Submitted:
01 October 2025
Posted:
01 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
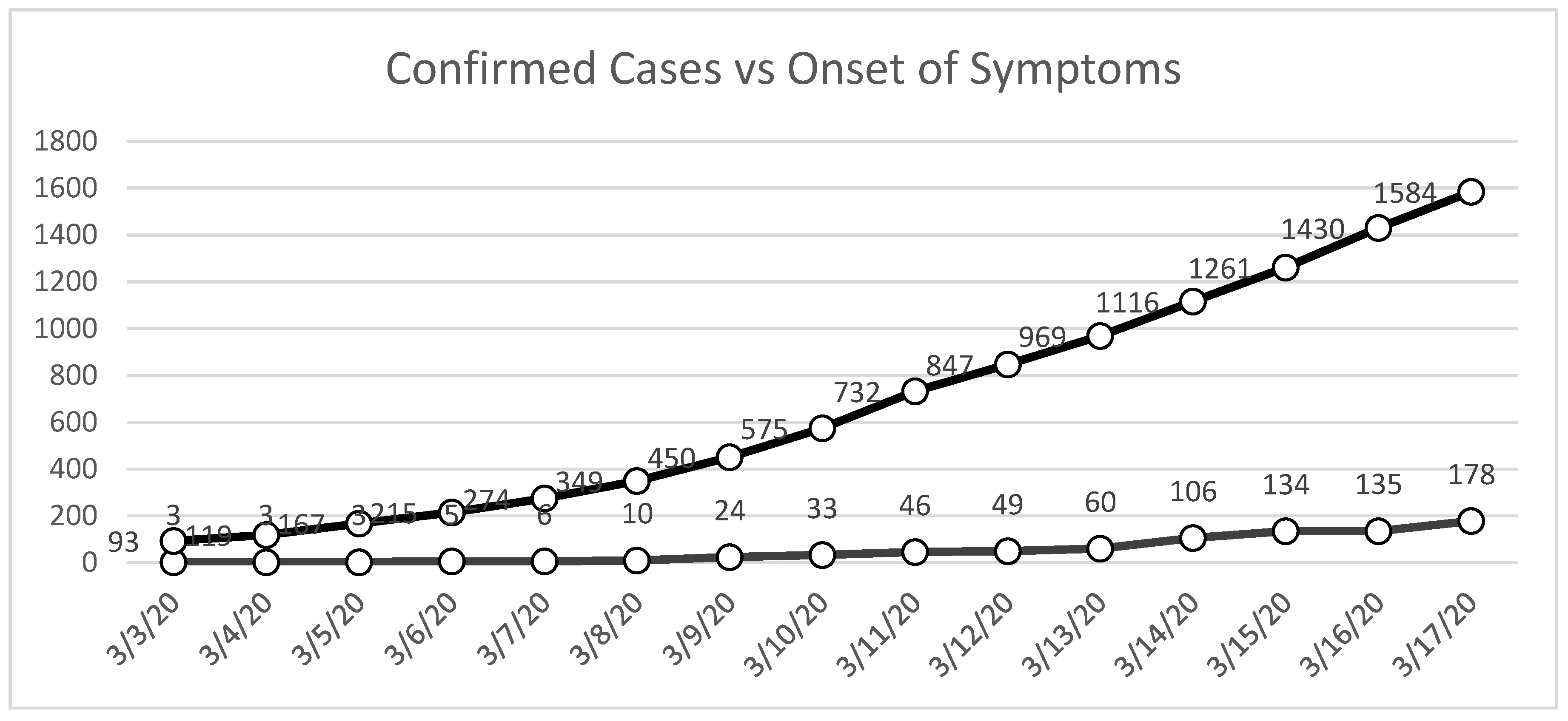
2. An Interoperability Problem
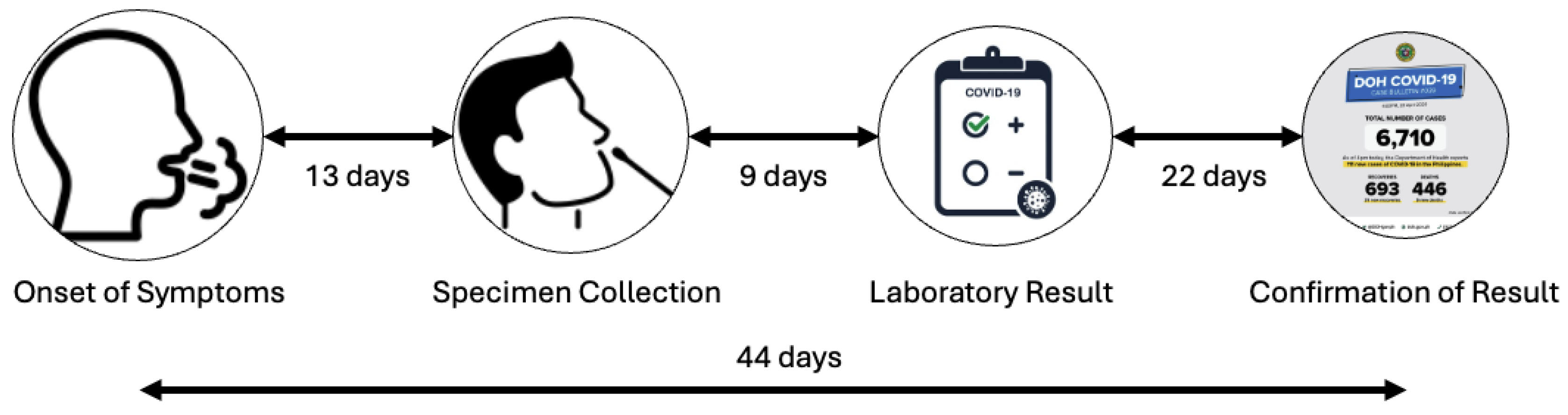
- Symptom onset to specimen collection: On average, 13 days passed between the start of symptoms and the collection of a COVID-19 specimen. This delay was partly due to limited awareness of testing protocols among patients and healthcare providers, as well as logistical barriers to reaching accredited testing centers.
- Specimen collection to release of results: The time between specimen collection, laboratory testing, and results release was about 9 days. This delay mainly resulted from the significant shortage of accredited COVID-19 testing labs during the early months of the pandemic, when testing capacity was focused primarily on the Research Institute for Tropical Medicine (RITM) and a few regional hubs.
- Result release to official case confirmation: The most significant bottleneck was likely the time between laboratory confirmation and official DOH case reporting, which averaged 22 days. This delay was not due to a lack of information systems but rather the need to validate patient records, reconcile laboratory results with case investigation forms, and start contact tracing. These tasks caused considerable administrative work, leading to further delays.
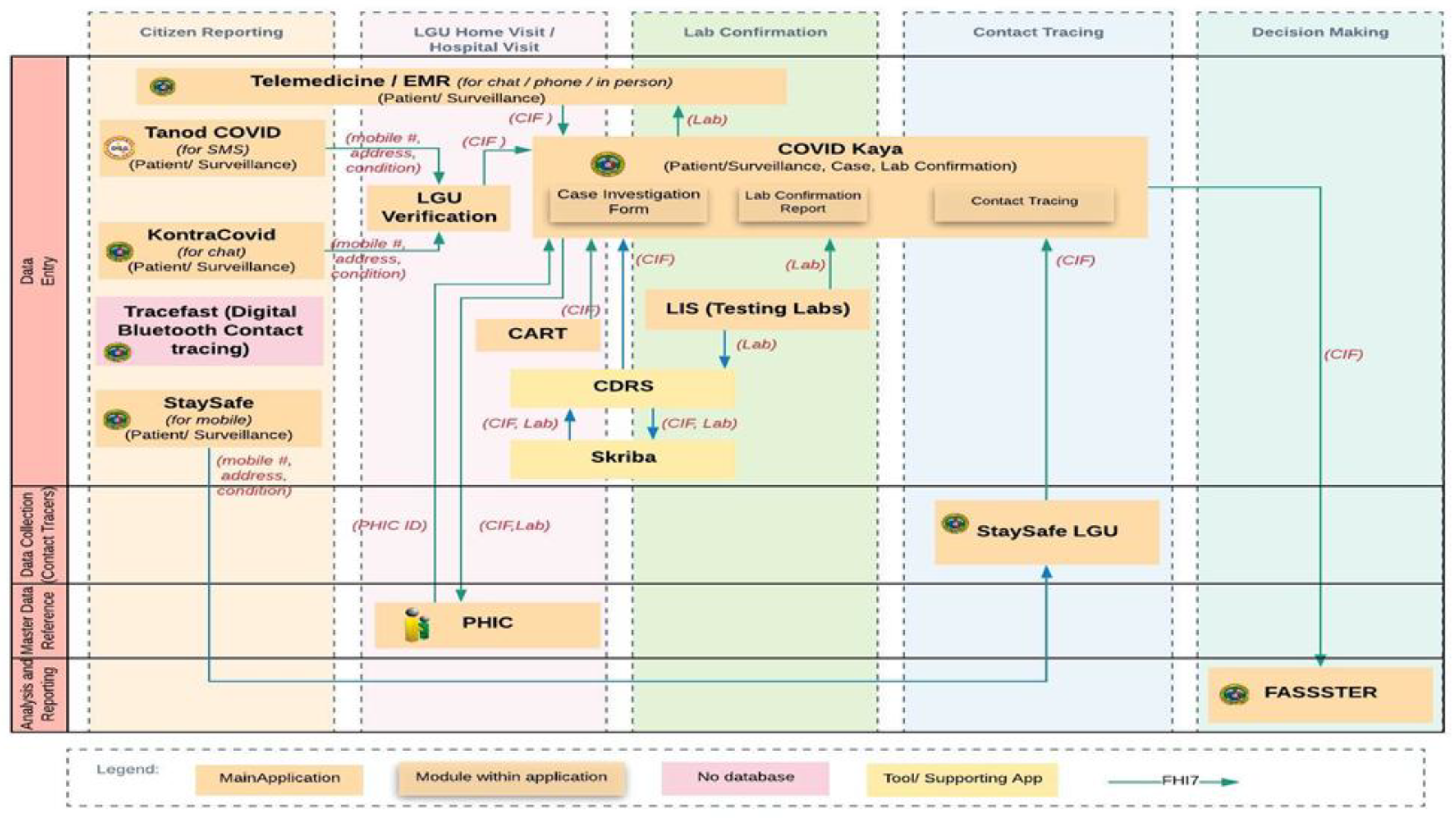
The COVID-19 Data Ecosystem
| System | Role | Description |
|---|---|---|
| COVID Kaya | Case reporting | Primary DOH platform for case investigation and reporting |
| EMR | Facility-level record | Electronic Medical Records used by hospitals and clinics |
| Tanod COVID | Community-level monitoring | Barangay-level surveillance tool for suspected cases |
| Kontra COVID | Public engagement | Citizen reporting and information platform |
| StaySafe | Contact tracing | Mobile application for exposure notification and symptom reporting |
| CART | Case assessment | DOH tool for rapid lab result generation |
| CDRS | Central data repository | Unified reporting platform for laboratory results |
| Skriba | Data entry support | Facilitates digitization of paper-based records |
| PHIC | Insurance-linked data | PhilHealth database for claims and patient records |
| LIS | Laboratory Information System | Manages specimen tracking and laboratory workflows |
| FASSSTER | Modeling and analytics | Forecasting tool for case trajectories and projections |
| StaySafe LGU | Localized variant of StaySafe | Customized deployments for local governments |
3. Interventions
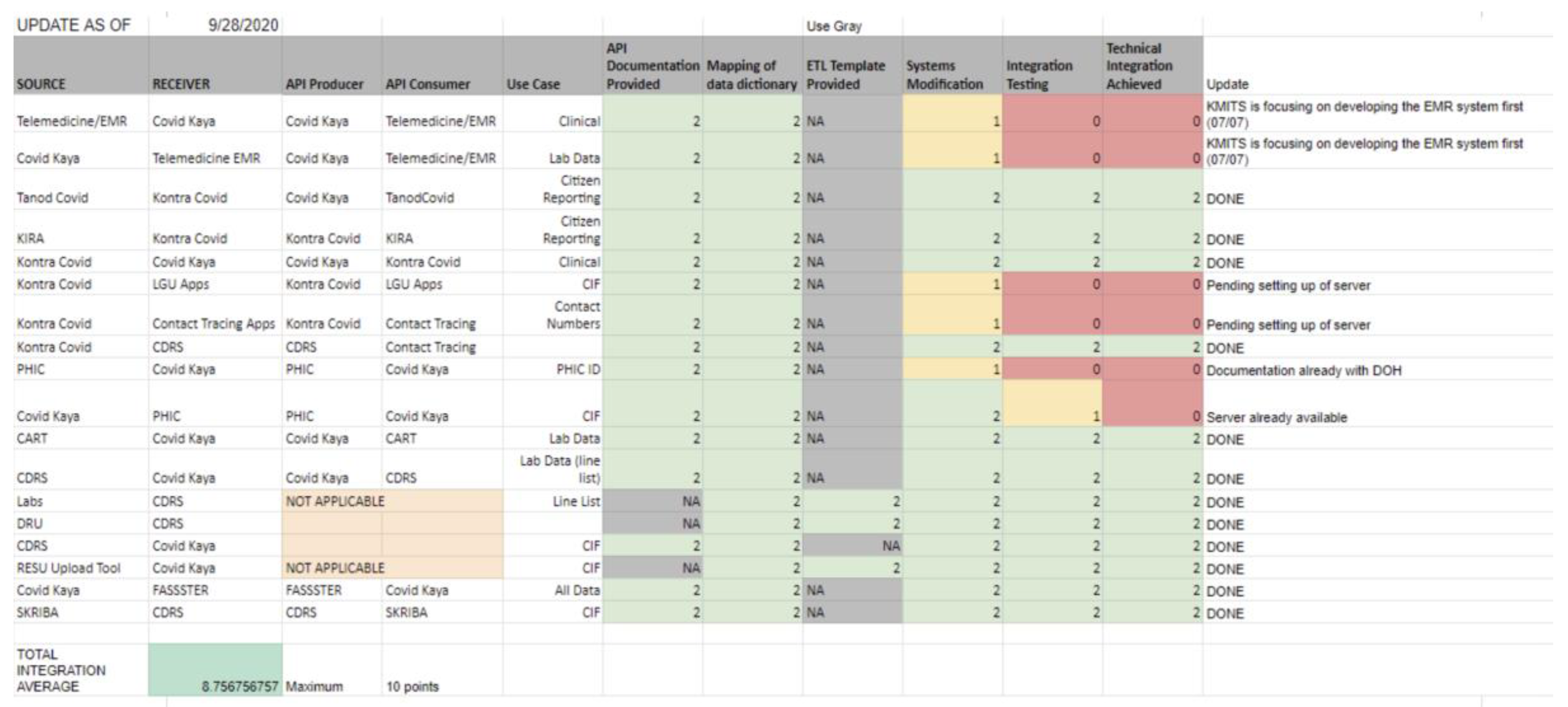
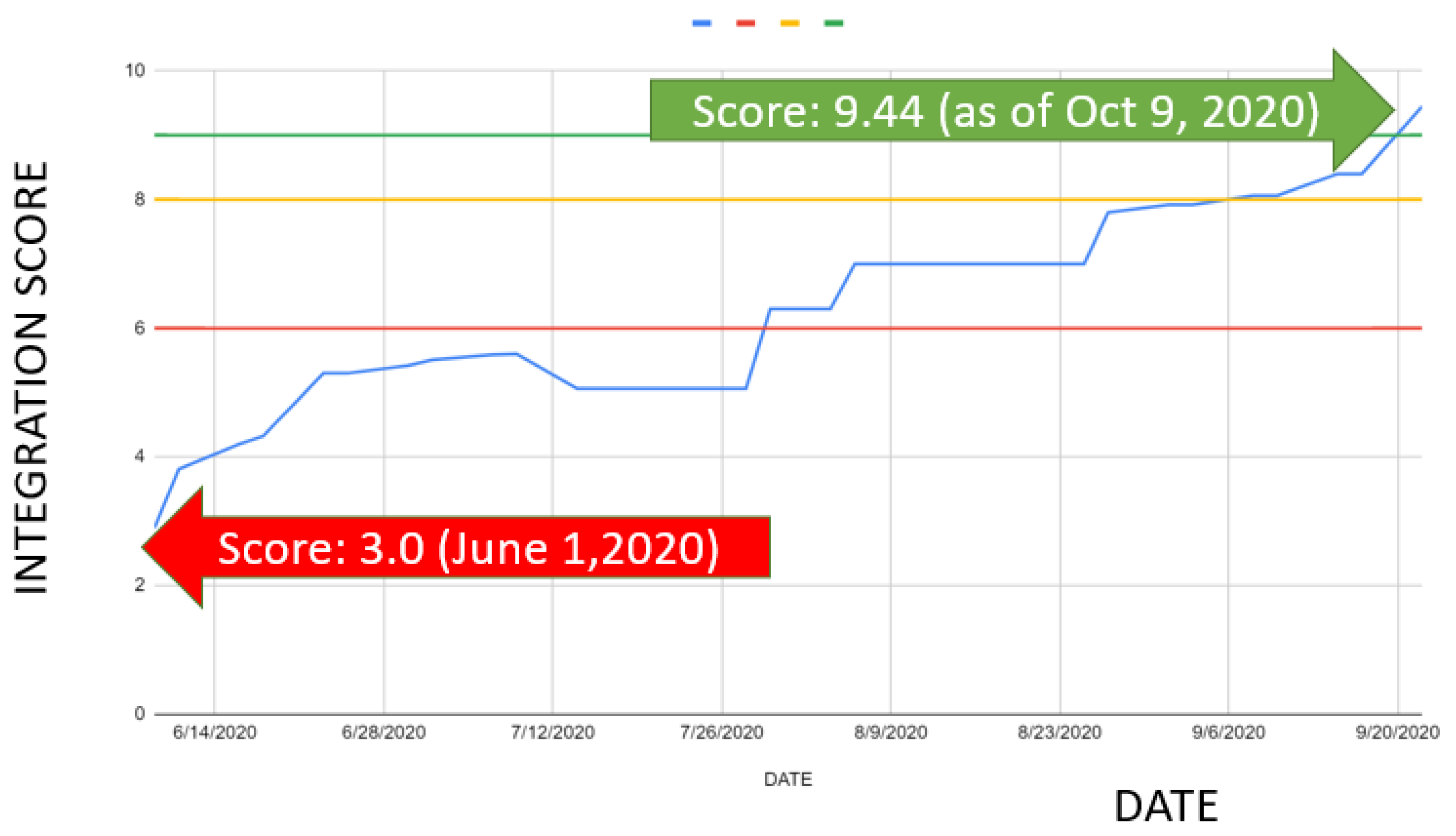
4. Monitoring Progress in Interoperability
- 1.
- API Documentation Provided.
- 2.
- Mapping of Data Dictionary.
- 3.
- ETL Template Provided.
- 4.
- System Modification.
- 5.
- Integration Testing.
- 6.
- Technical Integration Achieved.
5. Results and Discussion
6. Lessons and Recommendations
- 1.
- Interoperability as Both a Governance and Technical Challenge
- 2.
- Adopt a “Minimum, Then Mature” Strategy
- 3.
- The Value of Scorecards in Monitoring Progress
- 4.
- Designing for Heterogeneity and Low-Resource Settings
- 5.
- The Strategic Role of Interoperability Laboratories
Acknowledgements
Conflicts of Interest
References
- Vallejo, B. Policy responses and government science advice for the COVID 19 pandemic in the Philippines: January to April 2020; Progress in Disaster Science, 2020. [Google Scholar]
- Uy, F. From Blueprint to Breakthrough: Digital Transformation in the Philippines’ Pandemic Response Rapid Enterprise Architecture Deployment in National Crisis Management; The Open Group, 2025. [Google Scholar]
- Zuniga, P. C. Establishing a Regional Digital Health Interoperability Lab in the Asia-Pacific Region: Experiences and Recommendations. In Leveraging Data Science for Global Health; Springer, 2020. [Google Scholar]
- About SIL-Asia. Retrieved from SIL-Asia website. 2021. Available online: www.sil-asia.org.
- HL7 FHIR. Retrieved from HL7 FHIR website. n.d. Available online: https://www.hl7.org/fhir/.
- Philippine COVID 19 Implementation Guide. Retrieved from Philippine COVID 19 Implementation Guide. 2020. Available online: https://pczuniga.notion.site/Philippine-COVID-19-Implementation-Guide-b62d46afbfb14ee797e6cf9f14b05e1d.
- 2020 Revised IRR for RA 11332. The 2020 Implementing Rules and Regulations for RA 11332; Philippine Government, 2020. [Google Scholar]
- Kasunic, M. Measuring Systems Interoperability, Challenges and Opportunities. 2001, 40. [Google Scholar] [CrossRef]
| Stage | Baseline Time Interval (March 2020) | Time Interval (Oct 2020) |
|---|---|---|
| Onset of Symptoms to Specimen Collection | 13 days | 1 day |
| Specimen Collection to Release of Results | 9 days | 2 days |
| Release of Results to Confirmation of Case | 22 days | 3 days |
| Total | 44 days | 6 days |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




