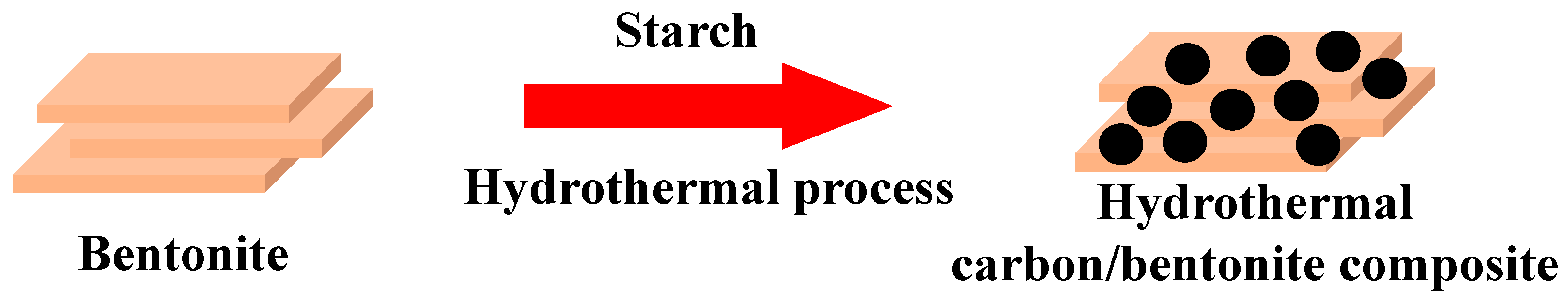
2.2. Preparation and Characterization of HCBCs
2.3. Drilling Fluid Preparation and Properties Measurement
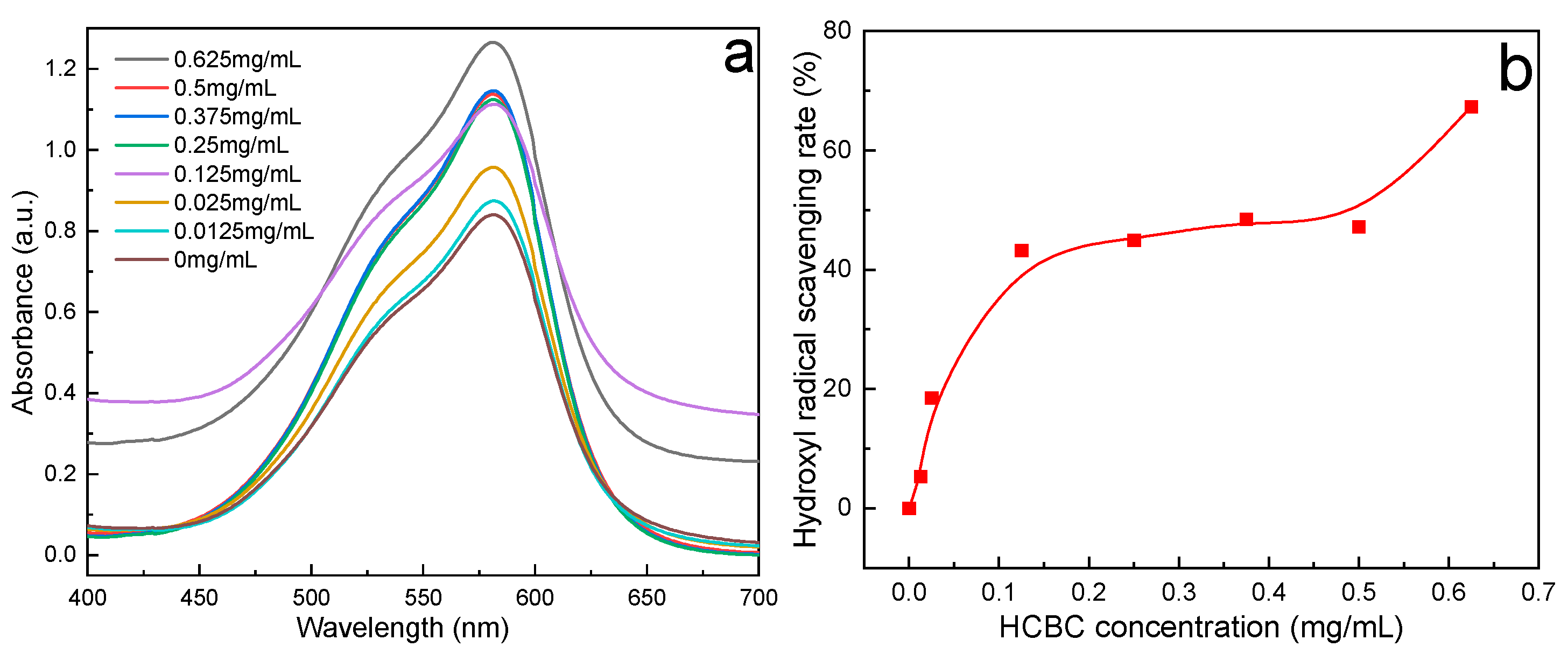
2.4. Hydroxyl Free Radicals Scavenging Experiment
3. Results
3.1. Characterization of HCBCs
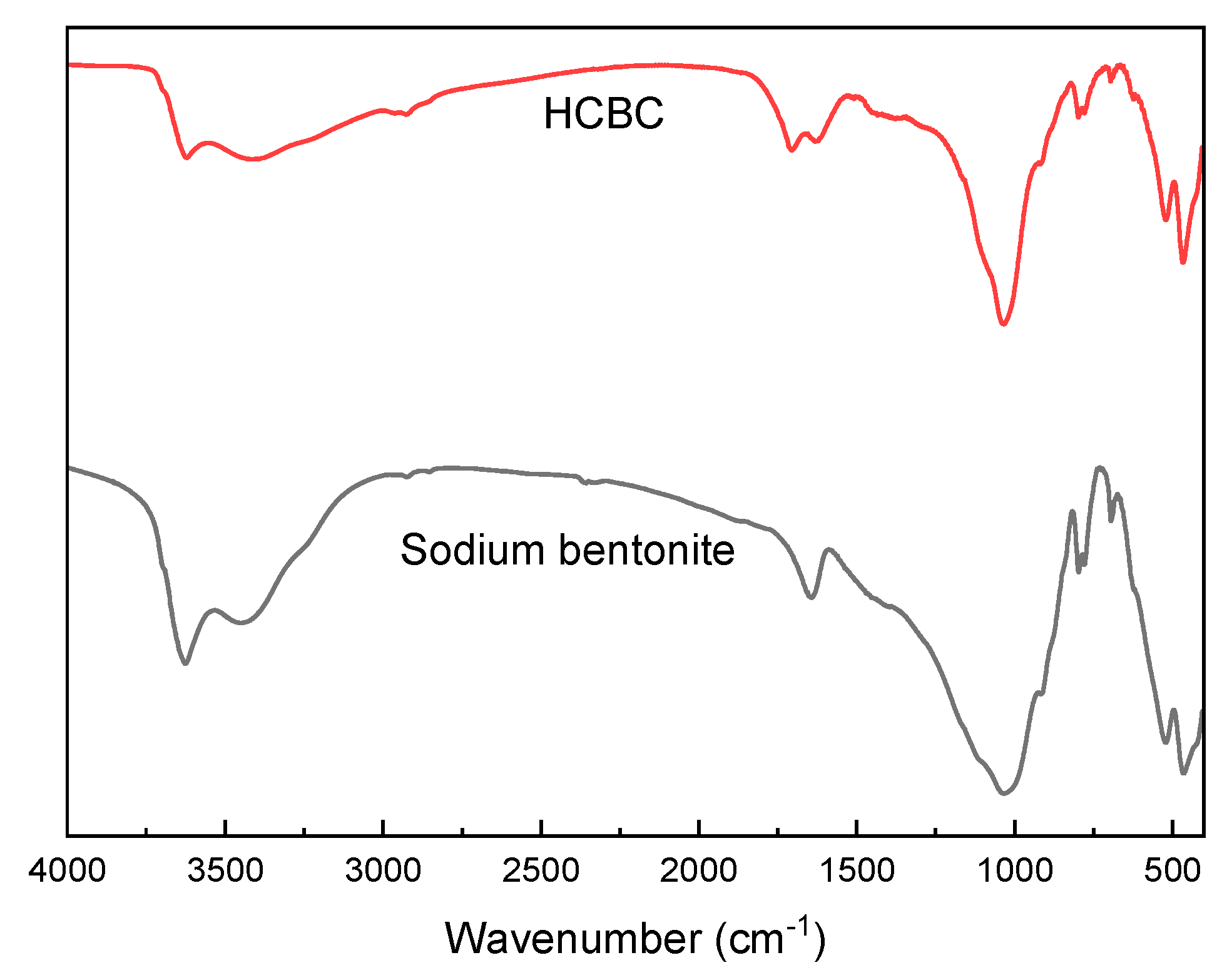
3.1.1. FT-IR
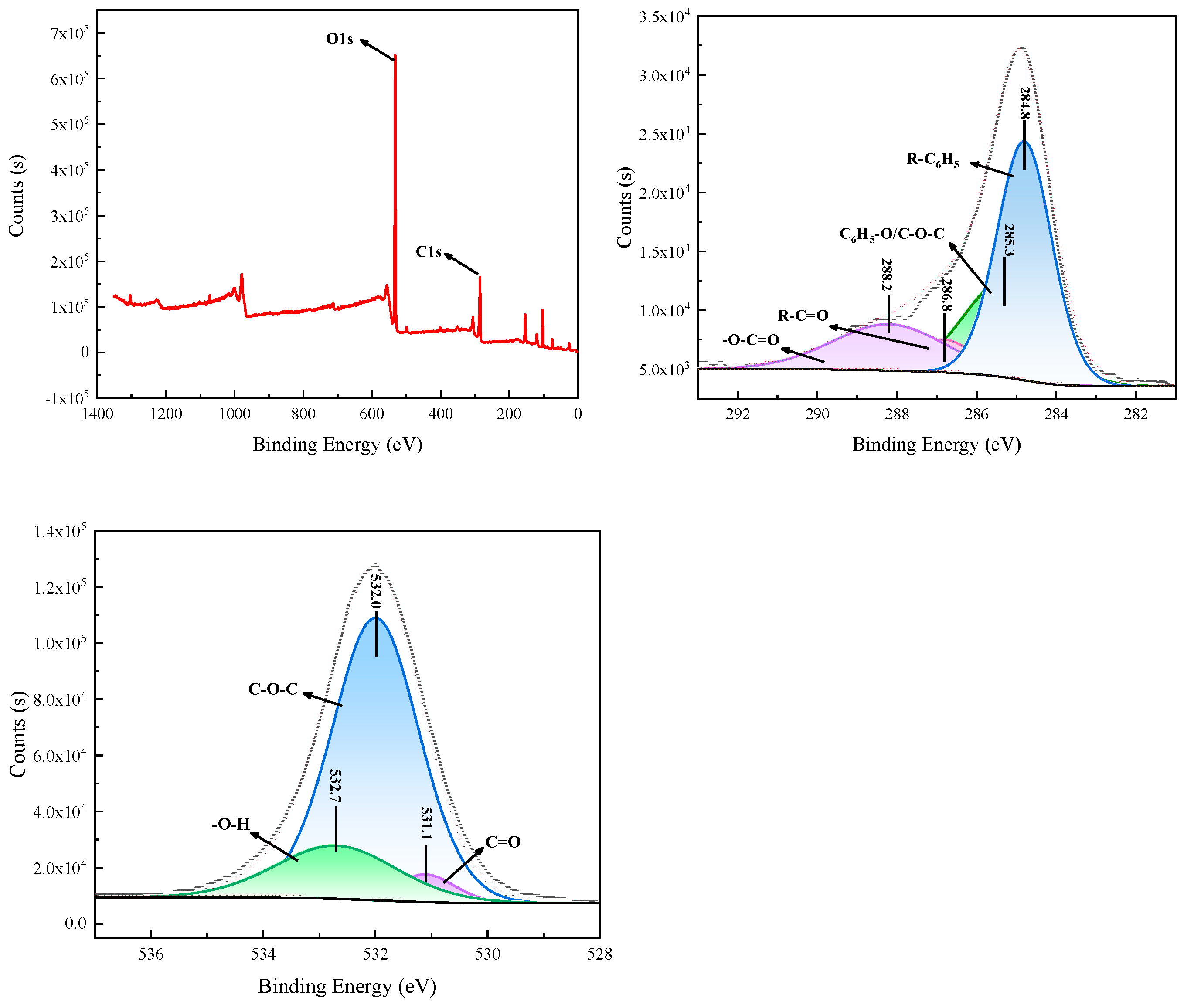
3.1.2. XPS
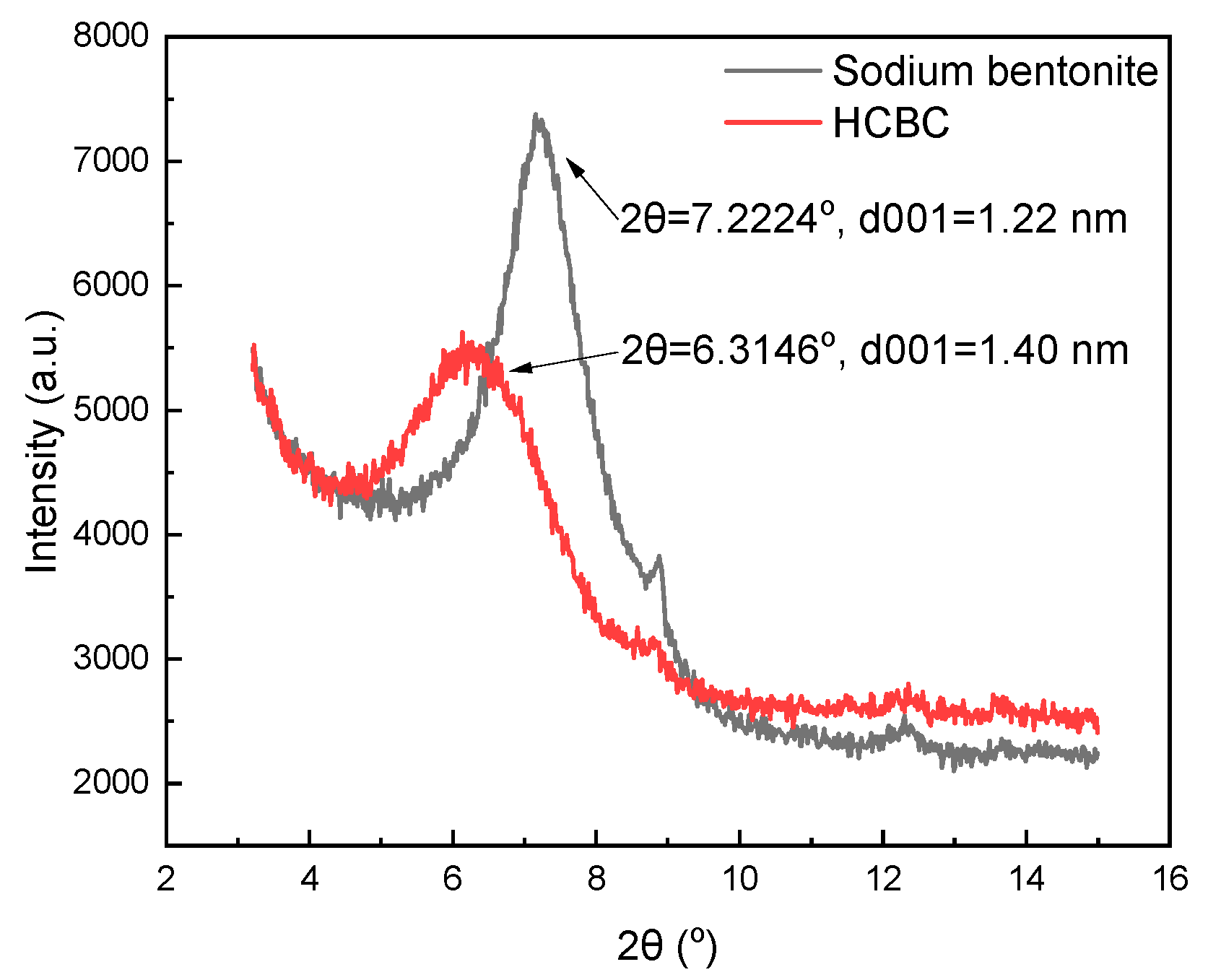
3.1.3. XRD
3.1.4. SEM and TEM
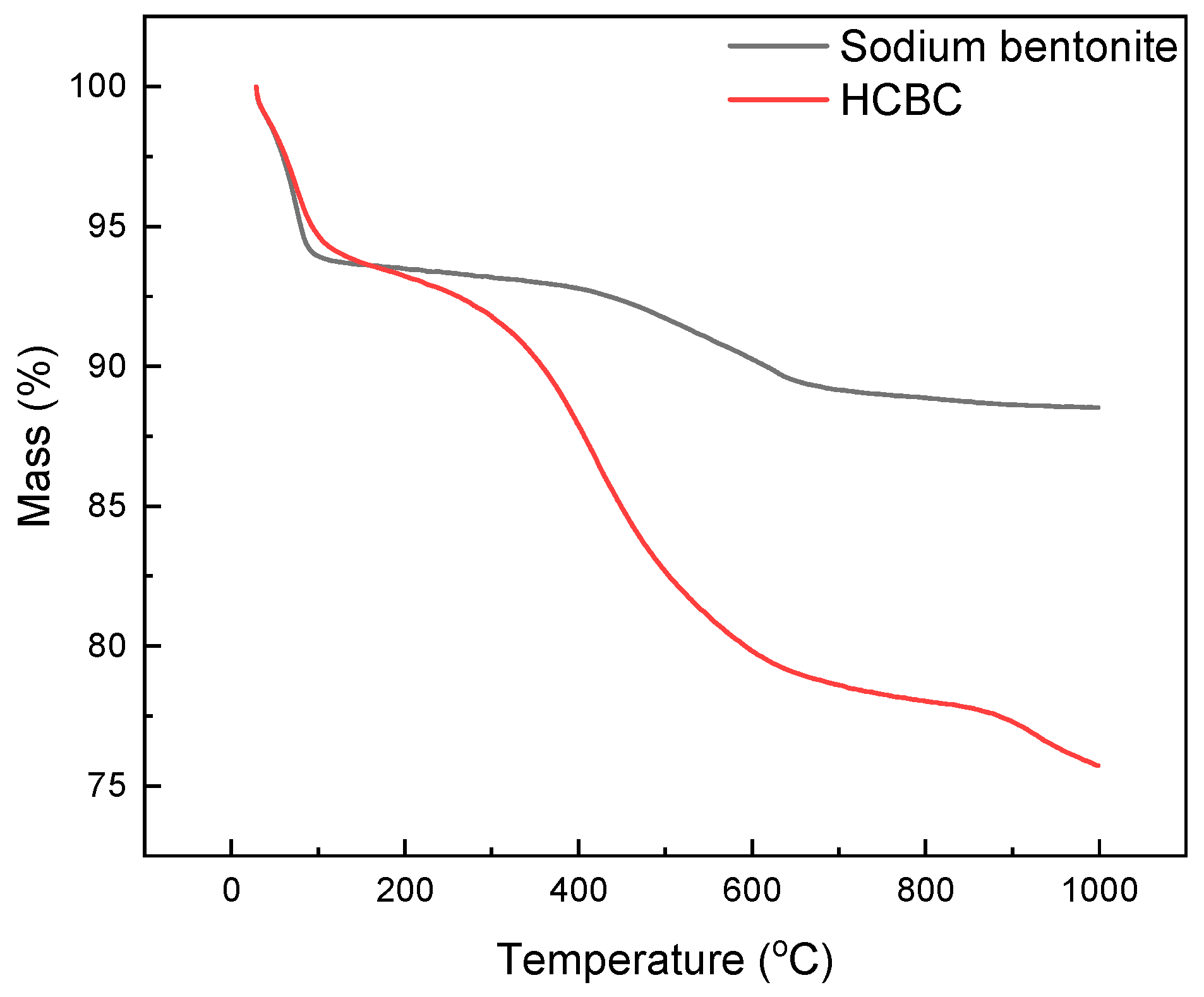
3.1.5. TGA
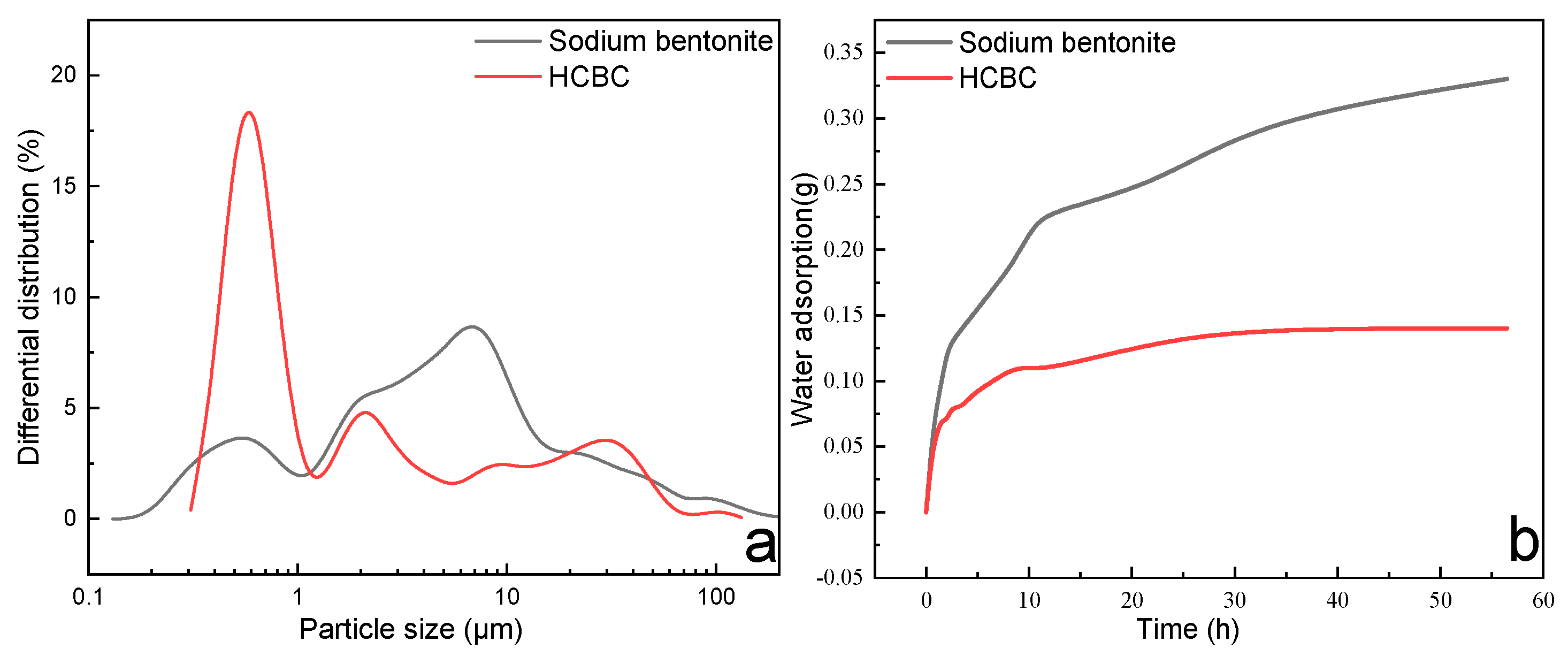
3.1.6. Particle Size Distribution and Water Adsorption
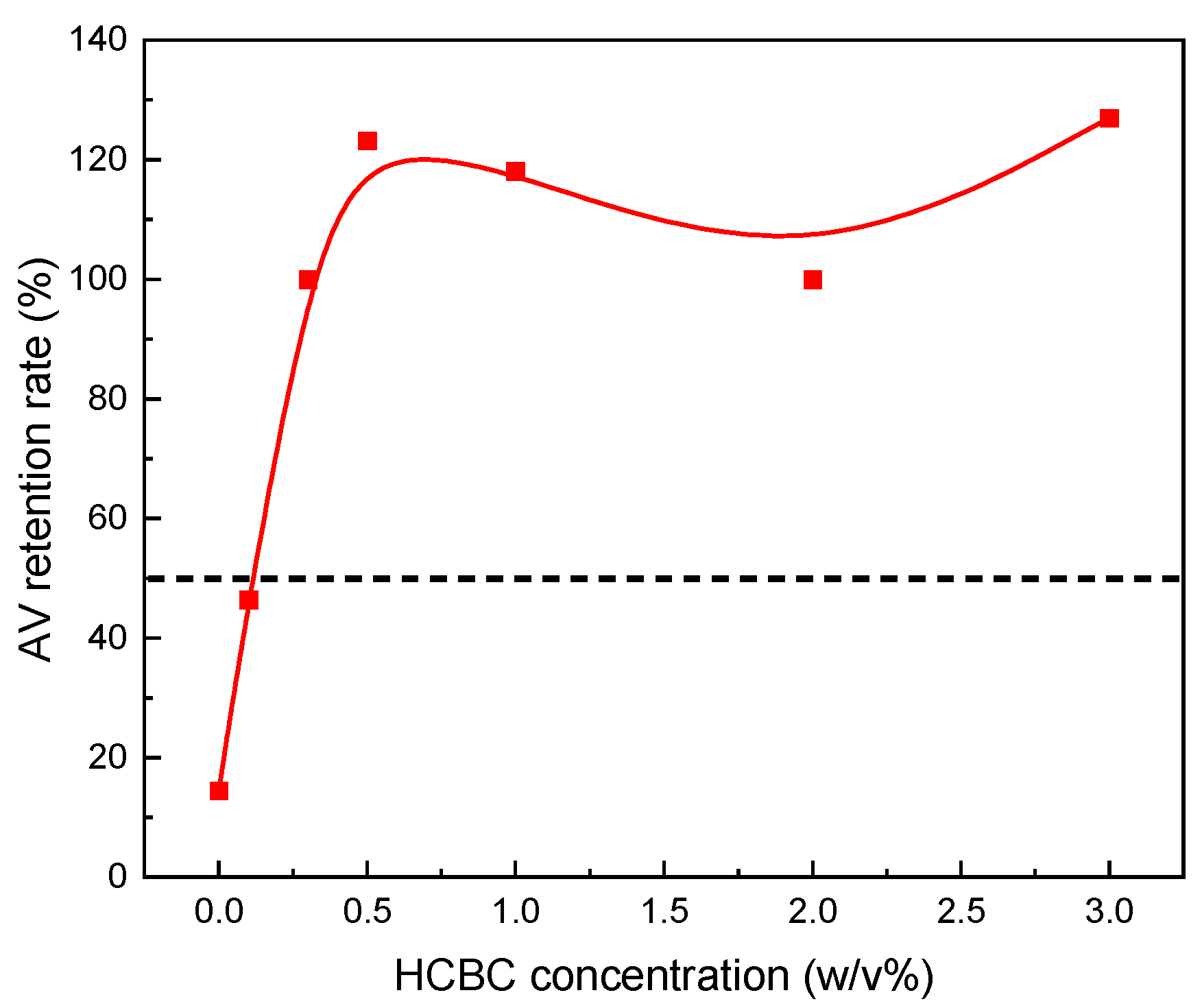
3.2. Properties Evaluation
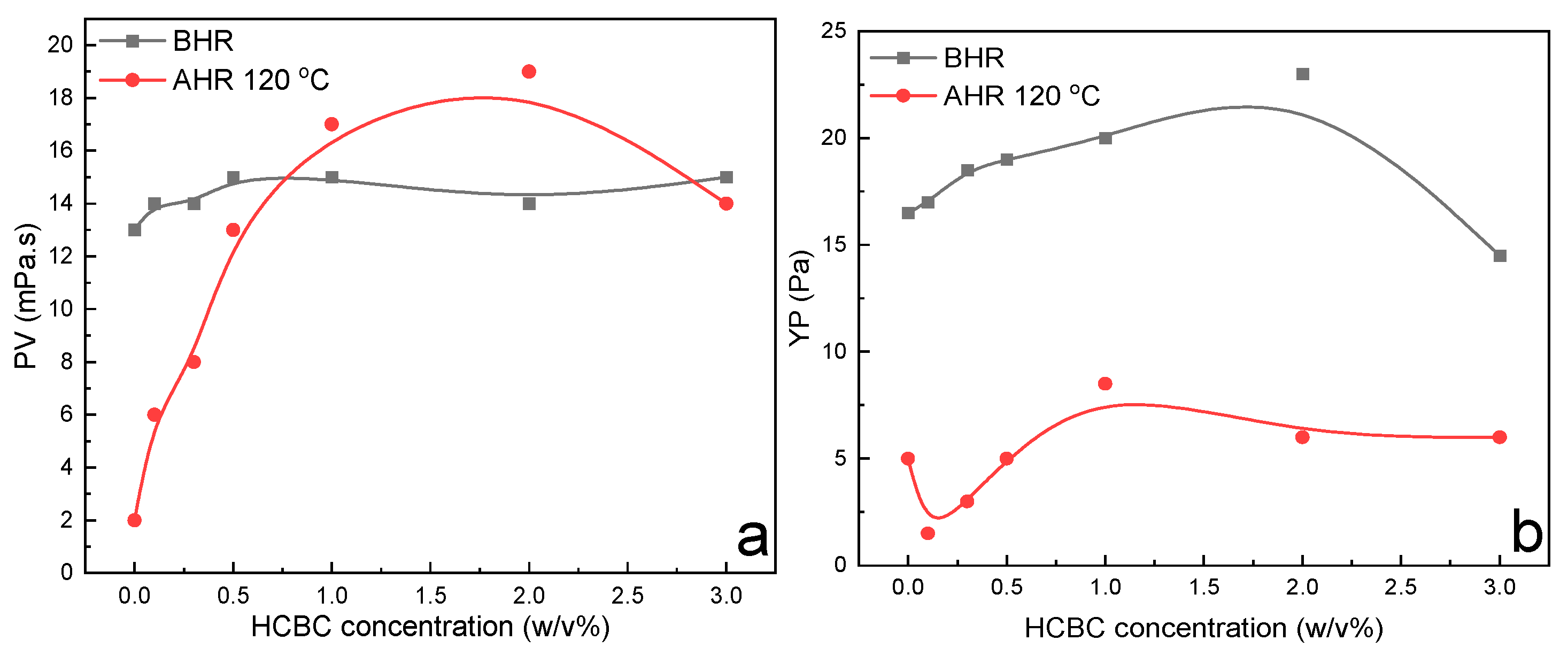
3.2.1. Xanthan Slurries
3.2.2. Modified Starch Slurries
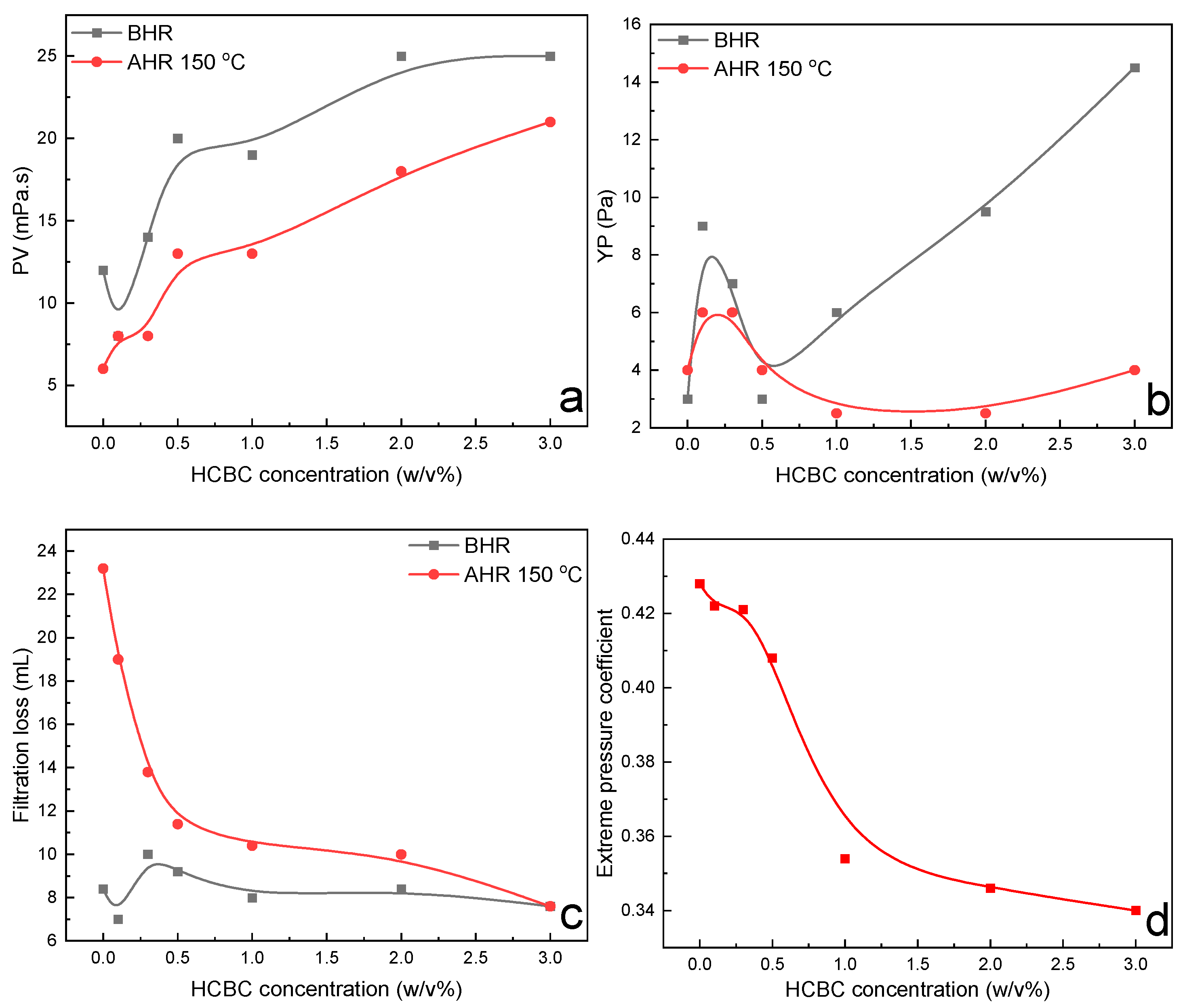
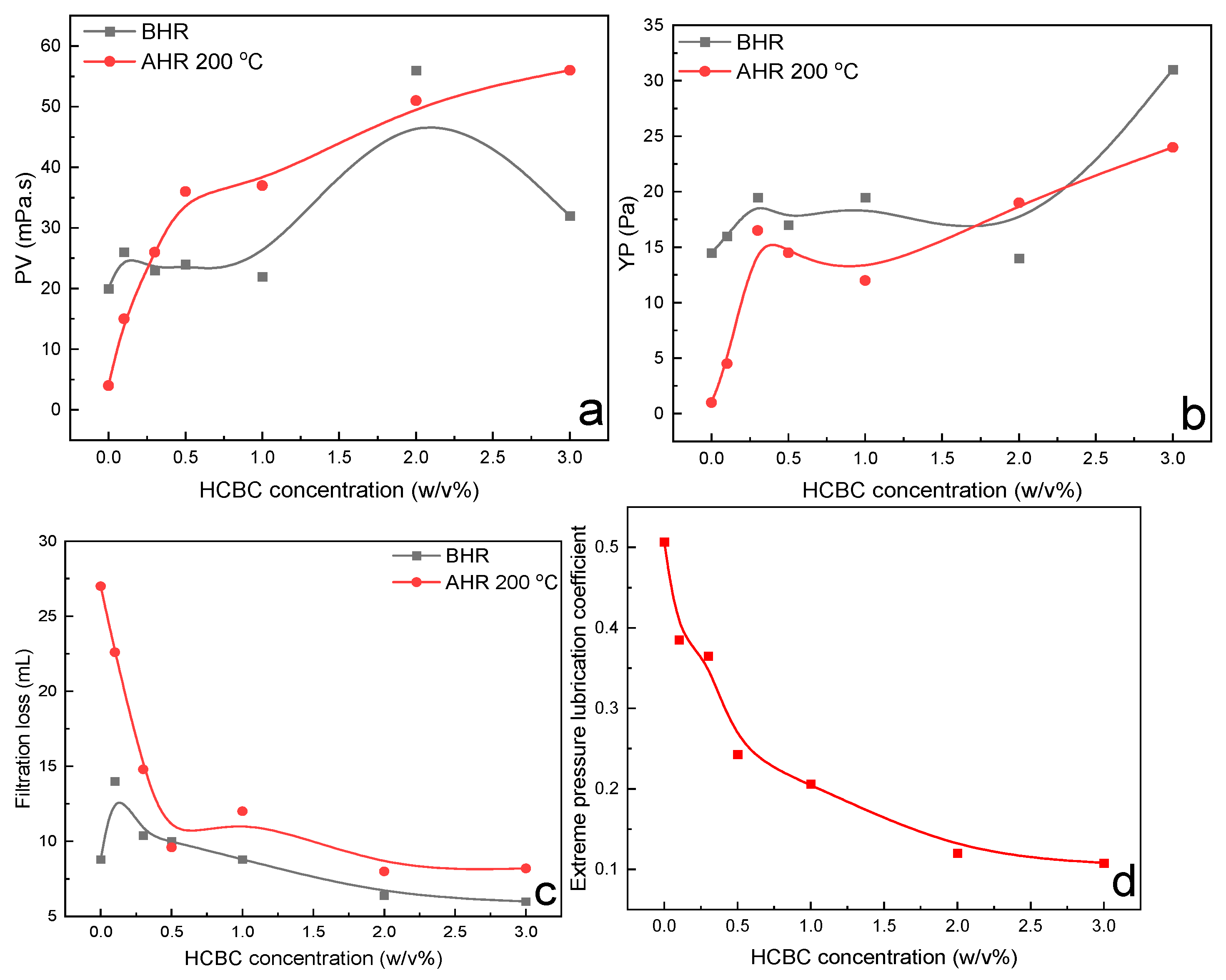
3.2.3. HT Polymer Slurries
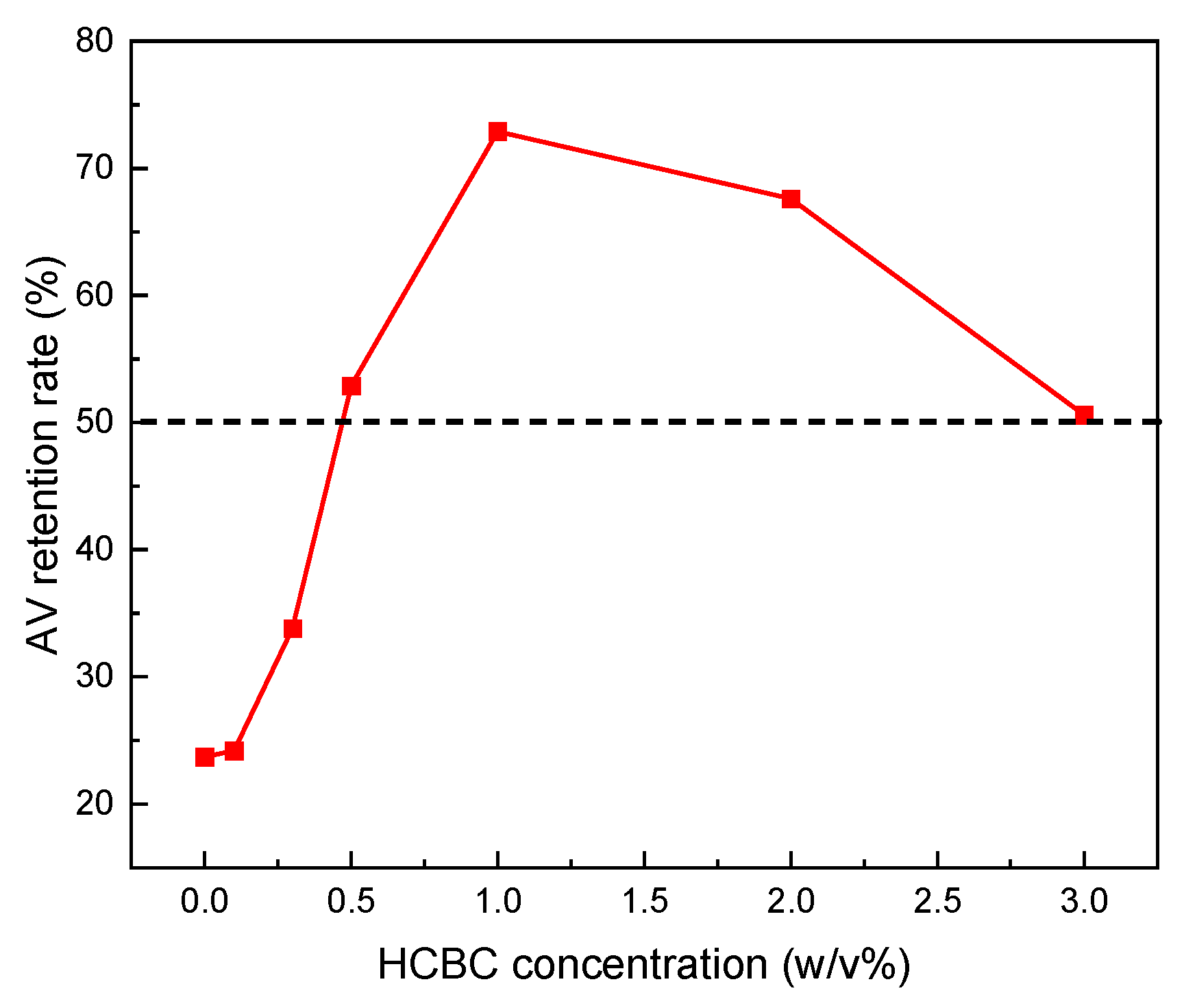
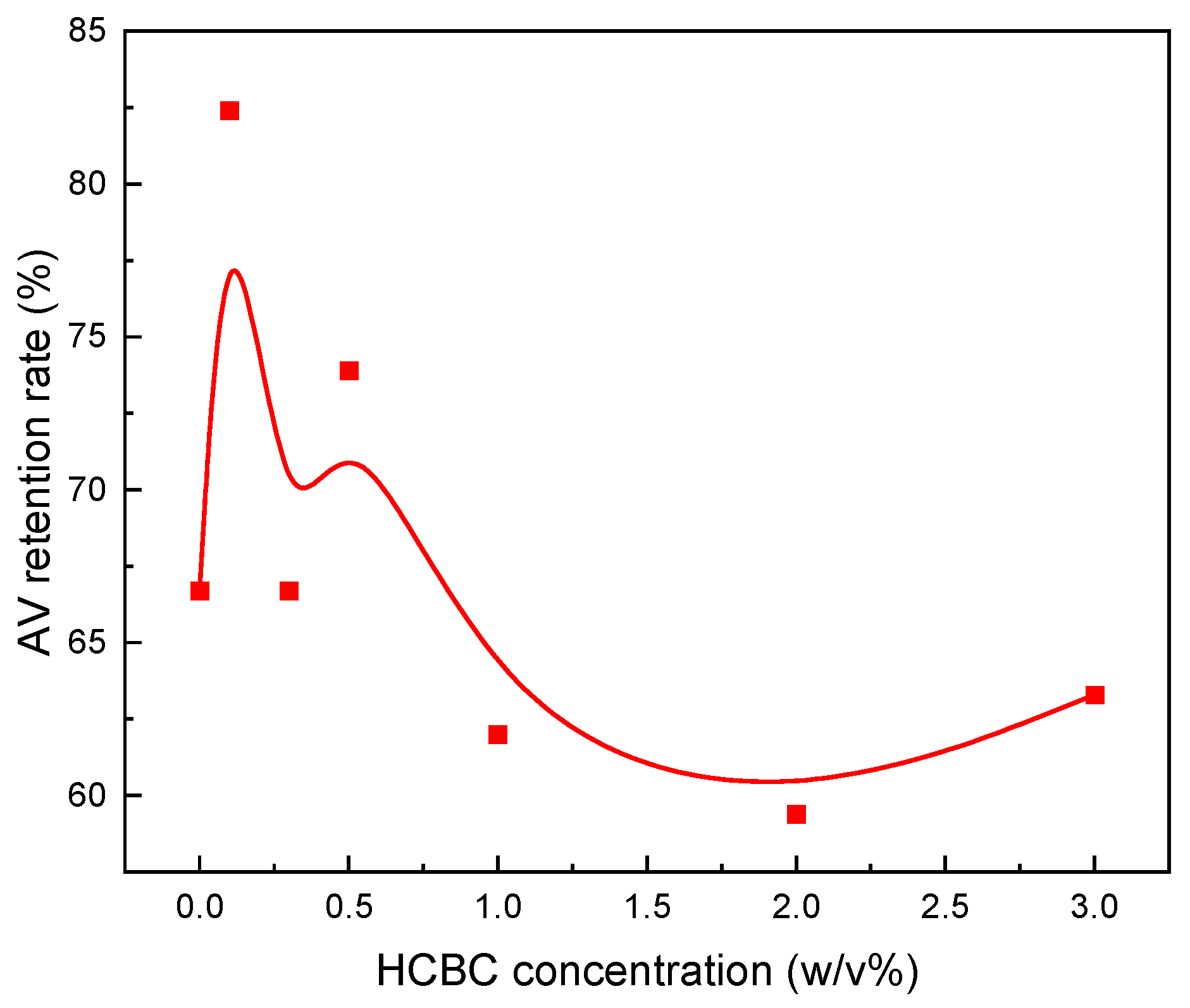
3.2.4. Free Radical Scavenging
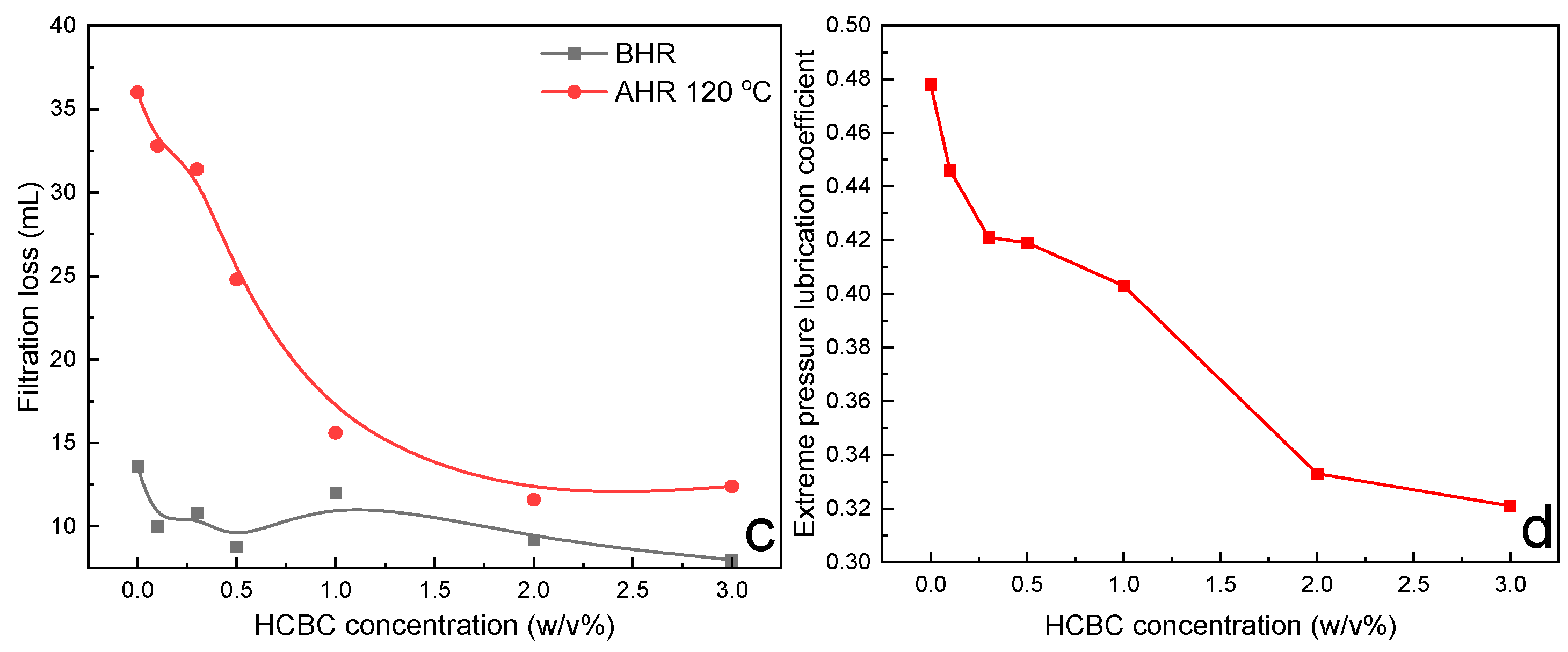
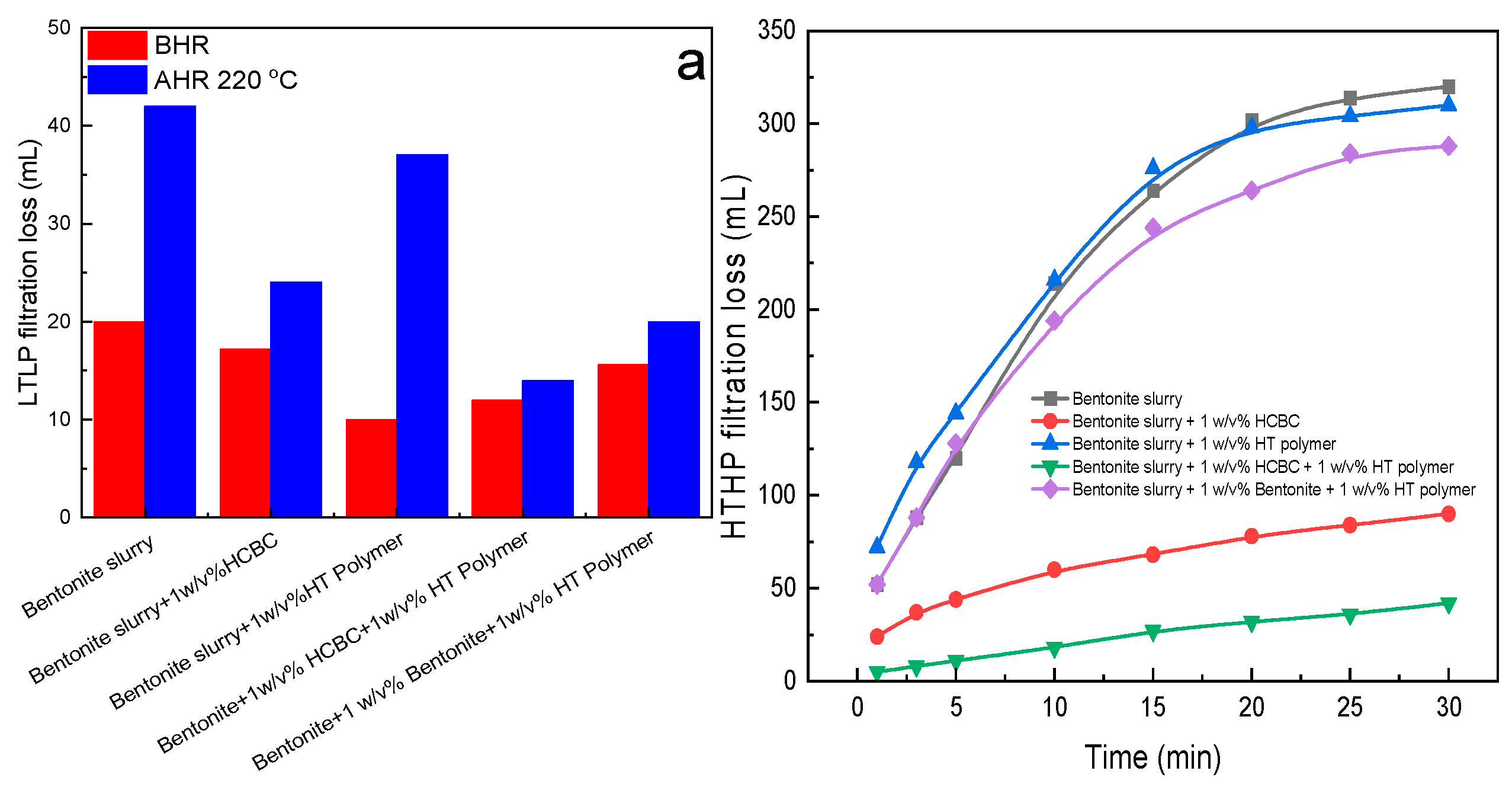
3.2.5. Filtration Loss
3.3. High Temperature Stabilizing and Filtration Control Mechanism
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AV | Apparent viscosity |
| FTIR | Fourier Transform infrared spectroscopy |
| HCBCs | Hydrothermal carbon/bentonite composites |
| HT | High-temperature |
| HTHP | High-temperature and High-pressure |
| LTLP | Low-temperature and Low-pressure |
| MV | Methyl violet |
| PV | Plastic viscosity |
| SEM | Scanning Electron Microscope |
| TEM | Transmission electron microscope |
| TGA | Thermogravimetry Analysis |
| XC | Xanthan gum |
| XPS | X-ray photoelectron spectroscopy |
| YP | Yield point |
References
- Galindo, K.A.; Zha, W.; Zhou, H.; Deville, J.P. Clay-free high performance water-based drilling fluid for extreme high temperature wells. SPE-173017-MS, SPE/IADC Drilling Conference and Exhibition, March 17-19, 2015.
- Tehrani, M.A.; Popplestone, A.; Guarneri, A.; Carminati, S. Water-based drilling fluid for HT/HP applications. SPE-105485-MS, International Symposium on Oilfield Chemistry, February 28-March 2, 2007.
- Rabaioli, M.R.; Miano, F.; Lockhart, T.P.; Burrafato, G. Physical/chemical studies on the surface interactions of bentonite with polymeric dispersing agents. SPE-25179-MS, SPE International Symposium on Oilfield Chemistry, March 2-5, 1993.
- Ahmad, H.M.; Kamal, M.S.; Al-Harthi, M.A. Effect of thermal aging and electrolyte on bentonite dispersions: Rheology and morphological properties. Journal of Molecular Liquids 2018, 269, 278–286. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, Z.; Han, F. Effects of pH on the gel properties of montmorillonite, palygorskite and montmorillonite-palygorskite composite clay. Applied Clay Science 2020, 190, 105543. [Google Scholar] [CrossRef]
- Dormán, J. Chemistry and field practice of high-temperature drilling fluids in Hungary. SPE-21940-MS, SPE/IADC Drilling Conference, March 11-14, 1991.
- Elward-Berry, J.; Darby, J.B. Rheologically sable, nontoxic, high-temperature, water-based drilling fluid. SPE Drilling & Completion 1997, 12, 158–162. [Google Scholar] [CrossRef]
- Zilch, H.E.; Otto, M.J.; Pye, D.S. The evolution of geothermal drilling fluid in the imperial valley. SPE 21786, Western Regional Meeting, California, March 20-22, 1991.
- Lozano, J.; Miska, N.; Takach, N.; Yu, M.; Saasen, A. The effect of elongational flow through the drill bit on the rheology of polymeric drilling fluids. SPE-99107-MS, IADC/SPE Drilling Conference, February 21-23, 2006.
- Zhou, H.; Deville, J.P.; Davis, C.L. Novel thermally stable high-density brine-based drill-in fluids for HP/HT applications. SPE-172659-MS, SPE Middle East Oil & Gas Show and Conference, March 8-11, 2015.
- Yang, J.; Sun, J.; Wang, R.; Liu, F.; Wang, J.; Qu, Y.; Wang, P.; Huang, H.; Liu, L.; Zhao, Z. Laponite-polymer composite as a rheology modifier and filtration loss reducer for water-based drilling fluids at high temperature. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022, 655, 130261. [Google Scholar] [CrossRef]
- Ahmed, A.; Pervaiz, E.; Abdullah, U.; Noor, T. Optimization of water based drilling fluid properties with the SiO2/g-C3N4 hybrid. ACS Omega 2024, 9, 15052–15064. [Google Scholar] [CrossRef]
- Ao, T.; Yang, L.; Xie, C.; Jiang, G.; Wang, G.; Liu, Z.; He, X. Zwitterionic silica-based hybrid nanoparticles for filtration control in oil drilling conditions. ACS Applied Nano Materials 2021, 4, 11052–11062. [Google Scholar] [CrossRef]
- Ikram, R.; Jan, B.M.; Vejpravova, J. Towards recent tendencies in drilling fluids: Application of carbon-based nanomaterials. Journal of Materials Research and Technology 2021, 15, 3733–3758. [Google Scholar] [CrossRef]
- Ospanov, Y.K.; Kudaikulova, G.A. A comprehensive review of carbon nanomaterials in the drilling industry. Journal of Polymer Science 2024, 1–20. [Google Scholar] [CrossRef]
- Rana, A.; Khan, I.; Saleh, T.A. Advances in carbon nanostructures and nanocellulose as additives for efficient drilling fluids: Trends and future perspective—A review. Energy & Fuels 2021, 35, 7319–7339. [Google Scholar] [CrossRef]
- Li, T.; Shen, J.; Huang, S.; Li, N.; Ye, M. Hydrothermal carbonization synthesis of a novel montmorillonite supported carbon nanosphere adsorbent for removal of Cr (VI) from waste water. Applied Clay Science 2014, 93–94, 48–55. [Google Scholar] [CrossRef]
- Liu, C.; Cai, W.; Liu, L. Hydrothermal carbonization synthesis of Al-pillared montmorillonite@ carbon composites as high performing toluene adsorbents. Applied Clay Science 2018, 162, 113–120. [Google Scholar] [CrossRef]
- Tian, S.; Liu, Y.; Liu, S.; Zeng, G.; Jiang, L.; Tan, X.; Huang, X.; Yin, Z.; Liu, N.; Li, J. Hydrothermal synthesis of montmorillonite/hydrochar nanocomposites and application for 17β-estradiol and 17α-ethynylestradiol removal. RSC advances 2018, 8, 4273–4283. [Google Scholar] [CrossRef]
- Lankone, R.S.; Deline, A.R.; Barclay, M.; Fairbrother, D.H. UV-Vis quantification of hydroxyl radical concentration and dose using principal component analysis. Talanta 2020, 218, 121148. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Luan, Q.; Yang, D.; Yao, X.; Zhou, K. Direct evidence for hydroxyl radical scavenging activity of Cerium oxide nanoparticles. The Journal of Physical Chemistry 2011, 115, 4433–4438. [Google Scholar] [CrossRef]
- Ai, L.; Li, L. Efficient removal of organic dyes from aqueous solution with ecofriendly biomass-derived carbon@montmroillonite nanocomposites by one-step hydrothermal process. Chemical Engineering Journal 2013, 223, 688–695. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides. Chemistry—A European Journal 2009, 15, 4195–4203. [Google Scholar] [CrossRef]
- Li, M.; Li, W.; Liu, S.X. Hydrothermal synthesis, characterization, and KOH activation of carbon spheres from glucose. Carbohydrate Research 2011, 346, 999–1004. [Google Scholar] [CrossRef]
- Titirici, M.M.; Antonietti, M.; Baccile, N. Hydrothermal carbon from biomass: A comparison of the local structure from poly- to monosaccharides and pentoses/hexoses. Green Chemistry 2008, 10, 1204–1212. [Google Scholar] [CrossRef]
- Yin, J.L.; Pei, M.S.; He, Y.J.; Du, Y.; Guo, W.; Wang, L. Hydrothermal and activated synthesis of adsorbent montmorillonite supported porous carbon nanospheres for removal of methylene blue from waste water. RSC Advances 2015, 5, 89839–89847. [Google Scholar] [CrossRef]
- Howard, S.; Kaminski, L.; Downs, J. Xanthan stability in formate brines-formulating non-damaging fluids for high temperature applications. SPE-174228-MS, SPE European Formation Damage Conference and Exhibition, Budapest, 3-5 June, 2015.
- Wang, Y.; Kong, W.; Wang, L.; Zhang, J.Z.; Li, Y.; Liu, X.; Li, Y. Optimizing oxygen functional groups in graphene quantum dots for improved antioxidant mechanism. Physical Chemistry Chemical Physics 2019, 21, 1336–1343. [Google Scholar] [CrossRef]
- Li, Q.; Shen, X.; Xing, D. Carbon quantum dots as ROS-generator and -scavenger: A comprehensive review. Dyes and Pigments 2023, 208, 110784. [Google Scholar] [CrossRef]
- Innocenzi, P.; Stagi, L. Carbon dots as oxidant-antioxidant nanomaterials, understanding the structure-properties relationship. A critical review. Nanotoday 2023, 50, 101837. [Google Scholar] [CrossRef]
- Qiu, Y.; Wang, Z.; Owens, A.C.E.; Kulaots, I.; Chen, Y.; Kane, A.B.; Hurt, R.H. Antioxidant chemistry of graphene-based materials and its role in oxidation protection technology. Nanoscale 2014, 6, 11744–11755. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



