Submitted:
01 October 2025
Posted:
02 October 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Theoretical Framework
3. Computational Details
4. Results and Discussion
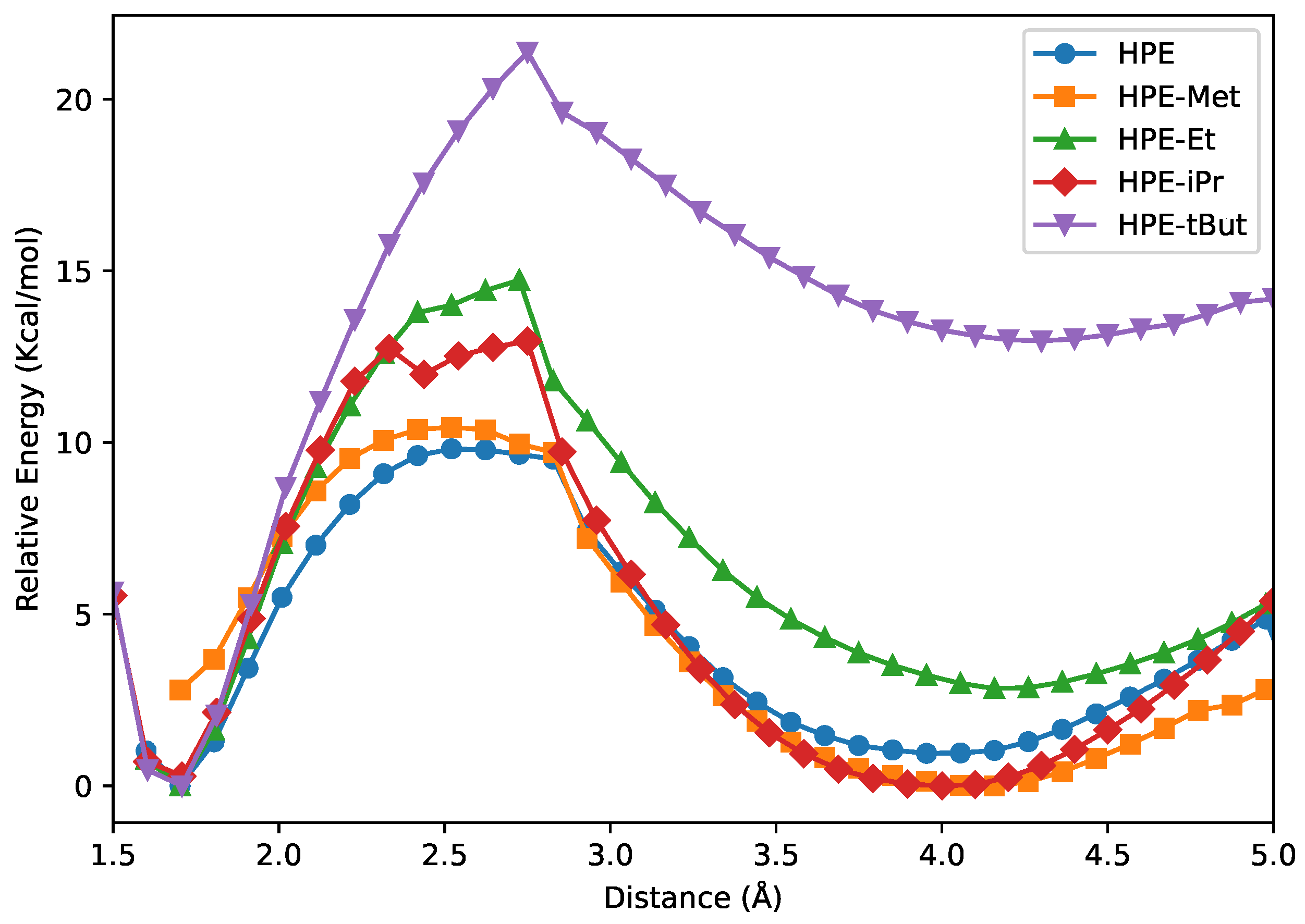
4.1. Potential Energy Curves and Thermodynamic Potentials
4.2. Quantum Theory of Atoms in Molecules Analysis
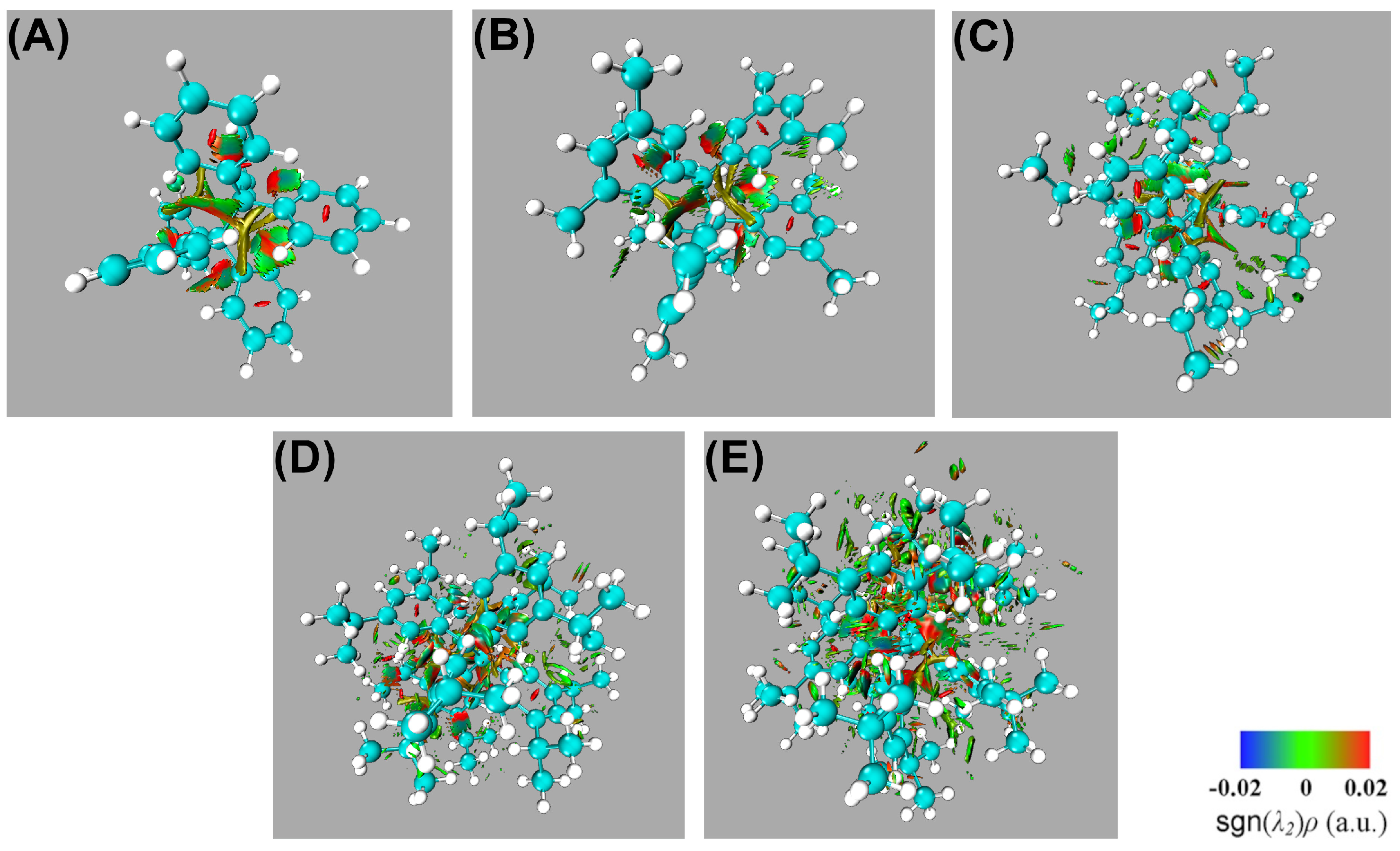
4.3. NCI Analyses
4.4. Interacting Quantum Atoms analyses
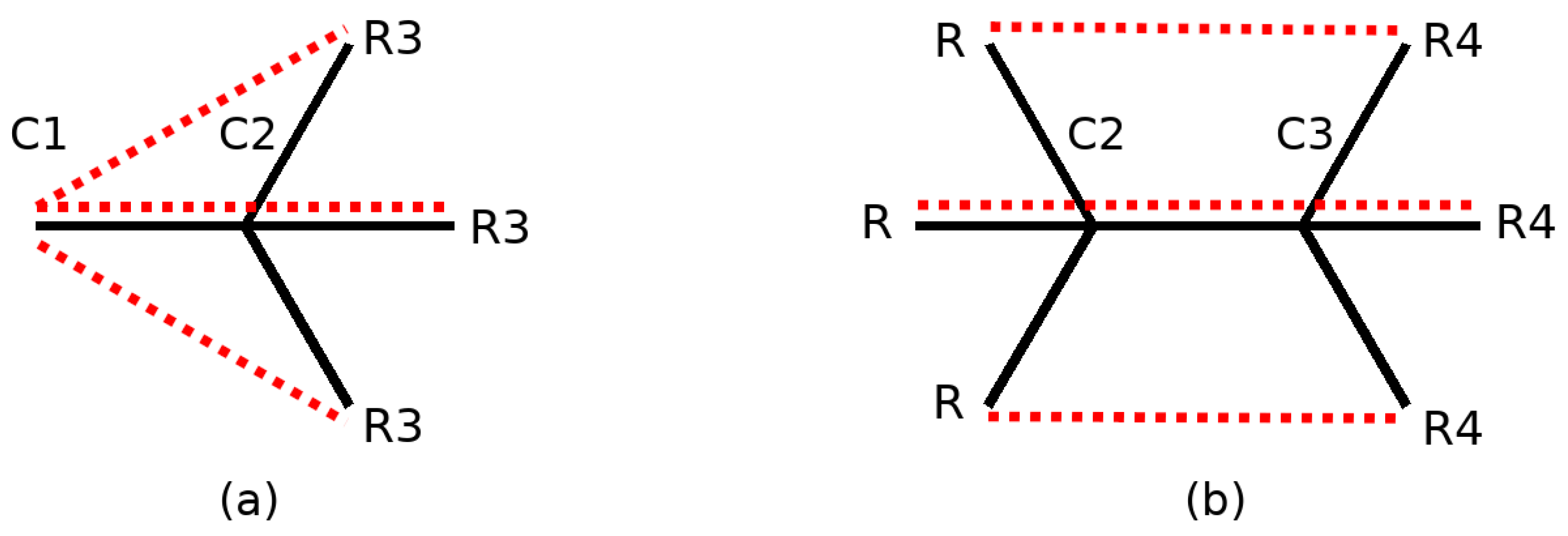
4.5. Collective 1-3 Interactions
4.6. Collective 1–4 Interactions
 | |||
| HPE | |||
| Phenyl groups | IQA partition | Dispersion correction | IQA-D |
| 1–4 | −15.934 | −3.586 | −19.520 |
| 1–5 | −7.340 | −1.372 | −8.713 |
| 1–6 | −0.546 | −0.587 | −1.133 |
| 2–4 | −0.540 | −0.585 | −1.124 |
| 2–5 | −15.933 | −3.639 | −19.572 |
| 2–6 | −7.391 | −1.368 | −8.760 |
| 3–4 | −7.341 | −1.367 | −8.709 |
| 3–5 | −0.544 | −0.590 | −1.134 |
| 3–6 | −15.866 | −3.636 | −19.502 |
| Total | −71.435 | −16.730 | −88.167 |
| C–C | −84.054 | ||
| HPEMe | |||
| Phenyl groups | IQA partition | Dispersion correction | IQA-D |
| 1–4 | −18.967 | −5.397 | −24.363 |
| 1–5 | −7.434 | −1.598 | −9.032 |
| 1–6 | −0.539 | −0.671 | −1.210 |
| 2–4 | −0.553 | −0.688 | −1.242 |
| 2–5 | −19.006 | −5.524 | −24.530 |
| 2–6 | −7.290 | −1.607 | −8.896 |
| 3–4 | −7.206 | −1.612 | −8.818 |
| 3–5 | −0.545 | −0.678 | −1.223 |
| 3–6 | −18.859 | −5.200 | −24.059 |
| Total | −80.399 | −22.975 | −103.373 |
| C–C | −82.055 | ||
| HPEEt | |||
| Phenyl groups | IQA partition | Dispersion correction | IQA-D |
| 1–4 | −24.763 | −6.914 | −31.677 |
| 1–5 | −8.542 | −1.962 | −10.503 |
| 1–6 | −0.538 | −0.693 | −1.231 |
| 2–4 | −0.551 | −0.714 | −1.265 |
| 2–5 | −26.218 | −7.562 | −33.780 |
| 2–6 | −8.503 | −1.958 | −10.462 |
| 3–4 | −9.453 | −2.259 | −11.711 |
| 3–5 | −0.532 | −0.680 | −1.212 |
| 3–6 | −21.566 | −6.345 | −27.911 |
| Total | −100.666 | −29.087 | −129.752 |
| C–C | −85.287 | ||
 | |||
| HPEiPr | |||
| Phenyl groups | IQA partition | Dispersion correction | IQA-D |
| 1–4 | −20.122 | −6.327 | −26.448 |
| 1–5 | −19.190 | −5.635 | −24.826 |
| 1–6 | −0.717 | −0.676 | −1.393 |
| 2–4 | −0.684 | −0.651 | −1.335 |
| 2–5 | −15.569 | −4.708 | −20.277 |
| 2–6 | −15.065 | −4.781 | −19.847 |
| 3–4 | −16.745 | −5.091 | −21.836 |
| 3–5 | −0.666 | −0.625 | −1.291 |
| 3–6 | −17.111 | −5.284 | −22.394 |
| Total | −105.869 | −33.778 | −139.647 |
| C–C | −85.514 | ||
| HPEtBut | |||
| 1–4 | −30.997 | −9.567 | −40.558 |
| 1–5 | −12.906 | −3.607 | −16.513 |
| 1–6 | −0.544 | −0.861 | −1.405 |
| 2–4 | −0.560 | −0.870 | −1.430 |
| 2–5 | −31.056 | −9.567 | −40.623 |
| 2–6 | −11.584 | −3.477 | −15.061 |
| 3–4 | −12.949 | −3.610 | −16.559 |
| 3–5 | −0.560 | −0.870 | −1.430 |
| 3–6 | −29.729 | −9.674 | −39.403 |
| Total | −130.885 | −42.103 | −172.982 |
| C–C | −86.155 | ||
 | |||||
| Phenyl groups | HPE | HPEMe | HPEEt | HPEiPr | HPEtBut |
| 1–4 | 0.298 | 0.364 | 0.473 | 0.377 | 0.593 |
| 1–5 | 0.111 | 0.112 | 0.129 | 0.354 | 0.216 |
| 1–6 | 0.016 | 0.017 | 0.017 | 0.022 | 0.017 |
| 2–4 | 0.016 | 0.017 | 0.017 | 0.021 | 0.017 |
| 2–5 | 0.300 | 0.367 | 0.504 | 0.281 | 0.594 |
| 2–6 | 0.111 | 0.111 | 0.129 | 0.270 | 0.194 |
| 3–4 | 0.111 | 0.110 | 0.149 | 0.308 | 0.216 |
| 3–5 | 0.016 | 0.017 | 0.016 | 0.020 | 0.017 |
| 3–6 | 0.299 | 0.358 | 0.414 | 0.313 | 0.574 |
| Total | 1.278 | 1.471 | 1.847 | 1.966 | 2.438 |
| C–C | 0.853 | 0.841 | 0.860 | 0.861 | 0.865 |
4.7. Comparison Among Covalent 1-2 Versus Collective 1–3 and 1–4 Interactions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
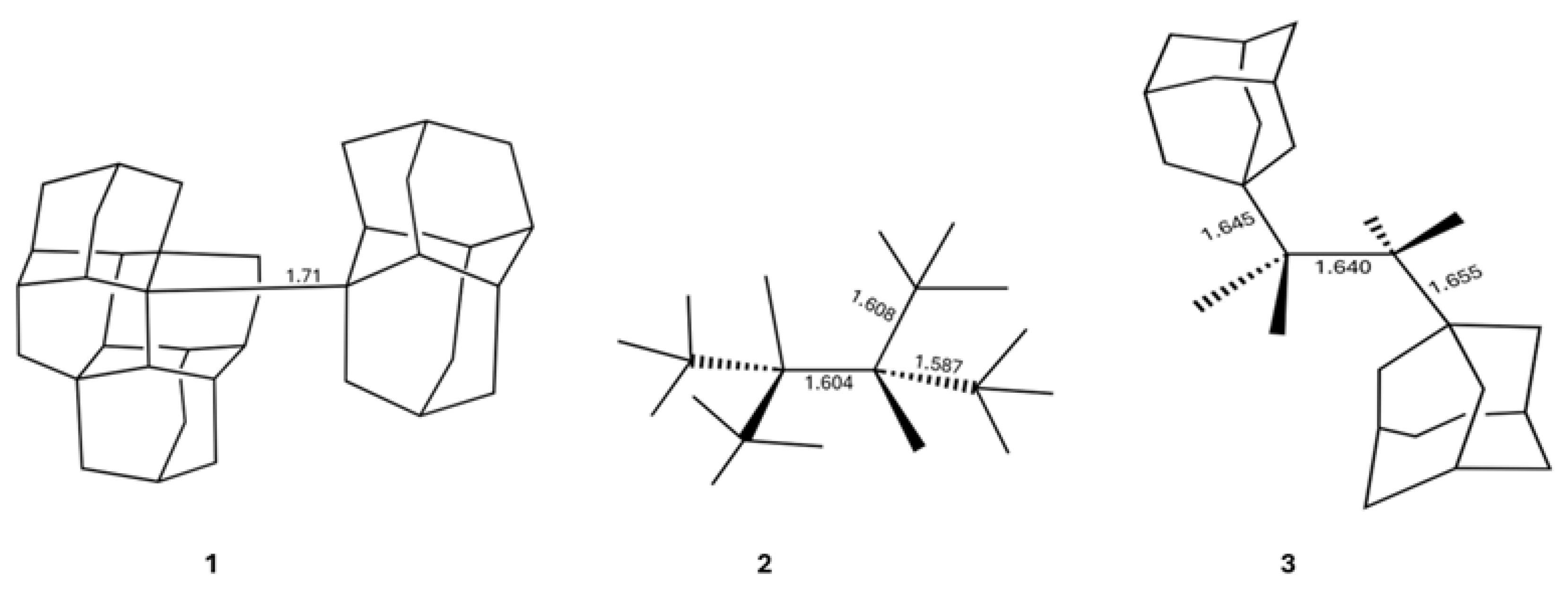
- Pauling, L. The nature of the chemical bond and the structure of molecules and crystals: an introduction to modern structural chemistry; Vol. 18, Cornell university press, 1960.
- Kaupp, M.; Danovich, D.; Shaik, S. Chemistry is about energy and its changes: A critique of bond-length/bond-strength correlations. Coord. Chem. Rev. 2017, 344, 355–362. [CrossRef]
- Blokker, E.; Sun, X.; Poater, J.; van der Schuur, J.M.; Hamlin, T.A.; Bickelhaupt, F.M. The chemical bond: when atom size instead of electronegativity difference determines trend in bond strength. Chem. Eur. J. 2021, 27, 15616–15622. [CrossRef]
- Lobato, A.; Salvadó, M.A.; Recio, J.M.; Taravillo, M.; Baonza, V.G. Highs and lows of bond lengths: Is there any limit? Angew. Chem. Int. Ed. 2021, 133, 17165–17173. [CrossRef]
- Zhao, L.; Zhi, M.; Frenking, G. The strength of a chemical bond. Int. J. Quantum Chem. 2022, 122, e26773. [CrossRef]
- Ishigaki, Y.; Shimajiri, T.; Takeda, T.; Katoono, R.; Suzuki, T. Longest C–C single bond among neutral hydrocarbons with a bond length beyond 1.8 Å. Chem 2018, 4, 795–806. [CrossRef]
- Shimajiri, T.; Suzuki, T.; Ishigaki, Y. Flexible C- C Bonds: Reversible Expansion, Contraction, Formation, and Scission of Extremely Elongated Single Bonds. Angew. Chem. Int. Ed. 2020, 59, 22252–22257. [CrossRef] [PubMed]
- Fokin, A.A. Long but strong C–C single bonds: challenges for theory. Chem. Rec. 2024, 24, e202300170. [CrossRef]
- Tyborski, C.; Hückstaedt, T.; Gillen, R.; Otto, T.; Fokina, N.A.; Fokin, A.A.; Schreiner, P.R.; Maultzsch, J. Vibrational signatures of diamondoid dimers with large intramolecular London dispersion interactions. Carbon 2020, 157, 201–207. [CrossRef]
- Brownstein, S.; Dunogues, J.; Lindsay, D.; Ingold, K. Conformation and rotational barriers in sym-tetra-tert-butylethane and sym-tetra (trimethylsilyl) ethane. J. Am. Chem. Soc. 1977, 99, 2073–2078. [CrossRef]
- Meer, M.A.F.t.; Beckhaus, H.D.; Peters, K.; von Schnering, H.G.; Rüchardt, C. Thermolabile hydrocarbons, XXVII. 2, 3-Di-1-adamantyl-2, 3-dimethylbutane; long bonds and low thermal stability. Chem. Ber. 1985, 118, 4665–4673. [CrossRef]
- Schreiner, P.R.; Chernish, L.V.; Gunchenko, P.A.; Tikhonchuk, E.Y.; Hausmann, H.; Serafin, M.; Schlecht, S.; Dahl, J.E.; Carlson, R.M.; Fokin, A.A. Overcoming lability of extremely long alkane carbon–carbon bonds through dispersion forces. Nature 2011, 477, 308–311. [CrossRef]
- Gravillier, L.A.; Cockroft, S.L. Context-dependent significance of London dispersion. Acc. Chem. Res. 2023, 56, 3535–3544. [CrossRef]
- Zhang, B.; Zarić, S.D.; Zrilić, S.S.; Gofman, I.; Heck, B.; Reiter, G. London dispersion forces and steric effects within nanocomposites tune interaction energies and chain conformation. Commun. Chem. 2025, 8, 21. [CrossRef]
- Rösel, S.; Balestrieri, C.; Schreiner, P.R. Sizing the role of London dispersion in the dissociation of all-meta tert-butyl hexaphenylethane. Chem. Sci. 2017, 8, 405–410. [CrossRef]
- Stein, M.; Winter, W.; Rieker, A. Hexakis (2, 6-di-tert-butyl-4-biphenylyl) ethane—The First Unbridged Hexaarylethane. Angew. Chem. Int. Ed. 1978, 17, 692–694. [CrossRef]
- Kahr, B.; Van Engen, D.; Mislow, K. Length of the ethane bond in hexaphenylethane and its derivatives. J. Am. Chem. Soc. 1986, 108, 8305–8307. [CrossRef]
- Phipps, M.J.; Fox, T.; Tautermann, C.S.; Skylaris, C.K. Energy decomposition analysis approaches and their evaluation on prototypical protein–drug interaction patterns. Chem. Soc. Rev. 2015, 44, 3177–3211. [CrossRef] [PubMed]
- Gimferrer, M.; Danes, S.; Andrada, D.M.; Salvador, P. Merging the energy decomposition analysis with the interacting quantum atoms approach. J. Chem. Theory Comput. 2023, 19, 3469–3485. [CrossRef]
- Oestereich, T.; Tonner-Zech, R.; Westermayr, J. Decoding energy decomposition analysis: Machine-learned Insights on the impact of the density functional on the bonding analysis. J. Comput. Chem. 2024, 45, 368–376. [CrossRef] [PubMed]
- Bader, R.F. Atoms in molecules. Acc. Chem. Res. 1985, 18, 9–15. [CrossRef]
- Blanco, M.; Martín Pendás, A.; Francisco, E. Interacting quantum atoms: a correlated energy decomposition scheme based on the quantum theory of atoms in molecules. J. Chem. Theory Comput. 2005, 1, 1096–1109. [CrossRef]
- Francisco, E.; Martín Pendás, A.; Blanco, M. A molecular energy decomposition scheme for atoms in molecules. J. Chem. Theory Comput. 2006, 2, 90–102. [CrossRef] [PubMed]
- Martín Pendás, A.; Blanco, M.; Francisco, E. The nature of the hydrogen bond: A synthesis from the interacting quantum atoms picture. J. Chem. Phys. 2006, 125. [CrossRef] [PubMed]
- Duarte, L.J.; Alves, E.H.; Braga, A.A. Energetic Origins of the Hydrogen-Bond Redshift: IQA Partitioning of Normal Mode Force Constants. J. Phys. Chem. A 2025, 129, 6281–6288. [CrossRef]
- Badri, Z.; Foroutan-Nejad, C.; Kozelka, J.; Marek, R. On the non-classical contribution in lone-pair–π interaction: IQA perspective. Phys. Chem. Chem. Phys. 2015, 17, 26183–26190. [CrossRef]
- Alkorta, I.; Silva, A.F.; Popelier, P.L. An Interacting Quantum Atoms (IQA) and Relative Energy Gradient (REG) Study of the Halogen Bond with Explicit Analysis of Electron Correlation. Molecules 2020, 25, 2674. [CrossRef]
- Triestram, L.; Falcioni, F.; Popelier, P.L. Interacting Quantum Atoms and Multipolar Electrostatic Study of XH··· π Interactions. ACS omega 2023, 8, 34844–34851. [CrossRef] [PubMed]
- Hercigonja, M.; Milovanović, B.; Etinski, M.; Petković, M. Decorated crown ethers as selective ion traps: Solvent’s role in crown’s preference towards a specific ion. J. Mol. Liq. 2023, 381, 121791. [CrossRef]
- Tiana, D.; Francisco, E.; Blanco, M.; Macchi, P.; Sironi, A.; Martín Pendás, A. Bonding in classical and nonclassical transition metal carbonyls: the interacting quantum atoms perspective. J. Chem. Theory Comput. 2010, 6, 1064–1074. [CrossRef]
- Tiana, D.; Francisco, E.; Macchi, P.; Sironi, A.; Martín Pendás, A. An Interacting Quantum Atoms Analysis of the Metal–Metal Bond in [M2 (CO) 8] n Systems. J. Phys. Chem. A 2015, 119, 2153–2160. [CrossRef]
- Guevara-Vela, J.M.; Chávez-Calvillo, R.; García-Revilla, M.; Hernández-Trujillo, J.; Christiansen, O.; Francisco, E.; Martín Pendás, Á.; Rocha-Rinza, T. Hydrogen-Bond Cooperative Effects in Small Cyclic Water Clusters as Revealed by the Interacting Quantum Atoms Approach. Chem. Eur. J. 2013, 19, 14304–14315. [CrossRef]
- Guevara-Vela, J.M.; Romero-Montalvo, E.; Gómez, V.A.M.; Chávez-Calvillo, R.; García-Revilla, M.; Francisco, E.; Pendás, Á.M.; Rocha-Rinza, T. Hydrogen bond cooperativity and anticooperativity within the water hexamer. Phys. Chem. Chem. Phys. 2016, 18, 19557–19566. [CrossRef] [PubMed]
- Guevara-Vela, J.M.; Hess, K.; Rocha-Rinza, T.; Pendás, Á.M.; Flores-Álamo, M.; Moreno-Alcántar, G. Stronger-together: the cooperativity of aurophilic interactions. Chem. Commun. 2022, 58, 1398–1401. [CrossRef]
- Ahirwar, M.B.; Gadre, S.R.; Deshmukh, M.M. On the short-range nature of cooperativity in hydrogen-bonded large molecular clusters. J. Phys. Chem. A 2023, 127, 4394–4406. [CrossRef] [PubMed]
- Cremer, D.; Kraka, E. Chemical bonds without bonding electron density—does the difference electron-density analysis suffice for a description of the chemical bond? Angew. Chem. Int. Ed. 1984, 23, 627–628. [CrossRef]
- Bader, R.F.W. Theory of atoms in molecules, 1995.
- Guevara-Vela, J.M.; Francisco, E.; Rocha-Rinza, T.; Martin Pendas, A. Interacting quantum atoms—a review. Molecules 2020, 25, 4028. [CrossRef]
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A.J.; Yang, W. Revealing noncovalent interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [CrossRef]
- Neese, F. Software update: The ORCA program system—version 6.0. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2025, 15, e70019. [CrossRef]
- Adamo, C.; Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 1999, 110, 6158–6170. [CrossRef]
- Becke, A.D.; Johnson, E.R. A density-functional model of the dispersion interaction. J. Chem. Phys. 2005, 123. [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132. [CrossRef]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [CrossRef]
- Vahtras, O.; Almlöf, J.; Feyereisen, M. Integral approximations for LCAO-SCF calculations. Chem. Phys. Lett. 1993, 213, 514–518. [CrossRef]
- Neese, F. An improvement of the resolution of the identity approximation for the formation of the Coulomb matrix. J. Comput. Chem. 2003, 24, 1740–1747. [CrossRef]
- Neese, F.; Wennmohs, F.; Hansen, A.; Becker, U. Efficient, approximate and parallel Hartree–Fock and hybrid DFT calculations. A ‘chain-of-spheres’ algorithm for the Hartree–Fock exchange. Chem. Phys. 2009, 356, 98–109. [CrossRef]
- Helmich-Paris, B.; de Souza, B.; Neese, F.; Izsák, R. An improved chain of spheres for exchange algorithm. J. Chem. Phys. 2021, 155. [CrossRef]
- Weigend, F. Accurate Coulomb-fitting basis sets for H to Rn. Phys. Chem. Chem. Phys. 2006, 8, 1057–1065. [CrossRef] [PubMed]
- Keith, T.A. AIMAll (Version 19.02.13). TK Gristmill Software, Overland Park KS, USA, 2019 (aim.tkgristmill.com).
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [CrossRef] [PubMed]
- Lu, T. A comprehensive electron wavefunction analysis toolbox for chemists, Multiwfn. J. Chem. Phys. 2024, 161. [CrossRef] [PubMed]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [CrossRef] [PubMed]
- Sowlati-Hashjin, S.; Šadek, V.; Sadjadi, S.; Karttunen, M.; Martín-Pendás, A.; Foroutan-Nejad, C. Collective interactions among organometallics are exotic bonds hidden on lab shelves. Nat. Commun. 2022, 13. [CrossRef] [PubMed]
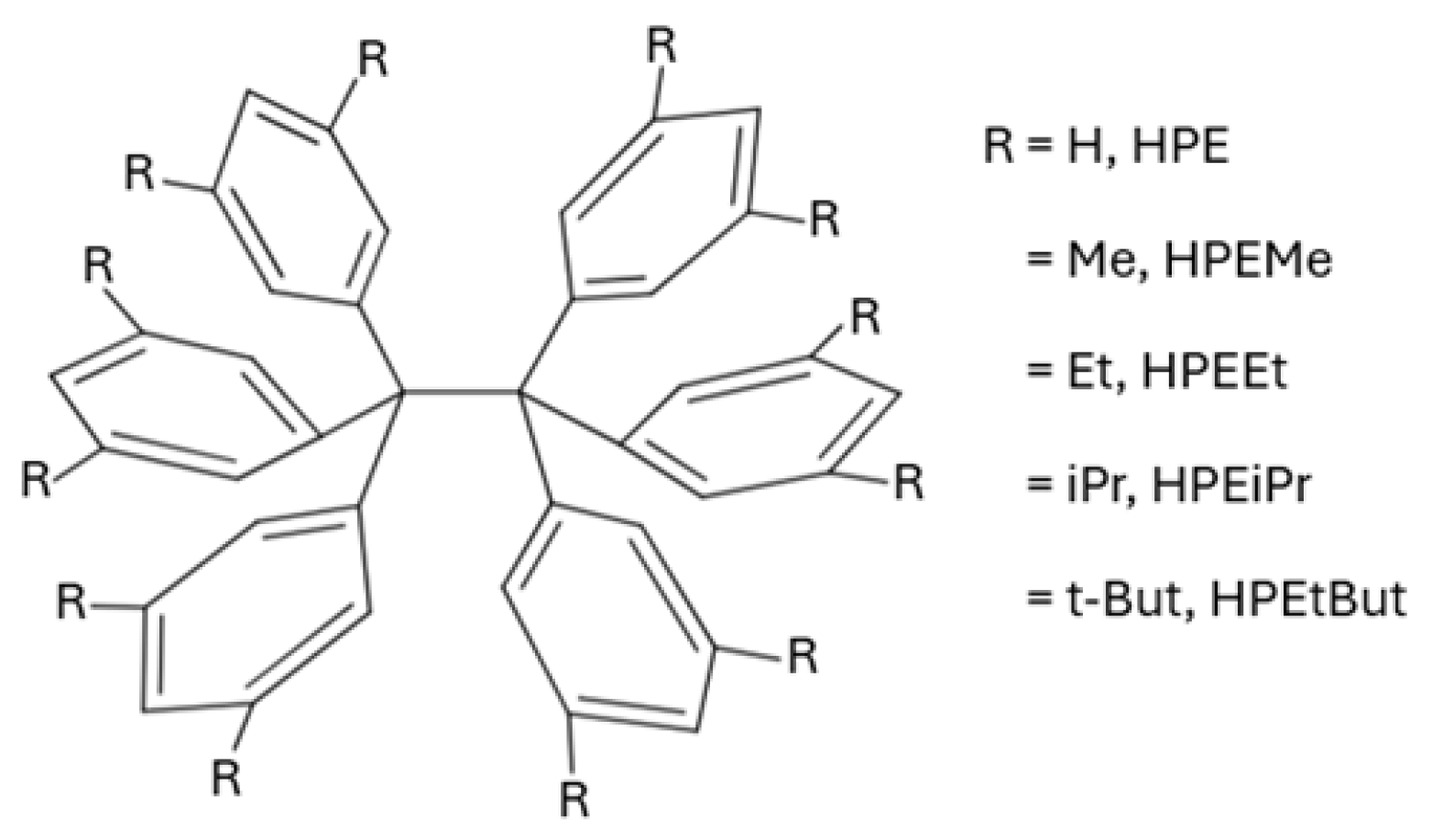
| Molecule | /kcal·mol−1 | /kcal·mol−1 | |
| Ethane | −92.32 | −81.42 | |
| HPE | −11.44 | 6.86 | |
| HPEMe | −15.90 | 2.56 | |
| HPEEt | −23.26 | −2.98 | |
| HPEiPr | −31.42 | −11.68 | |
| HPEtBut | −41.28 | −18.74 | |
| Molecule | ||||||
| H3C–CH3 | 1.006 | 0.244 | −0.5597 | −0.196 | 1.519 | −99.72 |
| HPE | 0.853 | 0.182 | −0.3325 | −0.121 | 1.685 | −14.85 |
| HPEMe | 0.841 | 0.176 | −0.3130 | −0.115 | 1.703 | −20.41 |
| HPEEt | 0.860 | 0.186 | −0.3451 | −0.125 | 1.674 | −26.86 |
| HPEiPr | 0.860 | 0.188 | −0.3531 | −0.128 | 1.667 | −36.49 |
| HPEtBut | 0.865 | 0.188 | −0.3528 | −0.127 | 1.669 | −45.64 |
| Ethane | |||
| IQA partition | Dispersion correction | IQA-D | |
| Interaction energy | −196.273 (−172.737) | −0.921 | −197.194 |
| Deformation energy | 97.248 | 0.092 | −97.340 |
| Formation energy | −99.026 | −0.829 | −99.854 |
| Hexaphenylethane | |||
| IQA partition | Dispersion correction | IQA-D | |
| Interaction energy | −291.233 (−147.689) | −20.192 | −311.425 |
| Deformation energy | 300.870 | −0.691 | 300.179 |
| Formation energy | 9.637 | −20.884 | −11.246 |
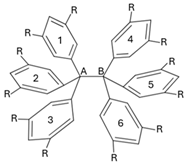 | |||
| HPE | |||
| Groups | IQA partition | Dispersion correction | IQA-D |
| A–4 | −3.395 | −0.551 | −3.945 |
| A–5 | −3.387 | −0.551 | −3.938 |
| A–6 | −3.417 | −0.553 | −3.970 |
| B–1 | −3.407 | −0.552 | −3.960 |
| B–2 | −3.371 | −0.549 | −3.921 |
| B–3 | −3.416 | −0.553 | −3.969 |
| Total | −20.393 | −3.309 | −23.703 |
| HPEMe | |||
| Groups | IQA partition | Dispersion correction | IQA-D |
| A–4 | −3.391 | −0.574 | −3.965 |
| A–5 | −3.372 | −0.574 | −3.946 |
| A–6 | −3.471 | −0.581 | −4.052 |
| B–1 | −3.440 | −0.580 | −4.020 |
| B–2 | −3.367 | −0.573 | −3.939 |
| B–3 | −3.470 | −0.581 | −4.051 |
| Total | −20.511 | −3.462 | −23.972 |
| HPEEt | |||
| Groups | IQA partition | Dispersion correction | IQA-D |
| A–4 | −3.399 | −0.592 | −3.991 |
| A–5 | −3.350 | −0.587 | −3.937 |
| A–6 | −3.460 | −0.597 | −4.057 |
| B–1 | −3.436 | −0.599 | −4.035 |
| B–2 | −3.363 | −0.591 | −3.954 |
| B–3 | −3.443 | −0.595 | −4.035 |
| Totla | −20.451 | −3.560 | −24.011 |
| HPEiPr | |||
| Groups | IQA partition | Dispersion correction | IQA-D |
| A–4 | −3.363 | −0.640 | −4.002 |
| A–5 | −3.443 | −0.603 | −4.046 |
| A–6 | −3.436 | −0.613 | −4.049 |
| B–1 | −3.443 | −0.615 | −4.058 |
| B–2 | −3.460 | −0.604 | −4.063 |
| B–3 | −3.350 | −0.585 | −3.935 |
| Total | −20.495 | −3.659 | −24.154 |
| HPEtBut | |||
| Groups | IQA partition | Dispersion correction | IQA-D |
| A–4 | −3.428 | −0.638 | −4.067 |
| A–5 | −3.280 | −0.630 | −3.910 |
| A–6 | −3.470 | −0.641 | −4.111 |
| B–1 | −3.429 | −0.638 | −4.067 |
| B–2 | −3.279 | −0.630 | −3.910 |
| B–3 | −3.469 | −0.641 | −4.110 |
| Total | −20.356 | −3.819 | −24.175 |
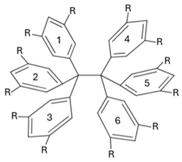 | |||
| Phenyl groups | IQA partition | Dispersion correction | IQA-D |
| 1–4 | −28.177 | −3.586 | −31.763 |
| 1–5 | −10.278 | −1.372 | −11.650 |
| 1–6 | −0.225 | −0.587 | −0.813 |
| 2–4 | −0.229 | −0.585 | −0.814 |
| 2–5 | −28.179 | −3.639 | −31.818 |
| 2–6 | −10.315 | −1.368 | −11.683 |
| 3–4 | −10.257 | −1.367 | −11.624 |
| 3–5 | −0.230 | −0.589 | −0.819 |
| 3–6 | −28.039 | −3.636 | −31.675 |
| Total | −115.928 | −16.731 | −132.659 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




