Submitted:
26 September 2025
Posted:
30 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
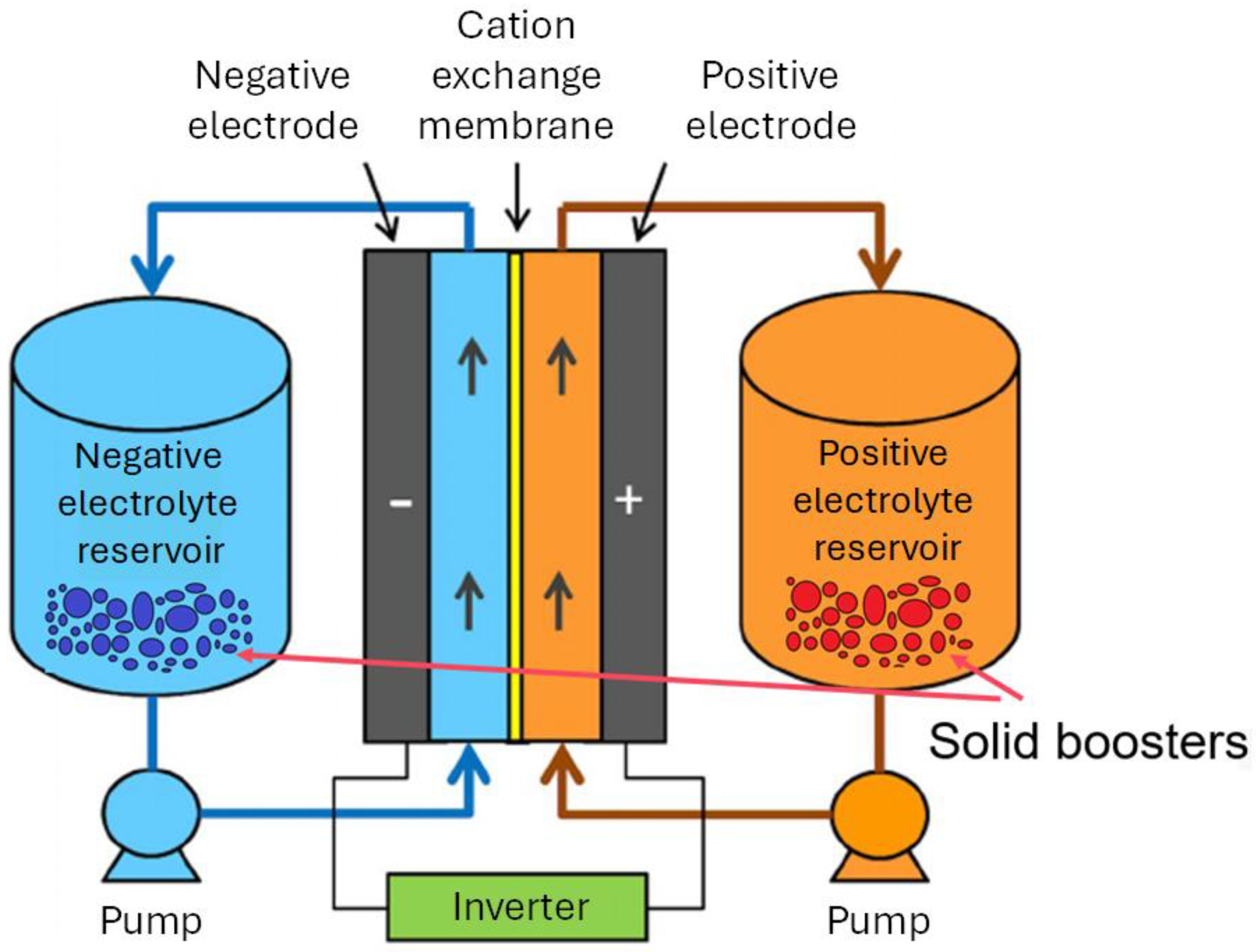
2. Materials and Methods
2.1. Materials
2.2. Electrode Preparation and Characterization
2.3. Electrochemical Measurements
3. Results
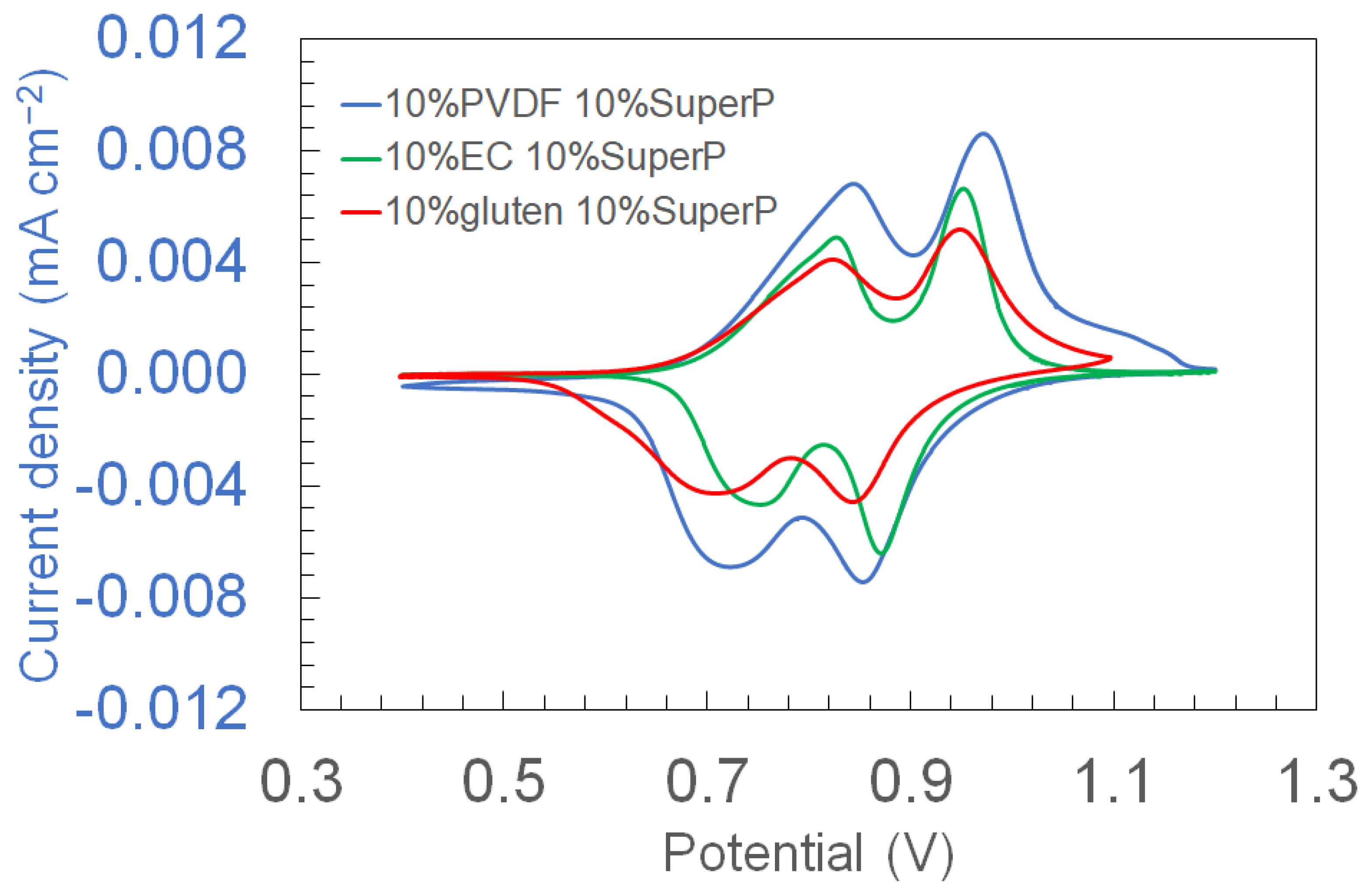
3.1. Cyclic Voltammetry of Electrodes
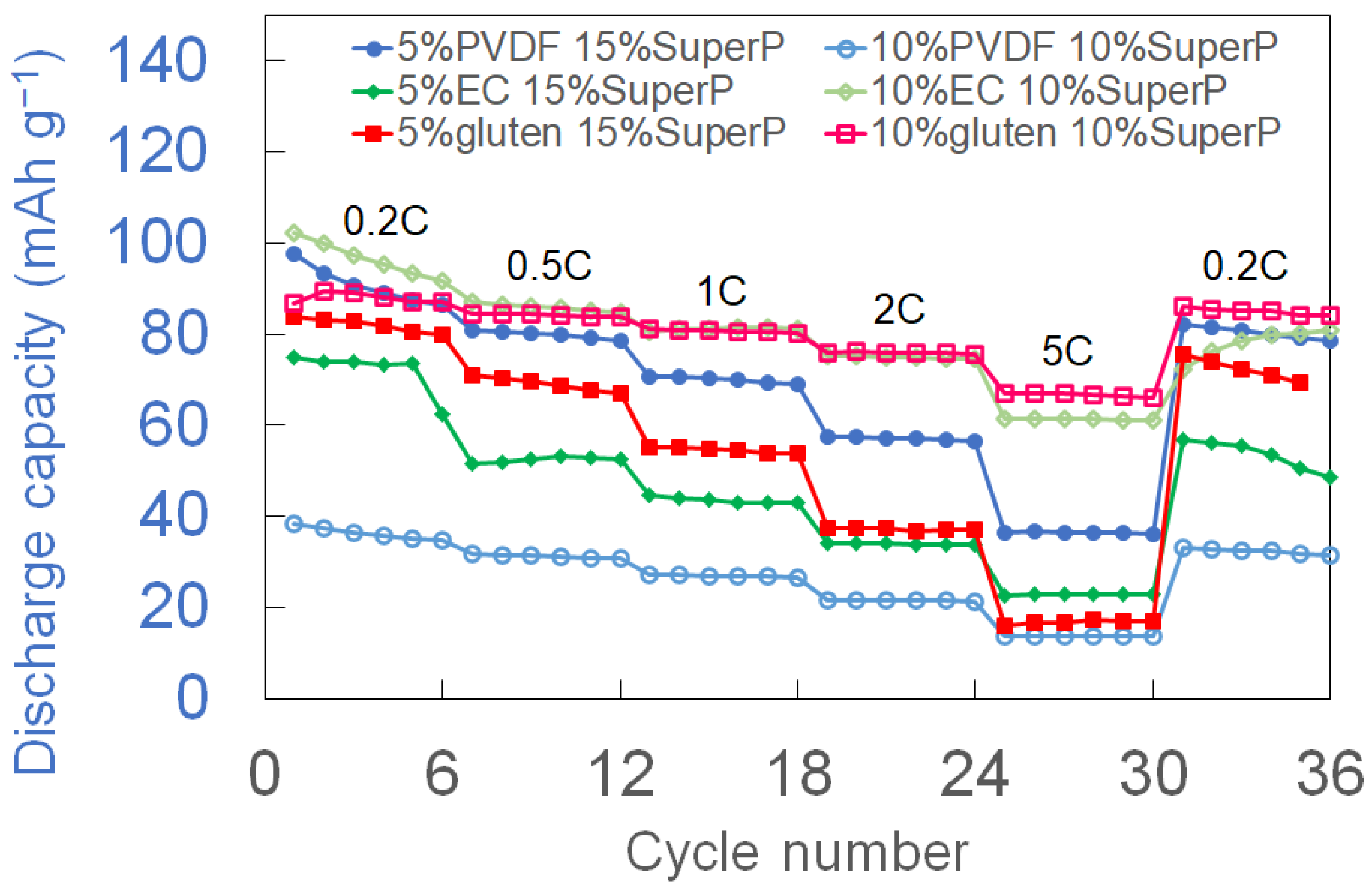
3.2. Rate Performance and Initial Capacity Comparison
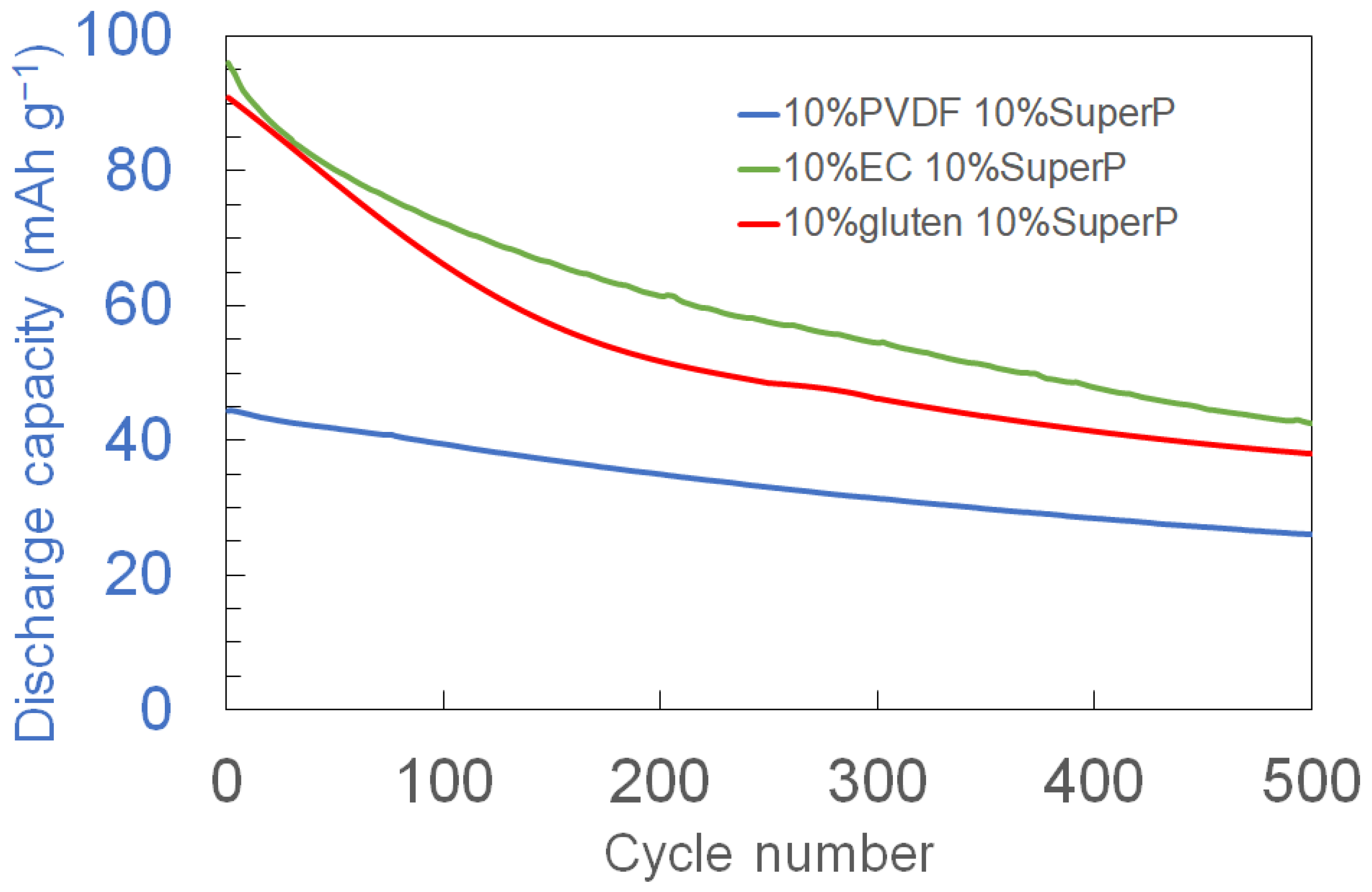
3.3. Long-Term Cycling Stability
3.4. Electrode morphology studies
4. Discussion
Mechanical and Interfacial Considerations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PVDF | Polyvinylidene Fluoride |
| ALIBs | Aqueous Lithium-Ion Batteries |
| FBs | Flow Batteries |
| LMO | Lithium Manganese Oxide (LiMn₂O₄) |
| NMP | N-Methyl-2-Pyrrolidone |
| PFAS | Per- and Polyfluoroalkyl Substances |
| EC | Ethyl Cellulose |
| CV | Cyclic Voltammetry |
| SEM | Scanning Electron Microscopy |
| EDS | Energy-Dispersive X-ray Spectroscopy |
References
- Moghaddam, M.; Sepp, S.; Wiberg, C.; Bertei, A.; Rucci, A.; Peljo, P. Thermodynamics, Charge Transfer and Practical Considerations of Solid Boosters in Redox Flow Batteries. Molecules 2021, 26, 2111. [CrossRef]
- Chen, Z.-Y.; He, Y.; Li, Z.-J.; Gao, L.-Z.; Jiang, Q.; Yu, Z.-L. Synthesis and Electrochemical Performance of Spinel LiMn2O4-x(SO4)x Cathode Materials. Chin. J. Chem. 2002, 20, 194–197. [CrossRef]
- Chen, Z.-Y.; Gao, L.-Z.; Liu, X.-Q.; Yu, Z.-L. Properties and Structure of Spinel Li-Mn-O-F Compounds for Cathode Materials of Secondary Lithium-Ion Battery. Chin. J. Chem. 2001, 19, 347–351. [CrossRef]
- Nastasi, L.; Fiore, S. Environmental Assessment of Lithium-Ion Battery Lifecycle and of Their Use in Commercial Vehicles. Batteries 2024, 10, 90. [CrossRef]
- Chen, B.; Zhang, Z.; Xiao, M.; Wang, S.; Huang, S.; Han, D.; Meng, Y. Polymeric Binders Used in Lithium Ion Batteries: Actualities, Strategies and Trends. ChemElectroChem 2024, 11, e202300651. [CrossRef]
- Chen, Z.; Kim, G.-T.; Chao, D.; Loeffler, N.; Copley, M.; Lin, J.; Shen, Z.; Passerini, S. Toward Greener Lithium-Ion Batteries: Aqueous Binder-Based LiNi0.4Co0.2Mn0.4O2 Cathode Material with Superior Electrochemical Performance. J. Power Sources 2017, 372, 180–187. [CrossRef]
- Chen, H.; Wang, Z.; Feng, Y.; Cai, S.; Gao, H.; Wei, Z.; Zhao, Y. Cellulose-Based Separators for Lithium Batteries: Source, Preparation and Performance. Chem. Eng. J. 2023, 471, 144593. [CrossRef]
- Dobryden, I.; Montanari, C.; Bhattacharjya, D.; Aydin, J.; Ahniyaz, A. Bio-Based Binder Development for Lithium-Ion Batteries. Materials 2023, 16, 5553. [CrossRef]
- Nordqvist, P.; Lawther, M.; Malmström, E.; Khabbaz, F. Adhesive Properties of Wheat Gluten after Enzymatic Hydrolysis or Heat Treatment – A Comparative Study. Ind. Crops Prod. 2012, 38, 139–145. [CrossRef]
- Li, J.; Lu, Y.; Yang, T.; Ge, D.; Wood, D.L.; Li, Z. Water-Based Electrode Manufacturing and Direct Recycling of Lithium-Ion Battery Electrodes—A Green and Sustainable Manufacturing System. iScience 2020, 23, 101081. [CrossRef]
- Yoon, J.; Lee, J.; Kim, H.; Kim, J.; Jin, H.-J. Polymeric Binder Design for Sustainable Lithium-Ion Battery Chemistry. Polymers 2024, 16, 254. [CrossRef]
- Srivastava, M.; M. R., A.K.; Zaghib, K. Binders for Li-Ion Battery Technologies and Beyond: A Comprehensive Review. Batteries 2024, 10, 268. [CrossRef]
- Cholewinski, A.; Si, P.; Uceda, M.; Pope, M.; Zhao, B. Polymer Binders: Characterization and Development toward Aqueous Electrode Fabrication for Sustainability. Polymers 2021, 13, 631. [CrossRef]
- Kuenzel, M.; Choi, H.; Wu, F.; Kazzazi, A.; Axmann, P.; Wohlfahrt-Mehrens, M.; Bresser, D.; Passerini, S. Co-Crosslinked Water-Soluble Biopolymers as a Binder for High-Voltage LiNi0.5Mn1.5O4|Graphite Lithium-Ion Full Cells. ChemSusChem 2020, 13, 2650–2660. [CrossRef]
- Dong, T.; Mu, P.; Zhang, S.; Zhang, H.; Liu, W.; Cui, G. How Do Polymer Binders Assist Transition Metal Oxide Cathodes to Address the Challenge of High-Voltage Lithium Battery Applications? Electrochem. Energy Rev. 2021, 4, 545–565. [CrossRef]
- Lee, S.; Koo, H.; Kang, H.S.; Oh, K.-H.; Nam, K.W. Advances in Polymer Binder Materials for Lithium-Ion Battery Electrodes and Separators. Polymers 2023, 15, 4477. [CrossRef]
- Wang, R.; Feng, L.; Yang, W.; Zhang, Y.; Zhang, Y.; Bai, W.; Liu, B.; Zhang, W.; Chuan, Y.; Zheng, Z.; et al. Effect of Different Binders on the Electrochemical Performance of Metal Oxide Anode for Lithium-Ion Batteries. Nanoscale Res. Lett. 2017, 12, 575. [CrossRef]
- Qin, T.; Yang, H.; Li, Q.; Yu, X.; Li, H. Design of Functional Binders for High-Specific-Energy Lithium-Ion Batteries: From Molecular Structure to Electrode Properties. Ind. Chem. Mater. 2024, 2, 191–225. [CrossRef]
- Bigoni, F.; Giorgio, F.D.; Soavi, F.; Arbizzani, C. Sodium Alginate: A Water-Processable Binder in High-Voltage Cathode Formulations. J. Electrochem. Soc. 2016, 164, A6171. [CrossRef]
- Versaci, D.; Nasi, R.; Zubair, U.; Amici, J.; Sgroi, M.; Dumitrescu, M.A.; Francia, C.; Bodoardo, S.; Penazzi, N. New Eco-Friendly Low-Cost Binders for Li-Ion Anodes. J. Solid State Electrochem. 2017, 21, 3429–3435. [CrossRef]
- Liu, G.; Zheng, H.; Song, X.; Battaglia, V.S. Particles and Polymer Binder Interaction: A Controlling Factor in Lithium-Ion Electrode Performance. J. Electrochem. Soc. 2011, 159, A214. [CrossRef]
- Kim, M.J.; Lee, C.H.; Jo, M.H.; Jeong, S.K. Electrochemical Decomposition of Poly(Vinylidene Fluoride) Binder for a Graphite Negative Electrode in Lithium-Ion Batteries. Mater. Sci. Forum 2017, 893, 127–131. [CrossRef]
- Gu, Y.; Yang, S.; Zhu, G.; Yuan, Y.; Qu, Q.; Wang, Y.; Zheng, H. The Effects of Cross-Linking Cations on the Electrochemical Behavior of Silicon Anodes with Alginate Binder. Electrochimica Acta 2018, 269, 405–414. [CrossRef]
- Lee, D.; Park, H.; Goliaszewski, A.; Byeun, Y.; Song, T.; Paik, U. In Situ Cross-Linked Carboxymethyl Cellulose-Polyethylene Glycol Binder for Improving the Long-Term Cycle Life of Silicon Anodes in Li Ion Batteries. Ind. Eng. Chem. Res. 2019, 58, 8123–8130. [CrossRef]
- Yoon, H.; Behera, P.; Lim, S.; Yun, T.G.; Hwang, B.; Cheong, J.Y. Review of the Mechanistic and Structural Assessment of Binders in Electrodes for Lithium-Ion Batteries. Int. J. Energy Res. 2024, 2024, 8893580. [CrossRef]
- Lagrain, B.; Thewissen, B.G.; Brijs, K.; Delcour, J.A. Mechanism of Gliadin–Glutenin Cross-Linking during Hydrothermal Treatment. Food Chem. 2008, 107, 753–760. [CrossRef]

| Property | PVDF (baseline) | Ethyl Cellulose (EC) | Cross-Linked Gluten |
| Binder Type | Synthetic fluoropolymer (non-renewable) |
Modified natural polymer (cellulose ether derivative) |
Natural protein polymer (wheat gluten), thermally cross-linked |
| Solvent Requirement |
NMP (toxic, high boiling point) | Ethanol or isopropanol (benign, low toxicity) | Water-based slurry (green process); heat curing required |
| Mechanical Properties | Highly flexible and tough; excellent adhesion to particles |
Good adhesion; moderate flexibility (sufficient for cohesion) | Flexible, rubbery network after crosslinking; strong cohesion |
| Water Stability | Hydrophobic and chemically inert in water (no swelling or dissolution) | Water-insoluble; minimal swelling (maintains structure) |
Water-insoluble after crosslink; slight swelling possible but no dissolution |
| Environmental Impact | Requires toxic solvent and energy-intensive drying; not biodegradable; fluorinated (hazardous disposal) | Renewable source; solvent is low-toxic and recyclable; biodegradable over time; no fluorine | Renewable and biodegradable; no organic solvent needed; no fluorine – safe disposal |
| Processing Complexity |
Established slurry casting with NMP, but needs strict safety controls and drying infrastructure | Simple slurry mixing in alcohol; fast drying; flammable solvent handling needed (standard lab safety) |
Easiest: aqueous slurry in ambient conditions; requires thermal cross-link step (can be integrated into drying) |
| Electrochemical Stability | Electrochemically inert; stable in wide potential window (up to ~4.2 V vs Li⁺ in organics) |
Inert within aqueous voltage window; no redox-active groups; stable in neutral pH |
Largely inert after curing; no significant degradation observed within water stability window (mild conditions) |
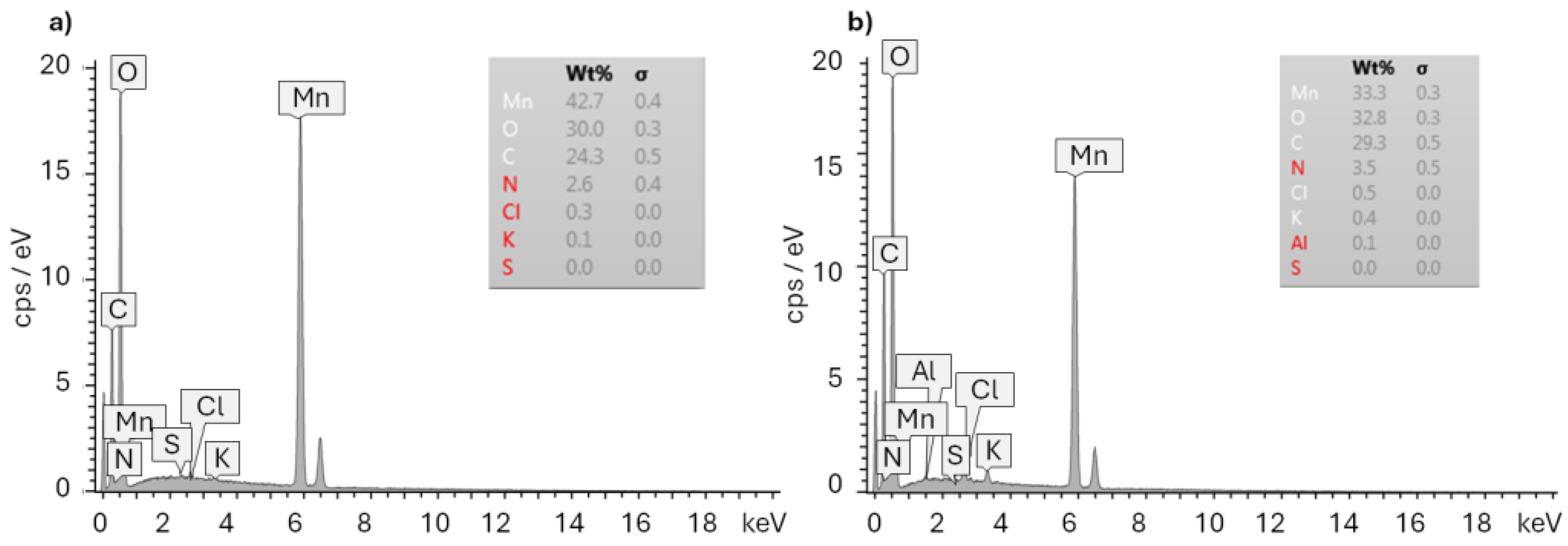
| Element | Fresh electrode | Post mortem electrode |
| Manganese (Mn) | 42 | 34 |
| Carbon (C) | 24 | 33 |
| Sulphur (S) | 0.09 | 0.01 |
| Nitrogen (N) | 3.1 | 2.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




