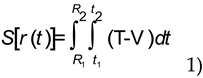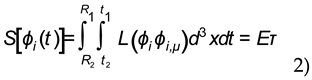1.0. Introduction
Although matrix mechanics and wave mechanics are believed to be equivalent formulations of quantum mechanics, they are not equal participants. They are based on widely separated initial assumptions, distinct experiments, and different mathematical models. Contributions to the foundations by wave mechanical models predominate, while matrix mechanics has not advanced significantly in its contributions to the foundations beyond initial assessments 100 years ago. Schrödinger gave a possible reason for the lack of progress [
1].
“Today there are not a few physicists who regard the task of physical theory as being merely a mathematical description (as economical as possible) of the empirical connection between observable quantities; that is, a description which reproduces the connection, as far as possible, without the intervention of unobservable elements. On this view, mathematical equivalence has almost the same meaning as physical equivalence. In the present case there might perhaps appear to be a certain superiority in the matrix representation because, through its stifling of intuition, it does not tempt us to form space-time pictures of atomic processes which must perhaps remain uncontrollable.” p. 58
During the time of Schrödinger and Heisenberg mathematics and experiment prevailed and no attempts were made to understand the underlying physical meaning. It was, in effect, a complete renunciation of the ability to “picture” the microscopic world. Rather than accept the idea that atomic processes are incomprehensible we will make use of “intervening unobservables” to reformulate matrix and wave mechanics, “unify” the mathematics, and enable the visualization of quantum systems.
2.0. Matrix Mechanics Reformulated
Classical mechanics is the study of the motion of objects and their causes; in other words, the equations of motion. Ideally the objective of quantum mechanics should be to find out the subatomic equations of motion. This is clearly what wave mechanics does when it plots the probability densities for the electron orbitals of a hydrogen atom. However, matrix mechanics makes no attempt at all for it describes the properties not of an atom but of the simultaneous emission of radiation by many hydrogen atoms. Measurements are performed in order to obtain matrices that describe black body radiation in terms of the frequency and transition probability of the spectral lines, which are the observables. The set of all possible observables are understood to be the set of eigenvalues of the matrix. Thus the purpose of matrices is to give a complete set of the observables of hydrogen gas for an arbitrary electron transition. Instead we will use a single hydrogen atom and quantum system to obtain a complete set of observables.
There are difficulties involved in deriving the laws of mechanics for the single hydrogen atom as noted by Nobel laureate Richard Feynman [
2].
But suppose we look at the whole hydrogen atom as a “particle.” If we didn’t know that the hydrogen atom was made out of a proton and an electron, we might have started out and said: “Oh, I know what the base states are—they correspond to a particular momentum of the hydrogen atom.” No, because the hydrogen atom has internal parts. It may, therefore, have various states of different internal energy, and describing the real nature requires more detail.
The internal energy of a hydrogen atom is described quantum mechanically by the Hamiltonian matrix H
ij because it has an infinite number of matrix elements and there are an infinite number of possible energy levels that an electron can transition between. However, it is impossible to determine the transition energies of all states. As energy states go higher, the energy differences between successive states become smaller, and the spectral lines converge and become continuous. Heisenberg chose to disregard spectral lines that cannot be observed stating that quantum mechanics should be “founded exclusively upon relationships between quantities which in principle are observable” [
3]. We will include all electron transitions in a theory of the hydrogen atom, irrespective of whether the corresponding spectral lines are observable.
The diagonalized matrix describing the hydrogen spectrum due to black body radiation is not the result of a single hydrogen atom, rather it is due to many atoms acting in unison. It is the result of many events, where each one occurs at an independent point in space and time. It is nevertheless possible for a solitary, electrically neutral hydrogen atom, isolated in space, to emit the entire hydrogen spectrum if we apply heat incrementally and describe the energy states sequentially as a series of Hamiltonian matrices, where each matrix is a snapshot of an atom’s energy state at a particular point in time. Initially the states are of infinitesimal energy and undetectable, but they eventually register lines in the infrared, the visible spectrum, and beyond to the ultraviolet spectrum. The complete spectrum as viewed by a single atom appears in the manner of a motion picture with each frame consisting of a Hamiltonian matrix. We observe the spectrum as a continuously increasing background thermal energy that surrounds the hydrogen atom and is periodically interrupted by spectral lines of increasing energy in the Lyman, Balmer. Paschen, and Brackett series. Thus the discrete energy eigenvalues of the atom appear sequentially as diagonalized elements of the Hamiltonian matrix. In other words, the complete spectral series of hydrogen is generated by integrating the emissions of a single atom over a sufficiently long time period.
3.0. Wave Mechanics
We proceed next to examine the methods used to derive another formulation of the equations of motion based on the Schrödinger wave equation, iℏ d/dt|Ψ(t)⟩=Ĥ|Ψ(t)⟩. The equation was arrived at by integrating over the entire coordinate space to obtain a wave function [
1].
“We now seek a function ψ such that for any arbitrary variation of it the integral . . . . taken over the whole coordinate space is stationary, ψ being everywhere real, single-valued, finite, and continuously differentiable up to the second order. The quantum conditions are replaced by this variation problem.” p. 2
Schrödinger seeks to derive a theory based on an action functional and variational principle, a method which is central to all of physics. A functional ψ(r,t) is introduced (the energy) whose variation over the coordinate space is stationary.
“We have always postulated up till now that the potential energy V is a pure function of the coordinates and does not depend explicitly on the time. There arises, however, an urgent need for the extension of the theory for non-conservative systems, because it is only in that way that we can study the behavior of the system under the influence of prescribed external forces, e.g. a light wave, or a strange atom flying past.” p. 103
Wave mechanics is derived with respect to a conservative electromagnetic system; that is, a proton-electron pair with a fixed potential. The probability density function of the orbiting electron for a fixed potential is defined by the square of the absolute value of the wave function at each point in coordinate space. Extending the theory to non-conservative systems would allow external forces to be introduced thereby enabling greater flexibility in the number and variety of applications. He concludes the first draft of his paper by repeating this same concern.
“In the pair of equations [given in the text], we have before us only the substitute . . . for a real wave equation . . . which, however, I have not succeeded in forming for the non-conservative case.” p. 123
He reaffirms here the need for a quantum theory of non-conservative systems which would include the “external forces” on a hydrogen atom and enable it to be merged with classical theory. His theory as it stands is incomplete. Years later Einstein realized as much [
4].
“The light quantum has a definite localization and a definite color. Naturally one cannot do justice to this by means of a wave function. Thus I incline to the opinion that the wave function does not (completely) describe what is real, but only a to us empirically accessible maximal knowledge regarding that which really exists. . . . This is what I mean when I advance the view that quantum mechanics gives an incomplete description of the real state of affairs.”
To accommodate Einstein’s objections we postulate that matrix mechanics provides the required “missing piece” of the puzzle that is needed to define a non-conservative theory that is complete and also real. It is mathematically equivalent to wave mechanics because it gives the same eigenvalues for the energy, but it also includes physical variables such as frequency that are lacking in the wave mechanical model.
4.0. Energy Conservation
It is well known that energy is not always conserved in quantum mechanics [
5]. “Wave functions collapse when they are observed. This collapse process is unpredictable, and doesn’t obey the Schrödinger equation. As a result, the average energy is not conserved in the process of quantum measurement.” In fact the original proof of energy conservation given by Heisenberg in his study of the anharmonic oscillator is not rigorous. He demonstrated energy conservation for a conservative quantum system; however, as shown in the previous section the atom is a non-conservative system since it absorbs thermal energy from the environment while emitting black body radiation. Neither he nor Schrödinger proved that energy is conserved for open, non-conservative quantum systems.
In order to prove that energy is conserved in non-conservative quantum systems the energy absorbed must be identical to the energy emitted. In fact due to energy conservation energy input equals energy output for all isolated material systems, whether the system is a hydrogen atom, a machine, a hydroelectric power plant, or a galaxy. The quantum system conserves energy for changes in electromagnetic potential in the same way that a gravitational system conserves energy for changes in gravitational potential. Therefore quantum systems must absorb an amount of energy as described matrix mechanically that is identical to the amount of energy emitted by means of the wave function in order to satisfy energy conservation.
We shall illustrate energy conservation in a non-relativistic quantum system to show that matrix and wave mechanics work together to describe the emission and absorption of radiation. We have described energy absorption at single points in time with matrices as incremental increases. It can also be described by one rotation of the wave function. Emission follows by means of a second rotation of the wave function demonstrating that absorption and emission are part of the same quantum process. We interpret the first rotation of the wave function not as a rotation in abstract space, but as the change in phase of electromagnetic fields from 0 to 2π in real space with phase information given by the off-diagonal matrix elements. Thus one “rotation” of the wave function is interpreted as one full cycle of an electromagnetic wave and an increase in the electron’s energy from the ground state to an excited state. The second rotation occurs as the electron returns to the ground state and is interpreted as a localization of electromagnetic field energy and emission of a photon. The dual wave-particle nature of the photon is thereby realized in a continuous transformation of fields. When a continuous function is integrated between two fixed endpoints, the orbitals, and two specified times, the period, the result is a relativistic invariant, the time integral of a Lagrangian ∫Ldt, or action function.
5.0. The Lagrangian in Quantum Mechanics
Quantum mechanics is formulated non-relativistically; that is, in an inertial system. To be formulated relativistically the paths of excitation and decay of electrons need to be described between two specific endpoints, the atomic orbitals, and two specific times, the period τ, which are the physical conditions that define an action function, Hamilton’s principle of least action [
6].
Thus the physical characteristics of a quantum system naturally define the correct equations of motion needed to describe it. The principle of least action simply means that of all the paths that an electron can take between two atomic orbitals, the ones actually taken are found by computing the action for each of the trajectories, and selecting the ones that have the least action. The paths selected will be the ones that have an action equal to the reduced Planck’s constant ћ.
The action is the time integral of the Lagrangian ∫Ldt, where L=T-V, T is the kinetic energy, and V is the potential energy. We compute the action of an electron transitioning from the ground state to an excited state by using generalized coordinates, three to describe its position on the electron shells R1 = (x1,y1,z1) and R2 = (x2,y2,z2), and three to describe its trajectory.
The action, S[r(t)], is a functional that describes the absorption process in four dimensions. It has as its argument an infinite number of functions, the possible electron paths r(t). We know that it is relativistically correct because gravitational fields influence the action identically to the way that they influence clocks; that is, by causing time dilation [
7].
Emission initiates from the excited state R2 = (x2,y2,z2) at time t2 and it finalizes at the ground state R1 = (x1,y1,z1) at time t1. Each of the electron shells R2 and R1 determines a locus of points where the fields vanish. The Lagrangian density over the region of space-time between the excited and ground states is stationary for all small variations of the coordinates inside the region. Changes in action during emission are evaluated by integrating the Lagrangian density four-dimensionally thereby yielding a relativistic formulation.
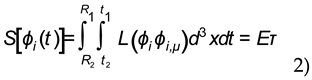
Solving 2) we obtain an expression for the localized fields of a photon Eτ=ћ, which is equivalent to the more familiar E=hν. The emission of a photon is described by the action integral of a Lagrangian density ∫£(
ϕi ,ϕi ,μ)dt in the region of space-time between the excited and ground states given by the fields φ
i and its first derivatives
ϕi,μ. This allows for a complete accounting of the energy interactions that occur during an electron transition within the volume ∫d
3x between the electron orbitals where
ϕi is the current density described
radially and
ϕi,μ is the electromagnetic field strength described
transversely that results in the creation of a photon. The photon is described therefore as a four-dimensional localization of fields contained within the volume ∫d
3x and the time interval t
2-t
1 = τ. The discrete and continuous properties of the emission process are described by the Lagrangian density over the time interval of a period which results in a photon being created. A more complete derivation following methods due to Dirac is given elsewhere [
8].
6.0. Conclusion
By his own admission Schrödinger did not complete a derivation of wave mechanics by formulating it in a non-conservative system. As shown in 4.0 it is accomplished by simply appending the output of matrix mechanics to the input of wave mechanics. The resulting “unified” theory is non-relativistic because the equations of motion are formulated in an inertial system. To be relativistic it must be formulated in a non-inertial frame so that the equations of motion vary continuously with respect to velocity and acceleration as derived in the previous section. Dirac recognized already in 1930 the significance and relative importance of a relativistic theory [
9]. “Quantum mechanics was built up on a foundation of analogy with the Hamiltonian theory of classical mechanics. Now there is an alternative formulation for classical dynamics provided by the Lagrangian. The two formulations are, of course closely related, but there are reasons for believing that the Lagrangian one is the more fundamental. In the first place the Lagrangian method allows one to collect together all the equations and express them as the stationary property of a certain action function. (This action function is just the time-integral of the Lagrangian.) There is no corresponding action principle in terms of the coordinates and momenta of the Hamiltonian theory. Secondly the Lagrangian method can easily be expressed relativistically, on account of the action function being a relativistic invariant; while the Hamiltonian method is essentially non-relativistic in form, since it marks out a particular time variable as the canonical conjugate of the Hamiltonian function. For these reasons it would seem desirable to take up the question of what corresponds in the quantum theory to the Lagrangian method of the classical theory.”
The Lagrangian method proves superior for describing quantum theory because it treats the time evolution of a quantum system as a continuous process. Classical heat is absorbed by a hydrogen atom creating a diagonalized Hamiltonian matrix with a specific frequency output and excited state (eqn 1). The electron spontaneously decays from the excited state to the ground state collapsing the wave function and emitting a photon (eqn 2). Of all the paths available to the photon it will select a trajectory that minimizes the action. Previous investigations of quantum systems are inaccurate because they describe quantization non-relativistically by means of observables at single points in time. However, time is not an observable and it must vary continuously to satisfy relativity theory.
References
- Schrödinger, E. Collected Papers on Wave Mechanics, 2nd edn; Blackie: London, 1928; Available online: https://mwolf.pracownicy.uksw.edu.pl/MK/Schrodinger_Collected_Papers_on_Wave_Mechanics.pdf.
- Feynman; Leighton; Sands. The Feynman Lectures on Physics, Vol III, Chap 8-3; “The Hamiltonian Matrix”, 1963. [Google Scholar]
- W. Heisenberg Z Phys 33 (1) (1925), in B.L. van der Waerden (ed.), Sources of Quantum Mechanics (Amsterdam, 1967). doi: http://www.mat.unimi.it/users/galgani/arch/heis25ajp.pdf.
- Einstein, A. Letter to Paul Epstein 11/10/1945 Sixty-Two Years of Uncertainty Edited by A. I. Miller; Plenum Press: New York, 1990; p. 103. [Google Scholar]
- Carroll, S.; Lodman, J. Energy non-conservation in quantum mechanics; 2021. [Google Scholar] [CrossRef]
- Lanczos, C. The Variational Principles of Mechanics, 4th edn; Dover, 1970. [Google Scholar]
- Bothwell, T.; et al. Resolving the gravitational red shift within a millimeter atomic sample. Nature 602 2022, arXiv:2109.12238Issue 7897, 420. [Google Scholar]
- Oldani, R. (2024) “Galactic symmetry” doi: preprints.org/manuscript/202009.0215/v8, p. 11.
- Dirac, P.A.M. The Lagrangian in quantum mechanics. Phys Zeit Sow 3(1933), 1. Available online: https://www.informationphilosopher.com/solutions/scientists/dirac/Lagrangian_1933.pdf.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).