Submitted:
19 September 2025
Posted:
22 September 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Historical Overview of Insecticide Use for Vector Control in India
Insecticide Resistance Among Mosquito Vectors in India
Mechanisms of Insecticide Resistance in Indian Vector Mosquitoes
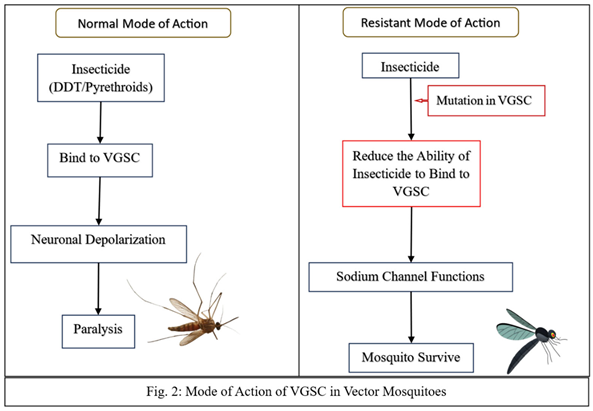
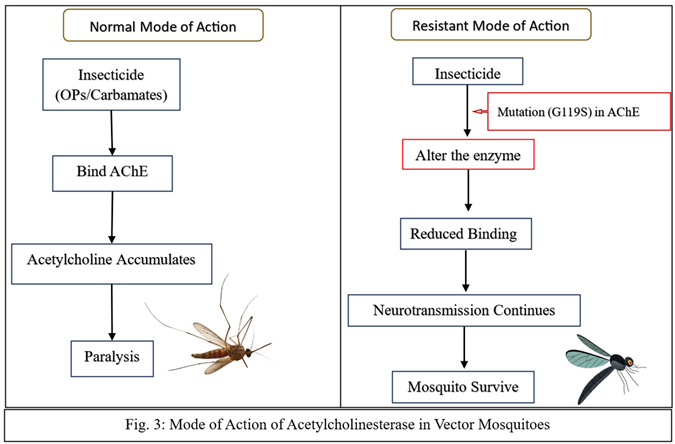
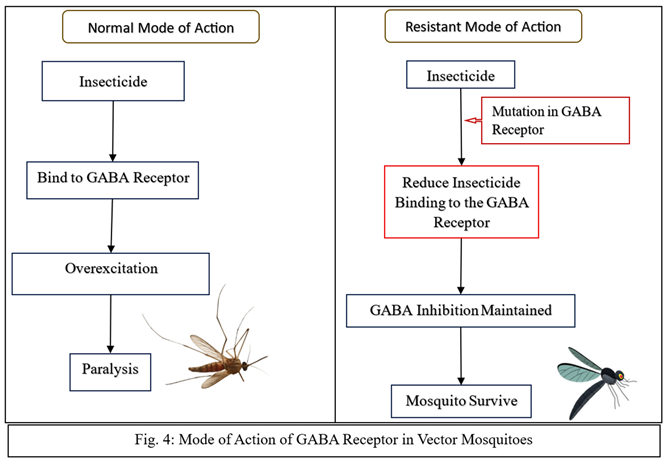
Target Site Resistance
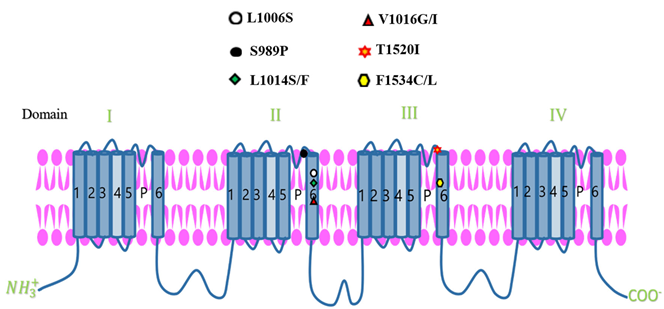
| Mosquito species | Mutation | Transmembrane Domain / gene | Reference(s) |
| A. Aegypti | V1016G, S989P, L1006S | ΙΙ | Kumawat et al., 2021; Saha et al., 2019; Kaura et al., 2022 |
| F1534C, F1534L, T1520I, | ΙΙΙ | Kumawat et al., 2021; Kushwah et al., 2015; Saha et al., 2019 |
|
| G119S | Ace-1 | Muthusamy and Shivakumar, 2015 | |
| A. albopictus | T1520I, F1534C | ΙΙΙ | Modak et al., 2022 |
| An. culicifacies | L1014F, L1014S | ΙΙ | Singh et al., 2009; Dykes et al., 2015 |
| An. subpictus | L1014F | ΙΙ | Sindhania et al., 2023 |
| An. stephensi | L1014F, L1014S | ΙΙ | Dykes et al., 2016 |
| Cx. quinquefasciatus | G119S | Ace-1 | Misra and Gore 2015 |
| L1014F, L1014S | ΙΙ | Rai & Saha, 2022; Modak et al., 2022 | |
| Culex tritaeniorhynchus | F331W | Ace-1 | Misra and Gore 2015 |
Metabolic Resistance
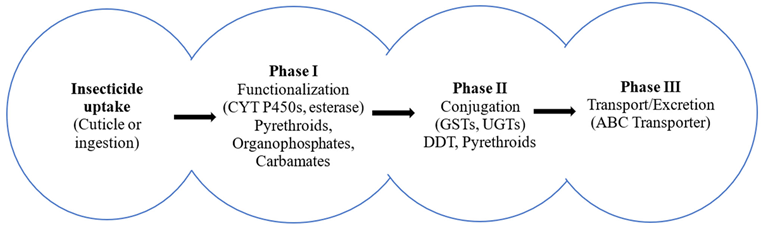
- Uptake: Insecticides penetrate the cuticle or are ingested.
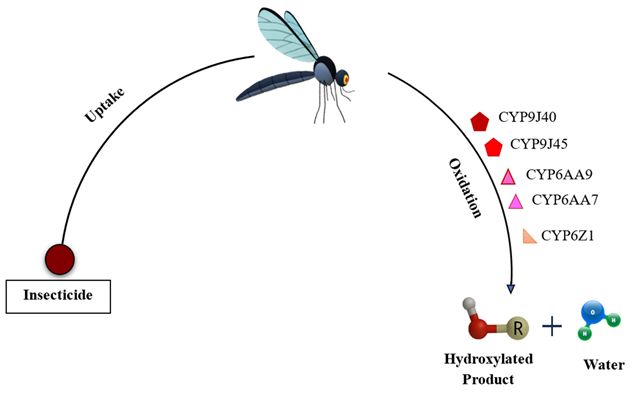
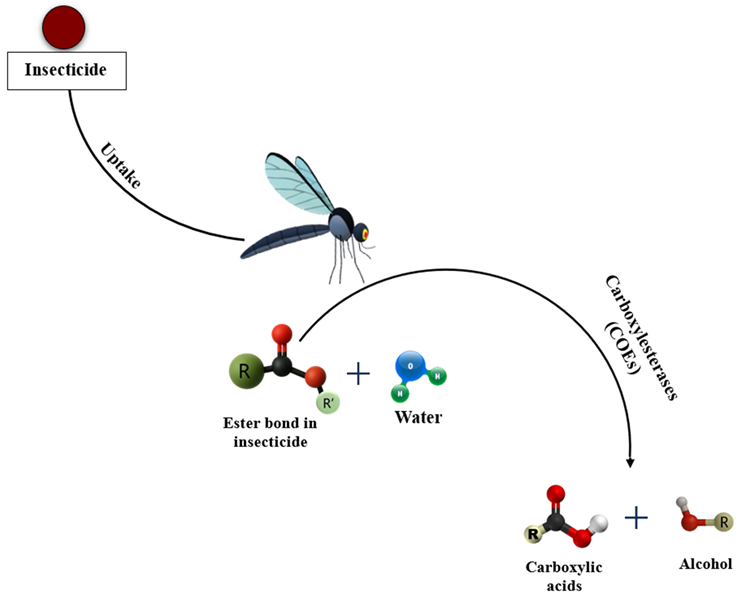
- Phase I (functionalization): Oxidation, reduction, or hydrolysis introduces or exposes polar groups to the substrate. The key actors are cytochrome P450 monooxygenases (P450s) and carboxyl/cholinesterases (esterases) (David et al., 2013).
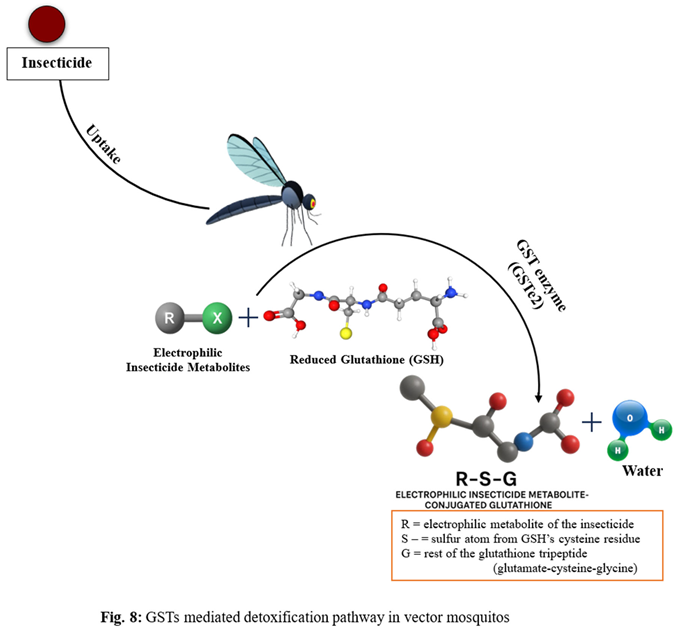
- Phase II (conjugation): Conjugating enzymes (notably glutathione S-transferases, GSTs) attach polar groups (e.g., glutathione) to Phase I products, increasing their solubility (Ranson and Hemingway, 2005).
- Phase III (transport/excretion): Transporter proteins (ATP-binding cassette (ABC) transporters and other efflux systems) move metabolites out of cells and across barriers for excretion (Dermauw and Van Leeuwen, 2014).
| Vector species | Metabolic mechanisms | Insecticide affected | Ref. |
| Anopheles stephensi | GSTe2 GSTe4 duplication, P450 overexpression, esterase | DDT, Pyrethroids |
Dykes et al., 2022 |
| Anopheles culicifacies | P450s (CYP6Z1), esterases, GSTs (GSTe2) | DDT, Pyrethroids, Organophosphates | Sahu et al., 2015; Kareemi et al., 2022 |
| Culex quinquefasciatus | P450s (CYP6/9 families), esterases | Pyrethroids, Organophosphates, Carbamates | Ramkumar et al., 2023 |
| Cx. pipiens | P450s (CYP6AA9) | Bharadwaj et al., 2025 |
Behavioral Resistance
Cuticular Resistance
Conclusions
References
- Adedeji, E. O., Ogunlana, O. O., Fatumo, S., Beder, T., Ajamma, Y., Koenig, R., and Adebiyi, E. (2020). Anopheles metabolic proteins in malaria transmission, prevention and control: a review. Parasites and vectors, 13(1), 465. [CrossRef]
- Baig, M. M., Panigrahi, D. K., Patnaik, M. K., Acharya, P., Choudhury, D., Vijayakumar, B., Sahu, S. S., and Kumar, A. (2021). Susceptibility status of A. aegypti (Linnaeus) and A. albopictus (Skuse) (Diptera: Culicidae) to insecticides in southern Odisha, India. International Journal of Mosquito Research, 8(3), 10–15.
- Balabanidou, V., Grigoraki, L., and Vontas, J. (2018). Insect cuticle: A critical determinant of insecticide resistance. Current Opinion in Insect Science, 27, 68–74. [CrossRef]
- Ballav, S., Chatterjee, M., Sardar, A. A., et al. (2022). Biochemical characterization of insecticide resistance in field population of major JE vectors from northern districts of West Bengal, India. International Journal of Tropical Insect Science, 42, 661–675. [CrossRef]
- Betne, R., and Rajankar, P. (2011, May). Factsheet 38: DDT losing grounds in India – Alternatives imminent. Toxics Link. https://toxicslink.org/wp-content/uploads/2022/08/Factsheet_38_DDT.pdf.
- Bharadwaj, N., Sharma, R., Subramanian, M., Ragini, G., Nagarajan, S. A., and Rahi, M. (2025). Omics approaches in understanding insecticide resistance in mosquito vectors. International Journal of Molecular Sciences, 26(5), 1854. [CrossRef]
- Bharati, M., Saha, P., and Saha, D. (2018). Variation in esterase activity among different A. aegypti L. populations from the Dooars and Terai regions of West Bengal, India. Proceedings of the Zoological Society, 71, 239–247. [CrossRef]
- David, J. P., Ismail, H. M., Chandor-Proust, A., and Paine, M. J. (2013). Role of cytochrome P450s in insecticide resistance: impact on the control of mosquito-borne diseases and use of insecticides on Earth. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 368(1612), 20120429. [CrossRef]
- Dermauw, W., and Van Leeuwen, T. (2014). The ABC gene family in arthropods: comparative genomics and role in insecticide transport and resistance. Insect biochemistry and molecular biology, 45, 89–110. [CrossRef]
- Dhiman, S., Rabha, B., Talukdar, P. K., Das, N. G., Yadav, K., Baruah, I., Singh, L., and Veer, V. (2013). DDT and deltamethrin resistance status of known Japanese encephalitis vectors in Assam, India. The Indian Journal of Medical Research, 138(6), 988–994.
- Dhiman, S., Yadav, K., Acharya, B. N., et al. (2021). Behavioural response of mosquito vectors A. aegypti, An. stephensi and Cx. quinquefasciatus to synthetic pyrethroid and organophosphorus-based slow-release insecticidal paint. Parasites and Vectors, 14, 259. [CrossRef]
- Dykes, C. L., Das, M. K., Eapen, A., Batra, C. P., Ghosh, S. K., Vijayan, V. A., Mishra, S., and Singh, O. P. (2016). Knockdown resistance (kdr) mutations in Indian An. stephensi (Diptera: Culicidae) populations. Journal of Medical Entomology, 53(2), 315–320. [CrossRef]
- Dykes, C. L., Kushwah, R. B., Das, M. K., Sharma, S. N., Bhatt, R. M., Veer, V., Agrawal, O. P., Adak, T., and Singh, O. P. (2015). Knockdown resistance (kdr) mutations in Indian An. culicifacies populations. Parasites and Vectors, 8, 333. [CrossRef]
- Dykes, C. L., Sharma, G., Behera, A. K., Kapoor, N., Paine, M. J. I., Donnelly, M. J., and Singh, O. P. (2022). Tandem duplication of a genomic region encoding glutathione S-transferase epsilon-2 and -4 genes in DDT-resistant Anopheles stephensi strain from India. Scientific reports, 12(1), 17872. [CrossRef]
- Elanga-Ndille, E., Tchouakui, M., et al. (2023). Impact of insecticide resistance on malaria vector competence: a literature review. Malaria Journal, 22, Article 19. [CrossRef]
- Epis, S., Porretta, D., Mastrantonio, V., Comandatore, F., Sassera, D., Rossi, P., Cafarchia, C., Otranto, D., Favia, G., Genchi, C., Bandi, C., and Urbanelli, S. (2014). ABC transporters are involved in defense against permethrin insecticide in the malaria vector Anopheles stephensi. Parasites and vectors, 7, 349. [CrossRef]
- Gan, S. C., Leong, Y. Q., Barhanuddin, M. F. A., Wong, S. T., Wong, S. F., Mak, J. W., and Ahmad, R. (2021). Dengue fever and insecticide resistance in Aedes mosquitoes in Southeast Asia: A review. Parasites and Vectors, 14, Article 315. [CrossRef]
- Ganesh, K. N., Urmila, J., & Vijayan, V. A. (2003). Pyrethroid susceptibility & enzyme activity in two malaria vectors, Anopheles stephensi (Liston) &. A. culicifacies (Giles) from Mysore, India. The Indian journal of medical research, 117, 30–38.
- Gunasekaran, K., Muthukumaravel, S., Sahu, S. S., Vijayakumar, T., & Jambulingam, P. (2011). Glutathione S transferase activity in Indian vectors of malaria: A defense mechanism against DDT. Journal of medical entomology, 48(3), 561–569. [CrossRef]
- Hemingway, J., Ranson, H., Magill, A., Kolaczinski, J., Fornadel, C., Gimnig, J., Coetzee, M., Simard, F., Roch, D. K., Hinzoumbe, C. K., Pickett, J., Schellenberg, D., Gething, P., Hoppé, M., and Hamon, N. (2016). Averting a malaria disaster: Will insecticide resistance derail malaria control? The Lancet, 387(10029), 1785–1788. [CrossRef]
- Insecticide Resistance Action Committee. (2023, August 29). Introduction to resistance. https://irac-online.org/training-centre/resistance/.
- Kareemi, T. I., Mishra, A. K., Chand, S. K., Nirankar, J. K., Vishwakarma, A. K., Tiwari, A., and Bharti, P. K. (2022). Analysis of the insecticide resistance mechanism in An. culicifacies sensu lato from a malaria-endemic state in India. Transactions of the Royal Society of Tropical Medicine and Hygiene, 116(3), 252–260. [CrossRef]
- Kaura, T., Devi, S., Mewara, A., Kaur, J., Singh, N. I., Lovleen, Sharma, S. K., Ratho, R. K., Sehgal, R., and Grover, G. S. (2022). Detection of insecticide susceptibility status and kdr mutation in field-collected A. aegypti from different districts of Punjab, India. Journal of Communicable Diseases, 54(4), 21–28. [CrossRef]
- Krishnan, J., Anbalagan, R., Shukla, A., Subramanian, V., and Srivastava, P. K. (2021). Monitoring of insecticide resistance and exploring the presence of virus in field populations of Cx. gelidus at Thiruvarur District of Tamil Nadu, India. Journal of Communicable Diseases, 53(4), 76–83. [CrossRef]
- Kumar, G., Gupta, S., Kaur, J., Pasi, S., Baharia, R., Mohanty, A. K., Goel, P., Sharma, A., and Rahi, M. (2024). Mapping malaria vectors and insecticide resistance in a high-endemic district of Haryana, India: Implications for vector control strategies. Malaria Journal, 23(1), 107. [CrossRef]
- Kumar, S., Kaushik, G., and Villarreal-Chiu, J. F. (2016). Scenario of organophosphate pollution and toxicity in India: A review. Environmental Science and Pollution Research, 23(10), 9480–9491. [CrossRef]
- Kumawat, N., Meena, S., and Kumari, V. (2021). Insecticide resistance status of Aedes mosquito vector in India: A review. International Journal of Mosquito Research, 8(4), 20–26.
- Kushwah, R. B. S., Kaur, T., Dykes, C. L., et al. (2020). A new knockdown resistance (kdr) mutation, F1534L, in the voltage-gated sodium channel of A. aegypti, co-occurring with F1534C, S989P and V1016G. Parasites and Vectors, 13, 327. [CrossRef]
- Kushwah, R. B., Dykes, C. L., Kapoor, N., Adak, T., and Singh, O. P. (2015). Pyrethroid-resistance and presence of two knockdown resistance (kdr) mutations, F1534C and a novel mutation T1520I, in Indian A. aegypti. PLoS Neglected Tropical Diseases, 9(1), e3332. [CrossRef]
- Liu, N. (2015). Insecticide resistance in mosquitoes: Impact, mechanisms, and research directions. Annual Review of Entomology, 60, 537–559. [CrossRef]
- Mastrantonio, V., Ferrari, M., Negri, A., Sturmo, T., Favia, G., Porretta, D., Epis, S., and Urbanelli, S. (2019). Insecticide Exposure Triggers a Modulated Expression of ABC Transporter Genes in Larvae of Anopheles gambiae s.s. Insects, 10(3), 66. [CrossRef]
- Mehrotra, K. N. (1989). Pesticide resistance in insect-pests Indian scenario. Pesticide Research Journal, 1(2), 95–103. http://www.spsindia.org.in/images/files/2.Mehorotra.pdf.
- Misra, B. R., and Gore, M. (2015). Malathion Resistance Status and Mutations in Acetylcholinesterase Gene (Ace) in Japanese Encephalitis and Filariasis Vectors from Endemic Area in India. Journal of medical entomology, 52(3), 442–446. [CrossRef]
- Modak, M. P., and Saha, D. (2022). First report of F1534C kdr mutation in deltamethrin resistant A. albopictus from northern part of West Bengal, India. Scientific Reports, 12(1), 13653. [CrossRef]
- Modak, P. M., Das, S., Saha, A., and Saha, D. (2024). Insecticide resistance and occurrence of L1014F kdr mutation in wild Cx. quinquefasciatus populations from sub-Himalayan region of West Bengal, India. Journal of Communicable Diseases, 56(3), 81–90. [CrossRef]
- Mukhopadhyay, A. K., Chakraborty, S., Karmakar, P., and Banerjee, P. L. (1996). Insecticidal susceptibility status of An. stephensi (Liston) in selected areas of Calcutta (West Bengal). Indian Journal of Public Health, 40(4), 130–133.
- Mukhopadhyay, A. K., Sinha, S. N., Yadav, R. L., and Narasimham, M. V. (1993). Susceptibility status of Cx. quinquefasciatus in Patna to insecticides. Indian Journal of Public Health, 37(2), 57–60.
- Muthusamy, R., and Shivakumar, M. S. (2015). Susceptibility status of Aedes aegypti (L.) (Diptera: Culicidae) to temephos from three districts of Tamil Nadu, India. Journal of vector borne diseases, 52(2), 159–165.
- Natarajan, E., Verma, V., Velamuri, P., Sreehari, U., and Kamaraju, R. (2014). Neonicotinoids in vector control: In silico approach. Asian Journal of Biomedical and Pharmaceutical Sciences, 4(39), 25–29. [CrossRef]
- National Center for Vector Borne Diseases Control. (2022, December 9). Insecticides: Formulations and dosage (IRS and larvicide). https://ncvbdc.mohfw.gov.in/Doc/tech-specification/Insecticides-Formulations_Dosage(IRS%20and%20Larvicide).pdf.
- National Institute of Malaria Research. (2006). Environmental management plan for the Vector Borne Disease Control Project India. Indian Council of Medical Research. https://nvbdcp.gov.in/Doc/EMP_final.pdf.
- Nayak, P., and Solanki, H. (2021). Pesticides and Indian agriculture – A review. International Journal of Research – Granthaalayah, 9(5), 250–263. [CrossRef]
- Nihad, P. P. M., Subramanian, M., Gunasekaran, K., & Kumar, A. (2023). Response to insecticides and underlying mechanisms of resistance in the field populations of Aedes aegypti Linnaeus (Diptera: Culicidae) in Puducherry, India. Journal of Vector Borne Diseases, 60(4), 393–400. [CrossRef]
- Paul, R. (2018, February 20). List of chemicals used for pest control: Agriculture. Essays, Research Papers and Articles on Agriculture in India. https://www.agricultureinindia.net/pest-control/chemicals-pest-control/list-of-chemicals-used-for-pest-control-agriculture.
- Raghavendra, K., Barik, T. K., Reddy, B. P., Sharma, P., and Dash, A. P. (2011). Malaria vector control: From past to future. Parasitology Research, 108(4), 757–779. [CrossRef]
- Raghavendra, K., Rahi, M., Verma, V., Velamuri, P. S., Kamaraju, D., Baruah, K., Chhibber-Goel, J., and Sharma, A. (2022). Insecticide resistance status of malaria vectors in the malaria endemic states of India: Implications and way forward for malaria elimination. Heliyon, 8(12), e11902. [CrossRef]
- Rahi, M., Mishra, A., Chand, G., Baharia, R., Hazara, R., Singh, S., Khan, S., Sreehari, U., Kamaraju, D., Kumar, G., Gupta, S., Sharma, A., Raghavendra, K., Gunasekaran, K., Singh, O., and Subbarao, S. (2024). Malaria vector bionomics: Countrywide surveillance study on implications for malaria elimination in India. JMIR Public Health and Surveillance, 10, e42050. https://publichealth.jmir.org/2024/1/e42050.
- Rai, P., and Saha, D. (2022). Occurrence of L1014F and L1014S mutations in insecticide resistant Cx. quinquefasciatus from filariasis endemic districts of West Bengal, India [Retracted]. PLoS Neglected Tropical Diseases, 16(1), e0010000. [CrossRef]
- Rai, P., and Saha, D. (2023). Retraction: Occurrence of L1014F and L1014S mutations in insecticide resistant Cx. quinquefasciatus from filariasis endemic districts of West Bengal, India. PLoS Neglected Tropical Diseases, 17(6), e0011417. [CrossRef]
- Rai, P., Bharati, M., Subba, A., and Saha, D. (2019). Insecticide resistance mapping in the vector of lymphatic filariasis, Cx. quinquefasciatus Say from northern region of West Bengal, India. PLoS ONE, 14(5), e0217706. [CrossRef]
- Ramkumar, G., Muthusamy, R., Narayanan, M., Shivakumar, M. S., and Kweka, E. J. (2023). Overexpression of cytochrome P450 and esterase genes involved in permethrin resistance in larvae and adults of Culex quinquefasciatus. Parasitology research, 122(12), 3205–3212. [CrossRef]
- Ranson, H., and Hemingway, J. (2005). Mosquito glutathione transferases. Methods in enzymology, 401, 226–241. [CrossRef]
- Rath, A., Mohanty, I., and Hazra, R. K. (2018). Insecticide susceptibility status of invasive A. albopictus across dengue endemic districts of Odisha, India. Pest Management Science, 74(6), 1431–1440. [CrossRef]
- Saha, A., Das, P., Das, S., and Saha, D. (2025). Overexpression of cytochrome P450 and esterase genes in pyrethroid-resistant wild Culex quinquefasciatus populations from sub-Himalayan West Bengal, India. Parasitology research, 124(6), 68. [CrossRef]
- Saha, P., Ballav, S., Chatterjee, M., Ganguly, S., Sarker, M., Biswas, A. K., Pramanik, T., Basu, N., and Maji, A. K. (2018). The status of susceptibility of Japanese encephalitis vectors to insecticides in endemic areas of northern districts of West Bengal, India. Japanese Journal of Infectious Diseases, 71(2), 91–98. [CrossRef]
- Saha, P., Chatterjee, M., Ballav, S., Chowdhury, A., Basu, N., and Maji, A. K. (2019). Prevalence of kdr mutations and insecticide susceptibility among natural population of A. aegypti in West Bengal. PLoS ONE, 14(4), e0215541. [CrossRef]
- Sahu, S. S., Gunasekaran, K., Vijayakumar, T., and Jambulingam, P. (2015). Triple insecticide resistance in Anopheles culicifacies: A practical impediment for malaria control in Odisha State, India. The Indian journal of medical research, 142 Suppl(Suppl 1), S59–S63. [CrossRef]
- Sahu, S. S., Sonia, T., Dash, S., Gunasekaran, K., and Jambulingam, P. (2019). Insecticide resistance status of three vectors of Japanese encephalitis in east central India. Medical and Veterinary Entomology, 33(2), 213–219. [CrossRef]
- Samal, R. R., and Kumar, S. (2021). Cuticular thickening associated with insecticide resistance in dengue vector, A. aegypti L. International Journal of Tropical Insect Science, 41, 809–820. [CrossRef]
- Sarkar, M., Bhattacharyya, I. K., Borkotoki, A., Goswami, D., Rabha, B., Baruah, I., and Srivastava, R. B. (2009). Insecticide resistance and detoxifying enzyme activity in the principal bancroftian filariasis vector, Cx. quinquefasciatus, in northeastern India. Medical and Veterinary Entomology, 23(2), 122–131. [CrossRef]
- Sharma, N., and Moin, S. (2021). Insecticide resistance, its origin and historical purview in reference to the mosquito vector. Journal of Environment, Science and Technology, 7(2). https://www.gyanvihar.org/journals/wp-content/uploads/2021/09/3.-Nitesh-Review-16-25.pdf.
- Sharma, V. P. (2003). DDT: The fallen angel. Current Science, 85(11), 1532–1537. http://www.jstor.org/stable/24110014.
- Shetty, V., Sanil, D., and Shetty, N. J. (2013). Insecticide susceptibility status in three medically important species of mosquitoes, An. stephensi, A. aegypti and Cx. quinquefasciatus, from Bruhat Bengaluru Mahanagara Palike, Karnataka, India. Pest Management Science, 69(2), 257–267. [CrossRef]
- Sindhania, A., Lohani, H. P., Kona, M. P., Kaur, T., Kaushal, B. R., and Singh, O. P. (2023). Molecular forms of the Indian An. subpictus complex differ in their susceptibility to insecticides and the presence of knockdown resistance (kdr) mutations in the voltage-gated sodium channel. PLoS ONE, 18(2), e0280289. [CrossRef]
- Singh, O. P., Bali, P., Hemingway, J., Subbarao, S. K., Dash, A. P., and Adak, T. (2009). PCR-based methods for the detection of L1014 kdr mutation in An. culicifacies sensu lato. Malaria Journal, 8, 154. [CrossRef]
- Singh, R. K., Kumar, G., and Mittal, P. K. (2014). Insecticide susceptibility status of malaria vectors in India: A review. International Journal of Mosquito Research, 1(1), 5–9.
- Srivastava, P. K., and Dhariwal, A. C. (2016). Compendium on entomological surveillance and vector control in India. National Center for Vector Borne Diseases Control (NCVBDC). https://ncvbdc.mohfw.gov.in/WriteReadData/l892s/Compendium-Entomological-SurveillanceandVector-Control-India.pdf.
- Suman, D. S., Devi, N., Tikar, S. N., Parashar, B. D., and Prakash, S. (2010). Development of insecticide resistance in Cx. quinquefasciatus mosquito from different locations in India. Journal of Pesticide Science, 35(1), 27–32. [CrossRef]
- Thomas, T. G., Prakash, V., Singh, S., Mandal, A. K., and Chauhan, L. S. (2013). Insecticide susceptibility status of Cx. quinquefasciatus Say, the vector of bancroftian filariasis against temephos in Delhi and National Capital Region. Japanese Journal of Infectious Diseases, 66(3), 238–240. [CrossRef]
- Thomas, T. G., Sharma, S. K., Prakash, A., Sokhey, J., and Sharma, B. R. (2000). Insecticide susceptibility status of Cx. tritaeniorhynchus Giles, vector of Japanese encephalitis in Delhi. Japanese Journal of Infectious Diseases, 53(1), 11–14.
- Viswan, K. A., Pushapalatha, E., and Azhahianambi, P. (2016). Application of synthetic insecticide and change in detoxifying enzyme levels in Cx. quinquefasciatus Say. International Journal of Mosquito Research, 3(6), 31–35.
- World Health Organization. (2015). Global plan for insecticide resistance management in malaria vectors (GPIRM). https://www.who.int/publications/i/item/WHO-HTM-GMP-2012.5.
- World Health Organization. (2015). Indoor residual spraying: An operational manual for indoor residual spraying (IRS) for malaria transmission control and elimination (2nd ed.). https://iris.who.int/handle/10665/177242.
- World Health Organization. (2017, September 20). Report of the twentieth WHOPES working group meeting, WHO/HQ, Geneva, 20–24 March 2017. https://www.who.int/publications/i/item/who-htm-ntd-whopes-2017.04.
- World Health Organization. (2020). Global plan for insecticide resistance management (GPIRM). https://www.who.int/publications/i/item/9789241564472.
| Insecticide | Formulation | Dosage | Application method | Duration of action | Target vector | Ref. |
| Organochlorines | ||||||
| DDT | 50% WP* | 1.0-1.5 g/m² | Indoor Residual Spraying (IRS) | Up to 6 months | Anopheles (malaria), Phlebotomus | WHO, 2015; NCVBDC, 2022 |
| Organophosphates | ||||||
| Malathion | 25% WP* | 1-2 g/m² | IRS | 2-3 months | Anopheles, Aedes, Culex | WHO, 2015; NCVBDC, 2022 |
| Malathion | 50% EC* | 1:100 (fogging dilution) | Fogging | 2-3 months | Anopheles, Aedes, Culex | NIMR, 2006; NCVBDC, 2022 |
| Temephos | 50% EC* | 2.5 cc in 10 L | Larvicide in Water | 1 Week | Anopheles, Aedes, Culex | NIMR, 2006; NCVBDC, 2022 |
| Fenthion | 100% EC* | 5 cc in 10 L | Larvicide in Water | 1 Week | Mosquitoes, flies | NIMR, 2006 |
| Fenitrothion | 50% or 25% WP* | 2 g/m² | IRS | 3-6 months | Anopheles, Aedes | WHO, 2015 |
| Pirimiphos-methyl | 50% WP*, 50% EC*, | 1-2 g/m² | IRS | 2-3 months | Anopheles, Aedes, Culex | WHO, 2015 |
| Pyrethroids | ||||||
| Deltamethrin | 2.5% WP*, 62.5 SC* | 20-25 mg/m² | IRS, ITN | 6 months | Anopheles, Phlebotomus | WHO, 2015; NCVBDC, 2022 |
| Alpha-Cypermethrin | 5% WP*, 10% SC* |
25 mg/m² | IRS | 4-6 months | Anopheles, Phlebotomus | WHO, 2015; NCVBDC, 2022 |
| Lambda-Cyhalothrin | 10% WP* | 25 mg/m² | IRS, ITN | 3-6 months | Anopheles | NIMR, 2006; WHO, 2015; NCVBDC, 2022 |
| Cyfluthrin | 10% WP* | 20-50 mg/m² | IRS, ITN | 3-6 months | Mosquitoes, Houseflies | NIMR, 2006; WHO, 2015; NCVBDC, 2022 |
| Bifenthrin | 10% WP* | 20-50 mg/m² | IRS | 3-6 months | Mosquitoes | WHO, 2015; NCVBDC, 2022 |
| Etofenprox | 5% WP* | 100-300 mg/m² | IRS | 3-6 months | Mosquitoes, flies | WHO, 2015 |
| Permethrin | 10% EC* | 1:100 dilution for fogging | Space spraying, Fogging | 1 month | Aedes | NIMR, 2006 |
| Cyphenothrin | 5% EC* | 0.50 mg/m² for spraying 0.35 mg/m² for fogging |
Space spraying, Fogging | 1-3 months | Mosquitoes, flies | NCVBDC, 2022 |
| Carbamates | ||||||
| Propoxur | 20% WP* | 1-2 g/m² | IRS | 3-6 months | Mosquitoes, Houseflies | WHO, 2015 |
| Bendiocarb | 80% WP* | 100-400 mg/m² | IRS | 2-6 months | Mosquitoes, flies | WHO, 2015 |
| Neonicotinoids | ||||||
| Clothianidin | 50% WDG* | IRS | up to 6-9 months | Mosquitoes | WHO, 2017 | |
| State | Mosquito Species | Insecticides Resistant | Reference(s) |
| Assam | An. minimus | Possible Resistant to Organochlorine (DDT) | Singh et al., 2014 |
| Chhattisgarh | An. culicifacies | Organochlorine (DDT), Organophosphate (Malathion), Pyrethroid (Deltamethrin) | Singh et al., 2014 |
| Gujarat | An. culicifacies | Organochlorine (DDT), Organophosphate (Malathion), Pyrethroid (Deltamethrin) | Raghavendra et al., 2022 |
| An. stephensi | Resistant to Organochlorine (DDT), Organophosphate (Malathion) and Possible Resistant to Pyrethroid (Deltamethrin) | Singh et al., 2014 | |
| Haryana | An. culicifacies | Organochlorine (DDT), Organophosphate (Malathion), Pyrethroid (Deltamethrin) | Raghavendra et al., 2022 |
| An. stephensi | Organochlorine (DDT) | Raghavendra et al., 2022 | |
| Jharkhand | An. culicifacies, An. annularis | Resistant to DDT (Organochlorine) and Possible Resistant to Organophosphate (Malathion) | Singh et al., 2014; Raghavendra et al., 2022 |
| An. fluviatilis, | DDT (Organochlorine) | Singh et al., 2014; Raghavendra et al., 2022 | |
| Karnataka | An. culicifacies | Organochlorine (DDT), Organophosphate (Malathion), Pyrethroid (Deltamethrin, alpha-cypermethrin) | Raghavendra et al., 2022 |
| An. stephensi | Organophosphate (Malathion) | Singh et al., 2014 | |
| Kerala | An. stephensi | DDT (Organochlorine) | Singh et al., 2014 |
| Madhya Pradesh | An. culicifacies | Resistant to Organochlorine (DDT), Organophosphate (Malathion) and Possible Resistant to Pyrethroid (Deltamethrin) | Singh et al., 2014; Raghavendra et al., 2022 |
| An. stephensi | Resistant to Organochlorine (DDT) and Possible Resistant to Organophosphate (Malathion) | Singh et al., 2014 | |
| Maharashtra | An. culicifacies | Resistant to Organochlorine (DDT), Organophosphate (Malathion), Pyrethroid (Deltamethrin, cyfluthrin, alpha-cypermethrin, and lambda-cyhalothrin) | Raghavendra et al., 2022 |
|
An. fluviatilis, An. annularis |
Organochlorine (DDT) | Singh et al., 2014 | |
| Odisha | An. culicifacies | Resistant to Organochlorine (DDT) and Possible Resistant to Pyrethroid (Deltamethrin) | Singh et al., 2014; Raghavendra et al., 2022 |
| An. fluviatilis | Possible Resistant to Organochlorine (DDT) | Singh et al., 2014; Raghavendra et al., 2022 | |
| An. minimus | Organochlorine (DDT) | Singh et al., 2014 | |
| Rajasthan | An. culicifacies, An. stephensi | Resistant to Organochlorine (DDT) and Possible Resistant to Organophosphate (Malathion) | Singh et al., 2014 |
| Tamil Nadu | An. culicifacies | Organochlorine (DDT), Pyrethroid (Deltamethrin) | Singh et al., 2014 |
| Tripura | An. minimus | Possible Resistant to Organochlorine (DDT) | Singh et al., 2014 |
| Uttar Pradesh | An. culicifacies | Resistant to Organochlorine (DDT) and Possible Resistant to Organophosphate (Malathion) | Singh et al., 2014 |
| West Bengal | An. stephensi | Organochlorine (DDT), Carbamate (Propoxur), Organophosphate (Malathion) | Mukhopadhyay et al., 1996 |
| State | Mosquito Species | Insecticides | Reference(s) |
| Andhra Pradesh | A. aegypti | DDT (Organochlorine) and Malathion (Organophosphate) | Kumawat et al., 2021 |
| Arunachal Pradesh |
A. Aegypti, A. albopictus |
DDT (Organochlorine) | Kumawat et al., 2021 |
| Assam |
A. aegypti, A. albopictus |
DDT (Organochlorine), Temephos (Organophosphate) | Kumawat et al., 2021 |
| A. albopictus (Guwahati) | Resistant to DDT (Organochlorine) and Incipient Resistant to Deltamethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 | |
|
A. Aegypti, A. albopictus |
DDT (Organochlorine) | Kumawat et al., 2021 | |
| Delhi | A. aegypti | Resistant to DDT and Dieldrin (Organochlorine) and Possible Resistant to Temephos (Organophosphate) | Kumawat et al., 2021 |
| A. albopictus | Resistant to DDT (Organochlorine) and Incipient Resistance to Deltamethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 | |
| Goa | A. aegypti | DDT (Organochlorine) | Kumawat et al., 2021 |
| Haryana (Gurgaon) | A. albopictus | Resistant to DDT (Organochlorine) and Incipient Resistance to Deltamethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 |
| Jharkhand |
A. Aegypti, A. albopictus |
DDT (Organochlorine) | Kumawat et al., 2021 |
| Karnataka | A. aegypti | DDT (Organochlorine), Propoxur (Carbamate), deltamethrin, and Permethrin (Pyrethroid) | Kumawat et al., 2021 |
| Kerala |
A. Aegypti, A. albopictus |
DDT and Dieldrin (Organochlorine) | Kumawat et al., 2021 |
| A. albopictus | Resistant to DDT (Organochlorine) and Incipient Resistance to Deltamethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 | |
| Maharashtra | A. albopictus | DDT (Organochlorine) | Kumawat et al., 2021 |
| A. aegypti | Temephos, Fenthion (Organophosphate) | Kumawat et al., 2021 | |
| Odisha | A. albopictus | DDT (Organochlorine) | Baig et al., 2021; Rath et al., 2018 |
| A. aegypti | Resistant to DDT (Organochlorine), Deltamethrin (Pyrethroids) and Possible Resistance Malathion (Organophosphate) | Baig et al., 2021; Rath et al., 2018 | |
| Rajasthan |
A. aegypti, A. vittatus, A. w-albus |
DDT and Dieldrin (Organochlorine) | Kumawat et al., 2021 |
| A. aegypti | Possible Resistant to Cypermethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 | |
| Tamil Nadu | A. aegypti | Permethrin (Pyrethroid) | Kumawat et al., 2021 |
| Uttarakhand (Haridwar) | A. albopictus | Resistant to DDT (Organochlorine) and Incipient Resistance to Deltamethrin, Permethrin (Pyrethroids) | Kumawat et al., 2021 |
| West Bengal | A. albopictus | DDT (Organochlorine), Permethrin (pyrethroid), propoxur (carbamate), and Temephos (Organophosphate) | Kumawat et al., 2021 |
| A. aegypti | Permethrin (Pyrethroid) and Propoxur (Carbamate) | Kumawat et al., 2021 |
| State | Mosquito Species | Insecticides | Reference(s) |
| Assam | Cx. gelidus, Cx. vishnui |
Suspected Resistant to DDT (Organochlorine), Sensitive to Deltamethrin (Pyrethroid) |
Dhiman et al., 2013 |
| Cx. quinquefasciatus | Resistant to DDT (Organochlorine), Susceptible to Deltamethrin (Pyrethroid) |
Sarkar et al., 2009 | |
| Bihar (Patna) | Cx. quinquefasciatus | Organochlorine (DDT and dieldrin) | Mukhopadhyay et al., 1993 |
| Delhi | Cx. tritaeniorhynchus | Organochlorine (DDT), Organophosphate (Malathion, Fenitrothion), Carbamate (Propoxur) | Thomas et al., 2000 |
| Cx. quinquefasciatus | Temephos (Organophosphate) | Thomas et al., 2013 | |
| Gujarat (Jamnagar) | Cx. quinquefasciatus | Lower resistant to Pyrethroid (alpha-cypermethrin) and Organophosphate (fenthion) | Suman et al., 2010 |
| Karnataka (Bengaluru) | Cx. quinquefasciatus | Highly resistant to Propoxur (Carbamate), Susceptible to Temephos (Organophosphate) | Paul, 2018 |
| Odisha | Cx. vishnui, Cx. tritaeniorhynchus |
DDT (Organochlorine), Deltamethrin (pyrethroids), and malathion (Organophosphate) | Sahu et al., 2019 |
| Cx. bitaeniorhynchus | Remains susceptible to DDT (organochlorine), deltamethrin (pyrethroids), and malathion (organophosphate). | ||
| Punjab (Bathinda) | Cx. quinquefasciatus | High larval Resistant to Organophosphate (temephos and fenthion) and pyrethroids (Lambda Cyhalothrin, alpha cypermethrin, and Cypermethrin) | Suman et al., 2010 |
| Rajasthan (Jodhpur) | Cx. quinquefasciatus | High Resistant to Organophosphate (Temephos, Fenthion), Neemarin (a neem-based insecticide), pyrethroids (Cypermethrin) | Suman et al., 2010 |
| Rajasthan (Bikaner) | Cx. quinquefasciatus | Moderately resistant to organophosphates (temephos, fenthion), Neemarin (a neem-based insecticide), and pyrethroids (alpha-cypermethrin). | |
| Tamil Nadu (Thiruvarur) | Cx. gelidus | Possible Resistant to Deltamethrin (pyrethroids) and malathion (Organophosphate) | Krishnan et al., 2021 |
| West Bengal | Cx. quinquefasciatus | organophosphate (temephos and malathion), carbamate (propoxur), synthetic pyrethroids (deltamethrin, lambdacyhalothrin and permethrin) and organochlorine (DDT) |
Rai et al., 2019 |
| Cx. tritaeniorhynchus | organochlorine (DDT), Deltamethrin (Pyrethroids), malathion (Organophosphate) | Saha et al., 2018 | |
| Cx. pseudovishnui | organochlorine (DDT), Deltamethrin (Pyrethroids), | Saha et al., 2018 | |
| Cx. vishnui | organochlorine (DDT), Deltamethrin (Pyrethroids) | Saha et al., 2018 | |
| Cx. gelidus | Resistant to organochlorine (DDT) and Possible Resistant to Deltamethrin (Pyrethroids) | Saha et al., 2018 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




