Submitted:
09 September 2025
Posted:
11 September 2025
You are already at the latest version
Abstract
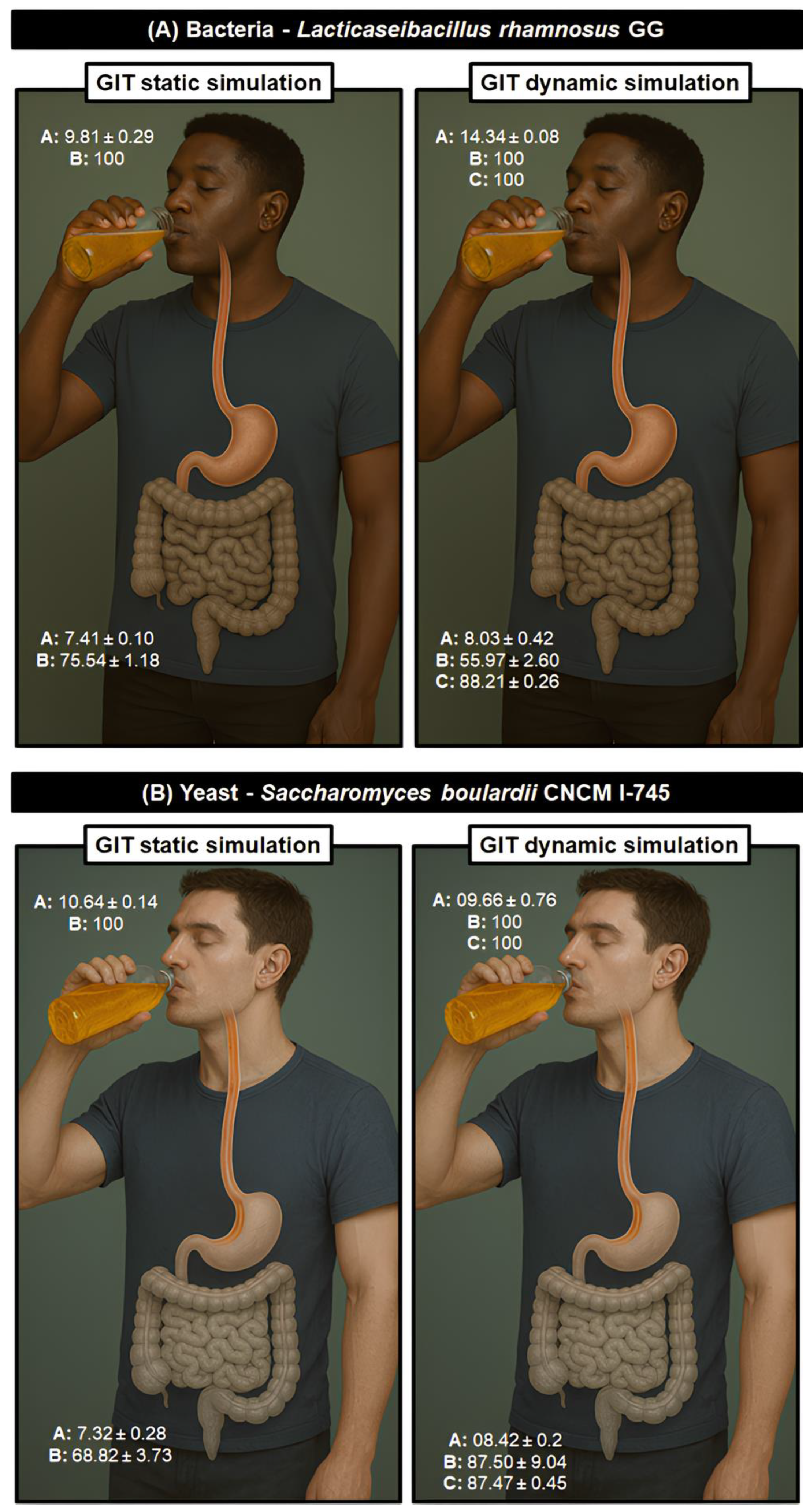
This study explored the prebiotic potential of africanized bee honeys from the Caatinga biome on the survival of Lacticaseibacillus rhamnosus GG in fermented beverages under refrigerated storage and gastrointestinal tract (GIT) simulation. Among the samples tested, honey derived predominantly from Myracrodruon urundeuva (aroeira-do-campo) exhibited the most pronounced protective effect. After 28 days of refrigeration, this formulation retained a viable bacterial load above 8 Log CFU/mL, compared to a drastic decline to 3 Log CFU/mL in honey analog control. Under static GIT simulation, probiotic survival exceeded 92 % post-gastric phase, with the aroeira-do-campo formulation achieving full retention. Dynamic simulations confirmed these findings, with survival rates up to 88.2 % by flow cytometry, even under complex digestive stress. Additionally, the bee honeys supported lactic acid production and maintained physicochemical integrity, confirming their synbiotic functionality. These results clearly show the potential prebiotic effect of Caatinga honeys, including the differences in their botanical origin as effective carriers of bacterial probiotics, opening the way for stable, dairy-free functional beverages adapted to arid ecosystems. Furthermore, it reinforces the importance of maintaining the sustainability of an exclusive biome and hotspot of biological diversity with a high degree of endemism. In other words, studies like this that reinforce the value of ecosystem services are of fundamental importance for the implementation of public policies in favor of the conservation of these environments.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Origin of Bee Honeys
2.2. Physicochemical and Microbiological Parameters of Bee Honey
2.3. Probiotic Bacteria and Cultivation Conditions
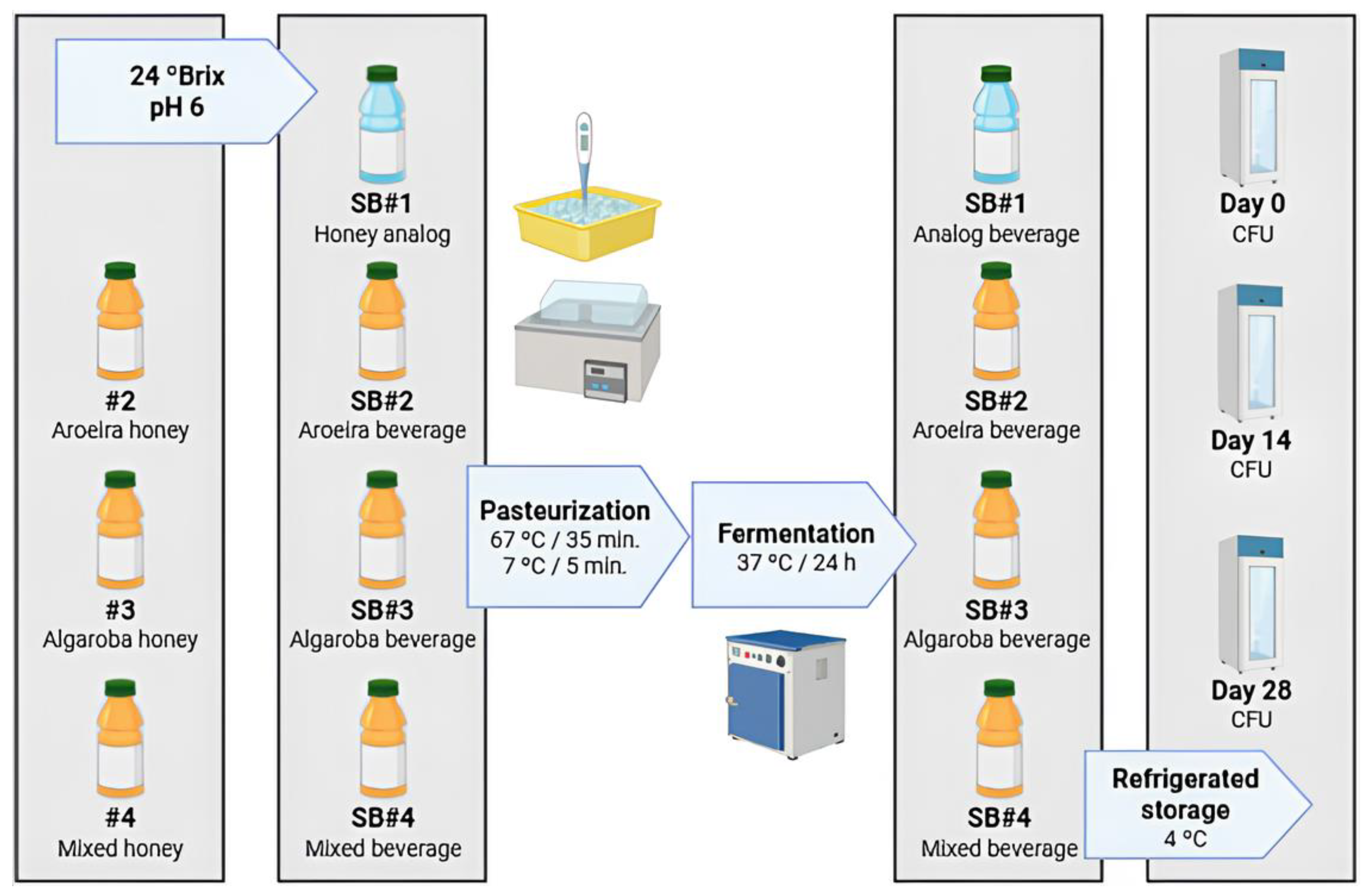
2.4. Experimental Design of the Formulated Bee Honey Beverages
2.5. Measuring the Metabolic Activity of the Probiotic
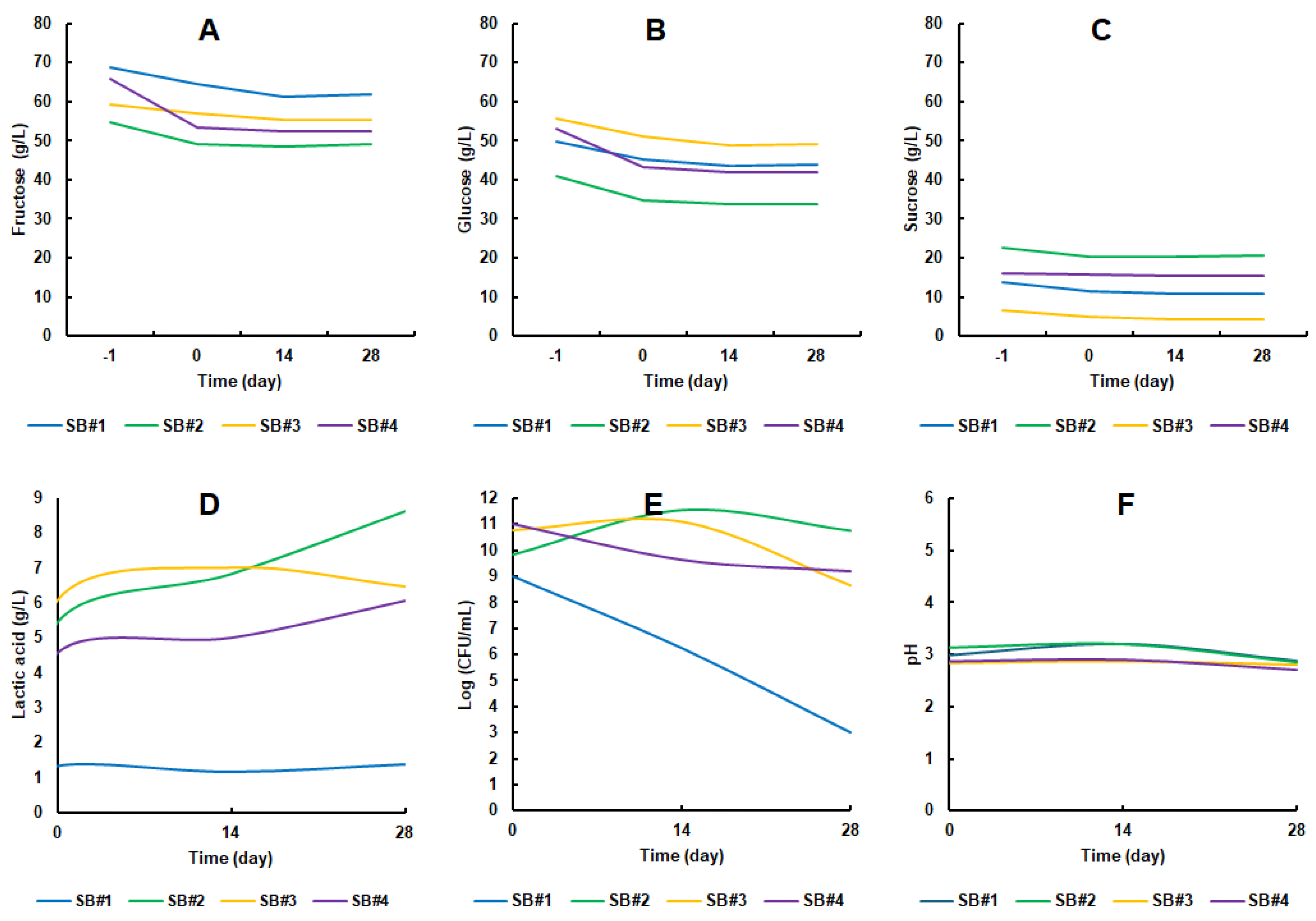
2.5.1. Carbohydrates (Sucrose, Fructose and Glucose) and Lactic Acid
2.5.2. Phenylethyl Alcohol Ester
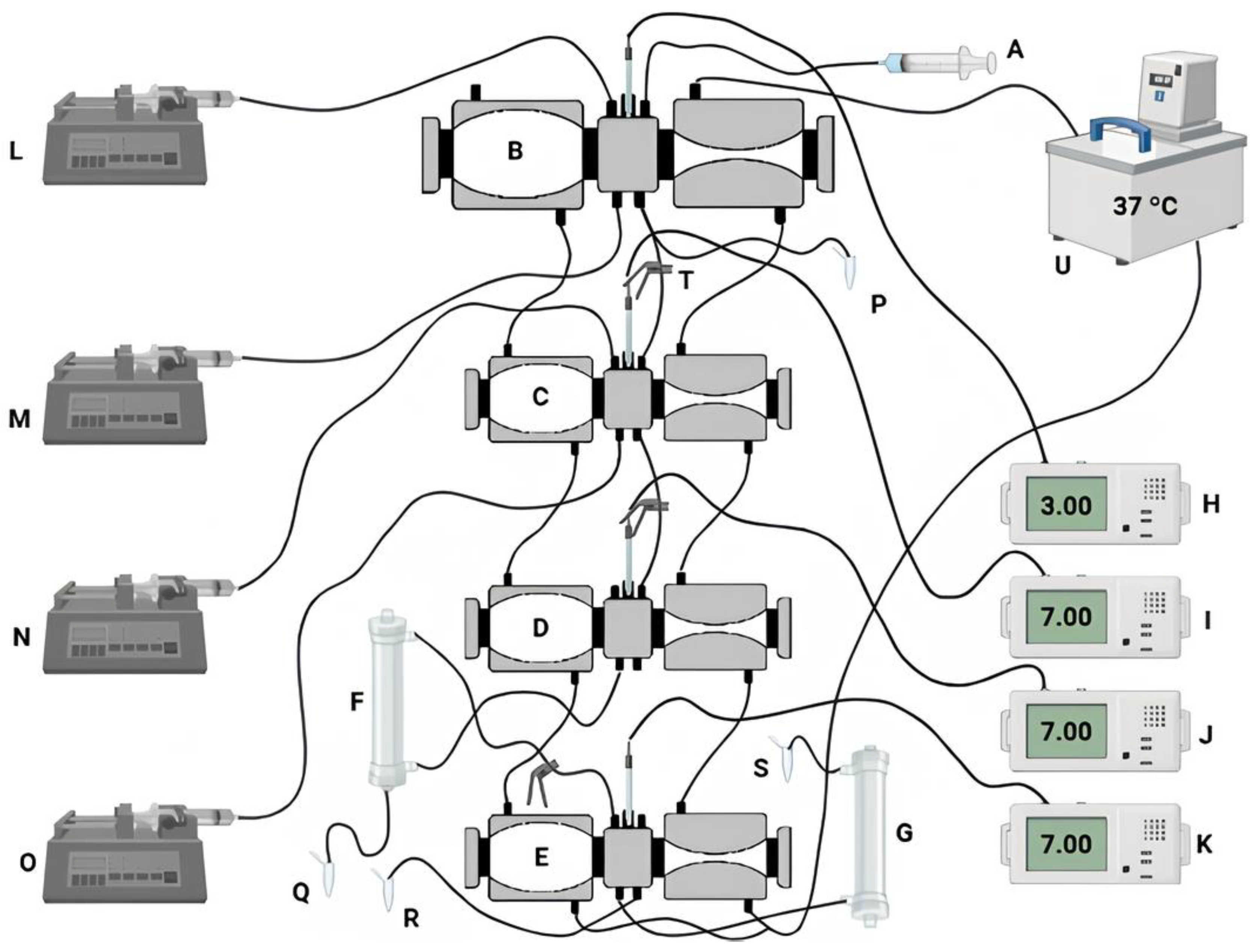
2.6. In Vitro Simulation Tests of the GIT
2.6.1. Static Simulation of the GIT Under Refrigerated Storage
2.6.2. Dynamic Simulation of the GIT
2.7. Measuring the Viability and Survival of the Probiotic
2.8. Measuring Probiotic Cell Membrane Integrity
2.9. In vitro Evaluation of Anti-Inflammatory Activity
2.10. Statistical Analysis
3. Results and Discussion
3.1. Physico-Chemical and Microbiological Characteristics of Bee Honeys
| Parameters | Aroeira-do-campo | Algaroba | Mixed |
|---|---|---|---|
| Colour (Pfund) | Light Ambar | Dark amber | Dark amber |
| Water activity (Aw) | 0,61 ± 0.00 | 0,56 ± 0.02 | 0,59 ± 0.01 |
| Humidity (%) | 14.01 ± 0.80 | 19.56 ± 0.52 | 19.71 ± 0.15 |
| Soluble solids (°Brix) | 85.00 ± 0.00 | 83.00 ± 0.00 | 81.50 ± 0.00 |
| Density (g/cm3) | 1.42 ± 0.06 | 1.42 ± 0.00 | 1.42 ± 0.00 |
| pH | 3.92 ± 0.05 | 3.61 ± 0.07 | 3.78 ± 0.00 |
| Free acidity (mEq/kg of honey) | 56.00 ± 0.01 | 34.50 ± 0.10 | 22.50 ± 0.06 |
| Lactonic acidity (mEq/kg of honey) | 11.00 ± 0.02 | 08.50 ± 0.00 | 25.00 ± 0.20 |
| Total acidity (mEq/kg of honey) | 67.00 ± 0.04 | 43.00 ± 0.02 | 47.50 ± 0.00 |
| Electric conductivity (µS/cm) | 436.25 ± 2.06 | 997.50 ± 0.71 | 270.50 ± 0.71 |
| Ashes (%) | 0.21 ± 0.02 | 0.57 ± 0.02 | 0.09 ± 0.01 |
| Firmness (g) | 57.74 ± 2.75 | 51,97 ± 0.13 | 66.95 ± 1.42 |
| Consistency (g/sec) | 646.61 ± 27.09 | 585.50 ± 3.18 | 753.78 ± 17.15 |
| Cohesiveness (g) | -29.52 ± 2.98 | -26.34 ± 0.59 | -40.50 ± 2.28 |
| Work of cohesion (g/sec) | -238.99 ± 25.55 | -204.36 ± 7.19 | -396.23 ± 17.79 |
| Viscosity (Pa/s) | 14.67 ± 0.37 | 18.46 ± 0.64 | 17.66 ± 0.50 |
| Hydroxymethyl furfural (mg/kg of honey) | 3.04 ± 0.09 | 9.45 ± 0.01 | 8.28 ± 0.11 |
| Diastase activity (Gothe units/g of honey) | 19.17 ± 0.73 | 4.10 ± 0.40 | 3.50 ± 0.20 |
| Glucose (g/100 g of honey) | 26.32 ± 0.23 | 29.83 ± 0.75 | 31.54 ± 0.16 |
| Fructose (g/100 g of honey) | 41.42 ± 0.56 | 32.41 ± 1.04 | 32.92 ± 0.65 |
| Apparent sucrose (g/100 g of honey) | 9.48 ± 0.68 | 9.82 ± 0.33 | 10.33 ± 0.11 |
| Total sugars (g/100 g of honey) | 85.90 ± 0.51 | 72.05 ± 1.45 | 74.79 ± 0.69 |
| Total proteins (g/100 g of honey) | 0.29 ± 0.03 | 0.22 ± 0.01 | 0.22 ± 0.02 |
| Antioxidant activity (FRAP method) (µM FeSO4/mL) | 403.92 ± 0.00 | 145.43 ± 1.62 | 301.38 ± 1.21 |
| Antioxidant activity (DPPH method) (%) | 52.7 ± 3.05 | 44.18 ± 2.32 | 39.32 ± 2.27 |
| Flavonoids (mg Rutin/100 g of honey) | 74.3 ± 0.00 | 102.6 ± 0.00 | 69.4 ± 0.00 |
| Flavonols (mg Quercetin/100 g of honey) | 54.3 ± 0.00 | 75.0 ± 0.00 | 59.5 ± 0.00 |
| Total phenolic (mg Tannic Acid/100 g of honey) | 185.72 ± 2.9 | 256.40 ± 0.64 | 191.31 ± 2.16 |
| Lund reaction | Positive | Positive | Positive |
| Lugol reaction | Negative | Negative | Negative |
| Coliforms (CFU/g) | <10 | <10 | <10 |
| Escherichia coli (CFU/g) | <10 | <10 | <10 |
| Salmonella | Negative | Negative | Negative |
3.2. The Metabolism of Lacticaseibacillus Rhamnosus GG During Fermentation and Storage Under Refrigeration
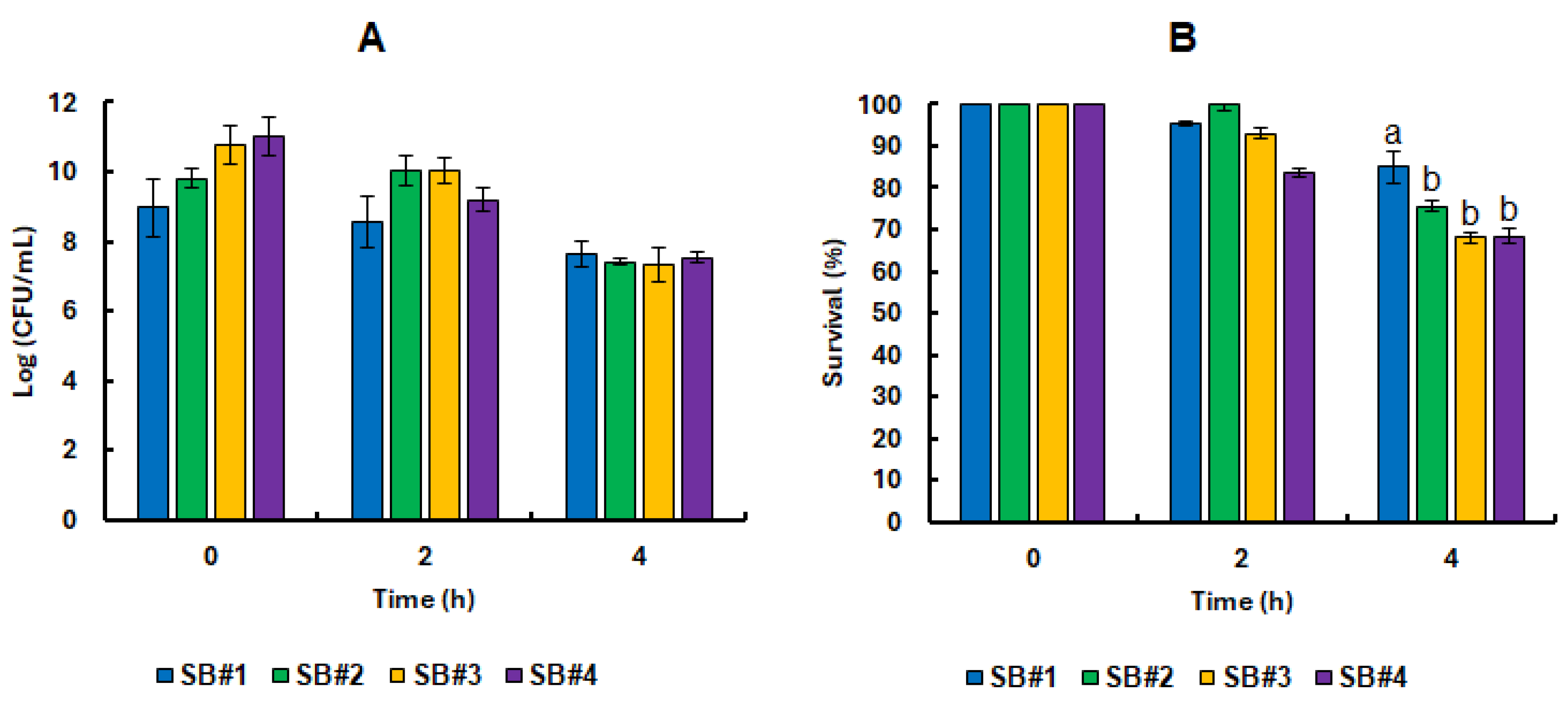
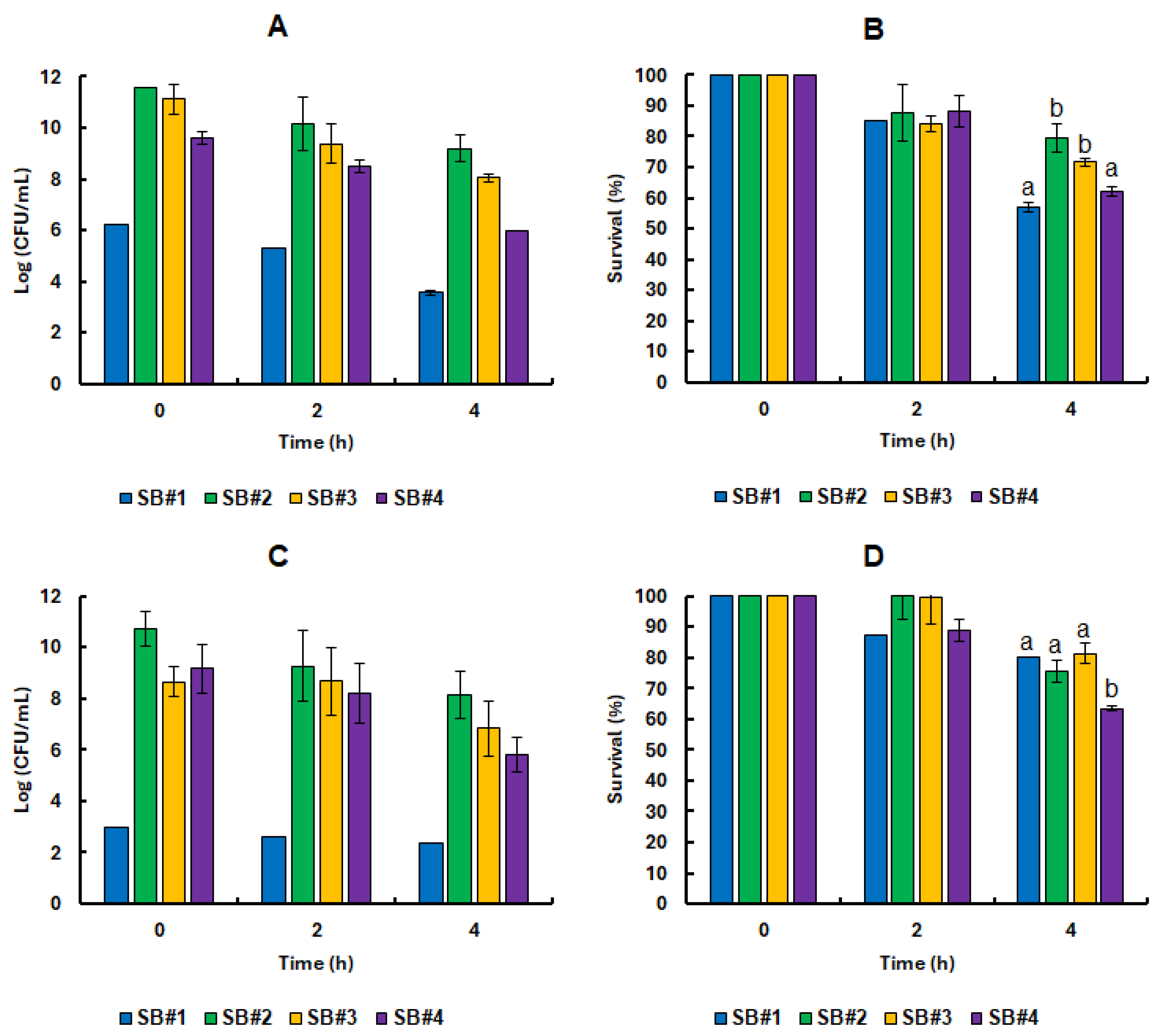
3.3. Survival of the Lacticaseibacillus Rhamnosus GG Cell in Synbiotic Beverages Under GIT Static Simulation
3.4. Impact of Bee Honeys from Different Blooms on the Tolerance of Probiotic Bacteria in Refrigerated Stock and Under Static Simulation of the GIT
3.5. Nature’s Shield: The Protective Role of Bee Honey on Lacticaseibacillus Rhamnosus GG Under Lifelike GIT Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO/WHO Guidelines for the Evaluation of Probiotics in Food; London, UK, 2002.
- Song, H.; Zhang, J.; Qu, J.; Liu, J.; Yin, P.; Zhang, G.; Shang, D. Lactobacillus Rhamnosus GG Microcapsules Inhibit Escherichia Coli Biofilm Formation in Coculture. Biotechnol Lett 2019, 41, 1007–1014. [Google Scholar] [CrossRef]
- Lopes, L.A.A.; Carvalho, R. de S.F.; Magalhães, N.S.S.; Madruga, M.S.; Athayde, A.J.A.A.; Araújo Portela, I.; Eduardo Barão, C.; Colombo Pimentel, T.; Magnani, M.; Christina Montenegro Stamford, T. Microencapsulation of Lactobacillus Acidophilus La-05 and Incorporation in Vegan Milks: Physicochemical Characteristics and Survival during Storage, Exposure to Stress Conditions, and Simulated Gastrointestinal Digestion. Food Research International 2020, 135, 109295. [Google Scholar] [CrossRef]
- Capurso, L. Thirty Years of Lactobacillus Rhamnosus GG: A Review. Journal of Clinical Gastroenterology 2019, 53, S1. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert Consensus Document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Prebiotics. Nat Rev Gastroenterol Hepatol 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Schell, K.R.; Fernandes, K.E.; Shanahan, E.; Wilson, I.; Blair, S.E.; Carter, D.A.; Cokcetin, N.N. The Potential of Honey as a Prebiotic Food to Re-Engineer the Gut Microbiome Toward a Healthy State. Front. Nutr. 2022, 9. [Google Scholar] [CrossRef]
- Pinto-Neto, W. de P.; Silva, R.K.; Lima, B. de S.; Acioli, G.F. de S.; Paixão, G.A. da; Muniz, B.C.; Silva, P.K.N. da; Costa, R.M.P.B.; Silva, F.S.B. da; Melo, H.F. de; et al. Bee Honey of the Pajeú Hinterland, Pernambuco, Brazil: Physicochemical Characterization and Biological Activity. Food Bioscience 2024, 60, 104289. [Google Scholar] [CrossRef]
- FAO/WHO Standard for Honey CXS 12-1981: Codex Alimentarius: International Food Standards; 2019.
- Miguel, M.; Antunes, M.; Faleiro, M. Honey as a Complementary Medicine. Integr Med�Insights 2017, 12, 1178633717702869. [Google Scholar] [CrossRef]
- Leal, I.R.; Silva, J.M.C. da; Tabarelli, M.; Lacher Jr., T.E. Changing the Course of Biodiversity Conservation in the Caatinga of Northeastern Brazil. 2005, 19.
- Araújo, T.A. de S.; Alencar, N.L.; de Amorim, E.L.C.; de Albuquerque, U.P. A New Approach to Study Medicinal Plants with Tannins and Flavonoids Contents from the Local Knowledge. Journal of Ethnopharmacology 2008, 120, 72–80. [Google Scholar] [CrossRef]
- Falcão, M.P.M.M.; Oliveira, T.K.B.; Ó, N.P.R. do; Sarmento, D.A.; Gadelha, N.C. Schinus terebinthifolius Raddi (Aroeira) e suas propriedades na Medicina Popular. Revista Verde de Agroecologia e Desenvolvimento Sustentável 2015, 10, 23–27. [Google Scholar] [CrossRef]
- Oliveira, P.P.; van den Berg, C.; Santos, F.D.A.R.D. Pollen Analysis of Honeys from Caatinga Vegetation of the State of Bahia, Brazil. Grana 2010, 49, 66–75. [Google Scholar] [CrossRef]
- Domingos, F.R.; Silva, M.A.P. da Uso, conhecimento e conservação de Myracrodruon urundeuva: uma revisão sistemática. Research, Society and Development 2020, 9, e2329118851–e2329118851. [Google Scholar] [CrossRef]
- Diógenes, É.S.G.; Silva, A.L.C. da; Neto, F.C. das C.; Silveira, E.R.; Leal, L.K.A.M.; Nicolete, R.; Araújo, T.G. de Evaluation of the Skin Whitening and Antioxidant Activity of Myracrodruon urundeuva Extract (Aroeira-Do-Sertão). Nat Prod Res 2024, 38, 3663–3668. [Google Scholar] [CrossRef] [PubMed]
- de Lemos, A.B.S.; Chaves, G.; Ribeiro, P.P.C.; da Silva Chaves Damasceno, K.S.F. Prosopis Juliflora: Nutritional Value, Bioactive Activity, and Potential Application in Human Nutrition. Journal of the Science of Food and Agriculture 2023, 103, 5659–5666. [Google Scholar] [CrossRef] [PubMed]
- González Galán, A.; Corrêa, A.D.; Patto de Abreu, C. maria; Piccolo Barcelos, M. de F. Caracterización química de la harina del fruto de Prosopis spp. procedente de Bolivia y Brasil. Archivos Latinoamericanos de Nutrición 2008, 58, 309–315. [Google Scholar]
- Nogueira, F. de C.; Pagotto, M.A.; Aragão, J.R.V.; Roig, F.A.; Ribeiro, A. de S.; Lisi, C.S. The Hydrological Performance of Prosopis Juliflora (Sw.) Growth as an Invasive Alien Tree Species in the Semiarid Tropics of Northeastern Brazil. Biol Invasions 2019, 21, 2561–2575. [Google Scholar] [CrossRef]
- Pinto-Neto, W. de P.; Loureiro, L.; Gonçalves, R.F.S.; Marques, M.C.T.; Rodrigues, R.M.M.; Abrunhosa, L.; de Barros, A.M.; Shinohara, N.K.S.; Pinheiro, A.C.; Vicente, A.A.; et al. Potential Prebiotic Effect of Caatinga Bee Honeys from the Pajeú Hinterland (Pernambuco, Brazil) on Synbiotic Alcoholic Beverages Fermented by Saccharomyces Boulardii CNCM I-745. Fermentation 2025, 11, 405. [Google Scholar] [CrossRef]
- Instituto Adolfo Lutz Métodos Físico-Químicos Para Análise de Alimentos; 1st digital ed.; Instituto Adolfo Lutz: São Paulo, Brazil, 2008.
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of Protein Using Bicinchoninic Acid. Analytical Biochemistry 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Analytical Biochemistry 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Orujei, Y.; Shabani, L.; Sharifi-Tehrani, M. Induction of Glycyrrhizin and Total Phenolic Compound Production in Licorice by Using Arbuscular Mycorrhizal Fungi. Russ J Plant Physiol 2013, 60, 855–860. [Google Scholar] [CrossRef]
- Woisky, R.G.; and Salatino, A. Analysis of Propolis: Some Parameters and Procedures for Chemical Quality Control. Journal of Apicultural Research 1998, 37, 99–105. [Google Scholar] [CrossRef]
- Bencherif, K.; Djaballah, Z.; Brahimi, F.; Boutekrabt, A.; Dalpè, Y.; Lounès-Hadj Sahraoui, A. Arbuscular Mycorrhizal Fungi Affect Total Phenolic Content and Antimicrobial Activity of Tamarix Gallica in Natural Semi-Arid Algerian Areas. South African Journal of Botany 2019, 125, 39–45. [Google Scholar] [CrossRef]
- AOAC International Official Methods of Analysis of AOAC International; 17th ed.; AOAC International: Gaithersburg, MD, USA, 2002.
- AOAC International Official Methods of Analysis of AOAC International; 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005.
- Vidal, E.E.; de Billerbeck, G.M.; Simões, D.A.; Schuler, A.; François, J.M.; de Morais, M.A. Influence of Nitrogen Supply on the Production of Higher Alcohols/Esters and Expression of Flavour-Related Genes in Cachaça Fermentation. Food Chemistry 2013, 138, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Vidal, E.E.; de Morais Jr, M.A.; François, J.M.; de Billerbeck, G.M. Biosynthesis of Higher Alcohol Flavour Compounds by the Yeast Saccharomyces cerevisiae: Impact of Oxygen Availability and Responses to Glucose Pulse in Minimal Growth Medium with Leucine as Sole Nitrogen Source. Yeast 2015, 32, 47–56. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST Static in Vitro Simulation of Gastrointestinal Food Digestion. Nat Protoc 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A Standardised Static in Vitro Digestion Method Suitable for Food – an International Consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef]
- Mulet-Cabero, A.-I.; Egger, L.; Portmann, R.; Ménard, O.; Marze, S.; Minekus, M.; Feunteun, S.L.; Sarkar, A.; Grundy, M.M.-L.; Carrière, F.; et al. A Standardised Semi-Dynamic in Vitro Digestion Method Suitable for Food – an International Consensus. Food Funct. 2020, 11, 1702–1720. [Google Scholar] [CrossRef]
- Pinheiro, A.C.; Coimbra, M.A.; Vicente, A.A. In Vitro Behaviour of Curcumin Nanoemulsions Stabilized by Biopolymer Emulsifiers – Effect of Interfacial Composition. Food Hydrocolloids 2016, 52, 460–467. [Google Scholar] [CrossRef]
- Pinheiro, A.C.; Gonçalves, R.F.; Madalena, D.A.; Vicente, A.A. Towards the Understanding of the Behavior of Bio-Based Nanostructures during in Vitro Digestion. Current Opinion in Food Science 2017, 15, 79–86. [Google Scholar] [CrossRef]
- Fernandes, J.-M.; Madalena, D.A.; Vicente, A.A.; Pinheiro, A.C. Influence of the Addition of Different Ingredients on the Bioaccessibility of Glucose Released from Rice during Dynamic in Vitro Gastrointestinal Digestion. Int J Food Sci Nutr 2021, 72, 45–56. [Google Scholar] [CrossRef]
- Günerken, E.; D’Hondt, E.; Eppink, M.; Elst, K.; Wijffels, R. Flow Cytometry to Estimate the Cell Disruption Yield and Biomass Release of Chlorella Sp. during Bead Milling. Algal Research 2017, 25, 25–31. [Google Scholar] [CrossRef]
- Loureiro, L.; Machado, L.; Geada, P.; Vasconcelos, V.; Vicente, A.A. Evaluation of Efficiency of Disruption Methods for Coelastrella Sp. in Order to Obtain High Yields of Biochemical Compounds Release. Algal Research 2023, 73, 103158. [Google Scholar] [CrossRef]
- Osman, N.I.; Sidik, N.J.; Awal, A.; Adam, N.A.M.; Rezali, N.I. In Vitro Xanthine Oxidase and Albumin Denaturation Inhibition Assay of Barringtonia racemosa L. and Total Phenolic Content Analysis for Potential Anti-Inflammatory Use in Gouty Arthritis. J Intercult Ethnopharmacol 2016, 5, 343–349. [Google Scholar] [CrossRef]
- Bakka, C.; Smara, O.; Hadjadj, M.; Dendougui, H.; Mahdjar, S.; Benzid, A. In Vitro Anti-Inflammatory Activity of Pistacia atlantica Desf. Extracts. Asian Journal of Research in Chemistry 2019, 12, 322–325. [Google Scholar] [CrossRef]
- Chakou, F.Z.; Boual, Z.; Hadj, M.D.O.E.; Belkhalfa, H.; Bachari, K.; El Alaoui-Talibi, Z.; El Modafar, C.; Hadjkacem, F.; Fendri, I.; Abdelkafi, S.; et al. Pharmacological Investigations in Traditional Utilization of Alhagi maurorum Medik. in Saharan algeria: In Vitro Study of Anti-Inflammatory and Antihyperglycemic Activities of Water-Soluble Polysaccharides Extracted from the Seeds. Plants 2021, 10, 2658. [Google Scholar] [CrossRef]
- Azeredo, L. da C.; Azeredo, M.A.A.; de Souza, S.R.; Dutra, V.M.L. Protein Contents and Physicochemical Properties in Honey Samples of Apis Mellifera of Different Floral Origins. Food Chemistry 2003, 80, 249–254. [Google Scholar] [CrossRef]
- Kadri, S.M.; Zaluski, R.; Pereira Lima, G.P.; Mazzafera, P.; de Oliveira Orsi, R. Characterization of Coffea arabica Monofloral Honey from Espírito Santo, Brazil. Food Chemistry 2016, 203, 252–257. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, C.; Li, C.; Huang, Z.Y.; Miao, X. Pathway of 5-Hydroxymethyl-2-Furaldehyde Formation in Honey. J Food Sci Technol 2019, 56, 2417–2425. [Google Scholar] [CrossRef]
- European Community (EC) Council Directive 2001/110/EC of 20 December 2001 Relating to Honey; 2002.
- Brasil Regulamento Técnico de Identidade e Qualidade Do Mel; 2000.
- Codex Alimentarius Revised Codex Standard for Honey; 2022.
- Terrab, A.; Recamales, A.F.; Hernanz, D.; Heredia, F.J. Characterisation of Spanish Thyme Honeys by Their Physicochemical Characteristics and Mineral Contents. Food Chemistry 2004, 88, 537–542. [Google Scholar] [CrossRef]
- Tafa, K.D.; Sundramurthy, V.P.; Subramanian, N. Rheological and Thermal Properties of Honey Produced in Algeria and Ethiopia: A Review. International Journal of Food Properties 2021, 24, 1117–1131. [Google Scholar] [CrossRef]
- Alevia, M.; Rasines, S.; Cantero, L.; Sancho, M.T.; Fernández-Muiño, M.A.; Osés, S.M. Chemical Extraction and Gastrointestinal Digestion of Honey: Influence on Its Antioxidant, Antimicrobial and Anti-Inflammatory Activities. Foods 2021, 10, 1412. [Google Scholar] [CrossRef] [PubMed]
- Alugoju, P.; Janardhanshetty, S.S.; Subaramanian, S.; Periyasamy, L.; Dyavaiah, M. Quercetin Protects Yeast Saccharomyces cerevisiae Pep4 Mutant from Oxidative and Apoptotic Stress and Extends Chronological Lifespan. Curr Microbiol 2018, 75, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Mongi, R.J.; Ruhembe, C.C. Sugar Profile and Sensory Properties of Honey from Different Geographical Zones and Botanical Origins in Tanzania. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Silva, J.; Lima, C.; Miranda, R.; Seraglio, S.; Barbosa, E.; Souza, A.; Cardoso, D. Sensorial Quality of Sugarcane Juice with the Addition of Fruits Pulp from the Semi-Arid. Research, Society and Development 2020, 9, 200973745. [Google Scholar] [CrossRef]
- Pinto Neto, W. de P.; Costa de Lucena, T.M.; Alves da Paixão, G.; Shinohara, N.K.S.; Pinheiro, A.C.; Vicente, A.A.; de Souza, R.B.; de Morais Junior, M.A. Symbiotic Honey Beverages: A Matrix Which Tells a Story of Survival and Protection of Human Health from a Gastronomic and Industrial Perspective. International Journal of Gastronomy and Food Science 2025, 40, 101183. [Google Scholar] [CrossRef]
- Mandha, J.; Shumoy, H.; Devaere, J.; Schouteten, J.J.; Gellynck, X.; de Winne, A.; Matemu, A.O.; Raes, K. Effect of Lactic Acid Fermentation of Watermelon Juice on Its Sensory Acceptability and Volatile Compounds. Food Chemistry 2021, 358, 129809. [Google Scholar] [CrossRef]
- Fiorda, F.A.; de Melo Pereira, G.V.; Thomaz-Soccol, V.; Medeiros, A.P.; Rakshit, S.K.; Soccol, C.R. Development of Kefir-Based Probiotic Beverages with DNA Protection and Antioxidant Activities Using Soybean Hydrolyzed Extract, Colostrum and Honey. LWT - Food Science and Technology 2016, 68, 690–697. [Google Scholar] [CrossRef]
- Magalhães, K.T.; de, M. Pereira, G.V.; Dias, D.R.; Schwan, R.F. Microbial Communities and Chemical Changes during Fermentation of Sugary Brazilian Kefir. World J Microbiol Biotechnol 2010, 26, 1241–1250. [Google Scholar] [CrossRef]
- Andrade, R.; Santos, E.; Azoubel, P.; Ribeiro, E. Increased Survival of Lactobacillus Rhamnosus ATCC 7469 in Guava Juices with Simulated Gastrointestinal Conditions during Refrigerated Storage. Food Bioscience 2019, 32, 100470. [Google Scholar] [CrossRef]
- Pereira, M.D.; Romeiro, T.C.O. da S.; Flores, A.V.; Severiano, R.L. Germinação e biometria de frutos e sementes de Prosopis juliflora (Sw) D.C. Ciência Florestal 2018, 28, 1271–1281. [Google Scholar] [CrossRef]
- Mahmoud, M.A.A.; Kılıç-Büyükkurt, Ö.; Aboul Fotouh, M.M.; Selli, S. Aroma Active Compounds of Honey: Analysis with GC-MS, GC-O, and Molecular Sensory Techniques. Journal of Food Composition and Analysis 2024, 134, 106545. [Google Scholar] [CrossRef]
- Camilleri, M. Integrated Upper Gastrointestinal Response to Food Intake. Gastroenterology 2006, 131, 640–658. [Google Scholar] [CrossRef] [PubMed]
- Guan, N.; Liu, L. Microbial Response to Acid Stress: Mechanisms and Applications. Appl Microbiol Biotechnol 2020, 104, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, A.A.; Pinto-Neto, W. de P.; da Paixão, G.A.; Santos, D. da S.; De Morais, M.A.; De Souza, R.B. Journey of the Probiotic Bacteria: Survival of the Fittest. Microorganisms 2023, 11, 95. [Google Scholar] [CrossRef]
- Orij, R.; Brul, S.; Smits, G.J. Intracellular pH Is a Tightly Controlled Signal in Yeast. Biochimica et Biophysica Acta (BBA) - General Subjects 2011, 1810, 933–944. [Google Scholar] [CrossRef]
- Santos, E.; Andrade, R.; Gouveia, E. Utilization of the Pectin and Pulp of the Passion Fruit from Caatinga as Probiotic Food Carriers. Food Bioscience 2017, 20, 56–61. [Google Scholar] [CrossRef]
- Rasika, D.M.; Vidanarachchi, J.K.; Rocha, R.S.; Balthazar, C.F.; Cruz, A.G.; Sant’Ana, A.S.; Ranadheera, C.S. Plant-Based Milk Substitutes as Emerging Probiotic Carriers. Current Opinion in Food Science 2021, 38, 8–20. [Google Scholar] [CrossRef]
- Machado, T.A.D.G.; de Oliveira, M.E.G.; Campos, M.I.F.; de Assis, P.O.A.; de Souza, E.L.; Madruga, M.S.; Pacheco, M.T.B.; Pintado, M.M.E.; Queiroga, R. de C.R. do E. Impact of Honey on Quality Characteristics of Goat Yogurt Containing Probiotic Lactobacillus Acidophilus. LWT 2017, 80, 221–229. [Google Scholar] [CrossRef]
- Caldeira, L.A.; Alves, É.E.; Ribeiro, A. de M.F.; Rocha Júnior, V.R.; Antunes, A.B.; Reis, A.F. dos; Gomes, J. da C.; Carvalho, M.H.R. de; Martinez, R.I.E. Viability of Probiotic Bacteria in Bioyogurt with the Addition of Honey from Jataí and Africanized Bees. Pesq. agropec. bras. 2018, 53, 206–211. [Google Scholar] [CrossRef]
- Melo, F.H.C.; Menezes, F.N.D.D.; de Sousa, J.M.B.; dos Santos Lima, M.; da Silva Campelo Borges, G.; de Souza, E.L.; Magnani, M. Prebiotic Activity of Monofloral Honeys Produced by Stingless Bees in the Semi-Arid Region of Brazilian Northeastern toward Lactobacillus Acidophilus LA-05 and Bifidobacterium Lactis BB-12. Food Research International 2020, 128, 108809. [Google Scholar] [CrossRef]
- Becerril-Sánchez, A.L.; Quintero-Salazar, B.; Dublán-García, O.; Escalona-Buendía, H.B. Phenolic Compounds in Honey and Their Relationship with Antioxidant Activity, Botanical Origin, and Color. Antioxidants 2021, 10, 1700. [Google Scholar] [CrossRef] [PubMed]
- Dżugan, M.; Tomczyk, M.; Sowa, P.; Grabek-Lejko, D. Antioxidant Activity as Biomarker of Honey Variety. Molecules 2018, 23, 2069. [Google Scholar] [CrossRef]
- Dimitriu, L.; Constantinescu-Aruxandei, D.; Preda, D.; Moraru, I.; Băbeanu, N.E.; Oancea, F. The Antioxidant and Prebiotic Activities of Mixtures Honey/Biomimetic NaDES and Polyphenols Show Differences between Honeysuckle and Raspberry Extracts. Antioxidants 2023, 12, 1678. [Google Scholar] [CrossRef]
- Coelho, B. de O.; Fiorda-Mello, F.; de Melo Pereira, G.V.; Thomaz-Soccol, V.; Rakshit, S.K.; de Carvalho, J.C.; Soccol, C.R. In Vitro Probiotic Properties and DNA Protection Activity of Yeast and Lactic Acid Bacteria Isolated from A Honey-Based Kefir Beverage. Foods 2019, 8, 485. [CrossRef]
- Chiron, C.; Tompkins, T.A.; Burguière, P. Flow Cytometry: A Versatile Technology for Specific Quantification and Viability Assessment of Micro-Organisms in Multistrain Probiotic Products. J Appl Microbiol 2018, 124, 572–584. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




