Submitted:
10 September 2025
Posted:
17 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. HSCs in Fibrosis
3. Role of the TGF-β Signaling Pathway in Liver Fibrosis
4. The Role of Oxidative Stress in Liver Fibrosis
5. Molecular Function of EWSR1 and Its Fibrosis Regulatory Potential
5.1. Structure and Biological Function of EWSR1
5.1.1. EWSR1 in Transcriptional Regulation
5.1.2. EWSR1 in Non-Coding RNA Regulation
5.2. EWSR1 and Its Interaction with TGF-β Signaling
5.3. Relationship Between EWSR1 and Oxidative Stress
5.3.1. Role of EWSR1 in Stress-Granule Formation
5.3.2. Regulation of EWSR1 by Oxidative Stress
5.4. The Potential Role of EWSR 1 in Fibrosis
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Allameh, A.; Niayesh-Mehr, R.; Aliarab, A.; Sebastiani, G.; Pantopoulos, K. J. A. Oxidative stress in liver pathophysiology and disease. 2023, 12(9), 1653. [Google Scholar] [CrossRef]
- Angelopoulou, E.; Pyrgelis, E.-S.; Ahire, C.; Suman, P.; Mishra, A.; Piperi, C. Functional implications of protein arginine methyltransferases (PRMTs) in neurodegenerative diseases. Biology 2023, 12(9), 1257. [Google Scholar] [CrossRef] [PubMed]
- Antar, S. A.; Ashour, N. A.; Marawan, M. E.; Al-Karmalawy, A. A. Fibrosis: types, effects, markers, mechanisms for disease progression, and its relation with oxidative stress, immunity, and inflammation. International Journal of Molecular Sciences 2023, 24(4), 4004. [Google Scholar] [CrossRef] [PubMed]
- Averill-Bates, D. Reactive oxygen species and cell signaling. Review. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research 2024, 1871(2), 119573. [Google Scholar] [CrossRef]
- Balakrishnan, M.; Rehm, J. A public health perspective on mitigating the global burden of chronic liver disease. Hepatology 2024, 79(2), 451–459. [Google Scholar] [CrossRef]
- Bao, Y.; Niu, T.; Zhu, J.; Mei, Y.; Shi, Y.; Meng, R.; Wang, Y. Evolution and Discovery of Matrine Derivatives as a New Class of Anti-Hepatic Fibrosis Agents Targeting Ewing Sarcoma Breakpoint Region 1 (EWSR1). Journal of Medicinal Chemistry 2023, 66(12), 7969–7987. [Google Scholar] [CrossRef]
- Baradaran-Heravi, Y.; Van Broeckhoven, C.; van der Zee, J. Stress granule mediated protein aggregation and underlying gene defects in the FTD-ALS spectrum. Neurobiology of Disease 2020, 134, 104639. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Wang, J.; Wang, J.; Zhou, Q.; Yang, B.; He, Q.; Weng, Q. Intercellular crosstalk of hepatic stellate cells in liver fibrosis: New insights into therapy. Pharmacol Res 2020, 155, 104720. [Google Scholar] [CrossRef]
- Chao, G.; Zhu, Y.; Bao, Y. A screening study of high-risk groups for liver fibrosis in patients with metabolic dysfunction-associated fatty liver disease. Sci Rep 2024, 14(1), 23714. [Google Scholar] [CrossRef]
- Chen, Z.; Lin, Z.; Yu, J.; Zhong, H.; Zhuo, X.; Jia, C.; Wan, Y. Mitofusin-2 Restrains Hepatic Stellate Cells’ Proliferation via PI3K/Akt Signaling Pathway and Inhibits Liver Fibrosis in Rats. J Healthc Eng 2022, 2022, 6731335. [Google Scholar] [CrossRef]
- Chothani, S.; Schäfer, S.; Adami, E.; Viswanathan, S.; Widjaja, A. A.; Langley, S. R.; Jian Pua, C. Widespread translational control of fibrosis in the human heart by RNA-binding proteins. Circulation 2019, 140(11), 937–951. [Google Scholar] [CrossRef]
- Chun Hao Ong, C. L. T.; Hanis Hazeera Harith; Nazmi Firdaus; Daud Ahmad Israf. TGF-β-induced fibrosis: A review on the underlying mechanism and potential therapeutic strategies, 2021.
- Danieau, G.; Morice, S.; Rédini, F.; Verrecchia, F.; Brounais-Le Royer, B. New insights about the Wnt/β-catenin signaling pathway in primary bone tumors and their microenvironment: a promising target to develop therapeutic strategies? International Journal of Molecular Sciences 2019, 20(15), 3751. [Google Scholar] [CrossRef] [PubMed]
- De Conti, L.; Baralle, M.; Buratti, E. Neurodegeneration and RNA-binding proteins. In Wiley Interdisciplinary Reviews: RNA; 2017; Volume 8, 2, p. e1394. [Google Scholar]
- Deng, Z.; Fan, T.; Xiao, C.; Tian, H.; Zheng, Y.; Li, C.; He, J. TGF-β signaling in health, disease, and therapeutics. Signal transduction and targeted therapy 2024, 9(1), 61. [Google Scholar] [CrossRef]
- Dewidar, B.; Meyer, C.; Dooley, S.; Meindl-Beinker, N. TGF-β in hepatic stellate cell activation and liver fibrogenesis—updated 2019. Cells 2019, 8(11), 1419. [Google Scholar] [CrossRef] [PubMed]
- Dhar, D.; Baglieri, J.; Kisseleva, T.; Brenner, D. A. Mechanisms of liver fibrosis and its role in liver cancer. Exp Biol Med (Maywood) 2020, 245(2), 96–108. [Google Scholar] [CrossRef]
- Duarte, S.; Baber, J.; Fujii, T.; Coito, A. J. J. M. B. Matrix metalloproteinases in liver injury, repair and fibrosis. 2015, 44, 147–156. [Google Scholar] [CrossRef]
- El-Kassas, M.; Cabezas, J.; Coz, P. I.; Zheng, M. H.; Arab, J. P.; Awad, A. Nonalcoholic Fatty Liver Disease: Current Global Burden. Semin Liver Dis 2022, 42(3), 401–412. [Google Scholar] [CrossRef]
- Elswefy, S. E. S.; Abdallah, F. R.; Atteia, H. H.; Wahba, A. S.; Hasan, R. A. J. I. j. o. e. p. Inflammation, oxidative stress and apoptosis cascade implications in bisphenol A-induced liver fibrosis in male rats. 2016, 97(5), 369–379. [Google Scholar] [CrossRef] [PubMed]
- Foot, O.; Hallin, M.; Jones, R. L.; Sumathi, V. P.; Thway, K. EWSR1-SMAD3-Positive fibroblastic tumor. International Journal of Surgical Pathology 2021, 29(2), 179–181. [Google Scholar] [CrossRef]
- Friedman, S. L. J. P. r. Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. 2008, 88(1), 125–172. [Google Scholar] [CrossRef]
- Fritz, D.; Stefanovic, B. RNA-binding protein RBMS3 is expressed in activated hepatic stellate cells and liver fibrosis and increases expression of transcription factor Prx1. Journal of molecular biology 2007, 371(3), 585–595. [Google Scholar] [CrossRef]
- Ghafouri-Fard, S.; Askari, A.; Shoorei, H.; Seify, M.; Koohestanidehaghi, Y.; Hussen, B. M.; Samsami, M. Antioxidant therapy against TGF-β/SMAD pathway involved in organ fibrosis. Journal of Cellular and Molecular Medicine 2024, 28(2), e18052. [Google Scholar] [CrossRef] [PubMed]
- Hahm, K.-B.; Cho, K.; Lee, C.; Im, Y.-H.; Chang, J.; Choi, S.-G.; Kim, S.-J. Repression of the gene encoding the TGF-β type II receptor is a major target of the EWS-FLI1 oncoprotein. Nature genetics 1999, 23(2), 222–227. [Google Scholar] [CrossRef]
- Hammerich, L.; Tacke, F. Hepatic inflammatory responses in liver fibrosis. Nature reviews Gastroenterology & hepatology 2023, 20(10), 633–646. [Google Scholar]
- Hassoun, Z. E. O. A Novel Role of EWSR1 Protein in RNA Translation Regulation. Doctoral dissertation, Universite de Liege (Belgium)), 2023. [Google Scholar]
- Heisey, D. A.; Lochmann, T. L.; Floros, K. V.; Coon, C. M.; Powell, K. M.; Jacob, S.; Maves, Y. K. The ewing family of tumors relies on BCL-2 and BCL-XL to escape PARP inhibitor toxicity. Clinical Cancer Research 2019, 25(5), 1664–1675. [Google Scholar] [CrossRef]
- Henderson, N. C.; Rieder, F.; Wynn, T. A. Fibrosis: from mechanisms to medicines. Nature 2020, 587(7835), 555–566. [Google Scholar] [CrossRef]
- Hernandez-Gea, V.; Friedman, S. L. Pathogenesis of liver fibrosis. Annual review of pathology: mechanisms of disease 2011, 6(1), 425–456. [Google Scholar] [CrossRef] [PubMed]
- Horii, Y.; Matsuda, S.; Toyota, C.; Morinaga, T.; Nakaya, T.; Tsuchiya, S.; Kasai, K. VGLL3 is a mechanosensitive protein that promotes cardiac fibrosis through liquid–liquid phase separation. Nature Communications 2023, 14(1), 550. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y. K.; Yim, H. J. J. T. K. j. o. i. m. Reversal of liver cirrhosis: current evidence and expectations. 2017, 32(2), 213. [Google Scholar] [CrossRef]
- Junghee Lee, P. T. N.; Hyun Soo Shim; Seung Jae Hyeon; Hyeonjoo Im; Mi-Hyun Choi; Sooyoung Chung; Neil W. Kowall; Sean Bong Lee; Hoon Ryu. EWSR1, a multifunctional protein, regulates cellular function and aging via genetic and epigenetic pathways. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2019, 1865(7), 1938–1945. [Google Scholar] [CrossRef]
- Liao, J.; He, Q.; Li, M.; Chen, Y.; Liu, Y.; Wang, J. LncRNA MIAT: myocardial infarction associated and more. Gene 2016, 578(2), 158–161. [Google Scholar] [CrossRef] [PubMed]
- Lin, M. H.; Li, H. Q.; Zhu, L.; Su, H. Y.; Peng, L. S.; Wang, C. Y.; Wang, Y. Liver Fibrosis in the Natural Course of Chronic Hepatitis B Viral Infection: A Systematic Review with Meta-Analysis. Dig Dis Sci 2022, 67(6), 2608–2626. [Google Scholar] [CrossRef]
- Luangmonkong, T.; Suriguga, S.; Mutsaers, H. A.; Groothuis, G. M.; Olinga, P.; Boersema, M. Targeting oxidative stress for the treatment of liver fibrosis. Reviews of Physiology, Biochemistry and Pharmacology 2018, 175, 71–102. [Google Scholar] [CrossRef]
- Masi, M.; Attanzio, A.; Racchi, M.; Wolozin, B.; Borella, S.; Biundo, F.; Buoso, E. Proteostasis deregulation in neurodegeneration and its link with stress granules: focus on the scaffold and ribosomal protein RACK1. Cells 2022, 11(16), 2590. [Google Scholar] [CrossRef]
- Meng, X.-m.; Nikolic-Paterson, D. J.; Lan, H. Y. TGF-β: the master regulator of fibrosis. Nature Reviews Nephrology 2016, 12(6), 325–338. [Google Scholar] [CrossRef]
- Mo, J.; Tan, K.; Dong, Y.; Lu, W.; Liu, F.; Mei, Y.; Ye, Y. Therapeutic targeting the oncogenic driver EWSR1:: FLI1 in Ewing sarcoma through inhibition of the FACT complex. Oncogene 2023, 42(1), 11–25. [Google Scholar] [CrossRef]
- Molliex, A.; Temirov, J.; Lee, J.; Coughlin, M.; Kanagaraj, A. P.; Kim, H. J.; Taylor, J. P. Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization. Cell 2015, 163(1), 123–133. [Google Scholar] [CrossRef] [PubMed]
- Omar, K. Protein-Protein Interaction Network Perturbations Caused by Oncogenic Fusion Events. Doctoral dissertation, (Doctoral dissertation, Cornell University, 2024. [Google Scholar]
- Ong, C. H.; Tham, C. L.; Harith, H. H.; Firdaus, N.; Israf, D. A. TGF-β-induced fibrosis: A review on the underlying mechanism and potential therapeutic strategies. European journal of pharmacology 2021, 911, 174510. [Google Scholar] [CrossRef]
- Pan, Z.; Zhao, R.; Li, B.; Qi, Y.; Qiu, W.; Guo, Q.; Li, M. EWSR1-induced circNEIL3 promotes glioma progression and exosome-mediated macrophage immunosuppressive polarization via stabilizing IGF2BP3. Molecular cancer 2022, 21(1), 16. [Google Scholar] [CrossRef] [PubMed]
- Parola, M.; Pinzani, M. J. M. a. o. m. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues. 2019, 65, 37–55. [Google Scholar] [CrossRef]
- Pei, R.; Zhao, L.; Ding, Y.; Su, Z.; Li, D.; Zhu, S.; Zhou, W. JMJD6–BRD4 complex stimulates lncRNA HOTAIR transcription by binding to the promoter region of HOTAIR and induces radioresistance in liver cancer stem cells. Journal of Translational Medicine 2023, 21(1), 752. [Google Scholar] [CrossRef]
- Qian, L.; Li, L.; Li, Y.; Li, S.; Zhang, B.; Zhu, Y.; Yang, B. LncRNA HOTAIR as a ceRNA is related to breast cancer risk and prognosis. Breast Cancer Research and Treatment 2023, 200(3), 375–390. [Google Scholar] [CrossRef]
- Rajan, S. S.; Ebegboni, V. J.; Pichling, P.; Ludwig, K. R.; Jones, T. L.; Chari, R.; Caplen, N. J. EWSR1’s visual modalities are defined by its association with nucleic acids and RNA polymerase II. bioRxiv: the preprint server for biology 2023, 09(16), 553246. [Google Scholar]
- Ramos-Tovar, E.; Muriel, P. Molecular mechanisms that link oxidative stress, inflammation, and fibrosis in the liver. Antioxidants 2020, 9(12), 1279. [Google Scholar] [CrossRef]
- Rodriguez, L.; Bui, S.; Beuschel, R.; Ellis, E.; Liberti, E.; Chhina, M.; Grant, G. J. M. M. Curcumin induced oxidative stress attenuation by N-acetylcysteine co-treatment: a fibroblast and epithelial cell in-vitro study in idiopathic pulmonary fibrosis. 2019, 25, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Roehlen, N.; Crouchet, E.; Baumert, T. F. Liver Fibrosis: Mechanistic Concepts and Therapeutic Perspectives. Cells 2020, 9(4), 875. [Google Scholar] [CrossRef] [PubMed]
- Said, M. M.; Azab, S. S.; Saeed, N. M.; El-Demerdash, E. J. A. o. H. Antifibrotic mechanism of pinocembrin: impact on oxidative stress, inflammation and TGF-β/Smad inhibition in rats. 2018, 17(2), 307–317. [Google Scholar] [CrossRef]
- Salem, E. S.; Vonberg, A. D.; Borra, V. J.; Gill, R. K.; Nakamura, T. RNAs and RNA-binding proteins in immuno-metabolic homeostasis and diseases. Frontiers in Cardiovascular Medicine 2019, 6, 106. [Google Scholar] [CrossRef]
- Sánchez-Valle, V.; C Chavez-Tapia, N.; Uribe, M.; Méndez-Sánchez, N. Role of oxidative stress and molecular changes in liver fibrosis: a review. Current medicinal chemistry 2012, 19(28), 4850–4860. [Google Scholar] [CrossRef]
- Schöpf, J.; Uhrig, S.; Heilig, C. E.; Lee, K.-S.; Walther, T.; Carazzato, A.; Hartmann, M. Multi-omic and functional analysis for classification and treatment of sarcomas with FUS-TFCP2 or EWSR1-TFCP2 fusions. Nature Communications 2024, 15(1), 51. [Google Scholar] [CrossRef]
- Shi, X.; Young, C. D.; Zhou, H.; Wang, X.-J. J. B. Transforming growth factor-β signaling in fibrotic diseases and cancer-associated fibroblasts. 2020, 10(12), 1666. [Google Scholar] [CrossRef] [PubMed]
- Svineng, G.; Ravuri, C.; Rikardsen, O.; Huseby, N.-E.; Winberg, J.-O. J. C. t. r. The role of reactive oxygen species in integrin and matrix metalloproteinase expression and function. 2008, 49(3-4), 197–202. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, C.; Grundevik, P.; Elias, P.; Ståhlberg, A.; Åman, P. A conserved N-terminal motif is required for complex formation between FUS, EWSR1, TAF15 and their oncogenic fusion proteins. The FASEB Journal 2013, 27(12), 4965–4974. [Google Scholar] [CrossRef]
- Tsuchida, T.; Friedman, S. L. Mechanisms of hepatic stellate cell activation. Nature reviews Gastroenterology & hepatology 2017, 14(7), 397–411. [Google Scholar]
- Tzavlaki, K.; Moustakas, A. TGF-β Signaling. Biomolecules 2020, 10(3), 487. [Google Scholar] [CrossRef]
- Udomsinprasert, W.; Sobhonslidsuk, A.; Jittikoon, J.; Honsawek, S.; Chaikledkaew, U. J. E. o. o. t. t. Cellular senescence in liver fibrosis: Implications for age-related chronic liver diseases. 2021, 25(9), 799–813. [Google Scholar] [CrossRef]
- Wachtel, M.; Surdez, D.; Grünewald, T. G.; Schäfer, B. W. Functional Classification of Fusion Proteins in Sarcoma. Cancers 2024, 16(7), 1355. [Google Scholar] [CrossRef]
- Wang, F.; Li, J.; Fan, S.; Jin, Z.; Huang, C. Targeting stress granules: A novel therapeutic strategy for human diseases. Pharmacological research 2020, 161, 105143. [Google Scholar] [CrossRef]
- Wang, G.; Tong, J.; Li, Y.; Qiu, X.; Chen, A.; Chang, C.; Yu, G. Overview of CircRNAs Roles and Mechanisms in Liver Fibrosis. Biomolecules 2023, 13(6), 940. [Google Scholar] [CrossRef]
- Wang, S.; Sun, Z.; Lei, Z.; Zhang, H.-T. RNA-binding proteins and cancer metastasis. Seminars in cancer biology; 2022. [Google Scholar]
- Wijayasiri, P.; Astbury, S.; Kaye, P.; Oakley, F.; Alexander, G. J.; Kendall, T. J.; Aravinthan, A. D. Role of Hepatocyte Senescence in the Activation of Hepatic Stellate Cells and Liver Fibrosis Progression. Cells 2022, 11(14). [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-C.; Luo, S.-Z.; Liu, T.; Lu, L.-G.; Xu, M.-Y. linc-SCRG1 accelerates liver fibrosis by decreasing RNA-binding protein tristetraprolin. The FASEB Journal 2019, 33(2), 2105–2115. [Google Scholar] [CrossRef]
- Wu, P.; Zuo, X.; Deng, H.; Liu, X.; Liu, L.; Ji, A. Roles of long noncoding RNAs in brain development, functional diversification and neurodegenerative diseases. Brain research bulletin 2013, 97, 69–80. [Google Scholar] [CrossRef]
- Xie, Z.; Wu, Y.; Liu, S.; Lai, Y.; Tang, S. LncRNA-SNHG7/miR-29b/DNMT3A axis affects activation, autophagy and proliferation of hepatic stellate cells in liver fibrosis. Clinics and research in hepatology and gastroenterology 2021, 45(2), 101469. [Google Scholar] [CrossRef]
- Xu, F.; Liu, C.; Zhou, D.; Zhang, L. TGF-β/SMAD pathway and its regulation in hepatic fibrosis. Journal of Histochemistry & Cytochemistry 2016, 64(3), 157–167. [Google Scholar]
- Yamada, A.; Toya, H.; Tanahashi, M.; Kurihara, M.; Mito, M.; Iwasaki, S.; Kawamura, Y. Species-specific formation of paraspeckles in intestinal epithelium revealed by characterization of NEAT1 in naked mole-rat. RNA 2022, 28(8), 1128–1143. [Google Scholar] [CrossRef] [PubMed]
- Yang, J. H.; Kim, S. C.; Kim, K. M.; Jang, C. H.; Cho, S. S.; Kim, S. J.; Ki, S. H. Isorhamnetin attenuates liver fibrosis by inhibiting TGF-β/Smad signaling and relieving oxidative stress. European journal of pharmacology 2016, 783, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.-L.; Chang, W.-T.; Hong, M.-Y.; Hung, K.-C.; Chuang, C.-C. J. P. O. Prevention of TGF-β-induced early liver fibrosis by a maleic acid derivative anti-oxidant through suppression of ROS, inflammation and hepatic stellate cells activation. 2017, 12(4), e0174008. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Fan, L.; Yin, Z.; Liu, Y.; Zhao, D.; Wang, Z.; Cheng, H. EWSR1:: SMAD3-rearranged fibroblastic tumor: A case with twice recurrence and literature review. Frontiers in Oncology 2022, 12, 1017310. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, M.; Li, W.; Liu, C.; Jiang, Z.; Gu, P.; medicine, t. Rebalancing TGF-β/Smad7 signaling via Compound kushen injection in hepatic stellate cells protects against liver fibrosis and hepatocarcinogenesis. 2021, 11(7), e410. [Google Scholar] [CrossRef]
- Zhang, J.; Han, C.; Song, K.; Chen, W.; Ungerleider, N.; Yao, L.; Wu, T. The long-noncoding RNA MALAT1 regulates TGF-β/Smad signaling through formation of a lncRNA-protein complex with Smads, SETD2 and PPM1A in hepatic cells. PLoS One 2020, 15(1), e0228160. [Google Scholar] [CrossRef]
- Zhang, X.; Yuan, L.; Zhang, W.; Zhang, Y.; Wu, Q.; Li, C.; Huang, Y. Liquid–liquid phase separation in diseases. MedComm 2024, 5(7), e640. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. E. Non-Smad pathways in TGF-β signaling. Cell research 2009, 19(1), 128–139. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yao, Z.; Wang, L.; Ding, H.; Shao, J.; Chen, A.; Zheng, S. Activation of ferritinophagy is required for the RNA-binding protein ELAVL1/HuR to regulate ferroptosis in hepatic stellate cells. Autophagy 2018, 14(12), 2083–2103. [Google Scholar] [CrossRef]
- Zhao, J.; Bai, D.; Qi, L.; Cao, W.; Du, J.; Gu, C.; Lu, N. The flavonoid GL-V9 alleviates liver fibrosis by triggering senescence by regulating the transcription factor GATA4 in activated hepatic stellate cells. Br J Pharmacol 2023, 180(8), 1072–1089. [Google Scholar] [CrossRef] [PubMed]
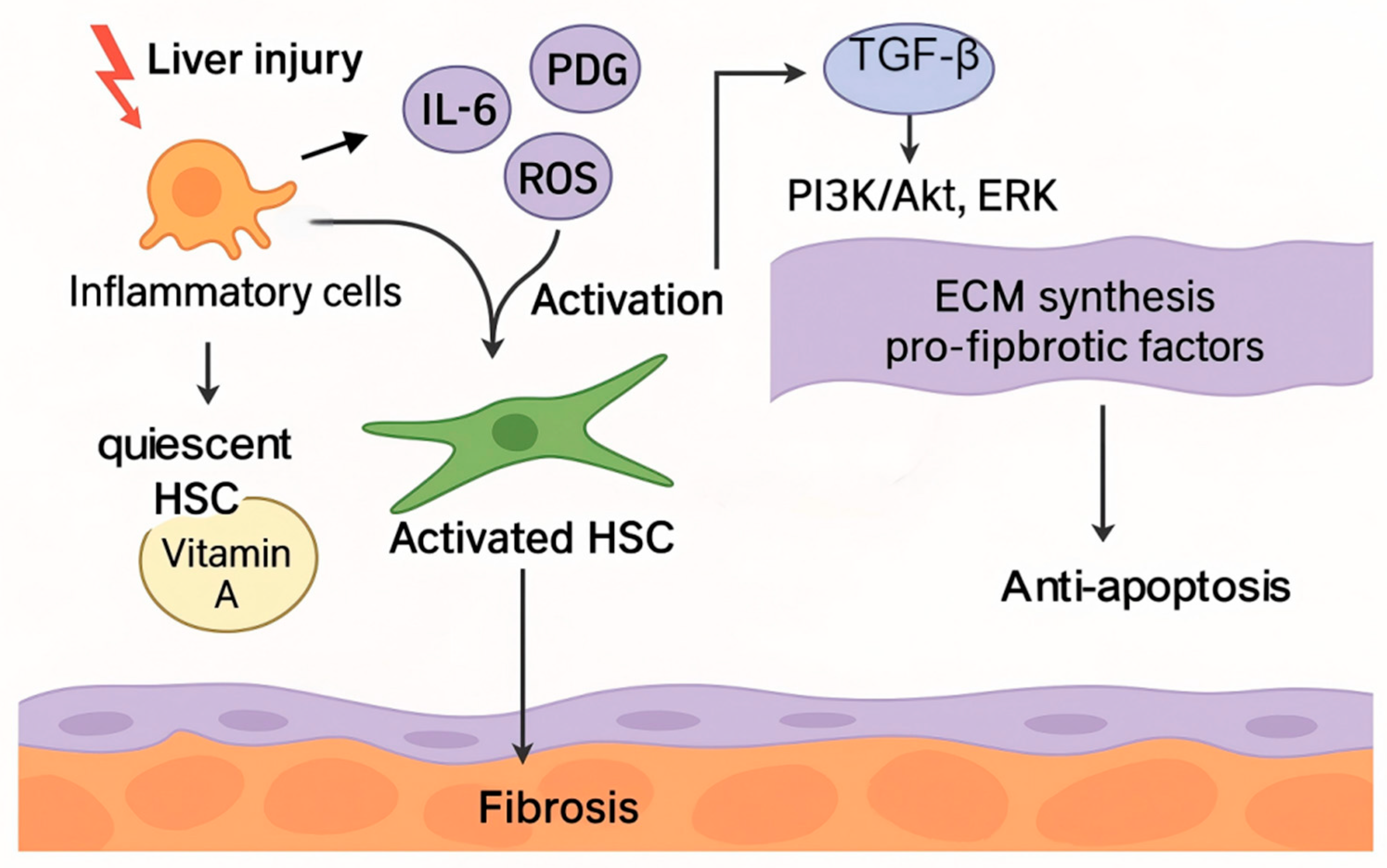
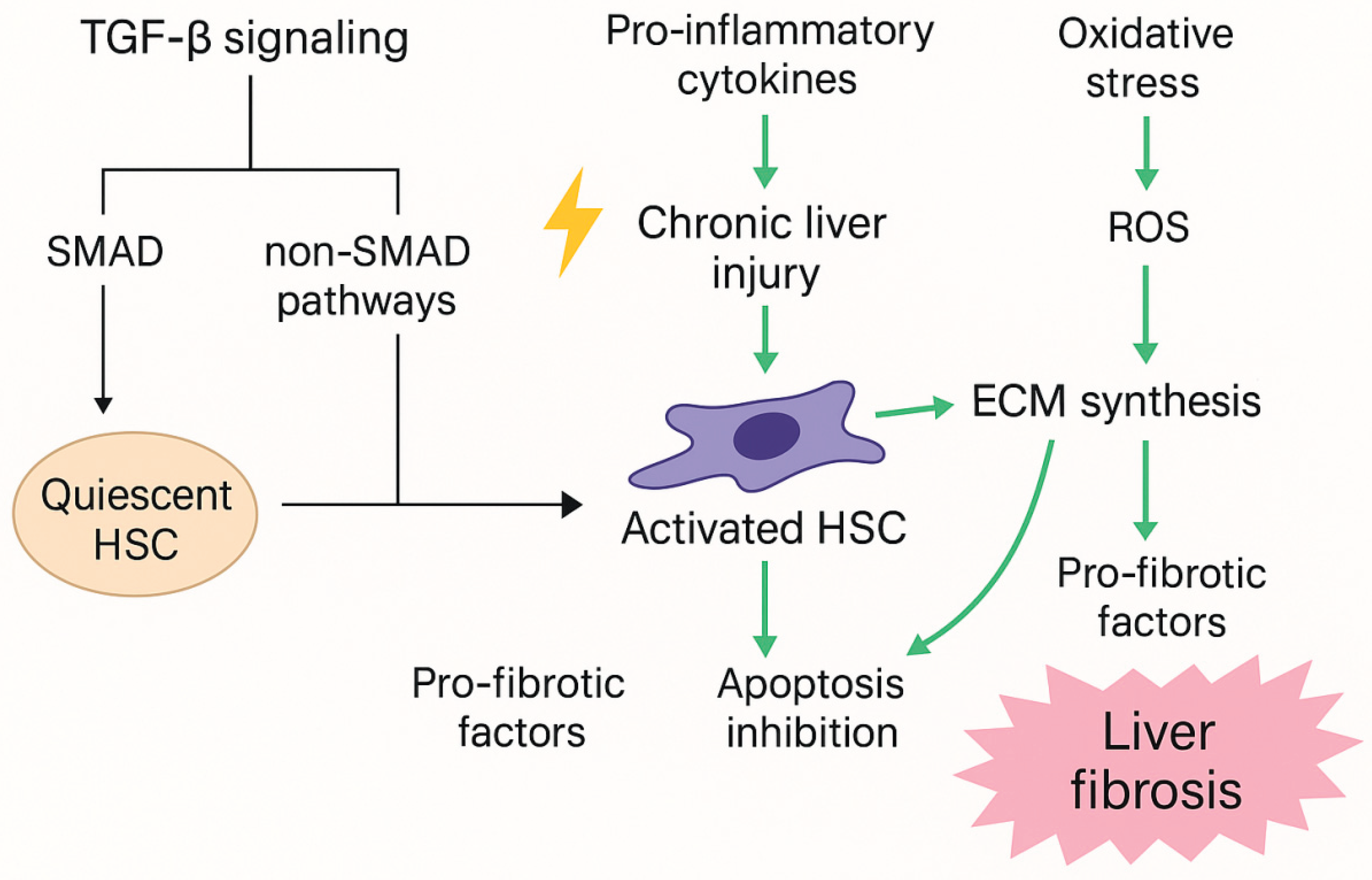
| Vital Mechanism | Role of TGF-β and ROS | The effects of fibrosis progression | Reference |
| TGF-β/SMAD signaling pathway | TGF-β binds to SMAD protein through its receptor and activates the SMAD2/3 pathway, leading to the accumulation of ECM and promoting the formation of fibrosis. ROS induces TGF-β signaling by inducing its expression and promoting latent TGF-β activation via LAP. |
The activated TGF-β/SMAD pathway promotes the activation and proliferation of HSCs, which ultimately accelerates fibrosis. | Deng et al., 2024 Dewidar et al., 2019 |
| Activation of HSCs | TGF-β stimulates HSCs to transform into myofibroblasts, resulting in more collagen and ECM components. ROS enhanced the activation of HSCs and further increased the synthesis of collagen and fibrosis factors. |
The activation of HSCs is central to the fibrotic process, promoting the expansion of fibrosis. | Shi et al., 2020 Yang et al., 2017 |
| ECM deposition | Upregulation of TGF-β receptors (TGF-βR1/2), CTGF, and SMAD promotes excessive ECM deposition and reduced degradation. ROS regulates ECM turnover by activating MMPs and suppressing TIMPs. | Excessive accumulation of ECM in fibrosis leads to liver sclerosis and destruction of normal liver function. | Antar et al., 2023 Svineng et al., 2008 |
| Oxidative stress and cell death | TGF-β may reduce oxidative stress by enhancing antioxidant response, but it can also induce cell death by inducing the generation of ROS. ROS oxidizes cellular components, triggering apoptosis or necrosis in HSCs. |
ROS-driven HSC death and hepatocyte injury exacerbate fibrotic progression. | Ghafouri-Fard et al., 2024 Ramos-Tovar & Muriel, 2020 |
| Signal feedback mechanism | The activation of TGF-β promotes the generation of ROS, forming a vicious circle and further activating the TGF-β/SMAD signaling pathway. ROS can enhance the TGF-β signaling pathway in a variety of ways, such as by enhancing the expression of TGF-β receptors or activating related kinases. |
ROS and TGF-β promote each other to form a feedback loop of fibrosis, leading to the aggravation of fibrosis. | Antar et al., 2023 |
| Functional Category | Molecular Mechanism | Downstream Effects | Key References |
| Transcriptional Regulation | Interacts with transcription factors and RNA polymerase II; modulates chromatin accessibility | Promotes transcription of target genes, including growth factors, cell cycle regulators, and pro-fibrotic factors | (Rajan et al., 2023); (Wachtel et al., 2024) |
| Co-activation in Transcription | Interacts with TATA-binding protein and transcription initiation complexes | Enhances transcription initiation efficiency; stabilizes the transcriptional machinery | (Hassoun, 2023) |
| TGF-β/SMAD Pathway Modulation | Binds SMAD3, a key mediator of TGF-β signaling | Regulates HSCs activation and expression of pro-fibrotic genes | (Yang et al., 2022) |
| Long Non-Coding RNA Regulation | Modulates expression, stability, and function of lncRNAs | Influences cell differentiation, proliferation, signal transduction, and fibrosis | (Hassoun, 2023); (Zhang et al., 2020) |
| Circular RNA Regulation | Binds specific circRNAs, regulates back-splicing and stability | Promotes cell proliferation, anti-apoptosis, and ECM synthesis | (Pan et al., 2022); (Wang et al., 2023) |
| MicroRNA Regulation | Binds miRNA precursors; influences maturation and function | Modulates HSCs proliferation, apoptosis, and fibrosis-related gene expression | (Horii et al., 2023); (Xie et al., 2021) |
| Fibrosis-Specific Effect | Downregulates anti-fibrotic miRNAs (e.g., miR-29b) | Increases collagen production and ECM deposition | (Horii et al., 2023) |
| Cross-Disease Regulatory Role | Regulates gene expression via ncRNAs in tumors, cardiovascular, and neurodegenerative diseases | Influences cell proliferation, apoptosis, inflammation, and ECM synthesis | (Pei et al., 2023; Qian et al., 2023); (Liao et al., 2016); (Wu et al., 2013). |
| Function/Role | Target/Mechanism | Experimental Model/Methods | Findings/Effects | References |
| Modulation of TGF-β signaling | Binding to Smad mRNA; interaction with Smad3 protein; regulation of TGF-β1, COL1A1, α-SMA, CTGF | LX-2 cells (HSC line), overexpression and knockdown experiments, qPCR, Western blot |
Overexpression increases fibrosis markers; knockdown decreases markers; stabilizes mRNA of fibrosis-related genes; promotes HSC activation | (Bao et al., 2023); (Hahm et al., 1999). |
| Interaction with non-classical TGF-β pathways | Modulation of PI3K/Akt, MAPK, JAK/STAT | Cell signaling assays | Enhances HSCs migration; promotes anti-apoptotic effects | (Mo et al., 2023). (Deng et al., 2024) |
| Stress granule formation under oxidative stress | Scaffolding via RRM and low-complexity domain (LCD); interaction with TIA-1, G3BP1, FUS; LLPS-mediated assembly | Stress-induced LX-2 cells, fluorescence imaging | Promotes SG assembly; inhibits translation temporarily; conserves energy; protects RNA; maintains cellular homeostasis | (Wang et al., 2020); (Zhang et al., 2024); (Molliex et al., 2015), |
| Regulation under oxidative stress | Nuclear-cytoplasmic translocation; post-translational modifications; interaction with NF-κB, PI3K/AKT, MAPK | ROS treatment in HSCs, Western blot, immunofluorescence | Enhances transcriptional regulatory capacity; modulates stress-related gene expression; maintains adaptive response | Averill-Bates, 2024 (Averill-Bates, 2024); Angelopoulou et al., 2023 (Angelopoulou et al., 2023) |
| Regulation of apoptosis in HSCs | Modulation of Bcl-2, Bax, Caspase-3; inhibition of PI3K/AKT | LX-2 cells and fibrotic mouse models; apoptosis assays (flow cytometry, TUNEL) | Promotes apoptosis by downregulating anti-apoptotic proteins and upregulating pro-apoptotic proteins; weakens PI3K/AKT survival signaling | Zhang et al., 2024 (Zhang et al., 2024) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




