Submitted:
11 September 2025
Posted:
12 September 2025
You are already at the latest version
Abstract
Chimeric antigen receptor T cell (CAR-T) therapy is a significant and costly immunotherapy strategy that has provided benefits to many patients with cancer and autoimmune diseases. As of January 2025, only a few FDA- and NMPA-approved CAR-T therapies were available for clinical use, targeting CD19 or B cell maturation antigen (BCMA) on cancer cells. The manufacturing process for these approved CAR-T cells mainly relies on lentiviral vectors (LVVs) due to their high efficiency in transducing functional genes in both dividing and nondividing cells, as they have the unique ability to integrate into the genomes of those types of cells stably, and post-mitotic mammalian cells, a capability that ɣ-retroviruses do not possess to the same extent. This review summarizes the main CAR-T therapies derived from LVVs, the basic biological principles of HIV derived LVVs, the LV structure, capacities, and functions in biotechnology, a comparison between different vectors, the CAR T structure, and a summary of manufacturing processes, with an emphasis on the microbiological perspective for human and environmental biosafety. Additionally, we outline the latest developments in LVV technology, providing insights into the production of next-generation CAR-T therapies by using in vivo approaches.
Keywords:
1. Introduction
2. Basic Biology of the Human Immunodeficiency Virus-1 (HIV-1) LVVs.
2.1. What Are LVVs?
2.2. What Are the LV Structure, Capacities, and Functions in Biotechnology?
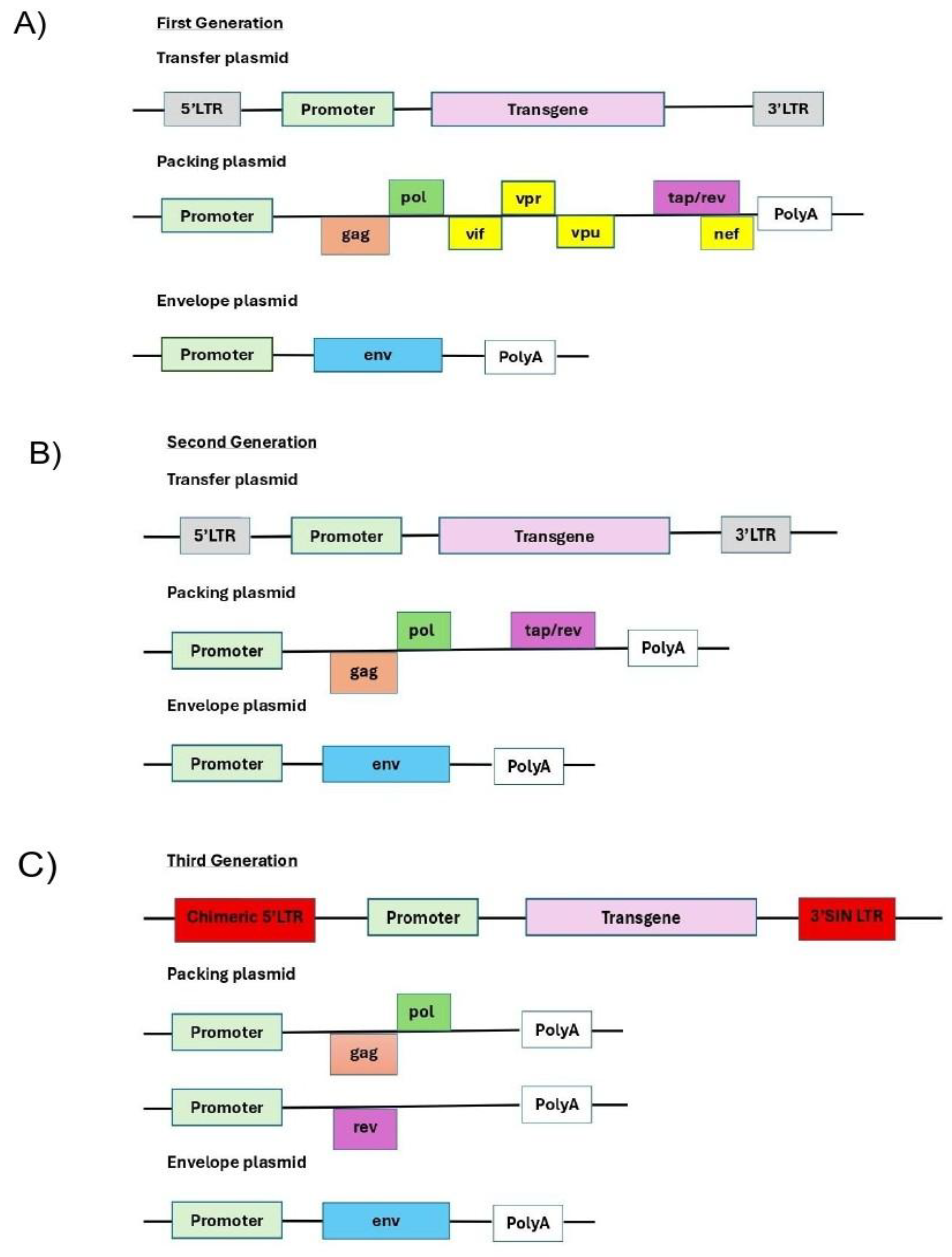
- (A)
- The first generation is composed by three plasmids: Transfer + Packaging + Envelope. Full HIV-1 genome minus env in packaging; accessory genes present (vif, vpr, vpu, nef). Tat-dependent LTR; non-SIN; high homology to wild-type..
- (B)
- The second generation is composed also by three plasmids:Transfer + gag/pol + tat + rev(packaging) + Envelope. The difference from the first is that accessory genesare deleted (Δvif, Δvpr, Δvpu, Δnef); often still non-SIN; tat + rev retained in packaging.
- (C)
- The third generation is composed by four plasmids: Transfer (SIN) + gag/pol + rev + Envelope. At a difference from previous generations rev is moved to a separate plasmid; SIN LTRs (ΔU3) standard; heterologous 5′ promoter; cPPT/CTS and WPRE commonly used. [12]
2.3. The Comparison Between Viral Vectors
| Characteristic | Lentivirus (LV) | ɣ Retrovirus | Adenovirus (AAVs) |
|---|---|---|---|
| Genome Integration Ability | Stable integration into the host genome | Less stable integration compared to LV | Cannot integrate into the host genome |
| Cell Type Target | Dividing, non-dividing, and post-mitotic cells | Primarily dividing cells | Dividing and non-dividing cells |
| Immunogenicity | Low | Moderate | High |
| Design and Preparation Time | Short | Moderate | Long |
| Application Range | Broad, suitable for various cell types and animal models | Limited, mainly used in dividing cells | Generally used for short-term gene expression or vaccine development |
| Vector Capacity | Relatively small | Relatively small | Large |
| Stability | High | Moderate | High |
2.4. LVs’ Summary of Advantages: [48]
- o
- High-efficiency gene delivery: LVs can efficiently deliver exogenous genes into target cells, including those that are difficult to transfect, such as primary and stem cells.
- o
- Long-term gene expression: Once integrated into the host genome, LVs can achieve long-term, stable gene expression, a crucial aspect for treating chronic diseases.
- o
- Broad host range: LVs can infect various cell types, including dividing and nondividing cells, expanding their range of applications.
- o
- Low immunogenicity: Modified LVVs typically have low immunogenicity, reducing the risk of host immune responses.
- o
- Safety: Modern LVVs have been genetically engineered to remove pathogenic genes, enhancing their safety.
2.5. Other Applications of LVs
- o
- Basic research: In molecular and cell biology, LLVs are utilized for gene overexpression, gene knockout, and knock-in experiments. Gene knockout involves inactivating a gene by replacing it with an artificial piece of DNA. In contrast, knock-in experiments involve the insertion of a gene into a specific location in the genome. These techniques aid in the study of gene functions and disease mechanisms. Large-scale collaborative efforts are underway to use LVs to block the expression of specific genes using RNA interference technology in high-throughput formats. Conversely, LVVs are also employed to stably over-express certain genes, thus allowing researchers to examine the effects of increased gene expression in a model system. For example, gene editing technologies mediated by LV, such as CRISPR/Cas9, can repair or replace mutated genes.[12]
- o
- Stable cell line construction: LV can be used to make stable cell lines in the same manner as standard retroviruses. The process involves infecting host cells with recombinant LLVs or pseudo-typed LVVs that carry selectable markers, such as the puromycin resistance gene. This gene confers antibiotic resistance to the infected host cells. When these antibiotics are added to the growth medium of the host cells, they kill any cells that have not incorporated the LV genome. [1]Those surviving cells can be expanded to create stable cell lines that include the lentiviral genome and harbor the genetic information encoded by it.[1] For instance, in vaccine development, LLVs can be vaccine carriers to express pathogen antigens, thereby inducing an immune response.[61] It is used in the development of HIV vaccines and other vaccines for infectious diseases. [14,16,62]
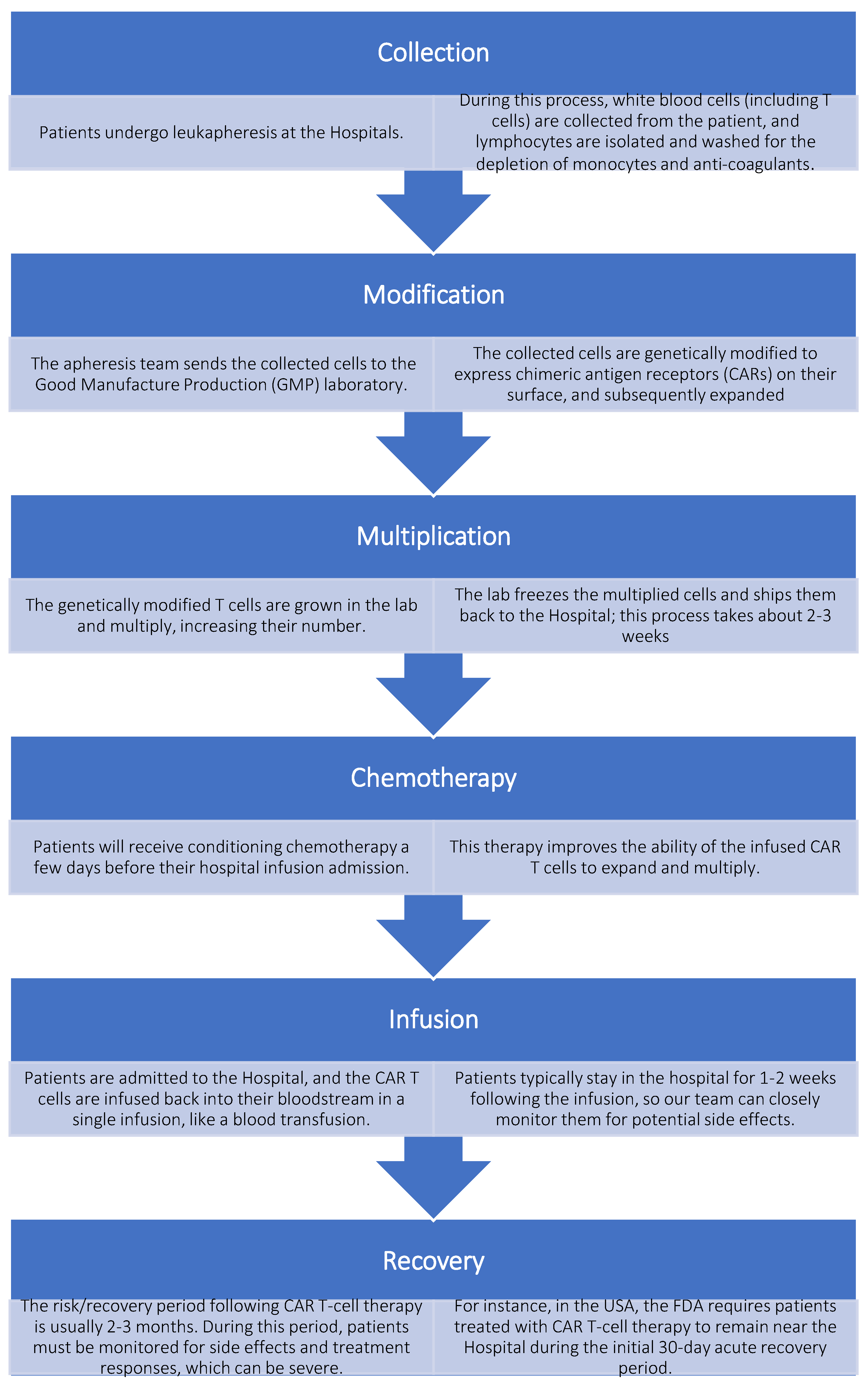
3. Manufacturing CAR T Cells Ex Vivo
3.1. How Is the CAR-T Cell Structure?
4. Risk Assessment While Working with LVs
4.1. Modes of Transmission
4.2. Containment Level
4.3. Project Review Process
4.4. Selection of a BSL-2+ Lab Space
5. Facility Considerations
5.1. Engineering Controls
- o
- Certified Class II BSCs;
- o
- Sealed centrifuge rotors and safety cups;
- o
- Vacuum lines with an in-line HEPA filter and a primary and secondary vacuum flask containing a 10% bleach solution.
5.2. Examples of Modifications to BSL-3 Practices
5.3. Personal Protective Equipment (PPE)
5.4. Waste Disposal Procedures
5.5. Additional Considerations for Implementing BSL-2+
5.6. Other Facility Infrastructure Requirements
| Cleanroom Standard | Cleanroom Classification Guidelines | |||||
|---|---|---|---|---|---|---|
| ISO 14644-1 | Class 3 | Class 4 | Class 5 | Class 6 | Class 7 | Class 8 |
| EU GMP (at rest) | - | - | A/B | - | C | D |
| US Federal Standard 209F (replaced by ISO 14644 in 2011 | 1 | -10 | 100 | 1,000 | 10,000 | 100,000 |
6. Recommendations for Working with LVVs
6.1. Practices and Procedures While Working with LVVs
6.2. Special Handling Procedures
6.3. Impatient Design Rooms for CAR-T Application to Human Subjects
7. Emergency Procedures and Exposure Management
7.1. Exposure Response and First Aid Measures
7.2. Spill Response and Decontamination Procedures
8. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Open Access
Abbreviations
| 4-1BB or CD137 | A surface glycoprotein that belongs to the tumor necrosis factor receptor family |
| AAVs | Adeno-associated virus |
| ALL | Acute lymphoblastic leukemia |
| BSCs | Biological Safety Cabinets |
| BSL-2+ | Biological Safety Level 2+ |
| BSL-3 | Biological Safety Level 3 |
| BSO | Biosafety Office |
| BCMA | Anti-B cell maturation antigen |
| CA | Capsid |
| CAR-CIK | CAR-cytokine-induced killer cell |
| CAR-MΦ | CAR-macrophage |
| CAR-NK | CAR-natural killer cell |
| CAR-T | Cell chimeric antigen receptor T cell |
| CD19 | Cluster of differentiation 19 . |
| CD28 | Cluster of differentiation 28. |
| CD3ζ | Accessory signaling molecule |
| CRISPR | Clustered regularly interspaced palindromic repeats |
| DLBCL | Diffuse large B-cell lymphoma. |
| Env | Envelop |
| EHS | Environmental Health and Safety |
| EIAV | Equine infectious anemia virus |
| FDA | Food and Drug Administration (USA) |
| FIV | Feline immunodeficiency virus |
| FL | Follicular lymphoma |
| Gag | Group-specific antigen |
| GMP | Good manufacturing practice |
| HIV | Human immunodeficiency virus |
| HSCT | Hematopoietic stem cell transplantation |
| IN | Integrase |
| IBC | Institutional Biosafety Committee |
| kb | Kilo bytes |
| KI | Knocking-In |
| KO | Knocking Out |
| LTR | Long terminal repeat |
| LV | Lentivirus |
| LVVs | Lentivirus vectors |
| MA | Matrix |
| MCL | Mantle cell lymphoma |
| MM | Multiple myeloma |
| NC | Nucleocapsid |
| NMPA | National Medical Products and Drug Administration (PR China) |
| PPE | Personal Protective Equipment |
| PA | Phosphatidic Acid |
| PI | Principal Investigator |
| Pol | Polymerase |
| Poly A | Poly-Adenine tail |
| PR | Protease |
| R | Repeat region |
| RCL | Replication-competent lentivirus |
| Rev | Regulator of the expression of viral protein |
| RNA | Ribonucleic acid |
| RRE | Rev response element |
| RT | Reverse transcriptase |
| sgRNA | Single guide RNA |
| shRNA | Short hairpin RNA |
| SOP | Standard Operating Procedure |
| SIV | Simian immunodeficiency virus |
| TAA | Tumor-associated antigens |
| Tat | Trans-activator of transcription |
| U3 | Unique 3’ region |
| U5 | Unique 5’ region |
| Vif | Viral infectivity factor |
| Vpr | Viral protein R |
| Vpu | Viral protein U |
| VSV-G | Vesicular stomatitis virus G |
| Ѱ | Retroviral psi packaging element |
References
- Yang G. Lentiviral Vectors in CAR-T Cell Manufacturing: Biological Principles, Manufacturing Process, and Frontier Development. Highl. Sci. Eng. Technol. 2023;74:172 80. [CrossRef]
- Sadelain M, Rivière I, Brentjens R. Targeting tumours with genetically enhanced T lymphocytes. Nat Rev Cancer 2003;3:35–45. [CrossRef]
- Zhao Z, Chen Y, Francisco NM, Zhang Y, Wu M. The Application of CAR-T Cell Therapy in Hematological Malignancies: Advantages and Challenges. Acta Pharm Sin B 2018;8:539–51. [CrossRef]
- Han D, Xu Z, Zhuang Y, Ye Z, Qian Q. Current Progress in CAR-T Cell Therapy for Hematological Malignancies. J Cancer 2021;12:326–34. [CrossRef]
- Chen Y-J, Abila B, Mostafa Kamel Y. CAR-T: What Is Next? Cancers (Basel) 2023;15:663. [CrossRef]
- FDA Package Insert-KYMRIAH. [(accessed on 13 December 2022)];2022 Available online: https://www.fda.gov/media/107296/download.
- Eshhar Z, Waks T, Gross G, Schindler DG. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci USA. 1993;90:720–4. [CrossRef]
- Heuser C, Hombach A, Lösch C, Manista K, Abken H. T-cell activation by recombinant immunoreceptors: Impact of the intracellular signalling domain on the stability of receptor expression and antigen-specific activation of grafted T cells. Gene Ther 2003;10:1408–19. [CrossRef]
- [FDA. Package Insert-YESCARTA. 2022. Available online: https://www.fda.gov/media/108377/download (accessed on 12 August 2025).
- [Bretscher PA. A two-step, two-signal model for the primary activation of precursor helper T cells. Proc Natl Acad Sci USA. 1999;96:185–90. [CrossRef]
- FDA. Package Insert-TERAKUS. 2022. Available online: https://www.fda.gov/media/140409/download (accessed on 12 August 2025).
- Dong W, Kantor B. Lentiviral Vectors for Delivery of Gene-Editing Systems Based on CRISPR/Cas: Current State and Perspectives. Viruses 2021;13:1288. [CrossRef]
- FDA. Package Insert-ABECMA. 2021. Available online: https://www.fda.gov/media/147055/download (accessed on 12 August 2025).
- van Heuvel Y, Schatz S, Rosengarten JF, Stitz J. Infectious RNA: Human Immunodeficiency Virus (HIV) Biology, Therapeutic Intervention, and the Quest for a Vaccine. Toxins (Basel) 2022;14:138. [CrossRef]
- FDA. Package Insert-BREYANZI. 2022. Available online: https://www.fda.gov/media/145711/download (accessed on 12 August 2025).
- Labbé RP, Vessillier S, Rafiq QA. Lentiviral Vectors for T Cell Engineering: Clinical Applications, Bioprocessing and Future Perspectives. Viruses 2021;13:1528. [CrossRef]
- FDA. Package Insert-CARVYKTI. 2022. Available online: https://www.fda.gov/media/156560/download (accessed on 12 August 2025).
- Meng B, Lever AM. Wrapping up the bad news – HIV assembly and release. Retrovirology 2013;10:5. [CrossRef]
- National Medical Products Administration. Equecabtagene Autoleucel Injection Approved with Conditions by China NMPA. https://english.nmpa.gov.cn/2023-06/30/c_940256.htm 2023, June 30 (Accessed 8 August 2025).
- Ying Z, Song Y, Yang H, Guo Y, Li W, Zou D, Zhou D, Wang Z, Zhang M, Wu J, Liu H, Wang Ch, Yang S, Zhou Z, Qin Y, Zhu J. Two-year follow-up result of RELIANCE study, a multicenter phase 2 trial of relmacabtagene autoleucel in Chinese patients with relapsed/refractory large B-cell lymphoma. J Clin Oncol. 2022;40:7529–7529.
- Addgene. Lentiviral Guide. https://www.AddgeneOrg/Guides/Lentivirus/ 2025.
- Gale RP. Chimeric antigen receptor-T-cell therapy: China leading the way. Blood Science 2022;4:176–176. [CrossRef]
- Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, Jäger U, Jaglowski S, Andreadis Ch, Westin JR, Fleury I, Bachanova V, Foley SR, Ho PJ, Mielke, Magenau JM, Holte H, Pantano S, Pacaud LB, Awasthi R, Chu J, Anak O, Salles G, Maziarz RT. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med. 2019;380:45–56. [CrossRef]
- Pasquini MC, Hu Z-H, Curran K, Laetsch T, Locke F, Rouce R, Pulsipher MA, Phillips Ch L, Keating A, Frigault MJ, Salzberg D, Jaglowski S, Sasine JP, Rosenthal J, Ghosh M, Landsburg D, Margossian S, Martin PL, Kamdar MK, Hematti P, Nikiforow S, Turtle C, Perales MA, Steinert P, Horowitz MM, Moskop A, Pacaud L, Yi L, Chawla R, Bleickardt E, Grupp S. Real-world evidence of tisagenlecleucel for pediatric acute lymphoblastic leukemia and non-Hodgkin lymphoma. Blood Adv 2020;4:5414–24. [CrossRef]
- Rezalotfi A, Fritz L, Förster R, Bošnjak B. Challenges of CRISPR-based gene editing in primary T cells. Int J Mol Sci. 2022;23:1689. 10.3390/ijms23031689.
- Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, Lin Y, Braunschweig I, Hill BT, Timmerman JM, Deol A, Reagan PM, Stiff P, Flinn IW, Farooq U, Goy A, McSweeney PA, Munoz J, Siddiqi T, Chavez JC, Herrera AF, Bartlett NL, Wiezorek JS, Navale L, Xue A, Jiang Y, Bot A, Rossi JM, Kim JK, Go WY, Neelapu SS. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet Oncol 2019;20:31–42. [CrossRef]
- Neelapu SS, Dickinson M, Munoz J, Ulrickson ML, Thieblemont C, Oluwole OO, Herrera AF, Ujjani Ch.S, Yi Lin Y, Riedell PA, Kekre N, de Vos S, Lui Ch, Milletti F, Dong J, Xu H, Chavez JC. Axicabtagene ciloleucel as first-line therapy in high-risk large B-cell lymphoma: the phase 2 ZUMA-12 trial. Nat Med 2022;28:735–42. [CrossRef]
- Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T, Lin Y, Timmerman JM, Stiff PJ, Friedberg JW, Flinn IW, Goy A, Hill BT, Smith MR, Deol A, Farooq U, McSweeney P, Munoz J, Avivi I, Castro JE, Westin JR, Chavez JC, Ghobadi A, Komanduri KV, Levy R, Jacobsen ED, Witzig TE, Reagan P, Bot A, Rossi J, Navale L, Jiang Y, Aycock J, Elias M, Chang D, Wiezorek J, Go WY. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N Engl J Med. 2017;377:2531–44. [CrossRef]
- Fosum Pharma. Fosun Kite’s First CAR-T Product Yescarta® (Axicabtagene Ciloleucel) Approved for Marketing. June 23, 2021. Available at: Fosun Kite’s First CAR-T Product Yescarta®(Axicabtagene Ciloleucel) Approved for Marketing_R&D News_R&D News_R&D_Fosun Pharma. (Accessed 13 August 2025).
- Andrea AE, Chiron A, Mallah S, Bessoles S, Sarrabayrouse G, Hacein-Bey-Abina S. Advances in CAR-T Cell Genetic Engineering Strategies to Overcome Hurdles in Solid Tumors Treatment. Front Immunol 2022;13. [CrossRef]
- Ball G, Lemieux C, Cameron D, Seftel MD. Cost-Effectiveness of Brexucabtagene Autoleucel versus Best Supportive Care for the Treatment of Relapsed/Refractory Mantle Cell Lymphoma following Treatment with a Bruton’s Tyrosine Kinase Inhibitor in Canada. Curr Oncol 2022;29:2021–45. [CrossRef]
- Frey N V. Approval of brexucabtagene autoleucel for adults with relapsed and refractory acute lymphocytic leukemia. Blood 2022;140:11–5. [CrossRef]
- Mann H, Comenzo RL. Evaluating the Therapeutic Potential of Idecabtagene Vicleucel in the Treatment of Multiple Myeloma: Evidence to Date. Onco Targets Ther 2022;Volume 15:799–813. [CrossRef]
- Munshi NC, Anderson LD, Shah N, Madduri D, Berdeja J, Lonial S, Raje N, Lin Y, Siegel D, Oriol A, Moreau Ph, Yakoub-Agha I, Delforge M, Cavo M, Einsele H, Goldschmidt H, Weisel K, Rambaldi A, Reece D, Petrocca F, Massaro M, Connarn JN, Kaiser S, Patel P, Huang L, Campbell TB, Hege K, San-Miguel J. Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma. N Engl J Med. 2021;384:705–16. [CrossRef]
- Kamdar M, Solomon SR, Arnason J, Johnston PB, Glass B, Bachanova V, Ibrahimi S, Mielke S, Mutsaers P, Hernandez-Ilizaliturri F, Izutsu K. Lisocabtagene maraleucel versus standard of care with salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment in patients with relapsed or refractory large B-cell lymphoma (TRANSFORM): results from an interim analysis of an open-label, randomised, phase 3 trial. The Lancet. 2022;399:2294–308. [CrossRef]
- Iragavarapu C, Hildebrandt G. Lisocabtagene Maraleucel for the treatment of B-cell lymphoma. Expert Opin Biol Ther 2021;21:1151–6. [CrossRef]
- Abramson JS, Palomba ML, Gordon LI, Lunning MA, Wang M, Arnason J, Mehta A, Purev E, Maloney DG, Andreadis C, Sehgal A. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. The Lancet 2020;396:839–52. [CrossRef]
- National Medical Products Administration. Ciltacabtagene Autoleucel Injection Approved for Marketing by China NMPA. 2025, Feb. 2025. https://english.nmpa.gov.cn/2025-02/19/c_1073597.htm (Accessed 8 August 2025).
- Berdeja JG, Madduri D, Usmani SZ, Jakubowiak A, Agha M, Cohen AD, Stewart AK, Hari P, Htut M, Lesokhin A, Deol A. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. The Lancet 2021;398:314–24. [CrossRef]
- Martin T, Usmani SZ, Berdeja JG, Agha M, Cohen AD, Hari P, Avigan D, Deol A, Htut M, Lesokhin A, Munshi NC. Ciltacabtagene autoleucel, an Anti–B-cell Maturation Antigen Chimeric Antigen Receptor T-Cell Therapy, for Relapsed/Refractory Multiple Myeloma: CARTITUDE-1 2-Year Follow-Up. J Clin Oncol. 2023;41:1265–74. [CrossRef]
- Legend Biotech. CARVYKTI® (ciltacabtagene autoleucel) Granted Conditional Approval by the European Commission for the Treatment of Patients with Relapsed and Refractory Multiple Myeloma. 2022 May 26. Available at: https://investors.legendbiotech.com/news-releases/news-release-details/carvyktir-ciltacabtagene-autoleucel-granted-conditional-approval.
- Hu Y, Feng J, Gu T, Wang L, Wang Y, Zhou L, et al. CAR T-cell therapies in China: rapid evolution and a bright future. Lancet Haematol 2022;9:e930–41. [CrossRef]
- Abu Haleeqa MI, El-Najjar I, Afifi YK, Ali SA, Alsaadawi N, Dennison JD, Sachdev M, Obeidat SA, Castillo-Aleman YM, Handgretinger R, Ibrahim A, Wahdan R, al Kaabi F, Ventura-Carmenate Y, Hadi LA, Hernández-Ramírez A, Al Karam M. Establishment of CD19-CAR-T Cell Production and Treatment of Non-Hodgkin Lymphoma in the United Arab Emirates. Blood 2024;144:7268–7268. [CrossRef]
- Stroncek DF, Tran M, Frodigh SE, David-Ocampo V, Ren J, Larochelle A, Sheikh V, Sereti I, Miller JL, Longin K, Sabatino M. Preliminary evaluation of a highly automated instrument for the selection of CD34+ cells from mobilized peripheral blood stem cell concentrates. Transfusion 2015; 56(2): 511–517. [CrossRef]
- Nickolay LE, Cheung GWC, Pule M, Thrasher A, Johnston I, Kaiser A, Pule M, Thrasher A, Qasim W. Automated lentiviral transduction of T cells with CARs using the CliniMACS Prodigy. European Society of Gene and Cell Therapy Collaborative Congress 2015, Helsinki, Finland. A26.
- Wang X, Rivière I. Clinical manufacturing of CAR T cells: foundation of a promising therapy. Mol Ther Oncolytics 2016;3:16015. [CrossRef]
- Lana, M.G., Strauss, B.E. Production of Lentivirus for the Establishment of CAR-T Cells. In: Swiech, K., Malmegrim, K., Picanço-Castro, V. (eds) Chimeric Antigen Receptor T Cells. Methods in Molecular Biology, 2020; 2086. Humana, New York, NY. [CrossRef]
- Dufait I, Liechtenstein T, Lanna A, Bricogne C, Laranga R, Padella A, Breckpot K, Escors D. Retroviral and Lentiviral Vectors for the Induction of Immunological Tolerance. Scientifica (Cairo) 2012;2012:1–14. [CrossRef]
- Lewis, P.F.; Emerman, M. Passage through mitosis is required for oncoretroviruses but not for the human immunodeficiency virus. J. Virol. 1994; 68, 510–516. [CrossRef]
- Kantor, B.; Bailey, R.M.; Wimberly, K.; Kalburgi, S.N.; Gray, S.J. Methods for Gene Transfer to the Central Nervous System. Agric. Food Prod. 2014, 87, 125–197. [CrossRef]
- Cronin J, Zhang XY, Reiser J. Altering the tropism of lentiviral vectors through pseudotyping. Curr Gene Ther. 2005 Aug;5(4):387-98. [CrossRef]
- Coffin JM, Hughes SH, Varmus HE. The Interactions of Retroviruses and their Hosts. In: Coffin JM, Hughes SH, Varmus HE, editors. Retroviruses. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press; 1997. PMID: 21433350.
- Naldini, L.; Blomer, U.; Gage, F.H.; Trono, D.; Verma, I.M. Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector. Proc. Natl. Acad. Sci. USA. 1996, 93, 11382–11388. [CrossRef]
- Blömer U, Naldini L, Kafri T, Trono D, Verma IM, Gage FH. Highly efficient and sustained gene transfer in adult neurons with a lentivirus vector. J Virol. 1997 Sep;71(9):6641-9. [CrossRef]
- Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D, Naldini L. A third-generation lentivirus vector with a conditional packaging system. J Virol. 1998 Nov;72(11):8463-71. [CrossRef]
- Kafri T, Blömer U, Peterson DA, Gage FH, Verma IM. Sustained expression of genes delivered directly into liver and muscle by lentiviral vectors. Nat Genet. 1997 Nov;17(3):314-7. [CrossRef]
- Zufferey R, Nagy D, Mandel RJ, Naldini L, Trono D. Multiply attenuated lentiviral vector achieves efficient gene delivery in vivo. Nat Biotechnol. 1997 Sep;15(9):871-5. [CrossRef]
- Cockrell AS, Ma H, Fu K, McCown TJ, Kafri T. A trans-lentiviral packaging cell line for high-titer conditional self-inactivating HIV-1 vectors. Mol Ther. 2006 Aug;14(2):276-84. [CrossRef]
- Zufferey R, Donello JE, Trono D, Hope TJ. The Woodchuck hepatitis virus post-transcriptional regulatory element enhances the expression of transgenes delivered by retroviral vectors. J Virol. 1999 Apr;73(4):2886-92. [CrossRef]
- Zennou V, Petit C, Guetard D, Nerhbass U, Montagnier L, Charneau P. HIV-1 genome nuclear import is mediated by a central DNA flap. Cell. 2000 Apr 14;101(2):173-85. [CrossRef]
- Di Nunzio F, Felix T, Arhel NJ, Nisole S, Charneau P, and Beignon AS. "HIV-derived vectors for therapy and vaccination against HIV." Vaccine 2012 30; 15: 2499-2509. [CrossRef]
- Beignon A, Mollier K, Liard C, Coutant F, Munier S, SRivière J, Souque P, Charneau P. Lentiviral Vector-Based Prime/Boost Vaccination against AIDS: Pilot Study Shows Protection against Simian Immunodeficiency Virus SIVmac251 Challenge in Macaques. .2009. J Virol 83. [CrossRef]
- Deichmann A, Schmidt M. Biosafety considerations using gamma-retroviral vectors in gene therapy. Curr Gene Ther. 2013 Dec;13(6):469-77. [CrossRef]
- U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institutes of Health. Biosafety in Microbiological and Biomedical Laboratories. 6th Edition. https://www.cdc.gov/labs/pdf/SF__19_308133-A_BMBL6_00-BOOK-WEB-final-3.pdf (Accessed: 8 August 2025).
- Cherkassky L, Morello A, Villena-Vargas J, Feng Y, Dimitrov DS, Jones DR, Sadelain M, Adusumilli PS. Human CAR T cells with cell-intrinsic PD-1 checkpoint blockade resist tumor-mediated inhibition. J. Clin. Investig. 2016, 126, 3130–3144. [Google Scholar] [CrossRef] [PubMed] [Green Version].
- Abu Dhabi Department of Health (DoH). DOH/RIC/ST/LAGRSP/V1/2023. Standard for Laboratory Accreditation for Genomic-Related Services and Products. 2023. https://www.doh.gov.ae/en/resources/standards (Accessed: 8 August 2025).
- Abu Dhabi Department of Health (DoH). DOH/GDL/RCT/V1/2024. Guidelines for Clinical & Translational Research in Genomics. Feb. 2024. https://www.doh.gov.ae/en/resources/guidelines (Accessed: 8 August 2025).
- Occupational Safety and Health Administration. Model Plans and Programs for the OSHA Bloodborne Pathogens and Hazard Communications Standards. https://www.osha.gov/sites/default/files/publications/osha3186.pdf.
- Environmental Health & Engineering. Guide: BSL-2+: A Guide to Safe Implementation in the Research Environment. https://eheinc.com/wp-content/uploads/2023/11/BSL2-Research-Env-Guide_3rd-Edition.pdf.
- Duane EG. A practical guide to implementing a BSL-2+ biosafety program in a research laboratory. Applied Biosafety 2013;18:30–6. www.liebertpub.com (Accessed 8 August 2025).
- Frank AM, Braun AH, Scheib L, Agarwal S, Schneider IC, Fusil F, Perian S, Sahin U, Thalheimer FB, Verhoeyen E, Buchholz CJ. Combining T-cell-specific activation and in vivo gene delivery through CD3-targeted lentiviral vectors. Blood Adv. 2020 Nov 24;4(22):5702-5715.
- Ahmed, S.O., El Fakih, R., Kharfan-Dabaja, M.A., Syed, F., Mufti, G., Chabannon, C., Rondelli, D., Mohty, M., Al Ahmari, A.A., Gauthier, J. and Ruella, M., 2025. Setting up a Chimeric Antigen Receptor T Cell Therapy Program: A Framework for Delivery from the Worldwide Network for Blood & Marrow Transplantation. Transplantation and Cellular Therapy, Official Publication of the American Society for Transplantation and Cellular Therapy.
- World Health Organization. Guidelines for HIV post-exposure prophylaxis [Internet]. Geneva: World Health Organization; 2024. Providing PEP. Available from: https://www.ncbi.nlm.nih.gov/books/NBK606993/.
- US Department of Health and Human Services. National Institutes of Health. NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules. April 2024. (NIH Guidelines) (accessed August 8, 2025).
- Barrett DM, Zhao Y, Liu X, Jiang S, Carpenito C, Kalos M, Carroll RG, June CH, Grupp SA. Treatment of advanced leukemia in mice with mRNA engineered T cells. Hum Gene Ther. 2011 Dec;22(12):1575-86. [CrossRef]
- Balke-Want H, Keerthi V, Cadinanos-Garai A, Fowler C, Gkitsas N, Brown AK, Tunuguntla R, Abou-El-Enein M, Feldman SA. Non-viral chimeric antigen receptor (CAR) T cells going viral. Immunooncol Technol. 2023 Mar 9;18:100375. [CrossRef]
- Moretti A, Ponzo M, Nicolette CA, Tcherepanova IY, Biondi A, Magnani CF. The Past, Present, and Future of Non-Viral CAR T Cells. Front Immunol. 2022 Jun 9;13:867013. [CrossRef]
- Rafiq S, Hackett CS, Brentjens RJ. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol. 2020 Mar;17(3):147-167. [CrossRef]
- Michels A, Ho N, Buchholz CJ. Precision medicine: In vivo CAR therapy as a showcase for receptor-targeted vector platforms. Mol Ther. 2022 Jul 6;30(7):2401-2415. [CrossRef]
- Chen Z, Hu Y, Mei H. Advances in CAR-Engineered Immune Cell Generation: Engineering Approaches and Sourcing Strategies. Adv Sci (Weinh). 2023 Dec;10(35):e2303215. [CrossRef]
| Item | Drug name | Targeted gene | Delivery Strategy | Targeted malignancy | Country | Data source |
|---|---|---|---|---|---|---|
| 1 |
KymriahTM (tisagenlecleucel) (Approved by FDA in 2017) |
CD19 | Lentiviral |
|
USA | [6,7,8,23,24,25] |
| 2 |
YescartaTM (axicabtagene ciloleucel) (Approved by FDA in 2017) |
CD19 | ɣ-retroviral |
|
USA China |
[9,10,26,27,28,29] |
| 3 |
TecartusTM (brexucabtagene autoleucel) (Approved by FDA in 2020) |
CD19 |
|
USA | [11,12,30,31,32] | |
| 4 |
Abecma® (idecabtagene vicleucel) (Approved by FDA in 2021) |
3BCMA | Lentiviral vector |
|
USA | [13,14,33,34] |
| 5 |
Breyanzi® (lisocabtagene maraleucel) Approved by the FDA in 2021) |
CD19 | Lentiviral vector |
|
USA | [15,16,35,36,37] |
| 6 |
CarvyktiTM (ciltacabtagene autoleucel) (Approved by FDA in 2022) |
3BCMA | Lentiviral vector |
|
USA China |
[17,18,38,39,40,41] |
| 7 |
Fucaso® (equecabtagene autoleucel) (Approved by China NMPA in 2023) |
BCMA | Lentiviral vector |
|
China | [19] |
| 8 |
Carteyva® (relmacabtagene autoleucel) (Approved by China NMPA in 2024) |
CD19 |
|
China | [20] |
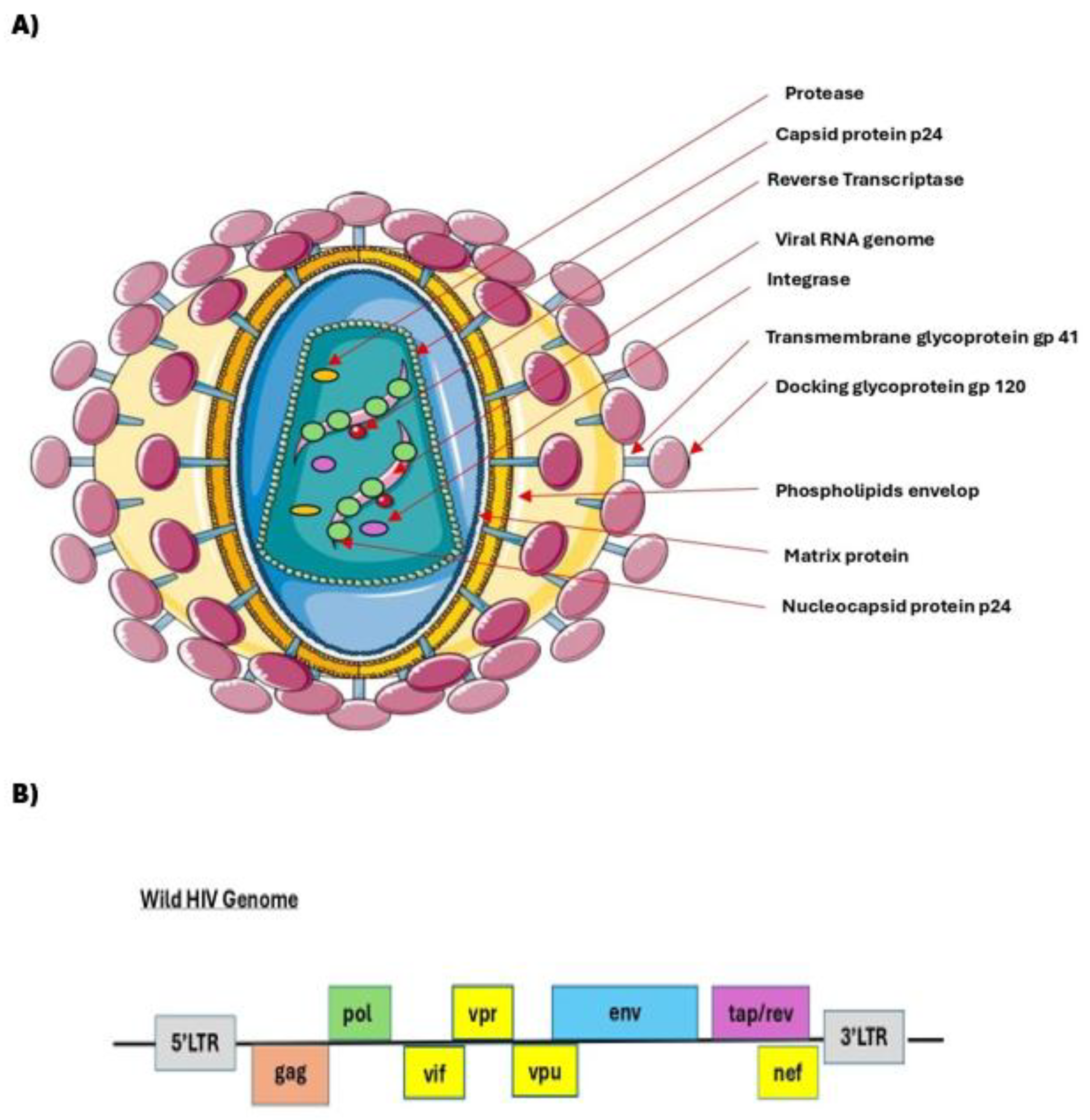
| Component | Gene | Proteins Encoded | Functions in Wild Type HIV | Role in Recombinant Vector System | Abbreviation |
|---|---|---|---|---|---|
| Structural gene | gag | MA (matrix), CA (capsid), NC (nucleocapsid), p6 | Structural core proteins: form the viral capsid and are essential for particle assembly and packaging. | Provided “in trans” by the “packaging plasmid”. Forms the viral core that encapsulates the vector RNA | gag |
| Enzymatic gene | pol | RT (reverse transcriptase), IN (integrase), PR (protease) | Reverse transcription, integration, polyprotein cleavage | Provided “in trans” by the “packaging plasmid”. Essential for creating an infectious vector particle | pol |
| Envelope gene | env | gp120, gp41 (envelope glycoproteins) | Mediates viral entry by binding to CD4 and CCR5/CXCR4 co-receptors on target cells | Deleted from the vector system. Replaced “in trans” by a heterologous envelope plasmid, most commonly “VSV-G”, which confers broad tropism | env |
| Vesicular Stomatitis Virus protein G | To support an extremely broad range of tropism | VSV-G | |||
| Regulatory element | RRE (in env RNA) | – (RNA motif, not protein) | Binding site for the Rev protein. Allows unspliced and partially spliced viral RNAs to be exported from the nucleus to the cytoplasm | Retained in the vector to enable nuclear export of the full-length vector RNA (especially important for genomes >4kb) | RRE |
| Regulatory gene |
rev | Rev protein | RNA-binding protein that binds to RRE to shuttle unspliced RNAs from the nucleus to the cytoplasm | Provided “in trans” on a “separate plasmid” (in 3rd/4th gen systems) or with packaging genes. Essential for high-titer production of vectors with RRE. | rev |
| tat | Tat protein | A key transcriptional activator that binds the TAR element to dramatically enhance transcription from the LTR. | Deleted. LTR in packaging plasmid is often replaced by a strong constitutive promoter (e.g., CMV). | tat | |
| Accessory gene |
nef | Nef protein | Immune evasion downmodulates CD4 and MHC-I, enhancing infectivity. | Deleted | nef |
| vif | Vif protein | Blocks APOBEC3G (antiviral restriction factor) | Deleted | vif | |
| vpr | Vpr protein | Cell cycle arrest, nuclear import of pre-integration complex | Deleted | vpr | |
| vpu | Vpu protein | CD4 degradation, virus release enhancement | Deleted | vpu | |
| LTR (5′ and 3′) |
U3 | – (cis-DNA/RNA element) | Promoter/enhancer region (drives transcription) | In “SIN vectors”, its U3 region is often deleted to be replaced by a specific promoter. | U3 |
| R | – | Repeat: transcription initiation and polyadenylation site | It drives transcription of the vector RNA. | R | |
| U5 | – | Needed for reverse transcription and integration | U5 | ||
| LTR (3′) | PolyA (in 3′ LTR) | – | Signals proper 3′ end processing and mRNA stability | In “SIN vectors”, the U3 region is “deleted”. This deletion is copied to the 5’ LTR during reverse transcription, “inactivating the viral promoter” in the provirus | PolyA |
| Generation | Key Genomic Feature | Plasmid System | Safety Profile | Primary Use |
|---|---|---|---|---|
| 1st. | Full HIV-1 genome minus env in packaging; accessory genes present (vif, vpr, vpu, nef). Tat-dependent LTR; non-SIN; high homology to wild-type. [21] | 3-Plasmid: Transfer + Packaging + Envelope. | Low | Basic research; historical. |
| 2nd. | Accessory genes deleted (Δvif, Δvpr, Δvpu, Δnef); often still non-SIN; tat + rev retained in packaging. | 3-Plasmid: Transfer + gag/pol + tat + rev (packaging) + Envelope. | Medium | In-vitro cell modification; preclinical. |
| 3rd. | rev moved to a separate plasmid; SIN LTRs (ΔU3) standard; heterologous 5′ promoter; cPPT/CTS and WPRE commonly used. | 4-Plasmid: Transfer (SIN) + gag/pol + rev + Envelope. | High | Gold standard for clinical applications (CAR-T, HSC). |
| 4th. | Further HIV sequence minimization; tat-independent with heterologous promoters; possible insulators/extra safety modules; may split gag from pol. | ≥4-Plasmid (extends 3rd-gen split; optional extra splits). | Very High | Cutting-edge trials/platforms where maximal safety is required. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




