Submitted:
15 September 2025
Posted:
17 September 2025
You are already at the latest version
Abstract
Keywords:
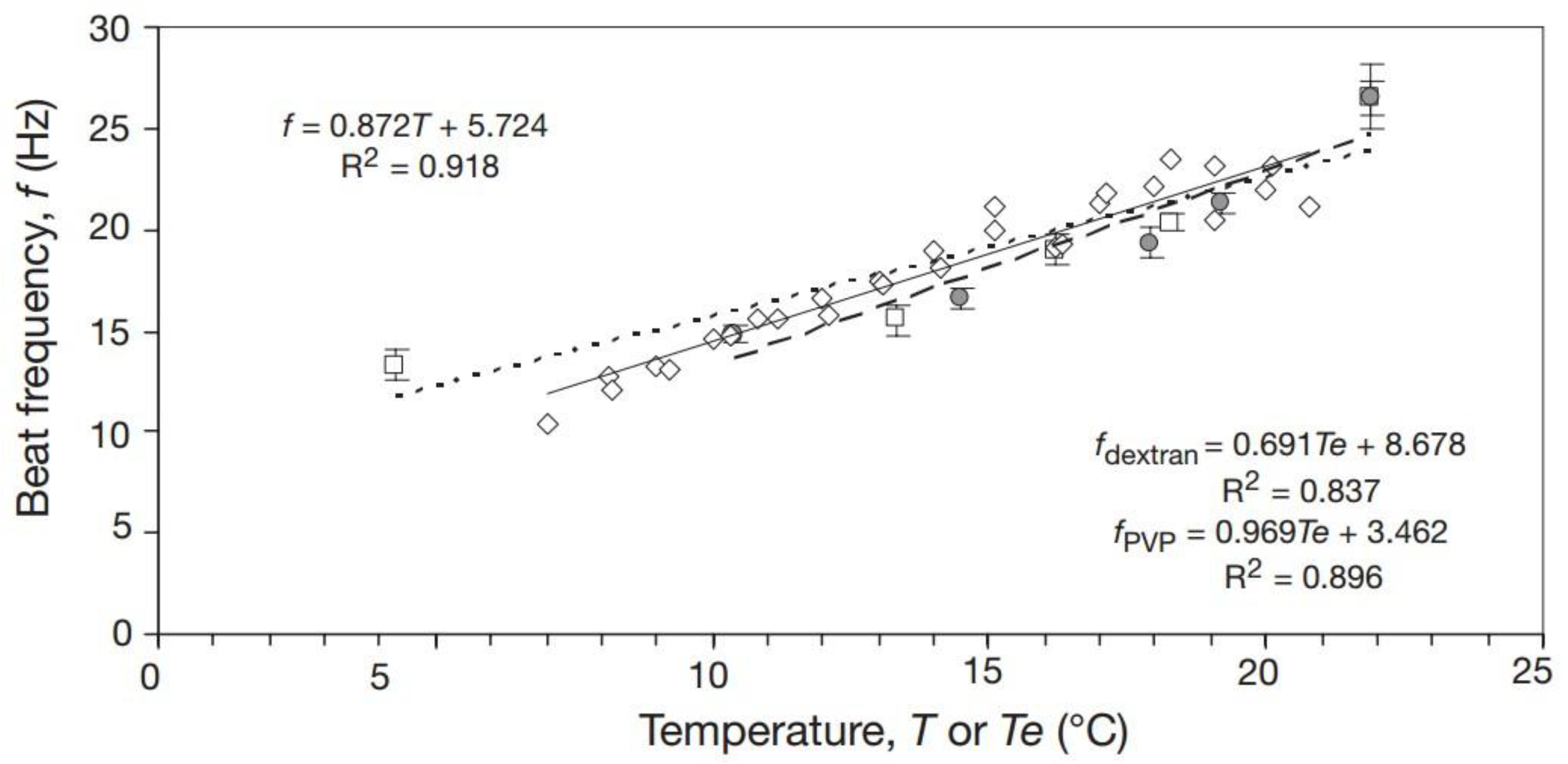
1. Introduction
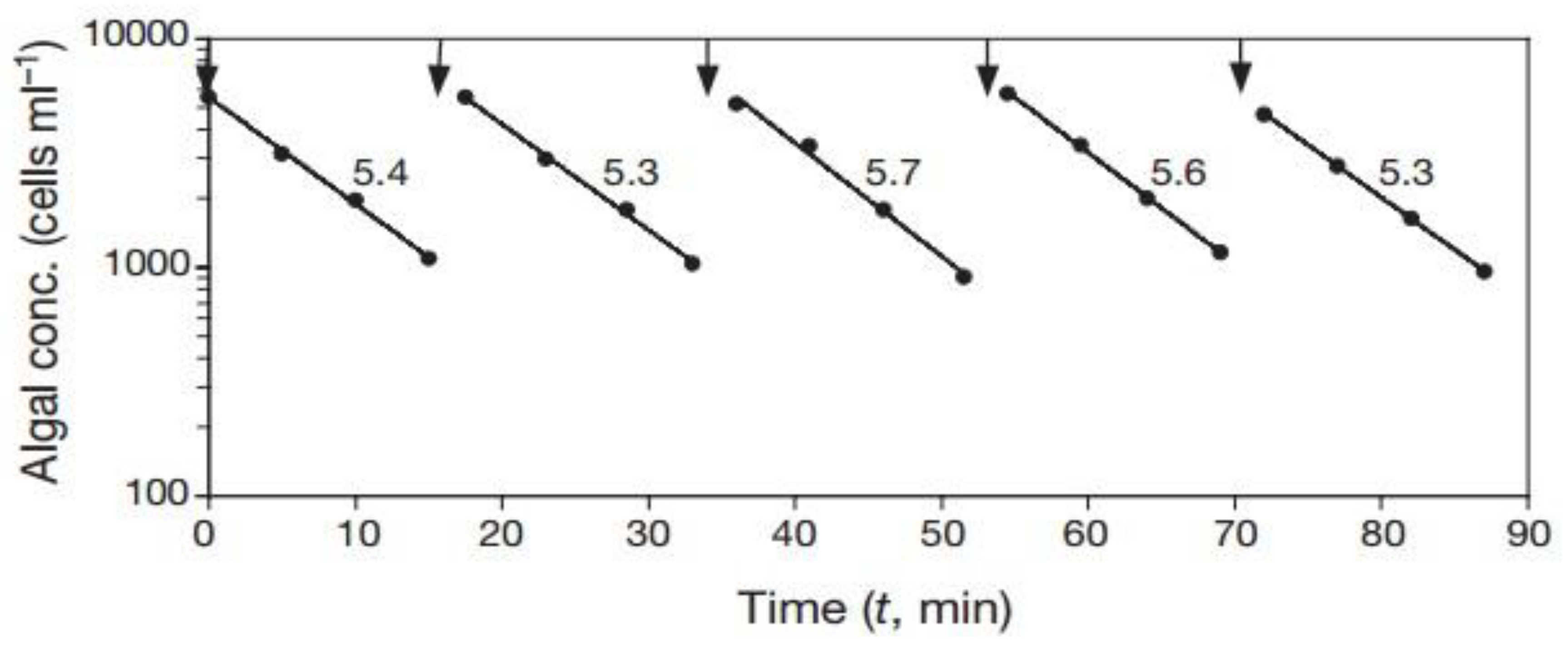
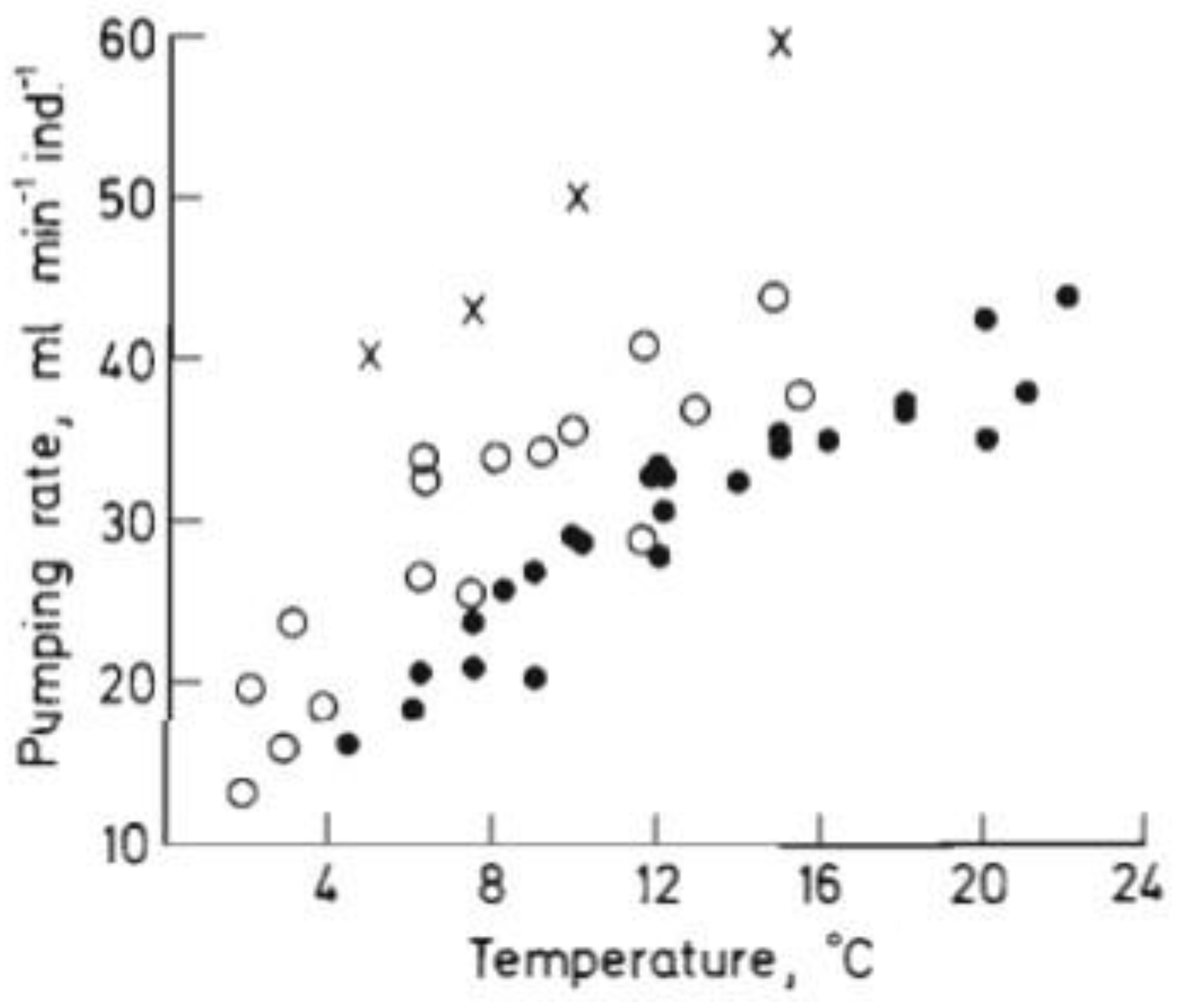
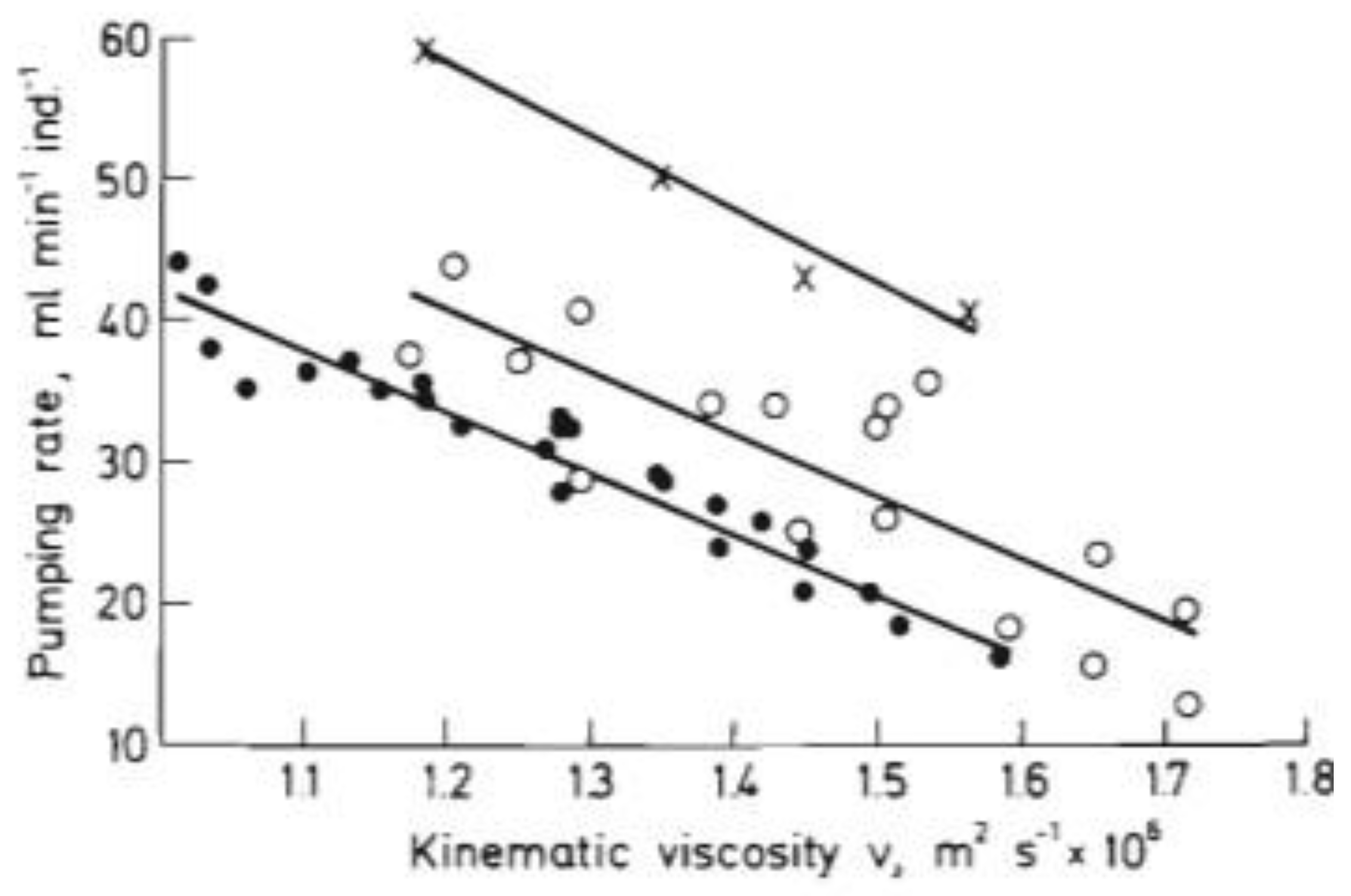
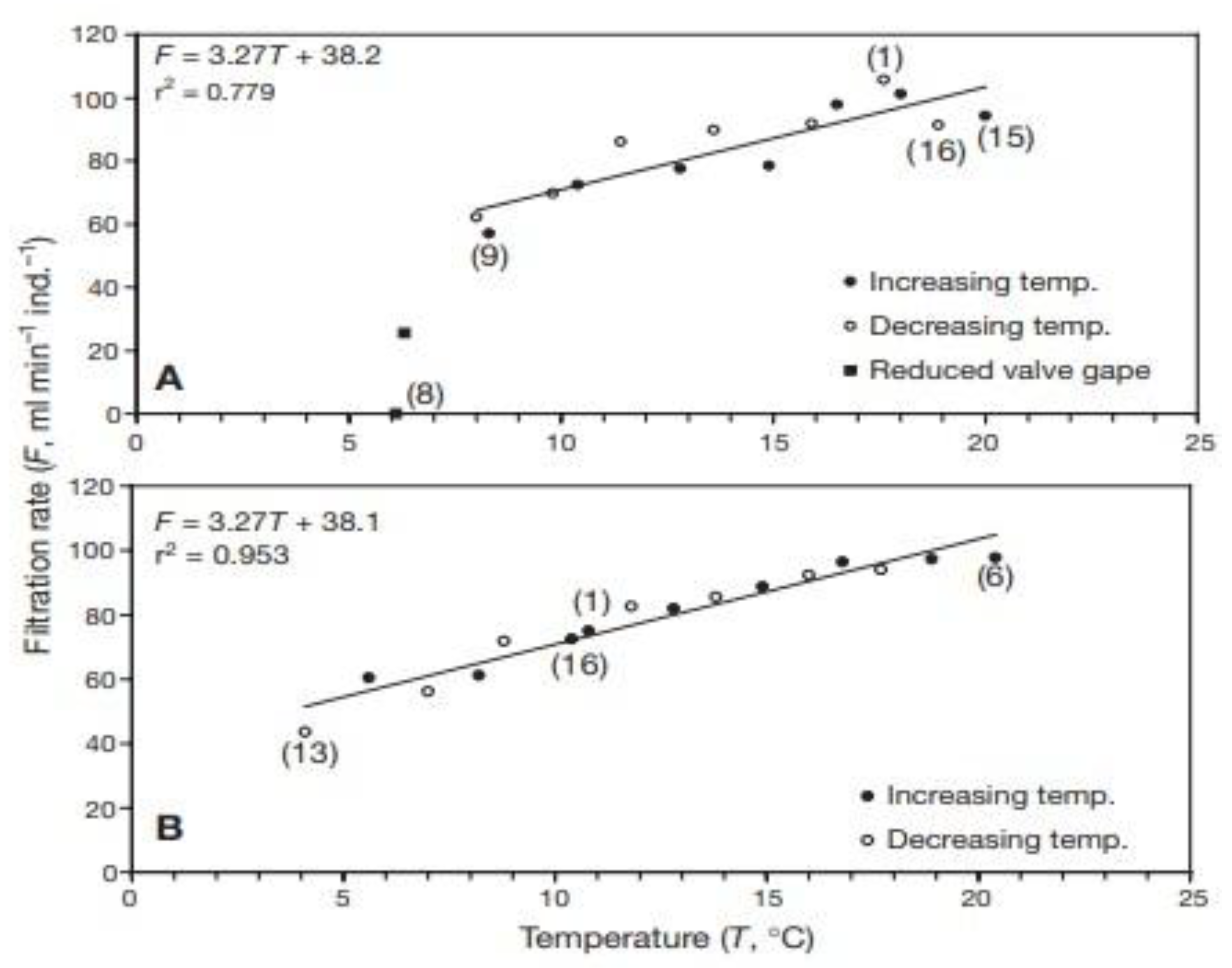
2. Our present Understanding
2.1. Recommendations, Critical Remarks and Pitfalls
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cranford, P. J., Ward, J. E., Shumway, S. E. (2011). Bivalve filter feeding: Variability and limits of the aquaculture biofilter. In S. E. Shumway (Ed.), Shellfish aquaculture and the environment (pp. 81–124). John Wiley & Sons Publ. [CrossRef]
- Gopalakrishnan, K. K., Kashian, D. R. (2020). Identification of optimal calcium and temperature conditions for quagga mussel filtration rates as a potential predictor of invasion. Environmental Toxicology and Chemistry 39, 410–418.
- Jørgensen, C. B., Larsen, P. S., Riisgård, H. U. (1990). Effects of temperature on the mussel pump. Marine Ecology Progress Series, 64, 89–97.
- Kittner, C., Riisgård, H. U. (2005). Effect of temperature on filtration rate in the mussel Mytilus edulis: No evidence for temperature compensation. Marine Ecology Progress Series, 305, 147-152.
- Larsen, P. S., Riisgård, H. U. (2009). Viscosity and not biological mechanisms often controls the effects of temperature on ciliary activity and swimming velocity of small aquatic organisms. Journal of Experimental Marine Biology and Ecology, 381, 67-73.
- Larsen, P. S, Riisgård, H. U. (2012). Validation of the flow-through chamber (FTC) and steady-state (SS) methods for clearance rate measurements in bivalves. Biology Open. 2012 Jan 15;1(1):6-11. [CrossRef]
- Lassen, J., Kortegård, M., Riisgård, H. U., Friedrichs, M., Graff, G., Larsen, P. S. (2006). Down-mixing of phytoplankton above filter-feeding mussels - interplay between water flow and biomixing. Marine Ecology Progress Series, 314, 77-88.
- Lüskow, F., Riisgård H. U. (2018). In situ filtration rates of blue mussels (Mytilus edulis) measured by an open-top chamber method. Open Journal of Marine Science, 8, 395-406.
- Marroni, S., Mezzeo, N., Iglesias, C. (2021). Effects of temperature and food availability on the filtration and excretion rates of Diplodon parallelopipedon (Hyriidae). International Review of Hydrobiology 106, 249-258. [CrossRef]
- Pestana, D., Ostrensky, A., Boeger, W. A. P., Pie, M. R. (2009). The effect of temperature and body size on filtration rates of Limnoperna fortunei (Bivalvia, Mytilidae) under laboratory conditions. Brazilian Archives of Biology and Technology, 52, 135-144.
- Petersen, J. K., Sejr, M, K., Larsen, J, E. N, (2003). Clearance rates in the Arctic bivalves Hiatella arctica and Mya sp. Polar Biology 26:334–341.
- Riisgård, H. U. (2001). On measurement of filtration rate in bivalves - the stony road to reliable data, review and interpretation. Marine Ecology Progress Series, 211, 275-291.
- Riisgård, H. U., Larsen, P. S. (2007). Viscosity of seawater controls beat frequency of water-pumping cilia and filtration rate of mussels Mytilus edulis. Marine Ecology Progress Series, 343, 141–150.
- Riisgård, H. U., Larsen, P. S. (2009). Ciliary-propelling mechanism, effect of temperature and viscosity on swimming speed, and adaptive significance of the 'jumping' ciliate Mesodinium rubrum. Marine Biology Research, 5, 585-595.
- Riisgård, H. U., Larsen, P. S. (2015). Physiologically regulated valve-closure makes mussels long-term starvation survivors: test of hypothesis. Journal of Molluscan Studies, 81, 303-307.
- Riisgård, H. U., Larsen, P. S. (2018). Thermal and viscous effects on ciliary suspension-feeding bivalves - no need for a new explanation: Comment on Specht & Fuchs (2018). Marine Ecology Progress Series, 596, 263-265.
- Riisgård, H. U., Seerup, D. F. (2003). Filtration rates in the soft clam Mya arenaria: Effects of temperature and body size. Sarsia, 88, 416-428.
- Riisgård, H. U., Kittner, C., Seerup, D.F. (2003). Regulation of opening state and filtration rate in filter-feeding bivalves (Cardium edule, Mytilus edulis, Mya arenaria) in response to low algal concentration. Journal of Experimental Marine Biology and Ecology, 284, 105-127.
- Riisgård, H. U., Lassen, J., Kittner, C. (2006). Valve-gape response times in mussels (Mytilus edulis) - effects of laboratory preceding-feeding conditions and in situ tidally induced variation in phytoplankton biomass. Journal of Shellfish Research, 25, 901-913.
- Riisgård, H. U., Egede, P. P., Saavedra, I. B. (2011). Feeding behaviour of mussels, Mytilus edulis, with a mini-review of current knowledge. Journal of Marine Biology, Volume 2011, Article ID 312459, 13 pages. [CrossRef]
- Riisgård, H. U., Pleissner, D., Lundgreen, K., Larsen P. S. (2013). Growth of mussels Mytilus edulis at algal (Rhodomonas salina) concentrations below and above saturation levels for reduced filtration rate. Mar. Biol. Res. 9(10): 1005-1017.
- Riisgård, H. U., Lundgreen, K., Larsen, P. S. (2014a). Potential for production of 'mini-mussels' in Great Belt (Denmark) evaluated on basis of actual growth of young mussels Mytilus edulis. Aquaculture International, 22, 859-885.
- Riisgård, H. U., Larsen, P. S., Pleissner, D. (2014b). Allometric equations for maximum filtration rate in blue mussels Mytilus edulis and importance of condition index. Helgoland Marine Research, 68, 193-198.
- Riisgård, H. U., Funch, P., Larsen, P. S. (2015). The mussel filter–pump - Present understanding, with a re-examination of gill preparations. Acta Zoologica, 96(3), 273–282.
- Rosa, M., Capriotti, M., Austin, K., Shumway, S. E., Ward, J. E. (2024). Effect of seasonal changes in temperature on capture efficiency in the blue mussel, Mytilus edulis, fed seston and microplastics. Invertebrate Biology, e12446. [CrossRef]
- Tang, B., Riisgård, H. U. (2016). Physiological regulation of valve-opening degree enables mussels Mytilus edulis to overcome starvation periods by reducing the oxygen uptake. Open Journal of Marine Science, 6, 341-352.
- Zhang, J. H., Xu, M. Z., Yang, Y. (2024). Assessing the effects of environmental factors on filtration rates of golden mussel (Limnoperna fortunei). Ecological Indicators, 158, 111544. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



