Introduction
Life’s molecular building blocks exhibit chirality (handedness); existing in left- or right-handed forms that are non-superimposable mirror images, like left vs. right gloves. Terrestrial biology is strikingly homochiral; DNA and RNA incorporate only right-handed sugars, while proteins consist exclusively of left-handed amino acids (Blackmond, 2019; Rohden et al., 2021). “Mirror life” refers to a hypothetical biology built from the opposite enantiomers, essentially a complete mirror-image version of life’s chemistry. The concept dates back to Louis Pasteur, who speculated in 1860 about a “mirror-image world of biology” after discovering molecular asymmetry (Sallembien et al., 2022). Although no mirror-based life has been found in nature, modern advances allow scientists to synthesize mirror versions of biomolecules and even contemplate entire mirror organisms in principle (Wang et al., 2016; Chen et al., 2022; Fan et al., 2021).
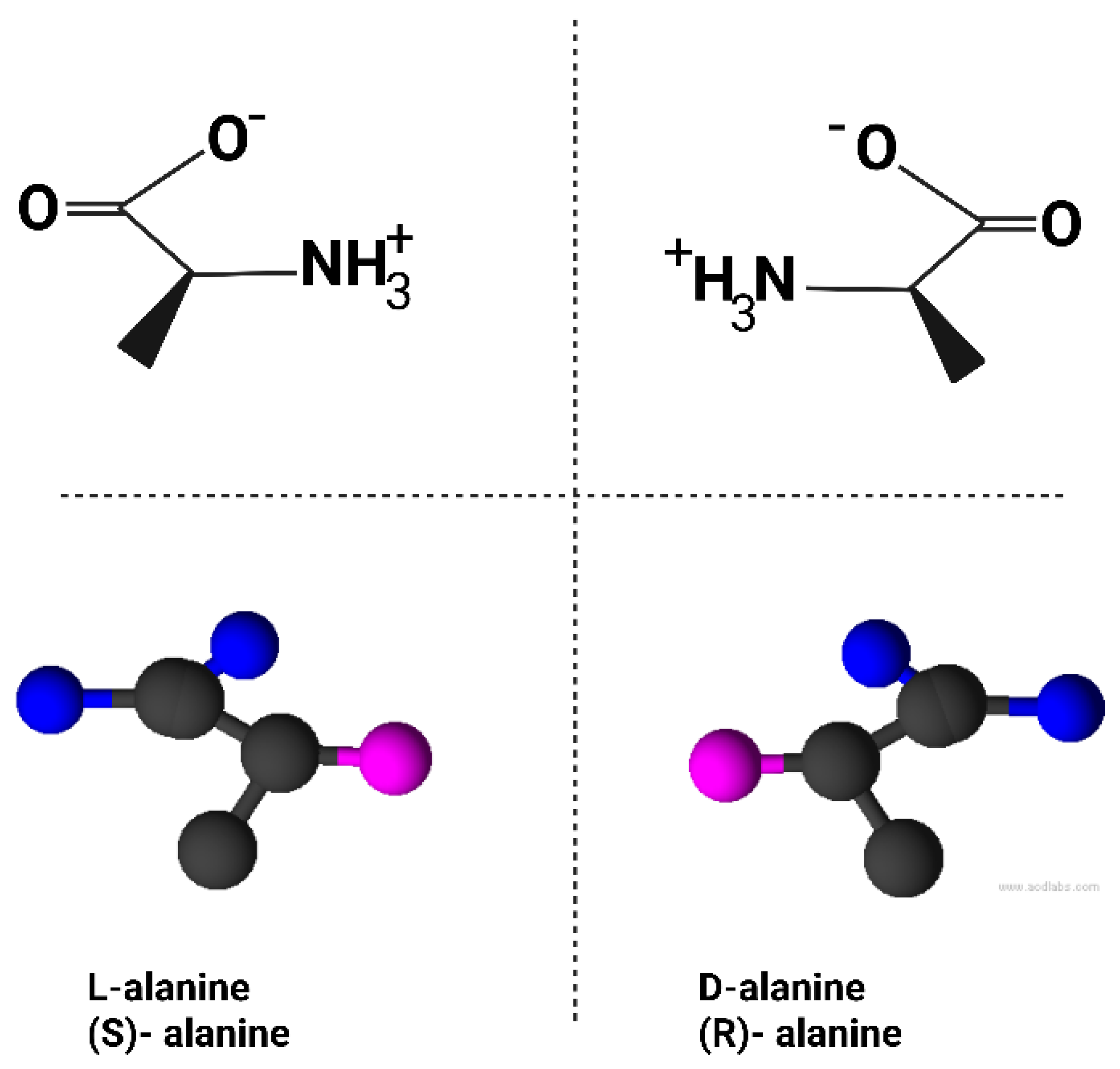
The chirality of the amino acid alanine is illustrated in Figure 1 as an example.
Chirality in Biological Systems
A molecule is chiral if it has a non-superimposable mirror image. The two mirror forms (enantiomers) have identical chemical formulas but differ in 3D orientation, often labeled “left-handed” (L) or “right-handed” (D) based on how they rotate polarized light or relate to a reference compound. This property is critical in biology; like a left hand vs. right hand, a chiral molecule will interact differently with other chiral structures, a principle analogous to how a left glove fits only a left hand. Life’s chemistry relies on this stereospecific recognition; enzymes, for example, typically bind only substrates of the correct chirality (Blackmond, 2019).
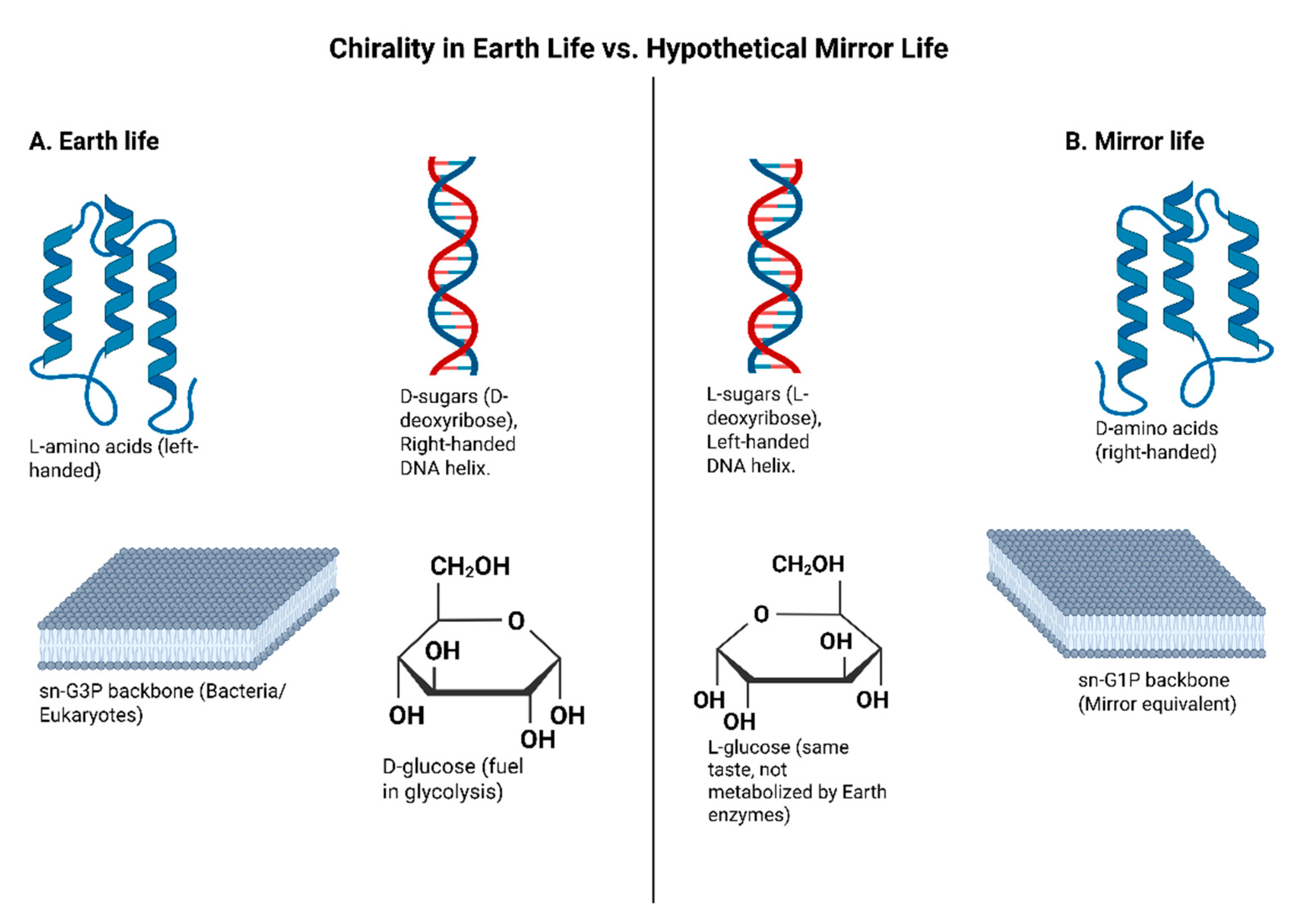
Remarkably, all known life uses only one chirality for each class of fundamental biomolecules. Specifically, virtually all amino acids in proteins are the L-form (left-handed), with rare exceptions like achiral glycine. Organisms built their proteins exclusively from L-amino acids; the D-enantiomers are not used in ribosomal protein synthesis (Rohden et al., 2021; Harrison et al., 2023). The sugars in DNA and RNA (ribose/deoxyribose) are D-form (right-handed). Thus, the sugar-phosphate backbone of nucleic acids has D-chirality. Likewise, many metabolic sugars (glucose, etc.) in biology are D-form (Blackmond, 2019; Sallembien et al., 2022). Many other chiral co-factors, vitamins, and metabolites in cells are also homochiral (e.g., only one enantiomer is biologically active). Even cell membranes reflect chirality, phospholipid glycerol backbones in bacteria/eukaryotes are one enantiomer (sn-glycerol-3-phosphate) while archaea uniquely use the opposite enantiomer; sn-glycerol-1-phosphate (Rohden et al., 2021). Notably, that is one rare case where life evolved the use of both mirror forms in different lineages, though each organism’s membrane is homochiral.
Using uniform handedness is thought to be essential for biopolymers to fold and function. Mixed-chirality polymers (containing both L- and D-monomers) generally do not fold into stable, functional structures (Harrison et al., 2023). For example, a peptide with alternating D- and L-amino acids tends to form irregular structures instead of the well-ordered helices or sheets that pure L-peptides form. Enzymes are highly shape-specific, so a wrong-handed substrate often won’t fit the active site, akin to a left hand not fitting a right-hand glove (Milton et al., 1992; Weinstock et al., 2014). Homochirality enables precise molecular recognition (DNA strands aligning, enzymes binding substrates, etc.), which is likely a prerequisite for self-replication and life’s origin (Blackmond, 2019; Rohden et al., 2021). Indeed, life’s replication machinery (DNA polymerases, ribosomes) requires consistent chirality; a D-sugar backbone in DNA will not pair properly with an L-sugar strand, and an L-amino acid tRNA cannot be read by a ribosome evolved for L-amino acids (Wang et al., 2016; Xu et al., 2017). Comparison of molecular chirality in Earth life versus hypothetical mirror life is summarized in
Table 1.
A direct consequence of homochirality is that mirror-image biomolecules are generally not recognized or utilized by Earth biology. For instance, L-glucose (the left-handed form of glucose) tastes sweet like normal D-glucose, but organisms cannot readily metabolize it as an energy source (Vater & Klussmann, 2015). Similarly, our enzymes that act on L-amino acid substrates won’t work on D-amino acids (Milton et al., 1992; Weinstock et al., 2014). This isolation cuts both ways; a “mirror” organism built from D-amino acids and L-sugars would find our nutrients, enzymes, and signals incompatible, and vice versa (Rohden et al., 2021; Harrison et al., 2023). We will later discuss how this fact underlies both potential uses of mirror systems (e.g., molecules that resist biodegradation) and dangers (e.g., mirror pathogens evading immune detection) (Adamala et al., 2024). Chirality in Earth life and hypothetical mirror life comparison is visualized in
Figure 2.
Why Does Earth’s Life Use One Handedness?
The dominance of L-amino acids and D-sugars in life raises the question: Was this choice arbitrary or driven by deeper forces? In principle, chemistry in the lab without chiral influences produces racemic mixtures (equal left/right) (Blackmond, 2019). So how and why did biology “break symmetry” to pick one hand? Scientists have explored several hypotheses.
Chance “Frozen Accident”
Life’s chirality may have been a random choice that became locked in. In a racemic primordial soup, if by chance one self-replicating system happened to use L-amino acids and gained a slight foothold, it would propagate that handedness. Competing proto-life using D-amino acids might have been outcompeted or simply failed to arise. This scenario suggests that mirror life was equally likely; Earth could have been a D-amino acid world if a few initial conditions were different (Blackmond, 2019). Many researchers lean toward chance combined with amplification mechanisms to explain homochirality (Sallembien et al., 2022).
Cosmic or Physical Bias
Alternatively, some asymmetry in physics or the environment might have biased early chemistry. One oft-cited idea is circularly polarized light in space (Bailey et al., 1998). Certain astronomical sources emit circularly polarized UV light, which can destroy one enantiomer of chiral molecules slightly more than the other. Meteorites reaching Earth have indeed shown significant enantiomeric excesses; often an excess of the same handedness used by life (e.g., an excess of left-handed amino acids in meteorite samples) (Cronin & Pizzarello, 1997). For example, analyses of carbon-rich meteorites found several with a surplus of L-aspartic and L-glutamic acid (up to ~15% ee), while their D-counterparts were depleted (Cronin & Pizzarello, 1997). This suggests an extraterrestrial asymmetry seeding Earth; “the molecule in excess was the left-handed amino acid that is found exclusively in life on Earth”. Such an excess could have been caused by polarized starlight preferentially destroying the D-form in space (Bailey et al., 1998). If true, early Earth’s organic inventory might already have had a slight L-bias, which life then amplified.
Parity Violation
On a fundamental physics level, the weak nuclear force violates mirror symmetry (P-symmetry). Calculations suggest this could make one enantiomer of certain amino acids ever so slightly more stable (on the order of 1 × 10⁻¹⁷in energy difference) (Quack, 2002). In theory, this parity-violating energy difference might favor L-amino acids and D-sugars (as observed in life). However, the effect is extremely small, and experiments have not confirmed it has any biochemical significance (Quack, 2002). Most scientists consider it unlikely to be the decisive factor in homochirality’s origin, given the vast gap between subatomic energy tweaks and the robust 100% homochirality of biology.
Terrestrial Chemical Processes
A variety of auto-amplification mechanisms have been demonstrated in the lab that can take a tiny initial enantiomeric excess and amplify it to near-pure homochirality. For instance, Frank’s autocatalysis model (1953) showed theoretically that a compound that catalyzes its own production while inhibiting its mirror image can drive a system to an enantiopure state. In 2006, Blackmond and colleagues experimentally achieved such amplification; starting from a small L-excess of an amino acid, they used crystallization cycles to amplify it to a large excess (Blackmond, 2019). They further showed that an L-excess amino acid could induce an excess in sugar chirality in an RNA precursor (Sallembien et al., 2022). Such chemical amplification could have occurred on early Earth in evaporation ponds, crystallizing mixtures, or surface catalysis on chiral minerals (Soai et al., 1995).
Given these possibilities, the prevailing view is that life’s handedness was likely a contingent outcome, a blend of some initial bias (possibly from meteorites or random fluctuation) followed by chemical and biological amplification (Blackmond, 2019; Sallembien et al., 2022). In other words, Earth’s biology might have turned out reversed if the dice had rolled differently. Importantly, nothing in known chemistry absolutely requires L-amino acids over D (or D-sugars over L), the choice could be seen as arbitrary from a purely chemical standpoint. Indeed, experimental models of prebiotic chemistry can produce either enantiomer; one experiment yielded a slight D-excess of certain amino acids under specific conditions, underscoring that homochirality could have gone either way (Soai et al., 1995).
Did Life Need Homochirality from the Start?
It’s debated whether a fully homochiral set of building blocks was necessary for the origin of life or a result of it. Some argue that molecular replication and the origin of genetic polymers (like RNA) demand a high degree of chirality purity; a “genetic takeover” can only happen once one handedness dominates (Blackmond, 2019). Others, like Blackmond, point out that life might tolerate some chirality heterogeneity in its earliest stages. Perhaps early self-replicating molecules worked with a majority chirality and gradually refined to exclusive use (Sallembien et al., 2022). Either way, once one form gained supremacy, life would become locked in because any inclusion of opposite-handed monomers in polymers tends to break function (Harrison et al., 2023). Thus, life likely selected one hand and stuck with it, leading to the universal homochirality we see today.
In summary, Earth’s use of L-amino acids and D-sugars appears to be a largely historical outcome, not an absolute requirement of biochemistry. A “mirror life” with reversed chirality is perfectly plausible chemically, a fact that motivates researchers to explore it further (Rohden et al., 2021).
Synthetic Mirror Biomolecules: D-Proteins, L-DNA, and More
Advances in synthetic chemistry and biotechnology now allow scientists to construct mirror versions of biomolecules and even envision assembling a mirror cell. This field, sometimes dubbed xenobiology or mirror biology, has achieved several milestones (Harrison et al., 2023; Rohden et al., 2021). Here are the key experiments and studies aimed at creating or investigating mirror life, from individual molecules to partial biological systems.
Researchers have chemically synthesized numerous mirror-image biomolecules by using enantiomerically flipped building blocks (D-amino acids in place of L, and L-nucleotides in place of D). These efforts demonstrate that, in isolation, many biological macromolecules have functional mirror twins.
In 2016, Ting Zhu and colleagues created a mirror-image DNA polymerase, a DNA-copying enzyme composed entirely of D-amino acids. They chemically synthesized a D-amino acid version of an enzyme and showed it could perform DNA replication and transcription on mirror-DNA templates (Wang et al., 2016). This was a landmark proof-of-concept of a “mirror central dogma” element. However, the initial mirror enzyme was small and low-efficiency. The group later synthesized a much larger, thermostable D-polymerase (Dpo4) ~352 amino acids in size (Xu et al., 2017; Pech et al., 2017; Peplow, 2016). By 2021, they reported a 90 kDa mirror version of Pfu DNA polymerase (from Pyrococcus furiosus), engineered in two fragments that self-assemble into a functional enzyme (Fan et al., 2021). This mirror Pfu polymerase can accurately amplify L-DNA (mirror DNA) in PCR-like reactions. Achieving such a large D-protein by total chemical synthesis was a major milestone, one of the largest D-proteins ever made, enabling “kilobase-sized mirror-gene” amplification.
With mirror polymerases in hand, researchers have begun constructing and using L-DNA (mirror DNA). L-DNA has the same double-helical structure as natural DNA but with left-handed deoxyribose sugars; it is sometimes called “enantiomeric DNA” or a Spiegel (mirror) polymer. A striking finding is that L-DNA is completely stable against natural nucleases and biochemistry; it’s invisible to normal enzymes. Fan et al. (2021) demonstrated this by encoding a piece of text (Louis Pasteur’s 1860 paragraph on molecular asymmetry) in an L-DNA sequence and showing it could be stored in the environment without degradation. An L-DNA “barcode” mixed into pond water remained intact and amplifiable a full year later, whereas a normal D-DNA control was destroyed within a day by microbes and nucleases. This illustrates how mirror genetic material can serve as robust “orthogonal” information storage, unreadable and uneditable by existing life. Researchers also developed mirror RNA transcription; e.g., a mirror version of T7 RNA polymerase (a ~100 kDa enzyme) was synthesized in 2022, capable of transcribing L-RNA from L-DNA templates (Chen et al., 2022; Fan et al., 2021; Rohden et al., 2021). This completes more steps of a mirror central dogma.
Beyond polymerases, other enzymes have been mirrored. In 2019, Weidmann et al. chemically synthesized a mirror-image DNA ligase entirely from D-amino acids. Remarkably, this D-ligase was enzymatically active; it could ligate L-DNA ends just as the normal ligase joins D-DNA (Weidmann et al., 2019). A commentary hailed this as “an important step for the creation of artificial life,” noting that preparing fully D-proteins is an “elegant approach to assemble completely orthogonal living systems”. Other examples include mirror versions of protein kinases, proteases, and metabolic enzymes constructed in research settings, often to test their folding and function. Each mirror enzyme typically functions only on substrates of the same chirality, highlighting strict stereospecificity (Meledin & Brik, 2019). For example, a natural L-enzyme that acts on an L-peptide will have a D-mirror counterpart that acts with equal proficiency on the corresponding D-peptide substrate (Milton et al., 1992; Weinstock et al., 2014).
An exciting application has been mirror-image aptamers (“Spiegelmers”). These are short DNA or RNA strands made of L-nucleotides that can bind targets like proteins. Because they are made of the unnatural mirror form, they resist degradation by nucleases. Companies like Noxxon Pharma have developed L-RNA aptamers that bind disease-related proteins, aiming for therapies that last longer in the body. One such L-aptamer drug (Spiegelmer NOX-A12) has progressed to clinical trials for cancer and other diseases (Vater & Klussmann, 2015). Until recently, producing mirror aptamers was laborious. In 2022, Chen et al. used a mirror-image PCR and selection system to evolve L-DNA aptamers directly from large random libraries. They performed iterative rounds of mirror PCR (using a mirror polymerase) to enrich L-DNA sequences that bind human thrombin. The selected L-DNA aptamers functioned as high-stability thrombin inhibitors, surviving even in human serum where normal DNA aptamers were quickly destroyed (Peplow, 2016). This demonstrated directed evolution in mirror space, greatly expanding the toolkit of mirror-life molecular technology (Chen et al., 2022).
A number of individual D-proteins have been synthesized to study their properties. Notably, a protein made of D-amino acids will fold into the exact mirror tertiary structure of the L-protein. X-ray crystallography of racemic mixtures (mixed D- and L-protein) can even yield better crystal packing, a technique called racemic crystallography used to solve structures that might be hard to crystallize otherwise (Yeates & Kent, 2012). Fewer than 30 distinct D-proteins had been reported by 2021. Examples include D-forms of enzymes like D-HIV protease (Milton et al., 1992), D-interleukin, and various D-peptide hormones (Weinstock et al., 2014). In all cases, the D-protein is a functional mirror of the L-protein, except that it is inert to normal biology (e.g., a D-enzyme won’t interact with L-substrates unless the substrate is achiral or via non-specific contacts) (Rohden et al., 2021).
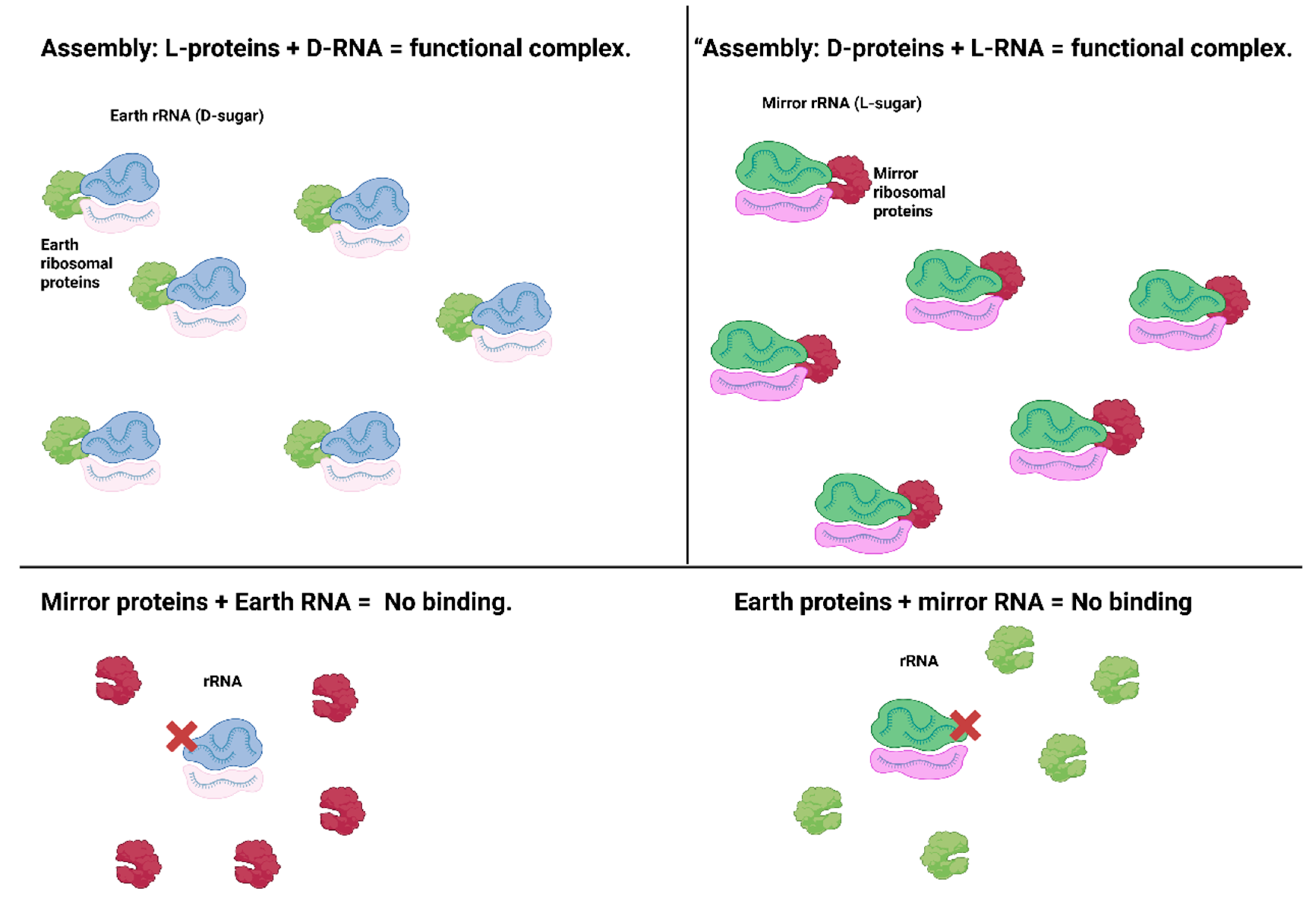
These achievements confirm a critical point. Biochemistry is symmetric with respect to chirality. Under the same physical conditions, a protein made of D-amino acids is just as viable in structure and catalysis as its L-counterpart, only oriented in reverse (Harrison et al., 2023). There appear to be no insurmountable internal barriers to mirror life’s chemistry; the challenge is more about engineering and assembling a complete mirror system (Rohden et al., 2021). Chiral orthogonality in partial ribosome assembly, displayed in
Figure 3.
Assembling Mirror-Life Systems
Constructing a fully autonomous “mirror organism” is a daunting task, but theoretical blueprints are emerging. It essentially requires rebuilding every major cellular component in opposite chirality: the genome (as L-DNA), the transcription/translation apparatus (L-RNA, D-ribosome, D-enzymes), and metabolic pathways (with mirror enzymes and co-factors) (Rohden et al., 2021; Harrison et al., 2023).
The ribosome, the molecular machine that translates mRNA into protein, is a top target for mirroring, but also one of the most complex (consisting of >50 proteins and 3 large RNA molecules). As an intermediate step, Zhu’s group synthesized mirror versions of several ribosomal proteins (D-amino acid versions of E. coli ribosomal proteins L5, L18, L25, each ~100–200 amino acids). They showed in 2019 that these mirror ribosomal proteins fold correctly and can assemble in vitro with a mirror (L-form) 5S rRNA to form partial mirror ribonucleoprotein complexes (Rohden et al., 2021). Crucially, they observed chiral specificity; the D-proteins did not bind normal 5S rRNA, only the L-5S, and vice versa. This proved that mixing chiral components in the ribosome doesn’t work; you cannot plug a mirror protein into a normal ribosome or vice versa, but a fully mirrored complex can assemble and is structurally analogous to the natural one. While a complete mirror ribosome (with all subunits and RNAs) has not been built yet, this was a step toward a mirror translation system. Simplified translation schemes are also being attempted; for example, using ribozymes called flexizymes to charge mirror tRNAs with D-amino acids, bypassing the need for dozens of mirror tRNA-synthetase enzymes (Peplow, 2016). In 2021, Chen et al. demonstrated charging L-form tRNAs with D-amino acids and even produced a small protein in a partially mirrored translation system (though not yet a fully functional mirror ribosome). These advances suggest it’s feasible to establish mirror protein synthesis, given enough engineered components (Chen et al., 2022; Peplow, 2016).
The simplest “organisms” to mirror might be viruses, since they have small genomes and few proteins (relying on a host to replicate). In theory, one could synthesize a mirror virus, e.g., an RNA virus with L-RNA genome and D-protein capsid. However, such a virus would not infect normal cells (the mirror RNA wouldn’t be recognized by L-ribosomes, and the D-capsid wouldn’t attach to L-receptors). It would require a mirror host cell to replicate. Thus, mirror viruses remain a thought experiment until mirror cells exist. One study in the 2000s did synthesize a polio virus from scratch (the natural form) to show de novo virus creation is possible; a mirror version would be the next leap. Some have proposed creating a mirror bacteriophage that could infect a mirror bacterium as part of a mirror ecosystem, but this remains speculative (Rohden et al., 2021).
Visionaries like genomics pioneer J. Craig Venter and synthetic biologist George Church have outlined how one might build a mirror microbe. One approach starts with a minimal cell (like Mycoplasma with ~500 genes, which Venter’s team has synthesized in normal form) and systematically replaces each component with its mirror version (Rohden et al., 2021). This would involve chemically synthesizing a mirror genome (L-DNA) and mirror ribosomes and enzymes, then encapsulating them in either a mirror membrane or an achiral membrane. Notably, membranes pose a special challenge; phospholipids are chiral molecules too. Church suggested using achiral lipid molecules for the first mirror cells to avoid needing mirror lipids. (Alternatively, one could use the archaeal lipid chirality as a “mirror” to bacterial, since life already tolerates that difference.) Another approach is building a proto-cell with a simplified metabolism that can copy, mirror DNA, and grow. Dr. Kate Adamala, for example, worked on assembling mirror versions of a protocell system (a liposome with a minimal gene set) as a testbed before ethical concerns shifted her focus (Adamala et al., 2024).
In parallel, computational biology has examined whether mirror proteins would interact with normal biomolecules. Predictably, they do not; a mirror enzyme’s binding pocket is the mirror image of the normal one, so it won’t bind the natural substrate. Computational docking studies confirm, for instance, that antibiotic drugs will not fit mirror bacterial targets (e.g., a mirror ribosome’s peptidyltransferase center has opposite chirality, so current antibiotics cannot bind it). This provides in silico evidence that a mirror microbe would be inherently resistant to our antibiotics and likely unrecognized by our immune proteins (Harrison et al., 2023; Adamala et al., 2024).
In summary, scientific progress has yielded individual mirror components of life: DNA, RNA, enzymes, even partial ribosomes, and shown that they can function properly in isolation. However, constructing a fully self-sustaining mirror organism has not yet been achieved. It remains a grand challenge requiring integration of all these pieces. Estimates vary, but some experts suspect it may take a decade or more of focused research to build even a simple mirror bacterium (Rohden et al., 2021). The efforts so far, though, have taught us a great deal about the feasibility and behavior of mirror-biochemistry, as discussed next.
Symmetry, Compatibility, and Constraints
Experiments confirm that for every biochemical mechanism in normal life, an equivalent mirror mechanism is possible. DNA replication, transcription, ligation, and even translation can, in principle, all operate with reversed chirality. Mirror polymerases accurately copy mirror DNA (Wang et al., 2016; Fan et al., 2021), mirror ligases join DNA ends (Weidmann et al., 2019), and mirror ribosomal proteins assemble with mirror RNA (Rohden et al., 2021; Meledin & Brik, 2019). There is no known biochemical reaction that inherently “fails” when done with opposite chirality, except that it must be done on consistently opposite substrates. This underscores a profound symmetry; the laws of chemistry (electromagnetism) are indifferent to chirality (Quack, 2002; Peplow, 2016). A mirror-image cell would obey the same biophysics as a normal cell, achieving the same complexity and function, just reflected. The only slight asymmetry comes from weak nuclear forces, but as noted, those effects are negligibly small for biochemistry (Quack, 2002).
While mirror life as a whole is viable, mixing chirality within one system is usually deleterious. The research uniformly shows that chiral specificity is strict; mirror enzymes do not interact productively with normal molecules and vice versa. For example, the mirror ribosomal proteins bound only mirror rRNA, not natural rRNA (Rohden et al., 2021). A mirror polymerase will not copy a normal DNA template (the geometry doesn’t match), and a normal polymerase won’t accept L-nucleotide substrates (Wang et al., 2016; Xu et al., 2017). If one tried to feed L-glucose to a normal bacterium, it would starve despite an energy-rich sugar being present, because its enzymes are specific for D-glucose (Vater & Klussmann, 2015; Fan et al., 2021; Peplow, 2016). These findings reinforce that any life form must internally choose one chirality consistently, otherwise its informational polymers and catalytic networks break down due to stereo-conflicts. This is likely why Earth life, once it “chose” L-amino acids and D-sugars, had to remain homochiral to thrive (Harrison et al., 2023).
One striking lesson is that a mirror biosphere would be biochemically isolated from our own. The experiments illustrate a strong form of orthogonality; normal organisms can’t eat or metabolize mirror-organic material efficiently, and mirror organisms couldn’t directly utilize normal nutrients (Harrison et al., 2023; Adamala et al., 2024). For instance, mirror cyanobacteria (if they existed), producing L-glucose and other mirror sugars, would create piles of energy-rich compounds that normal organisms cannot use. Conversely, a mirror microbe in our soil might sit surrounded by D-glucose and L-amino acids and yet starve, unless it has some racemase enzyme or a source of mirror nutrients. One exception noted is that some Earth bacteria have enzymes (racemases, epimerases) that can convert certain D-amino acids or L-sugars to the usable form. For example, a few strains can slowly metabolize L-glucose after converting it to D-glucose, and bacteria incorporate a few D-amino acids into cell walls via specialized enzymes (Rohden et al., 2021). These are rare capabilities, but they suggest that given enough time, one form of life could evolve to partially exploit the other form’s resources. Generally, however, mirror life and standard life would not compete for most biochemical resources; they occupy “orthogonal” niches.
A practical outcome of mirror experiments is recognizing that mirror biomolecules are often more stable in a normal environment. We saw how L-DNA resists nuclease degradation for orders of magnitude longer than regular DNA (Fan et al., 2021). Similarly, D-peptide drugs are not cleaved by our proteases (which target L-peptide bonds) (Milton et al., 1992; Weinstock et al., 2014). This stability is a double-edged sword; it makes mirror molecules attractive for drug design (longer half-life in the body) (Vater & Klussmann, 2015), and data storage (as DNA vaults that microbes can’t eat) (Fan et al., 2021), but it also means if mirror organisms or biochemicals were released, our ecosystem’s normal recycling pathways (decomposition, predation) wouldn’t eliminate them effectively. A mirror leaf falling from a tree might not be broken down by standard fungi or bacteria, potentially leading to the accumulation of litter that normal decomposers can’t digest (Adamala et al., 2024).
The step-by-step successes (polymerase, ligase, ribosomal subunits, etc.) show that mirror life is feasible; no known scientific law forbids it. The challenge is overwhelmingly one of engineering and labor. Chemical synthesis of large D-proteins is extremely time-consuming, often requiring solid-phase peptide synthesis in many segments and clever “ligation” chemistry to stitch segments together (Xu et al., 2017; Pech et al., 2017). For reference, the 90 kDa mirror Pfu polymerase had to be synthesized in two halves (each ~45 kDa), purified, and refolded together. The mirror T7 RNA polymerase (~100 kDa) was done in three fragments (Fan et al., 2021; Chen et al., 2022). To make a mirror E. coli (~1000 different proteins) by today’s methods is far beyond reach. However, each technological improvement (e.g., better chemical ligation methods, automated D-peptide synthesizers, or partial biological production of mirror blocks via engineered ribozymes) brings the goal closer (Harrison et al., 2023). Researchers ultimately imagine a bootstrapping scenario; once a minimal mirror ribosome or polymerase system is established, it could begin self-replicating mirror components, greatly accelerating progress. In essence, the first mirror cell will be hardest to build, but after that, it could propagate and perhaps even be engineered to produce other mirror biomaterials for us (Rohden et al., 2021).
In summary, the research so far teaches that mirror life is a realistic possibility in principle, with chemistry that mirrors our own. Yet it would be biochemically self-contained, unable to exchange genetic information or metabolites with standard life (Adamala et al., 2024). It validates that homochirality is a contingent feature, not a fixed requirement, since all critical biomolecules have functional mirror equivalents (Blackmond, 2019). And it highlights practical constraints; until we have mirror organisms to carry some load, creating large mirror systems will require enormous synthetic effort.