Submitted:
18 September 2025
Posted:
22 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
3. Taxonomy and Diversity of Neisseria gonorrhoeae
3.1. Taxonomic Classification
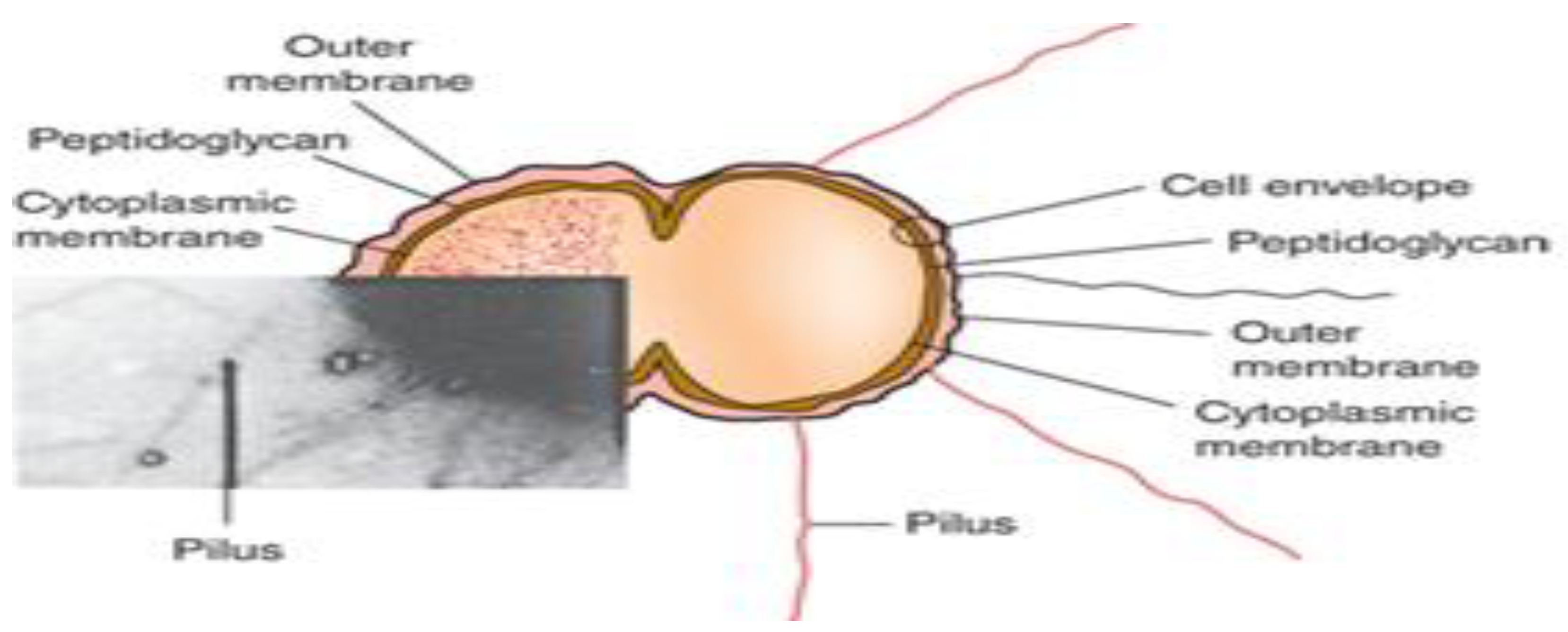
3.2. Morphology and Arrangement
3.3. Phenotypic and Genotypic Diversity of Neisseria gonorrhoeae
3.4. Neisseria gonorrhoeae
4. Virulence Factors of Neisseria gonorrhoeae
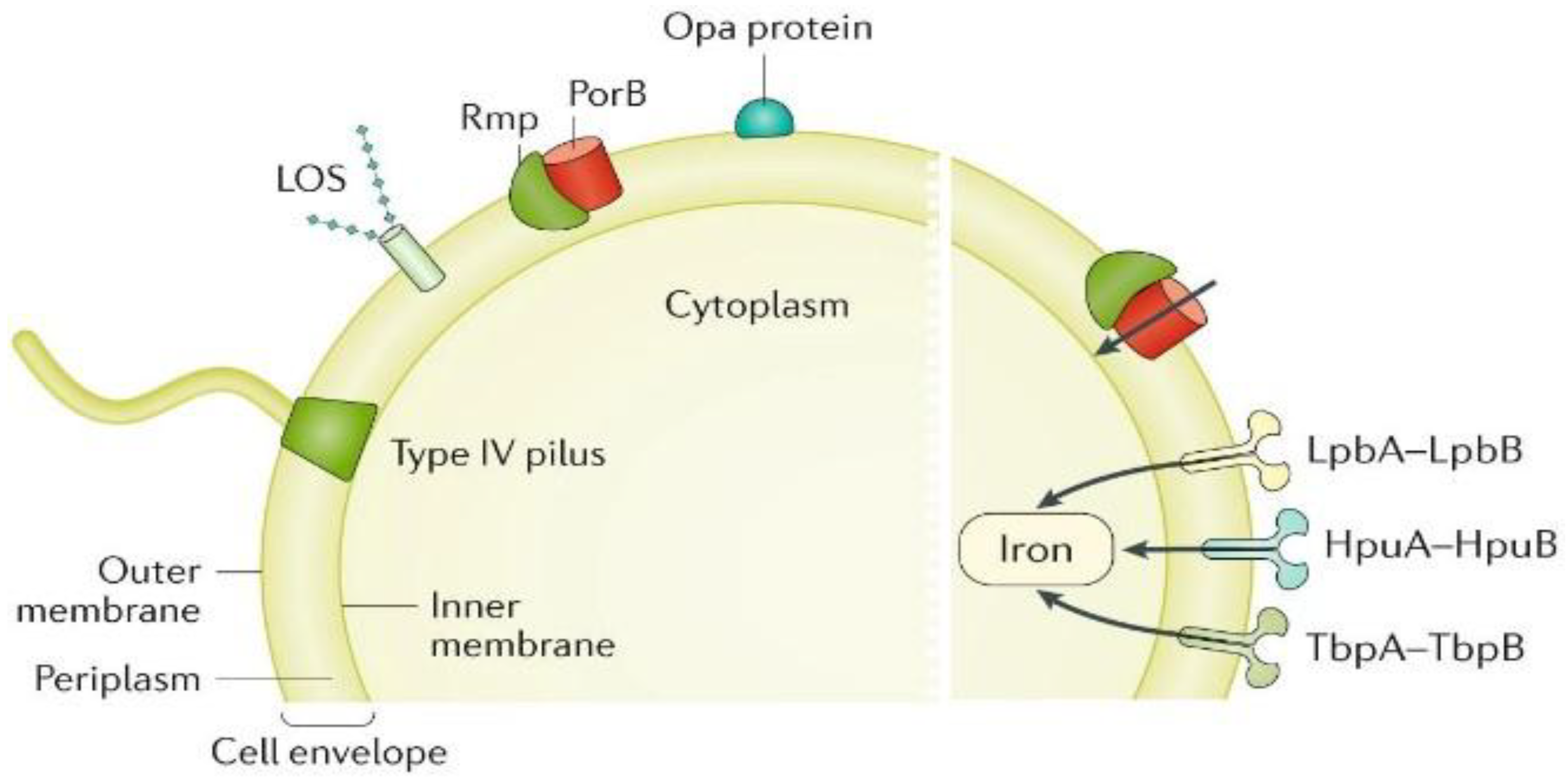
4.1. Pili and Type IV Fimbriae
4.2. Opa Proteins
4.3. Outer Membrane Porins (Por Proteins)
4.4. LOS (lipooligosaccharide)
4.5. Reduction Modifiable Protein (Rmp)
4.6. Transferrin Binding Proteins (Tbp1 and Tbp2)
4.7. IgA Protease
4.8. Phase and Antigenic Variation
5. Clinical Manifestations of Neisseria gonorrhoeae Infection
5.1. Urogenital Infections
5.2. Rectal and Pharyngeal Infections
5.3. Conjunctival Infections
5.4. Disseminated Gonococcal Infection (DGI)
5.5. Co-Infections and Synergistic Risks
5.6. Patient Risk Factors for Neisseria gonorrhoeae Infection
5.7. Age and Gender
5.8. Sexual Behavior
5.9. History of Previous STIs
5.9.1. Coinfection with HIV
5.9.2. Low Socioeconomic Status and Limited Access to Healthcare
5.9.3. Partner’s Risk Profile
5.9.4. Lack of Regular Screening
6. Molecular Characterization of Gonococcal Resistance
6.1. Gene-Based Typing Methods
6.2. Detection of Resistance Determinants
6.3. Rapid Molecular Diagnostics
| Method | Principle | Key Advantages | Limitations | Applications/ References |
|---|---|---|---|---|
| Conventional PCR | DNA amplification using sequence-specific primers under thermal cycling [Mullis and Faloona, 1987] | High sensitivity and specificity; rapid compared to culture; can detect AMR genes | Requires thermal cycler; contamination risk; post-PCR processing needed | Early detection of N. gonorrhoeae; AMR gene surveillance [Vasala et al., 2020] |
| Real-Time PCR (RT-PCR) | Monitors amplification in real time using fluorescent dyes (SYBR Green) or probes (TaqMan) [Tajadini et al., 2014] | Quantitative; rapid turnaround; high sensitivity/specificity; multiplexing possible | Higher cost; requires advanced equipment | Detection in urogenital, rectal, pharyngeal samples; clinical diagnosis and epidemiological studies [Man et al., 2021] |
| Loop-Mediated Isothermal Amplification (LAMP) | Isothermal DNA amplification using strand-displacing polymerase and 4–6 primers targeting multiple regions [Park et al., 2022] | Cost-effective; rapid (<1h); high specificity; minimal equipment; suitable for POC | Primer design complex; less widely standardized | Field-based/POC testing; resource-limited settings [Ahmadi et al., 2025] |
| CRISPR-based assays | Leverages Cas proteins (e.g., Cas12, Cas13) guided by gRNA to detect target DNA/RNA, often coupled with fluorescence or lateral-flow readouts [Kellner et al., 2019] | Ultra-sensitive; rapid (<1 h); portable; amenable to multiplexing and POC applications | Still under development for clinical adoption; may require pre-amplification | Rapid detection of N. gonorrhoeae and AMR markers directly from clinical specimens [Li et al., 2021; de Puig et al., 2021] |
| Sequencing-based platforms (NGS/WGS) | High-throughput sequencing of whole genomes or targeted regions to identify species and resistance markers [Goodwin et al., 2016] | Comprehensive; enables strain typing, AMR marker discovery, and epidemiological surveillance | Higher cost; requires bioinformatics expertise and infrastructure | Resistance mechanism discovery; outbreak investigation; global surveillance of AMR trends [Mortimer and Grad, 2019; Eyre et al., 2021] |
7. Emerging Diagnostic Technologies for Neisseria gonorrhoeae
8. Evolutionary Dynamics of Antimicrobial Resistance in Neisseria gonorrhoeae
8.1. Sulfonamides Resistance
8.2. Penicillin Resistance
8.3. Tetracycline Resistance
8.4. Spectinomycin Resistance
8.5. Quinolones Resistance
8.6. Macrolides Resistance
8.7. Azithromycin Resistance
8.8. Ceftriaxone Resistance
8.9. Cephalosporins Resistance
9. Global Epidemiology and Resistance Trends
10. Surveillance of Gonococcal AMR
11. Diagnostic Approaches: Conventional and Immuno-Molecular Methods
11.1. Immunodetection
11.2. MALDI-TOF Mass Spectrometry
12. Treatment of N. gonorrhoeae Infections
12.1. Emerging Therapeutics
12.2. Solithromycin
12.3. Delafloxacin
12.4. Zoliflodacin
12.5. Gepotidacin
13. Treatment of N. gonorrhoeae Infection
13.1. Historical Overview of Antimicrobials
13.2. Sulfonamides
13.3. Penicillin and other β-Lactams
13.4. Tetracyclines and Macrolides
13.5. Fluoroquinolones
13.6. Cephalosporins
13.7. Current Standard of Care
13.8. Dual therapy and Monotherapy Challenges
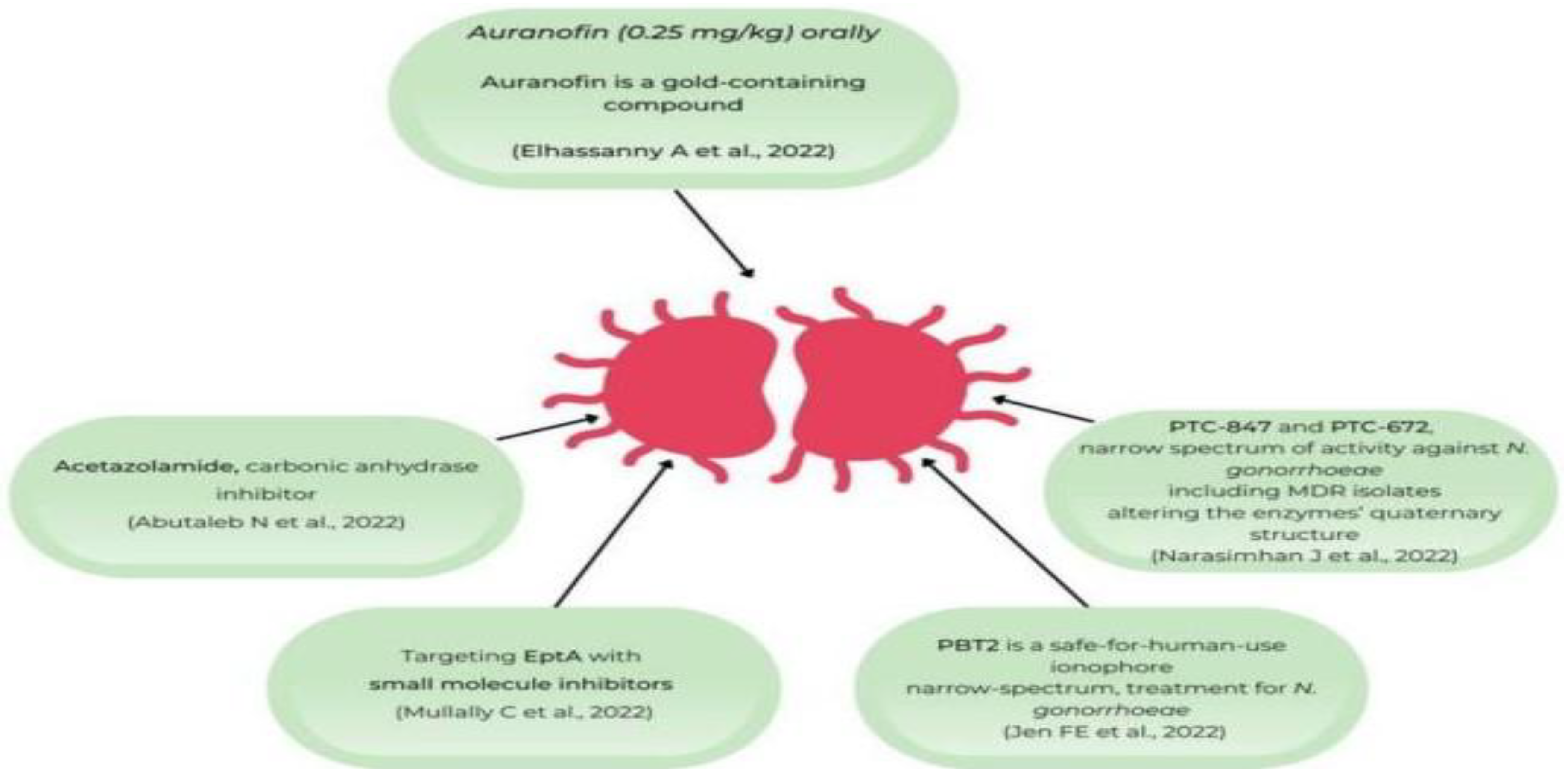
13.9. Novel / Alternatives to Antibiotics
14. Hypervirulent Neisseria gonorrhoeae
14.1. Defining Hypervirulence
14.2. Genetic and Phenotypic Determinants
14.3. Clinical Implications
15. Neisseria gonorrhoeae Amid the COVID-19 Pandemic
15.1. Impact of COVID-19 Measures on Epidemiology and Testing
15.2. Changes in Gonorrhea Incidence and Population Dynamics
15.3. Coinfection with COVID-19
15.4. Disruption of STI Services and Testing
15.5. Trends in Gonorrhea Post-COVID in India
15.6. Implications for Surveillance, Treatment, and Public Health
16. Policy and Public Health Imperatives in Gonorrhea Control
16.1. Core Public Health Measures
16.2. Surveillance Architecture
16.3. Policy, Stewardship, and Equity
16.4. Prevention and Innovation
16.5. Research and Development Priorities
17. Future Directions
18. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Declaration of Competing Interest
References
- Abraham, E.P.; Newton, G.G.F. The structure of cephalosporin C. Biochem. J. 1961, 79, 377–393. [Google Scholar] [CrossRef]
- Ahmadi, Y.; Yu, Y.; Cui, Z.; Huang, W.E.; Andersson, M.I. Loop-mediated isothermal amplification (LAMP) for the diagnosis of sexually transmitted infections: A review. Microb. Biotechnol. 2025, 18. [Google Scholar] [CrossRef]
- Akpomedaye, S.; Kenneth, E.P.; Imarenezor, P.; et al. Prevalence, molecular identification and antibiogram profile of Neisseria gonorrhoeae among individuals in Yenagoa, Bayelsa State, South South, Nigeria. Afr. J. Med. Surg. Public Health Res. 2024, 1, 66. [Google Scholar]
- Allan-Blitz, L.T.; et al. High prevalence of extra-genital chlamydial or gonococcal infections among men who have sex with men and transgender women in Lima, Peru. Int. J. STD AIDS. 2017, 28, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Allen, V.G.; et al. Neisseria gonorrhoeae treatment failure and susceptibility to cefixime in Toronto, Canada. JAMA. 2013, 309, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Amies, C.R. Development of resistance of gonococci to penicillin: An eight-year study. Can. Med. Assoc. J. 1967, 96, 33–35. [Google Scholar] [PubMed]
- Aravinda, A.; Sood, S.; Chaudhry, R.; Kapil, A.; Sharma, P.K.; Gupta, S. A pilot study to determine Neisseria gonorrhoeae–Chlamydia trachomatis coinfection rates in symptomatic patients attending STI clinics, New Delhi, India. Indian J. Dermatol. Venereol. Leprol. 2022, 88, 367–371. [Google Scholar] [CrossRef]
- Ashford, W.A.; Golash, R.G.; Hemming, V.G. Penicillinase-producing Neisseria gonorrhoeae. Lancet 1976, 308, 657–658. [Google Scholar] [CrossRef]
- Ayala, P.; Vasquez, B.; Wetzler, L.; So, M. Neisseria gonorrhoeae porin P1.B induces endosome exocytosis and a redistribution of Lamp1 to the plasma membrane. Infect. Immun. 2002, 70, 5965–5971. [Google Scholar] [CrossRef]
- Bala, M.; Kakran, M.; Singh, V.; Sood, S.; Ramesh, V.; Members of WHO GASP SEAR Network. Monitoring antimicrobial resistance in Neisseria gonorrhoeae in selected countries of the WHO South-East Asia Region between 2009 and 2012: A retrospective analysis. Sex. Transm. Infect. 2013, 89 (Suppl. 4), iv28–iv35. [Google Scholar] [CrossRef]
- Bala, M.; Sood, S. Cephalosporin resistance in Neisseria gonorrhoeae. J. Glob. Infect. Dis. 2010, 2, 284–290. [Google Scholar] [CrossRef]
- Balloux, F.; et al. From theory to practice: Translating whole-genome sequencing (WGS) into the clinic. Trends Microbiol. 2018, 26, 1035–1048. [Google Scholar] [CrossRef]
- Bauernfeind, A.; Jungwirth, R. Antibacterial activity of cefpodoxime in comparison with cefixime, cefdinir, cefetamet, and ceftibuten against clinical isolates, including Neisseria gonorrhoeae. Infection 1991, 19, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Berglund, T.; Unemo, M.; Olcén, P.; Giesecke, J.; Fredlund, H. One year of Neisseria gonorrhoeae isolates in Sweden: The prevalence study of antibiotic susceptibility shows relation to the geographic area of exposure. Int. J. STD AIDS 2002, 13, 109–114. [Google Scholar] [CrossRef]
- Biała, M.; Mączyńska, B.; Starzyński, K.; Rurańska-Smutnicka, D.; Secewicz, A.; Szuba, P.; Szetela, B. Antimicrobial resistance of Neisseria gonorrhoeae isolates among men who have sex with men in Lower Silesia, Poland. Pathogens 2024, 13, 890. [Google Scholar] [CrossRef]
- Bignell, C.; Fitzgerald, M. UK national guideline for the management of gonorrhoea in adults, 2011. Int. J. STD AIDS. 2011, 22, 541–547. [Google Scholar] [CrossRef]
- Bignell, C.; Unemo, M.; European STI Guidelines Editorial Board. 2012 European guideline on the diagnosis and treatment of gonorrhoea in adults. Int. J. STD AIDS 2013, 24, 85–92. [Google Scholar] [CrossRef]
- Bleich, A.T.; Sheffield, J.S.; Wendel, G.D., Jr.; Sigman, A.; Cunningham, F.G. Disseminated gonococcal infection in women. Obstet. Gynecol. 2012, 119, 597–602. [Google Scholar] [CrossRef]
- Boslego, J.W.; Tramont, E.C.; Takafuji, E.T.; Diniega, B.M.; Mitchell, B.S.; Small, J.W.; Khan, W.N.; Stein, D.C. Effect of spectinomycin use on the prevalence of spectinomycin-resistant and penicillinase-producing Neisseria gonorrhoeae. N. Engl. J. Med. 1987, 317, 272–278. [Google Scholar] [CrossRef] [PubMed]
- British Association for Sexual Health and HIV (BASHH). UK National Guideline for the Management of Gonorrhoea, Final Version, 1 April 2025. Available online: https://www.bashh.org.
- Brotzu, G. Ricerche su di un nuovo antibiotico. Lavori dell’Istituto d’Igiene di Cagliari 1948, 2, 1–11. [Google Scholar]
- Brunham, R.C.; Gottlieb, S.L.; Paavonen, J. Pelvic inflammatory disease. N. Engl. J. Med. 2015, 372, 2039–2048. [Google Scholar] [CrossRef] [PubMed]
- Camara, J.; Serra, J.; Ayats, J.; Bastida, T.; Carnicer-Pont, D.; Andreu, A. Molecular characterization of two high-level ceftriaxone-resistant Neisseria gonorrhoeae isolates detected in Catalonia, Spain. J. Antimicrob. Chemother. 2012, 67, 1858–1860. [Google Scholar] [CrossRef]
- Carannante, A.; Forcelli, N.; Spanu, T.; Sanguinetti, M.; Posteraro, B. Evaluation of matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) for identification and clustering of Neisseria gonorrhoeae. BMC Microbiol. 2015, 15, 142. [Google Scholar] [CrossRef]
- Carroll, K.C.; Hobden, J.A.; Miller, S. ; Morse, S et al., 28th ed.; McGraw-Hill Education: New York, NY, USA, 2019. [Google Scholar]
- CDC. Update to CDC's Treatment Guidelines for Gonococcal Infection. Weekly 2020, 69, 1911–1916. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Increases in fluoroquinolone-resistant Neisseria gonorrhoeae among men who have sex with men-United States, 2003, and revised recommendations for gonorrhea treatment, 2004. MMWR Morb. Mortal. Wkly. Rep. 2004, 53, 335–338. [Google Scholar]
- Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm. Rep. 2015, 64, 1–137. [Google Scholar]
- Centers for Disease Control and Prevention. Update to CDC's sexually transmitted diseases treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb. Mortal. Wkly. Rep. 2007, 56, 332–336. [Google Scholar]
- Chen, M.Y.; McNulty, A.; et al. Solithromycin versus ceftriaxone plus azithromycin for the treatment of uncomplicated genital gonorrhoea (SOLITAIRE-U): A randomised phase 3 non-inferiority trial. Lancet Infect. Dis. 2019, 19, 833–842. [Google Scholar] [CrossRef]
- Chico, R.M.; Mayaud, P.; Ariti, C.; Mabey, D.; Ronsmans, C.; Chandramohan, D. Prevalence of malaria and sexually transmitted and reproductive tract infections in pregnancy in sub-Saharan Africa: A systematic review. JAMA 2012, 307, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, S.A.; Dave, J.; Ison, C.A. High-level azithromycin resistance occurs in Neisseria gonorrhoeae as a result of a single point mutation in the 23S rRNA genes. Antimicrob. Agents Chemother. 2010, 54, 3812–3816. [Google Scholar] [CrossRef]
- Chow, E.P.F.; Ong, J.J.; Denham, I.; Fairley, C.K.; Huston, W.M.; Chen, M.Y. Sexually transmitted infection diagnoses and access to a sexual health service before and after the national lockdown for COVID-19 in Melbourne, Australia. Open Forum Infect. Dis. 2020, 8, ofaa536. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. A Study Evaluating Efficacy and Safety of Gepotidacin Compared with Ceftriaxone plus Azithromycin in the Treatment of Uncomplicated Urogenital Gonorrhea. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT04010539.
- Corander, J.; Connor, T.R.; O’Dwyer, C.A.; Kroll, J.S.; Hanage, W.P. Population structure in the Neisseria, and the biological significance of fuzzy species. J. R. Soc. Interface 2012, 9, 1208–1215. [Google Scholar] [CrossRef]
- Cornelissen, C.N.; Anderson, J.E.; Boulton, I.C.; Xiong, J.P.; Biswas, G.D.; Sparling, P.F. The transferrin receptor expressed by gonococcal strain FA1090 is required for the experimental infection of human male volunteers. Mol. Microbiol. 1998, 27, 611–616. [Google Scholar] [CrossRef]
- Costumbrado, J.; Ng, D.K.; Ghassemzadeh, S. Gonococcal Conjunctivitis. In StatPearls [Internet]; Updated 12 September 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459289/; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Cyr, S.S.; Barbee, L.A.; Workowski, K.A.; Bachmann, L.H.; Pham, C.; Schlanger, K.; Torrone, E.A.; Weinstock, H. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1911–1916. [Google Scholar] [CrossRef]
- Dadwal, R.; Paul, S.; Gupta, P.; et al. Stable isotope labeling as a promising tool for rapid drug susceptibility testing in Neisseria gonorrhoeae. Braz. J. Microbiol. 2023, 54, 1819–1825. [Google Scholar] [CrossRef]
- de Puig, H.; Lee, R.A.; Najjar, D.; Tan, X.; Soekensen, L.R.; et al. Minimally instrumented SHERLOCK (miSHERLOCK) for CRISPR-based point-of-care diagnosis of SARS-CoV-2 and emerging variants. Sci. Adv. 2021, 7. [Google Scholar] [CrossRef]
- Deguchi, T.; Nakane, K. Fluoroquinolone Resistance in Neisseria gonorrhoeae. Expert Rev. Anti-Infect. Ther. 2010, 8, 205–215. [Google Scholar]
- Demarco de Hormaeche, R.; Bundell, C.; Chong, H.; Taylor, D.W.; Wildy, P. Definition of a virulence-related antigen of Neisseria gonorrhoeae with monoclonal antibodies and lectins. J. Infect. Dis. 1986, 153, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Demarco, D.E.; Thornsberry, C.; McDougal, L.K. Monoclonal antibodies for the identification of Neisseria gonorrhoeae. J. Clin. Microbiol. 1986, 23, 357–362. [Google Scholar]
- Demczuk, W.; Sidhu, S.; Unemo, M.; Whiley, D.M.; Allen, V.G.; Dillon, J.R.; Martin, I. Neisseria gonorrhoeae Sequence Typing for Antimicrobial Resistance, a Novel Antimicrobial Resistance Multilocus Typing Scheme for Tracking Global Dissemination of N. gonorrhoeae Strains. J. Clin. Microbiol. 2017, 55, 1454–1468. [Google Scholar] [CrossRef] [PubMed]
- Dillon, J.A.; Pagotto, F. Importance of drug resistance in gonococci: From mechanisms to monitoring. Curr. Opin. Infect. Dis. 1999, 12, 35–40. [Google Scholar] [CrossRef]
- Dillon, J.A.; Trecker, M.A.; Thakur, S.D. Two decades of the gonococcal antimicrobial surveillance program in South America and the Caribbean: Challenges and opportunities. Sex. Transm. Dis. 2013, 89 (Suppl. 4), S36–S41. [Google Scholar] [CrossRef]
- Dobson, A.; Cotter, P.D.; Ross, R.P.; Hill, C. Bacteriocin production: A probiotic trait? Appl. Environ. Microbiol. 2012, 78, 1–7. [Google Scholar] [CrossRef]
- Easmon, C.S.; Forster, G.E.; Walker, G.D.; Ison, C.A.; Harris, J.R.; Munday, P.E. Spectinomycin as initial treatment for gonorrhoea. BMJ (Clin. Res. Ed.) 1984, 289, 1032–1034. [Google Scholar] [CrossRef]
- Echenique-Rivera, H.; Díaz, L.; Calderón, M.; et al. Characterization of Neisseria gonorrhoeae strains with hypervirulent phenotypes using whole-genome sequencing. J. Clin. Microbiol. 2011, 49, 3656–3663. [Google Scholar]
- Edwards, J.L.; Butler, E.K. The Pathobiology of Neisseria gonorrhoeae Lower Female Genital Tract Infection. Front. Microbiol. 2011, 2, 102. [Google Scholar] [CrossRef]
- Edwards, J.L.; Apicella, M.A. The molecular mechanisms used by Neisseria gonorrhoeae to initiate infection differ between men and women. Clin Microbiol Rev. 2004, 17, 965–981. [Google Scholar] [CrossRef]
- Enright, M.C.; Spratt, B.G. Multilocus sequence typing. Trends Microbiol. 1999, 7, 482–487. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Gonococcal Antimicrobial Susceptibility Surveillance in the EU/EEA (Euro-GASP). 2020 Euro-GASP results show low cefixime resistance and sporadic ceftriaxone resistance.
- European Centre for Disease Prevention and Control (ECDPC). Gonorrhoea Annual Epidemiological Report for 2017; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2019. [Google Scholar]
- Eyre, D.W.; Town, K.; Street, T.; Fifer, H. Detection in the United Kingdom of the Neisseria gonorrhoeae FC428 clone, with ceftriaxone resistance and intermediate azithromycin resistance, October to December 2018. Euro Surveill. 2019, 24, 1900147. [Google Scholar] [CrossRef]
- Ezewudo, M.N.; Joseph, S.J.; et al. Population structure of Neisseria gonorrhoeae based on whole genome data and its relationship with antibiotic resistance. PeerJ 2015, 3, e806. [Google Scholar] [CrossRef]
- Fifer, H.; Natarajan, U.; Jones, L.; Alexander, S.; Hughes, G.; Golparian, D.; Unemo, M. Failure of Dual Antimicrobial Therapy in Treatment of Gonorrhoea. N. Engl. J. Med. 2016, 374, 2504–2506. [Google Scholar] [CrossRef]
- Fifer, H.; Saunders, J.; Soni, S.; Sadiq, S.T.; FitzGerald, M. 2020 UK national guideline for the management of infection with Neisseria gonorrhoeae. Int. J. STD AIDS 2020, 31, 4–15. [Google Scholar] [CrossRef]
- Fischetti, V.A. Development of phage lysins as novel therapeutics: A historical perspective. Viruses 2018, 10, 310. [Google Scholar] [CrossRef]
- Fleming, D.T.; Wasserheit, J.N. From epidemiological synergy to public health policy and practice: The contribution of other sexually transmitted diseases to sexual transmission of HIV infection. Sex. Transm. Infect. 1999, 75, 3–17. [Google Scholar] [CrossRef]
- Fountain, H.; Migchelsen, S.J.; Charles, H.; Ram, T.; Fifer, H.; Mohammed, H.; Sinka, K. Rebound of Gonorrhea after Lifting of COVID-19 Preventive Measures, England. Emerg. Infect. Dis. 2024, 30, 329–332. [Google Scholar] [CrossRef]
- Fox, D.A.; et al. Structure of the Neisserial Outer Membrane Protein Opa60: Loop Flexibility Essential to Receptor Recognition and Bacterial Engulfment. J. Am. Chem. Soc. 2014, 136, 9938–9946. [Google Scholar] [CrossRef]
- Franks, A.G. Successful combined treatment of penicillin-resistant gonorrhea. Am. J. Med. Sci. 1946, 211, 553–555. [Google Scholar] [CrossRef]
- Freitag, N.E.; Seifert, H.S.; Koomey, M. Characterization of the pilF-pilD pilus-assembly locus of Neisseria gonorrhoeae. Mol. Microbiol. 1995, 16, 575–586. [Google Scholar] [CrossRef]
- Gaydos, C.A.; Hardick, J. Point of care diagnostics for sexually transmitted infections: perspectives and advances. Expert Rev. Anti Infect. Ther. 2014, 12, 657–672. [Google Scholar] [CrossRef]
- Goire, N.; Lahra, M.M.; Chen, M.; Donovan, B.; Fairley, C.K.; Guy, R.; Kaldor, J.; Regan, D.; Ward, J.; Nissen, M.D.; Sloots, T.P.; Whiley, D.M. Molecular approaches to enhance surveillance of gonococcal antimicrobial resistance. Nat. Rev. Microbiol. 2014, 12, 223–229. [Google Scholar] [CrossRef]
- Grad, Y.H.; Kirkcaldy, R.D.; Trees, D.; Dordel, J.; Harris, S.R.; Goldstein, E.; Rudy, M.; Ibiza, J.A.; Brown, W.; Abou Tayoun, A.; et al. Genomic epidemiology of Neisseria gonorrhoeae with reduced susceptibility to cefixime in the USA: a retrospective observational study. Lancet Infect. Dis. 2014, 14, 220–226. [Google Scholar] [CrossRef]
- Gransden, W.R.; Warren, C.A.; Phillips, I.; Hodges, M.; Barlow, D. Decreased susceptibility of Neisseria gonorrhoeae to ciprofloxacin. Lancet, 1990, 335, 51. [Google Scholar]
- Gulati, S.; Cox, A.D.; Jerse, A.E.; Rice, P.A. Lipooligosaccharide sialylation in Neisseria gonorrhoeae: mechanisms of immune evasion and systemic dissemination. Front. Microbiol. 2015, 6, 1268. [Google Scholar]
- Gupta, S.; Sharma, S.; Sharma, S.; Singh, N. The Trend of STD in COVID Times–A Retrospective Hospital-Based Study from Northeast India. IP Indian J. Clin. Exp. Dermatol. 2022, 8, 43–46. [Google Scholar]
- Handing, J.W.; Ragland, S.A.; Bharathan, U.V.; Criss, A.K. The MtrCDE efflux pump contributes to survival of Neisseria gonorrhoeae from human neutrophils and their antimicrobial components. Front. Microbiol. 2018, 9, 2688. [Google Scholar] [CrossRef]
- Handsfield, H.H.; Dalu, Z.A.; Martin, D.H.; Douglas, J.M., Jr.; McCarty, J.M.; Schlossberg, D.; Azithromycin Gonorrhea Study Group. Multicenter Trial of Single-Dose Azithromycin vs Ceftriaxone in the Treatment of Uncomplicated Gonorrhea. Sex. Transm. Dis. 1994, 21, 107–111. [Google Scholar] [CrossRef]
- Handsfield, H.H.; Lipman, T.O.; Harnisch, J.P.; Tronca, E.; Holmes, K.K. Asymptomatic Gonorrhea in Men. Diagnosis, Natural Course, Prevalence and Significance. N. Engl. J. Med. 1974, 290, 117–123. [Google Scholar] [CrossRef]
- Harris, S.R.; Cole, M.J.; Spiteri, G.; Sanchez-Buso, L.; Golparian, D.; Jacobsson, S.; Goire, N.; Unemo, M.; Grad, Y.H.; Read, T.D.; et al. Public health surveillance of multidrug-resistant clones of Neisseria gonorrhoeae in Europe: a genomic survey. Lancet Infect. Dis. 2018, 18, 758–768. [Google Scholar] [CrossRef] [PubMed]
- Harrison, W.O.; et al. A Trial of Minocycline Given after Exposure to Prevent Gonorrhea. N. Engl. J. Med. 1979, 300, 1074–1078. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.A.; Davies, J.K. Pilin gene variation in Neisseria gonorrhoeae: Reassessing the old paradigms. FEMS Microbiol. Rev. 2009, 33, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.A.; Masters, T.L.; Wachter, J. Gonorrhea–An Evolving Disease of the New Millennium. Microb. Cell, 2016, 3, 371–389. [Google Scholar] [CrossRef] [PubMed]
- Hook, E.W. III.; Kirkcaldy, R.D. A Brief History of Evolving Diagnostics and Therapy for Gonorrhea: Lessons Learned. Clin. Infect. Dis. 2018, 67, 1294–1299. [Google Scholar] [CrossRef]
- Hu, M.; Nandi, S.; Davies, C.; Nicholas, R.A. High-Level Chromosomally Mediated Tetracycline Resistance in Neisseria gonorrhoeae Results from a Point Mutation in the rpsJ Gene Encoding Ribosomal Protein S10 in Combination with the mtrR and penB Resistance Determinants. Antimicrob. Agents Chemother. 2005, 49, 4327–4334. [Google Scholar] [CrossRef]
- Hung, M.-C.; Christodoulides, M. The Biology of Neisseria Adhesins. Biology (Basel), 2013, 2, 1054–1109. [Google Scholar] [CrossRef]
- Imarenezor, E.P.K.; Ofiri, P.N. Assessment of Hepatitis B Virus Infection and Immunity Status amid Inhabitants of Wukari in Taraba State, North-East Nigeria: A Communal-Based Study. Int. J. Adv. Biol. Biomed. Res. 2024, 12, 319–330. [Google Scholar]
- Indian Council of Medical Research (ICMR). Treatment Guidelines for Antimicrobial Use in Common Syndromes; ICMR: New Delhi, India, 2019. [Google Scholar]
- Ison, C.A.; Littleton, K.; Shannon, K.P.; Easmon, C.S.; Phillips, I. Spectinomycin-resistant gonococci. Br. Med. J. (Clin. Res. Ed.) 1983, 287, 1827–1829. [Google Scholar] [CrossRef]
- Ito, M.; Deguchi, T.; et al. Emergence and Spread of Neisseria gonorrhoeae Clinical Isolates Harboring Mosaic-like Structure of Penicillin-Binding Protein 2 in Central Japan. Antimicrob. Agents Chemother. 2005, 49, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Yasuda, M.; Yokoi, S.; Ito, S.; Takahashi, Y.; Ishihara, S.; Maeda, S.; Deguchi, T. Remarkable Increase in Central Japan in 2001–2002 of Neisseria gonorrhoeae Isolates with Decreased Susceptibility to Penicillin, Tetracycline, Oral Cephalosporins, and Fluoroquinolones. Antimicrob. Agents Chemother. 2004, 48, 3185–3187. [Google Scholar] [CrossRef]
- Jadimurthy, R.; Anbarasu, A.; Ramaiah, S. Natural products as alternative therapeutics against antimicrobial resistance: Focus on gonorrhea. Front. Pharmacol. 2023, 14, 1124563. [Google Scholar]
- Jadimurthy, R.; Jagadish, S.; Nayak, S.C.; Kumar, S.; Mohan, C.D.; Rangappa, K.S. Phytochemicals as Invaluable Sources of Potent Antimicrobial Agents to Combat Antibiotic Resistance. Life 2023, 13, 948. [Google Scholar] [CrossRef] [PubMed]
- Jamaludin, N.; Gedye, K.; Collins-Emerson, J.; Benschop, J.; Nulsen, M. Phenotypic and Genotypic Characterization of Neisseria gonorrhoeae Isolates from New Zealand with Reduced Susceptibility to Ceftriaxone. Microb. Drug Resist. 2019, 25, 892–902. [Google Scholar] [CrossRef] [PubMed]
- Judson, F.N.; Ehret, J.M.; Handsfield, H.H. Comparative Study of Ceftriaxone and Spectinomycin for Treatment of Pharyngeal and Anorectal Gonorrhea. JAMA 1985, 253, 1417–1419. [Google Scholar] [CrossRef]
- Jünger, C.; Imkamp, F.; Balakrishna, S.; Gysin, M.; Haldimann, K.; Brugger, S.D.; Scheier, T.C.; et al. Phenotypic and genotypic characterization of Neisseria gonorrhoeae isolates among individuals at high risk for sexually transmitted diseases in Zurich, Switzerland. Int. J. STD AIDS 2024, 35, 725–734. [Google Scholar] [CrossRef]
- Kakooza, F.; Kiggundu, R.; Mboowa, G.; Kateete, P.D.; Nsangi, O.T.; Kabahita, J.M.; Ssentalo Bagaya, B.; Golparian, D.; Unemo, M. Antimicrobial susceptibility surveillance and antimicrobial resistance in Neisseria gonorrhoeae in Africa from 2001 to 2020: A mini-review. Front. Microbiol. 2023, 14, 1148817. [Google Scholar] [CrossRef]
- Kampmeier, R.H. Introduction of Sulfonamide Therapy for Gonorrhea. Sex. Transm. Dis. 1983, 10, 81–84. [Google Scholar] [CrossRef]
- Kannan, K.; Kanabar, P.; et al. The General Mode of Translation Inhibition by Macrolide Antibiotics. Proc. Natl. Acad. Sci. U.S.A. 2014, 111, 15958–15963. [Google Scholar] [CrossRef]
- Kellner, M.J.; Koob, J.G.; Gootenberg, J.S.; Abudayyeh, O.O.; Zhang, F. SHERLOCK: Nucleic acid detection with CRISPR nucleases. Nat. Protoc. 2019, 14, 2986–3012. [Google Scholar] [CrossRef]
- Kirkcaldy, R.D.; Schlanger, K.; Papp, J.R.; Torrone, E.A. Considerations for Strengthening Surveillance of Neisseria gonorrhoeae Antimicrobial Resistance and Interpreting Surveillance Data. Sex. Transm. Dis. 2017, 44, 154–156. [Google Scholar] [CrossRef]
- Kivata, M.W.; Mbuchi, M.; Eyase, F.L.; Bulimo, W.D.; Kyanya, C.K.; Oundo, V.; et al. gyrA and parC mutations in fluoroquinolone-resistant Neisseria gonorrhoeae isolates from Kenya. BMC Microbiol. 2019, 19. [Google Scholar]
- Klaper, K.; Tlapák, H.; Selb, R.; Jansen, K.; Heuer, D. Integrated Molecular, Phenotypic and Epidemiological Surveillance of Antimicrobial Resistance in Neisseria gonorrhoeae in Germany. Int. J. Med. Microbiol. 2024, 314, 151611. [Google Scholar] [CrossRef] [PubMed]
- Klausner, J.D.; Kohn, R.; Kent, C. Etiology of Clinical Proctitis among Men Who Have Sex with Men. Clin. Infect. Dis. 2004, 38, 300–302. [Google Scholar] [CrossRef]
- Latini, A.; Magri, F.; Donà, M.G.; Giuliani, M.; Cristaudo, A.; Zaccarelli, M. Is COVID-19 Affecting the Epidemiology of STIs? The Experience of Syphilis in Rome. Sex. Transm. Infect. 2021, 97, 78. [Google Scholar] [CrossRef]
- Laumen, J.G.E.; Hieu, V.N.; Nhung, P.H.; Vandelannoote, K.; Nguyen, T.T.; et al. High prevalence of ceftriaxone-resistant Neisseria gonorrhoeae in Hanoi, Vietnam, 2023–2024. J. Infect. Dis. 2025, 232, e73–e77. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D.A. New treatment options for Neisseria gonorrhoeae in the era of emerging antimicrobial resistance. Sex. Health 2019, 16, 449–456. [Google Scholar] [CrossRef]
- Lewis, D.A. The gonococcus fights back: Is this time a knock out? Sex. Transm. Infect. 2010, 86, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, S.; Wang, J.; Liu, G. CRISPR/Cas Systems towards Next-Generation Biosensing. Trends Biotechnol. 2021, 39, 730–747. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.Y.L.; Mullally, C.A.; et al. Anti-Virulence Therapeutic Approaches for Neisseria gonorrhoeae. Antibiotics 2021, 10, 103. [Google Scholar] [CrossRef]
- Lindberg, M.; Ringertz, O.; Sandström, E. Treatment of pharyngeal gonorrhoea due to β-lactamase-producing gonococci. Br. J. Vener. Dis. 1982, 58, 101–104. [Google Scholar]
- Lipsky, M.S.; Su, S.; Crespo, C.J.; Hung, M. Men and Oral Health: A Review of Sex and Gender Differences. Am. J. Mens Health 2021, 15, 15579883211016361. [Google Scholar] [CrossRef]
- Liu, S.; Yu, C.; Tu, Q.; Zhang, Q.; Fu, Z.; Huang, Y.; He, C.; Yao, L. Bacterial Co-Infection in COVID-19: A Call to Stay Vigilant. Peer J. 2024, 12, e18041. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, F.; Li, K.; Chen, S.; Hu, B.; Feng, Y.; Song, H.; Lu, X.; Wang, D.; Ge, C. From lab to clinic: A portable and automated microfluidic nucleic acid detection instrument for rapid STI pathogen identification. Biosens. Bioelectron. 2025, 282, 117498. [Google Scholar] [CrossRef]
- Maatouk, I.; Vumbugwa, P.; Cherdtrakulkiat, T.; et al. , WHO EGASP Study Group. Antimicrobial-resistant Neisseria gonorrhoeae in Europe in 2020 compared with in 2013 and 2018: a retrospective genomic surveillance study. Lancet Microbe 2024, 5, e478–e488. [Google Scholar]
- Mahapure, K.; Singh, A. A Review of Recent Advances in Our Understanding of Neisseria gonorrhoeae. Cureus 2023, 15, e43464. [Google Scholar] [CrossRef]
- Mahoney, J.; Ferguson, C.; Buchholtz, M.; Van Slyke, C. The Use of Penicillin Sodium in the Treatment of Sulfonamide-Resistant Gonorrhoea in Men. A Preliminary Report. Am. J. Syph. 1943, 27, 525–528. [Google Scholar]
- Mahoney, M.H.; Smith, J.M.; Brown, H.C. Clinical trials of sulfapyridine and sulfathiazole in gonorrhea therapy. JAMA 1941, 116, 1239–1245. [Google Scholar]
- Maiden, M.C.; Bygraves, J.A.; Feil, E.; Morelli, G.; Russell, J.E.; Urwin, R.; Spratt, B.G. Multilocus sequence typing: a portable approach to the identification of clones within populations of pathogenic microorganisms. Proc. Natl. Acad. Sci. U.S.A. 1998, 95, 3140–3145. [Google Scholar] [CrossRef] [PubMed]
- Maldonado Galdeano, C.; Cazorla, S.I.; Lemme Dumit, J.M.; Vélez, E.; Perdigón, G. Beneficial effects of probiotic consumption on the immune system. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef]
- Man, C.; Budka, J.; Wu, H.; Moon, J.; Jang, Y.; Kim, S.; El-Naggar, H.; Mulvey, M.R.; Kim, S.; Taha, M.-K.; Lee, H.; Chong, Y. Real-Time PCR Assay for Simultaneous Detection of Neisseria gonorrhoeae and Antimicrobial Resistance Markers Directly from Clinical Specimens. Front. Cell. Infect. Microbiol. 2021, 11, 679561. [Google Scholar]
- Man, O.M.; Ramos, W.E.; Vavala, G.; Goldbeck, C.; Ocasio, M.A.; et al. Optimizing screening for anorectal, pharyngeal, and urogenital Chlamydia trachomatis and Neisseria gonorrhoeae infections in at-risk adolescents and young adults in New Orleans, Louisiana and Los Angeles, California, United States. Clin. Infect. Dis. 2021, 73, e3201–e3209. [Google Scholar] [CrossRef]
- Manoharan-Basil, S.S.; Unemo, M.; Golparian, D.; Sánchez-Busó, L. Genomic mechanisms of antimicrobial resistance in Neisseria gonorrhoeae. Microb. Genom. 2021, 7, 000640. [Google Scholar]
- Martin, I.M.; Ison, C.A.; Aanensen, D.M.; Fenton, K.A.; Spratt, B.G. Rapid sequence-based identification of gonococcal transmission clusters in a large metropolitan area. J. Infect. Dis. 2004, 189, 1497–1505. [Google Scholar] [CrossRef]
- Martin, S.L.; Mortimer, T.D.; Grad, Y.H. Machine learning models for Neisseria gonorrhoeae antimicrobial susceptibility tests. Ann. N.Y. Acad. Sci. 2023, 20, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Massari, P.; Ram, S.; Macleod, H.; Wetzler, L.M. The role of Neisseria gonorrhoeae PorB porins in resistance to complement-mediated killing. J. Immunol. 2003, 171, 6191–6198. [Google Scholar]
- Mayor, M.T.; Roett, M.A.; Uduhiri, K.A. Diagnosis and management of gonococcal infections. Am. Fam. Physician 2012, 86, 931–938. [Google Scholar]
- Mayorga-Ramos, A.; Basallo, A.; Franco, A. CRISPR-Cas antimicrobials: A new paradigm to combat antimicrobial resistance. Front. Microbiol. 2023, 14, 1135798. [Google Scholar]
- Mayorga-Ramos, A.; Zúñiga-Miranda, J.; Carrera-Pacheco, S.E.; Barba-Ostria, C.; Guamán, L.P. CRISPR-Cas-based antimicrobials: design, challenges, and bacterial mechanisms of resistance. ACS Infect. Dis. 2023, 9, 1283–1302. [Google Scholar] [CrossRef]
- McGuire, J.; Conover, L.; Ploussard, J.H.; Raper, H.S. Erythromycin, a new antibiotic produced by Streptomyces erythraeus. Proc. Soc. Exp. Biol. Med. 1952, 80, 661–664. [Google Scholar]
- Mehta, N.; Verma, K. Impact of COVID-19 pandemic on sexually transmitted infections: Indian experience. Indian J. Dermatol. Venereol. Leprol. 2024, 90, 518–519. [Google Scholar] [CrossRef]
- Merrick, R.; Cole, M.; Pitt, R.; Enayat, Q.; Ivanov, Z.; Day, M.; Sun, S.; Sinka, K.; Woodford, N.; Mohammed, H.; Fifer, H. Antimicrobial-resistant gonorrhoea: the national public health response, England, 2013 to 2020. Euro Surveill. 2022, 27, 1–6. [Google Scholar] [CrossRef]
- Mitani, Y.; Lezhava, A.; Kawai, Y.; Kikuchi, T.; Oguchi-Katayama, A.; et al. Rapid SNP diagnostics using asymmetric isothermal amplification and a new mismatch-suppression technology. Nat. Methods 2013, 10, 891–895. [Google Scholar] [CrossRef]
- Mookherjee, N.; Anderson, M.A.; Haagsman, H.P.; Davidson, D.J. Antimicrobial host defence peptides: functions and clinical potential. Nat. Rev. Drug Discovery, 2020, 19, 311–332. [Google Scholar] [CrossRef]
- Moran, J.S.; Levine, W.C. Drugs of choice for the treatment of uncomplicated gonococcal infections. Clin. Infect. Dis. 1995, 20 (Suppl. 1), S47–S65. [Google Scholar] [CrossRef]
- Morse, S.A.; Johnson, S.R.; Biddle, J.; Roberts, M.C. High-level tetracycline resistance in Neisseria gonorrhoeae is due to acquisition of the tetM determinant. Antimicrob. Agents Chemother. 1986, 29, 169–172. [Google Scholar]
- Mortimer, T.D.; Grad, Y.H. Applications of genomics to slow the spread of multidrug-resistant Neisseria gonorrhoeae. Ann. N.Y. Acad. Sci. 2019, 1435, 93–109. [Google Scholar] [CrossRef]
- Mullis, K.B.; Faloona, F.A. Specific synthesis of DNA in vitro via a polymerase-catalyzed chain reaction. Methods Enzymol. 1987, 155, 335–350. [Google Scholar]
- Nagendra, G.; Carnevale, C.; Neu, N.; Cohall, A.; Zucker, J. The potential impact and availability of sexual health services during the COVID-19 pandemic. Sex. Transm. Dis. 2020, 47, 434–436. [Google Scholar] [CrossRef] [PubMed]
- Narain, J.P.; Mohan, N.; Vedhanaygam, M.; Swamiappan, M.; Rajagopalan, R. Resurgence of sexually transmitted infections in India. Indian J. Sex. Transm. Dis. AIDS, 2024, 45, 102–109. [Google Scholar] [CrossRef]
- Naumann, M.; Rudel, T.; Meyer, T.F. Host cell interactions and signalling with Neisseria gonorrhoeae. Curr. Opin. Microbiol. 1999, 2, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Noinaj, N.; Buchanan, S.K.; Cornelissen, C.N. The transferrin-iron import system from pathogenic Neisseria species. Mol. Microbiol. 2012, 86, 246–257. [Google Scholar] [CrossRef] [PubMed]
- NSW Health (New South Wales Health). GP Alert - Antimicrobial resistant gonococcal infection; update 22. NSW Health; GP Alert 2022, 1-4.
- Ofiri, P.N.; Imarenezor, E.P.K.; Anyiam, V.I.; Abhadionmhen, O.A. HIV/AIDS prevalence in Taraba State, North East, Nigeria; case study: Wukari and environs. Int. J. Adv. Multidiscip. Res. Stud. 2024, 4, 6–10. [Google Scholar] [CrossRef]
- Ohnishi, M.; et al. Is Neisseria gonorrhoeae initiating a future era of untreatable gonorrhea? Detailed characterization of the first strain with high-level resistance to ceftriaxone. Antimicrob. Agents Chemother. 2011, 55, 3538–3545. [Google Scholar] [CrossRef]
- Olawade, D.B.; Oladipo, E.K.; Ajayi, O.O.; Olayanju, A.O.; Aworunse, O.S. Therapeutic potential of bacteriophages and endolysins in combating multidrug-resistant bacteria: A review. Infect. Drug Resist. 2022, 15, 2267–2282. [Google Scholar]
- Olawade, D.B. ; Phage therapy: A targeted approach to overcoming antibiotic resistance. Microb. Pathog. 2024, 197, 107088. [Google Scholar] [CrossRef]
- Olesky, M.; Zhao, S.; Rosenberg, R.L.; Nicholas, R.A. Porin-mediated antibiotic resistance in Neisseria gonorrhoeae: Ion, solute, and antibiotic permeation through PIB proteins with penB mutations. J. Bacteriol. 2006, 188, 2300–2308. [Google Scholar] [CrossRef] [PubMed]
- Omeershffudin, U.N.M.; Kumar, S. Emerging threat of antimicrobial resistance in Neisseria gonorrhoeae: Pathogenesis, treatment challenges, and potential for vaccine development. Arch. Microbiol. 2023, 205, 330. [Google Scholar] [CrossRef] [PubMed]
- Oren, A.; da Costa, M.S.; Garrity, G.M.; Rainey, F.A.; Rosselló-Móra, R.; Schink, B.; Sutcliffe, I.; Trujillo, M.E.; Whitman, W.B. Proposal to include the rank of phylum in the International Code of Nomenclature of Prokaryotes. Int. J. Syst. Evol. Microbiol. 2015, 65, 4284–4287. [Google Scholar] [CrossRef]
- Oriel, J.D. The Scars of Venus: A History of Venereology; Springer: London, UK, 1994; pp. 187–189. [Google Scholar]
- Orzechowska, M.; Cybulski, M.; Krajewska-Kulak, E.; Sobolewski, M.; Gniadek, A.; Niczyporuk, W. Comparative analysis of the incidence of selected sexually transmitted viral infections in Poland in 2010–2015: A retrospective cohort study. J. Clin. Med. 2022, 11, 3448. [Google Scholar] [CrossRef] [PubMed]
- Ouk, V.; Heng, L.S.; Virak, M.; Deng, S.; Lahra, M.M.; Frankson, R.; Kreisel, K.; McDonald, R.; Escher, M.; Unemo, M.; Wi, T.; Maatouk, I.; EGASP Cambodia Working Group. High prevalence of ceftriaxone-resistant and XDR Neisseria gonorrhoeae in several cities of Cambodia, 2022–2023: WHO Enhanced Gonococcal Antimicrobial Surveillance Programme (EGASP). JAC Antimicrob. Resist. 2024, 6, dlae053. [Google Scholar] [CrossRef]
- Palermo, E.F.; Kuroda, K. Structural determinants of antimicrobial activity in polymers which mimic host defense peptides. Appl. Microbiol. Biotechnol. 2010, 87, 1605–1615. [Google Scholar] [CrossRef]
- Palermo, R.L.; Evans, H.F.; Rasko, D.A. Synthetic mimics of antimicrobial peptides with immunomodulatory functions. Curr. Pharm. Des. 2010, 16, 3371–3385. [Google Scholar]
- Papasian, C.J.; Bartholomew, W.R.; Amsterdam, D. Validity of an enzyme immunoassay for detection of Neisseria gonorrhoeae antigens. J. Clin. Microbiol. 1984, 19, 347–350. [Google Scholar] [CrossRef]
- Park, J.W. Principles and Applications of Loop-Mediated Isothermal Amplification to Point-of-Care Tests. Biosensors 2022, 12, 857. [Google Scholar]
- Pathela, P.; Braunstein, S.L.; Blank, S.; Schillinger, J.A. HIV incidence among men with and those without sexually transmitted rectal infections: Estimates from matching against an HIV case registry. Clin. Infect. Dis. 2013, 57, 1203–1209. [Google Scholar] [CrossRef]
- Phillips, I. β-lactamase-producing, penicillin-resistant gonococcus. Lancet 1976, 308, 656–657. [Google Scholar]
- Pinto, C.N.; Niles, J.K.; Kaufman, H.W.; Marlowe, E.M.; Alagia, D.P.; Chi, G.; Van Der Pol, B. Impact of the COVID-19 Pandemic on Chlamydia and Gonorrhea Screening in the U.S. Am. J. Prev. Med. 2021, 60, 847–855. [Google Scholar] [CrossRef]
- Poehlsgaard, J.; Douthwaite, S. The bacterial ribosome as a target for antibiotics. Nat. Rev. Microbiol. 2005, 3, 870–881. [Google Scholar] [CrossRef] [PubMed]
- Public Health Agency of Canada. Survey on the Impact of COVID-19 on the Delivery of STBBI Prevention, Testing and Treatment Including Harm Reduction Services in Canada; PHAC: Ottawa, ON, 2021. [Google Scholar]
- Quillin, S.J.; Seifert, H.S. Neisseria gonorrhoeae host adaptation and pathogenesis. Nat. Rev. Microbiol. 2018, 16, 226–240. [Google Scholar] [CrossRef]
- Radovanovic, M.; Kekic, D.; Jovicevic, M.; Kabic, J.; Gajic, I.; Opavski, N.; Ranin, L. Current Susceptibility Surveillance and Distribution of Antimicrobial Resistance in N. gonorrhoeae within WHO Regions. Pathogens 2022, 11, 1230. [Google Scholar] [CrossRef]
- Rasko, D.A.; Sperandio, V. Anti-virulence strategies to combat bacteria-mediated disease. Nat. Rev. Drug Discov. 2010, 9, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Reyn, A.; Korner, B.; Bentzon, M.W. Effects of penicillin, streptomycin, and tetracycline on N. gonorrhoeae isolated in 1944 and in 1957. Br. J. Vener. Dis. 1958, 34, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Rice, P.A.; et al. Serum resistance of Neisseria gonorrhoeae. Does it thwart the inflammatory response and facilitate the transmission of infection? Ann. N. Y. Acad. Sci. 1994, 730, 7–14. [Google Scholar] [CrossRef]
- Roberts, M.C.; Wagenvoort, J.H.; van Klingeren, B.; Knapp, J.S. tetM- and beta-lactamase-containing Neisseria gonorrhoeae (tetracycline resistant and penicillinase producing) in The Netherlands. Antimicrob. Agents Chemother. 1988, 32, 158. [Google Scholar]
- Rowley, J.; Vander Hoorn, S.; Korenromp, E.; Low, N.; Unemo, M.; Abu-Raddad, L.J.; Chico, R. Chlamydia, gonorrhoea, trichomoniasis and syphilis: Global prevalence and incidence estimates, 2016. Bull. World Health Organ. 2019, 97, 548–562. [Google Scholar]
- Roymans, R.; Onland, G.; Jansz, A.; Quint, W.; Boel, E. Evaluation of an in-house polymerase chain reaction for detection of Neisseria gonorrhoeae in urogenital samples. J. Clin. Pathol. 1999, 52, 411–414. [Google Scholar] [CrossRef]
- Sadarangani, M.; Pollard, A.J.; Gray-Owen, S.D. Opa proteins and CEACAMs: pathways of immune engagement for pathogenic Neisseria. FEMS Microbiol. Rev. 2011, 35, 498–514. [Google Scholar] [CrossRef]
- Sadiq, S.T.; Taylor, S.; Copas, A.J.; Bennett, J.; Kaye, S.; Drake, S.M.; et al. The effects of urethritis on seminal plasma HIV-1 RNA loads in homosexual men not receiving antiretroviral therapy. Sex. Transm. Infect. 2005, 81, 120–123. [Google Scholar] [CrossRef]
- Sánchez-Busó, L.; Golparian, D.; Corander, J.; Grad, Y.H.; Ohnishi, M.; Flemming, H.C.; Unemo, M. The impact of homologous recombination on the evolution of hypervirulent Neisseria gonorrhoeae lineages. Genome Biol. Evol. 2019, 11, 365–379. [Google Scholar]
- Sancta St., Cyr; et al. Update to CDC's Treatment Guidelines for Gonococcal Infection, 2020. MMWR Wkly. 2020, 69, 1911–1916. [Google Scholar]
- Savoia, D. Plant-derived antimicrobial compounds: Alternatives to antibiotics. Future Microbiol. 2012, 7, 979–990. [Google Scholar] [CrossRef]
- Sentís, A.; Prats-Uribe, A.; López-Corbeto, E.; et al. The impact of the COVID-19 pandemic on Sexually Transmitted Infections surveillance data: Incidence drop or artefact. BMC Public Health, 2021, 21, 1637. [Google Scholar] [CrossRef]
- Shaughnessy, J.; Ram, S.; Rice, P. Biology of the Gonococcus: Disease and Pathogenesis. Methods Mol. Biol. 2019, 1997, 1–27. [Google Scholar]
- Simms, A.N.; Jerse, A.E. Role of Phase and Antigenic Variation in Neisseria gonorrhoeae Colonization. In Colonization. In Colonization of Mucosal Surfaces; ASM: Washington, DC, 2005; pp. 327–350. [Google Scholar]
- Singh, V.; Bala, M.; Bhargava, A.; Kakran, M.; Bhatnagar, R. In vitro efficacy of 21 dual antimicrobial combinations comprising novel and currently recommended combinations for treatment of drug-resistant gonorrhoea in future era. PLoS ONE 2018, 13, e0193678. [Google Scholar]
- Soge, O.O.; Salipante, S.J.; No, D.; Duffy, E.; Roberts, M.C. In vitro activity of delafloxacin against clinical Neisseria gonorrhoeae isolates and selection of gonococcal delafloxacin resistance. Antimicrob. Agents Chemother. 2016, 60, 3106–3111. [Google Scholar] [CrossRef]
- Sood, S.; Gupta, S.; Verma, R.; Singh, R.; Agrawal, S.K.; Mahajan, N.; Sharma, V.K. Analysis of Neisseria gonorrhoeae susceptibility trends (MIC creep) from North India: A 15-years’ experience! Indian J. Dermatol. Venereol. Leprol. 2025, 91, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Sood, S.; Mahajan, N.; Singh, R.; Agrawal, S.K.; Shende, T.; Kapil, A.; Kar, H.K.; Sharma, V.K. Typing of Neisseria gonorrhoeae isolates by phenotypic and genotypic techniques in New Delhi, India. J. Lab. Physicians 2019, 11, 45–50. [Google Scholar] [CrossRef]
- Srikhanta, Y.N.; Dowideit, S.J.; Edwards, J.L.; Falsetta, M.L.; Wu, H.J.; Harrison, O.B.; Sikora, A.E.; Novotny, L.A.; Seib, K.L.; Tang, C.M.; et al. Phasevarions mediate random switching of gene expression in pathogenic Neisseria species. PLoS Pathog. 2009, 5, e1000400. [Google Scholar]
- Starnino, S.; Lefebvre, B.; Martin, I.; Michaud, S.; Tremblay, C.; Longtin, J.; Béland, N.; Charbonneau, C.; Girard, M.; Michaud, G. Emergence of azithromycin-resistant Neisseria gonorrhoeae in Latin America: epidemiology and molecular characterization. J. Antimicrob. Chemother. 2012, 67, 2698–2703. [Google Scholar]
- Stern, A.; Brown, M.; Nickel, P.; Meyer, T.F. Opacity genes in Neisseria gonorrhoeae: control of phase and antigenic variation. Cell 1986, 47, 61–71. [Google Scholar] [CrossRef]
- STI Guidelines Australia. Gonorrhoea. Accessed 15 September 2025. Available online: https://www.sti.guidelines.org.au.
- Stolz, E.; Zwart, H.G.; Michel, M.F. Activity of eight antimicrobial agents in vitro against Neisseria gonorrhoeae. Br. J. Vener. Dis. 1975, 51, 257–264. [Google Scholar]
- Su, X.; Lind, I. Molecular basis of high-level ciprofloxacin resistance in Neisseria gonorrhoeae strains isolated from Denmark from 1995–1998. Antimicrob. Agents Chemother. 2001, 45, 117–123. [Google Scholar]
- Swanson, J.; Belland, R.J.; Hill, S.A. Neisserial surface variation: How and why? Curr. Opin. Genet. Dev. 1992, 2, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Tajadini, M.; Panjehpour, M.; Javanmard, S.H. Comparison of SYBR Green and TaqMan methods in quantitative real-time polymerase chain reaction analysis of four adenosine receptor subtypes. Adv. Biomed. Res. 2014, 3, 85. [Google Scholar] [CrossRef]
- Taktikos, J.; Lin, Y.T.; Stark, H.; Biais, N.; Zaburdaev, V. Pili-induced clustering of Neisseria gonorrhoeae bacteria. PLoS One 2015, 10, e0137661. [Google Scholar] [CrossRef]
- Tam, M.R.; Buchanan, T.M.; Sandstrom, E.G.; Holmes, K.K.; Sadoff, J.C.; Perine, P.L. Serological classification of Neisseria gonorrhoeae with monoclonal antibodies. Infect. Immun. 1982, 36, 1042–1053. [Google Scholar] [CrossRef]
- Tang, J.; Crawley, A.; Jamison, K.; Wong, A.; Eng, S.; Zhang, C.; Navia, I.; Cubas, C.; Pathela, P. Delays in Sexual Health Care Among Patients Attending New York City Sexual Health Clinics During the COVID-19 Pandemic, March 2020 to February 2021. Sex Transm. Dis. 2025, 52, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Tapsall, J.W.; Ndowa, F.; Lewis, D.A.; Unemo, M. Meeting the public health challenge of multidrug- and extensively drug-resistant Neisseria gonorrhoeae. Expert Rev. Anti-Infect. Ther. 2009, 7, 821–834. [Google Scholar] [CrossRef]
- Tapsall, J.; Ndowa, F.; Lewis, D.A.; Unemo, M. Meeting the public health challenge of multidrug- and extensively drug-resistant Neisseria gonorrhoeae. Expert Rev. Anti-Infect. Ther. 2009, 7, 821–834. [Google Scholar]
- Tapsall, J. ; World Health Organization Anti-Infective Drug Resistance Surveillance and Containment Team. Antimicrobial Resistance in Neisseria gonorrhoeae; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Tayimetha, C.Y.; Njunda, L.A.; Akenji, B.; Founou, R.C.; Feteh, V.; et al. Phenotypic and genotypic characterization of Neisseria gonorrhoeae isolates from Yaoundé, Cameroon, 2019 to 2020. Microb. Genom. 2023, 9, mgen001091. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.N.; Marrazzo, J.; Batteiger, B.E.; et al. Single-dose zoliflodacin (ETX0914) for treatment of urogenital gonorrhea. N. Engl. J. Med. 2018, 379, 1835–1845. [Google Scholar]
- Teker, B.; Schim van der Loeff, M.F.; de Vries, H.J.C.; van Rooijen, M.S.; Hogewoning, A.A.; van Dam, A.P.; Bruisten, S.M. Increase in Neisseria gonorrhoeae infections after ending COVID-19 lockdown measures in Amsterdam, the Netherlands. Sex. Transm. Infect. 2025, 101, 152–159. [Google Scholar]
- Thorington, R.; Sawatzky, P.; Lefebvre, B.; Martin, I.; Mulvey, M.; Hoang, L.; Horsman, G.; Demczuk, W.; Allen, V.; Wong, T.; Choudhury, S.; for the Canadian Public Health Laboratory Network. Antimicrobial susceptibilities of Neisseria gonorrhoeae in Canada, 2020. Can. Commun. Dis. Rep. 2022, 48, 571–579. [Google Scholar] [CrossRef]
- Tong, Y.; Tang, W.; Kim, H.-J.; Pan, X.; Ranalli, T.A.; Kong, H.; Lemieux, B. Development of isothermal TaqMan assays for detection of Neisseria gonorrhoeae and Chlamydia trachomatis. J. Mol. Diagn. 2011, 13, 134–142. [Google Scholar]
- Tuddenham, S.; Stennett, C.A.; Ravel, J.; Ghanem, K.G. Vaginal cytokine profile and microbiota before and after lubricant use compared with condomless vaginal sex: a preliminary observational study. BMC Infect. Dis. 2021, 21, 1–13. [Google Scholar] [CrossRef]
- Unemo, M. Current and future antimicrobial treatment of gonorrhoea – the rapidly evolving Neisseria gonorrhoeae continues to challenge. BMC Infect. Dis. 2015, 15, 364. [Google Scholar] [CrossRef]
- Unemo, M.; Ballard, R.; Ison, C.; Lewis, D.; Ndowa, F.; Peeling, R. Laboratory diagnosis of sexually transmitted infections, including human immunodeficiency virus. WHO Press: Geneva, Switzerland, 2013. ISBN: 978-92-4-150584-0.
- Unemo, M.; Del Rio, C.; Shafer, W.M. Antimicrobial resistance expressed by Neisseria gonorrhoeae: a major global public health problem in the 21st century. Microbiol. Spectr. 2016, 4, 10. [Google Scholar] [CrossRef]
- Unemo, M.; Golparian, D.; Hestner, A. Ceftriaxone Treatment Failure of Pharyngeal Gonorrhoea Verified by International Recommendations, Sweden, July 2010. Euro Surveill. 2011, 16, 19792. [Google Scholar] [CrossRef]
- Unemo, M.; Golparian, D.; Limnios, A.; Whiley, D.; Ohnishi, M.; Lahra, M.M.; Tapsall, J.W. In vitro activity of ertapenem versus ceftriaxone against Neisseria gonorrhoeae isolates with highly diverse ceftriaxone MIC values and effects of ceftriaxone resistance determinants: Ertapenem for treatment of gonorrhea? Antimicrob. Agents Chemother. 2012, 56, 4226–4235. [Google Scholar] [CrossRef]
- Unemo, M.; Golparian, D.; Stary, A.; Eigentler, A. First Neisseria gonorrhoeae strain with resistance to cefixime causing gonorrhoea treatment failure in Austria, 2011. Euro Surveill. 2011, 16, 19998. [Google Scholar] [CrossRef]
- Unemo, M.; Lahra, M.M.; et al. World Health Organization Global Gonococcal Antimicrobial Surveillance Program (WHO GASP): review of new data and evidence to inform international collaborative actions and research efforts. Sex. Health 2019, 16, 412–425. [Google Scholar] [CrossRef]
- Unemo, M.; Lahra, M.M.; Escher, M.; Eremin, S.; Cole, M.J.; et al. WHO global antimicrobial resistance surveillance for Neisseria gonorrhoeae 2017–2018: A retrospective observational study. Lancet Microbe 2021, 2, e627–e636. [Google Scholar] [CrossRef]
- Unemo, M.; Nicholas, R.A. Emergence of multidrug-resistant, extensively drug-resistant and untreatable gonorrhea. Future Microbiol. 2012, 7, 1401–1422. [Google Scholar]
- Unemo, M.; Ross, J.; Serwin, A.B.; Gomberg, M.; Cusini, M.; Jensen, J.S. 2020 European guideline for the diagnosis and treatment of gonorrhoea in adults. Int. J. STD AIDS 2020, 31, 944–952. [Google Scholar] [CrossRef]
- Unemo, M.; Shafer, W.M. Antibiotic resistance in Neisseria gonorrhoeae: origin, evolution, and lessons learned for the future. Ann. N. Y. Acad. Sci. 2011, 1230, E19–E28. [Google Scholar] [CrossRef]
- Unemo, M.; Shafer, W.M. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: past, evolution, and future. Clin. Microbiol. Rev. 2014, 27, 587–613. [Google Scholar] [CrossRef]
- Unemo, M.; Workowski, K. Dual antimicrobial therapy for gonorrhoea: What is the role of azithromycin? Lancet Infect. Dis. 2018, 18, 486–488. [Google Scholar] [CrossRef]
- Ursu, R.G.; Luchian, I.; Damian, C.; Porumb-Andrese, E.; Cobzaru, R.G.; Nichitean, G.; Ripa, C.; Costin, D.; Sandu, D.; Andrioaie, I.M.; Iancu, L.S. The optimal management of Neisseria gonorrhoeae infections. Microorganisms 2022, 10, 2388. [Google Scholar] [CrossRef]
- Van Slyke, C.J.; Arnold, R.C.; Buchholtz, M. Penicillin therapy in sulfonamide-resistant gonorrhea in men. Am. J. Public Health Nations Health, 1943, 33, 1392–1394. [Google Scholar] [CrossRef]
- Van Slyke, D.D.; Talbot, A.; Schoen, H.E. Sulfapyridine and sulfathiazole in the treatment of gonorrhea. J. Clin. Investig. 1941, 20, 357–367. [Google Scholar]
- Vasala, A.; Hytonen, V.P.; Laitinen, O.H. Modern tools for rapid diagnostics of antimicrobial resistance. Front. Cell. Infect. Microbiol. 2020, 10, 308. [Google Scholar] [CrossRef]
- Veal, W.L.; Nicholas, R.A.; Shafer, W.M. Overexpression of the MtrC-MtrD-MtrE efflux pump due to an mtrR mutation is required for chromosomally mediated penicillin resistance in Neisseria gonorrhoeae. J. Bacteriol. 2002, 184, 5619–5624. [Google Scholar] [CrossRef]
- Vernel-Pauillac, F.; Colbus, B.; Merien, F.; Quentin-Millet, M.J. Transfer of quinolone resistance by transformation in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2008, 52, 2077–2081. [Google Scholar]
- Vernel-Pauillac, F.; Mérien, F. Characterization of quinolone resistance in Neisseria gonorrhoeae clinical isolates. J. Antimicrob. Chemother. 2006, 58, 466–470. [Google Scholar]
- Vincent, M.; Xu, Y.; Kong, H. Helicase-dependent isothermal DNA amplification. EMBO Rep. 2004, 5, 795–800. [Google Scholar] [CrossRef]
- Wainwright, M.; Swan, H.T. C.G. Paine and the earliest surviving clinical records of penicillin therapy. Med. Hist. 1986, 30, 42–56. [Google Scholar] [CrossRef]
- Walker, J.; Fairley, C.K.; Chow, E.P.F. Extragenital gonorrhoea: Silent reservoirs for ongoing transmission. Lancet Infect. Dis. 2023, 23, e295–e304. [Google Scholar]
- Waltmann, A.; Chen, J.S.; Duncan, J.A. Promising developments in gonococcal vaccines. Curr. Opin. Infect. Dis. 2024, 37, 63–69. [Google Scholar] [CrossRef]
- Weel, J.F.; Hopman, C.T.; van Putten, J.P. Bacterial entry and intracellular processing of Neisseria gonorrhoeae in epithelial cells: immunomorphological evidence for alterations in the major outer membrane protein P.IB. J. Exp. Med. 1991, 174, 705–715. [Google Scholar] [CrossRef]
- Whiley, D.M.; Jennison, A.V.; Pearson, J.; Lahra, M.M. Genetic characterization of Neisseria gonorrhoeae associated with disseminated gonococcal infection and antimicrobial resistance. J. Clin. Microbiol. 2017, 55, 1578–1585. [Google Scholar]
- WHO Guidelines for the Treatment of Neisseria Gonorrhoeae; World Health Organization: Geneva, Switzerland, 2016. Available online: https://www.who.int/publications-detail-redirect/9789241549691.
- Wi, T.; Lahra, M.M.; et al. Antimicrobial resistance in Neisseria gonorrhoeae: global surveillance and a call for international collaborative action. PLoS Med. 2017, 14, e1002344. [Google Scholar] [CrossRef]
- Willcox, R.R. The unsatisfactory treatment of gonorrhoea with penicillin. Br. J. Vener. Dis. 1970, 46, 217–219. [Google Scholar]
- Workowski, K.A.; Bolan, G.A. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm. Rep. 2015, 64, 1–137. [Google Scholar] [PubMed]
- Workowski, K.A.; Bachmann, L.H.; et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm. Rep. 2021, 70, 1–187. [Google Scholar] [CrossRef] [PubMed]
- Workowski, K.A.; Berman, S. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm. Rep. 2010, 59, 1–110. [Google Scholar]
- World Health Organization (WHO). Global Antimicrobial Resistance and Use Surveillance System (GLASS); WHO: Geneva, Switzerland, 2019; Available online: https://www.who.int/initiatives/glass. [Google Scholar]
- World Health Organization (WHO). Global Priority List of Antibiotic-Resistant Bacteria (GPLARB); WHO: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. Global Sexually Transmitted Infection Surveillance 2018. WHO; 2018.
- World Health Organization. Multi-drug resistant gonorrhoea; who fact sheet, 12 September 2025. Available online: https://www.who.int/news-room/fact-sheets/detail/multi-drug-resistant-gonorrhoea.
- Xu, S.X.; Leontyev, D.; Kaul, R.; Gray-Owen, S.D. Neisseria gonorrhoeae co-infection exacerbates vaginal HIV shedding without affecting systemic viral loads in human CD34+ engrafted mice. PLoS ONE 2018, 13, e0191672. [Google Scholar] [CrossRef]
- Yang, F.; Wang, D.; Zhang, Y.; Liu, Y.; Li, X.; Zhang, H.; Zhang, Q.; Li, X.; Zhang, W.; Wang, X.; et al. High-level ceftriaxone-resistant Neisseria gonorrhoeae strains associated with penA allele 60.001 in Hangzhou, China. Emerg. Infect. Dis. 2024, 30, 1234–1237. [Google Scholar]
- Yang, H.J.; Lee, H.M.; Lee, S.J.; Choi, J.B.; Bae, S.; Jung, J.H.; Kang, T.W.; Hwang, E.C. 2023 Korean Association of Urogenital Tract Infection and Inflammation guidelines for gonococcal infection. Investig. Clin. Urol. 2024, 65, 1–8. [Google Scholar] [CrossRef]
- Yang, Q.L.; Gotschlich, E.C. Variation of gonococcal lipooligosaccharide structure is due to alterations in poly-G tracts in lgt genes encoding glycosyl transferases. J. Exp. Med. 1996, 183, 323–327. [Google Scholar] [CrossRef]
- Yellman, M.A. Transportation risk behaviors among high school students – Youth Risk Behavior Survey, United States, 2019. MMWR Suppl. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- Yokoi, S.; et al. Threat to cefixime treatment of gonorrhea. Emerg. Infect. Dis. 2007, 13, 1275–1277. [Google Scholar] [CrossRef]
- Zhang, Q.; Peng, L.; Yuan, Y.; et al. High rates of Treponema pallidum, Neisseria gonorrhoeae, Chlamydia trachomatis, or Trichomonas vaginalis co-infection in people with HIV: a systematic review and meta-analysis. Eur. J. Clin. Microbiol. Infect. Dis. 2025, 44, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Xi, Y.; Gong, X.; Chen, S. Ceftriaxone-resistant gonorrhea, China, 2022. MMWR Morb. Mortal. Wkly. Rep. 2024, 73, 255–259. [Google Scholar] [PubMed]
- Zondag, H.C.A.; de Korne-Elenbaas, J.; Bruisten, S.M.; de Vries, H.J.C.; van Dam, A.P. Increased clonality among Neisseria gonorrhoeae isolates during the COVID-19 pandemic in Amsterdam, the Netherlands. Microb. Genom. 2023, 9, mgen000975. [Google Scholar] [CrossRef] [PubMed]
| Site of Infection | Uncomplicated Gonorrhea | Complicated Manifestations |
|---|---|---|
| Male urethra | Purulent or scant discharge, dysuria | Epididymitis, prostatitis, vesiculitis, infertility (rare) |
| Female cervix | Vaginal discharge, intermenstrual bleeding, dysuria, lower abdominal pain, dyspareunia | Pelvic inflammatory disease (PID), endometritis, salpingitis, tubo-ovarian abscess, infertility, ectopic pregnancy |
| Rectum (both sexes) | Often asymptomatic; anal pruritus, rectal pain, mucopurulent discharge, tenesmus | Proctitis (severe) |
| Pharynx (both sexes) | Usually asymptomatic; occasional sore throat | Rare systemic spread |
| Conjunctiva (adults) | Mucopurulent conjunctivitis, eyelid edema, conjunctival hyperemia | ----- |
| Disseminated gonococcal infection (DGI) | ------ | Fever, migratory polyarthritis, tenosynovitis, pustular skin lesions; rarely endocarditis or meningitis |
| Bartholin’s gland (females) | -------- | Painful labial swelling, localized abscess |
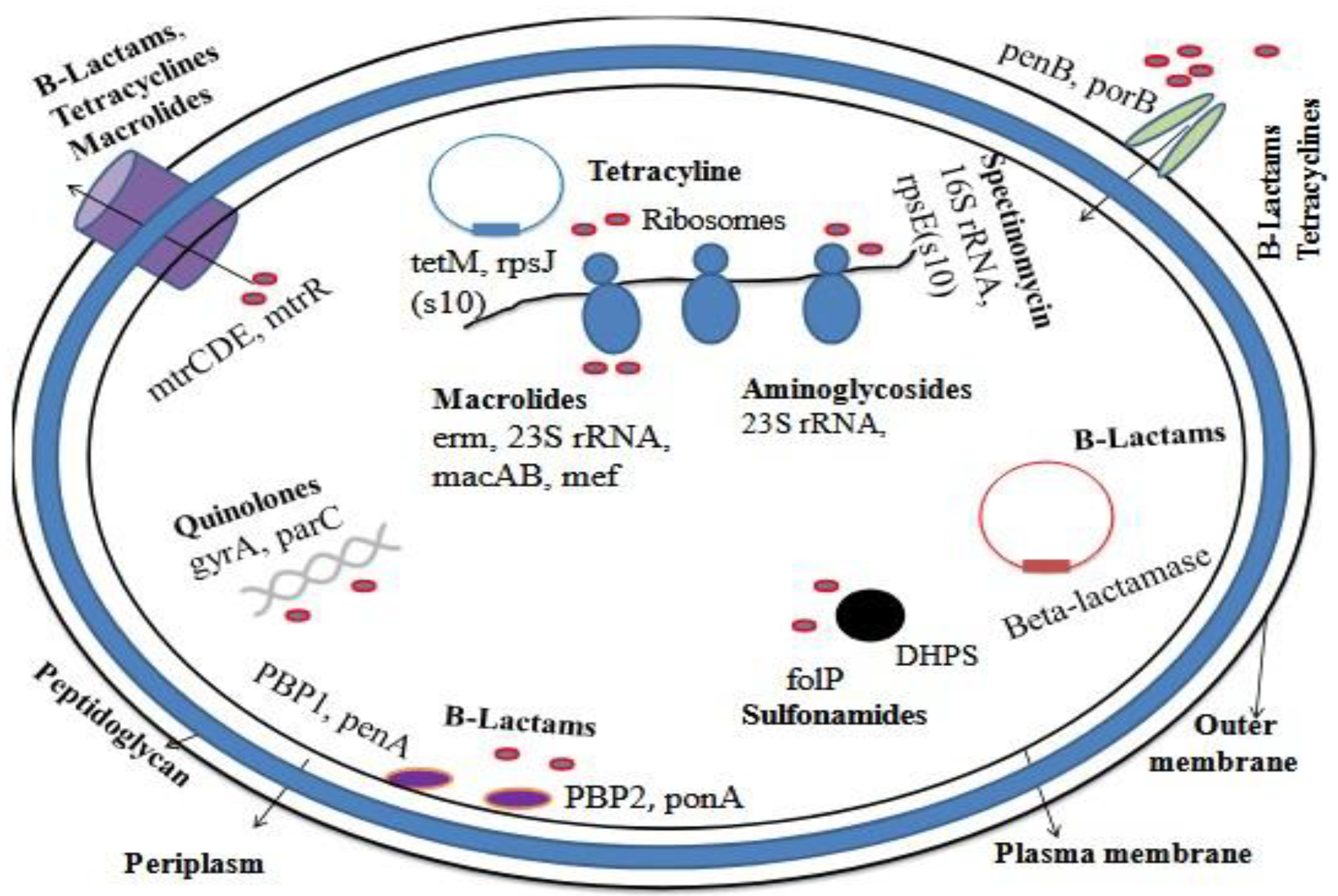
| Mechanism | Description | Examples |
|---|---|---|
| a) Antibiotic modification | Enzymatic degradation or chemical alteration of the antibiotic molecule, rendering it inactive. | β-lactamase production (penicillinase) |
| b) Target modification or protection | Structural alteration of antibiotic targets due to mutations or enzymatic modification, reducing drug affinity; in some cases, the target may be bypassed or replaced. | Mutations in penA, 23S rRNA, gyrA, parC |
| c) Reduced intracellular drug accumulation | Decreased drug uptake via porin loss or modification; increased drug efflux through active transport systems. | Overexpression of MtrCDE efflux pump, PorB mutations. [Handing et al., 2018]. |
| Antimicrobial Class | Representative Drug(S) | Year Introduced | Mechanism of Action | Resistance First Reported Year | Mechanism of Resistance | Current Status |
|---|---|---|---|---|---|---|
| Sulfonamides | Sulfanilamide, Sulfapyridine, Sulfathiazole | 1930s | Inhibit folate synthesis (folP) | ~1944 | Chromosomal mutations in folP | Obsolete; no longer used |
| Penicillins | Benzylpenicillin, Ampicillin | 1940s | Inhibit cell wall synthesis (PBPs) | 1970s–1980s | β-lactamase production, PBP alterations | Obsolete due to widespread resistance |
| Tetracyclines | Tetracycline | 1950s | Inhibit protein synthesis (30S ribosome) | 1980s | t etM plasmid, efflux pumps | No longer recommended |
| Macrolides | Erythromycin, Azithromycin | 1950s / 1990s | Inhibit protein synthesis (50S ribosome) | 1990s–2000s | 23S rRNA mutations, mtrCDE efflux pump overexpression | Azithromycin: previously part of dual therapy; resistance increasing |
| Aminocyclitols | Spectinomycin | 1960s | Inhibit protein synthesis (ribosomal binding) | 1980s | 16S rRNA or rpsE mutations | Rarely available; reserve/emergency option |
| Fluoroquinolones | Ciprofloxacin, Ofloxacin | 1980s | Inhibit DNA gyrase and topoisomerase IV | 1990s | gyrA and parC mutations | Not recommended since 2007 (CDC) |
| Cephalosporins | Ceftriaxone, Cefixime | 1980s | Inhibit cell wall synthesis (PBP2) | 2009–2011 | penA mosaic alleles (e.g., XXXIV) | Ceftriaxone remains last-line therapy; resistance emerging |
| Carbapenems (experimental) | Doripenem, Ertapenem, Imipenem, Meropenem | 1980s | Inhibit cell wall synthesis (PBPs) | ~2015–2017 | No high-level resistance documented | Experimental/emergency use in select XDR cases |
| Surveillance Program | Region | Objectives | Key Features |
|---|---|---|---|
| WHO-GASP (Global Gonococcal Antimicrobial Surveillance Programme) | Global | Monitor global trends in gonococcal AMR; inform WHO treatment guidelines | Standardized phenotypic testing; limited molecular data integration; capacity building in LMICs |
| GISP (Gonococcal Isolate Surveillance Project) | United States | Track national trends in AMR; guide CDC treatment recommendations | Longest-running national gonococcal AMR program; culture-based MIC testing |
| Euro-GASP (European Gonococcal Antimicrobial Surveillance Programme) | Europe | Provide EU-wide AMR data and detect treatment failures | Centralized reference testing; molecular typing integration |
| AGSP (Australian Gonococcal Surveillance Programme) | Australia | Monitor resistance trends and regional variation | High-resolution, culture-based testing across jurisdictions |
| National/Regional Programs (e.g., CAN-R, Japan’s national surveillance) | Country-specific | Support national guidelines; detect emerging resistance | Scope varies; increasingly using molecular and genomic tools |
| India (ICMR-led initiatives and academic studies) | India (fragmented, regional) | Generate local resistance data; inform empirical treatment | Lacks continuous national program; sporadic data; urgent need for WGS and coordinated national surveillance |
| Region / Program | Typical Recommended Regimen (uncomplicated urogenital) | Surveillance Snapshot / Issues |
|---|---|---|
| USA (CDC, 2020) MMWR | Ceftriaxone 500 mg IM once (1 g if ≥150 kg). If chlamydia not excluded: add doxycycline 100 mg bid × 7 d | Monotherapy (no routine azithromycin). Test-of-cure recommended for pharyngeal cases [CDC, 2020]. |
| UK (BASHH, 2024 draft) | Ceftriaxone 1 g IM once | Monotherapy; test-of-cure emphasized, especially for pharyngeal infection [BASHH, 2024]. |
| Australia (national guideline) | Ceftriaxone 500 mg IM + azithromycin 1 g PO stat (site- and risk-based variations) | Dual therapy still used; local resistance and public health considerations. Test-of-cure guidance provided [STI Guidelines, 2025]. |
| Europe (context via ECDC/Euro-GASP) | Country-specific (increasing uptake of ceftriaxone monotherapy) | Surveillance shows high azithromycin resistance in some settings; policies adapting accordingly [ECDC, 2020]. |
| Global LMIC settings (WHO/EGASP influence) | Ceftriaxone-based regimens; dual vs mono tailored to local data | EGASP supports capacity to align therapy with local susceptibility [Unemo et al., 2018; Maatouk et al, 2025]. |
| China | Ceftriaxone 1 g IM single dose (national practice) | Reports of ceftriaxone-resistant clones (FC428); national surveillance ongoing [Zhu et al., 2024]. |
| Japan / Korea | Ceftriaxone 500 mg IM (often used); alternatives (spectinomycin) in specific contexts | Historical emergence of resistance; continued national surveillance [Yang et al., 2024] |
| Southeast Asia (EGASP sentinel sites Cambodia) | Ceftriaxone 500 mg IM per WHO; local treatment updates advised where resistance high | EGASP (Cambodia) found high proportions of isolates with elevated ceftriaxone MICs, notable azithromycin non-susceptibility and XDR isolates, prompting urgent public health response [Ouk et al., 2024]. |
| Africa (general) | Ceftriaxone-based regimens per WHO; country variation | AMR data patchy; older drug resistance common; periodic reports of decreased susceptibility to ESCs/azithro in sentinel studies; surveillance strengthening required [Kakooza et al., 2023]. |
| India (national / programmatic) | Syndromic management widely used; many settings use cefixime 400 mg PO + azithromycin 1 g PO (“grey kit”) at primary care level; tertiary/ID centres may follow culture/AST | Increasing local reports of MIC creep to cefixime/ceftriaxone and rising azithromycin MICs; need for expanded quality-assured AMR surveillance and national guideline updates aligned with local data [Sood et al., 2025; ICMR, 2019]. |
| High-income settings (UK, USA, Australia) | UK/USA: ceftriaxone monotherapy (higher dose in UK/USA updates). Australia: many settings continued dual therapy historically; recent alerts about ceftriaxone failures in some regions. | Policy changes reflect local AMR trends (rise in azithro resistance; low but present ceftriaxone resistance). Test-of-cure emphasized for extragenital infections and suspected treatment failure [Sancta St. Cyr et al., 2020; Ouk et al., 2024; Unemo and Workowski, 2018]. |
| Approach | Mechanism of Action | Advantages | Current Limitations | References |
|---|---|---|---|---|
| Phage therapy and lysins | Bacteriophages infect and lyse gonococci; lysins enzymatically degrade cell walls | High specificity; active against MDR strains | Narrow host range; delivery and stability challenges | [Fischetti, 2018; Olawade et al., 2022] |
| CRISPR-based antimicrobials | Genome editing tools disrupt resistance genes and essential bacterial functions | Precision targeting; potential to reverse resistance | Delivery barriers in vivo; off-target risks | [Mayorga-Ramos et al., 2023] |
| AMPs and SMAMPs / IDR peptides | Disrupt bacterial membranes; modulate immune responses (IDR peptides) | Broad-spectrum; low resistance development | Toxicity, stability, and high production costs | [Mookherjee et al., 2020; Palermo et al., 2010] |
| Bacteriocins and probiotics | Natural microbial peptides and live microbes inhibit colonization via competition and secretion of antimicrobials | Generally safe; can restore microbiota balance | Limited strain-specific efficacy; variable colonization | [Dobson et al., 2012; Maldonado Galdeano et al., 2019] |
| Monoclonal antibodies | Neutralize gonococcal virulence factors (e.g., pili, outer membrane proteins) | High specificity; potential for vaccine-like protection | Costly; limited by antigenic variability of N. gonorrhoeae | [Tam et al., 1982; Demarco et al., 1986] |
| Host-directed therapies | Enhance innate immune pathways or block host mechanisms critical for bacterial survival | Less selective pressure for resistance | Risk of immune overactivation or side effects | [Rasko and Sperandio, 2010] |
| Anti-virulence agents | Disarm pathogens (e.g., block adhesins, toxins, quorum sensing) without killing bacteria | Reduced resistance selection; preserves microbiota | Many are still experimental; limited clinical validation | [Rasko and Sperandio, 2010] |
| Plant-derived phytochemicals | Bioactive metabolites (alkaloids, flavonoids, terpenoids) disrupt bacterial membranes, inhibit enzymes, and modulate | Abundant, low-cost, ethnomedicine-based; broad activity | Variability in extract composition; limited clinical validation | [Savoia, 2012; Jadimurthy et al., 2023] |
| Theme | Key Observations |
|---|---|
| Testing and diagnosis | 30–60% decreases during early 2020; elevated test positivity suggests missed asymptomatic cases |
| COVID-19 lockdown effect | Reduced sexual partner networks; genotype clustering in ST-9362 in Amsterdam; decreased gonorrhea diversity |
| Post-lockdown resurgence | Significant rise in diagnoses, especially in heterosexual and MSW/MSMW groups |
| Coinfection patterns | limited data; analogy with other bacteria suggests heightened risk in co-infected covid-19 patients |
| Surveillance and public health | Need for high-resolution genomic tracking and restoration of STI screening infrastructure |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




