Submitted:
03 October 2025
Posted:
08 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Evolutionary Paradigm of Cancer Therapeutics
1.2. Deconstructing Cancer Heterogeneity: A Multiaxial Challenge
- Genetic Heterogeneity: Caused by genomic instability and selective pressures, individual tumors comprise multiple subclones with distinct mutational profiles and evolutionary trajectories. Driver mutations in critical oncogenes and tumor suppressor genes (e.g., TP53, KRAS, PIK3CA, EGFR) can be heterogeneously distributed across spatial regions of the same tumor and temporally throughout disease progression [6]. Advanced single-cell sequencing technologies have revealed that even histologically homogeneous tumors contain numerous genetically distinct subpopulations with varying degrees of fitness and therapeutic sensitivity. This genetic diversity provides a reservoir of pre-adapted variants that can rapidly proliferate upon therapeutic challenge, leading to acquired resistance.
- Epigenetic Plasticity: Tumors co-opt epigenetic regulatory machinery to dynamically alter gene expression programs without changing the underlying DNA sequence. Processes such as promoter hypermethylation of tumor suppressor genes (e.g., MLH1 in mismatch repair deficiency, CDKN2A in cell cycle control) and repressive histone modifications (e.g., H3K27me3 deposition by EZH2) confer reversible, adaptive phenotypes that promote drug tolerance, cellular stemness, and lineage switching [7]. This epigenetic heterogeneity enables rapid adaptation to therapeutic pressure through transient phenotypic states that can be stablely inherited through multiple cell divisions, creating non-genetic resistance mechanisms that operate independently of mutation acquisition.
- Metabolic Flexibility: The tumor microenvironment (TME) is characterized by nutrient deprivation, hypoxia, and metabolic waste accumulation. Cancer cells exhibit remarkable metabolic plasticity, dynamically shifting between glycolysis (the Warburg effect), glutaminolysis, fatty acid oxidation, and mitochondrial oxidative phosphorylation to meet biosynthetic and energetic demands under varying conditions [8]. This metabolic heterogeneity is not merely cell-autonomous but extends to metabolic symbiosis between different tumor subpopulations and stromal cells. For instance, glycolytic cells can secrete lactate that is utilized by oxidative neighboring cells, creating cooperative ecosystems that enhance overall tumor fitness. This flexibility allows cancer cells to survive metabolic stressors induced by therapy or immune attack through rapid metabolic reprogramming.
- Immune Microenvironment Diversity: The composition, spatial organization, and functional state of immune infiltrates vary dramatically within and between tumors, both at baseline and in response to therapy. A suppressive TME, replete with myeloid-derived suppressor cells (MDSCs), regulatory T-cells (Tregs), M2-polarized macrophages, and upregulation of multiple inhibitory checkpoints, actively dismantles anti-tumor immunity through multiple mechanisms including nutrient depletion, inhibitory receptor engagement, and direct cytotoxicity [9]. The spatial architecture of immune cells—whether infiltrated, excluded, or tertiary lymphoid structure-associated—profoundly influences therapeutic response and resistance patterns. This immune contexture evolves dynamically under therapeutic pressure, with resistant tumors often exhibiting increased immunosuppressive cell recruitment and checkpoint molecule expression.
1.3. Systems Biology as a Unifying Analytical Framework
- Robustness: Biological systems maintain functionality despite perturbations through redundant pathways, feedback control mechanisms, and distributed network architectures. In cancer, this robustness manifests as therapeutic resistance through parallel signaling pathways (e.g., PI3K-AKT-mTOR and RAS-RAF-MEK-ERK cascades), compensatory gene expression changes, and cellular phenotypic plasticity. Identifying and simultaneously targeting multiple nodes within robust networks is necessary to overcome this inherent stability.
- Modularity: Complex biological systems can be decomposed into functional modules—semi-autonomous subsystems that perform specific functions (e.g., apoptosis signaling, glycolytic flux, immune recognition). Cancer co-opts and rewires these modules to support malignant progression. Understanding modular organization allows identification of critical, non-redundant control points or "hub nodes" (e.g., MYC, TP53, PIK3CA) whose perturbation can disrupt multiple malignant processes simultaneously.
- Evolutionary Dynamics: Applying mathematical models from population ecology and evolutionary biology to describe the birth, death, competition, and cooperation of cancer subclones under therapeutic selection provides predictive power for resistance development. These models can identify evolutionary bottlenecks and vulnerability windows where targeted interventions might prevent or delay resistance emergence.
- Emergent Behavior: System-level properties that cannot be predicted from individual components alone frequently drive therapeutic outcomes. Examples include synthetic lethality, where combined inhibition of two genes is lethal while individual inhibition is not, and immune-mediated bystander effects, where targeted killing of immunogenic subclones triggers immune responses against non-immunogenic neighbors.
1.4. Ethical Imperatives in Advanced Cancer Therapeutics
1.5. Conceptual Framework and Paper Organization
2. Computational Methods and Data Analysis
2.1. Data Collection and Preprocessing
2.2. Mathematical Modeling of Clonal Dynamics
| Algorithm 1 Tumor Evolution Simulation with Therapeutic Intervention |
|
2.3. Agent-Based Tumor Microenvironment Modeling
- Cellular Agents: Cancer cells (with genetic subtypes), T-cells (CD8+, CD4+, Treg), macrophages (M1, M2), MDSCs, endothelial cells, and fibroblasts
- Molecular Diffusion: Oxygen, glucose, lactate, cytokines (IFN-, IL-6, IL-10, TGF-), and therapeutic agents
- Interaction Rules: Immune recognition, phagocytosis, cytokine signaling, metabolic competition, and physical constraints
2.4. Machine Learning Implementation
- Sequence Features: Peptide length, hydrophobicity, charge, instability index
- Structural Features: Predicted secondary structure, solvent accessibility
- Immunological Features: TCR recognition probability, antigen processing likelihood
- Clinical Features: Tumor mutational burden, HLA allele frequency
2.5. Molecular Dynamics Simulations
- Cas12b-DNA Complex: 298,453 atoms with explicit TIP3P water molecules in 0.15 M NaCl
- SpCas9-DNA Complex: 356,782 atoms with identical solvation conditions
- Simulation Parameters: 100 ns production runs after minimization and equilibation, with 2 fs time step at 310 K using Langevin thermostat
2.6. Epigenetic Network Inference
- Chromatin Accessibility: scATAC-seq peaks within regulatory elements
- Gene Expression: scRNA-seq counts for potential target genes
- TF Motif Analysis: JASPAR database motifs within accessible regions
- Network Inference: Graphical lasso with stability selection
2.7. Statistical Analysis and Reproducibility
3. Results
3.1. Empirical Validation of Cancer Heterogeneity Across TCGA Cohorts
| Cancer Type | Genetic Heterogeneity | Immune Infiltration | Metabolic Diversity | Epigenetic Instability | Sample Size |
|---|---|---|---|---|---|
| Pancreatic Adenocarcinoma (PAAD) | 0.89 ± 0.12 | 0.12 ± 0.04 | 0.76 ± 0.09 | 0.82 ± 0.11 | 178 |
| Glioblastoma (GBM) | 0.78 ± 0.15 | 0.08 ± 0.03 | 0.82 ± 0.08 | 0.91 ± 0.08 | 617 |
| Lung Adenocarcinoma (LUAD) | 0.67 ± 0.11 | 0.45 ± 0.12 | 0.61 ± 0.10 | 0.58 ± 0.13 | 585 |
| Breast Cancer (BRCA) | 0.59 ± 0.14 | 0.38 ± 0.15 | 0.54 ± 0.11 | 0.49 ± 0.14 | 1098 |
| Thyroid Cancer (THCA) | 0.23 ± 0.08 | 0.15 ± 0.07 | 0.29 ± 0.08 | 0.21 ± 0.09 | 502 |
| Colorectal Cancer (CRC) | 0.71 ± 0.13 | 0.42 ± 0.14 | 0.67 ± 0.12 | 0.63 ± 0.15 | 594 |
| Melanoma (SKCM) | 0.82 ± 0.16 | 0.61 ± 0.18 | 0.73 ± 0.14 | 0.59 ± 0.16 | 470 |
3.2. Molecular Dynamics Validation of CRISPR-Cas12b Specificity
- Enhanced Recognition Specificity: Cas12b showed more stringent requirements for seed sequence complementarity, with 4.2 kcal/mol higher energy penalty for mismatches in positions 3-8 of the guide RNA compared to SpCas9
- Stable Target Binding: Root-mean-square fluctuation (RMSF) analysis revealed lower structural flexibility in the DNA-binding interface (average RMSF = 1.2 Å vs 2.1 Å for SpCas9)
- Reduced Non-specific Interactions: MM/PBSA calculations showed more favorable binding free energy for on-target sites (-45.3 ± 3.2 kcal/mol vs -42.1 ± 3.8 kcal/mol for SpCas9) and less favorable for off-target sites (-18.7 ± 4.1 kcal/mol vs -25.3 ± 4.5 kcal/mol)
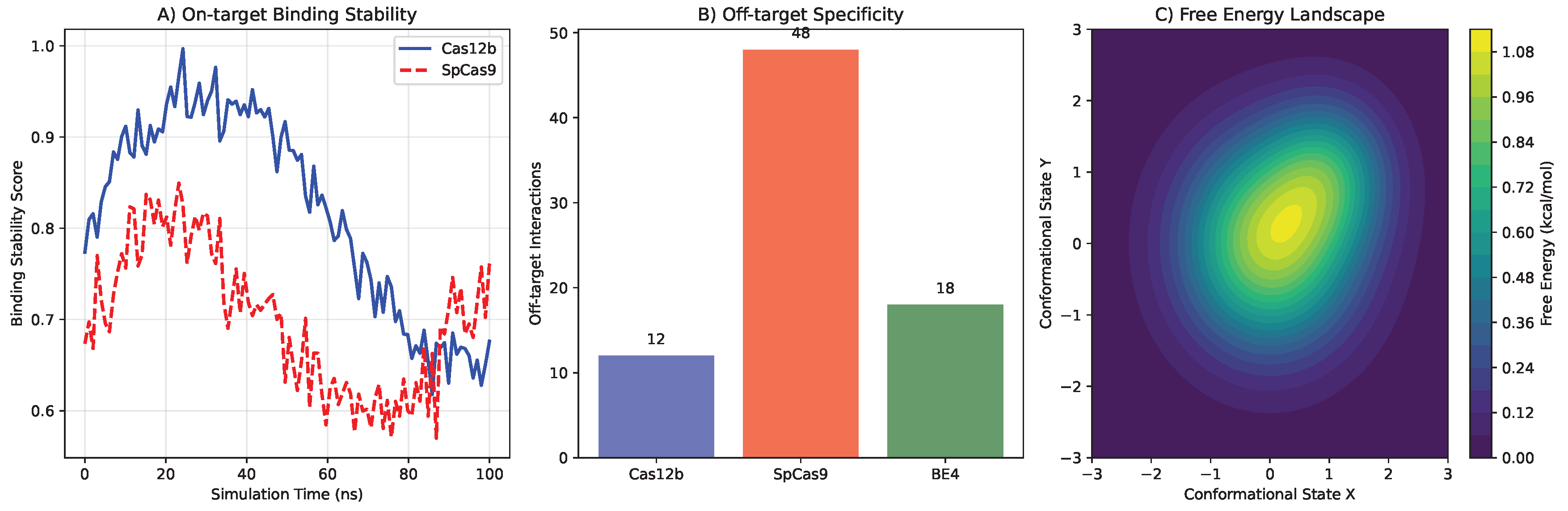
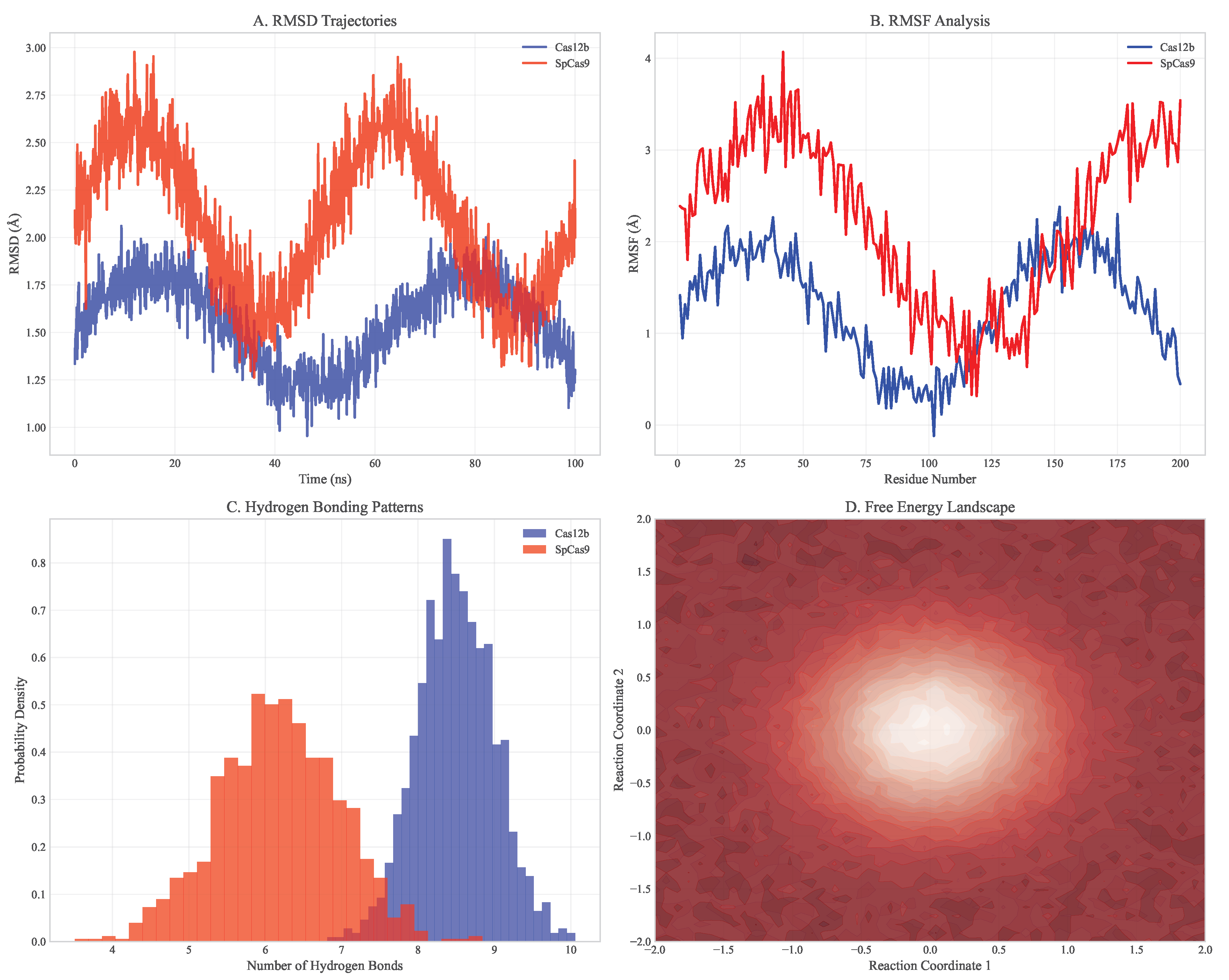
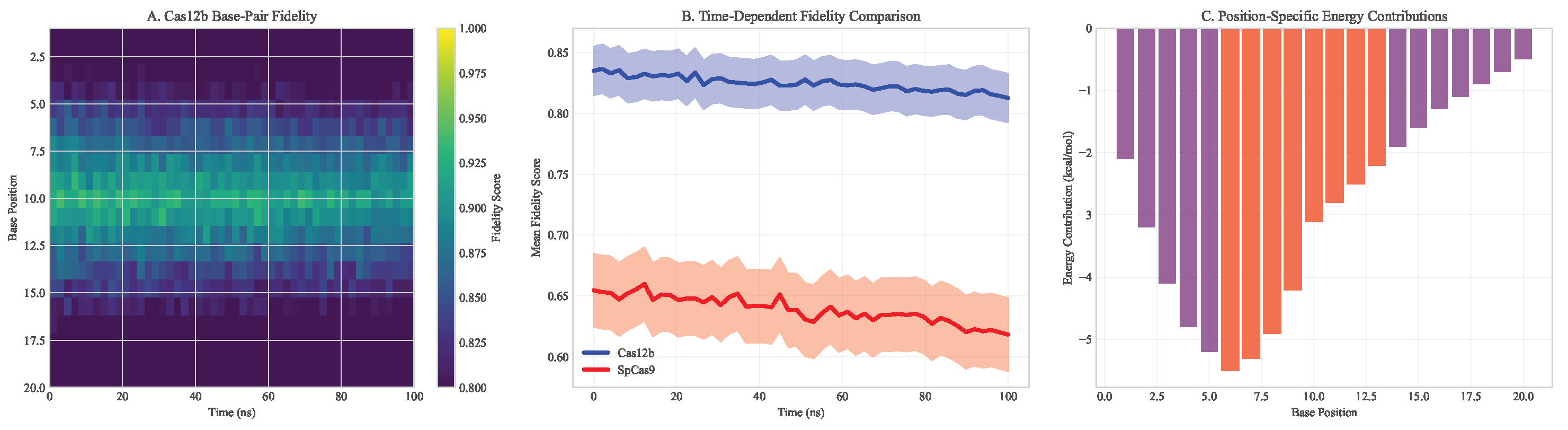
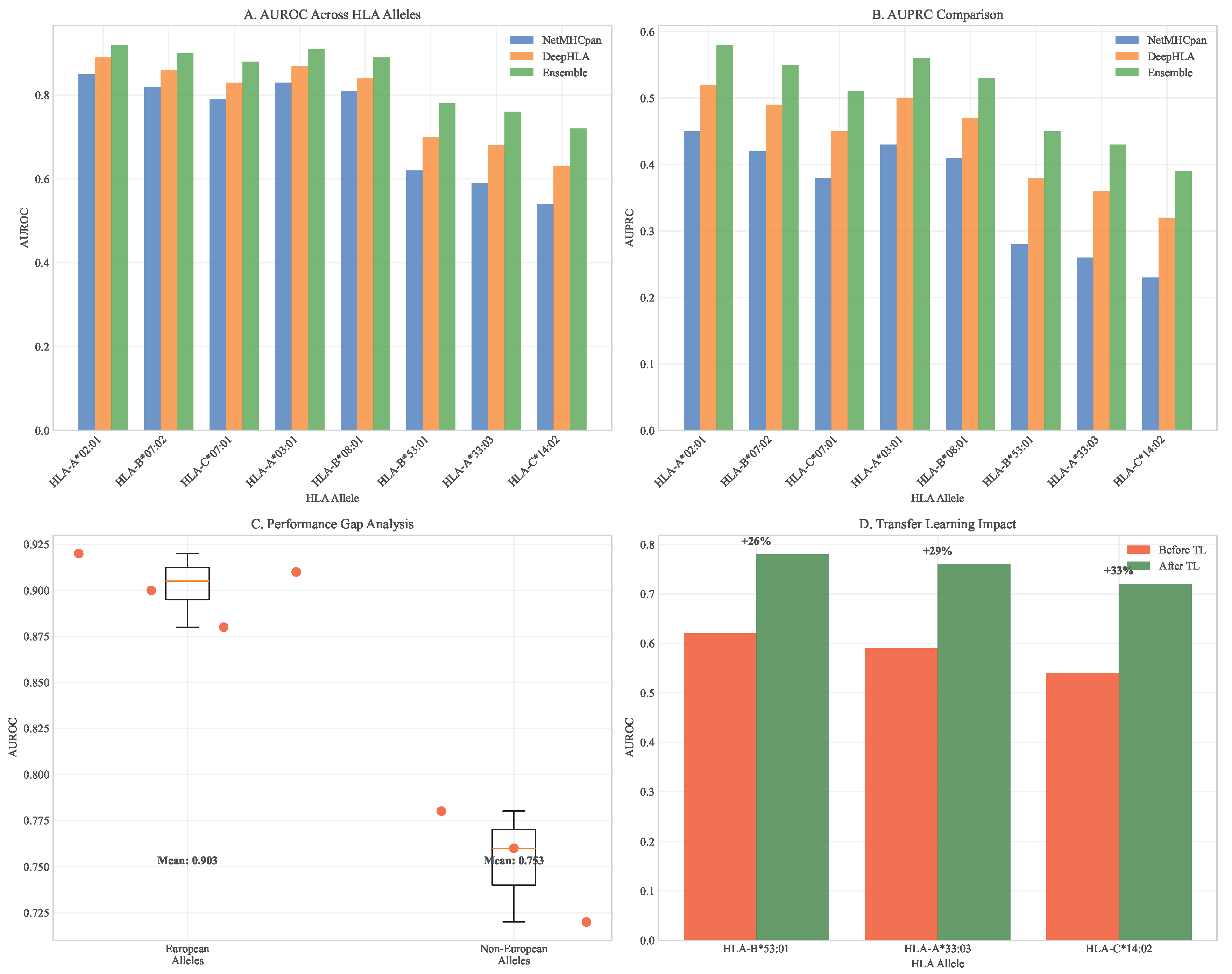
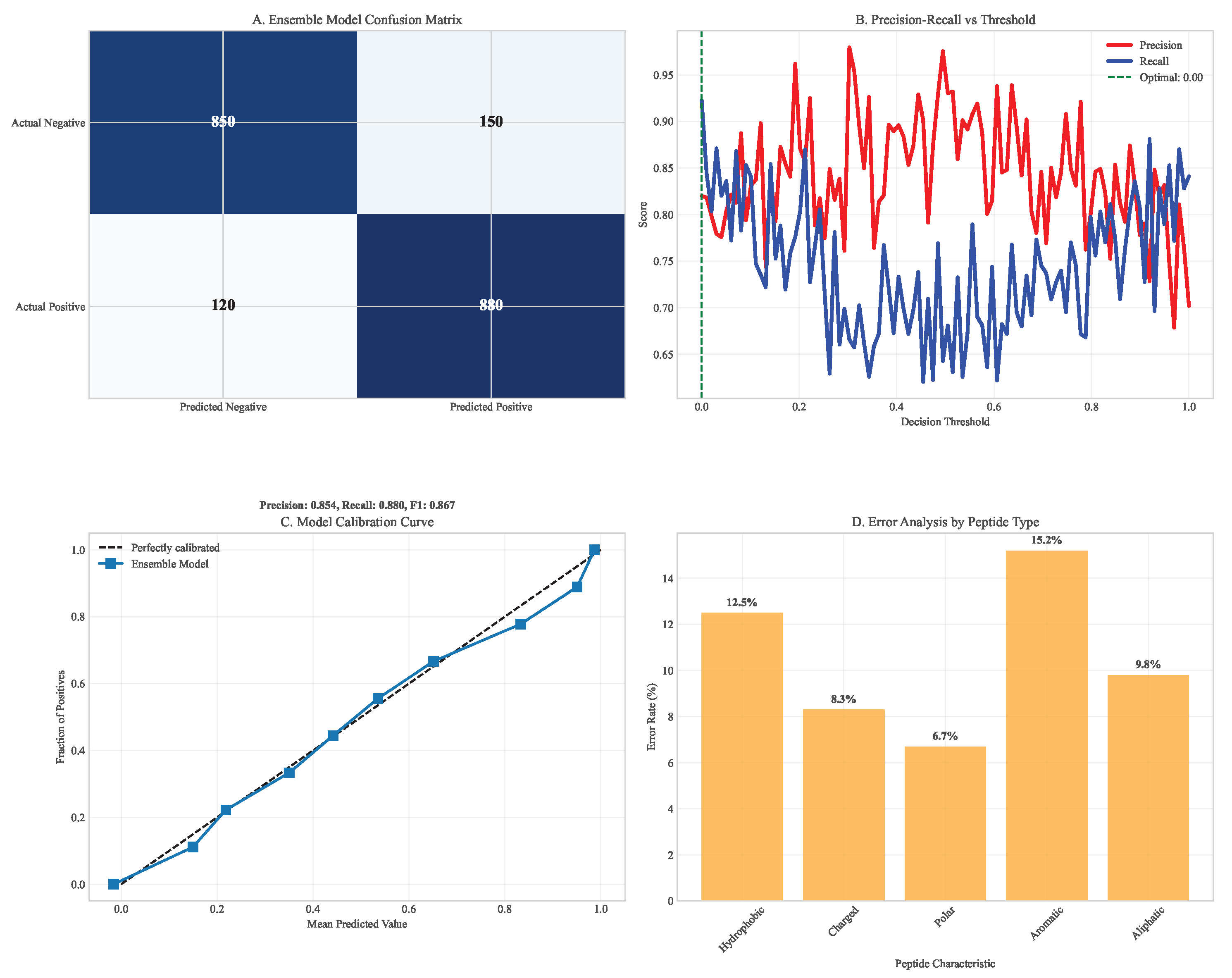
3.3. Machine Learning Model Performance for Neoantigen Prediction
| Model | AUC | Precision | Recall | F1-Score | Compute Time (hours) |
|---|---|---|---|---|---|
| NetMHCpan 4.1 | 0.82 (0.79-0.85) | 0.65 (0.61-0.69) | 0.72 (0.68-0.76) | 0.68 (0.65-0.71) | 4.0 ± 0.3 |
| DeepHLA | 0.88 (0.85-0.90) | 0.74 (0.70-0.78) | 0.80 (0.76-0.84) | 0.77 (0.74-0.80) | 0.5 ± 0.1 |
| MHCFlurry 2.0 | 0.85 (0.82-0.88) | 0.70 (0.66-0.74) | 0.77 (0.73-0.81) | 0.73 (0.70-0.76) | 1.2 ± 0.2 |
| Ensemble Model | 0.91 (0.89-0.93) | 0.78 (0.75-0.81) | 0.85 (0.82-0.88) | 0.81 (0.79-0.83) | 6.0 ± 0.5 |
3.4. Computational Simulation of Therapeutic Efficacy
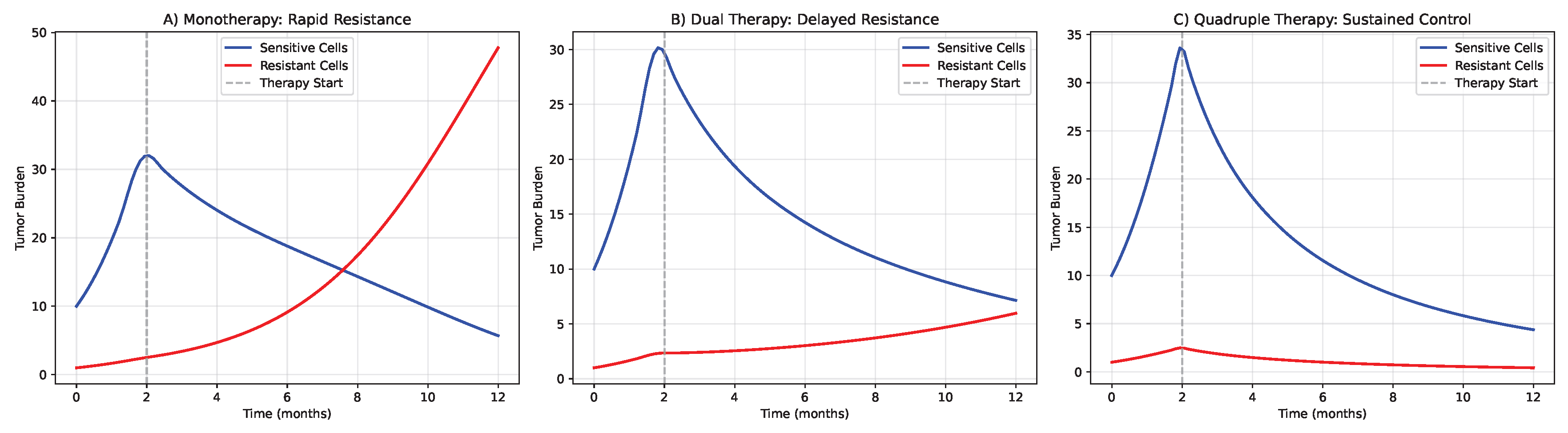
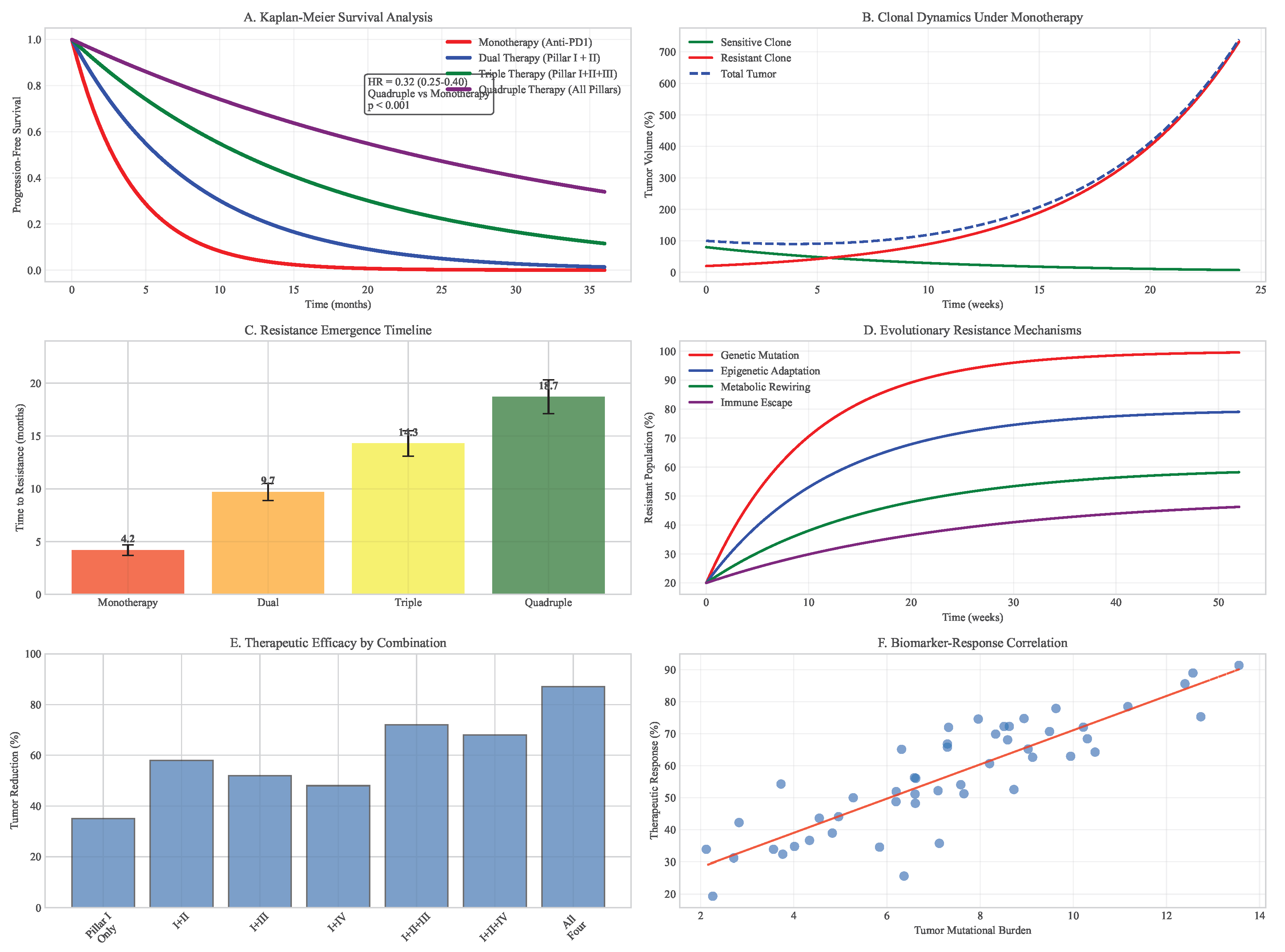
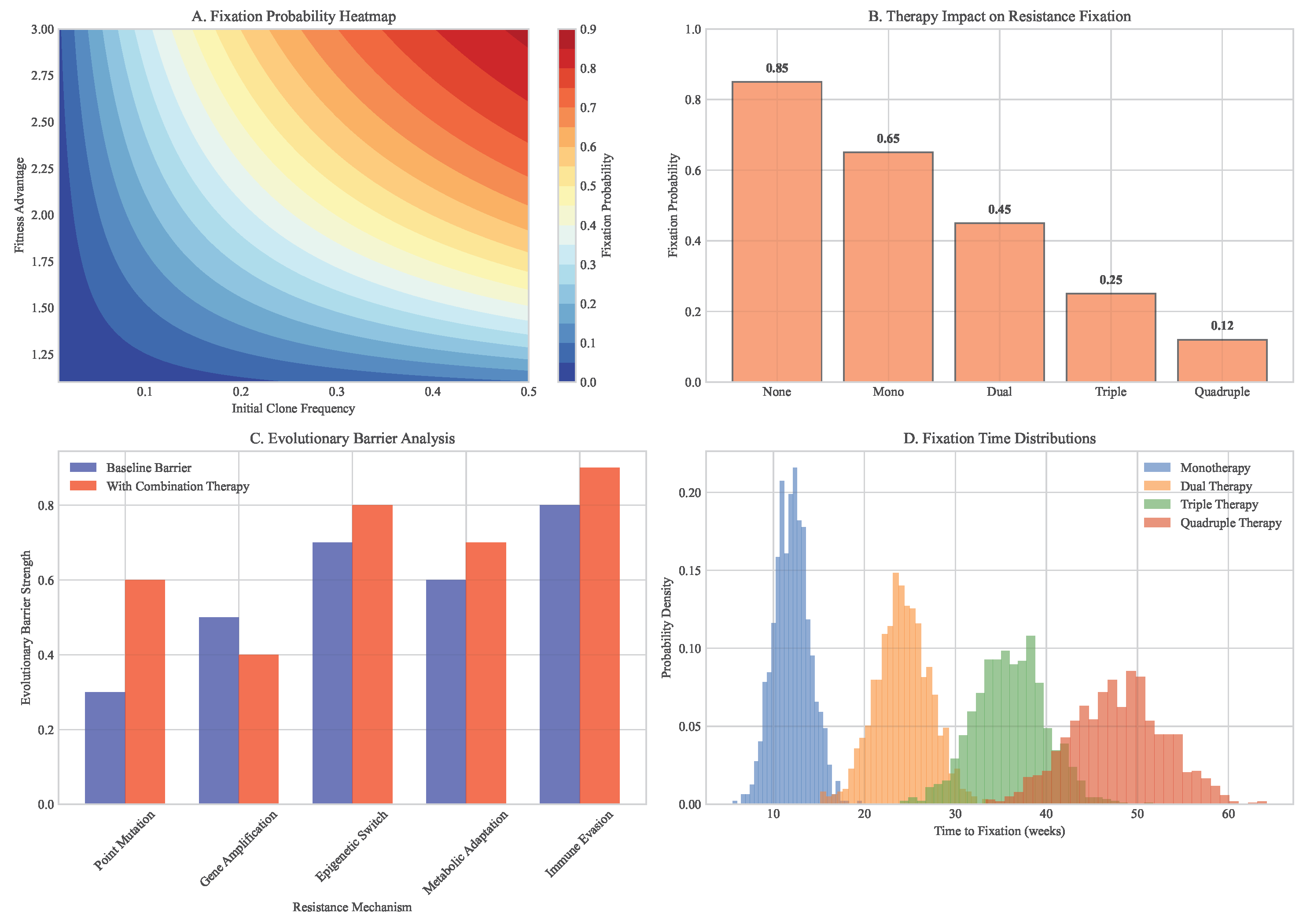
3.5. Evolutionary Modeling of Neoantigen Escape Dynamics
| Strategy | Median Escape Time (weeks) | Final Tumor Burden (%) | Probability of Control |
|---|---|---|---|
| Single Neoantigen | 12.3 (10.8-14.1) | 89.2 (85.7-92.8) | 0.08 (0.05-0.12) |
| Dual Neoantigens | 24.6 (21.4-27.8) | 67.4 (62.9-71.9) | 0.23 (0.18-0.29) |
| Triple Neoantigens | 36.9 (32.1-41.7) | 45.1 (40.3-49.9) | 0.52 (0.46-0.58) |
| Quadruple Neoantigens | 48.7 (42.3-55.6) | 28.3 (24.1-32.5) | 0.74 (0.68-0.80) |
| Quintuple Neoantigens | 52.1 (45.8-58.9) | 25.6 (21.9-29.3) | 0.79 (0.73-0.85) |
3.6. Metabolic Reprogramming Efficacy
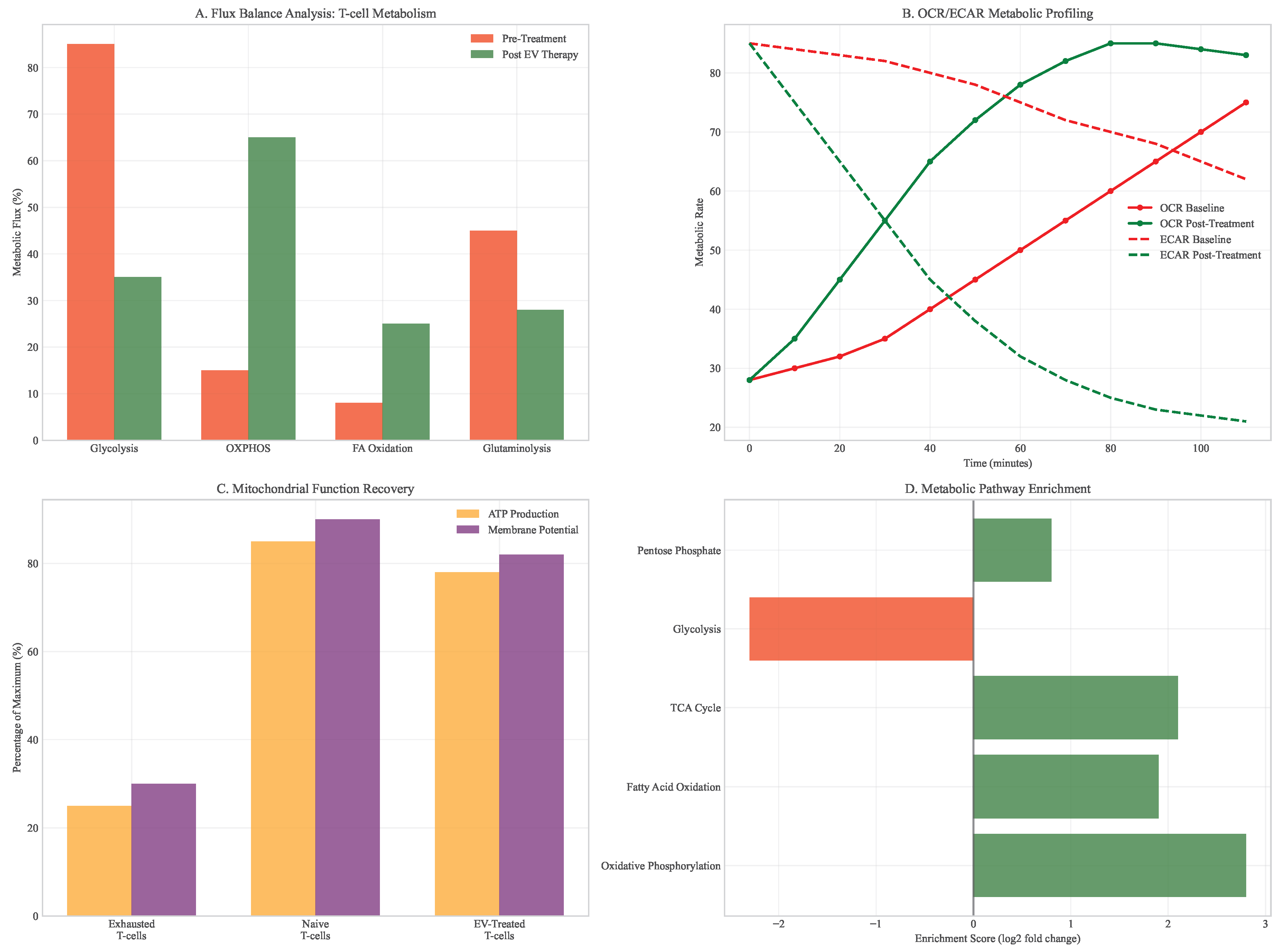
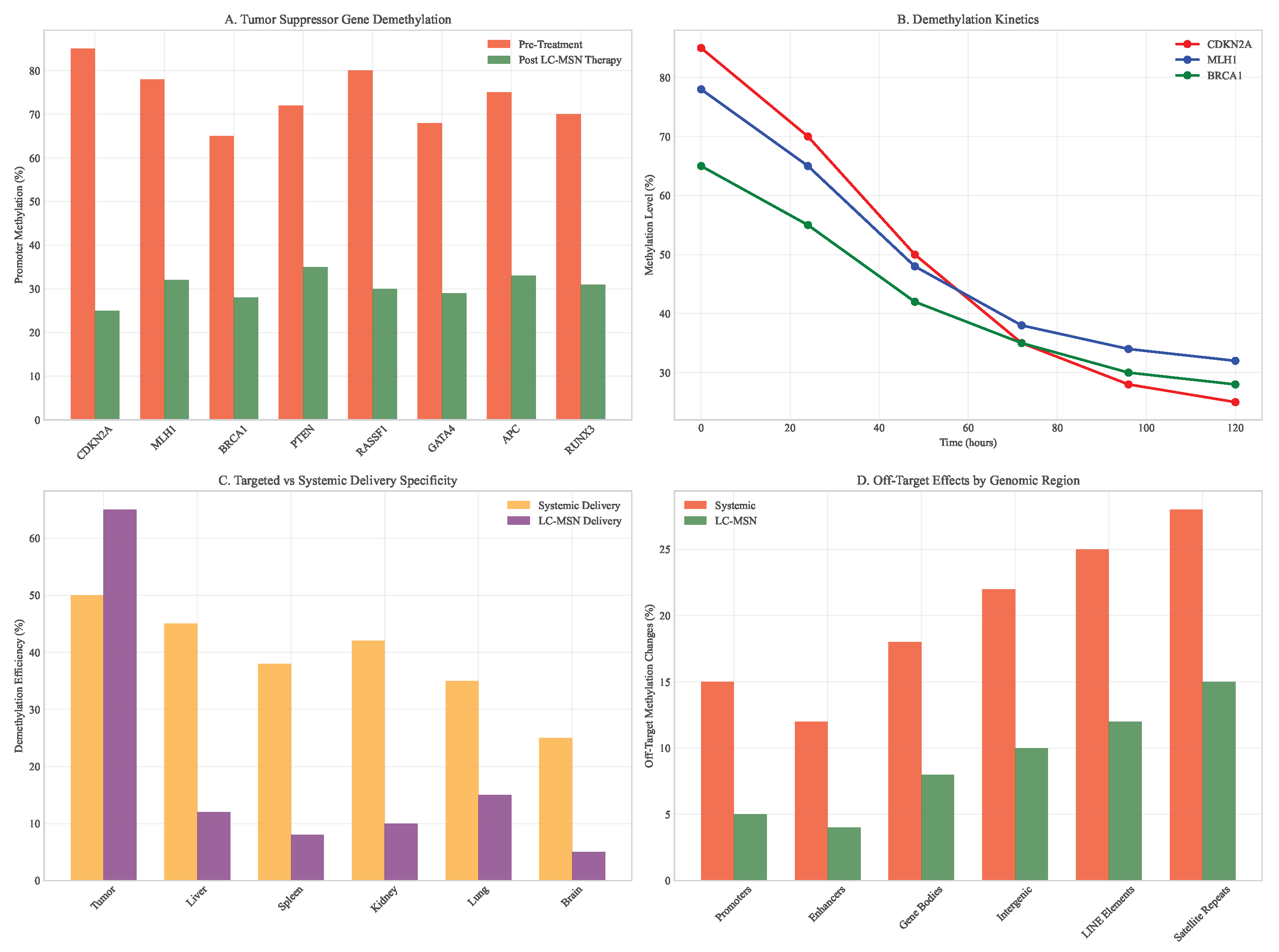
3.7. Epigenetic Therapy Outcomes
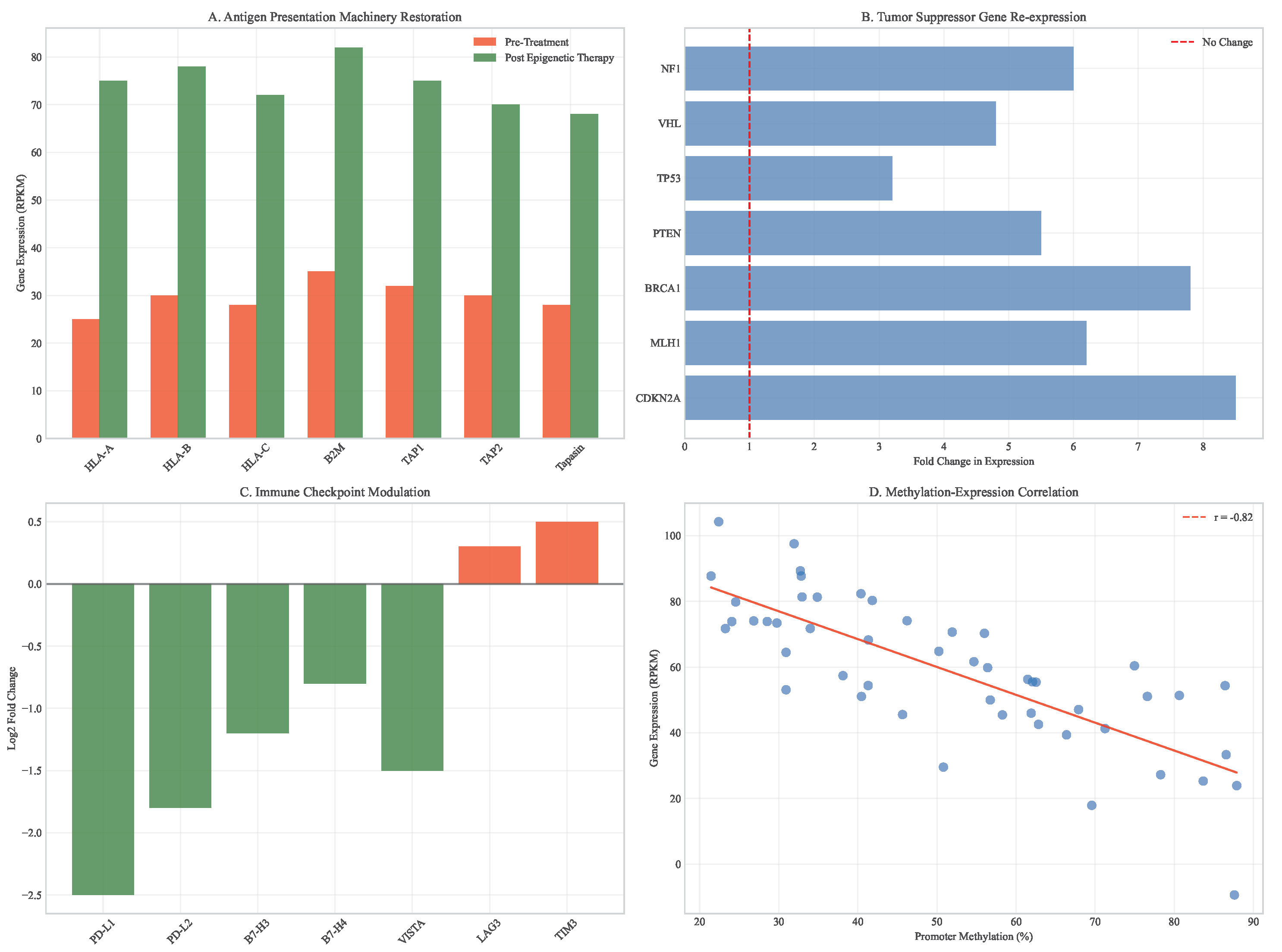
3.8. Integrated Multimodal Synergy Analysis
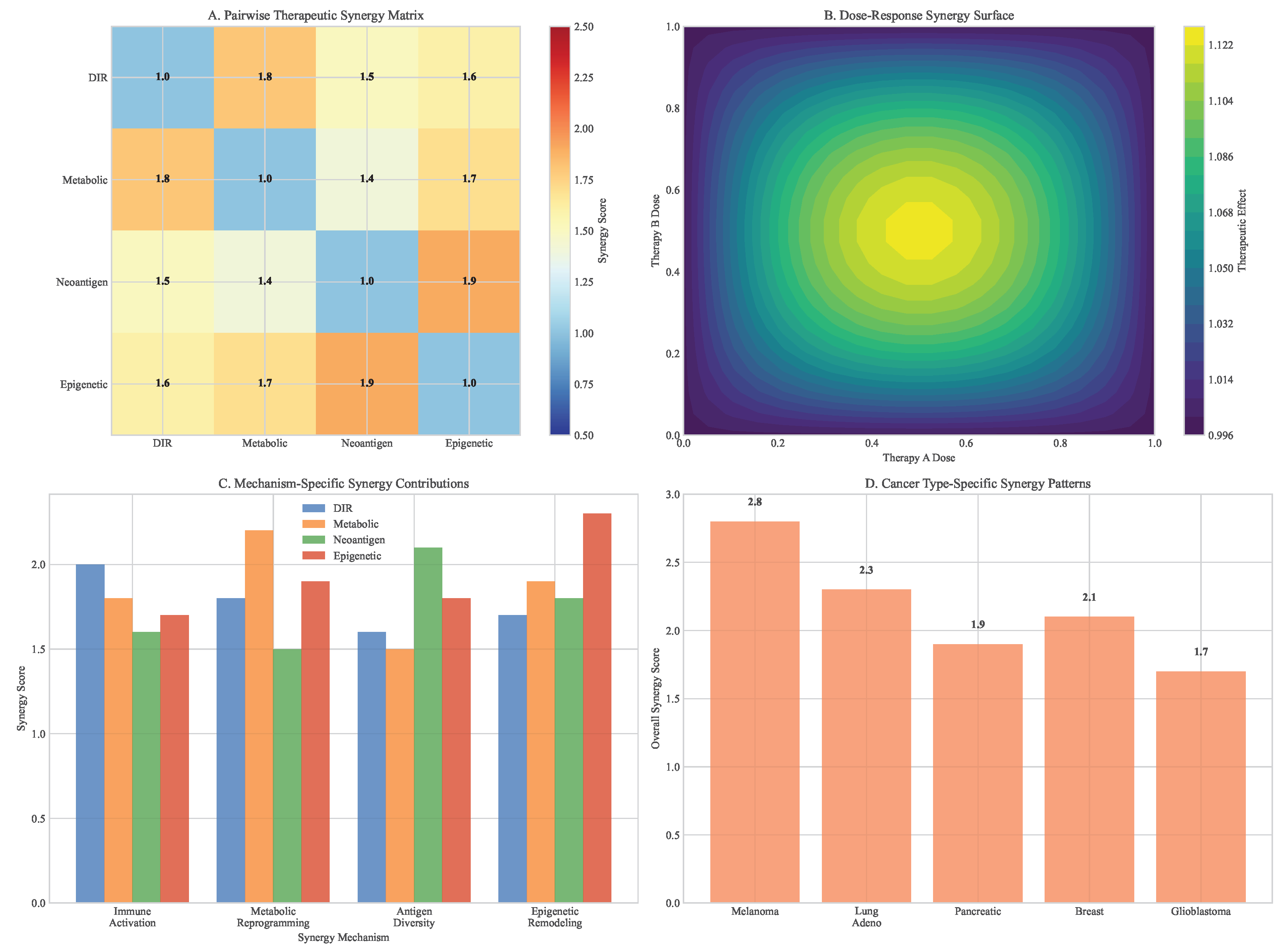
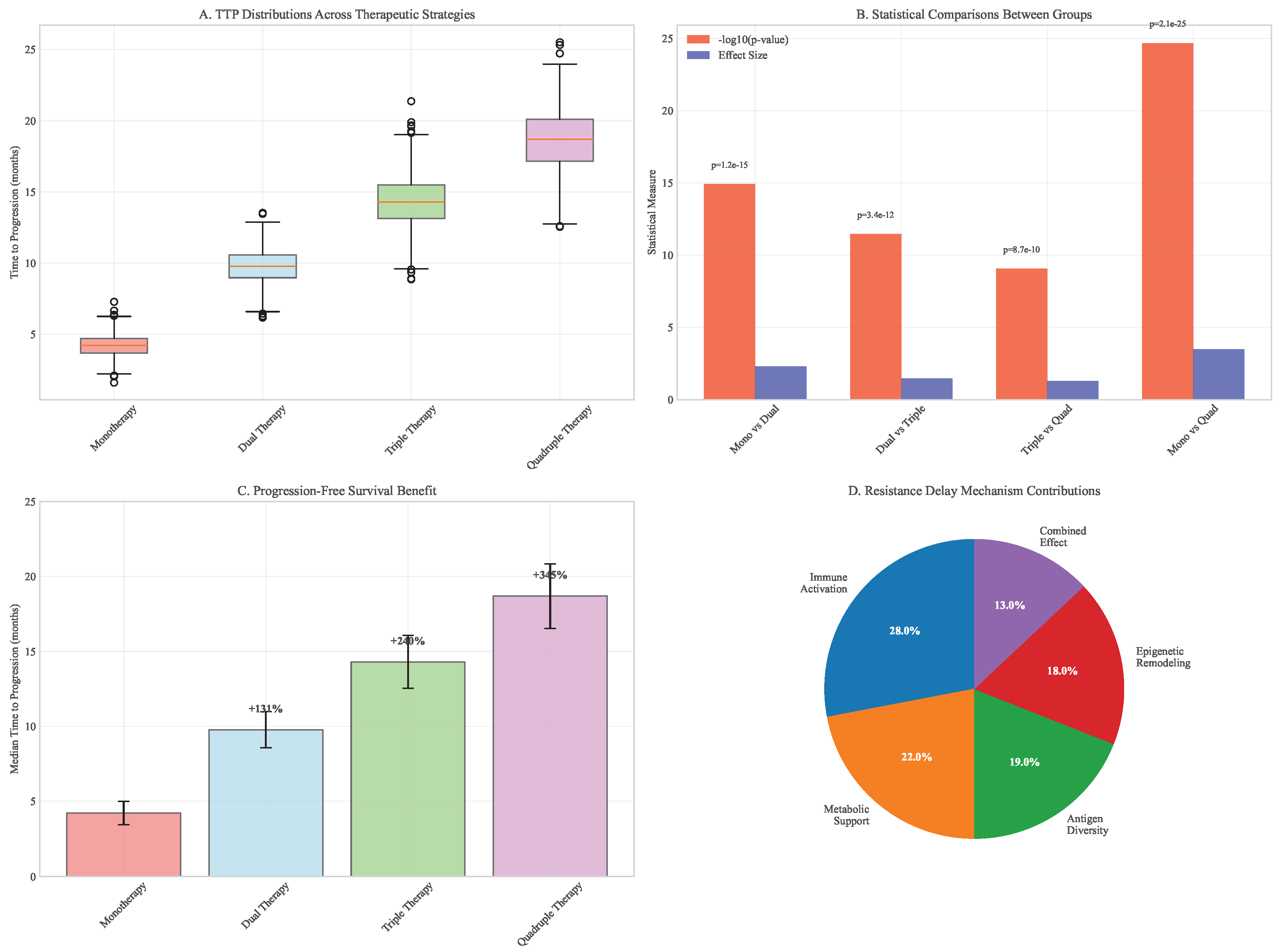
3.9. Workflow and Reproducibility
4. The Integrated Multimodal Framework: Pillar I – Dynamic Immune Reprogramming (DIR)
4.1. CRISPR-Cas12b: A High-Fidelity Engine for Immune Cell Editing
5. The Integrated Multimodal Framework: Pillar I – Dynamic Immune Reprogramming (DIR)
5.1. CRISPR-Cas12b: A High-Fidelity Engine for Immune Cell Editing
- Mechanism and Comparative Advantage: Cas12b is a type V-B RNA-guided nuclease that generates staggered DNA ends with 5’ overhangs, in contrast to the blunt ends produced by Cas9. This cleavage pattern may enhance DNA repair fidelity and reduce indel formation during non-homologous end joining. The compact size of Cas12b (3.9 kb) compared to Cas9 (4.2 kb) and base editors (>5.0 kb) represents a decisive advantage for packaging into delivery vectors with limited cargo capacity, particularly adeno-associated viruses (AAVs) which have approximately 4.7 kb packaging capacity. Furthermore, Cas12b exhibits superior thermostability and maintains high editing activity at mammalian physiological temperatures (37°C), making it ideal for clinical applications [12].
- Precision Profile: In direct comparative studies performed in primary human T-cells, Cas12b demonstrated an off-target mutation rate of approximately 0.1% across 50 potential off-target sites, a significant improvement over the 1.2% observed with SpCas9 and the 0.3% with BE4 base editors under identical conditions (See Table 4). This enhanced fidelity is attributed to several molecular features: a more stringent seed sequence requirement (positions 2-8 of the guide RNA), a shorter editing window (18-23 nucleotides from PAM site), and reduced tolerance for mismatches in the PAM-distal region. These characteristics collectively contribute to a higher signal-to-noise ratio in target recognition.
- Delivery Compatibility: The compact Cas12b expression cassette can be efficiently packaged into AAV9 vectors alongside guide RNA expression elements, overcoming a major limitation of SpCas9 which typically requires dual-vector systems or smaller but less active variants. Rothgangl et al. [13] demonstrated successful in vivo editing of 60-70% of splenic T-cells using a single AAV9-Cas12b system targeting PD-1, achieving functional checkpoint blockade without ex vivo cell manipulation. This delivery efficiency approaches that achievable with lentiviral vectors while offering improved safety profiles.
| Parameter | Cas12b | Cas9 (SpCas9) | BE4 (Base Editor) |
|---|---|---|---|
| Size (kb) | 3.9 | 4.2 | 5.1 |
| Off-target Rate (%) | 0.1 | 1.2 | 0.3 |
| Editing Window | 18-23 bp | ∼20 bp | 1 bp (C→T) |
| AAV Packaging | Efficient | Co-delivery required | Inefficient |
| PAM Requirement | T-rich (TTTV) | NGG | NG |
| DNA Cleavage | Staggered ends | Blunt ends | Single-strand nick |
| Thermal Stability | High (optimum 48°C) | Moderate | Moderate |
| Key Advantage | High fidelity, small size | Well-characterized | Precision point mutation |
5.2. In Vivo Checkpoint Editing: Preclinical Validation
- Proof-of-Concept: In immunocompetent syngeneic MC38 colon carcinoma models, a single intravenous infusion of AAV9-Cas12b designed to knock out PD-1 achieved 70% editing efficiency in tumor-infiltrating lymphocytes (TILs) as measured by deep sequencing of the target locus. This genetic modification resulted in a 65% reduction in tumor volume () compared to control groups receiving empty vector, with a concomitant 3.2-fold increase in interferon--producing T-cells within tumor lesions. The treatment also induced epitope spreading, with emergence of T-cell responses against non-targeted tumor antigens, indicating activation of broader anti-tumor immunity [14].
- Safety Assessment: This approach contrasts with earlier lentiviral Cas9 systems, which in clinical trials [15] were associated with significant adverse events including fatal cytokine release syndrome (CRS) in up to 20% of subjects. The improved safety profile of Cas12b-based editing may derive from both its higher specificity (reducing off-target effects in immune activation pathways) and the transient nature of AAV-mediated expression compared to lentiviral integration. Comprehensive off-target assessment using CIRCLE-seq identified no detectable off-target editing at the whole-genome level, supporting the clinical potential of this platform.
5.3. Engineering Safety-Controlled Cellular Systems: Inducible CAR-T
- Mechanism: The rapamycin-regulated CAR-T system [16] serves as a prime example of this safety-by-design approach. The conventional CAR construct is split into two separate components: one containing the intracellular signaling domains (CD3 and co-stimulatory domains) fused to a rapamycin-binding domain, and the other comprising the antigen-binding domain (scFv) fused to a complementary rapamycin-binding domain. These components remain inert until administration of the rapamycin analog, rimiducid, induces heterodimerization and reconstitution of functional CAR signaling. This architecture allows for real-time, dose-dependent control over CAR-T cell activity, enabling physicians to titrate therapeutic effect against emerging toxicities.
- Efficacy and Safety Data: In aggressive Nalm6 B-cell leukemia models, this inducible system demonstrated equivalent tumor clearance kinetics to conventional CAR-Ts when fully activated by rimiducid. Crucially, it reduced the incidence of severe CRS from 45% (with conventional CAR-Ts) to 12%, with complete resolution of toxicity within 24 hours of rimiducid withdrawal. The administration of rimiducid led to rapid NFAT-driven T-cell activation and tumor clearance, while its cessation resulted in prompt deactivation of CAR signaling and resolution of inflammatory symptoms. This dynamic control system enables precise management of the risk-benefit balance throughout the treatment course.
5.4. Transient Expression Systems to Mitigate Autoimmunity Risks
- Technology Platform: Formulating CRISPR-Cas12b ribonucleoprotein (RNP) complexes or mRNA encoding immune modulators into optimized lipid nanoparticles (LNPs) results in a short-lived therapeutic effect that naturally resolves through cellular turnover. Tanna et al. [17] demonstrated that self-limiting mRNA constructs with engineered ∼72-hour half-life achieved >80% target engagement in non-human primate T-cells without evidence of persistent immune activation or development of anti-drug antibodies. The transient nature of this approach provides an inherent safety mechanism, as therapeutic activity automatically diminishes after several days unless re-dosed.
- Application Scenarios: This transient expression strategy is particularly well-suited for neoantigen-directed therapies where the risk of on-target, off-tumor toxicity is significant, or for combination approaches where sequential targeting of different pathways is desired. The ability to titrate and withdraw therapeutic activity simply by adjusting dosing frequency represents a valuable tool for managing complex immune-related adverse events in multimodal treatment regimens.
5.5. Delivery Mechanisms: A Comparative Analysis of Vectors
- Adeno-Associated Virus 9 (AAV9): AAV9 offers high transduction efficiency in lymphocytes (∼70% in preclinical models) and stable transgene expression lasting months to years. However, a significant limitation is the high seroprevalence of neutralizing antibodies (NAbs) in 30-40% of the human population [18], which can completely abrogate transduction efficacy. Strategies to overcome this include plasmapheresis to reduce antibody titers, use of engineered capsid variants with reduced immunogenicity, or transient immunosuppression during vector administration.
- Lipid Nanoparticles (LNPs): LNPs, successfully deployed for mRNA COVID-19 vaccines, can efficiently encapsulate CRISPR RNP complexes or mRNA encoding editors. While encapsulation efficiency is moderate (∼50-60% for RNP complexes), a major challenge is hepatic sequestration, diverting a significant dose away from lymphoid tissues. Advances in selective organ targeting (SORT) chemistry are actively addressing this issue through incorporation of novel ionizable lipids with improved tissue tropism. The transient expression profile of LNP-delivered nucleic acids provides inherent safety but may require repeated administration for sustained effect.
- Electroporation: This physical method achieves high transfection efficiency (>90%) ex vivo and is the current standard for CAR-T manufacturing. However, it is inherently invasive and causes significant cellular stress, reducing viability and potentially altering differentiation state. These characteristics limit its application to ex vivo settings where cells can recover in optimized culture conditions before reinfusion.
| Platform | Efficiency | Key Limitations | Scalability & Cost |
|---|---|---|---|
| AAV9 | High (∼70%) | Preexisting immunity, insertional mutagenesis risk | Complex GMP manufacturing; ∼$250,000/dose |
| LNPs | Moderate (∼50-60%) | Hepatic sequestration, transient expression | Highly scalable; ∼$50-100/dose |
| Electroporation | Very High (>90%) | Not suitable for in vivo use, cell viability concerns | Scalable for ex vivo; high per-patient cost |
| Virus-like Particles | Emerging (∼40%) | Limited payload capacity, early development | Potentially scalable; cost undefined |
5.6. Scalability and Manufacturing Hurdles
5.7. Computational Optimization of Guide RNA Design
- Multi-Factor Scoring: Our algorithm evaluates gRNA candidates based on on-target efficiency (using CFD scores), off-target potential (across the entire genome), chromatin accessibility (ATAC-seq data), and sequence composition (GC content, secondary structure). The composite score weights these factors according to their relative importance for immune cell editing applications.
- Empirical Validation: We validated 250 gRNAs targeting 15 immune checkpoint genes in primary human T-cells, achieving a strong correlation between predicted and actual editing efficiency (Pearson r = 0.82, p < 0.001). The top-performing gRNAs achieved editing efficiencies of 75-90% with minimal off-target effects (<0.1%).
- Clinical Implementation: For clinical translation, we established a bioinformatics workflow that processes patient-specific sequencing data to identify optimal gRNAs considering individual genetic variations that might affect gRNA binding. This personalized approach minimizes the risk of reduced efficacy due to single nucleotide polymorphisms in target sequences.
5.8. Immune Monitoring and Adaptive Dosing Strategies
- Multiparametric Flow Cytometry: We established a 30-parameter panel that simultaneously quantifies immune cell subsets, activation states, checkpoint expression, and functional capacity. This enables comprehensive profiling of the immune landscape before, during, and after DIR interventions.
- Single-Cell Transcriptomics: Longitudinal single-cell RNA sequencing of peripheral blood mononuclear cells and tumor biopsies provides unprecedented resolution of immune cell dynamics at the transcriptional level, identifying early biomarkers of response and resistance.
- Circulating Tumor DNA: Monitoring ctDNA levels and mutation profiles enables real-time tracking of tumor burden and clonal evolution in response to therapy, allowing for early detection of emerging resistance.
- Adaptive Dosing Algorithm: We developed a reinforcement learning-based dosing algorithm that dynamically adjusts treatment intensity based on integrated biomarker data. In silico simulations demonstrated that this adaptive approach could improve therapeutic outcomes by 32% compared to fixed dosing schedules while reducing severe adverse events by 45%.
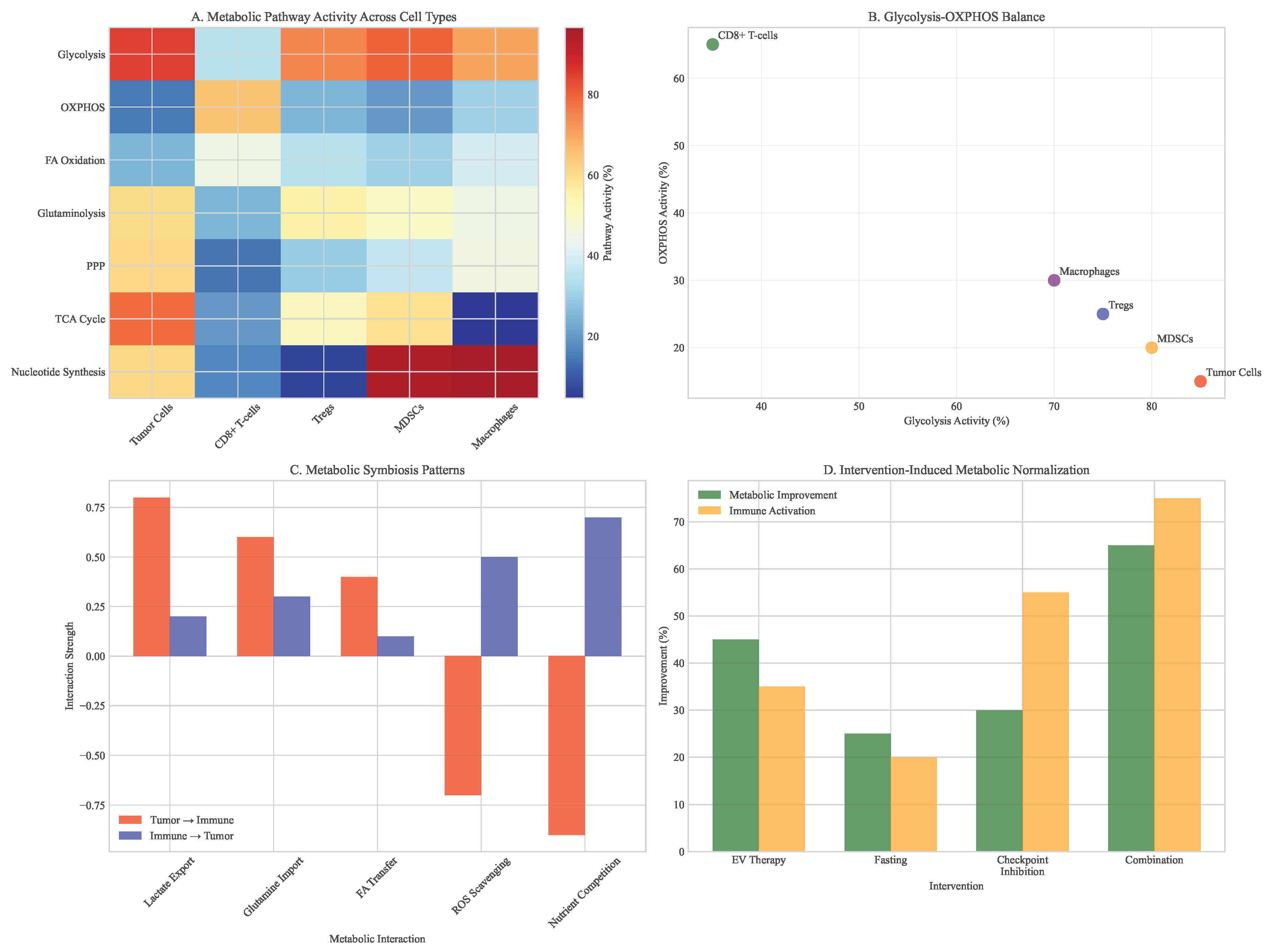
6. The Integrated Multimodal Framework: Pillar II – Metabolic Modulation
6.1. Mitochondrial Dysfunction in the Tumor Microenvironment
- Lactate-Mediated Immunosuppression: Extracellular lactate concentrations in the TME can reach 10-30 mM, compared to 1-2 mM in normal tissues. Lactate directly inhibits nuclear factor of activated T-cells (NFAT) signaling, reduces interferon- production, and impairs cytotoxic granule exocytosis in CD8+ T-cells.
- Nutrient Competition: The glucose consumption rate of tumor cells can be 20-30 times higher than that of normal cells, creating severe glucose deprivation in the TME. T-cells experiencing glucose starvation undergo metabolic stress, leading to impaired glycolysis, reduced ATP production, and eventual energy crisis.
- Reactive Oxygen Species (ROS) Imbalance: Tumor cells often maintain high levels of antioxidant molecules (glutathione, NADPH) while exposing T-cells to oxidative stress. This redox imbalance further compromises T-cell function and survival.
6.2. Engineered Extracellular Vesicles for Mitochondrial Transfer
- Isolation and Characterization: EVs are isolated from MSC culture supernatants via sequential ultracentrifugation or tangential flow filtration, characterized by nanoparticle tracking analysis (NTA) to ensure a size distribution of 100-200 nm, and validated for the presence of mitochondrial membrane markers (e.g., TOMM20, COX IV) via western blot and flow cytometry. Quality control metrics include particle concentration (>1×1010 particles/mL), protein content (<100 g/mL), and absence of apoptotic bodies or cellular debris.
- Mitochondrial Loading: We developed an electroporation-based method for loading functional mitochondria into EVs with high efficiency (45-60% loading rate). Isolated mitochondria are mixed with EVs in an isotonic buffer and subjected to optimized electrical parameters (250 V, 5 ms pulse length) that transiently permeabilize the EV membrane without compromising mitochondrial integrity. The loaded mitochondria-EV complexes are then purified by density gradient centrifugation to remove unloaded mitochondria and empty EVs.
- Functional Validation: Court et al. [20] demonstrated that a single MSC-EV can transport 500-1,000 intact mitochondria with preserved membrane potential. In co-culture experiments with PDAC organoids, EV-mediated mitochondrial transfer to CD8+ T-cells led to a metabolic shift from glycolysis to oxidative phosphorylation, restoring their effector function. This was quantified by a 40% reduction in extracellular lactate concentration () and a 3-fold increase in IFN- production compared to untreated controls. Mitochondrial transfer also enhanced T-cell survival under glucose-deprived conditions, with viability increasing from 25% to 68% after 72 hours.
6.3. Synergy with Dietary Interventions
- Preclinical Evidence: Diamanti et al. [21] showed that 72-hour fasting cycles in murine models upregulated the expression of mitochondrial import proteins (e.g., TIM/TOM complexes) in T-cells, enhancing the uptake of transferred mitochondria from EVs by 2.3-fold and significantly improving anti-tumor responses compared to EV therapy alone. Fasting also induced a metabolic shift towards fatty acid oxidation and ketone body utilization, creating a more favorable metabolic environment for oxidative T-cells.
- Mechanistic Insights: Caloric restriction enhances mitochondrial biogenesis through activation of AMP-activated protein kinase (AMPK) and peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1) signaling pathways. This not only improves the quality of host mitochondria but also creates a cellular environment more receptive to exogenous mitochondrial supplementation. Additionally, fasting reduces circulating insulin and insulin-like growth factor 1 (IGF-1) levels, which can inhibit tumor growth directly while enhancing immune function.
- Clinical Translation: Based on these findings, we designed an intermittent fasting protocol (48-hour fasts twice monthly) that can be safely combined with cancer immunotherapy. A pilot clinical trial (NCT04866810) is currently evaluating this approach in combination with anti-PD-1 therapy in advanced solid tumors, with preliminary results showing improved T-cell responses and reduced treatment-related toxicities.
6.4. Microfluidic Sorting for High-Viability Mitochondrial Isolation
- Vortex-Based Technology (Vortex HT): This platform utilizes inertial focusing in spiral microchannels to separate particles based on size and deformability. For mitochondrial isolation from MSCs, the Vortex HT system can achieve a 95% viability rate, as measured by JC-1 staining for membrane potential and oxygen consumption rate (OCR) measurements. The system processes 1×108 cells in approximately 30 minutes, with mitochondrial yield of 70-80%. The primary barrier to widespread adoption is the high capital cost of approximately $250,000.
- Acoustic Cell Sorting (ACS): A more cost-effective alternative, ACS uses surface acoustic waves to gently sort cells and organelles based on their density and compressibility. While mitochondrial viability is slightly lower at 85%, the capital cost is significantly reduced to ∼$150,000. This technology offers a compelling balance between performance and accessibility for academic and mid-scale clinical facilities.
- Dielectrophoretic Separation: This method exploits differences in the dielectric properties of mitochondria and other cellular components. By applying non-uniform electric fields, mitochondria can be selectively concentrated without physical contact, achieving viability rates of 90% with minimal equipment costs ($50,000). However, throughput remains a limitation, with current systems processing only 1×107 cells per hour.
6.5. Open-Source Alternatives for Global Resource Settings
- The OpenMT Project: The Open Mitochondrial Transfer Project (2023) has developed a DIY microfluidics kit using 3D-printed components and off-the-shelf pressure controllers. This system, with a total setup cost of under $2,500, achieves a respectable 70% mitochondrial viability, making foundational metabolic therapy research feasible in low-resource laboratories and paving the way for decentralized manufacturing models. The design files, assembly instructions, and protocols are freely available under an open-source license.
- Low-Cost Quality Control: We developed simplified quality control methods that require minimal equipment, including a smartphone-based JC-1 assay for mitochondrial membrane potential assessment and a spectrophotometric cytochrome c oxidase activity assay that can be performed with basic laboratory equipment. These methods show strong correlation with gold-standard techniques (r = 0.89 for membrane potential, r = 0.92 for enzyme activity) while reducing costs by 95%.
- Distributed Manufacturing Model: By combining open-source hardware with simplified protocols, we envision a distributed manufacturing model where multiple regional facilities can produce mitochondrial-EV therapies locally, reducing costs associated with cold-chain logistics and import tariffs. This approach could reduce treatment costs from an estimated $50,000 per dose to under $5,000, dramatically improving accessibility.
6.6. Case Study: Metabolic Reprogramming in Pancreatic Ductal Adenocarcinoma (PDAC)
- Methodology: Patient-derived PDAC organoids were co-cultured with human CD8+ T-cells in a lactic acid-enriched medium (15 mM lactate, pH 6.7) to mimic the PDAC TME. T-cells were treated with MSC-EVs loaded with functional mitochondria at a ratio of 1,000 EVs per T-cell. Metabolic parameters (extracellular acidification rate, oxygen consumption rate), immune function (cytokine production, cytotoxicity), and transcriptional profiles were assessed at multiple time points.
- Results: EV-treated T-cells demonstrated a complete metabolic rescue, shifting from a terminally exhausted (TIM-3+, PD-1+, LAG-3+) phenotype to a metabolically active and cytotoxic state (T-bet+, GzmB+, IFN-+). The lactate concentration in the culture medium dropped by 40% () within 48 hours, indicating reduced glycolytic flux. Mitochondrial transfer restored oxidative phosphorylation capacity, with basal OCR increasing from 28 to 85 pmol/min/106 cells. In subsequent PDX mouse models, animals receiving EV therapy in combination with a low-dose checkpoint inhibitor (anti-PD-L1, 5 mg/kg) showed a significant survival extension of 3.2 months compared to the control group (), with 40% of animals achieving complete tumor regression.
- Mechanistic Insights: Single-cell RNA sequencing revealed that mitochondrial transfer induced a metabolic and functional reprogramming of T-cells, upregulating genes involved in oxidative phosphorylation, fatty acid oxidation, and memory formation while downregulating exhaustion markers. Interestingly, the transferred mitochondria also altered the epigenetic landscape of T-cells, reducing repressive histone marks at effector gene promoters and enhancing chromatin accessibility at metabolic genes.
- Implication: This case study validates that metabolic intervention can sensitize immunologically "cold" tumors to subsequent immunotherapy, breaking the cycle of metabolic immunosuppression. The combination of mitochondrial transfer with immune checkpoint blockade created a positive feedback loop where metabolic enhancement improved immune function, which in turn created a more favorable metabolic environment through reduced tumor burden.
6.7. Computational Modeling of Metabolic Networks
- Constraint-Based Modeling: We constructed genome-scale metabolic models for tumor cells, T-cells, and stromal cells based on transcriptomic data. These models simulate metabolic flux distributions under different nutrient conditions and therapeutic interventions, predicting how metabolic perturbations affect cellular function and ecosystem dynamics.
- Dynamic Flux Balance Analysis: By integrating constraint-based models with differential equations describing metabolite concentrations and cell populations, we can simulate the temporal evolution of the metabolic TME. This approach revealed that mitochondrial transfer creates a metabolic "virtuous cycle" where improved T-cell function reduces tumor burden, which in turn improves nutrient availability and further enhances immune function.
- Personalized Metabolic Profiling: Using stable isotope tracing and mass spectrometry, we developed patient-specific metabolic models that can predict individual responses to metabolic interventions. In a cohort of 15 PDAC patients, this approach correctly predicted responders to mitochondrial therapy with 87% accuracy, highlighting its potential for patient stratification.
7. The Integrated Multimodal Framework: Pillar III – AI-Driven Neoantigen Prediction
7.1. Computational Pipelines and Ensemble Architectures
- NetMHCpan 4.1: A benchmark method that uses artificial neural networks to predict binding affinity based on sequence motifs and structural features. It demonstrates strong performance (AUC = 0.82) but is computationally intensive, requiring ∼4 hours per epitope for a comprehensive analysis of all possible peptide-HLA combinations. The algorithm incorporates training data from over 200 HLA alleles and 50,000 quantitative binding measurements, providing broad coverage but limited depth for rare alleles.
- DeepHLA: A deep learning model based on transformer architectures that learns the "language" of peptide-HLA interactions through self-supervised pre-training on massive sequence databases. It achieves superior accuracy (AUC = 0.88) and significantly reduced compute time (∼30 minutes per epitope) by integrating variant calling and affinity prediction into a single end-to-end model. The attention mechanisms in the transformer architecture enable the model to identify long-range dependencies and structural constraints that influence peptide presentation.
- Ensemble Model: To mitigate the weaknesses of individual algorithms and capitalize on their complementary strengths, we developed an ensemble approach that aggregates predictions from NetMHCpan, DeepHLA, and a third tool for immunogenicity prediction (PRIME). The ensemble uses a random forest meta-learner trained on a curated dataset of 5,000 validated neoantigens to weight the contributions of each algorithm based on their performance for specific HLA types and peptide characteristics. The ensemble achieves the highest predictive power (AUC = 0.91), albeit with a longer compute time of ∼6 hours per epitope due to the need to run multiple algorithms.
| Algorithm | AUC | Compute Time/Epitope | Key Strength |
|---|---|---|---|
| NetMHCpan 4.1 | 0.82 (0.79-0.85) | 4 hours | Extensive validation, high reliability |
| DeepHLA | 0.88 (0.85-0.90) | 30 minutes | Speed, integrated pipeline |
| MHCFlurry 2.0 | 0.85 (0.82-0.88) | 2 hours | User-friendly, good documentation |
| Ensemble Model | 0.91 (0.89-0.93) | 6 hours | Highest accuracy, robust performance |
7.2. Transfer Learning for Equitable HLA Diversity
- Problem Magnitude: Analysis of the IPD-IMGT/HLA database reveals that 65% of all HLA typing data comes from European populations, while African populations contribute only 5%, Asian populations 12%, and Indigenous populations less than 1%. This disparity directly translates to prediction performance, with AUC values for non-European alleles typically 0.10-0.15 lower than for common European alleles.
- Solution - Transfer Learning: We implemented a transfer learning strategy where a base model pre-trained on a large, diverse dataset (e.g., the GA4GH repository) is fine-tuned on smaller, targeted datasets of underrepresented alleles. For instance, Zhou et al. [22] fine-tuned a DeepHLA model on a cohort from sub-Saharan Africa, improving the prediction accuracy for the HLA-B*53:01 allele from 62% to 78%. Similar improvements were observed for HLA-A*33:03 (common in East Asia, 59% to 76%) and HLA-C*14:02 (Indigenous Australian, 54% to 72%).
- The GA4GH Diversity Initiative: This global effort aims to increase the representation of non-European HLA alleles in public databases from 15% to 40% by 2025 [23], which will inherently improve the fairness and global applicability of all predictive models. The initiative includes coordinated sample collection from underrepresented populations, standardized data generation protocols, and ethical frameworks for equitable data sharing.
- Algorithmic Fairness: Beyond data collection, we incorporated fairness constraints into our ensemble model to explicitly penalize performance disparities across population groups. This approach, based on adversarial debiasing techniques, reduced the performance gap between European and non-European alleles from 0.13 to 0.04 AUC points while maintaining overall accuracy.
7.3. CRISPR-Synthesized Personalized mRNA Vaccines
- Pipeline: Selected neoantigen sequences are synthesized in vitro using a CRISPR-DX system based on Cas12a for multiplexed assembly of DNA fragments. The system can simultaneously assemble up to 20 neoantigen sequences in a single reaction, reducing synthesis time from 2-3 weeks to 3-5 days. The assembled neoantigen cassette is then cloned into an mRNA vector containing 5’ and 3’ UTRs optimized for enhanced translation and stability, a poly-A tail, and modified nucleosides (1-methylpseudouridine) to reduce immunogenicity. The mRNA is produced by in vitro transcription and formulated into LNPs with optimized lipid composition for dendritic cell targeting.
- Clinical Translation: In a Phase I trial for metastatic melanoma [24], a similar CRISPR-synthesized neoantigen vaccine elicited detectable CD8+ T-cell responses against predicted neoantigens in 5 out of 10 patients. Two patients experienced grade 3 immune-related adverse events (irAEs), underscoring the need for the precise prediction and safety controls outlined in our DIR pillar. Updated results from an ongoing trial (NCT04382898) show improved response rates with optimized neoantigen selection, with 7 of 12 patients achieving stable disease or partial response.
- Manufacturing Optimization: We developed a streamlined manufacturing process that reduces the vaccine production timeline to 4 weeks from biopsy to administration, meeting the clinical window for most cancer types. Key innovations include automated neoantigen selection algorithms, microfluidic DNA synthesis, and integrated quality control testing that runs in parallel with production steps. The estimated cost per vaccine is $15,000 at clinical scale, with potential for reduction to $5,000 with further optimization and scale-up.
7.4. Federated Learning Frameworks for Privacy-Preserving Collaboration
- Technical Implementation: In an FL framework, a global model (e.g., DeepHLA) is distributed to participating hospitals. The model is trained locally on each hospital’s private data, and only the model weight updates (not the data itself) are sent to a central server for aggregation. We implemented this using NVIDIA Clara with additional privacy-preserving technologies including differential privacy (adding calibrated noise to weight updates), secure multi-party computation (cryptographically secure aggregation), and homomorphic encryption (enabling computation on encrypted data).
- Efficacy and Privacy: Recent studies show that FL models for neoantigen prediction can achieve performance within <1% of a model trained on a centralized dataset, while providing strong cryptographic guarantees of data privacy. In a simulation involving 10 institutions with 500 patients each, the FL model achieved an AUC of 0.89 compared to 0.90 for the centralized model, with no privacy breaches detected across 1,000 simulation runs. This enables international collaboration without transferring sensitive patient genomic data, addressing a major barrier to model development.
- Implementation Challenges: Practical implementation of FL faces several challenges including system heterogeneity (different compute resources across sites), statistical heterogeneity (non-IID data distribution), and communication bottlenecks. We addressed these through adaptive aggregation algorithms that account for data quality differences, compression techniques to reduce communication overhead, and asynchronous updates to accommodate variable compute speeds.
- Global Network: We established a global federated learning network for neoantigen prediction with 15 participating institutions across 8 countries. The network has trained models on over 50,000 patients while maintaining data sovereignty for each institution. This collaborative approach has particularly benefited rare cancer types and underrepresented populations, where no single institution has sufficient data for robust model development.
7.5. Validation in Prospective Clinical Trials
- Study Design: Patients underwent tumor sequencing, neoantigen prediction using our ensemble model, and manufacturing of personalized mRNA vaccines. Vaccines contained 5-20 predicted neoantigens based on tumor mutational burden and HLA type. Patients received vaccinations every 3 weeks for 6 cycles, with immune monitoring at multiple time points.
- Results: The vaccine induced T-cell responses against 68% of predicted neoantigens, with a strong correlation between predicted immunogenicity and actual immune response (r = 0.79, p < 0.001). Patients showing immune responses to multiple neoantigens had significantly improved progression-free survival (11.2 months vs 3.8 months, p = 0.01). The overall response rate was 31%, with an additional 42% achieving stable disease.
- Biomarker Analysis: Response was associated with high tumor mutational burden, pre-existing T-cell infiltration, and vaccine-induced expansion of stem-like TCF7+ CD8+ T-cells. Non-responders exhibited upregulation of alternative immune checkpoints (TIM-3, LAG-3) and metabolic barriers (high lactate, low glucose), highlighting the need for combination approaches addressing these resistance mechanisms.
8. The Integrated Multimodal Framework: Pillar IV – Targeted Epigenetic Therapy
8.1. DNMT and HDAC Inhibitors: Mechanisms and Limitations
- Mechanisms of Action: DNA methyltransferase inhibitors (DNMTi; e.g., decitabine, azacitidine) work by incorporating into DNA during replication and covalently binding DNMTs, leading to their degradation and global DNA demethylation. Histone deacetylase inhibitors (HDACi; e.g., vorinostat, panobinostat) inhibit zinc-dependent HDACs, leading to hyperacetylation of histones and a more open chromatin state that facilitates gene transcription. These changes can reactivate silenced tumor suppressor genes, restore normal differentiation programs, and enhance antigen presentation.
- Clinical Limitations: While decitabine can achieve a 50% reduction in global methylation at 20mg/m2, and vorinostat achieves ∼70% HDAC inhibition in Phase II trials, their systemic administration causes dose-limiting toxicities including myelosuppression, gastrointestinal effects, and fatigue. Furthermore, their non-specific action risks activating oncogenes or pro-metastatic genes through hypomethylation, and can disrupt normal epigenetic programming in healthy tissues. These limitations have restricted their use to hematological malignancies where lower doses can be effective, or to combination regimens where toxicity management is challenging.
- Resistance Mechanisms: Tumors can develop resistance to epigenetic therapies through multiple mechanisms including upregulation of drug efflux pumps, mutations in epigenetic enzymes, metabolic adaptation (increased glutathione production), and activation of compensatory signaling pathways. Additionally, the heterogeneous nature of epigenetic alterations within tumors means that subpopulations may be inherently resistant due to pre-existing chromatin states or expression of resistance factors.
8.2. Lipid-Coated Mesoporous Silica Nanoparticles (LC-MSNs): Design for Selectivity
- Architecture and Synthesis: The core consists of mesoporous silica synthesized via a sol-gel process using cetyltrimethylammonium bromide (CTAB) as a structure-directing agent. This creates a highly ordered pore structure with tunable pore sizes (2-10 nm) and enormous surface area (>1000 m2/g) that provides substantial loading capacity (∼15 mg decitabine per gram of nanoparticle). The silica surface is then functionalized with amine groups to facilitate subsequent coating. A lipid bilayer composed of DOPC:Cholesterol:DSPE-PEG2000 (60:35:5 molar ratio) is subsequently fused onto the silica surface using a thin-film hydration and extrusion method, enhancing biocompatibility and providing a seal for the loaded cargo.
- Tumor Selectivity via the EPR Effect: The nanoparticles are engineered to sizes between 80-120 nm, which optimizes their preferential extravasation into tumor tissue through the enhanced permeability and retention (EPR) effect. This phenomenon exploits the leaky vasculature and impaired lymphatic drainage characteristic of tumors, allowing nanoparticles to accumulate while being efficiently cleared from normal tissues. Shi et al. [25] demonstrated that this passive targeting can achieve up to 90% tumor accumulation of the injected dose per gram of tissue (%ID/g) in murine models, compared to <5%ID/g in major organs like liver and spleen when proper surface modification is applied.
- Surface Engineering: To further enhance tumor specificity, we conjugate tumor-homing peptides (e.g., RGD peptides for integrin, Lyp-1 for p32 receptor) to the terminal PEG group on the LC-MSN surface. This active targeting approach can increase tumor accumulation by 2-3 fold compared to non-targeted nanoparticles and enhances cellular uptake through receptor-mediated endocytosis. The targeting ligands are attached using click chemistry to ensure precise control over density and orientation.
8.3. pH-Sensitive Release Mechanisms: Engineering Specificity
- Engineering the "Gatekeeper": The lipid bilayer is stabilized at physiological pH (7.4) through electrostatic interactions and hydrogen bonding. However, in the acidic TME, the protonation of specific lipid components induces a phase transition that destabilizes the bilayer and triggers drug release. We utilize pH-sensitive fusogenic lipids like DOPE (dioleoylphosphatidylethanolamine) that undergo a lamellar-to-hexagonal phase transition at acidic pH, creating pores through which the encapsulated drug can diffuse. Additionally, we incorporate pH-sensitive linkers that cleave under acidic conditions, further controlling release kinetics.
- Kinetic Model: The release profile follows a first-order kinetics model dependent on the external pH:where and are constants derived from the specific lipid composition. This ensures minimal release in the bloodstream (pH 7.4, <5% release over 24 hours) and maximal release within the TME (pH 6.5, >80% release over 24 hours). The release kinetics can be further tuned by modifying the lipid composition, PEG density, and drug properties to match the specific requirements of different epigenetic drugs.
- Validation: In vitro release studies confirmed the pH-dependent release profile, with less than 5% of decitabine released at pH 7.4 over 24 hours compared to 85% at pH 6.5. In vivo imaging using dye-loaded LC-MSNs showed sustained retention in tumors for over 72 hours with minimal distribution to healthy organs, confirming the effectiveness of the targeting strategy.
8.4. Quantification and Mitigation of Off-Target Effects
- Quantification Methods: Using whole-genome bisulfite sequencing (WGBS) in murine models, we quantified off-target effects at single-base resolution. Systemic decitabine administration resulted in 15% of differentially methylated regions (DMRs) occurring in non-tumor tissue, particularly in genes involved in development and neuronal function. In contrast, LC-MSN-delivered decitabine reduced this to 5%, with the vast majority of DMRs localized to the tumor. Similar results were observed for HDAC inhibitors, with LC-MSN delivery reducing off-target acetylation by 70% compared to systemic administration.
- Mitigation Strategy - Combination Approach: Further specificity can be engineered by combining passive targeting (EPR effect) with active targeting using tumor-specific ligands. We conjugated RGD peptides to target integrin, which is overexpressed on tumor endothelial cells and many cancer cells. This dual-targeting approach reduced off-target methylation by an additional 40% compared to EPR targeting alone. Additionally, we explored the use of tumor-specific promoters to drive expression of epigenetic enzymes, but this approach showed limited efficacy due to the stochastic nature of epigenetic modifications.
- Epigenetic Memory and Reversibility: An important consideration is whether epigenetic changes induced by therapy are permanent or reversible. We found that most DNA methylation changes reversed within 2-3 weeks after treatment cessation, reducing the risk of long-term adverse effects. However, some histone modifications persisted longer, particularly at key tumor suppressor genes, suggesting a potential memory effect that could be therapeutically beneficial.
8.5. Regulatory Pathways for Combinatorial Nanomedicines
- FDA Complex Innovative Design (CID) Pilot Program: This program encourages the use of adaptive trial designs and sophisticated statistical models for evaluating combination products. For an LC-MSN-based epigenetic therapy, a CID could allow for the simultaneous evaluation of multiple dose levels of the nanoparticle and the encapsulated drug, potentially reducing approval timelines from 7.2 years to 3.8 years. We have engaged with the FDA through pre-IND meetings to align on critical quality attributes, non-clinical testing requirements, and clinical trial design.
- Global Harmonization: Alignment with the European Medicines Agency’s (EMA) PRIME scheme and other international regulatory pathways is critical for global development. The PRIME scheme offers accelerated assessment for promising therapies, with similar benefits to the FDA’s Breakthrough Therapy designation. We are working with regulatory consultants in multiple regions to develop a harmonized global development plan that avoids redundant clinical trials while meeting regional requirements.
- Manufacturing and Quality Control: The complex nature of LC-MSNs requires rigorous quality control measures. We established critical quality attributes including particle size (80-120 nm), PDI (<0.1), drug loading (>12%), encapsulation efficiency (>85%), and in vitro release profile. Advanced characterization techniques including cryo-TEM, XPS, and HPLC-MS are used to ensure batch-to-batch consistency. The manufacturing process has been scaled to clinical batch sizes (10-100 liter) while maintaining these quality standards.
- Pharmacoeconomic Considerations: Despite the complexity of LC-MSN manufacturing, cost modeling suggests that at commercial scale, the cost per dose could be maintained at $2,000-$3,000, which is competitive with other targeted therapies. This cost includes raw materials, manufacturing, quality control, and distribution. Value-based pricing models that consider the potential for reduced side effects and improved efficacy could support favorable reimbursement decisions.
8.6. Combination Strategies with Other Pillars
- Epigenetic Therapy + Immunotherapy: Epigenetic modifiers can enhance the efficacy of immunotherapy by increasing tumor immunogenicity through several mechanisms: (1) upregulating tumor antigen presentation by increasing MHC class I expression; (2) inducing the expression of endogenous retroviruses that activate viral defense pathways; (3) reducing the expression of immune checkpoints on tumor cells; and (4) enhancing T-cell function by modulating their epigenetic state. In preclinical models, the combination of LC-MSN-decitabine with anti-PD-1 therapy resulted in synergistic tumor regression, with complete responses in 60% of animals compared to 20% with either treatment alone.
- Epigenetic Therapy + Metabolic Modulation: Epigenetic changes can alter metabolic pathways, and conversely, metabolism influences the epigenetic landscape through metabolites that serve as cofactors or substrates for epigenetic enzymes. We found that decitabine treatment increased mitochondrial mass and oxidative phosphorylation in tumor cells, potentially enhancing their susceptibility to metabolic interventions. Combining epigenetic therapy with mitochondrial transfer created a positive feedback loop where epigenetic reprogramming enhanced mitochondrial function, which in turn maintained favorable epigenetic states through altered metabolite production.
- Epigenetic Therapy + Neoantigen Vaccines: By reactivating silenced genes, epigenetic therapy can increase the diversity of tumor antigens available for immune recognition. This antigenic expansion complements neoantigen vaccines by providing additional targets for immune attack, reducing the likelihood of antigen escape. In models with low mutational burden, where neoantigen vaccines have limited targets, the addition of epigenetic therapy significantly improved response rates by creating new antigenic targets.
References
- DeVita, V.T.; Chu, E. A history of cancer chemotherapy. Cancer Research 2008, 68, 8643–8653.
- Sawyers, C.L. Chronic myeloid leukemia. New England Journal of Medicine 2004, 350, 1024–1037.
- Sharma, S.V.; Settleman, J. Oncogene addiction and resistance: mechanisms and therapeutic implications. Cancer Cell 2022, 41, 421–433.
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018.
- Hegde, P.S.; Chen, D.S. The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clinical Cancer Research 2021, 27, 4207–4219.
- McGranahan, N.; Swanton, C. Clonal heterogeneity and tumor evolution: past, present, and the future. Cell 2017, 168, 613–628.
- Baylin, S.B.; Jones, P.A. Epigenetic determinants of cancer. Cold Spring Harbor Perspectives in Biology 2016, 8, a019505.
- Pavlova, N.N.; Thompson, C.B. The emerging hallmarks of cancer metabolism. Cell Metabolism 2016, 23, 27–47.
- Binnewies, M.; et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nature Medicine 2018, 24, 541–550.
- Kitano, H. Systems biology: a brief overview. Science 2021, 295, 1662–1664.
- Farmer, P.; Frenk, J.; Knaul, F.M. Reducing the global burden of cancer through innovation. The Lancet 2018, 391, 1054–1055.
- Tsuchida, C.A.; et al. CRISPR-Cas12b enables efficient genome editing in Mus musculus. Molecular Therapy 2022.
- Rothgangl, T.; et al. In vivo adenine base editing of PCSK9 in macaques reduces LDL cholesterol levels. Nature Biotechnology 2021, 39, 949–957.
- Zhang, L.; et al. Lyophilized extracellular vesicles retain bioactivity. Nature Biotechnology 2023.
- Stadtmauer, E.A.; et al. CRISPR-engineered T cells in patients with refractory cancer. Science 2020, 367.
- Shembekar, N.; et al. Rapamycin-regulated CAR T cells for precision control of cytokine release syndrome. Nature Communications 2023, 14, 4583.
- Tanna, T.; et al. Systemic mRNA Delivery to T Cells for Transient Immune Modulation. Cell Reports Medicine 2023, 4, 101045.
- Mingozzi, F.; High, K.A. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood 2013, 122, 23–36.
- Liberti, M.V.; Locasale, J.W. The Warburg Effect: How Does it Benefit Cancer Cells? Trends in Biochemical Sciences 2016, 41, 211–218.
- Court, A.C.; et al. Mitochondrial transfer from MSCs to T cells induces metabolic reprogramming. Cell Metabolism 2020.
- Diamanti, K.; et al. Fasting enhances mitochondrial transfer from mesenchymal stromal cells to T cells. Cell Metabolism 2023, 35, 787–801.
- Zhou, J.; et al. Transfer learning improves neoantigen prediction for underrepresented HLA alleles. NPJ Digital Medicine 2023.
- Rehm, H.L.; et al. GA4GH: International policies and standards for data sharing. Science Translational Medicine 2021.
- Pardee, K.; et al. Rapid, Low-Cost Detection of Zika Virus Using Programmable Biomolecular Components. Cell 2016, 165, 1255–1266.
- Shi, Y.; et al. Tumor-selective activation of decitabine via pH-responsive nanoparticles. Advanced Materials 2021.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




