Submitted:
17 September 2025
Posted:
19 September 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Multiple Mechanism of PCC1for Diverse Application
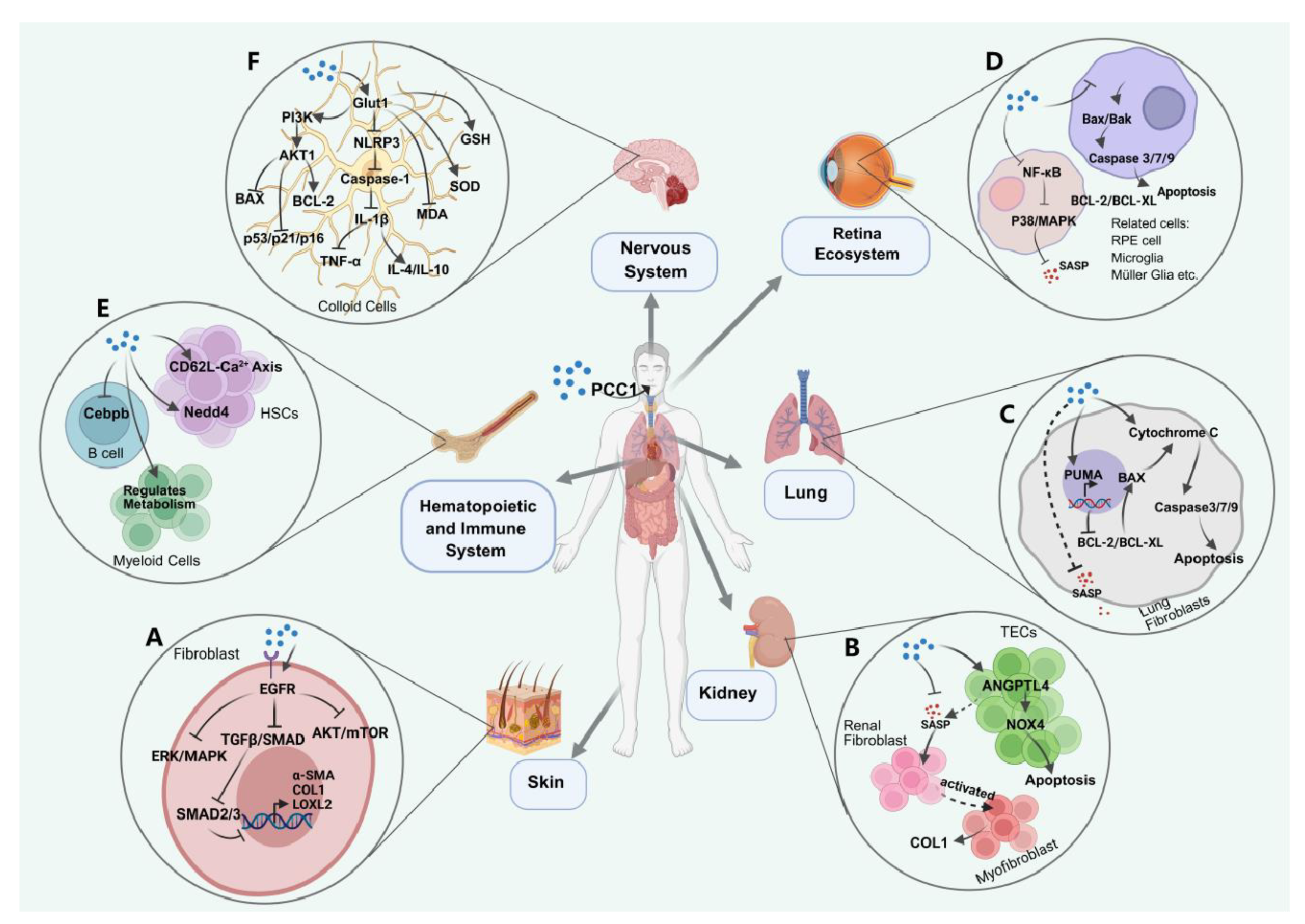
2.1. PCC1 as a Senotherapeutic Candidate
2.1.1. Dose-Dependent Dual Mechanism
2.1.2. PCC1 for Anti-Fibrosis
2.1.3. PCC1 for Retina Protection
2.1.4. PCC1 for HIS Modulation
2.1.5. PCC1 for Alzheimer’s Disease Mitigation
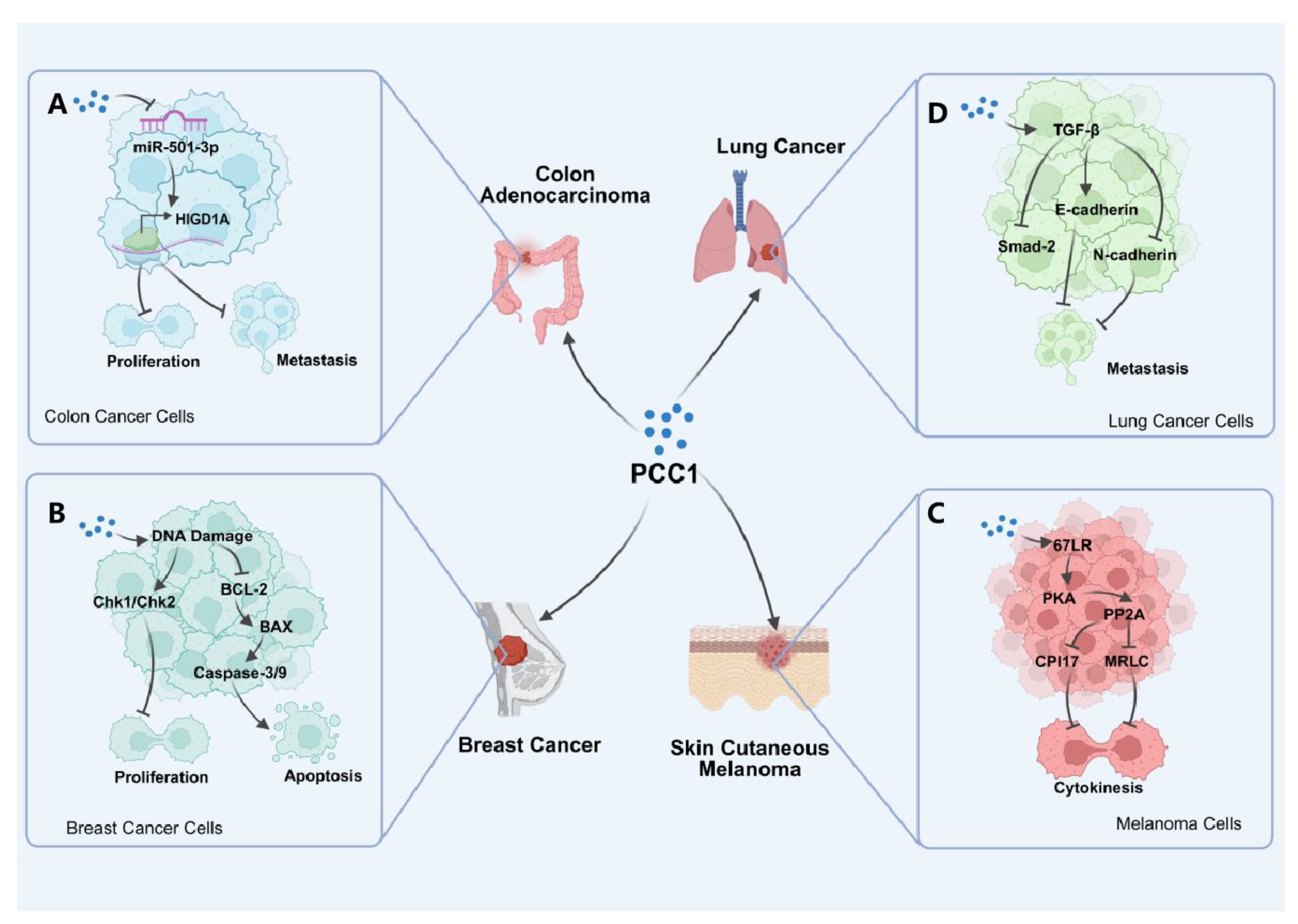
2.2. PCC1 for Anti-Cancer Application
2.3. PCC1 for Anti-Inflammation
2.4. PCC1 for Glucose Metabolism Regulation
2.5. Potential Antivirus Application
3. Safety Considerations
4. Future Directions
Author Contributions
Conflicts of Interest
References
- Guo, J. , et al. , Aging and aging-related diseases: from molecular mechanisms to interventions and treatments. Signal Transduct Target Ther 2022, 7, 391. [Google Scholar]
- Cai, Y. , et al. , The landscape of aging. Science China Life Sciences 2022, 65, 2354–2454. [Google Scholar] [CrossRef]
- Suryadevara, V. , et al. , SenNet recommendations for detecting senescent cells in different tissues. Nature Reviews Molecular Cell Biology 2024, 25, 1001–1023. [Google Scholar]
- Wong, C. , How to kill the ‘zombie’cells that make you age. 2024, Nature Publishing Group.
- Chaib, S., T. Tchkonia, and J. L. Kirkland, Cellular senescence and senolytics: the path to the clinic. Nature medicine 2022, 28, 1556–1568. [Google Scholar]
- Lelarge, V. , et al., Senolytics: from pharmacological inhibitors to immunotherapies, a promising future for patients’ treatment. npj Aging 2024, 10, 12. [Google Scholar] [CrossRef]
- Gasek, N.S. , et al. , Strategies for targeting senescent cells in human disease. Nature aging 2021, 1, 870–879. [Google Scholar]
- Boccardi, V. and P. Mecocci, Senotherapeutics: Targeting senescent cells for the main age-related diseases. Mechanisms of ageing and development 2021, 197, 111526. [Google Scholar] [CrossRef]
- Wang, B. , et al., The senescence-associated secretory phenotype and its physiological and pathological implications. Nature Reviews Molecular Cell Biology 2024, 25, 958–978. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q. , et al., The flavonoid procyanidin C1 has senotherapeutic activity and increases lifespan in mice. Nat Metab 2021, 3, 1706–1726. [Google Scholar] [CrossRef] [PubMed]
- Shao, M. , et al. Procyanidin C1 inhibits bleomycin-induced pulmonary fibrosis in mice by selective clearance of senescent myofibroblasts. FASEB J 2024, 38, e23749. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y. , et al., Senolytic and senomorphic agent procyanidin C1 alleviates structural and functional decline in the aged retina. Proc Natl Acad Sci U S A 2024, 121, e2311028121. [Google Scholar] [CrossRef]
- Lv, J.L. , et al. , Procyanidin C1 inhibits tumor growth and metastasis in colon cancer via modulating miR-501-3p/HIGD1A axis. J Adv Res 2024, 60, 215–231. [Google Scholar] [CrossRef]
- Kin, R. , et al. , Procyanidin C1 from Cinnamomi Cortex inhibits TGF-beta-induced epithelial-to-mesenchymal transition in the A549 lung cancer cell line. Int J Oncol 2013, 43, 1901–1906. [Google Scholar]
- Byun, E.B. , et al. , The procyanidin trimer C1 inhibits LPS-induced MAPK and NF-kappaB signaling through TLR4 in macrophages. Int Immunopharmacol 2013, 15, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Koteswari, L.L. , et al., A comparative anticancer study on procyanidin C1 against receptor positive and receptor negative breast cancer. Nat Prod Res 2020, 34, 3267–3274. [Google Scholar] [CrossRef]
- Sun, P. , et al., Procyanidin C1, a Component of Cinnamon Extracts, Is a Potential Insulin Sensitizer That Targets Adipocytes. J Agric Food Chem 2019, 67, 8839–8846. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H., H. J. Lee, and K.S. Kang, Procyanidin C1 Activates the Nrf2/HO-1 Signaling Pathway to Prevent Glutamate-Induced Apoptotic HT22. Cell Death. Int J Mol Sci 2019, 20. [Google Scholar]
- Duan, L. , et al., Glucose-modified BSA/procyanidin C1 NPs penetrate the blood-brain barrier and alleviate neuroinflammation in Alzheimer’s disease models. Int J Biol Macromol 2024, 268 Pt 1, 131739. [Google Scholar] [CrossRef]
- Chen, J. , et al., The Relationship between Procyanidin Structure and Their Protective Effect in a Parkinson’s Disease Model. Molecules 2022, 27. [Google Scholar]
- Wang, J.H. , et al., Procyanidin C1 ameliorates aging-related skin fibrosis through targeting EGFR to inhibit TGFbeta/SMAD pathway. Phytomedicine 2025, 142, 156787. [Google Scholar] [CrossRef]
- Gan, Y. , et al. , Senolytic procyanidin C1 alleviates renal fibrosis by promoting apoptosis of senescent renal tubular epithelial cells. Faseb j 2025, 39, e70362. [Google Scholar] [CrossRef] [PubMed]
- Liu, X. , et al., Single-cell profiling unveils a geroprotective role of Procyanidin C1 in hematopoietic immune system via senolytic and senomorphic effects. NPJ Aging 2025, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Bae, J. , et al., Procyanidin C1 Inhibits Melanoma Cell Growth by Activating 67-kDa Laminin Receptor Signaling. Mol Nutr Food Res 2020, 64, e1900986. [Google Scholar] [CrossRef]
- Kim, K.K. Sheppard, and H.A. Chapman, TGF-β1 signaling and tissue fibrosis. Cold Spring Harbor perspectives in biology 2018, 10, a022293. [Google Scholar] [CrossRef]
- Sisto, M. Ribatti, and S. Lisi, Organ fibrosis and autoimmunity: the role of inflammation in TGFβ-dependent EMT. Biomolecules 2021, 11, 310. [Google Scholar] [CrossRef]
- O’Reilly, S. -S. Tsou, and J. Varga, Senescence and tissue fibrosis: Opportunities for therapeutic targeting. Trends in molecular medicine 2024, 30, 1113–1125. [Google Scholar] [CrossRef]
- Pohlers, D. , et al., TGF-β and fibrosis in different organs—molecular pathway imprints. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2009, 1792, 746–756. [Google Scholar] [CrossRef]
- Sreekumar, P.G. R. Hinton, and R. Kannan, The Emerging Role of Senescence in Ocular Disease. Oxidative Medicine and Cellular Longevity 2020, 2020, 1–19. [Google Scholar] [CrossRef]
- Li, C. , et al., Age-related macular degeneration: a disease of cellular senescence and dysregulated immune homeostasis. Clinical Interventions in Aging, 2024: p. 939-951.
- Blasiak, J. , Senescence in the pathogenesis of age-related macular degeneration. Cellular and Molecular Life Sciences 2020, 77, 789–805. [Google Scholar] [CrossRef]
- Liao, Y.-L. , et al., Senescent endothelial cells: a potential target for diabetic retinopathy. Angiogenesis 2024, 27, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Crespo-Garcia, S. , et al., Therapeutic targeting of cellular senescence in diabetic macular edema: preclinical and phase 1 trial results. Nat Med 2024, 30, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Macha, N. , et al., Multifocal Electroretinography Changes after UBX1325 (Foselutoclax) Treatment in Neovascular Age-Related Macular Degeneration. J Clin Med. 2024, 13. [Google Scholar] [CrossRef]
- Klier, S. , et al. NEJM Evid 2025, 4, EVIDoa2400009. [Google Scholar]
- Wrona, M.V. , et al., The 3 I’s of immunity and aging: immunosenescence, inflammaging, and immune resilience. Front Aging 2024, 5, 1490302. [Google Scholar] [CrossRef] [PubMed]
- Geiger, H. Denkinger, and R. Schirmbeck, Hematopoietic stem cell aging. Curr Opin Immunol 2014, 29, 86–92. [Google Scholar] [CrossRef]
- Zheng, Q. and X. Wang, Alzheimer’s disease: insights into pathology, molecular mechanisms, and therapy. Protein Cell 2025, 16, 83–120. [Google Scholar] [CrossRef]
- Reddi Sree, R. , et al., Newer Therapeutic Approaches in Treating Alzheimer’s Disease: A Comprehensive Review. ACS Omega 2025, 10, 5148–5171. [Google Scholar] [CrossRef]
- Zhang, J. , et al., Recent advances in Alzheimer’s disease: Mechanisms, clinical trials and new drug development strategies. Signal Transduct Target Ther 2024, 9, 211. [Google Scholar] [CrossRef]
- Peng, Y. , et al., Current and future therapeutic strategies for Alzheimer’s disease: an overview of drug development bottlenecks. Front Aging Neurosci 2023, 15, 1206572. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S. , et al., Alzheimer’s disease: pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine 2019, 14, 5541–5554. [Google Scholar] [CrossRef]
- Martínez-Cué, C. and N. Rueda, Cellular Senescence in Neurodegenerative Diseases. Front Cell Neurosci 2020, 14, 16. [Google Scholar] [CrossRef]
- Liu, R.M. , Aging, Cellular Senescence, and Alzheimer’s Disease. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, M.M. , et al., Senolytic therapy in mild Alzheimer’s disease: a phase 1 feasibility trial. Nat Med 2023, 29, 2481–2488. [Google Scholar] [CrossRef]
- Srivastava, N. and A.K. Saxena, Novel small molecule inhibitors of cyclin-dependent kinases as anticancer agents. Current Medicinal Chemistry, 2025.
- Puxeddu, M., R. Silvestri, and G.L. Regina, Metabolism, a Blossoming Target for Small-Molecule Anticancer Drugs. Molecules 2025, 30, 3457. [Google Scholar] [CrossRef]
- Shi, Y. , et al., Procyanidin improves experimental colitis by regulating macrophage polarization. Biomed Pharmacother 2023, 165, 115076. [Google Scholar] [CrossRef]
- Hua, W. , et al. , Procyanidin C1 ameliorates acidic pH stress induced nucleus pulposus degeneration through SIRT3/FOXO3-mediated mitochondrial dynamics. J Transl Med 2024, 22, 1071. [Google Scholar]
- Ding, Y. , et al. , Grape seed proanthocyanidins ameliorate pancreatic beta-cell dysfunction and death in low-dose streptozotocin- and high-carbohydrate/high-fat diet-induced diabetic rats partially by regulating endoplasmic reticulum stress. Nutr Metab (Lond) 2013, 10, 51. [Google Scholar] [CrossRef]
- Zhang, Y. , et al., The application of procyanidins in diabetes and its complications: a review of preclinical studies. Frontiers in Pharmacology 2025, 16, 1532246. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y. , et al., Procyanidin promotes translocation of glucose transporter 4 in muscle of mice through activation of insulin and AMPK signaling pathways. PLoS One 2016, 11, e0161704. [Google Scholar] [CrossRef] [PubMed]
- El-Ashmawy, N.E. , et al., Upregulation of GLUT4 and PI3K, and downregulation of GSK3 mediate the anti-hyperglycemic effects of proanthocyanidins. Medicine International 2022, 2, 14. [Google Scholar] [CrossRef]
- Sun, P. , et al., Procyanidin C1, a component of cinnamon extracts, is a potential insulin sensitizer that targets adipocytes. Journal of Agricultural and Food chemistry 2019, 67, 8839–8846. [Google Scholar] [CrossRef]
- Maroli, N. , et al., The potential role of procyanidin as a therapeutic agent against SARS-CoV-2: a text mining, molecular docking and molecular dynamics simulation approach. J Biomol Struct Dyn 2022, 40, 1230–1245. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y. , et al., Senolytic drugs dasatinib and quercetin promote skin papilloma progression by eliminating senescent cells and impairing immune surveillance. Oral Science and Homeostatic Medicine 2025, 1. [Google Scholar] [CrossRef]
- Cheng, F. , et al. , Adverse reactions after treatment with dasatinib in chronic myeloid leukemia: Characteristics, potential mechanisms, and clinical management strategies. Front Oncol 2023, 13, 1113462. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, L. , et al., Phase I study of Navitoclax (ABT-263), a novel Bcl-2 family inhibitor, in patients with small-cell lung cancer and other solid tumors. J Clin Oncol 2011, 29, 909–916. [Google Scholar] [CrossRef]
- Farsad-Naeimi, A. , et al., Effect of fisetin supplementation on inflammatory factors and matrix metalloproteinase enzymes in colorectal cancer patients. Food & Function 2018, 9, 2025–2031. [Google Scholar]
- Heger, T. , et al., Procyanidin C1 from Viola odorata L. inhibits Na+, K+-ATPase. Scientific Reports 2022, 12, 7011. [Google Scholar] [CrossRef]
- Nie, F. , et al., Oligomeric proanthocyanidins: An updated review of their natural sources, synthesis, and potentials. Antioxidants 2023, 12, 1004. [Google Scholar] [CrossRef]
- Lu, M. , et al., Phyto-phospholipid complexes (phytosomes): A novel strategy to improve the bioavailability of active constituents. Asian journal of pharmaceutical sciences 2019, 14, 265–274. [Google Scholar] [CrossRef]
| Cells | Dosages | Mechanisms (index) | References |
| 661W,BV2,ARPE-19,HRMEC Cells | 50–200 μM | ↓:SA-β-GAL, p16/p21, IL-6, IL-1β, TNF-α, VEGF-A | [12] |
| Primary Pulmonary FibroblastsPSC27, WI-38,HUVEC, MSC Cells |
100-200 μM | ↓: SA-β-GAL, CXCL8, IL-1α ↑: NOXA, PUMA, ROS, MitoSOX |
[10] |
| 100 μM | ↓: SA-β-GAL, p16, p21, p53, ECM Deposition, Mitochondrial Membrane Potential (ΔΨm) ↑: Mitochondrial ROS levels |
[11] | |
| Colorectal Cancer Cell Lines | 5,10 μM | ↓: miR-501-3p, p-Rb, MMP-9, N-cadherin ↑: HIGD1A |
[13] |
| Lung Cancer Cell Line A549 | 5,10, 20 μM | ↓: N-cadherin, p-Smad2 ↑: E-cadherin |
[14] |
| RAW264.7,BMDMs | 29, 58, 115 μM | ↓: NO, TNF-α, IL-6, PGE2, CD80, CD86, MHC II, iNOS, COX-2 | [15] |
| MCF-7,MDA-MB-231 | 40 μM | ↑: BAX/Bcl-2, Caspase-3/9 | [16] |
| Murine Melanoma B16 Cells | 5 μM | ↑: PP2A, PKA | [16] |
| Mouse 3T3-L1 Preadipocytes | 25 μM | ↓: Glycerol Release ↑: TG, aP2/PPARγ, p-AKT, NO, GLUT4 |
[17] |
| HT22 Cells | 5,10 μM | ↓: p-ERK1/2,p-p38, ROS, Annexin V+ ↑: DPPH , Nrf2, HO-1 |
[18] |
| 58 μM | ↓: ROS, MDA, TNF-α, IL-1β, BAX ↑: GSH, SOD, Bcl-2, IL-4, IL-10 |
[19] | |
| PC12 Cells | 5 μM | ↓: ROS, MDA ↑: Nrf2, HO-1, NQO1 |
[20] |
| Murine fibroblast cell line L929 | 12.5, 25, 50 μM | ↓:p-EGFR, ERK/MARK, SA-β-Gal, LOXL2, COL1 ↑:EGFR |
[21] |
| HK2 | 100 μM | ↓:TGFB1, IL-1B, IL-6, MMP1, MMP3, FAS | [22] |
| Models | Dosages | Mechanisms (index) | References |
| NOD-SCID Mice | 20 mg/kg | ↓: SA-β-GAL, IL-6, CXCL8, WNT16B, tumor volume ↑: caspase-3 |
[10] |
| BALB/c Nude Mice | 20 mg/kg 40 mg/kg |
↓: miR-501-3p, Ki67+, p-Rb,MMP-9,N-cadherin ↑: HIGD1A |
[13] |
| C57BL/6J Mice | 8 mg/kg | ↓:SA-β-GAL, p16/p21, IL-6,IL-1β, TNF-α,Apoe, rem2, Spp1 | [12] |
| 20 mg/kg | ↓: SA-β-GAL, IL-6, CCL2, WNT16B , MCP-1 ↑:NOXA, PUMA, Caspase-3, Median Survival, Walking Speed, Balance Performance |
[10] | |
| 8 mg/kg | ↓:TNF, IL-6, IL-1α/β, CCL, CXCL2, MAPK ↑:Nedd4, Ube2r2, Ube2k, Cul3, Adipor1, Adipor2 |
[23] | |
| 2.5 mg/kg 5 mg/kg 10 mg/kg |
↓:p-EGFR, TGFβ1, α-SMA | [21] | |
| 20 mg/kg | ↓:SA-β-Gal, p16, p21 | [22] | |
| B16 Melanoma-Bearing Mice | 30 mg/kg | ↓: CPI17, MRLC tumor volume ↑: PP2A, PKA |
[24] |
| Zebrafish Larvae | 25 μM | ↓: ROS, MDA, GSH-Px, CAT ↑: Nrf2, HO-1, NQO1, SOD |
[20] |
| 5×FAD Transgenic Mice | 5 mg/kg | ↓:Aβ1-42, Tau, p-Tau (ser396,235), AChE, Bax, P21, P16, P53, TNF-α, IL-1β, MDA, NLRP3 ↑:BDNF, Ki67, NeuN, DCX, Bcl-2, IL-4, IL-10, GSH, SOD, PI3K/AKT |
[19] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




