Submitted:
29 September 2025
Posted:
30 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
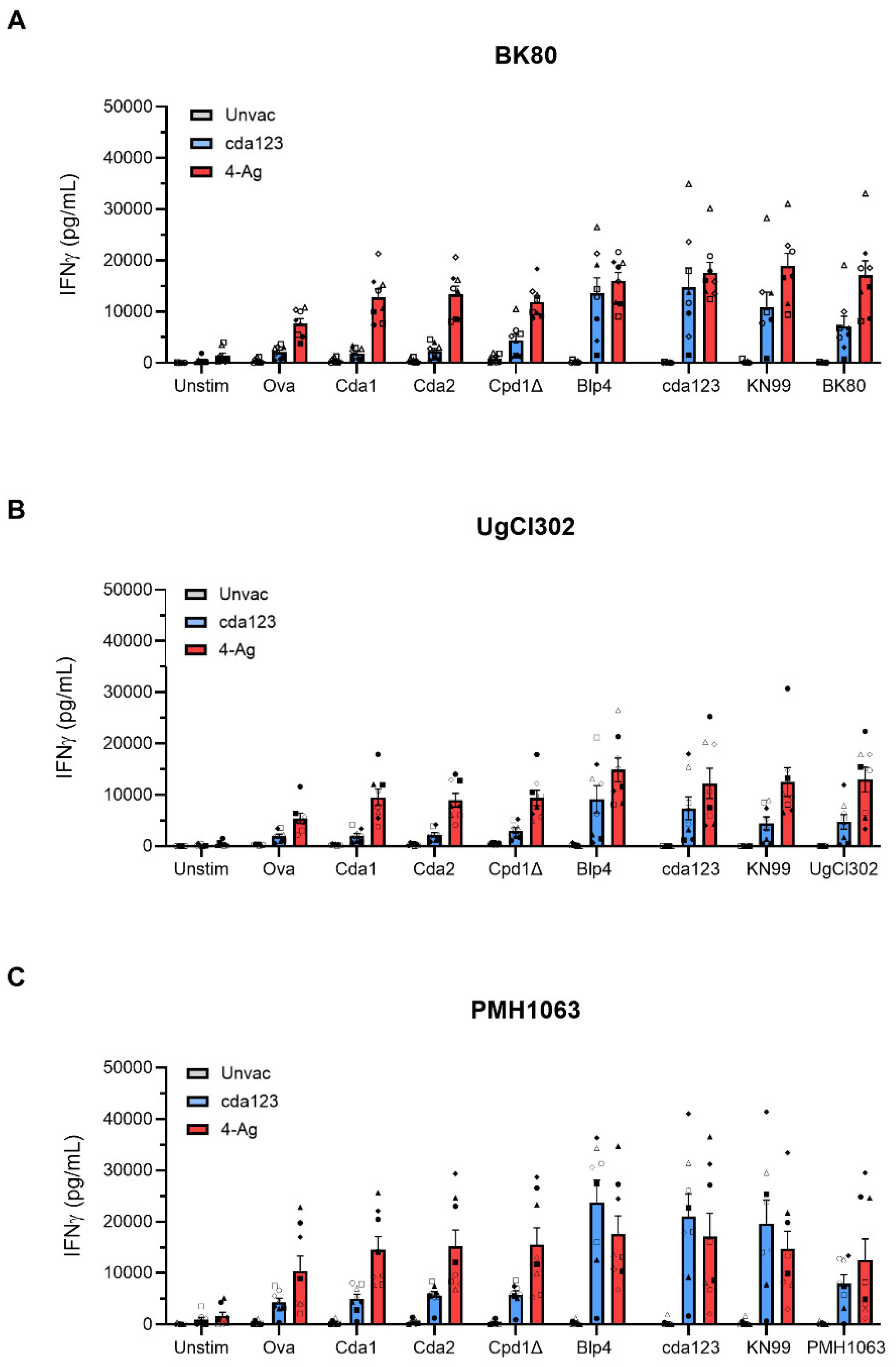
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Cda | chitin deacetylase |
| cda123 | cda1Δ2Δ3Δ avirulent Cryptococcus strain |
| CAF01 | Cationic Adjuvant Formulation 01 |
| 4-Ag | Four antigen Vaccine |
| Cpd1Δ | carboxypeptidase 1 trimmed of its region of human homology |
| Blp4 | Barwin-like domain protein 4 |
| YPD | yeast-peptone-dextrose |
| DPI | Days post infection |
References
- Meya, D.B.; Williamson, P.R. Cryptococcal Disease in Diverse Hosts. N Engl J Med 2024, 390, 1597-1610. [CrossRef]
- Rajasingham, R.; Govender, N.P.; Jordan, A.; Loyse, A.; Shroufi, A.; Denning, D.W.; Meya, D.B.; Chiller, T.M.; Boulware, D.R. The global burden of HIV-associated cryptococcal infection in adults in 2020: a modelling analysis. The Lancet Infectious Diseases 2022, 22, 1748-1755.
- Hagen, F.; Khayhan, K.; Theelen, B.; Kolecka, A.; Polacheck, I.; Sionov, E.; Falk, R.; Parnmen, S.; Lumbsch, H.T.; Boekhout, T. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet Biol 2015, 78, 16-48. [CrossRef]
- Kwon-Chung, K.J.; Bennett, J.E.; Wickes, B.L.; Meyer, W.; Cuomo, C.A.; Wollenburg, K.R.; Bicanic, T.A.; Castaneda, E.; Chang, Y.C.; Chen, J.; et al. The Case for Adopting the “Species Complex” Nomenclature for the Etiologic Agents of Cryptococcosis. mSphere 2017, 2. [CrossRef]
- Desjardins, C.A.; Giamberardino, C.; Sykes, S.M.; Yu, C.H.; Tenor, J.L.; Chen, Y.; Yang, T.; Jones, A.M.; Sun, S.; Haverkamp, M.R.; et al. Population genomics and the evolution of virulence in the fungal pathogen Cryptococcus neoformans. Genome Res 2017, 27, 1207-1219. [CrossRef]
- Litvintseva, A.P.; Thakur, R.; Vilgalys, R.; Mitchell, T.G. Multilocus Sequence Typing Reveals Three Genetic Subpopulations of Cryptococcus neoformans var. grubii (Serotype A), Including a Unique Population in Botswana. Genetics 2006, 172, 2223-2238. [CrossRef]
- Jackson, K.M.; Kono, T.J.Y.; Betancourt, J.J.; Wang, Y.; Kabbale, K.D.; Ding, M.; Kezh, P.; Ha, G.; Yoder, J.M.; Fulton, S.R.; et al. Single nucleotide polymorphisms are associated with strain-specific virulence differences among clinical isolates of Cryptococcus neoformans. Nature Communications 2024, 15, 10491. [CrossRef]
- Kassaza, K.; Wasswa, F.; Nielsen, K.; Bazira, J. Cryptococcus neoformans Genotypic Diversity and Disease Outcome among HIV Patients in Africa. Journal of Fungi 2022, 8, 734.
- Oliveira, L.V.N.; Wang, R.; Specht, C.A.; Levitz, S.M. Vaccines for human fungal diseases: close but still a long way to go. NPJ Vaccines 2021, 6, 33. [CrossRef]
- Specht, C.A.; Wang, R.; Oliveira, L.V.N.; Hester, M.M.; Gomez, C.; Mou, Z.; Carlson, D.; Lee, C.K.; Hole, C.R.; Lam, W.C.; et al. Immunological correlates of protection mediated by a whole organism, Cryptococcus neoformans, vaccine deficient in chitosan. mBio 2024, 15, e0174624. [CrossRef]
- Upadhya, R.; Lam, W.C.; Maybruck, B.; Specht, C.A.; Levitz, S.M.; Lodge, J.K. Induction of Protective Immunity to Cryptococcal Infection in Mice by a Heat-Killed, Chitosan-Deficient Strain of Cryptococcus neoformans. mBio 2016, 7, e00547-00516. [CrossRef]
- Wang, R.; Oliveira, L.V.N.; Hester, M.M.; Carlson, D.; Christensen, D.; Specht, C.A.; Levitz, S.M. Protection against experimental cryptococcosis elicited by Cationic Adjuvant Formulation 01-adjuvanted subunit vaccines. PLOS Pathogens 2024, 20, e1012220. [CrossRef]
- Hester, M.M.; Lee, C.K.; Abraham, A.; Khoshkenar, P.; Ostroff, G.R.; Levitz, S.M.; Specht, C.A. Protection of mice against experimental cryptococcosis using glucan particle-based vaccines containing novel recombinant antigens. Vaccine 2020, 38, 620-626. [CrossRef]
- Specht, C.A.; Lee, C.K.; Huang, H.; Hester, M.M.; Liu, J.; Luckie, B.A.; Torres Santana, M.A.; Mirza, Z.; Khoshkenar, P.; Abraham, A.; et al. Vaccination with Recombinant Cryptococcus Proteins in Glucan Particles Protects Mice against Cryptococcosis in a Manner Dependent upon Mouse Strain and Cryptococcal Species. MBio 2017, 8, e01872-01817. [CrossRef]
- Pedersen, G.K.; Andersen, P.; Christensen, D. Immunocorrelates of CAF family adjuvants. Semin Immunol 2018, 39, 4-13. [CrossRef]
- Nielsen, K.; Cox, G.M.; Wang, P.; Toffaletti, D.L.; Perfect, J.R.; Heitman, J. Sexual cycle of Cryptococcus neoformans var. grubii and virulence of congenic a and alpha isolates. Infection and immunity 2003, 71, 4831-4841.
- Janbon, G.; Ormerod, K.L.; Paulet, D.; Byrnes, E.J., 3rd; Yadav, V.; Chatterjee, G.; Mullapudi, N.; Hon, C.C.; Billmyre, R.B.; Brunel, F.; et al. Analysis of the genome and transcriptome of Cryptococcus neoformans var. grubii reveals complex RNA expression and microevolution leading to virulence attenuation. PLoS genetics 2014, 10, e1004261. [CrossRef]
- Litvintseva, A.P.; Mitchell, T.G. Most environmental isolates of Cryptococcus neoformans var. grubii (serotype A) are not lethal for mice. Infection and immunity 2009, 77, 3188-3195. [CrossRef]
- Mukaremera, L.; McDonald, T.R.; Nielsen, J.N.; Molenaar, C.J.; Akampurira, A.; Schutz, C.; Taseera, K.; Muzoora, C.; Meintjes, G.; Meya, D.B.; et al. The Mouse Inhalation Model of Cryptococcus neoformans Infection Recapitulates Strain Virulence in Humans and Shows that Closely Related Strains Can Possess Differential Virulence. Infect Immun 2019, 87. [CrossRef]
- Thanh, L.T.; Toffaletti, D.L.; Tenor, J.L.; Giamberardino, C.; Sempowski, G.D.; Asfaw, Y.; Phan, H.T.; Van Duong, A.; Trinh, N.M.; Thwaites, G.E.; et al. Assessing the virulence of Cryptococcus neoformans causing meningitis in HIV infected and uninfected patients in Vietnam. Med Mycol 2020, 58, 1149-1161. [CrossRef]
- Wiesner, D.L.; Moskalenko, O.; Corcoran, J.M.; McDonald, T.; Rolfes, M.A.; Meya, D.B.; Kajumbula, H.; Kambugu, A.; Bohjanen, P.R.; Knight, J.F.; et al. Cryptococcal genotype influences immunologic response and human clinical outcome after meningitis. mBio 2012, 3. [CrossRef]
- Ashton, P.M.; Thanh, L.T.; Trieu, P.H.; Van Anh, D.; Trinh, N.M.; Beardsley, J.; Kibengo, F.; Chierakul, W.; Dance, D.A.B.; Rattanavong, S.; et al. Three phylogenetic groups have driven the recent population expansion of Cryptococcus neoformans. Nature Communications 2019, 10, 2035. [CrossRef]
- Boulware, D.R.; von Hohenberg, M.; Rolfes, M.A.; Bahr, N.C.; Rhein, J.; Akampurira, A.; Williams, D.A.; Taseera, K.; Schutz, C.; McDonald, T.; et al. Human Immune Response Varies by the Degree of Relative Cryptococcal Antigen Shedding. Open Forum Infectious Diseases 2015, 3. [CrossRef]
- Nielsen, K.; Marra, R.E.; Hagen, F.; Boekhout, T.; Mitchell, T.G.; Cox, G.M.; Heitman, J. Interaction between genetic background and the mating-type locus in Cryptococcus neoformans virulence potential. Genetics 2005, 171, 975-983. [CrossRef]
- Yu, C.-H.; Sephton-Clark, P.; Tenor, J.L.; Toffaletti, D.L.; Giamberardino, C.; Haverkamp, M.; Cuomo, C.A.; Perfect, J.R. Gene Expression of Diverse Cryptococcus Isolates during Infection of the Human Central Nervous System. mBio 2021, 12, e02313-02321, doi:doi:10.1128/mBio.02313-21.
- Specht, C.A.; Wang, R.; Oliveira, L.V.N.; Hester, M.M.; Gomez, C.; Mou, Z.; Carlson, D.; Lee, C.K.; Hole, C.R.; Lam, W.C.; et al. Immunological correlates of protection mediated by a whole organism, Cryptococcus neoformans, vaccine deficient in chitosan. mBio 2024, e0174624. [CrossRef]
- Oliveira, L.V.N.; Hargarten, J.C.; Wang, R.; Carlson, D.; Park, Y.-D.; Specht, C.A.; Williamson, P.R.; Levitz, S.M. Peripheral blood CD4+ and CD8+ T cell responses to Cryptococcus candidate vaccine antigens in human subjects with and without cryptococcosis. Journal of Infection 2025, 91, 106521. [CrossRef]
- Wang, R.; Oliveira, L.V.N.; Lourenco, D.; Gomez, C.L.; Lee, C.K.; Hester, M.M.; Mou, Z.; Ostroff, G.R.; Specht, C.A.; Levitz, S.M. Immunological correlates of protection following vaccination with glucan particles containing Cryptococcus neoformans chitin deacetylases. NPJ Vaccines 2023, 8, 6. [CrossRef]
- Chen, Y.; Litvintseva, A.P.; Frazzitta, A.E.; Haverkamp, M.R.; Wang, L.; Fang, C.; Muthoga, C.; Mitchell, T.G.; Perfect, J.R. Comparative analyses of clinical and environmental populations of Cryptococcus neoformans in Botswana. Molecular Ecology 2015, 24, 3559-3571. [CrossRef]
- Coelho, M.A.; David-Palma, M.; Aylward, J.; Pham, N.Q.; Visagie, C.M.; Fuchs, T.; Yilmaz, N.; Roets, F.; Sun, S.; Taylor, J.W.; et al. Decoding Cryptococcus: From African biodiversity to worldwide prevalence. PLOS Pathogens 2025, 21, e1012876. [CrossRef]
- Ferreira-Paim, K.; Andrade-Silva, L.; Fonseca, F.M.; Ferreira, T.B.; Mora, D.J.; Andrade-Silva, J.; Khan, A.; Dao, A.; Reis, E.C.; Almeida, M.T.G.; et al. MLST-Based Population Genetic Analysis in a Global Context Reveals Clonality amongst Cryptococcus neoformans var. grubii VNI Isolates from HIV Patients in Southeastern Brazil. PLOS Neglected Tropical Diseases 2017, 11, e0005223. [CrossRef]
- Day, J.N.; Qihui, S.; Thanh, L.T.; Trieu, P.H.; Van, A.D.; Thu, N.H.; Chau, T.T.H.; Lan, N.P.H.; Chau, N.V.V.; Ashton, P.M.; et al. Comparative genomics of Cryptococcus neoformans var. grubii associated with meningitis in HIV infected and uninfected patients in Vietnam. PLOS Neglected Tropical Diseases 2017, 11, e0005628. [CrossRef]
- Van Dyke, M.C.C.; Chaturvedi, A.K.; Hardison, S.E.; Leopold Wager, C.M.; Castro-Lopez, N.; Hole, C.R.; Wozniak, K.L.; Wormley, F.L. Induction of Broad-Spectrum Protective Immunity against Disparate Cryptococcus Serotypes. Frontiers in Immunology 2017, 8, 1-16. [CrossRef]
- Yauch, L.E.; Lam, J.S.; Levitz, S.M. Direct inhibition of T-cell responses by the Cryptococcus capsular polysaccharide glucuronoxylomannan. PLoS Pathog 2006, 2, e120. [CrossRef]
- Kelly, R.M.; Chen, J.; Yauch, L.E.; Rottman, J.B.; Levitz, S.M. Opsonic requirements for dendritic cell-mediated responses to Cryptococcus neoformans. Infect Immun 2005, 73, 592-598.
- Wormley, F.L., Jr.; Perfect, J.R.; Steele, C.; Cox, G.M. Protection against cryptococcosis by using a murine gamma interferon-producing Cryptococcus neoformans strain. Infection and immunity 2007, 75, 1453-1462. [CrossRef]
- Jarvis, J.N.; Meintjes, G.; Rebe, K.; Williams, G.N.; Bicanic, T.; Williams, A.; Schutz, C.; Bekker, L.G.; Wood, R.; Harrison, T.S. Adjunctive interferon-gamma immunotherapy for the treatment of HIV-associated cryptococcal meningitis: a randomized controlled trial. AIDS 2012, 26, 1105-1113. [CrossRef]
- Pappas, P.G.; Bustamante, B.; Ticona, E.; Hamill, R.J.; Johnson, P.C.; Reboli, A.; Aberg, J.; Hasbun, R.; Hsu, H.H. Recombinant interferon- gamma 1b as adjunctive therapy for AIDS-related acute cryptococcal meningitis. J Infect Dis 2004, 189, 2185-2191. [CrossRef]
- Levitz, S.M.; Golenbock, D.T. Beyond empiricism: informing vaccine development through innate immunity research. Cell 2012, 148, 1284-1292. [CrossRef]
- Li, Y.; Ambati, S.; Meagher, R.B.; Lin, X. Developing mRNA lipid nanoparticle vaccine effective for cryptococcosis in a murine model. npj Vaccines 2025, 10, 24. [CrossRef]
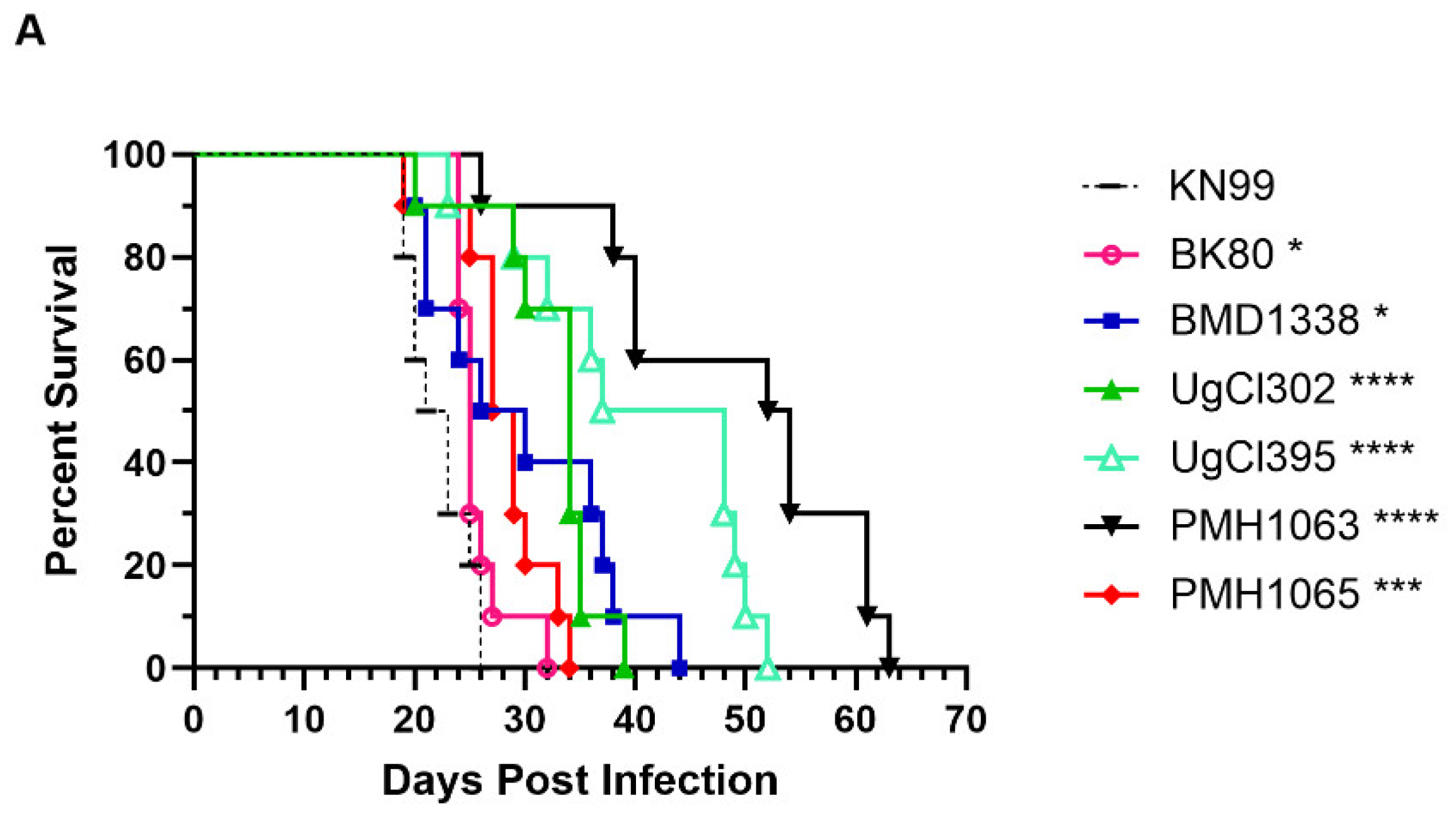
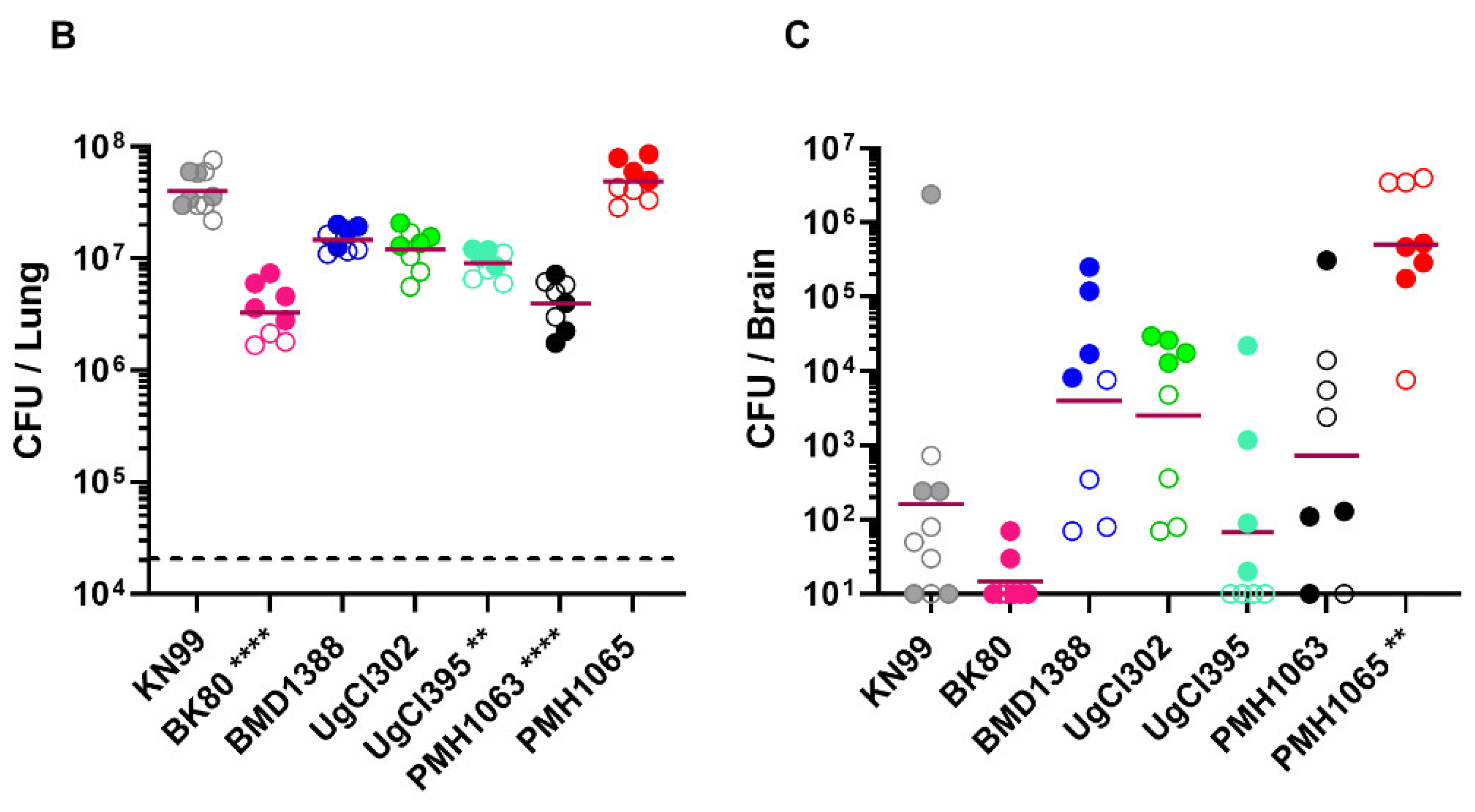
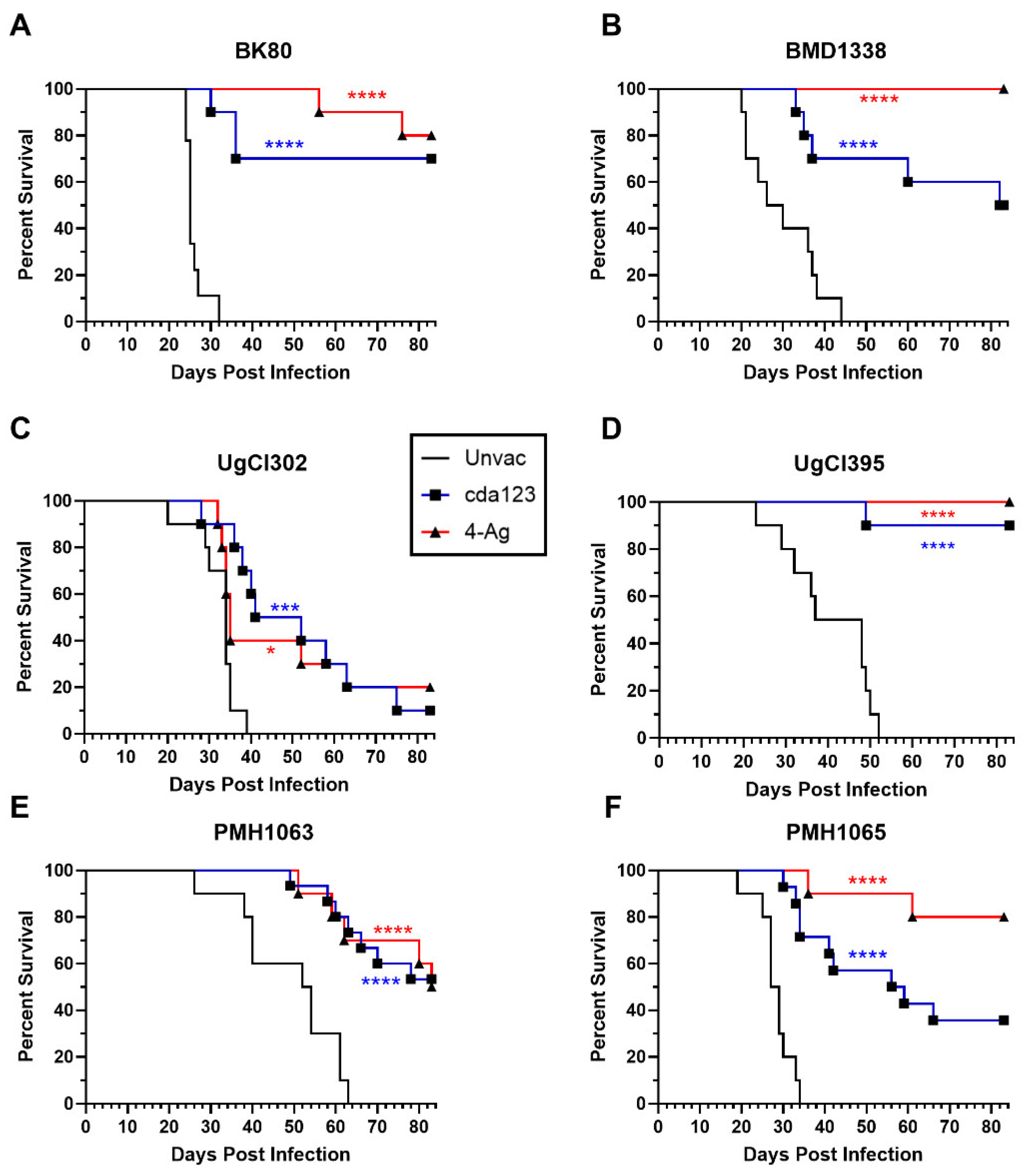
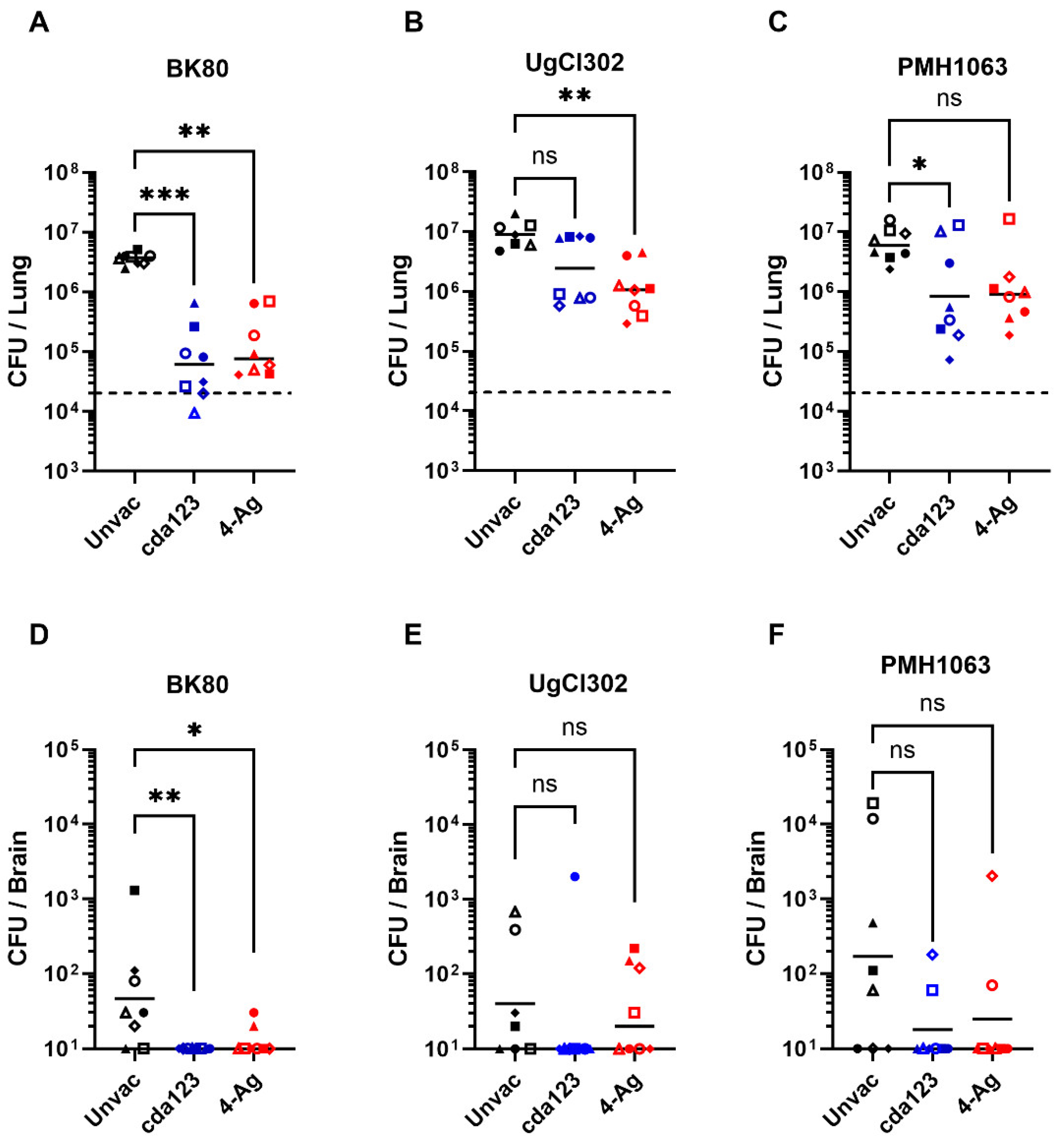
| Strain | Country | Lineage |
|---|---|---|
| KN99 (Control) | USA | VNIb |
| BK80 | Vietnam | VNIa ST4 |
| BMD1338 | Vietnam | VNIa ST5 |
| UgCl302 | Uganda | VNIa ST93 |
| UgCl395 | Uganda | VNIa ST93 |
| PMH1063 | Botswana | VNBII |
| PMH1065 | Botswana | VNI |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




