Submitted:
23 September 2025
Posted:
24 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
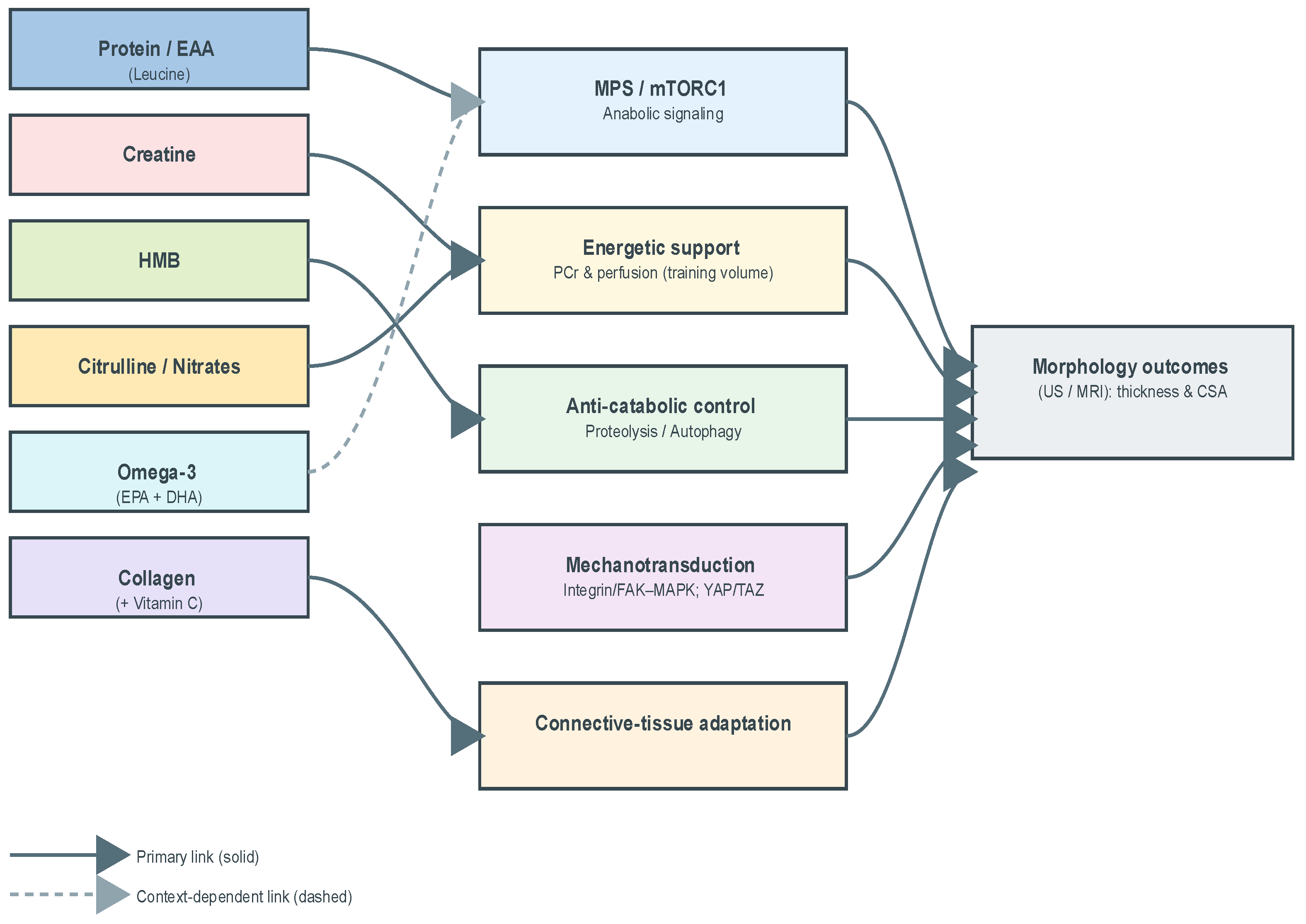
2.1. Conceptual Framework
2.2. Scope and Eligibility
- Protein/EAA (leucine)—direct stimulation of MPS via mTORC1-linked mechanisms (leucine “threshold”/per-meal distribution);
- Creatine monohydrate—energetic support (PCr buffering) enabling higher training volume/quality with downstream morphological accrual;
- β-hydroxy-β-methylbutyrate (HMB)—anti-catabolic candidate with putative benefit under high training stress or energy deficit;
- Adjuncts (omega-3, citrulline/nitrates, collagen )—plausible facilitators (anabolic sensitivity/recovery, perfusion/tolerance, connective-tissue adaptation), for which morphology-direct evidence remains limited or context-dependent.
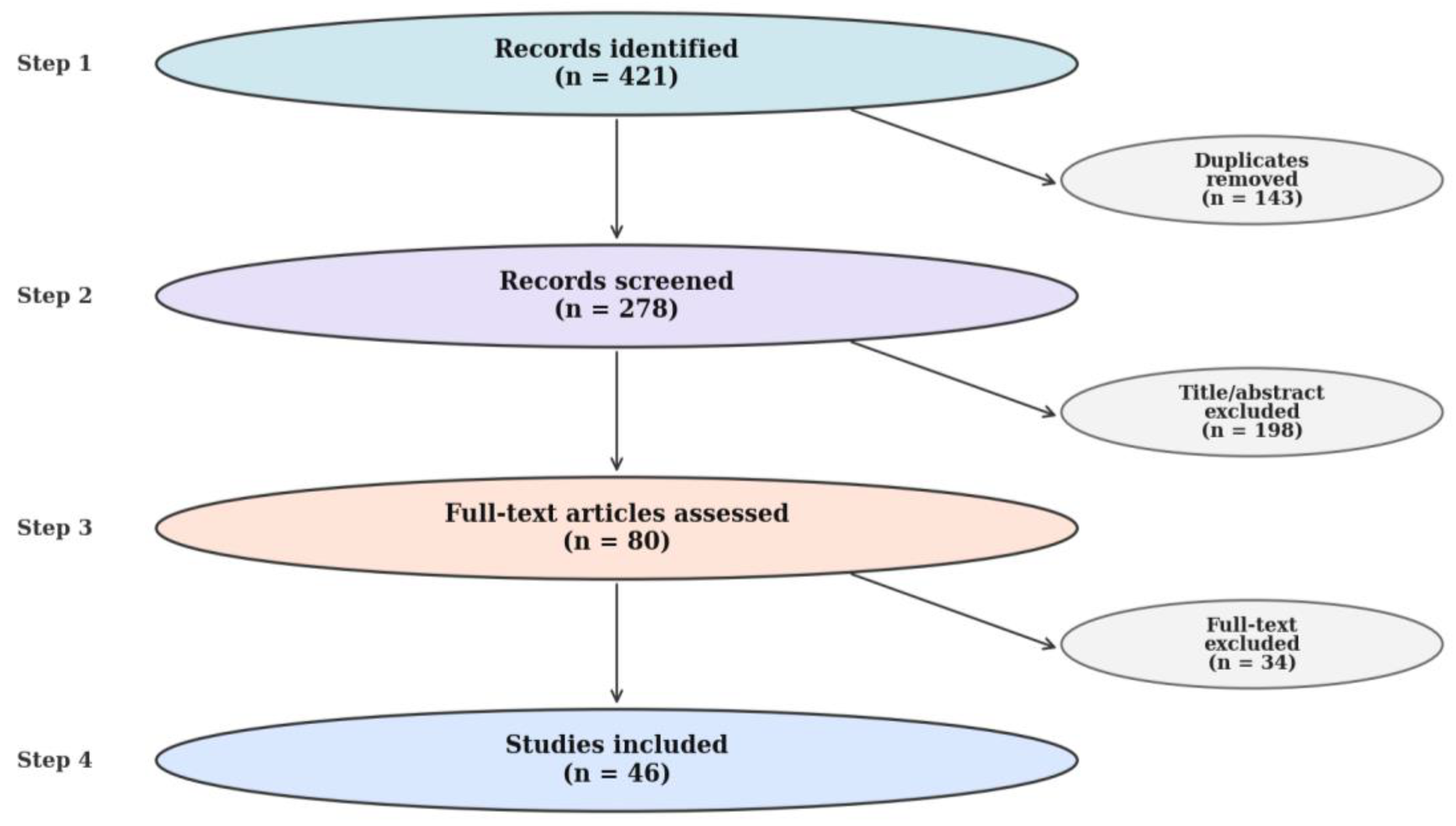
2.3. Information Sources and Search Strategy
- Trained = ≥6 months of supervised resistance training or structured equivalent;
- Untrained = <3 months of resistance training or irregular/unstructured exposure.
2.4. Study Selection and Data Extraction
2.5. Quality Appraisal and Risk of Bias (RoB 2)
2.6. Evidence Appraisal and Synthesis Approach
3. Results
3.1. Proteins and Essential Amino Acids (EAA; Leucine)
3.2. Creatine Monohydrate
3.3. β-Hydroxy-β-Methylbutyrate (HMB)
3.4. Adjuncts: Omega-3 Fatty Acids, Citrulline/Nitrates, Collagen
3.5. Evidence Synthesis Across Supplement Classes
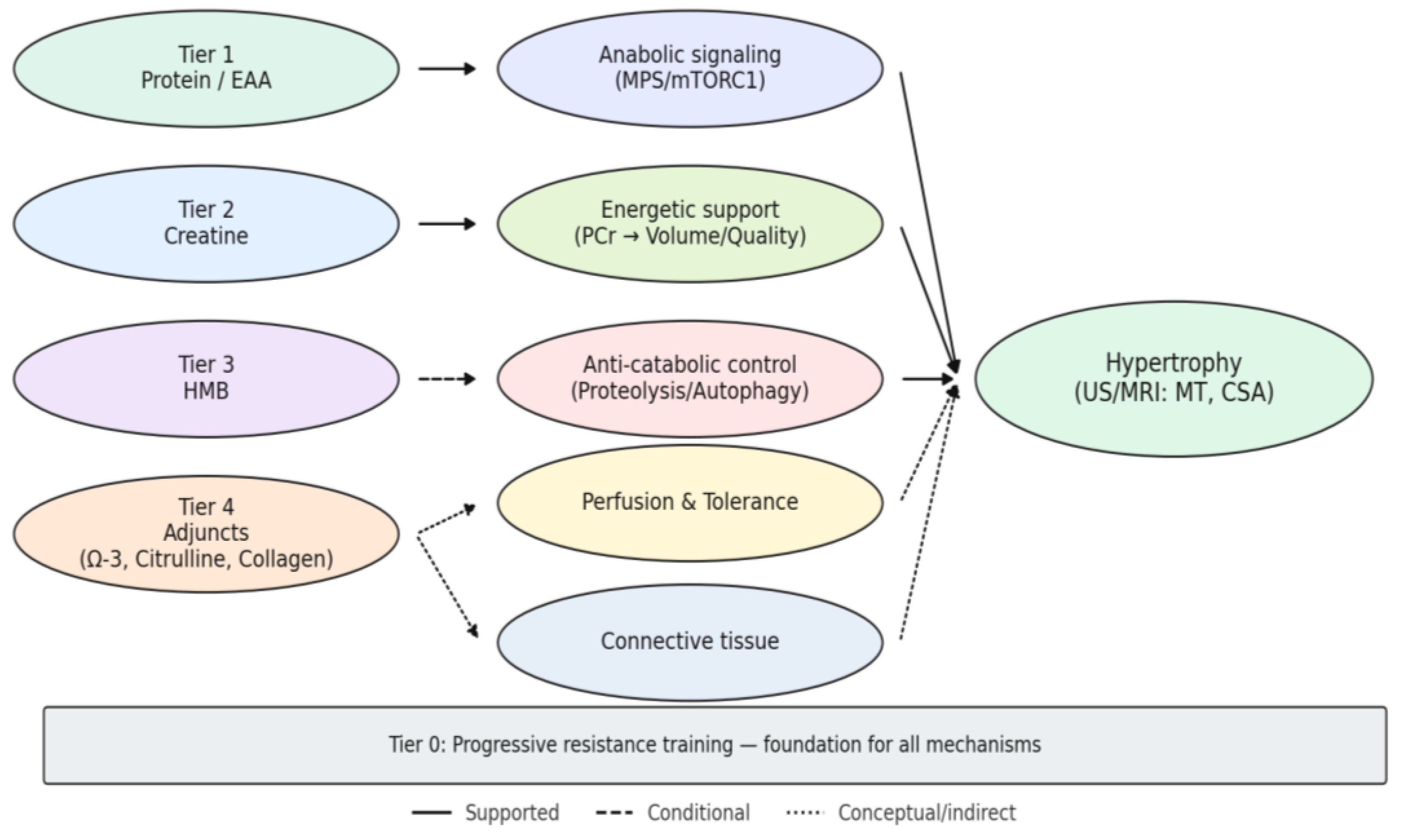
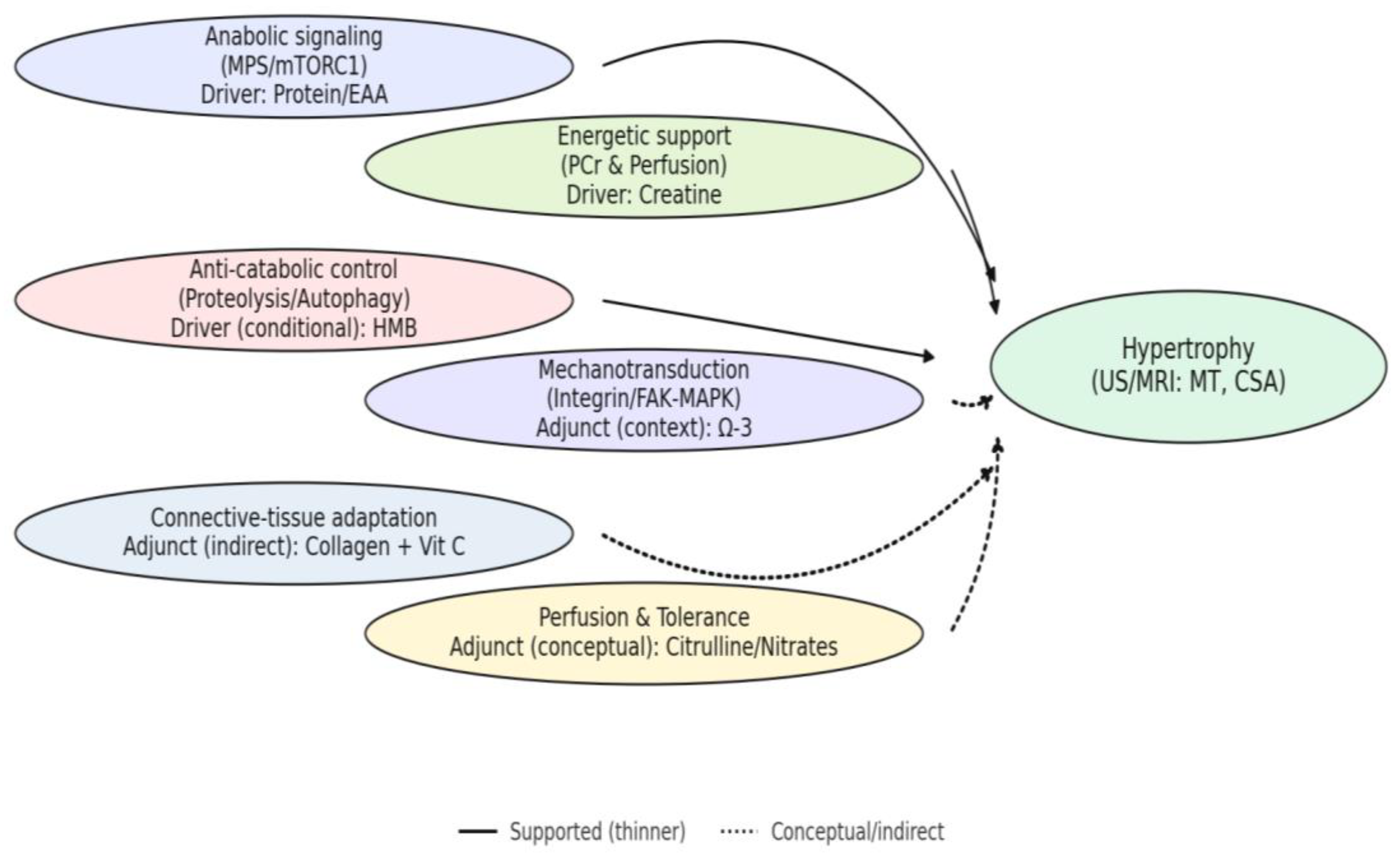
3.6. Cross-Walk to Working Hypotheses (H1–H5)
3.7. Quality and Risk-of-Bias Snapshot (Rolled-Up)
4. Discussion
4.1. Integrating Findings with the Working Hypotheses (H1–H5)
4.2. Practical Translation for Resistance-Trained Populations
4.3. Safety and Ethical Considerations
4.4. Limitations, Measurement Issues, and Research Gaps
4.5. Standardizing Morphological Assessments (Recommendations)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mazzilli, M.; Macaluso, F.; Zambelli, S.; Picerno, P.; Iuliano, E. The Use of Dietary Supplements in Fitness Practitioners: A Cross-Sectional Observation Study. Int. J. Environ. Res. Public Health 2021, 18, 5005. [Google Scholar] [CrossRef]
- Finamore, R.; et al. Sportsmen’s Attitude towards Dietary Supplements and Nutrition Knowledge: An Investigation in Selected Roman Area Gyms. Nutrients 2022, 14, 945. [Google Scholar] [CrossRef]
- Isenmann, E.; Tolle, P.; Geisler, S.; Flenker, U.; Diel, P. Differences in Consumption Behaviour of Dietary Supplements in Competitive Athletes Depends on Sports Discipline. Nutrients 2024, 16, 374. [Google Scholar] [CrossRef] [PubMed]
- Manescu, D.C. Nutritional tips for muscular mass hypertrophy. Marathon 2016, 8(1), 79–83. [Google Scholar]
- Herrmann, M.; et al. Interactions between Muscle and Bone—Where Physics Meets Biology. Biomolecules 2020, 10, 432. [Google Scholar] [CrossRef]
- Millward, D.J. Interactions between Growth of Muscle and Stature: Mechanisms of Nutritional Regulation of Postnatal Growth. Nutrients 2021, 13, 729. [Google Scholar] [CrossRef]
- Mechelli, F.; Arendt-Nielsen, L.; Stokes, M.; Agyapong-Badu, S. Validity of Ultrasound Imaging Versus Magnetic Resonance Imaging for Measuring Anterior Thigh Muscle, Subcutaneous Fat, and Fascia Thickness. Methods Protoc. 2019, 2, 58. [Google Scholar] [CrossRef]
- Ashir, A.; et al. Skeletal Muscle Assessment Using Quantitative Ultrasound: A Review. Sensors 2023, 23, 4763. [Google Scholar] [CrossRef]
- Katakis, S.; et al. Muscle Cross-Sectional Area Segmentation in Transverse Ultrasound Images Using Deep Learning. Diagnostics 2023, 13, 217. [Google Scholar] [CrossRef]
- López-Gómez, J.J.; et al. Nutritional Ultrasonography, a Method to Evaluate Muscle Mass and Quality. Nutrients 2023, 15, 3923. [Google Scholar] [CrossRef]
- Manescu, D.C. Alimentaţia în fitness şi bodybuilding. 2010, Editura ASE.
- Cuesta-Vargas, A.; et al. Automatic Analysis of Ultrasound Images to Estimate Subcutaneous and Visceral Fat and Muscle Tissue. Diagnostics 2025, 15, 988. [Google Scholar] [CrossRef]
- Shiose, K.; Takahashi, H.; Yamada, Y. Muscle Glycogen Assessment and Relationship with Body Hydration Status: A Narrative Review. Nutrients 2023, 15, 155. [Google Scholar] [CrossRef]
- Bae, K.C.; et al. Assessment of Quadriceps Muscle in Advanced Knee Osteoarthritis Using MRI. Medicina 2024, 60, 1983. [Google Scholar] [CrossRef] [PubMed]
- Meza-Valderrama, D.; et al. Musculoskeletal Ultrasound Shows Muscle Mass Changes during Hospitalization. Int. J. Environ. Res. Public Health 2022, 19, 15150. [Google Scholar] [CrossRef]
- Alamoudi, N.H.; et al. Handheld Ultrasound Parameters of Lower Limb Muscles and Correlations with Anthropometry and BIA. Diagnostics 2024, 14, 1582. [Google Scholar] [CrossRef]
- Voskuil, C.; et al. Skeletal Muscle Ultrasonography and Muscle Fitness: Effects of Scanning Plane and Echogenicity Correction. Muscles 2023, 2, 10. [Google Scholar] [CrossRef] [PubMed]
- Mănescu, D.C.; Mănescu, A.M. Artificial Intelligence in the Selection of Top-Performing Athletes for Team Sports: A Proof-of-Concept Predictive Modeling Study. Appl. Sci. 2025, 15, 9918. [Google Scholar] [CrossRef]
- Mănescu, D.C. Computational Analysis of Neuromuscular Adaptations to Strength and Plyometric Training: An Integrated Modeling Study. Sports 2025, 13, 298. [Google Scholar] [CrossRef]
- Sidiropoulos, G.K.; et al. Measurement Reliability and Anatomical Characteristics of Cervical Muscles Using Musculoskeletal Ultrasound. Muscles 2025, 4, 4. [Google Scholar] [CrossRef]
- Sánchez-Cubillo, J.; et al. Comparison between Short, Medium, and Long Fields of View in Estimating Biceps Femoris Fascicle Length. Muscles 2024, 3, 31. [Google Scholar] [CrossRef]
- Nasr, A.J.; Byrnes, T.J.; Shah, N.P. The Use of Dixon MRI Methods to Quantify Muscle Fatty Infiltration in Rotator Cuff Tears—A Systematic Review. Muscles 2024, 3, 13. [Google Scholar] [CrossRef]
- Bird, S.P.; Zwolski, C. Supplementation Strategies for Strength and Power Athletes: A Narrative Review. Nutrients 2024, 16, 1886. [Google Scholar] [CrossRef]
- Takegaki, J.; et al. Leucine-Enriched Essential Amino Acids Enhance mTORC1 Signal after Exercise. Nutrients 2020, 12, 2421. [Google Scholar] [CrossRef]
- Lixandrão, M.E.; et al. Daily Leucine Intake Is Positively Associated with Skeletal Muscle Mass and Strength in the Elderly. Nutrients 2021, 13, 3536. [Google Scholar] [CrossRef]
- Mănescu, A.M.; Grigoroiu, C.; Smîdu, N.; Dinciu, C.C.; Mărgărit, I.R.; Iacobini, A.; Mănescu, D.C. Biomechanical Effects of Lower Limb Asymmetry During Running: An OpenSim Computational Study. Symmetry 2025, 17, 1348. [Google Scholar] [CrossRef]
- Mănescu, D.C. Big Data Analytics Framework for Decision-Making in Sports Performance Optimization. Data 2025, 10, 116. [Google Scholar] [CrossRef]
- Oh, G.S.; et al. Leucine-Rich Protein Supplement plus Resistance Exercise: Body Composition and Function. Nutrients 2022, 14, 4501. [Google Scholar] [CrossRef]
- Massimino, E.; et al. Protein and Leucine Intake at Main Meals in Elderly with T2D. Nutrients 2023, 15, 1345. [Google Scholar] [CrossRef]
- Desai, I.; et al. The Effect of Creatine Supplementation on Lean Body Mass: RCT. Nutrients 2025, 17, 1081. [Google Scholar] [CrossRef]
- Forbes, S.C.; Candow, D.G.; Ostojic, S.M.; Roberts, M.D.; Chilibeck, P.D. Meta-Analysis of Creatine Ingestion Strategies in Older Adults. Nutrients 2021, 13, 1912. [Google Scholar] [CrossRef]
- Manescu, D.C. Interferențe biochimice în sport. 2025, Editura Risoprint.
- Azevedo, K.S.; et al. Creatine Supplementation Improves Muscular Performance in Trained Women. Muscles 2022, 1, 13. [Google Scholar] [CrossRef]
- Mabrey, G.; et al. Creatine Nitrate and Caffeine, Alone or Combined, on Performance. Nutrients 2024, 16, 766. [Google Scholar] [CrossRef]
- Aguiar Bonfim Cruz, A.J.; et al. Creatine Improves Sleep Duration on Resistance Training Days in Females. Nutrients 2024, 16, 2772. [Google Scholar] [CrossRef]
- Santanielo, N.; et al. Creatine Enhances the Effects of Cluster-Set Resistance Training. Nutrients 2021, 13, 2303. [Google Scholar] [CrossRef]
- Koch, A.J.; et al. The Effects of Creatine Monohydrate Supplementation on Recovery from Resistance Exercise. Nutrients 2025, 17, 1772. [Google Scholar] [CrossRef]
- Manescu, D.C. Powerlifting. 2025, Editura Risoprint.
- Jakubowski, J.S.; et al. HMB Does Not Improve RET-Induced Changes in Young Subjects: Systematic Review & Meta-Analysis. Nutrients 2020, 12, 1523. [Google Scholar] [CrossRef]
- Fairfield, W.D.; et al. HMB plus Vitamin D3 in Middle-Aged Women: Small-Scale RCT. Nutrients 2022, 14, 4674. [Google Scholar] [CrossRef]
- Manescu, D.C. Nutriție ergogenă, suplimentație și performanță. 2025, Editura Risoprint.
- Moreira-Velasco, J.E.; et al. Beyond Calcium and Vitamin D: Creatine, HMB, and Others for Muscle Health. Nutrients 2025, 17, 2332. [Google Scholar] [CrossRef]
- Zhou, S.; et al. Physiological Benefits and Applications of HMB: Review. Foods 2025, 14, 1294. [Google Scholar] [CrossRef]
- Mirzoev, T.M.; et al. Skeletal Muscle Recovery from Disuse Atrophy: Protein Synthesis and Regulation. Int. J. Mol. Sci. 2020, 21, 7940. [Google Scholar] [CrossRef]
- Gallardo, F.S.; et al. Denervation Drives YAP/TAZ Activation in Muscular Fibro/Adipogenic Progenitors. Int. J. Mol. Sci. 2023, 24, 5585. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, F.; et al. Integrating Physical and Biochemical Cues for Muscle Tissue Engineering (FAK/MAPK focus). Bioengineering 2024, 11, 1245. [Google Scholar] [CrossRef]
- Berdiaki, A.; et al. Extracellular Matrix Components and Mechanosensing/Mechanotransduction. Biomolecules 2024, 14, 1186. [Google Scholar] [CrossRef]
- Manescu, D.C. Fitness. 2025, Editura Risoprint.
- Kamal, K.Y.; et al. The Interplay between Focal Adhesions and Oxidative Stress in Skeletal Muscle. Int. J. Mol. Sci. 2025, 26, 2802. [Google Scholar] [CrossRef]
- Tan, R.; et al. Dietary Nitrate and Resistance Exercise Performance: Systematic Review & Meta-Analysis. Nutrients 2023, 15, 2493. [Google Scholar] [CrossRef]
- Gonzalez, A.M.; et al. Supplementation with Nitric Oxide Precursors for Strength Performance: Meta-Analysis. Nutrients 2023, 15, 660. [Google Scholar] [CrossRef]
- Gonzalez, A.M.; Escobar, K.A.; et al. Acute Effect of L-Citrulline on Resistance Exercise in Men and Women. Sports 2023, 8, 88. [Google Scholar] [CrossRef]
- Fernández-Lázaro, D.; et al. Omega-3 Fatty Acid Supplementation on Post-Exercise Recovery: Review. Nutrients 2024, 16, 2044. [Google Scholar] [CrossRef]
- Manescu, D.C. Bodybuilidng. 2025, Editura Risoprint.
- da Cruz Alves, N.M.; et al. Fish Oil Supplementation and Responsiveness to RET in Sarcopenic Women: RCT. Nutrients 2022, 14, 2844. [Google Scholar] [CrossRef]
- Jendricke, P.; Centner, C.; Zdzieblik, D.; Gollhofer, A.; König, D. Specific Collagen Peptides + RET Improve Body Composition and Strength in Premenopausal Women: RCT. Nutrients 2019, 11, 892. [Google Scholar] [CrossRef]
- Kirmse, M.; Oertzen-Hagemann, V.; de Marées, M.; Bloch, W.; Platen, P. Prolonged Collagen Peptide Supplementation and RET. Nutrients 2019, 11, 1154. [Google Scholar] [CrossRef]
- Paul, C.; Leser, S.; Oesser, S. Significant Amounts of Functional Collagen Peptides Can Be Incorporated in the Diet While Maintaining Indispensable Amino Acid Balance. Nutrients 2019, 11, 1079. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.A.; et al. Sarcopenia Identification Using Alternative Vertebral Landmarks in Individuals with Lung Cancer. Muscles 2024, 3(2), 12. [Google Scholar] [CrossRef] [PubMed]
| Supplement & Comparator |
Population (N / Sex / Age/ Training Status) |
RT Program & Duration |
Imaging (site) |
Dose & Timing |
ΔMT/CSA (Primary outcome) |
|---|---|---|---|---|---|
|
Creatine vs placebo |
30 M; 22±2 y; Trained |
Split 5 d/ wk supervised; 6 wks |
US multisite (upper/lower) |
~0.1 g·kg−1·d−1 intra-workout | ↑ MT over time in both; no between-group difference; strength ↑ with creatine |
|
Creatine vs caffeine vs combo vs placebo |
32 M/F; 24±3 y; Trained |
Total-body 4 d/wk; 6 wks |
US (knee extensors; elbow flexors/extensors) |
~0.1 g·kg−1 pre-workout; caffeine 3 mg·kg−1 |
↑ MT knee extensors with creatine; others ≈ NS |
|
HMB-FA vs placebo |
40 M; 23±2 y; Trained |
Periodized RT withover reaching; 12 wks |
US (quadriceps MT) |
3 g·d−1 (split doses) |
↑ MT and ↑ LBM vs placebo; strength/power ↑ |
|
Whey/Soy/Leucine vs placebo |
28 M; 21±3 y; Untrained |
Supervised RT; 12 wks |
US (VL thickness) |
2×/day; ~3 g leucine/serving |
Time effect on VL; no supplement effect; satellite cells ↑ with whey |
|
Whey vs placebo |
26 M/F; 25±4 y; Mixed |
≥3 mo RT; supervised; 12 wks |
US (biceps; VL/VI/RF) | Post-exercise; daily dose | Local VI ↑ with whey; otherwise NS |
|
Pea protein vs Whey vs placebo |
24 M; 20±2 y; Untrained |
Upper-body focus; 12 wks |
US (biceps MT) |
25 g ×2/day (incl. post-session) |
↑ biceps all groups; Pea > placebo in weakest subgroup; Pea ≈ Whey |
|
Hydrolyzed whey vs CHO |
18 M; 24±3 y; Trained |
Concentric vs eccentric emphasis; 12 wks | MRI (quadriceps CSA; tendon) |
~0.3 g·kg−1 post-session |
Whey H > CHO for muscle & tendon hypertrophy |
|
Greek yogurt vs CHO |
20 M; 19±2 y; Untrained |
Supervised RT; 12 wks |
US (biceps MT) |
~20 g protein/serving peri-workout & snacks |
↑ biceps MT > CHO; strength ↑ |
|
Whey vs collagen (leucine-equated) |
22 M/F; 21±2 y; Untrained |
Supervised RT; 10 wks |
US (arm/thigh MT) |
Iso-leucine doses |
Whey > collagen for ↑ MT during RT |
| Study | Imaging (site[s]) | Δ Treat (summary) | Δ Control (summary) | Between-group outcome (ΔΔ) | Effect size (approx.) | Reliability (ICC/TE) | Assessor blinded | Overall RoB2 |
|---|---|---|---|---|---|---|---|---|
|
Creatine vs placebo |
US multisite (upper/lower) | ↑MT both groups | ↑MT both groups | n.s. | small, n.s. | NR | Y | Some concerns |
|
Creatine vs caffeine vs placebo |
US knee extensors + EF |
↑MT (knee extensors) | ≈NS | small ΔΔ | small | ICC NR | NR | Some concerns |
|
HMB-FA vs placebo |
US quadriceps |
↑MT, ↑LBM, ↑strength | slight ↑MT | significant ΔΔ (p<0.05) | medium | ICC NR | NR | High |
|
Whey/Soy/ Leucine vs placebo |
US VL | ↑VL | ↑VL | n.s. | small, n.s. | Single-site US | Y | Some concerns |
|
Whey vs placebo |
US biceps + VL/VI/RF |
↑VI thickness |
≈NS | small ΔΔ (VI) | small | Variable reliability | NR | Some concerns |
|
Pea vs whey vs placebo |
US biceps |
↑biceps MT; Pea > placebo | ↑biceps MT | Pea > placebo; Pea ≈ whey | small | Reported consistent | Y | Low |
|
Hydrolyzed whey vs CHO |
MRI quadriceps CSA + tendon | ↑CSA, tendon hypertrophy | smaller ↑ | Whey > CHO | medium | Standardized MRI | Y | Low |
|
Greek yogurt vs CHO |
US biceps |
↑biceps MT; ↑strength | ↑biceps MT (less) | Yogurt > CHO | small–medium | NR | NR | Some concerns |
|
Whey vs collagen |
US arm + thigh |
↑MT (whey) | smaller ↑MT | Whey > collagen | small–medium | ICC NR | NR | Some concerns |
| Supplement | Primary mechanistic node(s) |
Typical dosing (range) | Typical timing | Morphology (US/MRI) evidence in trained adults |
Consistency | Key caveats |
|---|---|---|---|---|---|---|
| Protein/ EAA (Leucine) | MPS/mTORC1; translational efficiency; leucine threshold |
~0.3 g·kg−1· meal−1 or 2–3 g leucine/meal; 3–5 meals/day |
Pre/ Post RT; distributed |
Positive when baseline intake/distribution is inadequate; diminishing returns when already optimized |
Moderate to High |
Endpoint choice; per-meal distribution; baseline intake; ceiling effect at high protein |
| Creatine Monohydrate | PCr buffering → set quality & training volume |
3–5 g/day (± short loading ~0.3 g·kg−1·day−1 × 5–7 days) |
Daily; align with RT blocks |
Small–moderate gains with long, progressive RT; neutral when blocks are short or under-progressed |
Moderate | Program duration; early water shifts; inter-individual variability; imaging site |
|
HMB (FA or Ca-salt) |
Anti-catabolic (proteolysis/ autophagy); membrane stability |
~3 g /day (FA or Ca-salt) |
Daily; consider high-stress or deficit | Context-dependent; mixed/neutral in eucaloric, highly trained settings |
Low to Moderate |
Context-dependent; formulation differences; energy status; industry sponsorship |
| Omega-3 Fatty Acids | Membrane/ inflammation; anabolic sensitivity |
~1–3 g /day EPA + DHA |
Daily | Limited/heterogeneous direct morphology outcomes; may facilitate training/recovery | Low | Dependent on adequate protein and overload; small samples; heterogeneous dosing |
|
Citrulline / Nitrates |
NO pathway; perfusion/ tolerance |
6–8 g L-citrulline; dietary nitrates | Pre - exercise |
Inconsistent morphology; performance effects more common | Low | Large inter-study variation; acute vs chronic effects; weak morphology linkage |
| Collagen (+Vitamin C) | Connective-tissue support | 10–15 g /day |
Daily; ± pre-rehab / RT |
Limited direct CSA gains; indirect support to training continuity |
Low | Outcome alignment; Vit C timing; often inferior to whey comparators; targeted use-cases; |
| Supplement class | Imaging RCTs (n) | Median duration (wk) | Typical ΔΔ (US/MRI) | Typical Δ% hypertrophy | Signal strength |
|---|---|---|---|---|---|
| Protein / EAA (Leucine) | ~9 | 10–12 | +1–2 mm MT (vastus lateralis, biceps) | +3–5% vs controls (when baseline intake <1.6 g·kg−1·day−1) |
Strong, but conditional |
| Creatine monohydrate | ~7 | 8–12 | +1.5–2.0 mm MT or +0.3–0.5 cm2 CSA (KE, EF) |
+5–7% hypertrophy over controls in long programs |
Moderate-to-strong |
|
HMB (FA/Ca-salt) |
~3 | 12 | FA trial: +2–3 mm quadriceps MT; others neutral |
+4–6% in stressed/deficit states; ≈0% in eucaloric trained |
Weak-to-moderate, context-dependent |
|
Omega-3 fatty acids |
2 | 8–12 | ≈ n.s. for MT/CSA in young trained adults |
≤1% Δ vs controls | Weak |
| Citrulline / Nitrates | 1–2 | 6–8 | No reproducible MT/CSA effect |
0% | Very weak |
|
Collagen (+Vit C) |
2 | 8–10 | Neutral for MT; ↑ tendon CSA ~5–10% |
0% muscle; +5–10% tendon |
Weak (connective tissue support) |
| Trial | Randomization | Outcome measurement |
Reporting bias | Notes (Sponsorship / Blinding) |
Overall risk |
|---|---|---|---|---|---|
| Creatine vs placebo | Low | Some concerns (single-site US) |
Low | Independent; assessor blinded | Some concerns |
| Creatine vs caffeine vs combo vs placebo | Some concerns |
Some concerns (limited sites; ICC NR) |
Low | Independent; blinding NR | Some concerns |
|
HMB-FA vs placebo |
Some concerns |
Some concerns (industry involvement; ICC NR) | High | Industry-sponsored; assessor NR | High |
|
Whey/Soy/Leucine vs placebo |
Low | Some concerns (single-muscle US) |
Low | Independent; assessor blinded | Some concerns |
| Whey vs placebo | Low | Some concerns (multi-site US; variable reliability) | Low | Independent; blinding NR | Some concerns |
|
Pea protein vs Whey vs placebo |
Low | Low (US; biceps thickness; consistent reporting) | Low | Independent | Low |
|
Hydrolyzed whey vs CHO |
Low | Low (MRI standardized, tendon + muscle CSA) | Low | Independent; assessor blinded | Low |
|
Greek yogurt vs CHO |
Low | Some concerns (single-site US) |
Low | Independent; blinding NR | Some concerns |
|
Whey vs collagen (leucine-equated) |
Low | Some concerns (US; variable sites; ICC NR) | Low | Independent; blinding NR | Some concerns |
| Domain | Minimum reporting requirement |
|---|---|
| Anatomical site | Exact landmark (e.g., vastus lateralis at 50% femur length); specify side and limb dominance. |
|
Probe handling (US) |
Angle of insonation, applied pressure, coupling medium (gel, stand-off pad). |
| Timing / participant state | Standardize time of day, hydration, prior exercise, and nutrition before scans. |
| Repetition / averaging | Aqcuire ≥3 images per site recommended; report procedure for averaging values. |
| Reliability metrics | Provide intra- and inter-rater ICC; Typical Error (TE) or coefficient of variation (CV). |
| Assessor blinding | State explicitly whether imaging assessor was blinded to allocation and time point. |
| Data reporting | Report baseline and post values (mean ± SD), within-group Δ, between-group ΔΔ with 95% CI. |
| Units & transparency | Use standardized units (mm for MT; cm2 for CSA); pre-register measurement protocol; report device make/model. |
| Domain | Reporting requirement |
|---|---|
| Training load & volume | Total sets, reps, and load lifted per week (absolute and relative to baseline). |
| Intensity & effort | %1RM or RIR (repetitions in reserve) for each session. |
| Session density | Rest intervals and total session duration. |
| Progression | Week-to-week changes in load, volume, or intensity. |
| Performance outcomes | Strength (1RM, isometric MVC), endurance, power (jump, sprint). |
| Morphological outcomes | US/MRI: MT, CSA with exact site and reliability indices. |
| Linking variables | Paired reporting of workload and morphology to permit mediation analyses. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




