Submitted:
05 September 2025
Posted:
05 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Participants
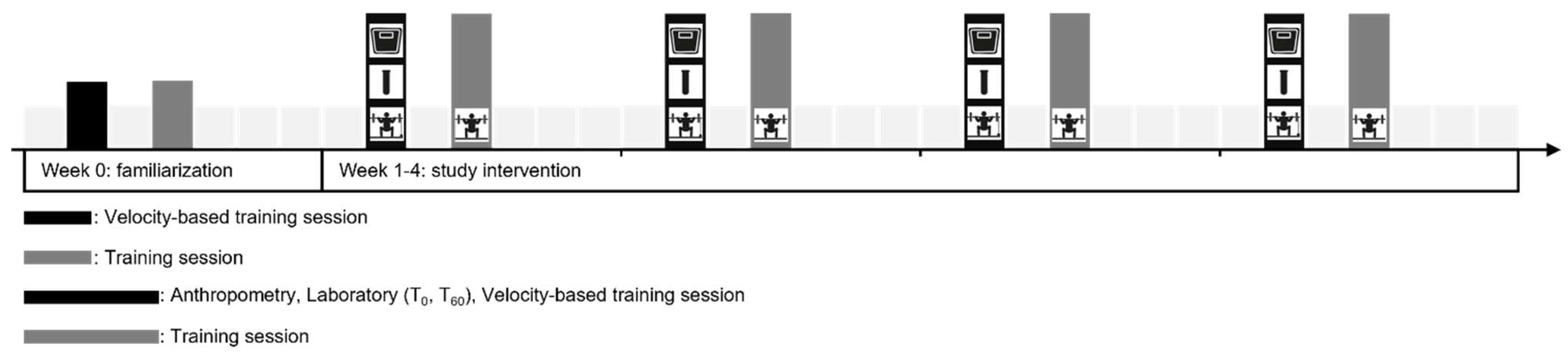
2.2. Study Design
2.3. Measurements
2.3.1. Testing Procedures
2.3.2. Anthropometric Measurements
2.3.3. Estimated One Repetition Maximum and Intra-Set Velocity Loss of the Back Squat Exercise
2.3.4. Blood Samples and Biochemical Analyses
2.4. Menstrual Cycle Phase Determination
2.5. Resistance Training
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Hormonal Responses to Resistance Training
3.2.1. Acute Post-Exercise Alteration in the Composite Androgen Profile
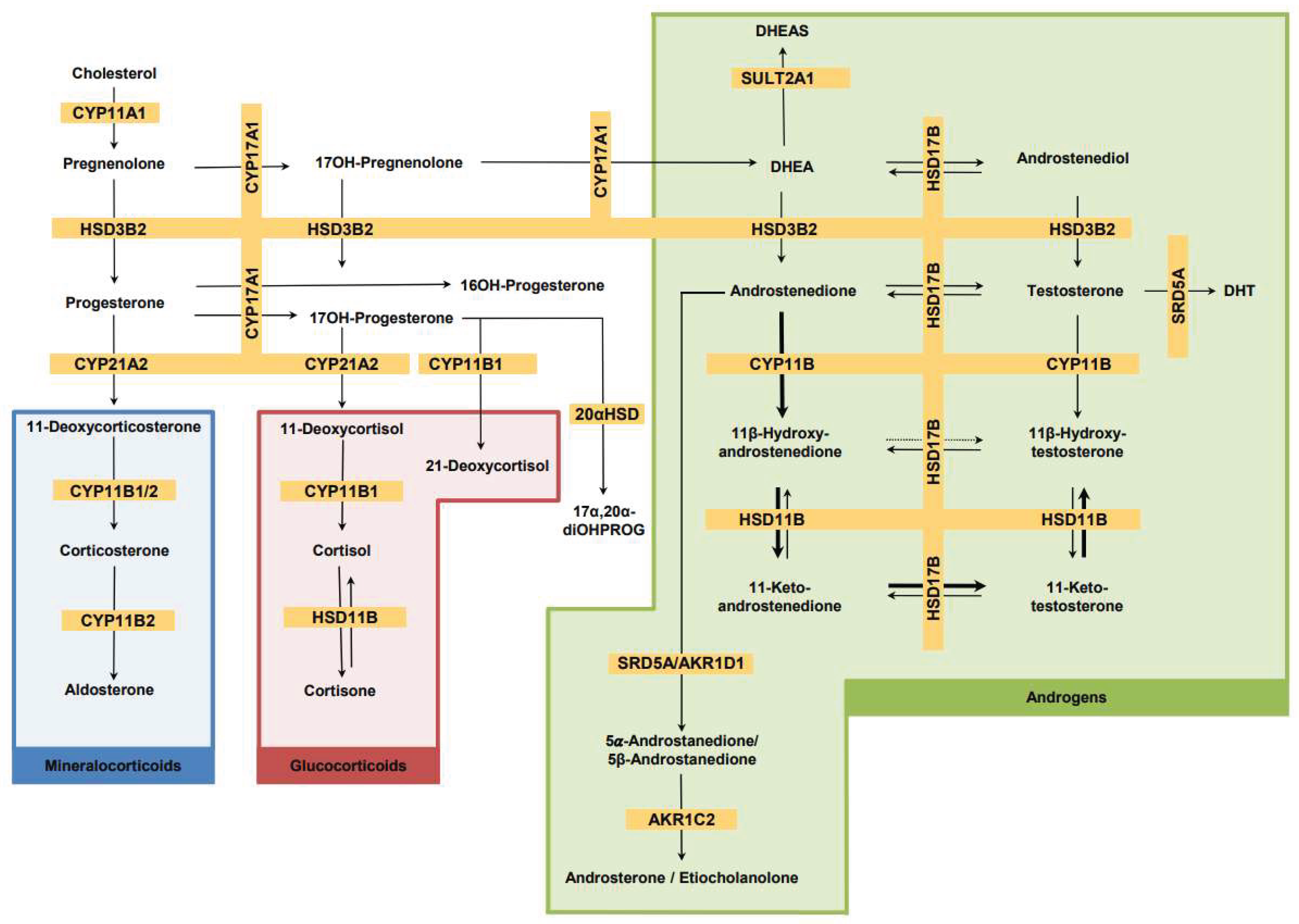
3.2.2. Acute Post-Exercise Alteration in Adrenal-Derived Androgens
3.2.3. Acute Post-Exercise Alteration of Androgen Metabolites Depending on the Androgen Biosynthesis Pathway
3.2.4. Acute Post-Exercise Alteration in Adrenal 11-Oxygenated Adrenal Steroids
3.2.5. Acute Post-Exercise Alteration in Glucocorticoids and Mineralocorticoids
| Metabolite |
Androgens (exclusively adrenal androgens marked by xx) |
Miner- alcorti-coids |
Gluco- corticoids |
Progestins, unclassi-fiable steroids |
Classical pathway |
11-oxy pathway |
11-oxy- pathway with their direct precursors |
Backdoor pathway |
| 11-DOC | x | |||||||
| S | x | |||||||
| 11KA4 | xx | x | x | |||||
| 11KT | xx | x | x | |||||
| 11OHA4 | xx | x | x | |||||
| 11OHT | xx | x | x | |||||
| 16αOHP4 | x | |||||||
| 17α20α-diOHP4 | x | |||||||
| 17OHP4 | x | x | ||||||
| 21DF | x | |||||||
| DHT | x | x | x | |||||
| ALDO | x | |||||||
| A4 | x | x | x | |||||
| AST | x | x | x | |||||
| E | x | |||||||
| CORT | x | |||||||
| F | x | |||||||
| DHEA | x | x | ||||||
| DHEAS | xx | |||||||
| ETIO | x | x | ||||||
| P5 | x | |||||||
| P4 | x | x | ||||||
| T | x | x | x |
3.2.6. Alterations in Additional Hormonal Related Parameters
3.3. Velocity-Based Training Measures
4. Discussion
4.1. Post-Exercise Change in Androgens
4.2. Post-Exercise Change in Glucocorticoids
4.3. Post-Exercise Change in Mineralocorticoids
4.4. Mechanism of Hormonal Change
4.5. Intra-Set Velocity Measures of the Back Squat Exercise During Menstrual Cycle
4.6. Methodological Considerations
4.7. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Makaruk, H.; Starzak, M.; Tarkowski, P.; Sadowski, J.; Winchester, J. The Effects of Resistance Training on Sport-Specific Performance of Elite Athletes: A Systematic Review with Meta-Analysis. J. Hum. Kinet. 2024, 91, 135–155. [Google Scholar] [CrossRef] [PubMed]
- Zouita, A.; Darragi, M.; Bousselmi, M.; Sghaeir, Z.; Clark, C.C.T.; Hackney, A.C.; Granacher, U.; Zouhal, H. The Effects of Resistance Training on Muscular Fitness, Muscle Morphology, and Body Composition in Elite Female Athletes: A Systematic Review. Sports Med. 2023, 53, 1709–1735. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Hakkinen, K.; Triplett-Mcbride, N.T.; Fry, A.C.; Koziris, L.P.; A Ratamess, N.; E Bauer, J.; Volek, J.S.; McConnell, T.; Newton, R.U.; et al. Physiological Changes with Periodized Resistance Training in Women Tennis Players. Med. Sci. Sports Exerc. 2003, 35, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Staron, R.S.; Karapondo, D.L.; Kraemer, W.J.; Fry, A.C.; Gordon, S.E.; Falkel, J.E.; Hagerman, F.C.; Hikida, R.S. Skeletal muscle adaptations during early phase of heavy-resistance training in men and women. J. Appl. Physiol. 1994, 76, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Qin, S.; Yan, B.; Girard, O. Metabolic and hormonal responses to acute high-load resistance exercise in normobaric hypoxia using a saturation clamp. Front. Physiol. 2024, 15, 1445229. [Google Scholar] [CrossRef]
- Poderoso, R.; Cirilo-Sousa, M.; Júnior, A.; Novaes, J.; Vianna, J.; Dias, M.; Leitão, L.; Reis, V.; Neto, N.; Vilaça-Alves, J. Gender Differences in Chronic Hormonal and Immunological Responses to CrossFit®. Int. J. Environ. Res. Public Heal. 2019, 16, 2577. [Google Scholar] [CrossRef]
- Arazi, H.; Khanmohammadi, A.; Asadi, A.; Haff, G.G. The effect of resistance training set configuration on strength, power, and hormonal adaptation in female volleyball players. Appl. Physiol. Nutr. Metab. 2018, 43, 154–164. [Google Scholar] [CrossRef]
- Kyröläinen, H.; Hackney, A.C.; Salminen, R.; Repola, J.; Häkkinen, K.; Haimi, J. Effects of Combined Strength and Endurance Training on Physical Performance and Biomarkers of Healthy Young Women. J. Strength Cond. Res. 2018, 32, 1554–1561. [Google Scholar] [CrossRef]
- Libardi, C.A.; Nogueira, F.R.D.; Vechin, F.C.; Conceição, M.S.; Bonganha, V.; Chacon-Mikahil, M.P.T. Acute hormonal responses following different velocities of eccentric exercise. Clin. Physiol. Funct. Imaging 2013, 33, 450–454. [Google Scholar] [CrossRef]
- Haff GG, Jackson JR, Kawamori N, Carlock JM, Hartman MJ, Kilgore JL, et al. Force-time curve characteristics and hormonal alterations during an eleven-week training period in elite women weightlifters. J Strength Cond Res. 2008;22(2):433-46.
- Linnamo, V.; Pakarinen, A.; Komi, P.V.; Kraemer, W.J.; Häkkinen, K. Acute Hormonal Responses to Submaximal and Maximal Heavy Resistance and Explosive Exercises in Men and Women. J. Strength Cond. Res. 2005, 19, 566–71. [Google Scholar] [CrossRef]
- Copeland, J.L.; Consitt, L.A.; Tremblay, M.S. Hormonal Responses to Endurance and Resistance Exercise in Females Aged 19-69 Years. Journals Gerontol. Ser. A 2002, 57, B158–B165. [Google Scholar] [CrossRef]
- Marx, J.O.; Ratamess, N.A.; Nindl, B.C.; Gotshalk, L.A.; Volek, J.S.; Dohi, K.; Bush, J.A.; Gómez, A.L.; Mazzetti, S.A.; Fleck, S.J.; et al. Low-volume circuit versus high-volume periodized resistance training in women. Med. Sci. Sports Exerc. 2001, 33, 635–643. [Google Scholar] [CrossRef]
- Consitt, L.A.; Copeland, J.L.; Tremblay, M.S. Hormone Responses to Resistance vs. Endurance Exercise in Premenopausal Females. Can. J. Appl. Physiol. 2001, 26, 574–587. [Google Scholar] [CrossRef]
- Häkkinen, K.; Pakarinen, A.; Kraemer, W.J.; Newton, R.U.; Alen, M. Basal concentrations and acute responses of serum hormones andstrength development during heavy resistance training in middle-aged andelderly men and women. Journals Gerontol. Ser. A 2000, 55, B95–105. [Google Scholar] [CrossRef] [PubMed]
- Hickson, R.C.; Hidaka, K.; Foster, C.; Falduto, M.T.; Chatterton, R.T. Successive time courses of strength development and steroid hormone responses to heavy-resistance training. J. Appl. Physiol. 1994, 76, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Kraemer WJ, Fleck SJ, Dziados JE, Harman EA, Marchitelli LJ, Gordon SE, et al. Changes in hormonal concentrations after different heavy-resistance exercise protocols in women. J Appl Physiol (1985). 1993;75(2):594-604.
- Häkkinen, K.; Pakarinen, A.; Kallinen, M. Neuromuscular adaptations and serum hormones in women during short-term intensive strength training. Eur. J. Appl. Physiol. 1992, 64, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Iemitsu, M.; Matsutani, K.; Kurihara, T.; Hamaoka, T.; Fujita, S. Resistance training restores muscle sex steroid hormone steroidogenesis in older men. FASEB J. 2014, 28, 1891–1897. [Google Scholar] [CrossRef]
- Ahtiainen, J.P.; Pakarinen, A.; Kraemer, W.J.; Hakkinen, K. Acute Hormonal Responses to Heavy Resistance Exercise in Strength Athletes Versus Nonathletes. Can. J. Appl. Physiol. 2004, 29, 527–543. [Google Scholar] [CrossRef]
- Raastad, T.; Bjøro, T.; Hallén, J. Hormonal responses to high- and moderate-intensity strength exercise. Eur. J. Appl. Physiol. 2000, 82, 121–128. [Google Scholar] [CrossRef]
- Buresh, R.; Berg, K.; French, J. The Effect of Resistive Exercise Rest Interval on Hormonal Response, Strength, and Hypertrophy With Training. J. Strength Cond. Res. 2009, 23, 62–71. [Google Scholar] [CrossRef]
- Migiano, M.J.; Vingren, J.L.; Volek, J.S.; Maresh, C.M.; Fragala, M.S.; Ho, J.-Y.; A Thomas, G.; Hatfield, D.L.; Häkkinen, K.; Ahtiainen, J.; et al. Endocrine Response Patterns to Acute Unilateral and Bilateral Resistance Exercise in Men. J. Strength Cond. Res. 2010, 24, 128–134. [Google Scholar] [CrossRef]
- West, D.W.D.; Phillips, S.M. Associations of exercise-induced hormone profiles and gains in strength and hypertrophy in a large cohort after weight training. Eur. J. Appl. Physiol. 2011, 112, 2693–2702. [Google Scholar] [CrossRef]
- Bell, G.J.; Syrotuik, D.; Martin, T.P.; Burnham, R.; Quinney, H.A. Effect of concurrent strength and endurance training on skeletal muscle properties and hormone concentrations in humans. Eur. J. Appl. Physiol. 2000, 81, 418–427. [Google Scholar] [CrossRef]
- Benini, R.; Nunes, P.R.P.; Orsatti, C.L.; Barcelos, L.C.; Orsatti, F.L. Effects of acute total body resistance exercise on hormonal and cytokines changes in men and women. 2015, 55, 337–344.
- Kraemer, W.J.; A Ratamess, N. Hormonal Responses and Adaptations to Resistance Exercise and Training. Sports Med. 2005, 35, 339–361. [Google Scholar] [CrossRef]
- Häkkinen, K.; Newton, R.U.; Walker, S.; Häkkinen, A.; Krapi, S.; Rekola, R.; Koponen, P.; Kraemer, W.J.; Haff, G.G.; Blazevich, A.J.; et al. Effects of Upper Body Eccentric versus Concentric Strength Training and Detraining on Maximal Force, Muscle Activation, Hypertrophy and Serum Hormones in Women. J. Sports Sci. Med. 2022, 21, 200–213. [Google Scholar] [CrossRef]
- Häkkinen, K.; Pakarinen, A.; Kraemer, W.J.; Häkkinen, A.; Valkeinen, H.; Alen, M. Selective muscle hypertrophy, changes in EMG and force, and serum hormones during strength training in older women. J. Appl. Physiol. 2001, 91, 569–580. [Google Scholar] [CrossRef]
- Häkkinen, K.; Pakarinen, A.; Kyröläinen, H.; Cheng, S.; Kim, D.; Komi, P. Neuromuscular Adaptations and Serum Hormones in Females During Prolonged Power Training. Int. J. Sports Med. 1990, 11, 91–98. [Google Scholar] [CrossRef]
- Häkkinen, K.; Pakarinen, A. Acute Hormonal Responses to Heavy Resistance Exercise in Men and Women at Different Ages. Int. J. Sports Med. 1995, 16, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Häkkinen, K.; Kraemer, W.J.; Pakarinen, A.; Tripleltt-Mcbride, T.; Mcbride, J.M.; Häkkinen, A.; Alen, M.; Mcguigan, M.R.; Bronks, R.; Newton, R.U. Effects of Heavy Resistance/Power Training on Maximal Strength, Muscle Morphology, and Hormonal Response Patterns in 60-75-Year-Old Men and Women. Can. J. Appl. Physiol. 2002, 27, 213–231. [Google Scholar] [CrossRef] [PubMed]
- Heavens, K.R.; Szivak, T.K.; Hooper, D.R.; Dunn-Lewis, C.; Comstock, B.A.; Flanagan, S.D.; Looney, D.P.; Kupchak, B.R.; Maresh, C.M.; Volek, J.S.; et al. The Effects of High Intensity Short Rest Resistance Exercise on Muscle Damage Markers in Men and Women. J. Strength Cond. Res. 2014, 28, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Keizer, H.; Beckers, E.; de Haan, J.; Janssen, G.; Kuipers, H.; van Kranenburg, G.; Geurten, P. Exercise-Induced Changes in the Percentage of Free Testosterone and Estradiol in Trained and Untrained Women*. Int. J. Sports Med. 1987, 08, S151–S153. [Google Scholar] [CrossRef]
- Keizer, H.; Kuipers, H.; de Haan, J.; Beckers, E.; Habets, L. Multiple Hormonal Responses to Physical Exercise in Eumenorrheic Trained and Untrained Women*. Int. J. Sports Med. 1987, 08, S139–S150. [Google Scholar] [CrossRef]
- Kemmler, W.; Wildt, L.; Engelke, K.; Pintag, R.; Pavel, M.; Bracher, B.; Weineck, J.; Kalender, W. Acute hormonal responses of a high impact physical exercise session in early postmenopausal women. Eur. J. Appl. Physiol. 2003, 90, 199–209. [Google Scholar] [CrossRef]
- Kostka, T.; Patricot, M.C.; Mathian, B.; Lacour, J.-R.; Bonnefoy, M. Anabolic and catabolic hormonal responses to experimental two-set low-volume resistance exercise in sedentary and active elderly people. Aging Clin. Exp. Res. 2003, 15, 123–130. [Google Scholar] [CrossRef]
- Le Panse, B.; Labsy, Z.; Baillot, A.; Vibarel-Rebot, N.; Parage, G.; Albrings, D.; Lasne, F.; Collomp, K. Changes in steroid hormones during an international powerlifting competition. Steroids 2012, 77, 1339–1344. [Google Scholar] [CrossRef]
- Luk, H.-Y.; Levitt, D.E.; Boyett, J.C.; Rojas, S.; Flader, S.M.; McFarlin, B.K.; Vingren, J.L. Resistance exercise-induced hormonal response promotes satellite cell proliferation in untrained men but not in women. Am. J. Physiol. Metab. 2019, 317, E421–E432. [Google Scholar] [CrossRef] [PubMed]
- Merrigan, J.J.; Tufano, J.J.; Fields, J.B.; Oliver, J.M.; Jones, M.T. Rest Redistribution Does Not Alter Hormone Responses in Resistance-Trained Women. J. Strength Cond. Res. 2020, 34, 1867–1874. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Aizawa, K.; Imai, T.; Kono, I.; Mesaki, N. Hormonal Responses to Resistance Exercise during Different Menstrual Cycle States. Med. Sci. Sports Exerc. 2011, 43, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, W.; Xirouchaki, C.E.; El-Gilany, A.-H. The Comparative Effects of High-Intensity Interval Training and Traditional Resistance Training on Hormonal Responses in Young Women: A 10-Week Intervention Study. Sports 2025, 13, 67. [Google Scholar] [CrossRef]
- Shahid, W.; Noor, R.; Bashir, M.S. Effects of exercise on sex steroid hormones (estrogen, progesterone, testosterone) in eumenorrheic females: A systematic to review and meta-analysis. BMC Women's Heal. 2024, 24, 1–16. [Google Scholar] [CrossRef]
- Umlauff, L.; Weil, P.; Zimmer, P.; Hackney, A.C.; Bloch, W.; Schumann, M. Oral Contraceptives Do Not Affect Physiological Responses to Strength Exercise. J. Strength Cond. Res. 2021, 35, 894–901. [Google Scholar] [CrossRef]
- Nindl, B.C.; Kraemer, W.J.; Gotshalk, L.A.; Marx, J.O.; Volek, J.S.; Bush, J.A.; Häkkinen, K.; Newton, R.U.; Fleck, S.J. Testosterone Responses after Resistance Exercise in Women: Influence of Regional Fat Distribution. Int. J. Sport Nutr. Exerc. Metab. 2001, 11, 451–465. [Google Scholar] [CrossRef]
- Cumming DC, Wall SR, Galbraith MA, Belcastro AN. Reproductive hormone responses to resistance exercise. Med Sci Sports Exerc. 1987;19(3):234-8.
- Weiss, L.W.; Cureton, K.J.; Thompson, F.N. Comparison of serum testosterone and androstenedione responses to weight lifting in men and women. Eur. J. Appl. Physiol. 1983, 50, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Staron, R.S.; Hagerman, F.C.; Hikida, R.S.; Fry, A.C.; Gordon, S.E.; Nindl, B.C.; Gothshalk, L.A.; Volek, J.S.; Marx, J.O.; et al. The effects of short-term resistance training on endocrine function in men and women. Eur. J. Appl. Physiol. 1998, 78, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Borst, S.E.; Vincent, K.R.; Lowenthal, D.T.; Braith, R.W. Effects of Resistance Training on Insulin-Like Growth Factor and its Binding Proteins in Men and Women Aged 60 to 85. J. Am. Geriatr. Soc. 2002, 50, 884–888. [Google Scholar] [CrossRef] [PubMed]
- Bosco, C.; Colli, R.; Bonomi, R.; VON Duvillard, S.P.; Viru, A. Monitoring strength training: neuromuscular and hormonal profile. Med. Sci. Sports Exerc. 2000, 32, 202–8. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Gordon, S.E.; Fleck, S.J.; Marchitelli, L.J.; Mello, R.; Dziados, J.E.; Friedl, K.; Harman, E.; Maresh, C.; Fry, A.C. Endogenous Anabolic Hormonal and Growth Factor Responses to Heavy Resistance Exercise in Males and Females. Int. J. Sports Med. 1991, 12, 228–235. [Google Scholar] [CrossRef]
- Kraemer, R.R.; Heleniak, R.J.; Tryniecki, J.L.; Kraemer, G.R.; Okazaki, N.J.; Castracane, V.D. Follicular and luteal phase hormonal responses to low-volume resistive exercise. Med. Sci. Sports Exerc. 1995, 27, 809–817. [Google Scholar] [CrossRef]
- Izquierdo, M.; Ibañez, J.; González-Badillo, J.J.; Häkkinen, K.; Ratamess, N.A.; Kraemer, W.J.; French, D.N.; Eslava, J.; Altadill, A.; Asiain, X.; et al. Differential effects of strength training leading to failure versus not to failure on hormonal responses, strength, and muscle power gains. J. Appl. Physiol. 2006, 100, 1647–1656. [Google Scholar] [CrossRef]
- A Herold, D.; Fitzgerald, R.L. Immunoassays for Testosterone in Women: Better than a Guess? Clin. Chem. 2003, 49, 1250–1251. [Google Scholar] [CrossRef]
- Zhang, X.; Feng, S.; Peng, R.; Li, H. The Role of Velocity-Based Training (VBT) in Enhancing Athletic Performance in Trained Individuals: A Meta-Analysis of Controlled Trials. Int. J. Environ. Res. Public Heal. 2022, 19, 9252. [Google Scholar] [CrossRef]
- Włodarczyk, M.; Adamus, P.; Zieliński, J.; Kantanista, A. Effects of Velocity-Based Training on Strength and Power in Elite Athletes—A Systematic Review. Int. J. Environ. Res. Public Heal. 2021, 18, 5257. [Google Scholar] [CrossRef]
- Rauch, J.T.; Loturco, I.; Cheesman, N.; Thiel, J.; Alvarez, M.; Miller, N.; Carpenter, N.; Barakat, C.; Velasquez, G.; Stanjones, A.; et al. Similar Strength and Power Adaptations between Two Different Velocity-Based Training Regimens in Collegiate Female Volleyball Players. Sports 2018, 6, 163. [Google Scholar] [CrossRef]
- Montalvo-Pérez, A.; Alejo, L.B.; Valenzuela, P.L.; Gil-Cabrera, J.; Talavera, E.; Lucia, A.; Barranco-Gil, D. Traditional Versus Velocity-Based Resistance Training in Competitive Female Cyclists: A Randomized Controlled Trial. Front. Physiol. 2021, 12. [Google Scholar] [CrossRef]
- Rossi, C.; Vasiljevic, I.; Manojlovic, M.; Trivic, T.; Ranisavljev, M.; Stajer, V.; Thomas, E.; Bianco, A.; Drid, P. Optimizing strength training protocols in young females: A comparative study of velocity-based and percentage-based training programs. Heliyon 2024, 10, e30644. [Google Scholar] [CrossRef]
- Hermes, M.J.; Fry, A.C. Intentionally Slow Concentric Velocity Resistance Exercise and Strength Adaptations: A Meta-Analysis. J. Strength Cond. Res. 2023, 37, e470–e484. [Google Scholar] [CrossRef] [PubMed]
- Lorenzetti, S.; Lamparter, T.; Lüthy, F. Validity and reliability of simple measurement device to assess the velocity of the barbell during squats. BMC Res. Notes 2017, 10, 707–707. [Google Scholar] [CrossRef]
- Achermann, B.; Oberhofer, K.; Ferguson, S.J.; Lorenzetti, S.R. Velocity-Based Strength Training: The Validity and Personal Monitoring of Barbell Velocity with the Apple Watch. Sports 2023, 11, 125. [Google Scholar] [CrossRef] [PubMed]
- Jukic, I.; Prnjak, K.; Helms, E.R.; McGuigan, M.R. Modeling the repetitions-in-reserve-velocity relationship: a valid method for resistance training monitoring and prescription, and fatigue management. Physiol. Rep. 2024, 12, e15955. [Google Scholar] [CrossRef]
- Declaration of Helsinki [Available from: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects.
- Consitt, L.A.; Copeland, J.L.; Tremblay, M.S. Endogenous Anabolic Hormone Responses to Endurance Versus Resistance Exercise and Training in Women. Sports Med. 2002, 32, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Banyard, H.G.; Nosaka, K.; Haff, G.G. Reliability and Validity of the Load–Velocity Relationship to Predict the 1RM Back Squat. J. Strength Cond. Res. 2017, 31, 1897–1904. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.G. Resistance Training Intensity Prescription Methods Based on Lifting Velocity Monitoring. Int. J. Sports Med. 2023, 45, 257–266. [Google Scholar] [CrossRef]
- Pareja-Blanco, F.; Walker, S.; Häkkinen, K. Validity of Using Velocity to Estimate Intensity in Resistance Exercises in Men and Women. Int. J. Sports Med. 2020, 41, 1047–1055. [Google Scholar] [CrossRef]
- Andrieu, T.; du Toit, T.; Vogt, B.; Mueller, M.D.; Groessl, M. Parallel targeted and non-targeted quantitative analysis of steroids in human serum and peritoneal fluid by liquid chromatography high-resolution mass spectrometry. Anal. Bioanal. Chem. 2022, 414, 7461–7472. [Google Scholar] [CrossRef]
- Liimatta, J.; du Toit, T.; Voegel, C.D.; Jääskeläinen, J.; Lakka, T.A.; Flück, C.E. Multiple androgen pathways contribute to the steroid signature of adrenarche. Mol. Cell. Endocrinol. 2024, 592, 112293. [Google Scholar] [CrossRef]
- Janse De Jonge X, Thompson B, Han A. Methodological Recommendations for Menstrual Cycle Research in Sports and Exercise. Medicine & Science in Sports & Exercise. 2019;51(12).
- Mann, JB. Developing Explosive Athletes: Use of Velocity Based Training in Athletes2016.
- Suchomel, T.J.; Nimphius, S.; Bellon, C.R.; Stone, M.H. The Importance of Muscular Strength: Training Considerations. Sports Med. 2018, 48, 765–785. [Google Scholar] [CrossRef]
- Baumgartner, S.; Bitterlich, N.; Geboltsberger, S.; Neuenschwander, M.; Matter, S.; Stute, P. Contraception, female cycle disorders and injuries in Swiss female elite athletes—a cross sectional study. Front. Physiol. 2023, 14, 1232656. [Google Scholar] [CrossRef]
- Af, T.; J, R.; Rj, A.; We, R. 8.20 11-Oxygenated androgens in health and disease. Yearb. Paediatr. Endocrinol. 2020. [Google Scholar] [CrossRef]
- Auchus, R.J. Overview of Dehydroepiandrosterone Biosynthesis. Semin. Reprod. Med. 2004, 22, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Naamneh Elzenaty R, du Toit T, Flück CE. Basics of androgen synthesis and action. Best Pract Res Clin Endocrinol Metab. 2022;36(4):101665.
- Heaney, J.L.J.; Carroll, D.; Phillips, A.C. DHEA, DHEA-S and cortisol responses to acute exercise in older adults in relation to exercise training status and sex. AGE 2011, 35, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Le Panse, B.; Vibarel-Rebot, N.; Parage, G.; Albrings, D.; Amiot, V.; De Ceaurriz, J.; Collomp, K. Cortisol, DHEA, and testosterone concentrations in saliva in response to an international powerlifting competition. Stress 2010, 13, 528–532. [Google Scholar] [CrossRef]
- Riechman, S.E.; Fabian, T.J.; Kroboth, P.D.; Ferrell, R.E. Steroid sulfatase gene variation and DHEA responsiveness to resistance exercise in MERET. Physiol. Genom. 2004, 17, 300–306. [Google Scholar] [CrossRef]
- Zouhal, H.; Jayavel, A.; Parasuraman, K.; Hayes, L.D.; Tourny, C.; Rhibi, F.; Laher, I.; Ben Abderrahman, A.; Hackney, A.C. Effects of Exercise Training on Anabolic and Catabolic Hormones with Advanced Age: A Systematic Review. Sports Med. 2021, 52, 1353–1368. [Google Scholar] [CrossRef]
- Schwartz, AG. Dehydroepiandrosterone, Cancer, and Aging. Aging Dis. 2022;13(2):423-32.
- Fang, Z.; Jiang, C.; Feng, Y.; Chen, R.; Lin, X.; Zhang, Z.; Han, L.; Chen, X.; Li, H.; Guo, Y.; et al. Effects of G6PD activity inhibition on the viability, ROS generation and mechanical properties of cervical cancer cells. Biochim. et Biophys. Acta (BBA) - Mol. Cell Res. 2016, 1863, 2245–2254. [Google Scholar] [CrossRef]
- TaheriChadorneshin H, Motameni S, Golestani A. Effects of resistance exercise type on cortisol and androgen cross talk in resistance-trained women. Journal of Exercise & Organ Cross Talk. 2021;1(1):8-14.
- Kotikangas, J.; Walker, S.; Toivonen, S.; Peltonen, H.; Häkkinen, K. Acute Neuromuscular and Hormonal Responses to Power, Strength, and Hypertrophic Protocols and Training Background. Front. Sports Act. Living 2022, 4, 919228. [Google Scholar] [CrossRef]
- Dote-Montero M, De-la OA, Jurado-Fasoli L, Ruiz JR, Castillo MJ, Amaro-Gahete FJ. The effects of three types of exercise training on steroid hormones in physically inactive middle-aged adults: a randomized controlled trial. Eur J Appl Physiol. 2021;121(8):2193-206.
- Szivak, T.K.; Hooper, D.R.; Dunn-Lewis, C.; Comstock, B.A.; Kupchak, B.R.; Apicella, J.M.; Saenz, C.; Maresh, C.M.; Denegar, C.R.; Kraemer, W.J. Adrenal Cortical Responses to High-Intensity, Short Rest, Resistance Exercise in Men and Women. J. Strength Cond. Res. 2013, 27, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Uchida MC, Bacurau R, Navarro F, Pontes F, Tessutti V, Moreau R, et al. Alteration of testosterone: Cortisol ratio induced by resistance training in women. Revista Brasileira de Medicina do Esporte. 2004;10.
- Turcu A, Smith JM, Auchus R, Rainey WE. Adrenal androgens and androgen precursors-definition, synthesis, regulation and physiologic actions. Compr Physiol. 2014;4(4):1369-81.
- Baffour-Awuah B, Man M, Goessler KF, Cornelissen VA, Dieberg G, Smart NA, et al. Effect of exercise training on the renin-angiotensin-aldosterone system: a meta-analysis. J Hum Hypertens. 2024;38(2):89-101.
- Boone CH, Hoffman JR, Gonzalez AM, Jajtner AR, Townsend JR, Baker KM, et al. Changes in Plasma Aldosterone and Electrolytes Following High-Volume and High-Intensity Resistance Exercise Protocols in Trained Men. J Strength Cond Res. 2016;30(7):1917-23.
- Patlar, S.; Ünsal, S. RAA System and Exercise Relationship. Turk. J. Sport Exerc. 2019, 21, 261–269. [Google Scholar] [CrossRef]
- Liu, J.; Xiao, Q.; Xiao, J.; Niu, C.; Li, Y.; Zhang, X.; Zhou, Z.; Shu, G.; Yin, G. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct. Target. Ther. 2022, 7, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Walczak, E.M.; Kuick, R.; Finco, I.; Bohin, N.; Hrycaj, S.M.; Wellik, D.M.; Hammer, G.D. Wnt Signaling Inhibits Adrenal Steroidogenesis by Cell-Autonomous and Non–Cell-Autonomous Mechanisms. Mol. Endocrinol. 2014, 28, 1471–1486. [Google Scholar] [CrossRef] [PubMed]
- Spillane, M.; Schwarz, N.; Willoughby, D.S. Upper-body resistance exercise augments vastus lateralis androgen receptor–DNA binding and canonical Wnt/β-catenin signaling compared to lower-body resistance exercise in resistance-trained men without an acute increase in serum testosterone. Steroids 2015, 98, 63–71. [Google Scholar] [CrossRef]
- Hackney, A.C. Stress and the neuroendocrine system: the role of exercise as a stressor and modifier of stress. Expert Rev. Endocrinol. Metab. 2006, 1, 783–792. [Google Scholar] [CrossRef]
- Song, Z.; Moore, D.R.; Hodson, N.; Ward, C.; Dent, J.R.; O’lEary, M.F.; Shaw, A.M.; Hamilton, D.L.; Sarkar, S.; Gangloff, Y.-G.; et al. Resistance exercise initiates mechanistic target of rapamycin (mTOR) translocation and protein complex co-localisation in human skeletal muscle. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Meignié, A.; Duclos, M.; Carling, C.; Orhant, E.; Provost, P.; Toussaint, J.-F.; Antero, J. The Effects of Menstrual Cycle Phase on Elite Athlete Performance: A Critical and Systematic Review. Front. Physiol. 2021, 12. [Google Scholar] [CrossRef]
- McNulty, K.L.; Elliott-Sale, K.J.; Dolan, E.; Swinton, P.A.; Ansdell, P.; Goodall, S.; Thomas, K.; Hicks, K.M. The Effects of Menstrual Cycle Phase on Exercise Performance in Eumenorrheic Women: A Systematic Review and Meta-Analysis. Sports Med. 2020, 50, 1813–1827. [Google Scholar] [CrossRef] [PubMed]
- Colenso-Semple, L.M.; D'SOuza, A.C.; Elliott-Sale, K.J.; Phillips, S.M. Current evidence shows no influence of women's menstrual cycle phase on acute strength performance or adaptations to resistance exercise training. Front. Sports Act. Living 2023, 5. [Google Scholar] [CrossRef] [PubMed]
- Wolken, J.K.; Peterson, M.M.; Cao, W.; Challoner, K.; Jin, Z. Sensitive LC-MS/MS Assay for Total Testosterone Quantification on Unit Resolution and High-Resolution Instruments. J. Clin. Med. 2024, 13, 7056. [Google Scholar] [CrossRef]
- Gervasio, J.; Zheng, S.; Skrotzki, C.; Pachete, A. The effect of oral contraceptive use on cortisol reactivity to the Trier Social Stress Test: A meta-analysis. Psychoneuroendocrinology 2022, 136, 105626. [Google Scholar] [CrossRef]

| Week | 1 | 2 | 3 | 4 | |||||||||||||||
| Football | Sets | Reps | MPV (m/s) | % 1RM | Sets | Reps | MPV (m/s) | % 1RM | Sets | Reps | MPV (m/s) | % 1RM |
Sets | Reps | MPV (m/s) | % 1RM | |||
| Back squat | 4 | 4,4,3,3 | 0.6-0.9 | 4 | 4,3,3,2 | 0.4-0.7 | 4 | 3,3,2,2 | 0.4-0.7 | 4 | 3,2,2,2 | 0.4-0.6 | |||||||
| Single leg jumps | 3 | 4 | 3 | 5 | 3 | 6 | 3 | 6 | |||||||||||
| Hip thrust | 3 | 17 | 60 | 3 | 18 | 60 | 3 | 19 | 60 | 3 | 20 | 60 | |||||||
| Eccentric single leg calf raises | 3 | 10 | 3 | 10 | 3 | 10 | 3 | 10 | |||||||||||
| Nordic hamstring curls | 4 | 4 | 4 | 5 | 4 | 6 | 4 | 6 | |||||||||||
| Push-up | 4(CS) | 6,8,10,12 | 4 | 6,8,10,12 | 4 | 6,8,10,12 | 4 | 6,8,10,12 | |||||||||||
| Pull-up (with elastic band) | 4(CS) | 4 | 4 | 4 | 4 | 4 | 4 | 4 | |||||||||||
| Dips | 4(CS) | 8 | 4 | 8 | 4 | 8 | 4 | 8 | |||||||||||
| Incline pull-up | 4(CS) | 4 | 4 | 4 | 4 | 4 | 4 | 4 | |||||||||||
| Track and field | |||||||||||||||||||
| Overhead squat | 4 | 6 | 60 | 4 | 6 | 60 | 4 | 6 | 60 | 4 | 6 | 60 | |||||||
| Hang power clean | 4 | 4,4,2,2 | 80-85 | 4 | 4,3,2,2 | 80-85 | 5 | 3,3,2,1,1 | 85-90 | 5 | 3,2,1,1,1 | 90-95 | |||||||
| Back squat | 4 | 4,4,3,3 | 0.4-0.7 | 4 | 4,3,3,2 | 0.4-0.7 | 4 | 3,3,2,2 | 0.4-0.7 | 4 | 3,2,2,2 | 0.4-0.6 | |||||||
| Single leg step up | 4 | 6,6,4,4 | 80-85 | 4 | 6,4,4,3 | 80-85 | 4 | 4,4,3,3 | 85-90 | 4 | 4,3,3,2 | 85-90 | |||||||
| Eccentric single leg press | 3 | 5,4,3 | 100-105 | 3 | 4,4,3 | 105-110 | 3 | 4,3,2 | 105-110 | 3 | 3,3,2 | 110 | |||||||
| Hip thrust | 4 | 14 | 60 | 4 | 16 | 60 | 4 | 18 | 60 | 4 | 20 | 60 | |||||||
| Eccentric leg press calf raises | 3 | 8 | 100 | 3 | 8 | 100 | 3 | 8 | 100 | 3 | 8 | 100 | |||||||
| Bench press | 5(CS) | 3,4,5,6,7 | 70 | 5(CS) | 3,4,5,6,7 | 70 | 5(CS) | 3,4,5,6,7 | 70 | 5(CS) | 3,4,5,6,7 | 70 | |||||||
| Push-up with dumbbells | 5 | 12 | 5 | 12 | 5 | 12 | 5 | 12 | |||||||||||
| Pull-up with elastic band | 5(CS) | 3,4,5,6,7 | 5(CS) | 3,4,5,6,7 | 5(CS) | 3,4,5,6,7 | 5(CS) | 3,4,5,6,7 | |||||||||||
| Characteristic | Football (n=8) (mean, +/- SD, median (1st quartile, 3rd quartile) | Track and field (n=11) (mean, +/- SD, median (1st quartile, 3rd quartile) | Total group (n=19) (mean, +/- SD, median (1st quartile, 3rd quartile) |
| Age (y) | 18.9 ± 0.8 19.0 (18.0 ; 19.8) |
25.0 ± 3.5 24.0 (21.0 ; 28.0) |
22.4 ± 4.1 21.0 (19.0 ; 26.0) |
| Height (cm) | 168.9 ± 3.2 169.5 (166.0 ; 171.0) |
172.4 ± 7.3 171.0 (168.0 ; 177.0) |
170.9 ± 6.1 170.5 (166.0 ; 172.0) |
| Weight (kg) | 61.2 ± 4,7 60.8 (56.4 ; 64.3) |
67.3 ± 6.5 66.1 (64.1 ; 73.3) |
64.9 ± 6.5 64.7 (59.5 ; 69.6) |
| BMI (kg/m2) | 21.9 ± 2.1 20.9 (19.6 ; 23.8) |
22.8 ± 1.6 22.3 (21.8 ; 23.8) |
22.4 ± 1.8 22.3 (20.6 ; 23.8) |
| Training hours | 10.9 ± 1.2 11.0 (10.0 ; 11.0) |
16.7 ± 2.4 18.0 (14.0 ; 18.0) |
14.3 ± 3.6 14.0 (11.0 ; 18.0) |
| 1RMest for the back squat (kg) | 101.7 ± 18.6 96.5 (75.6 ; 141.7) |
118.0 ± 19.8 110.2 (93.0 ; 153.3) |
110.8 ± 20.9 106.8 (75.6 ; 153.3) |
| ROM for the back squat (cm) | 45.3 ± 7.3 42.5 (38.0 ; 59.0) |
62.2 ± 6.1 61.0 (54.0 ; 77.0) |
54.7 ± 10.8 57.5 (38.0 ; 77.0) |
|
Parameter [nmol/l] |
Pre | Post |
Absolute changes |
Pwil [pt] Effect size |
Relative changes |
Pwil [pt] Effect size |
|
Mean (SD) Median |
Mean (SD) Median |
Mean (SD) Median |
Mean (SD) Median |
|||
| 11-Deoxycorticosterone (11-DOC) |
0.084 (0.040) 0.089 |
0.061 (0.031) 0.089 |
-0.023 (0.027) -0.011 |
0.001 [0.001] 0.86 |
77.7% (29.1%) 80.6% |
0.002 [0.004] 0.77 |
| 11-Deoxycortisol (S) |
0.530 (0.241) 0.568 |
0.254 (0.184) 0.205) |
-0.275 (0.270) -0.206 |
< 0.001 [< 0.001] 1.02 |
53.5% (34.7%) 45.3% |
< 0.001 [< 0.001] 1.34 |
| 11-Ketoandrostenedione (11KA4) | 0.414 (0.160) 0.369 |
0.493 (0.185) 0.476 |
0.079 (0.139) 0.081 |
0.023 [0.023] 0.57 |
124.3% (45.0%) 120.3% |
0.023 [0.030] 0.54 |
| 11-Ketotestosterone (11KT) |
1.220 (0.465) 1.184 |
1.334 (0.711) 1.221 |
0.114 (0.492) 0.023 |
0.651 [0.326] 0.23 |
108.8% (39.3%) 101.9% |
0.515 [0.341] 0.22 |
| 11β-Hydroxyandrostenedione (11OHA4) | 3.122 (1.052) 2.990 |
2.415 (1.139) 1.883 |
-0.707 (1.083) -0.589 |
0.012 [0.011] 0.65 |
79.9% (31.5%) 76.7% |
0.011 [0.013] 0.64 |
| 11β-Hydroxytestosterone (11OHT) | 0.228 (0.096) 0.214 |
0.260 (0.162) 0.188 |
0.032 (0.121) 0.015 |
0.352 [0.258] 0.27 |
114.5% (52.5%) 110.8% |
0.465 [0.243] 0.28 |
| 16α-Hydroxyprogesterone (16αOHP4) | 0.257 (0.121) 0.240 |
0.175 (0.082) 0.156 |
-0.082 (0.120) -0.053 |
0.002 [0.008] 0.69 |
78.0% (34.4%) 76.2% |
0.009 [0.012] 0.64 |
| 17α,20α-Dihydroxyprogesterone (17α20α-diOHP4) | 0.625 (0.220) 0.643 |
0.600 (0.243) 0.653 |
-0.025 (0.153) -0.022 |
0.312 [0.486] 0.16 |
96.6% (24.7%) 91.2% |
0.595 [0.552] 0.14 |
| 17α-Hydroxyprogesterone (17OHP4) | 1.461 (0.661) 1.461 |
1.321 (0.640) 1.414 |
-0.140 (0.381) -0.151 |
0.241 [0.126] 0.37 |
91.6% (30.2%) 91.8% |
0.312 [0.242] 0.28 |
| 21-Deoxycortisol (21DF) |
0.043 (0.042) 0.028 |
0.010 (0.011) 0.008 |
-0.032 (0.040) -0.021 |
< 0.001 [0.003] 0.80 |
37.2% (46.2%) 19.2% |
< 0.001 [< 0.001] 1.36 |
| 5α-Dihydrotestosterone (5DHT) |
0.554 (0.256) 0.463 |
0.488 (0.237) 0.469 |
-0.067 (0.208) -0.050 |
0.275 [0.181] 0.32 |
90.0% (25.3%) 91.4% |
0.169 [0.102] 0.40 |
| Aldosterone (ALDO) |
0.275 (0.183) 0.233 |
0.247 (0.118) 0.277 |
-0.028 (0.154) 0.002 |
0.776 [0.435] 0.18 |
108.7% (54.0%) 100.3% |
0.623 [0.493] 0.16 |
| Androstenedione (A4) |
3.098 (1.074) 2.846 |
2.839 (1.208) 2.807 |
-0.260 (0.557) -0.245 |
0.080 [0.057] 0.53 |
91.9% (19.0%) 89.3% |
0.080 [0.079] 0.43 |
| Androsterone (AST) |
0.758 (0.594) 0.697 |
0.557 (0.274) 0.478 |
-0.201 (0.582) -0.045 |
0.049 [0.150] 0.34 |
85.2% (29.1%) 93.1% |
0.045 [0.039] 0.51 |
| Cortisone (E) |
46.993 (8.902) 46.620 |
42.629 (12.292) 39.617 |
-4.364 (10.318) -1.731 |
0.123 [0.082] 0.42 |
91.3% (21.6%) 96.5% |
0.113 [0.096] 0.40 |
| Corticosterone (CORT) |
9.059 (4.883) 8.739 |
4.052 (3.260) 3.022 |
-5.007 (5.763) -6.146 |
0.001 [0.001] 0.87 |
56.9% (46.5%) 37.2% |
0.001 [0.001] 0.93 |
| Cortisol (F) |
309.30 (93.23) 288.04 |
206.58 (75.93) 208.84 |
-102.72 (83.93) -122.40 |
< 0.001 [< 0.001] 1.22 |
69.1% (24.0%) 67.1% |
< 0.001 [< 0.001] 1.29 |
| Dehydroepiandrosterone (DHEA) |
19.460 (7.793) 18.515 |
15.647 (5.485) 16.094 |
-3.813 (6.342) -1.909 |
0.006 [0.017] 0.60 |
82.9% (22.2%) 89.5% |
0.006 [0.004] 0.77 |
| Dehydroepiandrosteronsulfate (DHEAS) |
6847.7 (4279.5) 5700.9 |
7198.4 (3886.5) 6216.2 |
350.7 (1580.8) 488.5 |
0.145 [0.346] 0.22 |
109.9% (19.0%) 107.0% |
0.045 [0.036] 0.52 |
| Etiocholanolone (ETIO) |
0.961 (0.718) 0.670 |
0.832 (0.596) 0.700 |
-0.129 (0.444) -0.073 |
0.490 [0.221] 0.29 |
96.1% (32.8%) 85.8% |
0.395 [0.609] 0.12 |
| Pregnenolone (P5) |
1.010 (0.901) 1.060 |
0.980 (0.893) 0.771 |
-0.116 (0.736) -0.092 |
0.651 [0.502] 0.16 |
129.7% (112.3%) 94.1% |
0.615 [0.264] 0.26 |
| Progesterone (P4) |
4.119 (4.022) 2.396 |
4.625 (4.731) 2.685 |
0.506 (1.420) 0.013 |
0.293 [0.138] 0.36 |
105.7% (23.6%) 106.0% |
0.293 [0.304] 0.24 |
| Testosterone (T) |
0.771 (0.383) 0.674 |
0.726 (0.401) -0.042 |
-0.046 (0.111) -0.042 |
0.080 [0.089] 0.41 |
93.8% 16.5%) 92.9% |
0.080 [0.119] 0.38 |
|
Profile (artithmetic sum) [nmol/l] |
Mean (SD) Median |
Pwil [pt] |
Effect size |
Mean (SD) Median |
Pwil [pt] |
Effect size |
| Androgenic hormones (11OHA4, 11OHT, 11KA4, 11KT, DHT, A4, AST, DHEA, DHEAS, T and ETIO) | 345.7 (1582.5) 483.9 |
0.145 [0.354] |
0.22 | 109.7% (18.9%) 107.0% |
0.055 [0.038] |
0.51 |
| Adrenal androgenic hormones (11KA4, 11KT, 11OHA4, 11OHT, DHEAS) | 349.8 (1580.8) 486.0 |
0.145 [0.348] |
0.22 | 109.8% (19.0%) 107.0% |
0.045 [0.037] |
0.52 |
| Glucocorticoids (S, 21DF, F, E) | -107.39 (88.09) -134.46 |
< 0.001 [< 0.001] |
1.22 | 71.8% (22.8%) 74.8% |
< 0.001 [< 0.001] |
1.23 |
| Mineralocorticoids (11-DOC, CORT, ALDO) | -5.058 (5.841) -6.172 |
0.001 [0.001] |
0.87 | 58.5% (45.5%) 39.4% |
0.001 [0.001] |
0.91 |
| Classic androgen pathway (DHT, A4, AST, DHEA, ETIO) | -4.515 (6.861) -2.622 |
0.005 [0.010] |
0.66 | 84.6% (19.7%) 84.1% |
0.005 [0.003] |
0.78 |
| 11-oxy pathway (11OHA4, 11OHT, 11KA4, 11KT, A4 and T) | -0.787 (2.242) -1.266 |
0.096 [0.143] |
0.35 | 92.3% (27.3%) 85.7% |
0.113 [0.143] |
0.28 |
| Backdoor pathway (17OHP4, DHT, AST, P4) | 0.040 (2.000) 0.072 |
0.953 [0.932] |
0.02 | 98.9% (23.0%) 101.5% |
0.922 [0.835] |
0.05 |
| Weighted profile of the classic androgen pathway | -1.000 (0.582) 0.985 |
<0.001 [<0.001] |
1.72 | |||
| Weighted profile of the 11-oxy pathway | -1.000 (1.032) -1.232 |
0.001 [0.001] |
0.97 | |||
| Weighted profile of the backdoor pathway | -0.999 (1.390) -0.818 |
0.007 [0.006] |
0.72 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




