Submitted:
07 October 2025
Posted:
08 October 2025
You are already at the latest version
Abstract
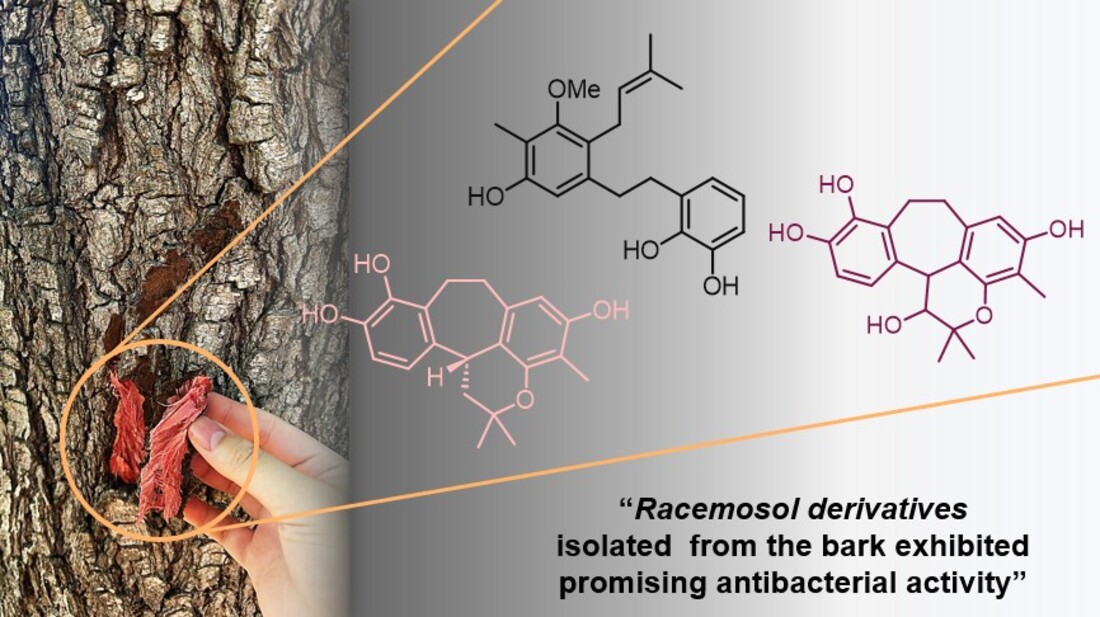
Keywords:
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedure
3.2. Plant Material
3.3. Extraction and Isolation
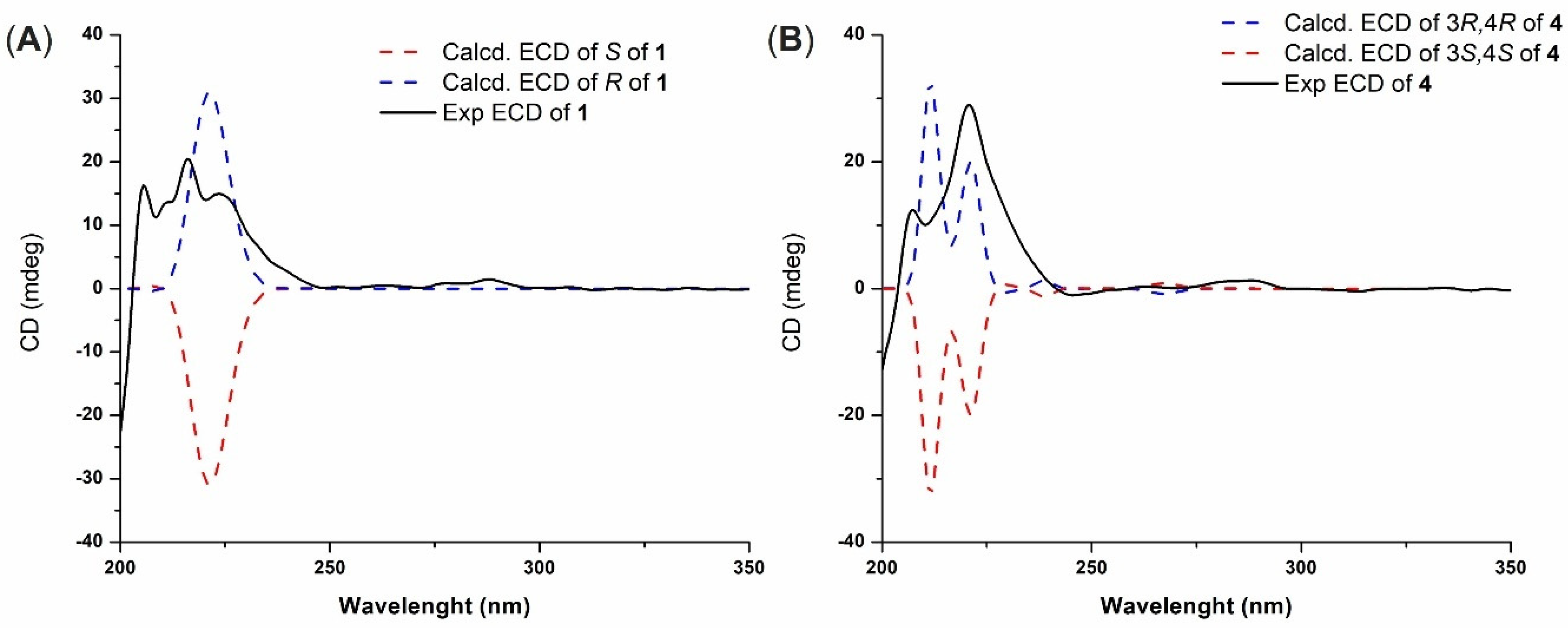
3.4. ECD Calculations
3.5. Antimicrobial Properties
3.5.1. Determination of Antimicrobial Activity
3.5.2. Overlay Spot Assay for Antimicrobial Screening
3.5.3. Microdilution Method for MIC and MBC
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thetsana, P.; Chaowuttikul, C.; Palanuvej, C.; Ruangrungsi, N. Pharmacognostic Specifications, Quercetin and Quercitrin Quantification in Bauhinia Malabarica Leaf. Pharmacognosy Journal 2019, 11, 155–160. [CrossRef]
- Kaewamatawong, R.; Kitajima, M.; Kogure, N.; Takayama, H. Flavonols from Bauhinia malabarica. J Nat Med 2008, 62, 364–365. [CrossRef]
- Kittakoop, P.; Kirtikara, K.; Tanticharoen, M.; Thebtaranonth, Y. Antimalarial Preracemosols A and B, Possible Biogenetic Precursors of Racemosol from Bauhinia malabarica Roxb. Phytochemistry 2000, 55, 349–352. [CrossRef]
- Songarsa, S.; Rajviroongit, S.; Sae-Tang, D.; Hannongbua, S.; Kirtikara, K.; Kittakoop, P. New Racemosol Derivatives as Potent Cyclooxygenase (COX) Inhibitors. Chem Biodivers 2005, 2, 1635–1647. [CrossRef]
- Verma, R.; Dash, S.; Ankita; Thakur, S.; Kumar, R.; Singh, G.; Kaur, C. Genus Bauhinia (Fabaceae): A Review from Phytochemistry to Pharmacology- Exploring Traditional Uses and Toxicological Insights across Asia. Phytomedicine 2024, 135, 156246. [CrossRef]
- Rashed, K.; Butnariu, M. Antimicrobial and Antioxidant Activities of Bauhinia racemosa Lam. and Chemical Content; 2014; Vol. 13.
- Schevenels, F.T.; Jadsadajerm, S.; Lekphrom, R.; Yodsin, N.; Suebrasri, T.; Senawong, T.; Wisetsai, A. Siamfuranones A–C, Three Novel Furanone Derivatives from the Flowers of Uvaria siamensis and Their Biological Activities. Nat Prod Res 2024, 1–10. [CrossRef]
- Wongwad, E.; Jadsadajerm, S.; Mungmai, L.; Wisetsai, A. Antioxidant, Cytotoxic, Anti-Glycation, and Anti-Tyrosinase Compounds from the Leaves of Uvaria siamensis. Chem Biodivers 2024, 21. [CrossRef]
- Tabtimmai, L.; Phonchan, T.; Thongprik, N.; Kaennakam, S.; Yodsin, N.; Choowongkomon, K.; Sonklin, C.; Jadsadajerm, S.; Wisetsai, A. New Oxepin and Dihydrobenzofuran Derivatives from Bauhinia saccocalyx Roots and Their Anti-Inflammatory, Cytotoxic, and Antioxidant Activities. J Nat Med 2025, 79, 543–555. [CrossRef]
- Jain, R.; Alam, S.; Saxena, U. A New Tetracyclic Phenol and Other Constituents from the Roots of Bauhinia racemosa. ChemInform 2002, 33, 219–219. [CrossRef]
- Anjaneyulu, A.S.R.; Reddy, A.V.R.; Reddy, D.S.K.; Cameron, T.S.; Roe, S.P. Racemosol: A Novel Tetracyclic Phenol from Bauhimia racemosa Lamk. Tetrahedron 1986, 42, 2417–2420. [CrossRef]
- Maillard, M.P.; Recio-Iglesias, M.; Saadou, M.; Stoeckli-Evans, H.; Hostettmann, K. Novel Antifungal Tetracyclic Compounds from Bauhinia rufescens LAM. Helv Chim Acta 1991, 74, 791–799. [CrossRef]
- Grimblat, N.; Zanardi, M.M.; Sarotti, A.M. Beyond DP4: An Improved Probability for the Stereochemical Assignment of Isomeric Compounds Using Quantum Chemical Calculations of NMR Shifts. J Org Chem 2015, 80, 12526–12534. [CrossRef]
- Lobiuc, A.; Pavăl, N.-E.; Mangalagiu, I.I.; Gheorghiță, R.; Teliban, G.-C.; Amăriucăi-Mantu, D.; Stoleru, V. Future Antimicrobials: Natural and Functionalized Phenolics. Molecules 2023, 28, 1114. [CrossRef]
- Kamal, G.M.; Uddin, J.; Asmari, M.; Noreen, A.; Liaqat, A.; Sabir, A.; Khan, J.; Iqbal, R.; Sharma, A.; Nayik, G.A.; et al. Natural Polyphenols as a Promising Aquatic Food Preservative: A Concurrent Review. J Agric Food Res 2025, 22, 102046. [CrossRef]
- Kumar, R.S.; Sivakumar, T.; Sunderam, R.S.; Gupta, M.; Mazumdar, U.K.; Gomathi, P.; Rajeshwar, Y.; Saravanan, S.; Kumar, M.S.; Murugesh, K.; et al. Antioxidant and Antimicrobial Activities of Bauhinia racemosa L. Stem Bark. BJMBR 2005, 38, 1015–1024. [CrossRef]
- Ju, J.-H.; Li, X.-T.; Shen, Q.-K.; Guo, H.-Y.; Quan, Z.-S.; Yin, X.-M. Advances in Structural Modification of Natural Products with Antimicrobial Activity. Fitoterapia 2025, 186, 106815. [CrossRef]
- Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V,Mennucci B, Petersson GA, et al. 2013. Gaussian 09, revision D.01. Wallingford (CT): Gaussian Inc.
- Sirisarn, W.; Mordmuang, A.; Kerdkumthong, K.; Saeheng, S. Exploring Bougainvillea Glabra Flowers: A Promising Source of Natural Antimicrobial and Anticancer Agents. J Appl Biol Biotechnol 2024. [CrossRef]
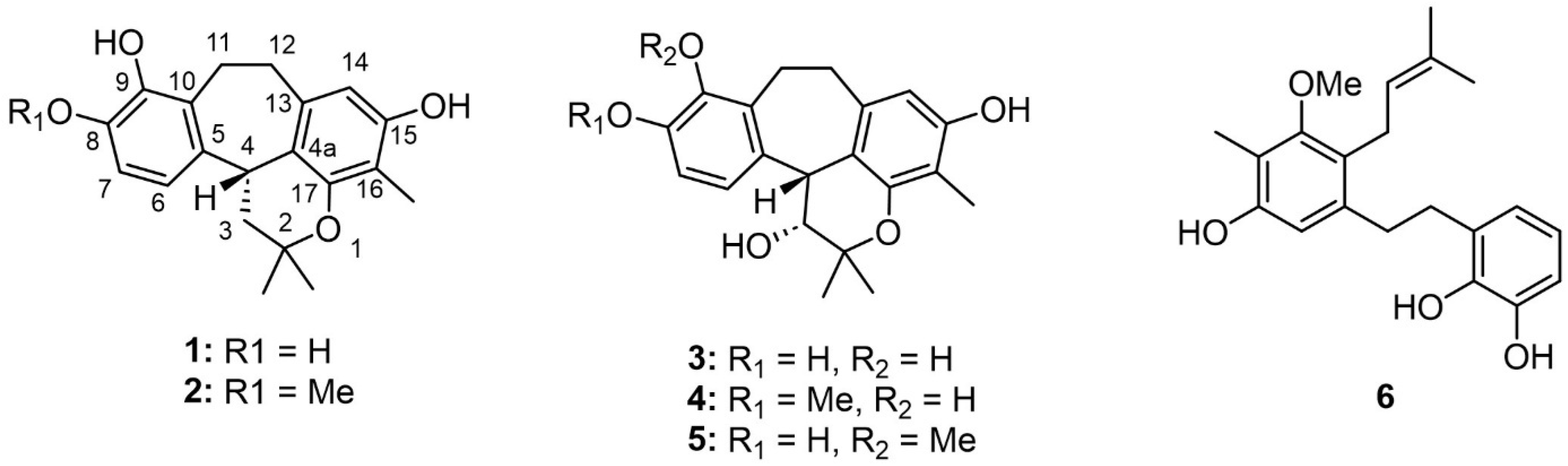
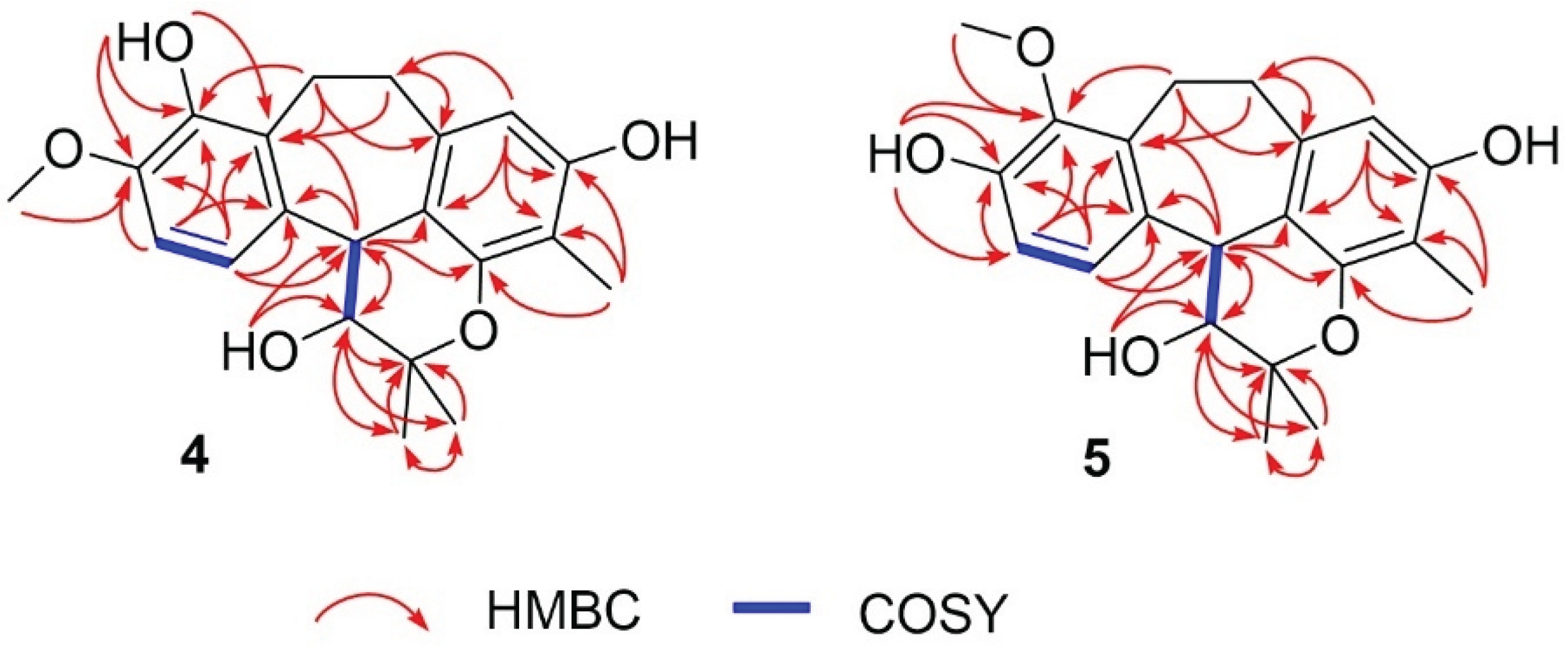
| No. | 3-Hydroxy-8-O-methylracemosol (4) | 3-Hydroxy-9-O-methylracemosol (5) | |||
|---|---|---|---|---|---|
| δC | δH (mult, J in Hz) | δC | δH (mult, J in Hz) | ||
| 2 | 76.9 | - | 76.9 | - | |
| 3 | 71.9 | 4.31 (m) | 71.9 | 4.28 (m) | |
| 4 | 41.4 | 4.31 (m) | 41.3 | 4.28 (m) | |
| gem-Me | 27.5 | 1.55 (s) | 27.5 | 1.55 (s) | |
| 17.7 | 1.23 (s) | 17.7 | 1.23 (s) | ||
| 4a | 115.7 | - | 115.5 | - | |
| 5 | 135.5 | - | 134.1 | - | |
| 6 | 116.0 | 6.90 (d, 8.6) | 114.1 | 6.68 (d, 8.4) | |
| 7 | 109.0 | 6.72 (d, 8.6) | 121.3 | 7.04 (d, 8.4) | |
| 8 | 146.5 | - | 149.2 | - | |
| 9 | 143.5 | - | 145.4 | - | |
| 10 | 129.6 | - | 137.2 | - | |
| 11 | 22.3 | 3.37 (m) 3.04 (m) |
23.1 | 3.25 (m) 3.15 (m) |
|
| 12 | 34.3 | 3.12 (m) 2.76 (m) |
34.9 | 3.17 (m) 2.80 (m) |
|
| 13 | 136.4 | - | 136.1 | - | |
| 14 | 110.7 | 6.11 (s) | 110.7 | 6.11 (s) | |
| 15 | 154.7 | - | 154.7 | - | |
| 16 | 110.1 | - | 110.2 | - | |
| 17 | 152.1 | - | 152.1 | - | |
| 16-Me | 8.7 | 1.97 (s) | 8.7 | 1.97 (s) | |
| 8-OMe | 56.3 | 3.80 (s) | - | - | |
| 9-OMe | - | - | 61.3 | 3.78 (s) | |
| 3-OH | - | 4.09 (d, 4.8) | - | 4.17 (s) | |
| 8-OH | - | - | - | 7.87 (s) | |
| 9-OH | - | 7.30 (s) | - | - | |
| 15-OH | - | 7.78 (s) | - | 7.80 (s) | |
| Pathogenic isolates | Antimicrobial activity of compounds 1–6 (100 ug) | Positive control (amp/gen) | Negative control (1% DMSO) | |||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |||
| Gram-positive bacteria | ||||||||
| Bacillus subtilis 7988 (Clinical isolate) | + | + | + | + | - | + | +* | - |
| Bacillus subtilis ATCC6051 | + | + | + | + | - | + | +* | - |
| Enterococcus faecalis 4232 (Clinical isolates) | + | + | + | + | - | + | +* | - |
| Listeria monocytogenes (Clinical isolates) | + | + | + | + | - | + | +* | - |
| Staphylococcus aureus ATCC25923 | + | + | + | + | - | + | +* | - |
| Staphylococcus aureus ATCC29213 | + | + | + | + | - | + | +* | - |
| Methicillin Resistant Staphylococcus aureus (Clinical isolate) | + | + | + | + | - | + | −* | - |
| Staphylococcus epidermitis 35984 | + | + | + | + | - | + | +* | - |
| Streptococcus pyogenes ATCC49619 | + | + | + | + | - | + | +* | - |
| Gram-negative bacteria | - | |||||||
| Acinetobacter baumannii ATCC19606 | + | - | - | + | - | + | +** | - |
| Multidrug-resistant Acinetobacter baumannii (MDR) | + | + | - | + | - | + | −** | - |
| Escherichia coli ATCC25922 | + | + | - | + | - | + | +** | - |
| Escherichia coli O157:H7 | + | + | + | + | - | + | +** | - |
| Klebsiella pneumoniae ATCC70063 | + | + | - | + | - | + | +** | - |
| Pseudomonas aeruginosa ATCC27853 | - | - | - | + | - | + | +** | - |
| Salmonella enterica serotype Typhi (Clinical isolates) | + | + | - | + | - | + | +** | - |
| Shigella enteritis (Clinical isolates) | - | - | - | - | - | - | +** | - |
| Pathogenic isolates | Minimum Inhibitory Concentration (MIC)/ Minimum Bactericidal Concentration (MBC) in µg/µL of compounds 1–6 | Minimum Inhibitory Concentration (MIC) of Ampicillin/Gentamycin (µg/ml) | Negative control (1% DMSO) | |||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |||
| Gram-positive bacteria | ||||||||
| Bacillus subtilis 7988 (Clinical isolate) | 0.625/0.625 | 0.625/2.5 | 2.5/2.5 | 0.625/0.625 | NA | 0.15625/0.15625 | 0.5/0.5 | R |
| Bacillus subtilis ATCC6051 | 0.15625/>5 | 0.3125/>5 | 1.25/>5 | 0.3125/>5 | NA | 0.15625/>5 | 0.125/0.125 | R |
| Enterococcus faecalis 4232 (Clinical isolates) | 0.15625/>5 | 0.625/>5 | 2.5/>5 | 1.25/>5 | NA | 0.3125/0.625 | 0.25/NA | R |
| Listeria monocytogenes (Clinical isolates) | 0.15625/0.3125 | 0.625/>5 | 0.625/>5 | 0.3125/1.25 | NA | 0.3125/0.3125 | 0.25/2 | R |
| Staphylococcus aureus ATCC25923 | 0.15625/0.3125 | 0.625/>5 | 0.625/>5 | 0.625/>5 | NA | 0.3125/0.3125 | 0.25/0.5 | R |
| Staphylococcus aureus ATCC29213 | 0.15625/0.3125 | 0.625/>5 | 0.625/>5 | 0.3125/>5 | NA | 0.3125/0.625 | 0.25/0.125 | R |
| Methicillin Resistant Staphylococcus aureus (Clinical isolate) | 0.15625/0.15625 | 0.625/>5 | 0.625/>5 | 0.3125/>5 | NA | 0.3125/0.3125 | NA/0.25 | R |
| Staphylococcus epidermitis 35984 | 0.15625/0.625 | 1.25/>5 | 1.25/>5 | 0.15625/>5 | NA | 0.3125/0.625 | 0.5/0.25 | R |
| Streptococcus pyogenes ATCC49619 | 0.3125/0.3125 | 1.25/>5 | 1.25/>5 | 0.3125/>5 | NA | 0.3125/0.625 | 0.25/0.125 | R |
| Gram-negative bacteria | ||||||||
| Acinetobacter baumannii ATCC19606 | 0.3125/0.625 | NA | NA | 0.3125/5 | NA | 0.15625/0.3125 | NA/1 | R |
| Multidrug-resistant Acinetobacter baumannii (MDR) | 1.25/5 | 1.25/>5 | NA | 1.25/5 | NA | 1.25/5 | NA/NA | R |
| Escherichia coli ATCC25922 | 2.5/>5 | 2.5/>5 | NA | 2.5/5 | NA | 2.5/>5 | NA/0.031 | R |
| Escherichia coli O157:H7 | 2.5/5 | 2.5/5 | 2.5/5 | 2.5/>5 | NA | 2.5/5 | NA/0.063 | R |
| Klebsiella pneumoniae ATCC70063 | 2.5/>5 | 2.5/>5 | NA | 2.5/>5 | NA | 2.5/>5 | NA/0.062 | R |
| Pseudomonas aeruginosa ATCC27853 | NA | NA | NA | 2.5/>5 | NA | 2.5/5 | NA/4 | R |
| Salmonella enterica serotype Typhi (Clinical isolates) | 2.5/>5 | 2.5/>5 | NA | 2.5/>5 | NA | 2.5/5 | NA/2 | R |
| Shigella enteritis (Clinical isolates) | NA | NA | NA | NA | NA | NA | NA/1 | R |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




