Submitted:
02 October 2025
Posted:
03 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Patient Characteristics
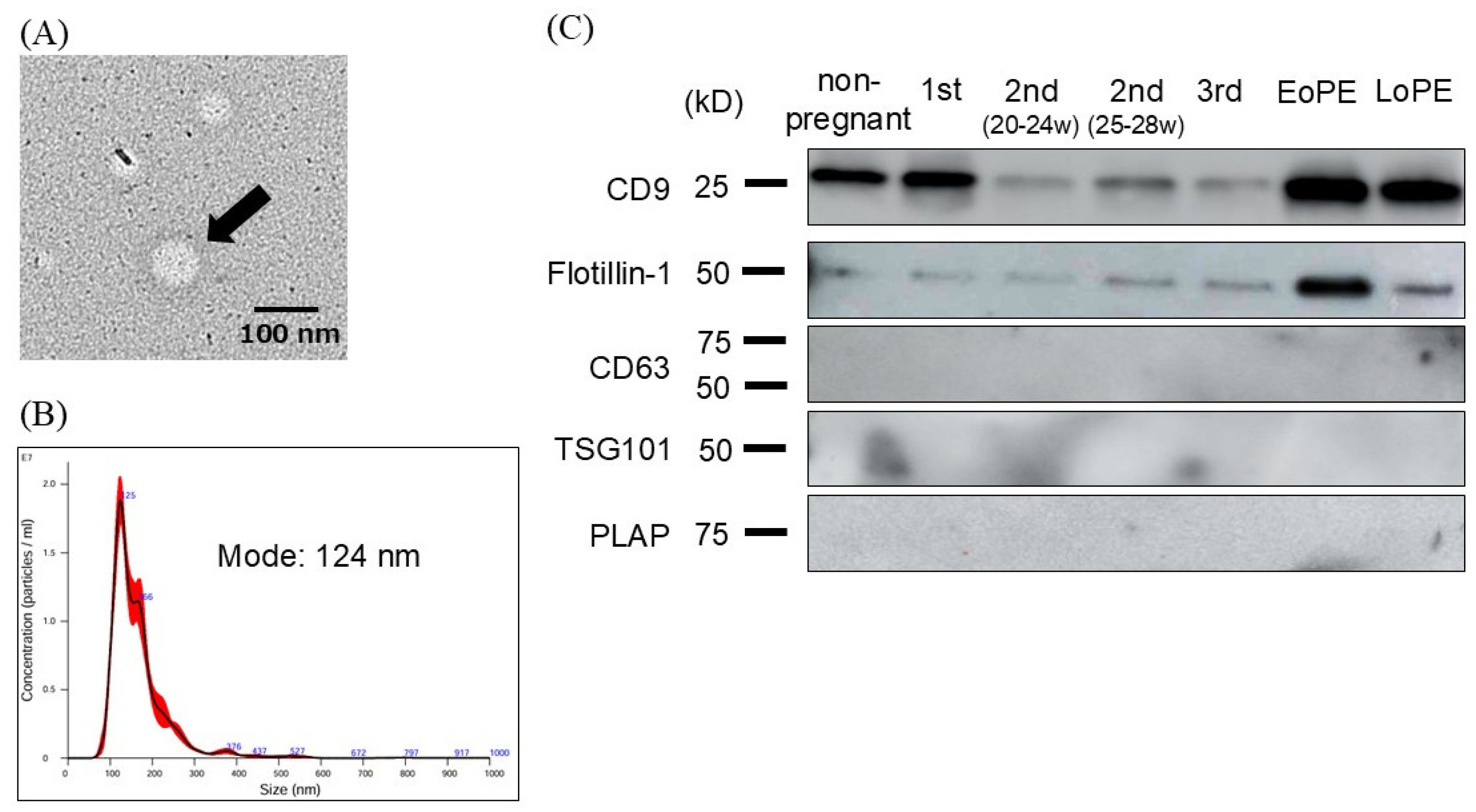
2.2. Characterization and Evaluation of Circulating Small EVs
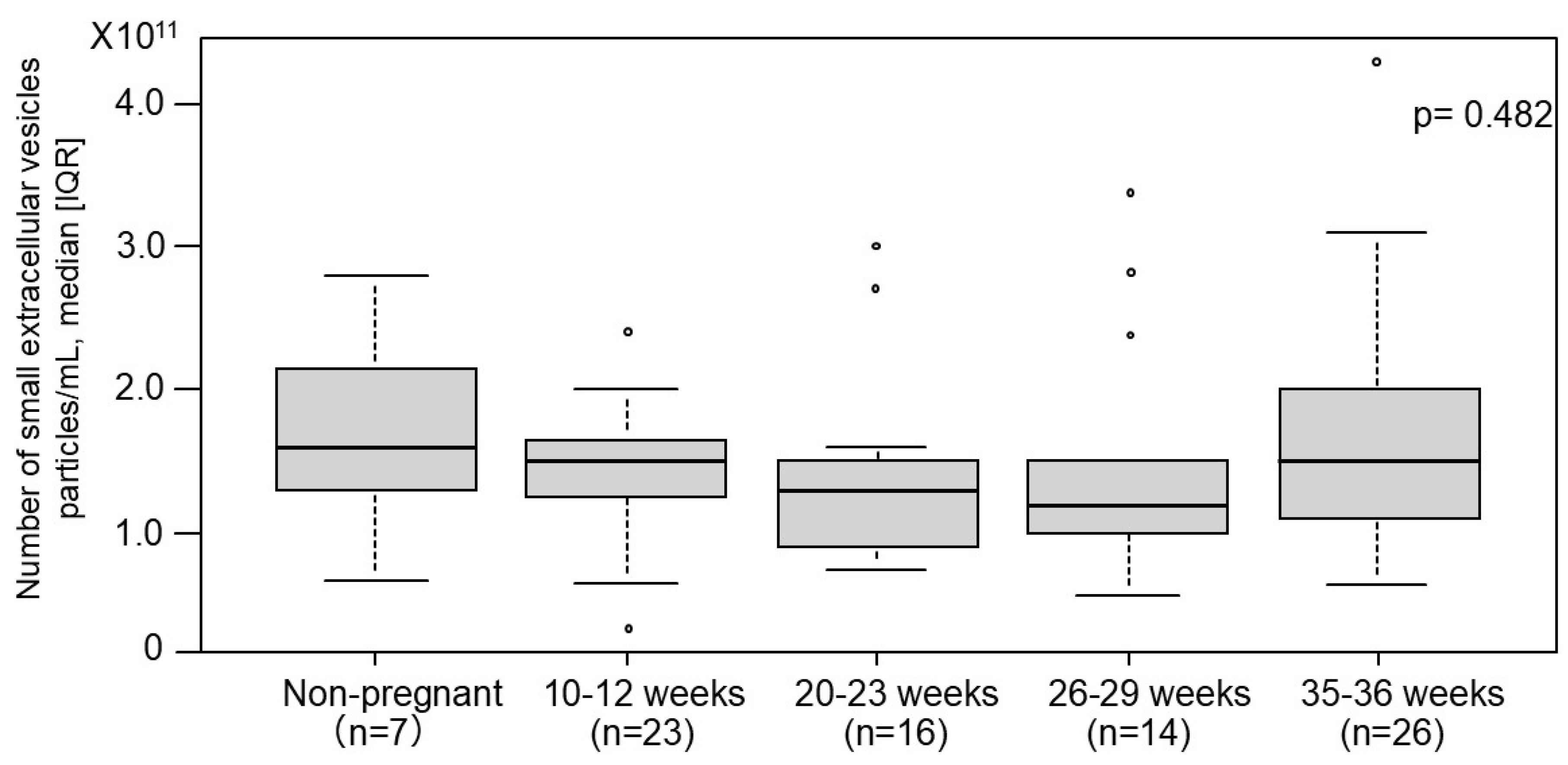
2.3. The Number of Small EVs in Non-Pregnant Women and Normotensive Controls
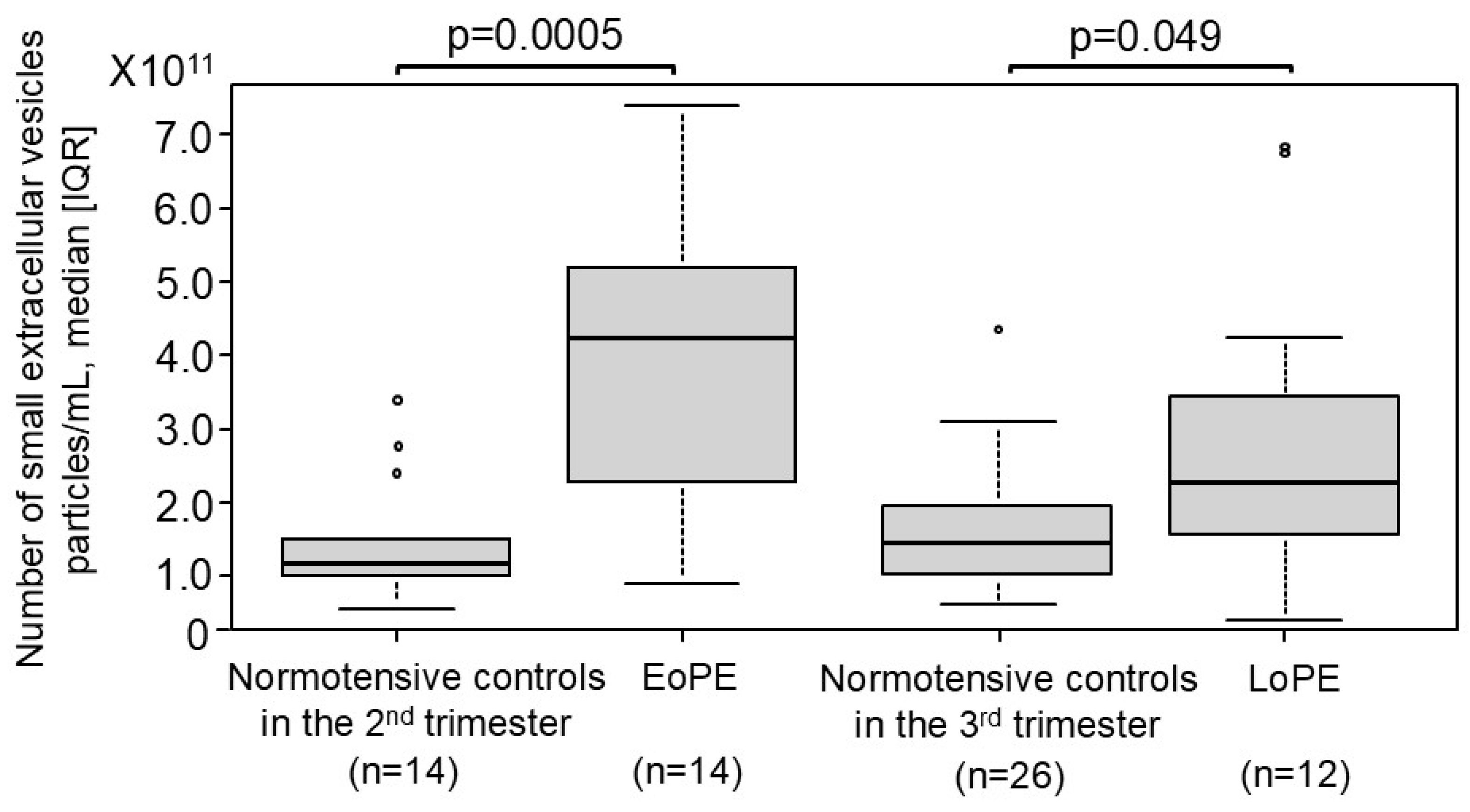
2.4. Comparison of the Number of Small EVs Between Patients with PE and Normotensive Controls
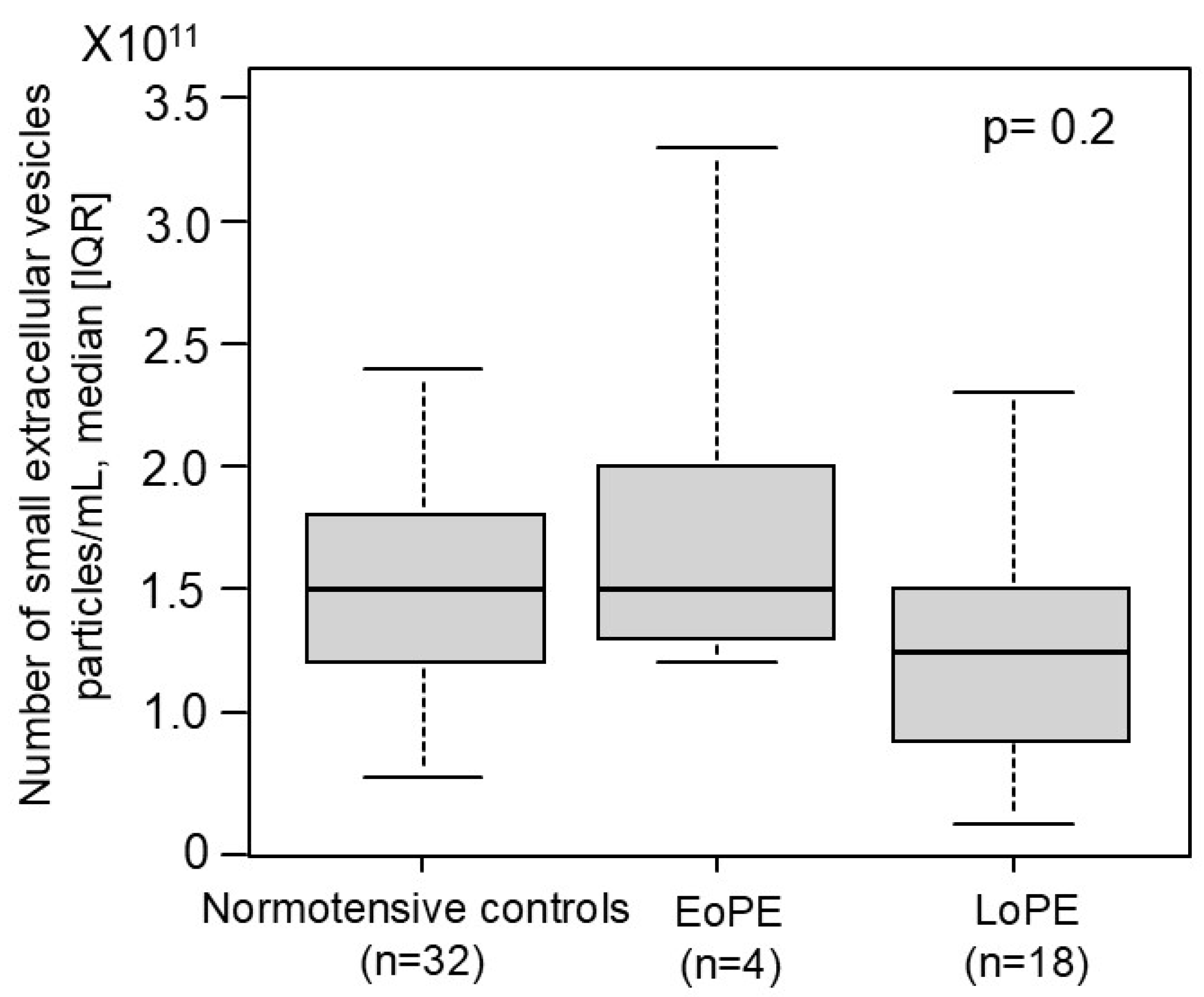
2.5. Comparison of the Number of Small EVs in the First Trimester Between Women Who Later Developed PE and Normotensive Controls in the Prospective Nested Case-Control Study
3. Discussion
4. Materials and Methods
4.1. Study Participants and Sample Collection
4.2. Purification of Small EVs
4.3. Western Blotting
4.4. Nanoparticle Tracking Analysis (NTA)
4.5. Transmission Electron Microscopy (TEM)
4.6. Measurement of Small EV Numbers
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EVs | Extracellular vesicles |
| EoPE | Early-onset preeclampsia |
| LoPE | Late-onset preeclampsia |
| NTA | Nanoparticle tracking analysis |
| HAECs | Human aortic endothelial cells |
| HUVECs | Human Umbilical Vein Endothelial Cells |
| NIPT | Non-invasive prenatal testing |
| TEM | Transmission electron microscopy |
References
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Small EV-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Thakur, B.K.; Zhang, H.; Becker, A.; Matei, I.; Huang, Y.; Costa-Silva, B.; Zheng, Y.; Hoshino, A.; Brazier, H.; Xiang, J.; et al. Double-stranded DNA in small EVs: A novel biomarker in cancer detection. Cell Res. 2014, 24, 766–769. [Google Scholar] [CrossRef]
- Maas, S.L.N.; Breakefield, X.O.; Weaver, A.M. Extracellular Vesicles: Unique Intercellular Delivery Vehicles. Trends Cell Biol. 2017, 27, 172–188. [Google Scholar] [CrossRef]
- Hashimoto, A.; Sugiura, K.; Hoshino, A. Impact of small EV-mediated feto-maternal interactions on pregnancy maintenance and development of obstetric complications. J. Biochem. 2021, 169, 163–171. [Google Scholar] [CrossRef]
- Mitchell, M.D.; Peiris, H.N.; Kobayashi, M.; Koh, Y.Q.; Duncombe, G.; Illanes, S.E.; Rice, G.E.; Salomon, C. Placental small EVs in normal and complicated pregnancy. Am J Obstet Gynecol. 2015, 213, 173–181. [Google Scholar] [CrossRef]
- Yu, Y.; Min, Z.; Zhou, Z.; Linhong, M.; Tao, R.; Yan, L.; Song, H. Hypoxia-induced small EVs promote hepatocellular carcinoma proliferation and metastasis via miR-1273f transfer. Exp. Cell Res. 2019, 385, 111649. [Google Scholar] [CrossRef]
- Huang, C.; Fisher, K.P.; Hammer, S.S.; Navitskaya, S.; Blanchard, G.J.; Busik, J.V. Plasma Small EVs Contribute to Microvascular Damage in Diabetic Retinopathy by Activating the Classical Complement Pathway. Diabetes. 2018, 67, 1639–1649. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Kano, M.; Akutsu, Y.; Hanari, N.; Hoshino, I.; Murakami, K.; Usui, A.; Suito, H.; Takahashi, I.; Otsuka, R.; et al. Quantification of plasma exosome is a potential prognostic marker for esophageal squamous cell carcinoma. Oncol Rep. 2016, 36, 2535–2543. [Google Scholar] [CrossRef]
- Silva, J.; Garcia, V.; Rodriguez, M.; Compte, M.; Cisneros, E.; Veguillas, P.; Garcia, J.M.; Dominguez, G.; Campos-Martin, Y.; Cuevas, J.; et al. Analysis of Small EV Release and Its Prognostic Value in Human Colorectal Cancer. Genes Chromosomes Cancer. 2012, 51, 409–418. [Google Scholar] [CrossRef]
- Lee, J.Y.; Park, J.K.; Lee, E.Y.; Lee, E.B.; Song, Y.W. Circulating exosomes from patients with systemic lupus erythematosus induce a proinflammatory immune response. Arthritis Res Ther. 2016, 18, 264. [Google Scholar] [CrossRef]
- Salomon, C.; Torres, M.J.; Kobayashi, M.; Scholz-Romero, K.; Sobrevia, L.; Dobierzewska, A.; Illanes, S.E.; Mitchell, M.D.; Rice, G.E. A Gestational Profile of Placental Small EVs in Maternal Plasma and Their Effects on Endothelial Cell Migration. PLoS ONE. 2014, 9, e98667. [Google Scholar] [CrossRef]
- Arias, M.; Monteiro, L.J.; Acuna-Gallardo, S.; Varas-Godoy, M.; Rice, G.E.; Monckeberg, M.; Diaz, P.; Illanes, S.E. Extracellular vesicle concentration in maternal plasma as an early marker of gestational diabetes. Rev Med Chil. 2019, 147, 1503–1509. [Google Scholar] [CrossRef]
- Salomon, C.; Guanzou, D.; Scholz-Romero, K.; Longo, S.; Correa, P.; Illanes, S.E.; Rice, G.E. Placental Exosomes as Early Biomarker of Preeclampsia: Potential Role of Exosomal MicroRNAs Across Gestation. J Clin Endocrinol Metab. 2017, 102, 3182–3194. [Google Scholar] [CrossRef]
- Pillay, P.; Maharaj, N.; Moodley, J.; Mackraj, I. Placental small EVs and pre-eclampsia: Maternal circulating levels in normal pregnancies and, early and late onset pre-eclamptic pregnancies. Placenta. 2016, 46, 18–25. [Google Scholar] [CrossRef]
- Murugesan, S.; Hussey, H.; Saravanakumar, L.; Sinkey, R.G.; Sturdivant, A.B.; Powell, M.F.; Berkowitz, D.E. Extracellular Vesicles From Women With Severe Preeclampsia Impair Vascular Endothelial Function. Anesth Analg. 2022, 134, 713–723. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, P.; Li, J.; Liu, L.; Zhao, Z.; Gao, Y.; Oin, Y.; Xu, Q.; Yan, Y.; et al. Association of placenta-derived extracellular vesicles with pre-eclampsia and associated hypercoagulability: A clinical observational study. BJOG. 2021, 128, 1037–1046. [Google Scholar] [CrossRef]
- Karimi, N.; Dalirfardouel, R.; Dias, T.; Lotcall, J.; Lasser, C. Tetraspanins distinguish separate extracellular vesicle subpopulations in human serum and plasma– Contributions of platelet extracellular vesicles in plasma samples. J Extracell Vesicles. 2022, 11, e12213. [Google Scholar] [CrossRef]
- Hoshino, A.; Kim, H.S.; Bojmar, L.; Gyan, K.E.; Cioffi, M.; Hernandez, J.; Zambirinis, C.P.; Rodrigues, G.; Molina, H.; Heissel, S.; et al. Extracellular Vesicle and Particle Biomarkers Define Multiple Human Cancers. Cell. 2020, 182, 1044–1061. [Google Scholar] [CrossRef]
- Kilinc, S.; Paisner, R.; Camarda, R.; Gupta, S.; Momcilovic, O.; Kohnz, R.A.; Avsaroglu, B.; L’Etoile, N.D.; Perera, R.M.; Nomura, D.K.; et al. Oncogene Regulated Release of Extracellular Vesicles. Dev Cell. 2021, 56, 1989–2006. [Google Scholar] [CrossRef]
- Bebelman, M.P.; Smit, M.J.; Pegtel, D.M.; Baglio, S.R. Biogenesis and function of extracellular vesicles in cancer. Pharmacol Ther. 2018, 188, 1–11. [Google Scholar] [CrossRef]
- Xu, F.; Wang, K.; Zhu, C.; Fan, L.; Zhu, Y.; Wang, J.F.; Zhao, Y.; Zhu, C.; Zhang, W.; Yang, F.; et al. Tumor-derived extracellular vesicles as a biomarker for breast cancer diagnosis and metastasis monitoring. iScience. 2024, 27, 109506. [Google Scholar] [CrossRef]
- Choi, B.H.; Quan, Y.H.; Rho, J.; Hong, S.; Park, Y.; Choi, Y.; Park, J.; Yong, H.S.; Han, K.N.; Choi, Y.H.; et al. Levels of Extracellular Vesicles in Pulmonary and Peripheral Blood Correlate with Stages of Lung Cancer Patients. World J Surg. 2020, 44, 3522–3529. [Google Scholar] [CrossRef] [PubMed]
- Dimitrakopoulos, F.; Kottorou, A.E.; Rodgers, K.; Sherwood, J.T.; Koliou, G.; Lee, B.; Yang, A.; Brahmer, J.R.; Baylin, S.B.; Yang, S.C.; et al. Clinical Significance of Plasma CD9-Positive Exosomes in HIV Seronegative and Seropositive Lung Cancer Patients. Cancers. 2021, 13, 5193. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.J.; Welton, J.; Staffurth, J.; Court, J.; Mason, M.D.; Tabi, Z.; Clayton, A. Can urinary exosomes act as treatment response markers in prostate cancer? J Transl Med. 2009, 7, 1–13. [Google Scholar] [CrossRef]
- Logozzi, M.; Mizzoni, D.; Raimo, R.D.; Giuliani, A.; Maggi, M.; Sciarra, A.; Fais, S. Plasmatic Exosome Number and Size Distinguish Prostate Cancer Patients From Healthy Individuals: A Prospective Clinical Study. Front Oncol. 2021, 11, 727317. [Google Scholar] [CrossRef]
- Welsh, J.A.; Goberdhan, D.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Vizio, D.D.; Driedonks, T.P.; Erdbrügger, U.; et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024, 13, e12404. [Google Scholar] [CrossRef]
- Bowman-Gibson, S.; Chandiramani, C.; Stone, M.L.; Waker, C.A.; Maxwell, R.A.; Dhanraj, D.N.; Brown, T.L. Streamlined Analysis of Maternal Plasma Indicates Small Extracellular Vesicles are Significantly Elevated in Early-Onset Preeclampsia. Reprod Sci. 2024, 31, 2771–2782. [Google Scholar] [CrossRef]
- Truong, G.; Guanzon, D.; Kinhal, V.; Elfeky, O.; Lai, A.; Longo, S.; Nuzhat, Z.; Palma, C.; Scholz-Romero, K.; Menon, R.; et al. Oxygen tension regulates the miRNA profile and bioactivity of exosomes released from extravillous trophoblast cells – Liquid biopsies for monitoring complications of pregnancy. PLoS ONE. 2017, 12, e0174514. [Google Scholar] [CrossRef]
- Rice, G.E.; Scholz-Romero, K.; Sweeney, E.; Peiris, H.; Kobayashi, M.; Duncombe, G.; Mitchell, M.D.; Salomon, C. The Effect of Glucose on the Release and Bioactivity of Exosomes From First Trimester Trophoblast Cells. J Clin Endocrinol Metab. 2015, 100, E1280–E1288. [Google Scholar] [CrossRef]
- Shen, L.; Li, Y.; Li, R.; Diao, Z.; Yany, M.; Wu, M.; Sun, H.; Yan, G.; Hu, Y. Placenta-associated serum exosomal miR-155 derived from patients with preeclampsia inhibits eNOS expression in human umbilical vein endothelial cells. Int J Mol Med. 2018, 41, 1731–1739. [Google Scholar] [CrossRef]
- Chang, X.; Yao, J.; He, Q.; Liu, M.; Duan, T.; Wang, K. Exosomes from women with preeclampsia induced vascular dysfunction by delivering sFlt (soluble fms-like tyrosine kinase)-1 and sEng (soluble endoglin) to endothelial cells. Hypertension 2018, 72, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Hestand, M.S.; Bessem, M.; Rijn, P.; Menezes, R.X.; Sie, D.; Bakker, I.; Boon, E.M.J.; Sistermans, E.A.; Weiss, M.M. Fetal fraction evaluation in non-invasive prenatal screening (NIPS). Eur J Hum Genet. 2019, 27, 198–202. [Google Scholar] [CrossRef]
- Wang, E.; Batey, A.; Struble, C.; Musci, T.; Song, K.; Oliphant, A. Gestational age and maternal weight effects on fetal cell-free DNA in maternal plasma. Prenat Diagn. 2013, 33, 662–666. [Google Scholar] [CrossRef]
- Sarker, S.; Scholz-Romero, K.; Perez, A.; Illanes, S.E.; Mitchell, M.D.; Rice, G.E.; Salomon, C. Placenta-derived exosomes continuously increase in maternal circulation over the first trimester of pregnancy. J Transl Med. 2014, 12, 204. [Google Scholar] [CrossRef]
- Watanabe, K.; Matsubara, K.; Nakamoto, O.; Ushijima, J.; Ohkuchi, A.; Koide, K.; Makino, S.; Mimura, K.; Morikawa, M.; Naruse, K.; et al. Outline of the new definition and classification of “Hypertensive Disorders of Pregnancy (HDP)”; a revised JSSHP statement of 2005. Hypertens Res Pregnancy. 2018. [Google Scholar] [CrossRef]
 |
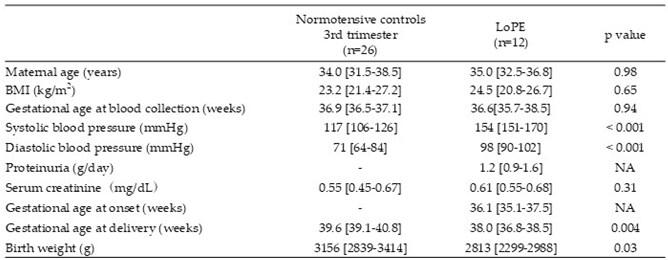 |
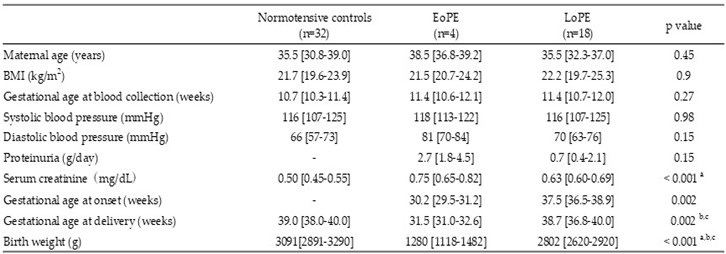 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




