Submitted:
02 October 2025
Posted:
03 October 2025
Read the latest preprint version here
Abstract
Keywords:
Key Points
- Strong evidence shows MDMA-AT can produce large, lasting reductions in PTSD symptoms.
- Blinding remains problematic because MDMA’s psychoactive effects are obvious to participants.
- TAML model explains how trauma memories are stored, reactivated, and reinforced in brain circuits.
- MDMA creates a “therapeutic window” by lowering fear signals and enhancing emotional regulation.
- Different trauma types respond differently: acute trauma may resolve quickly, while complex trauma needs extended care.
- Introduction of BMPP, a new clinical trial design uses role-based masking to reduce bias while supporting effective therapy.
1. Introduction
2. A Functional Model of Dysregulated Memory Recall in Trauma
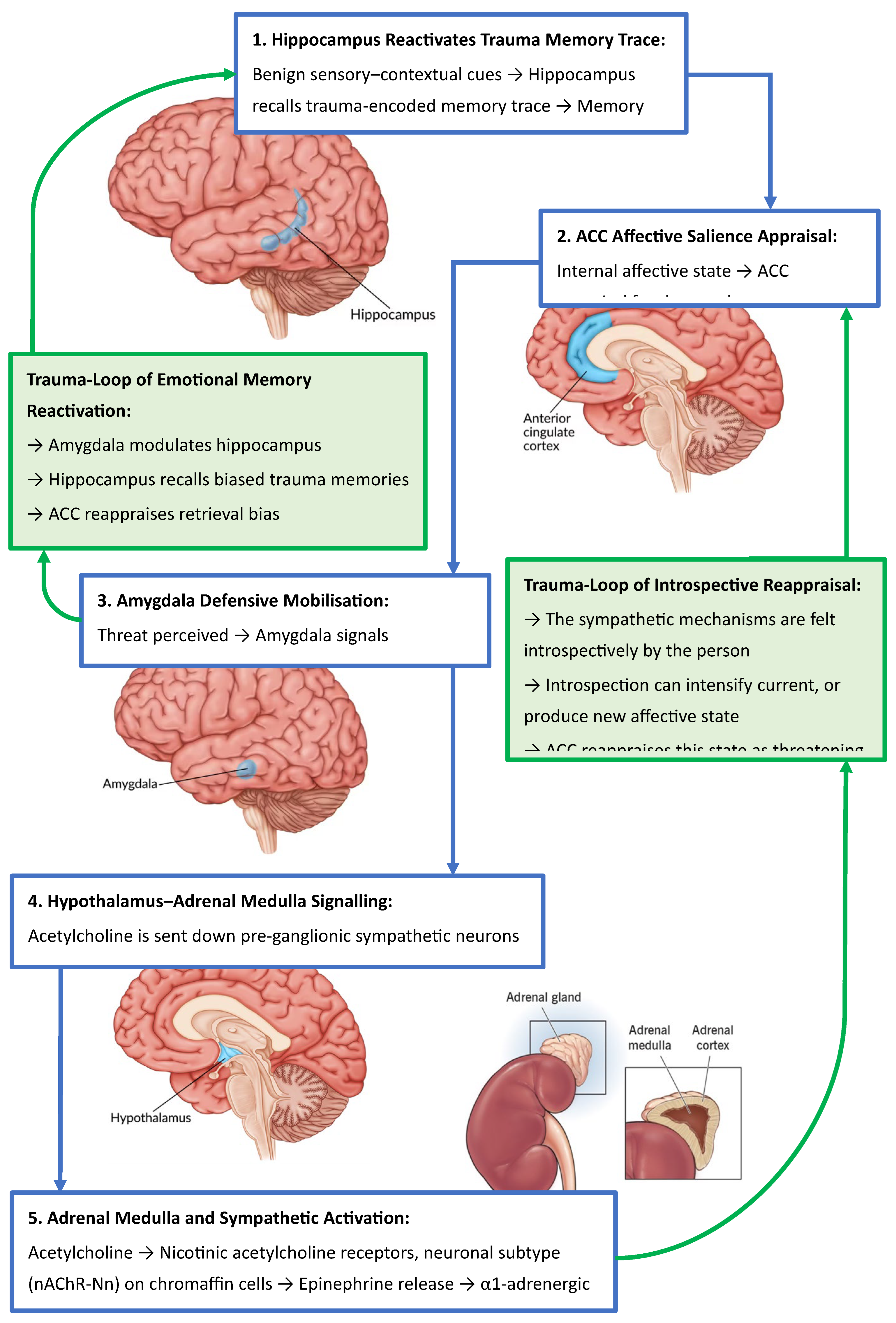
2.1. Mapping the Trauma-Affective .Memory Loop
2.2. From Context to Crisis: Memory Retrieval and Affective Appraisal
2.3. Trauma as an Adaptive Response of the Amygdala
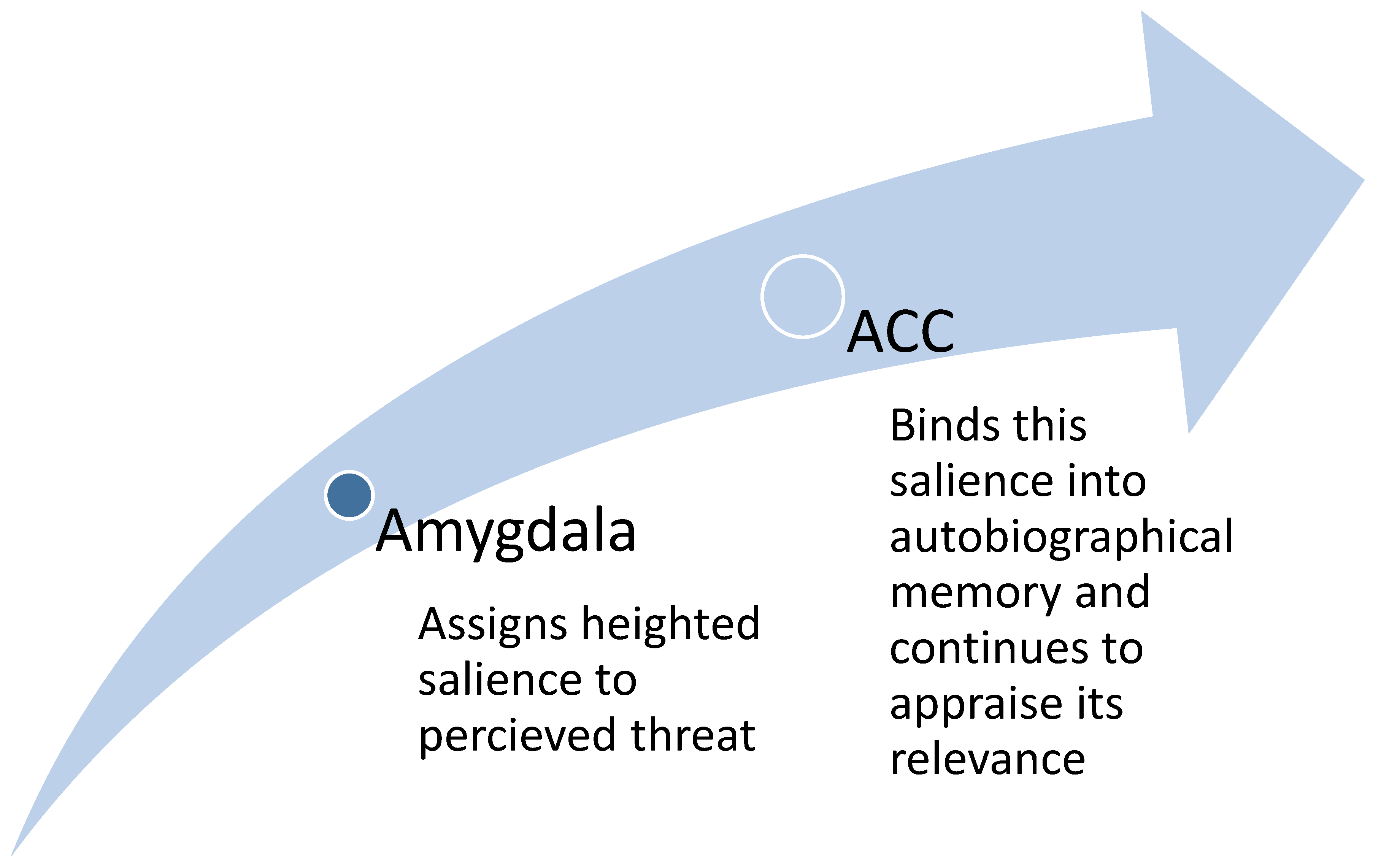
2.4. The Role of the ACC in Triggering the Adaptive Response of the Amygdala
2.5. Affective Fusion: When Emotion Becomes Identity
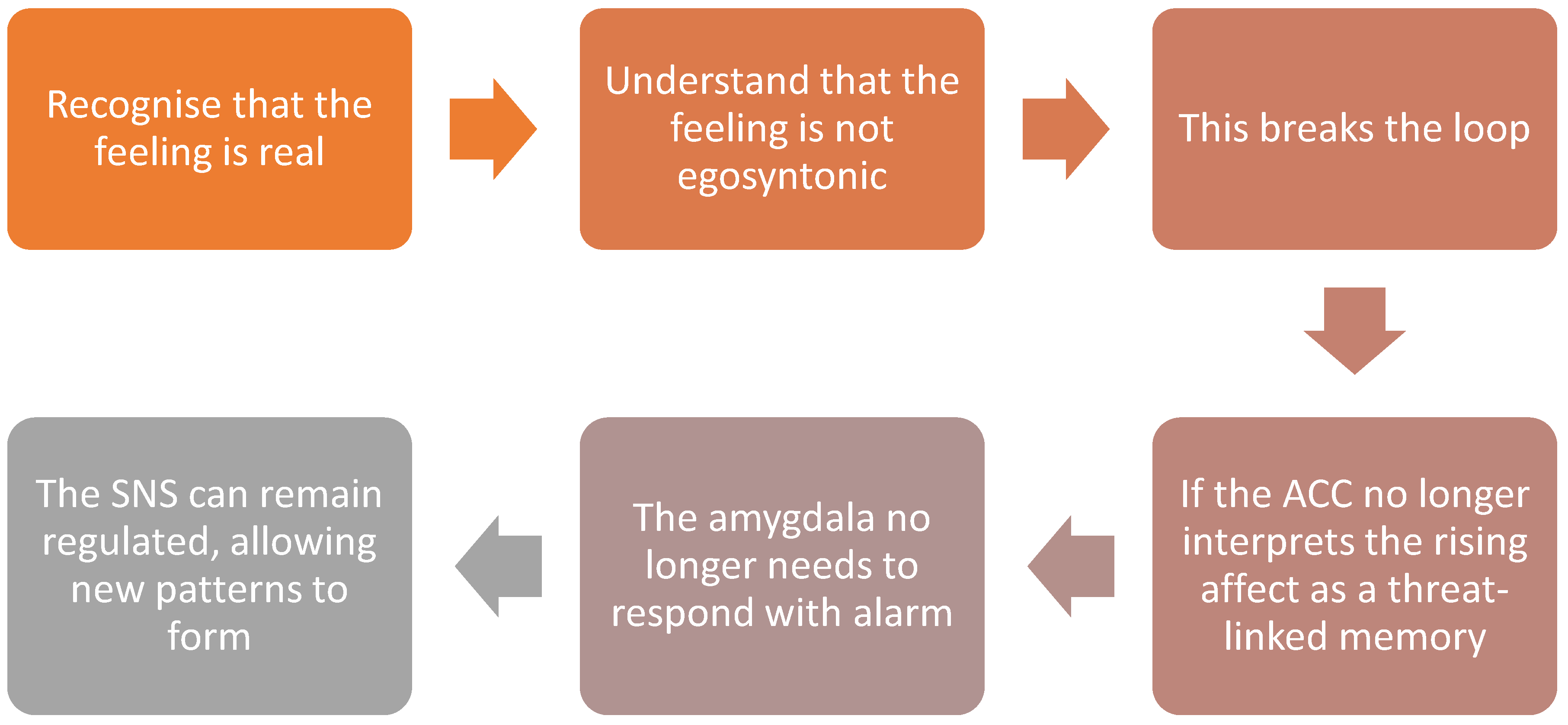
2.6. Dis-identification and the Deconstruction of Affective Imprints
3. Neuropsychopharmacology of MDMA
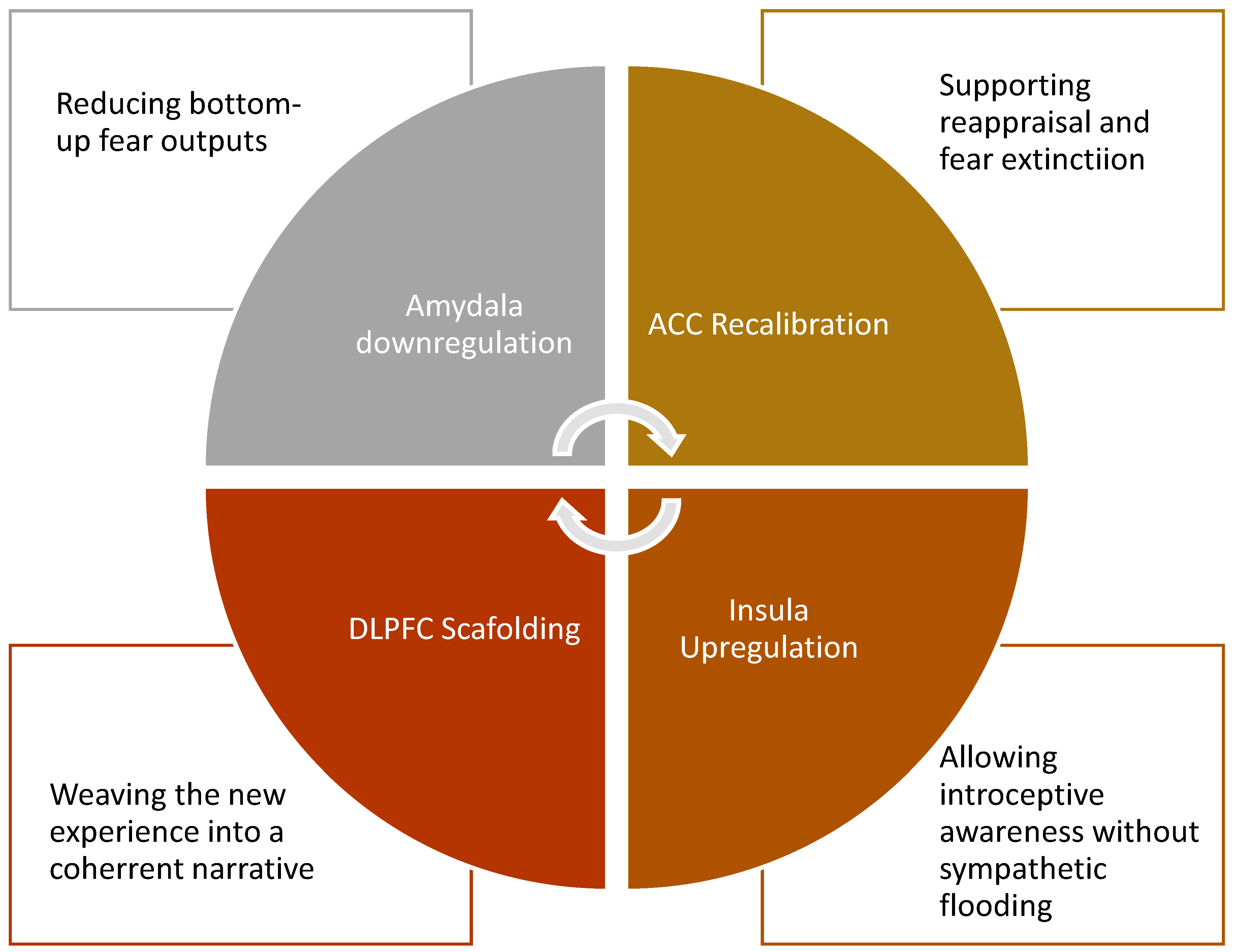
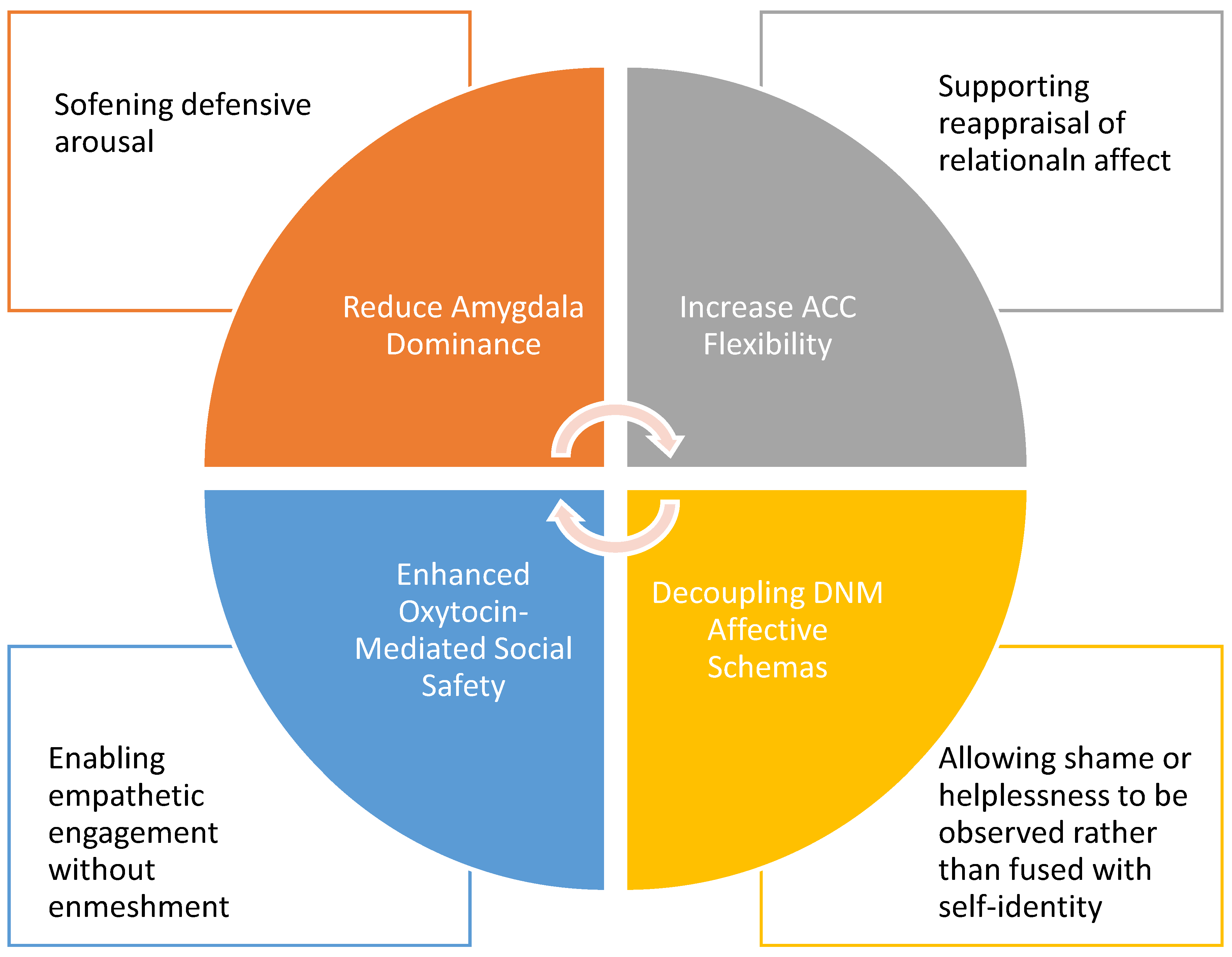
3.1. Affective Circuitry Modulation in MDMA-Assisted Therapy
3.2. Corticolimbic Decoupling and Salience Recalibration
- Decreasing amygdala hyperactivity: MDMA directly dampens the amygdala’s excitability and weakens its bottom-up output to the hypothalamus (Sottile and Vida, 2022), reducing the body’s automatic fear-driven reactions.
- Enhancing fear extinction: By modulating amygdala–ACC FC, MDMA raises the threshold for the ACC to classify a stimulus as threatening (Doss et al., 2018; Luoma and Lear, 2021; Walpola et al., 2017). This recalibration allows traumatic memories to be revisited without triggering the defensive sympathetic cascade, creating the conditions for extinction learning.
3.3. The Therapeutic Window: Self-Referential Network Remapping and Affective Decoupling
- This is the therapeutic window: a brain state where emotional and affective states can be observed rather than fused with, enabling metacognitive awareness. Within this altered context, traumatic memories can be re-encountered without defensive mobilisation. Instead, they are re-encoded against a backdrop of coherence, empathy, and agency. Over repeated exposures, the traumatic imprint shifts from a danger-laden signal to a survivorship narrative, supporting integration.
3.4. Role of the Prefrontal Cortex in Supporting Integration
4. The Brinzei MDMA-PTSD Protocol
4.1. Formulating the Hypothesis
4.2. Differentiating Trauma Exposure: Neurobiological Implications for MDMA-Assisted Therapy
4.2.1. Neurophenomenological Stratification of Trauma Exposure
4.2.2. Direct Exposure and Affective Traceability
4.2.3. Indirect Exposure and Affective Enmeshment
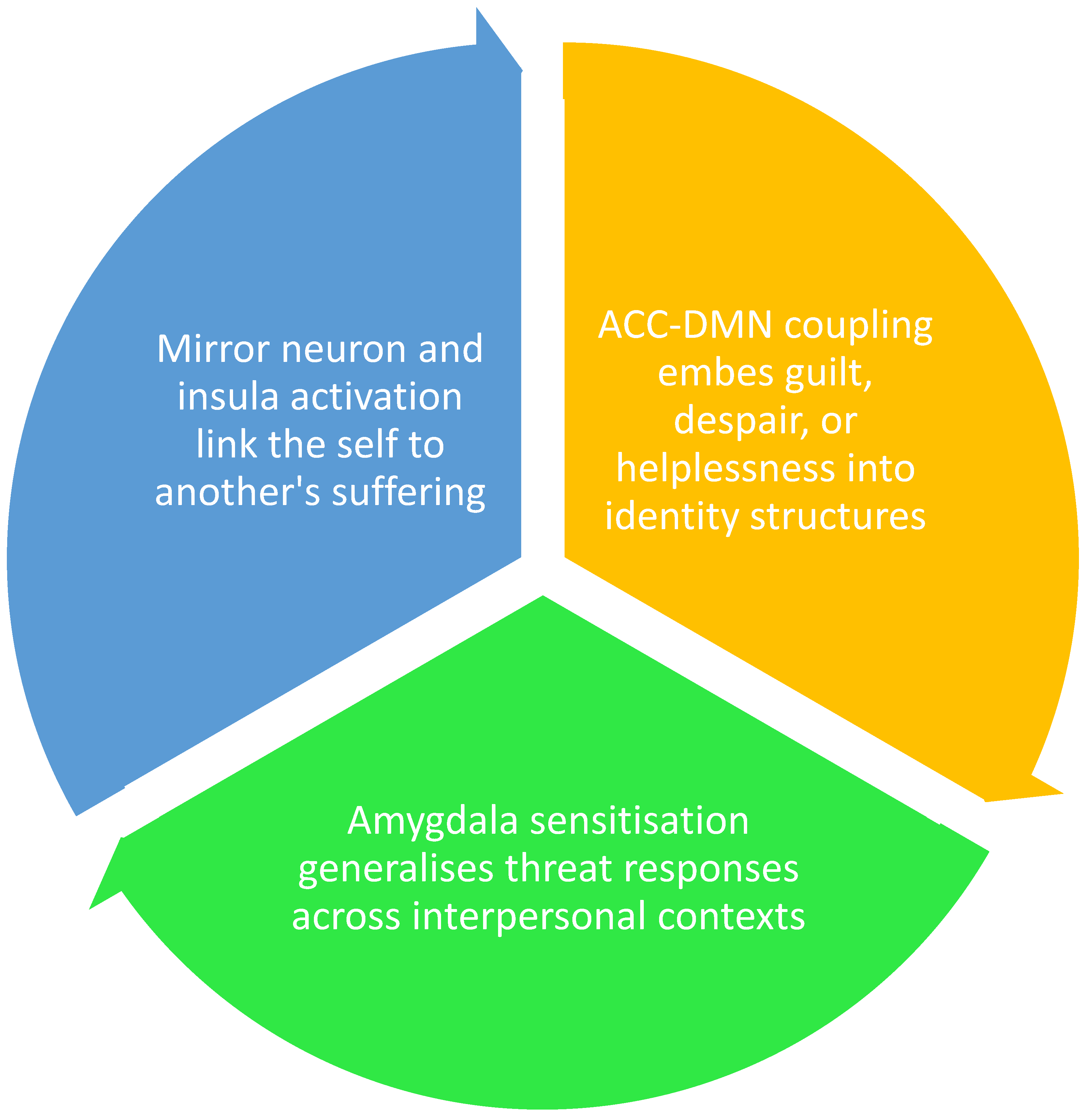
4.2.4. Clinical Implications
| Trauma Type | PTSD Phenotype | Nature of Exposure | Affective Encoding Profile | MDMA Therapeutic Potential |
|---|---|---|---|---|
| Acute direct trauma | Classic PTSD | First-hand, time-bound | Amygdala–ACC circuit; discrete imprint | High; rapid decoupling and re-encoding |
| Chronic direct trauma | Complex PTSD | Sustained, developmental | Multi-network dysregulation (ACC, DMN) | Moderate; requires extended integration |
| Witnessed traumaa | Secondary PTSD | Vicarious, empathic | Partial co-activation, less personal | Moderate; depends on identity fusion |
| Witnessed traumab | Relational PTSD | Deeply personal, empathic | Affective enmeshment, insula/ACC load | Variable; depends on healing in the other |
| Repeated exposurec | Occupational PTSD | Chronic, cumulative | Layered micro-trauma, moral injury | Moderate; may require phased intervention |
5. Methodological Foundations of Masking Design
5.1. Clinical Rationale for Expectancy Control
5.2. Role-Based Masking Justification
| Role | Masked To | Rationale |
|---|---|---|
| Participants | Phenotype classification; Therapeutic hypothesis |
Reduces expectancy effects and demand characteristics |
| Care Providers | Phenotype classification | Prevents bias in supportive care delivery |
| Outcome Assessors | Phenotype classification | Ensures objectivity in clinical outcome assessment |
| Therapists | Not blinded | Must know phenotype and rationale to deliver targeted psychotherapy |
| Investigators | Phenotype classification | Preserves analytic objectivity while maintaining scientific oversight |
5.3. Masking Integrity: Role Conflict Management
| Role | Excluded Function | Access Restrictions |
|---|---|---|
| Therapist | Cannot act as investigator, recruiter, care provider, or outcome assessor | Access to trauma phenotype and rationale; excluded from assessment and analysis |
| Recruiter | Cannot act as therapist, care provider, outcome assessor, or data-analysing | Blinded to phenotype where possible; firewalled if also acting in investigator role |
| Investigator | Cannot act as therapist, recruiter, care provider, or outcome assessor | Masked to phenotype; may access treatment data for monitoring; no direct participant contact |
6. Discussion
7. Conclusions
Funding
Acknowledgements
Declaration of conflicting interests
References
- Aday JS, Heifets BD, Pratscher SD, et al. (2022) Great Expectations: recommendations for improving the methodological rigor of psychedelic clinical trials. Psychopharmacology 239(6): 1989-2010. [CrossRef]
- Akiki TJ, Averill CL and Abdallah CG (2017) A Network-Based Neurobiological Model of PTSD: Evidence From Structural and Functional Neuroimaging Studies. Curr Psychiatry Rep 19(11): 81. [CrossRef]
- Albert AE and Back AL (2025) Psychoanalytically informed MDMA-assisted therapy for pathological narcissism: a novel theoretical approach. Front Psychiatry 16: 1529427.
- Arluk S, Matar MA, Carmi L, et al. (2022) MDMA treatment paired with a trauma-cue promotes adaptive stress responses in a translational model of PTSD in rats. Transl Psychiatry 12(1): 181. [CrossRef]
- Arnsten AFT, Raskind MA, Taylor FB, et al. (2015) The effects of stress exposure on prefrontal cortex: Translating basic research into successful treatments for post-traumatic stress disorder. Neurobiology of Stress 1: 89-99. [CrossRef]
- Avanceña ALV, Kahn JG and Marseille E (2022) The Costs and Health Benefits of Expanded Access to MDMA-assisted Therapy for Chronic and Severe PTSD in the USA: A Modeling Study. Clinical Drug Investigation 42(3): 243-252. [CrossRef]
- Barber M, Evans S, Marks R, et al. (2025) “I am not pain, I have pain”: A pilot study examining iRest yoga nidra as a mind-body intervention for persistent pain. Complementary Therapies in Clinical Practice 59: 101955. [CrossRef]
- Baroncelli CMC, Lodder P, van der Lee M, et al. (2025) The role of enmeshment and undeveloped self, subjugation and self-sacrifice in childhood trauma and attachment related problems: The relationship with self-concept clarity. Acta Psychologica 254: 104839. [CrossRef]
- Bedi G, Phan KL, Angstadt M, et al. (2009) Effects of MDMA on sociability and neural response to social threat and social reward. Psychopharmacology (Berl) 207(1): 73-83. [CrossRef]
- Belkin MR and Schwartz TL (2015) Alpha-2 receptor agonists for the treatment of posttraumatic stress disorder. Drugs Context 4: 212286.
- Bexis S and Docherty JR (2005) Role of alpha2A-adrenoceptors in the effects of MDMA on body temperature in the mouse. Br J Pharmacol 146(1): 1-6.
- Birkeland MS, Skar AS and Jensen TK (2022) Understanding the relationships between trauma type and individual posttraumatic stress symptoms: a cross-sectional study of a clinical sample of children and adolescents. J Child Psychol Psychiatry 63(12): 1496-1504. [CrossRef]
- Borissova A, Ferguson B, Wall MB, et al. (2021) Acute effects of MDMA on trust, cooperative behaviour and empathy: A double-blind, placebo-controlled experiment. Journal of Psychopharmacology 35(5): 547-555. [CrossRef]
- Brinzei O, Donley CN and Dixon Ritchie G (2023) From prohibited to prescribed: The rescheduling of MDMA and psilocybin in Australia. Drug Science, Policy and Law 9: 20503245231198472. [CrossRef]
- Brinzei OV (2025a) The Brinzei MDMA-PTSD Protocol. Available at: https://brinzei.solutions/blogs/neuroscience/brinzei-mdma-ptsd-protocol.
- Brinzei OV (2025b) Trauma-Effective Memory Loop. Available at: https://brinzei.solutions/blogs/neuroscience/trauma-effective-memory-loop. [CrossRef]
- Burek GA, Waite MR, Heslin K, et al. (2021) Low-dose clonidine in veterans with Posttraumatic stress disorder. Journal of Psychiatric Research 137: 480-485. [CrossRef]
- Busso DS, McLaughlin KA and Sheridan MA (2014) Media exposure and sympathetic nervous system reactivity predict PTSD symptoms after the Boston marathon bombings. Depress Anxiety 31(7): 551-558. [CrossRef]
- Butler M, Jelen L and Rucker J (2022) Expectancy in placebo-controlled trials of psychedelics: if so, so what? Psychopharmacology 239(10): 3047-3055. [CrossRef]
- Carhart-Harris RL, Murphy K, Leech R, et al. (2015) The Effects of Acutely Administered 3,4-Methylenedioxymethamphetamine on Spontaneous Brain Function in Healthy Volunteers Measured with Arterial Spin Labeling and Blood Oxygen Level-Dependent Resting State Functional Connectivity. Biol Psychiatry 78(8): 554-562. [CrossRef]
- Carhart-Harris RL, Wall MB, Erritzoe D, et al. (2014) The effect of acutely administered MDMA on subjective and BOLD-fMRI responses to favourite and worst autobiographical memories. Int J Neuropsychopharmacol 17(4): 527-540. [CrossRef]
- Cheung LK (2025) Guanfacine as an Adjunct Treatment for Complex Post-Traumatic Stress Disorder: A Case Report. J Korean Acad Child Adolesc Psychiatry 36(2): 78-82. [CrossRef]
- Chiamulera C, Hinnenthal I, Auber A, et al. (2014) Reconsolidation of maladaptive memories as a therapeutic target: pre-clinical data and clinical approaches. Front Psychiatry 5: 107. [CrossRef]
- Christie D, Yazar-Klosinski B, Nosova E, et al. (2022) MDMA-assisted therapy is associated with a reduction in chronic pain among people with post-traumatic stress disorder. Front Psychiatry 13: 939302. [CrossRef]
- Doblin R (2002) A Clinical Plan for MDMA (Ecstasy) in the Treatment of Posttraumatic Stress Disorder (PTSD): Partnering with the FDA. Journal of Psychoactive Drugs 34(2): 185-194. [CrossRef]
- Doblin RE, Christiansen M, Jerome L, et al. (2019) The Past and Future of Psychedelic Science: An Introduction to This Issue. Journal of Psychoactive Drugs 51(2): 93-97. [CrossRef]
- Doss MK, Weafer J, Gallo DA, et al. (2018) MDMA Impairs Both the Encoding and Retrieval of Emotional Recollections. Neuropsychopharmacology 43(4): 791-800. [CrossRef]
- Dumont GJ, Sweep FC, van der Steen R, et al. (2009) Increased oxytocin concentrations and prosocial feelings in humans after ecstasy (3,4-methylenedioxymethamphetamine) administration. Soc Neurosci 4(4): 359-366. [CrossRef]
- Edmondson D (2014) An Enduring Somatic Threat Model of Posttraumatic Stress Disorder Due to Acute Life-Threatening Medical Events. Soc Personal Psychol Compass 8(3): 118-134. [CrossRef]
- Etkin A, Egner T and Kalisch R (2011) Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci 15(2): 85-93. [CrossRef]
- Feduccia AA and Mithoefer MC (2018) MDMA-assisted psychotherapy for PTSD: Are memory reconsolidation and fear extinction underlying mechanisms? Prog Neuropsychopharmacol Biol Psychiatry 84(Pt A): 221-228. [CrossRef]
- Fisher J (2017) Trauma-informed stabilisation treatment: A new approach to treating unsafe behaviour. Australian Clinical Psychologist 3(1): 1744.
- Food and Drugs Administration (FDA) (2023) Psychedelic Drugs: Considerations for Clinical Investigations. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/psychedelic-drugs-considerations-clinical-investigations.
- Ford JD and Courtois CA (2021) Complex PTSD and borderline personality disorder. Borderline Personality Disorder and Emotion Dysregulation 8(1): 16.
- Fox AS, Oler JA, Tromp do PM, et al. (2015) Extending the amygdala in theories of threat processing. Trends Neurosci 38(5): 319-329. [CrossRef]
- García-Cabezas M and Barbas H (2017) Anterior Cingulate Pathways May Affect Emotions Through Orbitofrontal Cortex. Cereb Cortex 27(10): 4891-4910. [CrossRef]
- Gattuso JJ, Perkins D, Ruffell S, et al. (2023) Default Mode Network Modulation by Psychedelics: A Systematic Review. Int J Neuropsychopharmacol 26(3): 155-188. [CrossRef]
- Green B (2014) Prazosin in the treatment of PTSD. J Psychiatr Pract 20(4): 253-259.
- Griffiths RR, Johnson MW, Carducci MA, et al. (2016) Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: A randomized double-blind trial. J Psychopharmacol 30(12): 1181-1197. [CrossRef]
- Griffiths RR, Richards WA, McCann U, et al. (2006) Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance. Psychopharmacology (Berl) 187(3): 268-283; discussion 284-292. [CrossRef]
- Harris E (2023) FDA Proposes First Guidance for Researchers Studying Psychedelics. JAMA 330(4): 307-307. [CrossRef]
- Hovmand OR, Poulsen ED, Arnfred S, et al. (2023) Risk of bias in randomized clinical trials on psychedelic medicine: A systematic review. Journal of Psychopharmacology 37(7): 649-659. [CrossRef]
- Huestis MA, Smith WB, Leonowens C, et al. (2025) MDMA pharmacokinetics: A population and physiologically based pharmacokinetics model-informed analysis. CPT: Pharmacometrics & Systems Pharmacology 14(2): 376-388. [CrossRef]
- Jardim AV, Jardim DV, Chaves BR, et al. (2021) 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy for victims of sexual abuse with severe post-traumatic stress disorder: an open label pilot study in Brazil. Braz J Psychiatry 43(2): 181-185. [CrossRef]
- Johansen P and Krebs T (2009) How could MDMA (ecstasy) help anxiety disorders? A neurobiological rationale. Journal of Psychopharmacology 23(4): 389-391. [CrossRef]
- Karp Barnir E, Rubinstein Z, Abend R, et al. (2025) Peri-traumatic consumption of classic psychedelics is associated with lower anxiety and post-traumatic responses 3 weeks after exposure. J Psychopharmacol 39(9): 1031-1036. [CrossRef]
- Kensinger EA, Addis DR and Atapattu RK (2011) Amygdala activity at encoding corresponds with memory vividness and with memory for select episodic details. Neuropsychologia 49(4): 663-673. [CrossRef]
- Kiser LJ, Nurse W, Lucksted A, et al. (2008) Understanding the Impact of Trauma on Family Life From the Viewpoint of Female Caregivers Living in Urban Poverty. Traumatology (Tallahass Fla) 14(3): 77-90. [CrossRef]
- Kredlow MA, Fenster RJ, Laurent ES, et al. (2022) Prefrontal cortex, amygdala, and threat processing: implications for PTSD. Neuropsychopharmacology 47(1): 247-259. [CrossRef]
- Lanius RA, Bluhm RL and Frewen PA (2011) How understanding the neurobiology of complex post-traumatic stress disorder can inform clinical practice: a social cognitive and affective neuroscience approach. Acta Psychiatrica Scandinavica 124(5): 331-348. [CrossRef]
- Liberzon I and Abelson JL (2016) Context Processing and the Neurobiology of Post-Traumatic Stress Disorder. Neuron 92(1): 14-30. [CrossRef]
- Luoma J and Lear MK (2021) MDMA-Assisted Therapy as a Means to Alter Affective, Cognitive, Behavioral, and Neurological Systems Underlying Social Dysfunction in Social Anxiety Disorder. Front Psychiatry 12: 733893.
- Maddox SA, Hartmann J, Ross RA, et al. (2019) Deconstructing the Gestalt: Mechanisms of Fear, Threat, and Trauma Memory Encoding. Neuron 102(1): 60-74. [CrossRef]
- Mahoney D and Markel B (2016) An Integrative Approach to Conceptualizing and Treating Complex Trauma. Psychoanalytic Social Work 23(1): 1-22. [CrossRef]
- Martinelli P, Sperduti M, Devauchelle AD, et al. (2013) Age-related changes in the functional network underlying specific and general autobiographical memory retrieval: a pivotal role for the anterior cingulate cortex. PLoS One 8(12): e82385. [CrossRef]
- Metzner R, Litwin G and Weil G (1965) The relation of expectation and mood to psilocybin reactions: a questionnaire study. Psychedelic Rev 5: 3-39.
- Mitchell JM, Bogenschutz M, Lilienstein A, et al. (2021) MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study. Nat Med 27(6): 1025-1033. [CrossRef]
- Mitchell JM, Ot'alora GM, van der Kolk B, et al. (2023) MDMA-assisted therapy for moderate to severe PTSD: a randomized, placebo-controlled phase 3 trial. Nat Med 29(10): 2473-2480.
- Mithoefer MC, Mithoefer AT, Feduccia AA, et al. (2018) 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy for post-traumatic stress disorder in military veterans, firefighters, and police officers: a randomised, double-blind, dose-response, phase 2 clinical trial. Lancet Psychiatry 5(6): 486-497. [CrossRef]
- Mithoefer MC, Wagner MT, Mithoefer AT, et al. (2011) The safety and efficacy of {+/-}3,4-methylenedioxymethamphetamine-assisted psychotherapy in subjects with chronic, treatment-resistant posttraumatic stress disorder: the first randomized controlled pilot study. J Psychopharmacol 25(4): 439-452. [CrossRef]
- Mocanu V, Mackay L, Christie D, et al. (2022) Safety considerations in the evolving legal landscape of psychedelic-assisted psychotherapy. Substance Abuse Treatment, Prevention, and Policy 17(1): 37. [CrossRef]
- Morris KR, Jaeb M, Dunsmoor JE, et al. (2025) Decoding threat neurocircuitry representations during traumatic memory recall in PTSD. Neuropsychopharmacology 50(3): 568-575. [CrossRef]
- Mueser KT, Gottlieb JD, Xie H, et al. (2015) Evaluation of cognitive restructuring for post-traumatic stress disorder in people with severe mental illness. Br J Psychiatry 206(6): 501-508. [CrossRef]
- Murty VP, Ritchey M, Adcock RA, et al. (2010) fMRI studies of successful emotional memory encoding: A quantitative meta-analysis. Neuropsychologia 48(12): 3459-3469. [CrossRef]
- Nirmalani-Gandhy A, Sanchez D and Catalano G (2015) Terazosin for the treatment of trauma-related nightmares: a report of 4 cases. Clin Neuropharmacol 38(3): 109-111.
- O’Brien S and Nutt D (2025) MDMA-assisted therapy: challenges, clinical trials, and the future of MDMA in treating behavioral disorders. CNS spectrums 30(1): e15.
- Oehen P and Gasser P (2022) Using a MDMA- and LSD-Group Therapy Model in Clinical Practice in Switzerland and Highlighting the Treatment of Trauma-Related Disorders. Front Psychiatry 13: 863552. [CrossRef]
- Oehen P, Traber R, Widmer V, et al. (2013) A randomized, controlled pilot study of MDMA (+/- 3,4-Methylenedioxymethamphetamine)-assisted psychotherapy for treatment of resistant, chronic Post-Traumatic Stress Disorder (PTSD). J Psychopharmacol 27(1): 40-52. [CrossRef]
- Ot'alora GM, Grigsby J, Poulter B, et al. (2018) 3,4-Methylenedioxymethamphetamine-assisted psychotherapy for treatment of chronic posttraumatic stress disorder: A randomized phase 2 controlled trial. J Psychopharmacol 32(12): 1295-1307. [CrossRef]
- Pitts BL, Eisenberg ML, Bailey HR, et al. (2022) PTSD is associated with impaired event processing and memory for everyday events. Cognitive Research: Principles and Implications 7(1): 35. [CrossRef]
- Recchia A, Tonti MP, Mirabella L, et al. (2023) The Pharmacological Class Alpha 2 Agonists for Stress Control in Patients with Respiratory Failure: The Main Actor in the Different Acts. Stresses 3(1): 1-10. [CrossRef]
- Rolls ET (2019) The cingulate cortex and limbic systems for emotion, action, and memory. Brain Struct Funct 224(9): 3001-3018. [CrossRef]
- Roseman L (2025) A reflection on paradigmatic tensions within the FDA advisory committee for MDMA-assisted therapy. Journal of Psychopharmacology 39(4): 313-315. [CrossRef]
- Sar V (2011) Developmental trauma, complex PTSD, and the current proposal of DSM-5. Eur J Psychotraumatol 2. [CrossRef]
- Schenberg EE (2025) From Efficacy to Effectiveness: Evaluating Psychedelic Randomized Controlled Trials for Trustworthy Evidence-Based Policy and Practice. Pharmacology Research & Perspectives 13(2): e70097. [CrossRef]
- Schmid Y and Bershad AK (2024) Altered States and Social Bonds: Effects of MDMA and Serotonergic Psychedelics on Social Behavior as a Mechanism Underlying Substance-Assisted Therapy. Biol Psychiatry Cogn Neurosci Neuroimaging 9(5): 490-499. [CrossRef]
- Schott GD (2015) Pictures of pain: their contribution to the neuroscience of empathy. Brain 138(Pt 3): 812-820. [CrossRef]
- Sessa B, Higbed L and Nutt D (2019) A Review of 3,4-methylenedioxymethamphetamine (MDMA)-Assisted Psychotherapy. Front Psychiatry 10: 138. [CrossRef]
- Sharot T, Riccardi AM, Raio CM, et al. (2007) Neural mechanisms mediating optimism bias. Nature 450(7166): 102-105. [CrossRef]
- Šimić G, Tkalčić M, Vukić V, et al. (2021) Understanding Emotions: Origins and Roles of the Amygdala. Biomolecules 11(6). [CrossRef]
- Singleton SP, Wang JB, Mithoefer M, et al. (2022) Altered brain activity and functional connectivity after MDMA-assisted therapy for post-traumatic stress disorder. Front Psychiatry 13: 947622. [CrossRef]
- Slomski A (2021) MDMA-Assisted Therapy Highly Effective for PTSD. JAMA 326(4): 299-299. [CrossRef]
- Smith C and Koola MM (2016) Evidence for Using Doxazosin in the Treatment of Posttraumatic Stress Disorder. Psychiatr Ann 46(9): 553-555. [CrossRef]
- Sottile RJ and Vida T (2022) A proposed mechanism for the MDMA-mediated extinction of traumatic memories in PTSD patients treated with MDMA-assisted therapy. Front Psychiatry 13: 991753.
- Taylor F and Raskind MA (2002) The alpha1-adrenergic antagonist prazosin improves sleep and nightmares in civilian trauma posttraumatic stress disorder. J Clin Psychopharmacol 22(1): 82-85. [CrossRef]
- Thomaes K, Dorrepaal E, Draijer N, et al. (2013) Increased anterior cingulate cortex and hippocampus activation in Complex PTSD during encoding of negative words. Soc Cogn Affect Neurosci 8(2): 190-200. [CrossRef]
- Thomason ME and Marusak HA (2017) Toward understanding the impact of trauma on the early developing human brain. Neuroscience 342: 55-67. [CrossRef]
- van der Kolk B (2000) Posttraumatic stress disorder and the nature of trauma. Dialogues in Clinical Neuroscience 2(1): 7-22.
- van der Kolk BA (2006) Clinical Implications of Neuroscience Research in PTSD. Annals of the New York Academy of Sciences 1071(1): 277-293.
- van der Kolk BA, Wang JB, Yehuda R, et al. (2024) Effects of MDMA-assisted therapy for PTSD on self-experience. PLoS One 19(1): e0295926.
- Vaslavski A, Gross AH, Israel S, et al. (2025) The effect of MDMA administration on oxytocin concentration levels: systematic review and a multilevel meta-analysis in humans. Neuroscience & Biobehavioral Reviews 177: 106324. [CrossRef]
- Wagner G, Koch K, Schachtzabel C, et al. (2013) Self-referential processing influences functional activation during cognitive control: an fMRI study. Soc Cogn Affect Neurosci 8(7): 828-837. [CrossRef]
- Walpola IC, Nest T, Roseman L, et al. (2017) Altered insula connectivity under MDMA. Neuropsychopharmacology 42(11): 2152-2162. [CrossRef]
- Wang J, John Y and Barbas H (2021a) Pathways for Contextual Memory: The Primate Hippocampal Pathway to Anterior Cingulate Cortex. Cereb Cortex 31(3): 1807-1826. [CrossRef]
- Wang JB, Lin J, Bedrosian L, et al. (2021b) Scaling Up: Multisite Open-Label Clinical Trials of MDMA-Assisted Therapy for Severe Posttraumatic Stress Disorder. Journal of Humanistic Psychology. [CrossRef]
- Wilkinson ST and Sanacora G (2025) Issues in Clinical Trial Design—Lessons From the FDA’s Rejection of MDMA. JAMA Psychiatry 82(6): 545-546.
- Yadav G, McNamara S and Gunturu S (2025) Trauma-Informed Therapy. StatPearls. Treasure Island (FL).
- Yehuda R (2004) Understanding Heterogeneous Effects of Trauma Exposure: Relevance to Postmortem Studies of PTSD. Psychiatry 67(4): 391-397. [CrossRef]
- Young DA, Chao L, Neylan TC, et al. (2018) Association among anterior cingulate cortex volume, psychophysiological response, and PTSD diagnosis in a Veteran sample. Neurobiol Learn Mem 155: 189-196. [CrossRef]
- Yu Y, Li Y, Han D, et al. (2023) Effect of Dexmedetomidine on Posttraumatic Stress Disorder in Patients Undergoing Emergency Trauma Surgery: A Randomized Clinical Trial. JAMA Network Open 6(6): e2318611-e2318611.
- Zhang X, Hack LM, Bertrand C, et al. (2025) Negative Affect Circuit Subtypes and Neural, Behavioral, and Affective Responses to MDMA: A Randomized Clinical Trial. JAMA Network Open 8(4): e257803-e257803.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




