Submitted:
22 September 2025
Posted:
24 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Main Challenges in Table Grape Production and Postharvest
2.1. Fungal Incidence and Decay
2.2. Cracking
2.3. Berry Shattering
2.4. Colour Loss and Browning
2.5. Dehydration and Rachis Browning
3. Current Strategies for Quality Control of Table Grapes
3.1. Preharvest Strategies
3.1.1. Girdling
3.1.2. Hormone Applications
3.2. Postharvest Strategies
3.2.1. Modified Atmosphere Packaging Technology
3.2.2. Sulfure Dioxide Applications: Benefits and Limitations
3.2.3. Alternative Solutions
3.3. The Need for Sustainable and Innovative Solutions
4. The Crucial Role of Ca in Fruit Quality at Harvest and Postharvest
4.1. Ca as a Structural Component, Signaling Agent, and Its Mobility in the Plant
4.2. Physiological Disorders Associated with Ca Deficiency
4.3. Effect of Ca Application on Fruit Quality
4.4. Effect of Ca Application on Table Grapes Quality
5. Polyols as Physiological Tools and Nutrients Vector
5.1. Sorbitol: Properties and Role in Plant Physiology
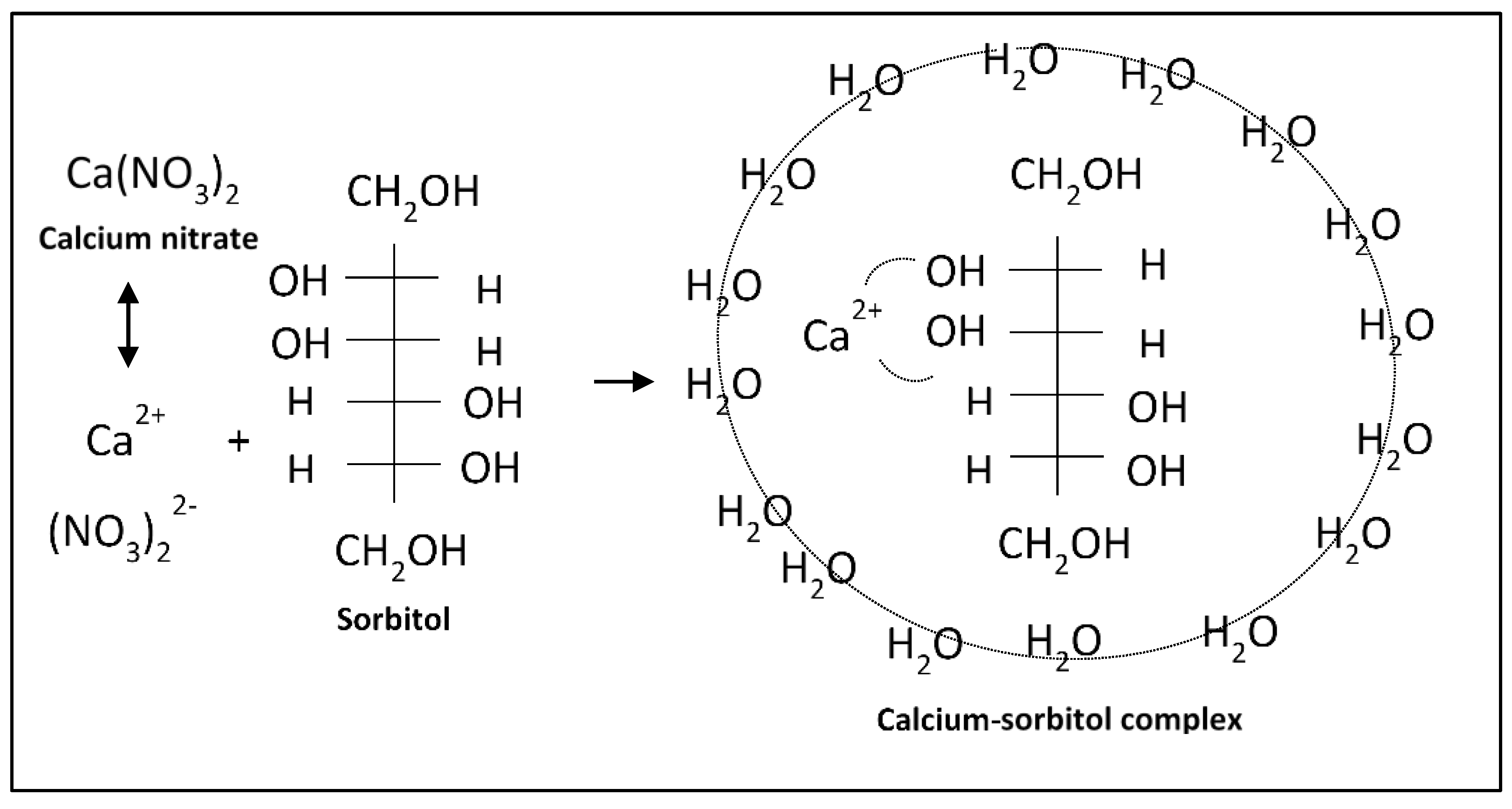
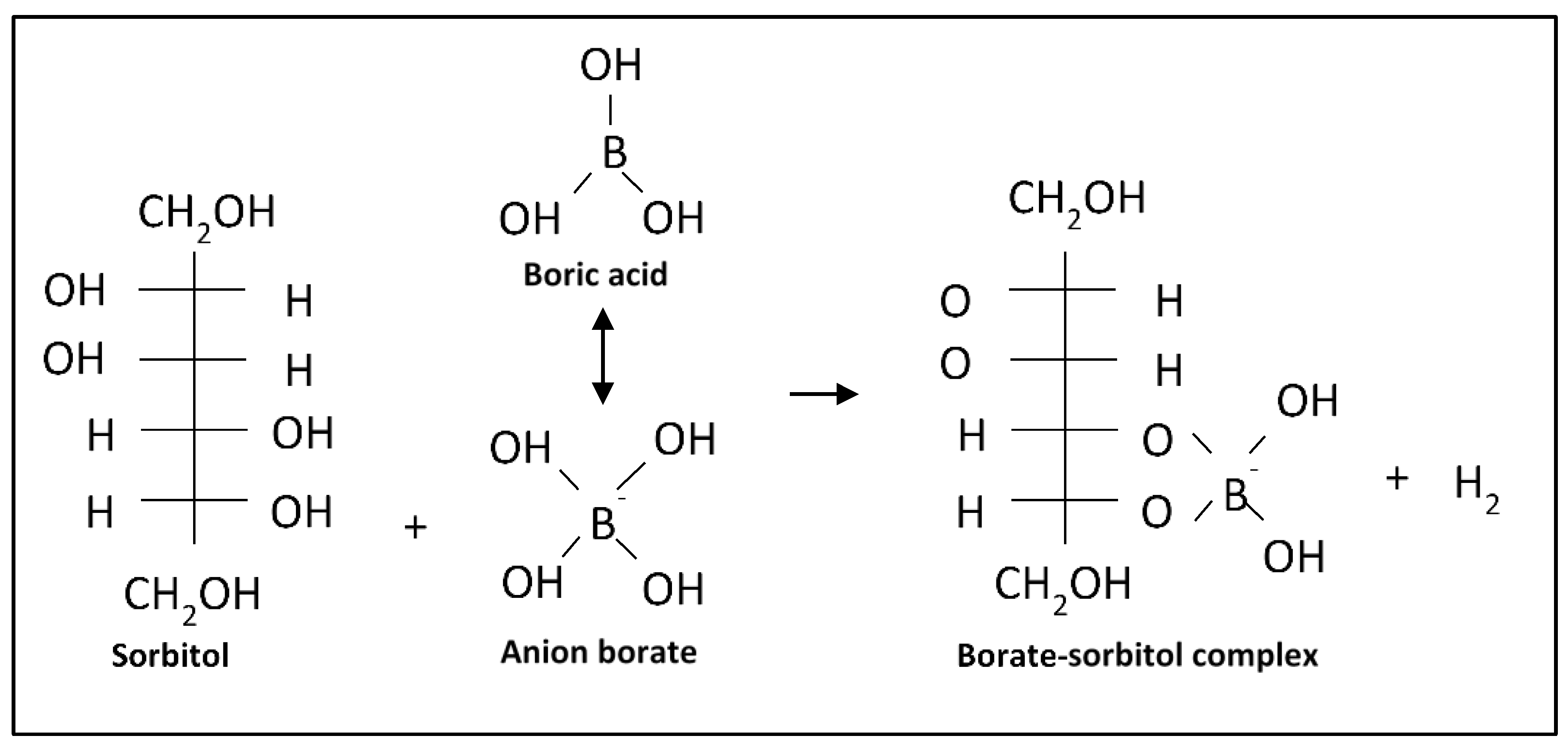
5.2. Complexation Mechanism and Transport of Sorbitol
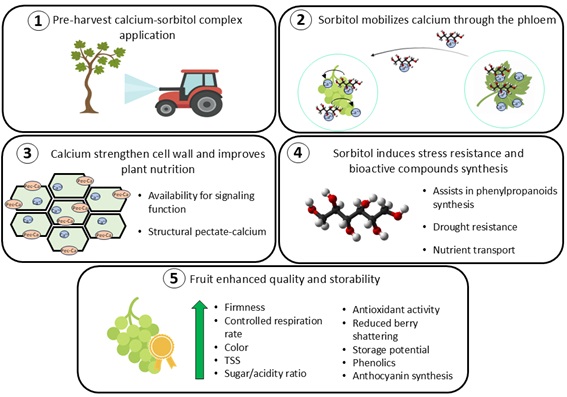
6. Applications and Effect of Calcium-Sorbitol Complexes in Table Grape Quality
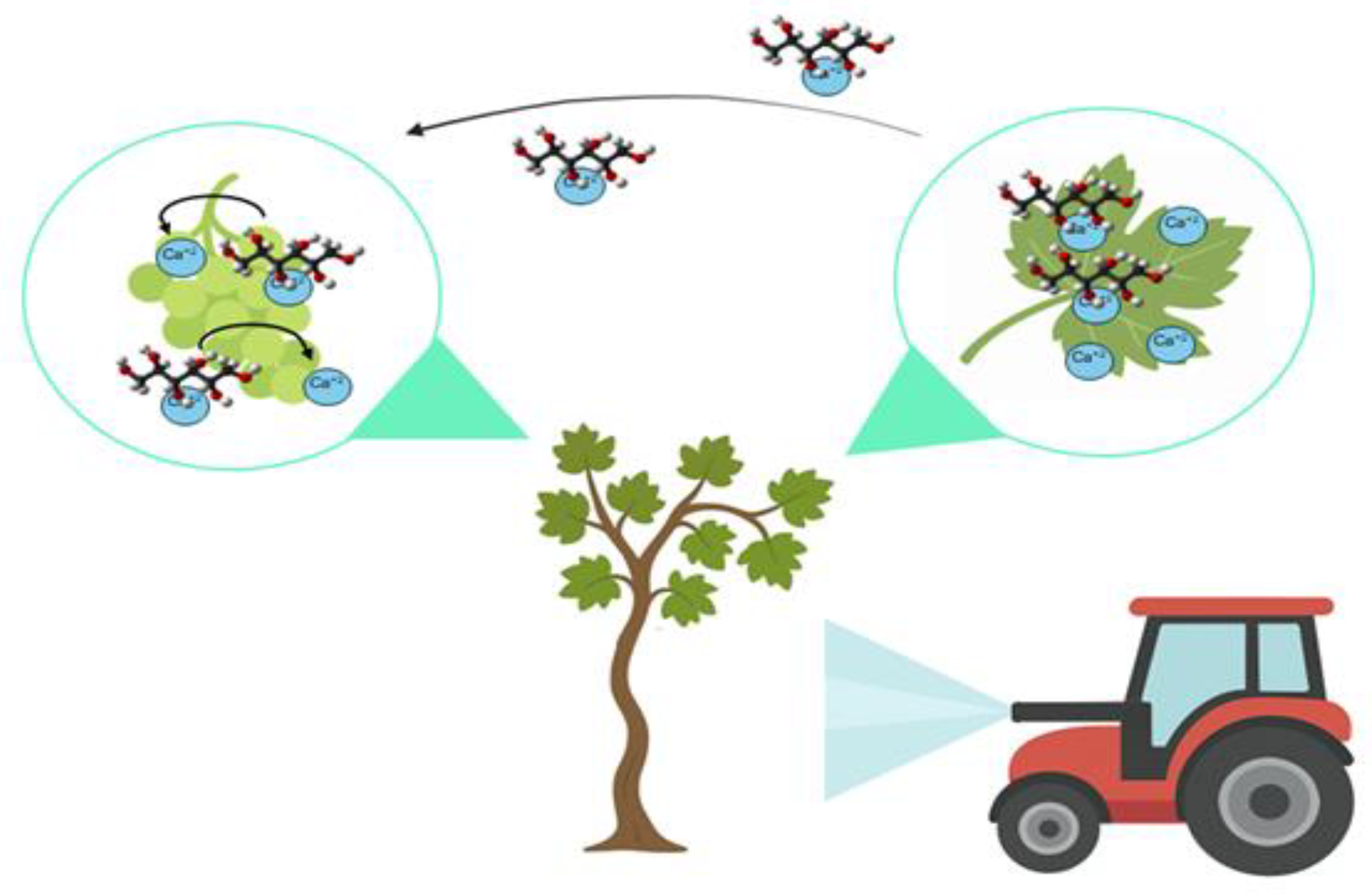
6.1. Enhancing Ca Transport and Other Nutrients
6.2. Effects on Fruit Quality and Stress Resistance
7. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moradi, S.; Koushesh Saba, M.; Sadeghi, S.; Inglese, P.; Liguori, G. Changes in Biochemical and Bioactive Compounds in Two Red Grape Cultivars during Ripening and Cold Storage. Agronomy 2024, 14. [Google Scholar] [CrossRef]
- Akram, M.T.; Qadri, R.; Khan, M.A.; Atak, A.; Liaquat, M.; Hussain, T.; Khan, M.M.; Azam, M.; Hasan, M. Ul Comparative Assessment of Bioactive Compounds, Fruit Quality Attributes and Sugar Profiling in Early Maturing Table Grape (Vitis Vinifera L.) Cultivars from Pothohar, Pakistan. Applied Fruit Sci 2024, 66, 983–995. [Google Scholar] [CrossRef]
- Conde, A.; Badim, H.; Dinis, L.T.; Moutinho-Pereira, J.; Ferrier, M.; Unlubayir, M.; Lanoue, A.; Gerós, H. Stimulation of Secondary Metabolism in Grape Berry Exocarps by a Nature-Based Strategy of Foliar Application of Polyols. Oeno One 2024, 58. [Google Scholar] [CrossRef]
- Ban, Z.; Zhang, S.; Niu, C.; Liu, L.; Cao, K.; Li, L.; Wu, Z.; Wang, L.; Chen, C.; Zhu, Y. Potential Role of Exogenous Melatonin Involved in Postharvest Quality Maintenance of Vitis Labrusca × Vinifera ‘Kyoho. ’ J Sci Food Agric 2023, 103, 6243–6251. [Google Scholar] [CrossRef]
- Admane, N.; Genovese, F.; Altieri, G.; Tauriello, A.; Trani, A.; Gambacorta, G.; Verrastro, V.; Di Renzo, G.C. Effect of Ozone or Carbon Dioxide Pre-Treatment during Long-Term Storage of Organic Table Grapes with Modified Atmosphere Packaging. LWT 2018, 98, 170–178. [Google Scholar] [CrossRef]
- Solairaj, D.; Guillaume Legrand, N.N.; Yang, Q.; Zhang, H. Isolation of Pathogenic Fungi Causing Postharvest Decay in Table Grapes and in Vivo Biocontrol Activity of Selected Yeasts against Them. Physiol Mol Plant Pathol 2020, 110. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, J.; Wang, S.; Ke, Y.; Ren, T.; Wang, Y. Mechanism Analysis of Amphotericin B Controlling Postharvest Gray Mold in Table Grapes. Foods 2025, 14. [Google Scholar] [CrossRef]
- Pedrotti, C.; Marcon, Â.R.; Delamare, A.P.L.; Echeverrigaray, S.; da Silva Ribeiro, R.T.; Schwambach, J. Alternative Control of Grape Rots by Essential Oils of Two Eucalyptus Species. J Sci Food Agric 2019, 99, 6552–6561. [Google Scholar] [CrossRef] [PubMed]
- Quintieri, L.; Fancello, F.; Caputo, L.; Sorrentino, A.; Zara, S.; Lippolis, V.; Cervellieri, S.; Fanelli, F.; Corvino, A.; Pace, B.; et al. Effect of Gaseous Citral on Table Grapes Contaminated by Rhizopus Oryzae ITEM 18876. Foods 2022, 11. [Google Scholar] [CrossRef]
- Torres-Palazzolo, C.; Ferreyra, S.; Iribas, F.; Chimeno, V.; Rojo, M.C.; Casalongue, C.; Fontana, A.; Combina, M.; Ponsone, M.L. Biocontrol of Alternaria Alternata in Cold-Stored Table Grapes Using Psychrotrophic Yeasts and Bioactive Compounds of Natural Sources. Int J Food Microbiol 2024, 415. [Google Scholar] [CrossRef]
- Chen, L.; Zhao, C.; Yan, T.; Li, B.; Wang, S.; Gong, D.; Long, D. Antifungal Potentiality of Non-Volatile Compounds Produced from Hanseniaspora Uvarum against Postharvest Decay of Table Grape Fruit Caused by Botrytis Cinerea and Penicillium Expansum. Postharvest Biol Technol 2025, 222. [Google Scholar] [CrossRef]
- Mundy, D.C.; Elmer, P.; Wood, P.; Agnew, R. A Review of Cultural Practices for Botrytis Bunch Rot Management in New Zealand Vineyards. Plants 2022, 11. [Google Scholar] [CrossRef]
- Soldateli, F.J.; Both, V.; Thewes, F.R.; Wendt, L.M.; Ludwig, V.; Rossato, F.P.; Thewes, F.R.; Batista, C.B.; Brackmann, A.; Wagner, R. Overall Quality, Phenolic Compounds, and Volatile Profile of ‘BRS Isis’ Seedless Table Grapes after Long-Term Storage: Effect of Ethanol and High CO2. Sci Hortic 2023, 312. [Google Scholar] [CrossRef]
- Zoffoli, J.P.; Latorre, B.A.; Naranjo, P. Hairline, a Postharvest Cracking Disorder in Table Grapes Induced by Sulfur Dioxide. Postharvest Biol Technol 2008, 47, 90–97. [Google Scholar] [CrossRef]
- Shi, Y.; Li, B.J.; Su, G.; Zhang, M.; Grierson, D.; Chen, K.S. Transcriptional Regulation of Fleshy Fruit Texture. J Integr Plant Biol 2022, 64, 1649–1672. [Google Scholar] [CrossRef]
- Yu, J.; Li, J.; Hong, Z.; Zeng, Q.; Fu, Y.; Deng, R.; Xu, K.; Huang, Z.; Zhu, M. VvPIP1;1 Plays a Role in Grape Berry Cracking by Regulating Water Uptake. Hortic Plant J 2025, 11, 1506–1516. [Google Scholar] [CrossRef]
- Khadivi-Khub, A. Physiological and Genetic Factors Influencing Fruit Cracking. Acta Physiol Plant 2015, 37. [Google Scholar] [CrossRef]
- Santos, M.; Egea-Cortines, M.; Gonçalves, B.; Matos, M. Molecular Mechanisms Involved in Fruit Cracking: A Review. Front Plant Sci 2023, 14. [Google Scholar] [CrossRef]
- Simon, G. Review on Rain Induced Fruit Cracking of Sweet Cherries (Prunus Avium L.), Its Causes and Possibilities of Prevention. Int J Hortic Sci 2006, 12, 3–17. [Google Scholar] [CrossRef]
- Dashrath, S.; Nrcg, R.; Parhe, S.D.; Bhagwat, S.; Rajya, M.; Bagaitdar Sangh Pune, D. Berry Cracking; Its Causes and Remedies in Grapes-A Review. Trend in Biosciences 2017, 10, 2. [Google Scholar]
- La Spada, P.; Continella, A.; Dominguez, E.; Heredia, A.; Gentile, A. Improving Cuticle Thickness and Quality Traits in Table Grape Cv. ‘Italia’ Using Pre-Harvest Treatments. Plants 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Brüggenwirth, M.; Knoche, M. Cell Wall Swelling, Fracture Mode, and the Mechanical Properties of Cherry Fruit Skins Are Closely Related. Planta 2017, 245, 765–777. [Google Scholar] [CrossRef]
- Yang, Z.; Wu, Z.; Zhang, C.; Hu, E.; Zhou, R.; Jiang, F. The Composition of Pericarp, Cell Aging, and Changes in Water Absorption in Two Tomato Genotypes: Mechanism, Factors, and Potential Role in Fruit Cracking. Acta Physiol Plant 2016, 38. [Google Scholar] [CrossRef]
- Wang, J.; Gao, X.; Ma, Z.; Chen, J.; Liu, Y. Analysis of the Molecular Basis of Fruit Cracking Susceptibility in Litchi Chinensis Cv. Baitangying by Transcriptome and Quantitative Proteome Profiling. J Plant Physiol 2019, 234–235, 106–116. [Google Scholar] [CrossRef]
- Guirao, A.; Valverde, J.M.; Díaz-Mula, H.M.; Valero, D.; Serrano, M.; Martínez-Romero, D. Role of Pre-Harvest Sorbitol–Calcium Treatments in Controlling Berry Drop in Bagged Table Grapes of the “Doña María” Variety. Horticulturae 2024, 10. [Google Scholar] [CrossRef]
- Deng, Y.; Wu, Y.; Li, Y.; Zhang, P.; Yang, M.; Shi, C.; Zheng, C.; Yu, S. A Mathematical Model for Predicting Grape Berry Drop during Storage. J Food Eng 2007, 78, 500–511. [Google Scholar] [CrossRef]
- Meneses, M.; García-Rojas, M.; Muñoz-Espinoza, C.; Carrasco-Valenzuela, T.; Defilippi, B.; González-Agüero, M.; Meneses, C.; Infante, R.; Hinrichsen, P. Transcriptomic Study of Pedicels from GA3-Treated Table Grape Genotypes with Different Susceptibility to Berry Drop Reveals Responses Elicited in Cell Wall Yield, Primary Growth and Phenylpropanoids Synthesis. BMC Plant Biol 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Calderón, Á.; Falagán, N.; Terry, L.A.; Alamar, M.C. Biomarkers of Postharvest Resilience: Unveiling the Role of Abscisic Acid in Table Grapes during Cold Storage. Front Plant Sci 2023, 14. [Google Scholar] [CrossRef]
- Rizzuti, A.; Aguilera-Sáez, L.M.; Gallo, V.; Cafagna, I.; Mastrorilli, P.; Latronico, M.; Pacifico, A.; Matarrese, A.M.S.; Ferrara, G. On the Use of Ethephon as Abscising Agent in Cv. Crimson Seedless Table Grape Production: Combination of Fruit Detachment Force, Fruit Drop and Metabolomics. Food Chem 2015, 171, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. lin; Zhang, R. guang Effects of ABA Content on the Development of Abscission Zone and Berry Falling After Harvesting of Grapes. Agric Sci China 2009, 8, 59–67. [Google Scholar] [CrossRef]
- Zhu, M.; Zheng, L.; Zeng, Y.; Yu, J. Susceptibility of Two Grape Varieties to Berry Abscission. Sci Hortic 2022, 304. [Google Scholar] [CrossRef]
- Teker, T.; Soltekin, O. Berry Shattering Phenomena in Vineyards: The Influence of Maximum Temperatures during Flowering Period in an Extreme Year. Sci Hortic 2023, 321. [Google Scholar] [CrossRef]
- Khalil, U.; Rajwana, I.A.; Razzaq, K.; Farooq, U.; Saleem, B.A.; Brecht, J.K. Quality Attributes and Biochemical Changes in White and Colored Table Grapes as Influenced by Harvest Maturity and Ambient Postharvest Storage. South African Journal of Botany 2023, 154, 273–281. [Google Scholar] [CrossRef]
- Sabir, F.K.; Sabir, A.; Payli, T.; Unal, S. Exogenous Melatonin Treatments Maintain Berry Quality and Bioactive Compounds of Minimally Processed Table Grapes (‘Crimson Seedless’) During Cold Storage. Applied Fruit Science 2024, 66, 1609–1617. [Google Scholar] [CrossRef]
- Zenoni, S.; Savoi, S.; Busatto, N.; Tornielli, G.B.; Costa, F. Molecular Regulation of Apple and Grape Ripening: Exploring Common and Distinct Transcriptional Aspects of Representative Climacteric and Non-Climacteric Fruits. J Exp Bot 2023, 74, 6207–6223. [Google Scholar] [CrossRef] [PubMed]
- Ovadia, R.; Oren-Shamir, M.; Kaplunov, T.; Zutahy, Y.; Lichter, A.; Lurie, S. Effects of Plant Growth Regulators and High Temperature on Colour Development in “Crimson Seedless” Grapes. J Hortic Sci Biotech 2013, 88, 387–392. [Google Scholar] [CrossRef]
- Shinomiya, R.; Fujishima, H.; Muramoto, K.; Shiraishi, M. Impact of Temperature and Sunlight on the Skin Coloration of the ‘Kyoho’ Table Grape. Sci Hortic 2015, 193, 77–83. [Google Scholar] [CrossRef]
- Bahar, A.; Kaplunov, T.; Alchanatis, V.; Lichter, A. Evaluation of Methods for Determining Rachis Browning in Table Grapes. Postharvest Biol Technol 2017, 134, 106–113. [Google Scholar] [CrossRef]
- Lichter, A. Rachis Browning in Tablegrapes. Aust J Grape Wine Res 2016, 22, 161–168. [Google Scholar] [CrossRef]
- Hamie, N.; Zoffoli, J.P.; Tarricone, L.; Verrastro, V.; Pérez-Donoso, A.G.; Gambacorta, G. Rachis Browning and Water Loss Description during Postharvest Storage of ‘Krissy’ and ‘Thompson Seedless’ Table Grapes. Postharvest Biol Technol 2022, 184. [Google Scholar] [CrossRef]
- Lichter, A.; Kaplunov, T.; Zutahy, Y.; Daus, A.; Alchanatis, V.; Ostrovsky, V.; Lurie, S. Physical and Visual Properties of Grape Rachis as Affected by Water Vapor Pressure Deficit. Postharvest Biol Technol 2011, 59, 25–33. [Google Scholar] [CrossRef]
- Lo’ay, A.A.; Taha, N.A. Evaluation Rachis Browning Phenomena of ‘Superior Seedless’ Vines Grafted on Different Rootstocks during Shelf Life. Sci Hortic 2020, 261. [Google Scholar] [CrossRef]
- Tóth, A.M. Precision Canopy Management of the Grapevine: Early Defoliation and Girdling. Acta Carolus Robertus 2020, 107–118. [Google Scholar] [CrossRef]
- Timpanaro, N.; Ferlito, F.; Amenta, M.; Torrisi, B.; Allegra, M.; Rapisarda, P.; Romeo, F. V. Effect of Branch Girdling on Berry Traits, Chemical and Sensory Characteristics of ‘Italia’ and ‘Victoria’ Table Grapes. N Z J Crop Hortic Sci 2023, 51, 52–68. [Google Scholar] [CrossRef]
- Tóth, A.M.; Zsófi, Z.; Veres, S. Cane Girdling Influence on the Berry Texture Properties of Three Table Grape Varieties. Horticulturae 2022, 8. [Google Scholar] [CrossRef]
- Goren, R.H.M.; Goldsmichmidt, E.E. Girdling: Physiological Horticultural Aspects, Janick, John Wiley & Sons, Inc, J Horticultural Reviews 2004, 30 ISBN 9780471354208.
- Reynolds, A.G.; De Savigny, C. Influence of Girdling and Gibberellic Acid on Yield Components, Fruit Composition, and Vestigial Seed Formation of ‘Sovereign Coronation’ Table Grapes. Hort Science 2004, 39, 3–541. [Google Scholar] [CrossRef]
- Abu-Zahra, T. Berry Size of Thompson Seedless as Influenced by the Application of Gibberellic Acid and Cane Girdling. Pak J Bot 2010, 42, 3–1755. [Google Scholar]
- Chen, T.; Xu, T.; Shen, L.; Zhang, T.; Wang, L.; Chen, Z.; Wu, Y.; Yang, J. Effects of Girdling and Foliar Fertilization with K on Physicochemical Parameters, Phenolic and Volatile Composition in ‘Hanxiangmi’ Table Grape. Horticulturae 2022, 8. [Google Scholar] [CrossRef]
- Ferrara, G.; Mazzeo, A.; Netti, G.; Pacucci, C.; Matarrese, A.M.S.; Cafagna, I.; Mastrorilli, P.; Vezzoso, M.; Gallo, V. Girdling, Gibberellic Acid, and Forchlorfenuron: Effects on Yield, Quality, and Metabolic Profile of Table Grape Cv. ‘Italia’. Am J Enol Vitic 2014, 65, 381–387. [Google Scholar] [CrossRef]
- Soltekin, O.; Candemir, A.; Altindisli, A. Effects of Cane Girdling on Yield, Fruit Quality and Maturation of (Vitis Vinifera L.) Cv. ‘Flame Seedless’. BIO Web Conf 2016, 7, 01032. [Google Scholar] [CrossRef]
- Yamane, T.; Shibayama, K. Effects of Trunk Girdling and Crop Load Levels on Fruit Quality and Root Elongation in ‘Aki Queen’ Grapevines. J Japan Soc Hort Sci 2006, 75, 6–439. [Google Scholar] [CrossRef]
- İşçi, B.; Kacar, E.; Altındişli, A. The Effects of Some Exogenous Applications on Quality in ‘Crimson Seedless’ Grape. Erwerbs-Obstbau 2020, 62, 87–100. [Google Scholar] [CrossRef]
- Zhu, M.; Yu, J.; Xu, Y.; Yang, G. Effect of Girdling on Anthocyanin Content and Quality of Spine Grape Berries. J Plant Growth Regul 2022, 41, 65–73. [Google Scholar] [CrossRef]
- Crupi, P.; Antonacci, D.; Savino, M.; Genghi, R.; Perniola, R.; Coletta, A. Girdling and Gibberellic Acid Effects on Yield and Quality of a Seedless Red Table Grape for Saving Irrigation Water Supply. Eur J Agron 2016, 80, 21–31. [Google Scholar] [CrossRef]
- Xi, X.; Zha, Q.; He, Y.; Tian, Y.; Jiang, A. Influence of Cluster Thinning and Girdling on Aroma Composition in ‘Jumeigui’ Table Grape. Sci Rep 2020, 10. [Google Scholar] [CrossRef]
- Trentham, W.R.; Sams, C.E.; Conway, W.S. Histological Effects of Calcium Chloride in Stored Apples. J Amer Soc Hort Sci 2008, 133, 4–487. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Serrano, M.; Guillén, F.; Castillo, S.; Martínez-Romero, D.; Valero, D.; Zapata, P.J. Methyl Jasmonate Effects on Table Grape Ripening, Vine Yield, Berry Quality and Bioactive Compounds Depend on Applied Concentration. Sci Hortic 2019, 247, 380–389. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Zapata, P.J.; Castillo, S.; Martínez-Romero, D.; Valero, D.; Serrano, M.; Guillén, F. Preharvest Salicylate Treatments Enhance Antioxidant Compounds, Color and Crop Yield in Low Pigmented-Table Grape Cultivars and Preserve Quality Traits during Storage. Antioxidants 2020, 9, 1–17. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Giménez, M.J.; Zapata, P.J.; Guillén, F.; Valverde, J.M.; Serrano, M.; Valero, D. Preharvest Application of Methyl Salicylate, Acetyl Salicylic Acid and Salicylic Acid Alleviated Disease Caused by Botrytis Cinerea through Stimulation of Antioxidant System in Table Grapes. Int J Food Microbiol 2020, 334. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Giménez, M.J.; Serna-Escolano, V.; Guillén, F.; Valero, D.; Serrano, M.; García-Martínez, S.; Terry, L.A.; Alamar, M.C.; Zapata, P.J. Oxalic Acid Preharvest Treatment Improves Colour and Quality of Seedless Table Grape ‘Magenta’ Upregulating on-Vine Abscisic Acid Metabolism, Relative VvNCED1 Gene Expression, and the Antioxidant System in Berries. Front Plant Sci 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Wan, Y.; Qin, G.; Xu, Y. Induction of Defense Responses against Alternaria Rot by Different Elicitors in Harvested Pear Fruit. Appl Microbiol Biotechnol 2006, 70, 729–734. [Google Scholar] [CrossRef]
- Kawano, T.; Furuichi, T. Salicylic Acid: a Plant Hormone, first edition, Hayat, S; A. A. Springer Dordrecht, 2007, 10, ISBN–10. [Google Scholar]
- Zheng, X.; Jing, G.; Liu, Y.; Jiang, T.; Jiang, Y.; Li, J. Expression of Expansin Gene, MiExpA1, and Activity of Galactosidase and Polygalacturonase in Mango Fruit as Affected by Oxalic Acid during Storage at Room Temperature. Food Chem 2012, 132, 849–854. [Google Scholar] [CrossRef]
- Giménez, M.J.; Valverde, J.M.; Valero, D.; Guillén, F.; Martínez-Romero, D.; Serrano, M.; Castillo, S. Quality and Antioxidant Properties on Sweet Cherries as Affected by Preharvest Salicylic and Acetylsalicylic Acids Treatments. Food Chem 2014, 160, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Giménez, M.J.; Valverde, J.M.; Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Serrano, M.; Moral, J.; Castillo, S. Methyl Salicylate Treatments of Sweet Cherry Trees Improve Fruit Quality at Harvest and during Storage. Sci Hortic 2015, 197, 665–673. [Google Scholar] [CrossRef]
- Giménez, M.J.; Serrano, M.; Valverde, J.M.; Martínez-Romero, D.; Castillo, S.; Valero, D.; Guillén, F. Preharvest Salicylic Acid and Acetylsalicylic Acid Treatments Preserve Quality and Enhance Antioxidant Systems during Postharvest Storage of Sweet Cherry Cultivars. J Sci Food Agric 2017, 97, 1220–1228. [Google Scholar] [CrossRef]
- Dar, T.A.; Uddin, M.; Khan, M.M.A.; Hakeem, K.R.; Jaleel, H. Jasmonates Counter Plant Stress: A Review. Environ Exp Bot 2015, 115, 49–57. [Google Scholar] [CrossRef]
- Martínez-Esplá, A.; Serrano, M.; Valero, D.; Martínez-Romero, D.; Castillo, S.; Zapata, P.J. Enhancement of Antioxidant Systems and Storability of Two Plum Cultivars by Preharvest Treatments with Salicylates. Int J Mol Sci 2017, 18. [Google Scholar] [CrossRef]
- Martínez-Esplá, A.; Zapata, P.J.; Valero, D.; Martínez-Romero, D.; Díaz-Mula, H.M.; Serrano, M. Preharvest Treatments with Salicylates Enhance Nutrient and Antioxidant Compounds in Plum at Harvest and after Storage. J Sci Food Agric 2018, 98, 2742–2750. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Zhang, D.; Zhang, H.; Jiang, G.; Su, X.; Qu, H.; Jiang, Y.; Duan, X. Physiological and Biochemical Response of Harvested Plum Fruit to Oxalic Acid during Ripening or Shelf-Life. Food Research International 2011, 44, 1299–1305. [Google Scholar] [CrossRef]
- Kumar, D. Salicylic Acid Signaling in Disease Resistance. Plant Science 2014, 228, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Min, D.; Li, F.; Ji, N.; Meng, D.; Li, L. Synergistic Effects of l -Arginine and Methyl Salicylate on Alleviating Postharvest Disease Caused by Botrysis Cinerea in Tomato Fruit. J Agric Food Chem 2017, 65, 4890–4896. [Google Scholar] [CrossRef]
- Chen, J.Y.; Wen, P.F.; Kong, W.F.; Pan, Q.H.; Zhan, J.C.; Li, J.M.; Wan, S.B.; Huang, W.D. Effect of Salicylic Acid on Phenylpropanoids and Phenylalanine Ammonia-Lyase in Harvested Grape Berries. Postharvest Biol Technol 2006, 40, 64–72. [Google Scholar] [CrossRef]
- Oraei, M.; Panahirad, S.; Zaare-Nahandi, F.; Gohari, G. Pre-Véraison Treatment of Salicylic Acid to Enhance Anthocyanin Content of Grape (Vitis Vinifera L.) Berries. J Sci Food Agric 2019, 99, 5946–5952. [Google Scholar] [CrossRef]
- Blanch, G.P.; Gómez-Jiménez, M.C.; del Castillo, M.L.R. Exogenous Salicylic Acid Improves Phenolic Content and Antioxidant Activity in Table Grapes. Plant Foods for Human Nutrition 2020, 75, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Habibi, F.; Ramezanian, A.; Rahemi, M.; Eshghi, S.; Guillén, F.; Serrano, M.; Valero, D. Postharvest Treatments with γ-Aminobutyric Acid, Methyl Jasmonate, or Methyl Salicylate Enhance Chilling Tolerance of Blood Orange Fruit at Prolonged Cold Storage. J Sci Food Agric 2019, 99, 6408–6417. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Esplá, A.; Zapata, P.J.; Valero, D.; García-Viguera, C.; Castillo, S.; Serrano, M. Preharvest Application of Oxalic Acid Increased Fruit Size, Bioactive Compounds, and Antioxidant Capacity in Sweet Cherry Cultivars (Prunus Avium L.). J Agric Food Chem 2014, 62, 3432–3437. [Google Scholar] [CrossRef]
- Flores, G.; Ruiz del Castillo, M.L. Variations in Ellagic Acid, Quercetin and Myricetin in Berry Cultivars after Preharvest Methyl Jasmonate Treatments. Journal of Food Composition and Analysis 2015, 39, 55–61. [Google Scholar] [CrossRef]
- Flores, G.; Ruiz del Castillo, M.L. Accumulation of Anthocyanins and Flavonols in Black Currants (Ribes Nigrum L.) by Pre-Harvest Methyl Jasmonate Treatments. J Sci Food Agric 2016, 96, 4026–4031. [Google Scholar] [CrossRef]
- Portu, J.; Santamaría, P.; López-Alfaro, I.; López, R.; Garde-Cerdán, T. Methyl Jasmonate Foliar Application to Tempranillo Vineyard Improved Grape and Wine Phenolic Content. J Agric Food Chem 2015, 63, 2328–2337. [Google Scholar] [CrossRef]
- Portu, J.; López, R.; Baroja, E.; Santamaría, P.; Garde-Cerdán, T. Improvement of Grape and Wine Phenolic Content by Foliar Application to Grapevine of Three Different Elicitors: Methyl Jasmonate, Chitosan, and Yeast Extract. Food Chem 2016, 201, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Gil-Muñoz, R.; Fernández-Fernández, J.I.; Crespo-Villegas, O.; Garde-Cerdán, T. Elicitors Used as a Tool to Increase Stilbenes in Grapes and Wines. Food Research International 2017, 98, 34–39. [Google Scholar] [CrossRef]
- Sun, C.; Zhu, C.; Tang, Y.; Ren, D.; Cai, Y.; Zhou, G.; Wang, Y.; Xu, L.; Zhu, P. Inhibition of Botrytis Cinerea and Control of Gray Mold on Table Grapes by Calcium Propionate. Food Quality and Safety 2021, 5. [Google Scholar] [CrossRef]
- Young, P.R.; Lashbrooke, J.G.; Alexandersson, E.; Jacobson, D.; Moser, C.; Velasco, R.; Vivier, M.A. The Genes and Enzymes of the Carotenoid Metabolic Pathway in Vitis Vinifera L. BMC Genomics 2012, 13. [Google Scholar] [CrossRef]
- Castellarin, S.D.; Gambetta, G.A.; Wada, H.; Krasnow, M.N.; Cramer, G.R.; Peterlunger, E.; Shackel, K.A.; Matthews, M.A. Characterization of Major Ripening Events during Softening in Grape: Turgor, Sugar Accumulation, Abscisic Acid Metabolism, Colour Development, and Their Relationship with Growth. J Exp Bot 2016, 67, 709–722. [Google Scholar] [CrossRef]
- Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Castillo, S.; Guillén, F.; Martínez-Romero, D.; Serrano, M. Postharvest Treatments with Salicylic Acid, Acetylsalicylic Acid or Oxalic Acid Delayed Ripening and Enhanced Bioactive Compounds and Antioxidant Capacity in Sweet Cherry. J Agric Food Chem 2011, 59, 5483–5489. [Google Scholar] [CrossRef]
- Deng, J.; Bi, Y.; Zhang, Z.; Xie, D.; Ge, Y.; Li, W.; Wang, J.; Wang, Y. Postharvest Oxalic Acid Treatment Induces Resistance against Pink Rot by Priming in Muskmelon (Cucumis Melo L.) Fruit. Postharvest Biol Technol 2015, 106, 53–61. [Google Scholar] [CrossRef]
- Razzaq, K.; Khan, A.S.; Malik, A.U.; Shahid, M.; Ullah, S. Effect of Oxalic Acid Application on Samar Bahisht Chaunsa Mango during Ripening and Postharvest. LWT 2015, 63, 152–160. [Google Scholar] [CrossRef]
- Razavi, F.; Hajilou, J. Enhancement of Postharvest Nutritional Quality and Antioxidant Capacity of Peach Fruits by Preharvest Oxalic Acid Treatment. Sci Hortic 2016, 200, 95–101. [Google Scholar] [CrossRef]
- Zhu, Y.; Yu, J.; Brecht, J.K.; Jiang, T.; Zheng, X. Pre-Harvest Application of Oxalic Acid Increases Quality and Resistance to Penicillium Expansum in Kiwifruit during Postharvest Storage. Food Chem 2016, 190, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Kok, D.; Bal, E. Changes on Bioactive Compounds and Electrochemical Characteristics of Cv. ‘Horoz Karası’ Table Grape (V. Vinifera L.) Induced by Various Doses of Preharvest Applications of Benzoic Acid, Citric Acid and Oxalic Acid at Berry Setting and Verasion Periods. Erwerbs-Obstbau 2019, 61, 17–24. [Google Scholar] [CrossRef]
- Zheng, X.; Tian, S.; Gidley, M.J.; Yue, H.; Li, B. Effects of Exogenous Oxalic Acid on Ripening and Decay Incidence in Mango Fruit during Storage at Room Temperature. Postharvest Biol Technol 2007, 45, 281–284. [Google Scholar] [CrossRef]
- Abdusattarov, B.; Safarov, A.; Toshmatov, B. Determination of the Influence of Packaging Measures of a Variety of Husayni Grapes. In Proceedings of the E3S Web of Conferences; EDP Sciences, April 14 2023, 381. [Google Scholar] [CrossRef]
- Kulapichitr, F.; Asensio, C.; Arpaia, M.L.; Walse, S.; Obenland, D. Effect of Controlled Atmosphere Storage on Key Volatiles and Sensory Perception of Muscat Grapes. ACS Food Science and Technology 2024, 4, 766–772. [Google Scholar] [CrossRef]
- Liguori, G.; Sortino, G.; Gullo, G.; Inglese, P. Effects of Modified Atmosphere Packaging and Chitosan Treatment on Quality and Sensorial Parameters of Minimally Processed Cv. ‘Italia’ Table Grapes. Agronomy 2021, 11. [Google Scholar] [CrossRef]
- Martínez-Romero, D.; Guillén, F.; Castillo, S.; Valero, D.; Serrano, M. Modified Atmosphere Packaging Maintains Quality of Table Grapes. J Food Sci 2003, 68, 1838–1843. [Google Scholar] [CrossRef]
- Matera, A.; Altieri, G.; Genovese, F.; Scarano, L.; Genovese, G.; Pinto, P.; Rashvand, M.; Elshafie, H.S.; Ippolito, A.; Mincuzzi, A.; et al. Impact of the Pre-Harvest Biocontrol Agent and Post-Harvest Massive Modified Atmosphere Packaging Application on Organic Table Grape (Cv. ‘Allison’) Quality during Storage. Appl Sci (Switzerland) 2024, 14. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, J.; Chen, Y.; Wei, J.; Wu, B. Nitric Oxide Treatment Maintains Postharvest Quality of Table Grapes by Mitigation of Oxidative Damage. Postharvest Biol Technol 2019, 152, 9–18. [Google Scholar] [CrossRef]
- Pretel, M.T.; Martínez-Madrid, M.C.; Martínez, J.R.; Carreño, J.C.; Romojaro, F. Prolonged Storage of ‘Aledo’ Table Grapes in a Slightly CO2 Enriched Atmosphere in Combination with Generators of SO2. LWT 2006, 39, 1109–1116. [Google Scholar] [CrossRef]
- Xing, S.; Wang, M.; Zhang, Z.; Yuan, Y.Y.; Song, Z.; Wu, B.; Wei, J. Sulfur Dioxide Enhances Postharvest Grape Resistance to Botrytis Cinerea by Promoting Glutathione Level. Sci Hortic 2024, 334. [Google Scholar] [CrossRef]
- Xing, S.; Tian, Q.; Zheng, Y.; Yuan, Y.Y.; Zhang, Z.; Zhang, Y.; Zhang, H.; Wei, J.; Wu, B. Sulfur Dioxide Enhances the Resistance of Postharvest Grape Berries to Gray Mold through Hydrogen Peroxide Signaling. Postharvest Biol Technol 2025, 221. [Google Scholar] [CrossRef]
- Yan, D.; Yi, H. Transcriptome Analysis Provides Insights into Preservation Mechanism of Postharvest Muscat Hamburg Grapes Treated with SO2. Sci Hortic 2024, 331. [Google Scholar] [CrossRef]
- Li, Z.; Tan, N.; Huang, J.; Wang, J.; Xiao, Y.; Xu, J.; Wang, Q.; Wu, B.; Luo, Z.; Xu, Y. H2O2-Mediated Cell Wall Remodeling and Pectin Demethylesterification Are Involved in Maintaining Postharvest Texture of Table Grape by Sulfur Dioxide. Food Chem 2025, 464. [Google Scholar] [CrossRef]
- de Aguiar, A.C.; Higuchi, M.T.; Yamashita, F.; Roberto, S.R. SO2-Generating Pads and Packaging Materials for Postharvest Conservation of Table Grapes: A Review. Horticulturae 2023, 9. [Google Scholar] [CrossRef]
- Reuveni, M.; Arroyo, C.J.; Ovadia, S. An Effective Hybrid Fungicide Containing Tea Tree Oil and Difenoconazole for Grape Powdery Mildew Management. Agric (Switzerland) 2023, 13. [Google Scholar] [CrossRef]
- El-Abbasy, U.K.; Abdel-Hameed, M.A.; Hatterman-Valenti, H.M.; El-Shereif, A.R.; Abd El-Khalek, A.F. Effectiveness of Oregano and Thyme Essential Oils as Alternatives for Sulfur Dioxide in Controlling Decay and Gray Mold and Maintaining Quality of ‘Flame Seedless’ Table Grape (Vitis Vinifera L.) during Cold Storage. Agronomy 2023, 13. [Google Scholar] [CrossRef]
- Guillén, F.; Zapata, P.J.; Martínez-Romero, D.; Castillo, S.; Serrano, M.; Valero, D. Improvement of the Overall Quality of Table Grapes Stored under Modified Atmosphere Packaging in Combination with Natural Antimicrobial Compounds. J Food Sci 2007, 72. [Google Scholar] [CrossRef]
- Loh, M.Y.; Adzahan, N.M.; Azman, E.M.; Koh, S.P.; Yusof, N.L. Enhancing Cold Tolerance and Quality Characteristics of Carica Papaya Linn through the Application of 1-Methylcyclopropene, Geranium and Lavender Oil. Int J Food Sci Technol 2024, 59, 3245–3257. [Google Scholar] [CrossRef]
- Dhanasekaran, S.; Liang, L.; Gurusamy, S.; Yang, Q.; Zhang, H. Chitosan Stabilized Lemon Essential Oil Nanoemulsion Controls Black Mold Rot and Maintains Quality of Table Grapes. Int J Biol Macromol 2024, 277. [Google Scholar] [CrossRef]
- Walsh, K.B. Postharvest Handling, third edition, Florkowski, W. J.; Banks, N.H; Shewfelt, R.L.; Prussia, S.E. Elsevier Inc., 2014, 8, ISBN–978. [Google Scholar]
- Nedjimi, B. Plant Nutrients and Abiotic Stress Tolerance; Hasanuzzaman, M.; Fujita, M.; Oku, H.; Nahar, K.; Hawrylak-Nowak, B. Springer, Singapore, 2018, 9, pp. 205-220, ISBN 9789811090448.
- Fernández, V.; Brown, P.H. From Plant Surface to Plant Metabolism: The Uncertain Fate of Foliar-Applied Nutrients. Front Plant Sci 2013, 4. [Google Scholar] [CrossRef]
- Hocking, B.; Tyerman, S.D.; Burton, R.A.; Gilliham, M. Fruit Calcium: Transport and Physiology. Front Plant Sci 2016, 7. [Google Scholar] [CrossRef]
- Navarro-León, E.; López-Moreno, F.J.; Fernández, M.A.; Maldonado, J.J.; Yánez, J.; Blasco, B.; Ruiz, J.M. A New Calcium Vectoring Technology: Concentration and Distribution of Ca and Agronomic Efficiency in Pepper Plants. Agronomy 2022, 12. [Google Scholar] [CrossRef]
- Kim, Y.; Castroverde, C.D.M.; Kim, J.H. Natural Allelic Diversity of the Calcium Signaling Regulators in Plants. Mol Cells 2024, 47. [Google Scholar] [CrossRef]
- Shao, H.B.; Song, W.Y.; Chu, L.Y. Advances of Calcium Signals Involved in Plant Anti-Drought. C R Biol 2008, 331, 587–596. [Google Scholar] [CrossRef]
- Halling, D.B.; Liebeskind, B.J.; Hall, A.W.; Aldrich, R.W. Conserved Properties of Individual Ca2+-Binding Sites in Calmodulin. Proc Natl Acad Sci U S A 2016, 113, E1216–E1225. [Google Scholar] [CrossRef] [PubMed]
- Kiselev, K. V.; Dubrovina, A.S. The Role of Calcium-Dependent Protein Kinase (CDPK) Genes in Plant Stress Resistance and Secondary Metabolism Regulation. Plant Growth Regul 2025, 105, 535–552. [Google Scholar] [CrossRef]
- Camm, E.L.; Towers, G.H.N. Phenylalanine Ammonia Lyase. Phytochemistry 1973, 12, 961–973. [Google Scholar] [CrossRef]
- Griffith, C.; Einhorn, T.C. The Effect of Plant Growth Regulators on Xylem Differentiation, Water and Nutrient Transport, and Bitter Pit Susceptibility of Apple. Sci Hortic 2023, 310. [Google Scholar] [CrossRef]
- Reitz, N.F.; Mitcham, E.J. Lignification of Tomato (Solanum Lycopersicum) Pericarp Tissue during Blossom-End Rot Development. Sci Hortic 2021, 276. [Google Scholar] [CrossRef]
- Reitz, N.F.; Shackel, K.A.; Mitcham, E.J. Differential Effects of Excess Calcium Applied to Whole Plants vs. Excised Fruit Tissue on Blossom-End Rot in Tomato. Sci Hortic 2021, 290. [Google Scholar] [CrossRef]
- Loekito, S.; Afandi; Afandi, A. ; Nishimura, N.; Koyama, H.; Senge, M. The Effects of Calcium Fertilizer Sprays during Fruit Development Stage on Pineapple Fruit Quality under Humid Tropical Climate. International Journal of Agronomy 2022, 2022. [Google Scholar] [CrossRef]
- Pusittigul, I.; Siriphanich, J.; Juntee, C. Role of Calcium on Internal Browning of Pineapples. Acta Hortic 2014, 1024. [Google Scholar] [CrossRef]
- Youryon, P.; Supapvanich, S.; Kongtrakool, P.; Wongs-Aree, C. Calcium Chloride and Calcium Gluconate Peduncle Infiltrations Alleviate the Internal Browning of Queen Pineapple in Refrigerated Storage. Hortic Environ Biotechnol 2018, 59, 205–213. [Google Scholar] [CrossRef]
- Hou, Y.; Liu, Y.; Zhao, L.; Zhao, Y.; Wu, Z.; Zheng, Y.; Jin, P. EjCML19 and EjWRKY7 Synergistically Function in Calcium Chloride-Alleviated Chilling Injury of Loquat Fruit. Postharvest Biol Technol 2023, 203. [Google Scholar] [CrossRef]
- Li, Z.; Wang, L.; Xie, B.; Hu, S.; Zheng, Y.; Jin, P. Effects of Exogenous Calcium and Calcium Chelant on Cold Tolerance of Postharvest Loquat Fruit. Sci Hortic 2020, 269. [Google Scholar] [CrossRef]
- Talang, H.D.; Dutta, P.; Mukhim, C.; Patil, S. Effect of Calcium, Boron and Sorbitol on Fruit-Set, Yield and Quality in Mango Cv. Himsagar. J Hort Sci, 2017, 11, 2, 166-169.
- Hawkesford, M.; Horst, W.; Kichey, T.; Lambers, H.; Schjoerring, J.; Møller, I.S.; White, P. Functions of Macronutrients. In Marschner’s Mineral Nutrition of Higher Plants, Third Edition, Elsevier Inc., 2011, pp. 135–189 ISBN 9780123849052.
- Maletsika, P.; Liava, V.; Sarrou, E.; Titeli, V.S.; Nasiopoulou, E.; Martens, S.; Karagiannis, E.; Grigoriadou, K.; Molassiotis, A.; Nanos, G.D. Foliar Calcium Effects on Quality and Primary and Secondary Metabolites of White-Fleshed ‘Lemonato’ Peaches. Horticulturae 2023, 9. [Google Scholar] [CrossRef]
- Manganaris, G.A.; Vasilakakis, M.; Diamantidis, G.; Mignani, I. Effect of Calcium Additives on Physicochemical Aspects of Cell Wall Pectin and Sensory Attributes of Canned Peach (Prunus Persica (L) Batsch Cv Andross). J Sci Food Agric 2005, 85, 1773–1778. [Google Scholar] [CrossRef]
- Shah, S.T.; Sajid, M.; Basit, A.; Haleema, B.; Khan, A.; Mohamed, H.I. Revealing Effects of Calcium-Based Formulations Focusing on Membrane Instability, Fruit Quality, Phytochemical Properties, and Disease Incidence in Peach (Prunus Persica) During Storage. Applied Fruit Science 2025, 67. [Google Scholar] [CrossRef]
- Wu, Y.; Dong, X.; Zhu, Y.; Chen, H. Influence of Pre-Harvest Sprays of Calcium Chloride on Fruit Storage Quality and Softening of ‘Fengtangli’ (Prunus Salicina L.) Plum. CYTA - Journal of Food 2025, 23. [Google Scholar] [CrossRef]
- Michailidis, M.; Polychroniadou, C.; Kosmidou, M.A.; Petraki-Katsoulaki, D.; Karagiannis, E.; Molassiotis, A.; Tanou, G. An Early Calcium Loading during Cherry Tree Dormancy Improves Fruit Quality Features at Harvest. Horticulturae 2021, 7. [Google Scholar] [CrossRef]
- Erogul, D. Effect of Preharvest Calcium Treatments on Sweet Cherry Fruit Quality. Not Bot Horti Agrobo 2014, 42, 1–150. [Google Scholar] [CrossRef]
- Guerra, M.; Sanz, M.Á.; Rodríguez-González, Á.; Casquero, P.A. Effect of Sustainable Preharvest and Postharvest Techniques on Quality and Storability of High-Acidity ‘Reinette Du Canada’ Apple. Horticulturae 2022, 8. [Google Scholar] [CrossRef]
- Reitz, N.F.; Mitcham, E.J. Impact of Postharvest Dips with Abscisic Acid, Prohexadione, Calcium, or Water on Bitter Pit Incidence and Apple Physiology. Postharvest Biol Technol 2025, 219. [Google Scholar] [CrossRef]
- Li, S.; Tao, S.; Zhang, J.; Wu, L.; Huan, C.; Zheng, X. Effect of Calcium Treatment on the Storability and Disease Resistance in Preharvest Bagging Mango Fruit during Room Temperature Storage. J Food Process Preserv 2020, 44. [Google Scholar] [CrossRef]
- Asgarian, Z.S.; Karimi, R.; Palou, L. Pre-Harvest Foliar Spraying of Calcium and Zinc Preserves Berries Quality and Mitigates Chilling Injury of Grape during Cold Storage. Sci Hortic 2024, 338. [Google Scholar] [CrossRef]
- Ciccarese, A.; Stellacci, A.M.; Gentilesco, G.; Rubino, P. Effectiveness of Pre- and Post-Veraison Calcium Applications to Control Decay and Maintain Table Grape Fruit Quality during Storage. Postharvest Biol Technol 2013, 75, 135–141. [Google Scholar] [CrossRef]
- Abd El-Wahab, S.M.; Abd El-Zaher, M.H.; Hussein, M.M. Prolonging Shelf Life of Flame Seedless Table Grapes with Pre-Harvest Calcium and Magnesium Treatment. In Proceedings of the BIO Web of Conferences; EDP Sciences, April 23 2025; Vol. 173. [Google Scholar] [CrossRef]
- Zhu, M.; Li, J.; Liu, Y.; Wang, Q.; Fan, Z.; Zeng, J.; Yu, J. Preharvest Nano-Calcium Reduces the Table Grape Berry Abscission by Regulating Ethylene Production During Storage. J Plant Growth Regul 2024, 43, 1400–1409. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.; Han, X.; Chen, R.; Xue, X. Effects of Spraying Calcium Fertilizer on Photosynthesis, Mineral Content, Sugar–Acid Metabolism and Fruit Quality of Fuji Apples. Agronomy 2022, 12. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, C.; Du, H.; Li, W. Impact of Preharvest Foliar Application of Calcium Ascorbate on Quality, Health Promoting Phytochemicals and Postharvest Life of Thompson Seedless Table Grape. J Plant Growth Regul 2025, 44, 879–890. [Google Scholar] [CrossRef]
- Park, Y.S.; Lee, J.C.; Kim, J.H.; Jeong, H.N.; Heo, J.Y. Effect of Calcium Nitrate Treatment on Reduction of Berry Shattering in ‘Cheongsoo’ Grape Cultivars. Not Sci Biol 2022, 14. [Google Scholar] [CrossRef]
- Lang, A.; During, H. Grape Berry Splitting and Some Mechanical Properties of the Skin. Vitis 1990, 29, 61–70. [Google Scholar]
- Zhang, C.; Wu, J. Yun; Cui, L. wen; Fang, J. Gui Mining of Candidate Genes for Grape Berry Cracking Using a Genome-Wide Association Study. J Integr Agric 2022, 21, 2291–2304. [Google Scholar] [CrossRef]
- Huang, X.M.; Wang, H.C.; Lu, X.J.; Yuan, W.Q.; Lu, J.M.; Li, J.G.; Huang, H.B. Cell Wall Modifications in the Pericarp of Litchi (Litchi Chinensis Sonn.) Cultivars That Differ in Their Resistance to Cracking. J Hortic Sci Biotech 2006, 81, 231–237. [Google Scholar] [CrossRef]
- Giné-Bordonaba, J.; Echeverria, G.; Ubach, D.; Aguiló-Aguayo, I.; López, M.L.; Larrigaudière, C. Biochemical and Physiological Changes during Fruit Development and Ripening of Two Sweet Cherry Varieties with Different Levels of Cracking Tolerance. Plant Physiol Biochem 2017, 111, 216–225. [Google Scholar] [CrossRef]
- Yu, J.; Zhu, M.; Bai, M.; Xu, Y.; Fan, S.; Yang, G. Effect of Calcium on Relieving Berry Cracking in Grape (Vitis Vinifera L.) “Xiangfei. ” PeerJ 2020, 8. [Google Scholar] [CrossRef]
- Lara, I.; Heredia, A.; Domínguez, E. Shelf Life Potential and the Fruit Cuticle: The Unexpected Player. Front Plant Sci 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.; Bres, C.; Mauxion, J.P.; Bakan, B.; Rothan, C. Breeding for Cuticle-Associated Traits in Crop Species: Traits, Targets, and Strategies. J Exp Bot 2017, 68, 5369–5387. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.; Nguyen, N.; Hykkerud, A.L.; Häggman, H.; Martinussen, I.; Jaakola, L.; Karppinen, K. Developmental and Environmental Regulation of Cuticular Wax Biosynthesis in Fleshy Fruits. Front Plant Sci 2019, 10. [Google Scholar] [CrossRef]
- Winkler, A.; Brüggenwirth, M.; Ngo, N.S.; Knoche, M. Fruit Apoplast Tension Draws Xylem Water into Mature Sweet Cherries. Sci Hortic 2016, 209, 270–278. [Google Scholar] [CrossRef]
- Adel, A.-Q.; Awad, M.A. Effect of Pre-Harvest Calcium Chloride and Ethanol Spray on Quality of “El-Bayadi” Table Grapes during Storage. Vitis 2013, 52, 2–61. [Google Scholar]
- Chardonnet, C.O.; Sams, C.E.; Trigiano, R.N.; Conway, W.S. Variability of Three Isolates of Botrytis Cinerea Affects the Inhibitory Effects of Calcium on This Fungus. APS, 2000, 90, 7–769. [Google Scholar]
- Zhu, M.; Yu, J.; Tang, W.; Fan, S.; Bai, M.; Chen, M.; Yang, G. Role of Calcium in Regulating Anthocyanin Accumulation in ‘Manicure Finger’ Grape Berries. Sci Hortic 2019, 256. [Google Scholar] [CrossRef]
- Grembecka, M. Sugar Alcohols - Their Role in the Modern World of Sweeteners: A Review. European Food Research and Technology 2015, 241. [Google Scholar] [CrossRef]
- Shankar, P.; Ahuja, S.; Sriram, K. Non-Nutritive Sweeteners: Review and Update. Nutrition 2013, 29, 1293–1299. [Google Scholar] [CrossRef]
- Pleyerová, I.; Hamet, J.; Konrádová, H.; Lipavská, H. Versatile Roles of Sorbitol in Higher Plants: Luxury Resource, Effective Defender or Something Else? Planta 2022, 256. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.; Huang, B. Mechanism of Salinity Tolerance in Plants: Physiological, Biochemical, and Molecular Characterization. Int J Genomics 2014, 2014. [Google Scholar] [CrossRef]
- Tian, G.; Liu, C.; Xu, X.; Xing, Y.; Liu, J.; Lyu, M.; Feng, Z.; Zhang, X.; Qin, H.; Jiang, H.; et al. Effects of Magnesium on Nitrate Uptake and Sorbitol Synthesis and Translocation in Apple Seedlings. Plant Physiol Biochem 2023, 196, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Shen, Y.; Liu, D.H.; Liu, J.; Zhang, J.; Wei, J.; Wang, C.L. A Sorbitol Transporter Gene Plays Specific Role in the Occurrence of Watercore by Modulating the Level of Intercellular Sorbitol in Pear. Plant Science 2022, 317. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, X.; Wu, J.; Li, Y.; Deng, W.; Cao, K.; Li, Z.; Wang, L. Effect of Postharvest Cold Storage and Subsequent Shelf-Life on Fruit Quality and Endogenous Phytohormones in Nectarine Fruit. Postharvest Biol Technol 2024, 218. [Google Scholar] [CrossRef]
- Gómez-Martínez, H. Variation of Fruit Quality Traits in Apricot as Sources for Nutraceutical Breeding. Ph.D. Thesis, IVIA, Valencia, October 2021. [Google Scholar]
- Niu, X.X.; Deng, L.Z.; Wang, H.; Wang, Q.H.; Xu, M.Q.; Li, S. Bin; Okaiyeto, S.A.; Xiao, H.W. Transformation of Cell Wall Pectin Profile during Postharvest Ripening Process Alters Drying Behavior and Regulates the Sugar Content of Dried Plums. Food Chem 2024, 458. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Su, M.; Du, J.; Zhang, X.; Li, X.; Zhang, M.; Hu, Y.; Huan, C.; Ye, Z. Crucial Roles of Sorbitol Metabolism and Energy Status in the Chilling Tolerance of Yellow Peach. Plant Physiol Biochem 2023, 204. [Google Scholar] [CrossRef]
- Lee, J. Sorbitol, Rubus Fruit, and Misconception. Food Chem 2015, 166, 616–622. [Google Scholar] [CrossRef]
- Conde, A.; Soares, F.; Breia, R.; Gerós, H. Postharvest Dehydration Induces Variable Changes in the Primary Metabolism of Grape Berries. Food Res Int 2018, 105, 261–270. [Google Scholar] [CrossRef]
- Guirao, A.; Valverde, J.M.; Solana-Guilabert, A.; Díaz-Mula, H.M.; Valero, D.; Martínez-Romero, D. Impact of Pre-Harvest Sorbitol and Sorbitol-Ca Complex Treatments on the Quality of Fresh Blood Oranges and their Juice after Storage at 8°C. Postharvest Biol Technol 2025, 227. [Google Scholar] [CrossRef]
- Guirao, A.; Martínez-Romero, D.; Solana-Guilabert, A.; Agulló, V.; Díaz-Mula, H.M.; Valverde, J.M. Influence of Preharvest Sorbitol and Calcium-Sorbitol Applications on the Ripening Process and Anthocyanin Biosynthesis in Blood Orange (Citrus Sinensis Cv. Sanguinelli). Food Chem 2025, 481. [Google Scholar] [CrossRef] [PubMed]
- Freitas, J.R.L.; Vendramini, P.H.; Melo, J.O.F.; Eberlin, M.N.; Augusti, R. An Appraisal on the Source-to-Sink Relationship in Plants: An Application of Desorption Electrospray Ionization Mass Spectrometry Imaging. J Braz Chem Soc 2018, 29, 17–23. [Google Scholar] [CrossRef]
- Doehlert, D.C. Ketose Reductase Activity in Developing Maize Endosperm. Plant Physiol 1987, 84, 830–834. [Google Scholar] [CrossRef]
- Singh, M.; Kumar, J.; Singh, S.; Singh, V.P.; Prasad, S.M. Roles of Osmoprotectants in Improving Salinity and Drought Tolerance in Plants: A Review. Rev Environ Sci Biotechnol 2015, 14, 407–426. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive Oxygen Species and Antioxidant Machinery in Abiotic Stress Tolerance in Crop Plants. Plant Physiol Biochem 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Conde, A.; Regalado, A.; Rodrigues, D.; Costa, J.M.; Blumwald, E.; Chaves, M.M.; Gerós, H. Polyols in Grape Berry: Transport and Metabolic Adjustments as a Physiological Strategy for Water-Deficit Stress Tolerance in Grapevine. J Exp Bot 2015, 66, 889–906. [Google Scholar] [CrossRef]
- Fernández, V.; Brown, P.H. Foliar Fertilization: Scientific Principles and Field Pratices, First edition, International Fertilizer Industry Association, Paris, France, 2013, ISBN 979-10-92366-00-6.
- Carreón, Y.J.P.; Pereyra Zarate, A.A.; Pérez Sánchez, A.E.; Díaz-Hernández, O.; González-Gutiérrez, J. Surface Coating with Foliar Fertilizers. Coatings 2024, 14. [Google Scholar] [CrossRef]
- Wu, H.; Xu, H.; Marivingt-Mounir, C.; Bonnemain, J.L.; Chollet, J.F. Vectorizing Agrochemicals: Enhancing Bioavailability via Carrier-Mediated Transport. Pest Manag Sci 2019, 75, 1507–1516. [Google Scholar] [CrossRef]
- Bondada, B.R.; Matthews, M.A.; Shackel, K.A. Functional Xylem in the Post-Veraison Grape Berry. J Exp Bot 2005, 56, 2949–2957. [Google Scholar] [CrossRef]
- Dinant, S.; Lemoine, R. The Phloem Pathway: New Issues and Old Debates. C R Biol 2010, 333, 307–319. [Google Scholar] [CrossRef]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations and Biological Applications. Chem Rev 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Miguel, M.G.; Lourenço, J.P.; Faleiro, M.L. Superparamagnetic Iron Oxide Nanoparticles and Essential Oils: A New Tool for Biological Applications. Int J Mol Sci 2020, 21, 1–24. [Google Scholar] [CrossRef]
- Deng, X.; Li, L.; Zhang, G.; Zhao, X.; Hao, J.; Han, C.; Li, B. Anode Chemistry in Calcium Ion Batteries: A Review. Energy Storage Mater 2022. [CrossRef]
- Junio, J.B.; Chirawatkul, P.; Conato, M.T.; Mercado, C.C. Substitution of Ca2+ in Calcite by Sn2+ and Sr2+ Cations through Ion Exchange Characterized by X-Ray Absorption and Photoelectron Spectroscopies. MRS Adv 2021, 6, 342–349. [Google Scholar] [CrossRef]
- Zabozlaev, A.A.; Oganesyan, É.T.; Pogorelov, V.I. Drug Synthesis Methods and Manufacturing Technology. Solubilization of Poorly Soluble Calcium Salts of Organic Acids with Sorbitol. Pharm Chem J 2007, 41, 8. [Google Scholar] [CrossRef]
- Li, P.; Geng, C.; Li, L.; Li, Y.; Li, T.; Wei, Q.; Yan, D. Calcium-Sorbitol Chelating Technology and Application in Potatoes. Am J Biochem Biotechnol 2020, 16, 96–102. [Google Scholar] [CrossRef]
- Mäikinen, K.K.; Söderling, E. Solubility of Calcium Salts, Enamel, and Hydroxyapatite in Aqueous Solutions of Simple Carbohydrates. Calcif Tissue Int 1984, 36, 67–71. [Google Scholar] [CrossRef]
- Restrepo-Diaz, H.; Benlloch, M.; Fernández-Escobar, R. Plant Water Stress and K+ Starvation Reduce Absorption of Foliar Applied K+ by Olive Leaves. Sci Hortic 2008, 116, 409–413. [Google Scholar] [CrossRef]
- Gholami, A.; Akhlaghi, S.; Shahsavani, S.; Farrokhi, N. Effects of Urea Foliar Application on Grain Yield and Quality of Winter Wheat. Commun Soil Sci Plant Anal 2011, 42, 719–727. [Google Scholar] [CrossRef]
- Asif, M.; Asad, M.S.; Safdar, M.E.; Khan, I.; Akhtar, N.; Hassan, M.A.; Moosa, M.; Baoyi, Z.; Almoallim, H.S.; Ansari, M.J. Agronomic Bio-Fortification of Zinc Improves the Yield and Quality of Fodder Oat. J Ecol Eng 2024, 25, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, U.; Sattar, A.; Sher, A.; Ijaz, M.; Ul-Allah, S.; Ashraf, J.; Shah, A.N.; Nawaz, M. Mineral Biofortification in Crop Plants for Ensuring Food Security; Hasanuzzaman, M. , Muhammad, S.T., Mohsin, T., Adnan, N.S. Springer, 2023, 6, pp.139-157, ISBN 978-981-99-4090-5. 6.
- Bonasia, A.; Lazzizera, C.; Elia, A.; Conversa, G. Pre-Harvest Strategy for Improving Harvest and Post-Harvest Performance of Kale and Chicory Baby Leaves. Plants 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Mangena, P. A Review on the Role of Algae-Nanoparticles as a Sustainable Agricultural Strategy to Improve Resilience against Abiotic Stresses in Leguminous Crops. Plant Nano Biol 2025, 11. [Google Scholar] [CrossRef]
- Xie, P.; Yang, Y.; Li, Y.; Wang, Y.; Bai, B.; Prusky, D.; Li, Y.; Bi, Y. Preharvest Phenylalanine Spraying Alleviates Chilling Injury in Harvested Muskmelons by Maintaining Reactive Oxygen Species Homeostasis. Food Chem 2025, 466. [Google Scholar] [CrossRef]
- Li, S.; Guo, S.; Xu, J.; Cai, Z.X.; Ma, R.; Yu, M.; Shen, Z. Preharvest Calcium Chloride Treatments Enhance Post-Harvest Peach Fruit Resistance to Monilinia Fructicola-Induced Brown Rot Disease. Postharvest Biol Technol 2025, 222. [Google Scholar] [CrossRef]
- Ssemugenze, B.; Ocwa, A.; Kuunya, R.; Gumisiriya, C.; Bojtor, C.; Nagy, J.; Széles, A.; Illés, Á. Enhancing Maize Production Through Timely Nutrient Supply: The Role of Foliar Fertiliser Application. Agronomy 2025, 15. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, W.; Li, D.; Zhou, F.; Chen, X.; Li, C.; Yu, S.; Brestic, M.; Liu, Y.; Yang, X. Glycinebetaine: A Versatile Protectant to Improve Rice Performance against Aluminium Stress by Regulating Aluminium Uptake and Translocation. Plant Cell Rep 2021, 40, 2397–2407. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.H.; Hu, H. Phloem Mobility of Boron Is Species Dependent : Evidence for Phloem Mobility in Sorbitol-Rich Species. Ann Bot 1996, 77, 497–505. [Google Scholar] [CrossRef]
- Hu, H.; Penn, S.C.; Lebrilla, C.B.; Brown, P.H. lsolation and Characterization of Soluble Boron Complexes in Higher Plants' The Mechanism of Phloem Mobility of Boron. Plant physiol 1997, 113, 649–655. [Google Scholar] [CrossRef]
- Raposo, J.C.; Zuloaga, O.; Olazabal, M.A.; Madariaga, J.M. Development of a Modified Bromley Methodology for the Estimation of Ionic Media Effects on Solution Equilibria: Part 5. The Chemical Model of Boric Acid in Aqueous Solution at 25 °C and Comparison with Arsenious Acid. Fluid Phase Equilib 2003, 207, 81–95. [Google Scholar] [CrossRef]
- Broadley, M.; Brown, P.; Cakmak, I.; Rengel, Z.; Zhao, F. Function of Nutrients: Micronutrients. In Marschner’s Mineral Nutrition of Higher Plants, Third Edition, Elsevier Inc., 2011; pp. 191–248 ISBN 9780123849052.
- Will, S.; Eichert, T.; Fernández, V.; Müller, T.; Römheld, V. Boron Foliar Fertilization of Soybean and Lychee: Effects of Side of Application and Formulation Adjuvants. J Plant Nutr Soil Sci 2012, 175, 180–188. [Google Scholar] [CrossRef]
- Al-Jburi, S.M.Y.; Alshammari, M.F. The Effect of Spraying Zinc, Calcium, and Sorbitol on Qualitative Yield Traits of Potato Solanum Tuberosum L. Bionatura 2023, 8. [Google Scholar] [CrossRef]
- Will, S.; Eichert, T.; Fernández, V.; Möhring, J.; Müller, T.; Römheld, V. Absorption and Mobility of Foliar-Applied Boron in Soybean as Affected by Plant Boron Status and Application as a Polyol Complex. Plant Soil 2011, 344, 283–293. [Google Scholar] [CrossRef]
- Liu, C.; Li, Y.; Huo, W.; Li, T.; Wei, Q.; Huang, M.; Geng, C.; Yan, D. Effect of Sorbitol Calcium Chelate on Yield and Calcium Nutrient Absorption of Peanut. Am J Biochem Biotechnol 2021, 17, 160–173. [Google Scholar] [CrossRef]
- Abd El-Azeiz, E.H.; El-Sonbaty, A.E.; El-Sherpiny, M.A. Enhancing the Quantitative and Qualitative Traits of Wheat Grown with Low Mineral Nitrogen Level through Zeolite, Sorbitol and Copper. Egypt J Soil Sci 2024, 64, 1053–1067. [Google Scholar] [CrossRef]
- Maibam, M.; Subba, S.; Chhetri, S.; Majhi, D.; Lepcha, L. Effect of Micronutrient on Quality, Yield and Shelf Life of Mango (Mangifera Indica L.) Cv. Himsagar. Proggressive Horticulture 2024, 56. [Google Scholar] [CrossRef]
- Nasif, S.; Soliman, G.; Farag, M.; Ibrahim, M.; Abd, M.; Nasser, E.-H. Application of Exogenous Polyols, Amino Acids and Girdling to Improve (Vitis Vinifera L.) Crimson Seedless Cv. Berries Coloration, and Postharvest Quality. Am-Eurasian J Agric Environ Sci 2023, 23, 70–80. [Google Scholar] [CrossRef]
- Ma, T.; Hui, Y.; Zhang, L.; Su, B.; Wang, R. Foliar Application of Chelated Sugar Alcohol Calcium Fertilizer for Regulating the Growth and Quality of Wine Grapes. Int J Agric Biol Eng 2022, 15, 153–158. [Google Scholar] [CrossRef]
- Alvarez, R.C.F.; Prado, R.M.; Souza Júnior, J.P.; Oliveira, R.L.L.; Felisberto, G.; Deus, A.C.F.; Cruz, F.J.R. Effects of Foliar Spraying with New Zinc Sources on Rice Seed Enrichment, Nutrition, and Productivity. Acta Agric Scand B Soil Plant Sci 2019. [Google Scholar] [CrossRef]
- Alabdaly, M.M.M.; Abdullah, S.K. Influence of Potassium, Sugar Alcohol, and Boron on Growth and Chemical Composition of Melons under Protected Cultivation. Anbar J Agric Sci 2024, 22, 160–172. [Google Scholar] [CrossRef]
- Lester, G.E.; Grusak, M.A. Field Application of Chelated Calcium: Postharvest Effects on Cantaloupe and Honeydew Fruit Quality. Hort Technol 2004, 14, 1. [Google Scholar] [CrossRef]
| Fruit | Ca Products | Effects | References |
| Apple | Prohexadione-Ca CaCl2 CaCO3 |
-Alleviates bitter pit incidence | [121,137,138] |
| Tomato | CaCl2 | -Reduced blossom-end rot incidence and severity | [122,123] |
| Pineapple | CaCl2 Ca gluconate Ca oxide (CaO) Ca(NO3)2 Ca-boron |
-Reduced IL -Reduced MDA content -Reduced internal browning incidence |
[124,126] |
| Loquat | CaCl2 | -Reduced IL -Reduced MDA content -Reduced internal browning incidence -Promoted accumulation of proline, GABA and polyamines |
[127,128] |
| Mango | CaCl2 | -Enhanced antioxidant enzymatic activity | [139] |
| Peaches | Calcium-silicate CaCl2 Ca(NO3)2 Calcium sulphate (CaSO4) |
-Improved firmness -Reduced weight loss -Promoted bioactive compound content -Delayed softening -Limited rise of PG, PME, Cx, and β-Gal activities |
[131,132,133,134] |
| Sweet cherry | CaCl2 Ca(OH)2 Ca(NO3)2 Calcium caseinate |
-Improved firmness -Enhanced bioactive compounds content and antioxidant activity -Reduced cracking incidence |
[135,136] |
| Table grapes | Ca(NO3)2 CaCl₂ CaAs |
-Reduced weight loss -Reduced berry shattering -Reduced malic acid degradation -Reduced Botrytis cinerea incidence -Higher activity of antioxidant enzymes -Enhanced bioactive compounds content -Reduced MDA -Reduced IL -Inhibed ABA synthesis - Increased fruit firmness and Ca pectate -Inhibed ethylene production by supressing VvACO1 expression -Reduced cracking incidence |
[25,30,140,141,142,143,144,145,146] |
| Plant | Minerals Applied | Effect | References |
| Mango | B Ca |
+ Total soluble solids +Carotenoids +Ascorbic acid +C: N ratio +Shelf-life +Yield -Acidity - Total soluble solids /acidity ratio |
[129,209] |
| Table grapes | Ca | + Total soluble solids + Total soluble solids /acidity ratio +Total Phenols +Colour and anthocyanidins -Berry shattering +Improved firmness -Respiration rate |
[3,25,177,210,211] |
| Wheat | Cu | +Enhanced nutrient uptake | [208] |
| Peanut | Ca | +Higher crop yield +Fat content +Mineral content +Dry matter |
[207] |
| Blood oranges | Ca | +Red colour of peel and pulp +Anthocyanin content +Naruritin and hesperidin content +Individual sugars accumulation +Enhanced organoleptic properties -K: Ca ratio |
[171,172] |
| Potatoes | Ca Zn |
+Tubers per plant +Tuber weight +Total yield +N, P, and K content |
[188,205] |
| Rice | Zn | +Yield +Protein synthesis +Seed production +Dry matter |
[212] |
| Melon | B | +Plant growth +Dry matter |
[213] |
| Lychee | B | +B absorption | [204] |
| Soybeans | B | +B absorption | [204,206] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




