Submitted:
03 September 2025
Posted:
04 September 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Liquid Immunogenic Fiducial Eluter (LIFE) Biomaterial Assembly
2.3. Cell Culture Preparations for Mice Inoculation
2.4. Data Collection and Image Analysis
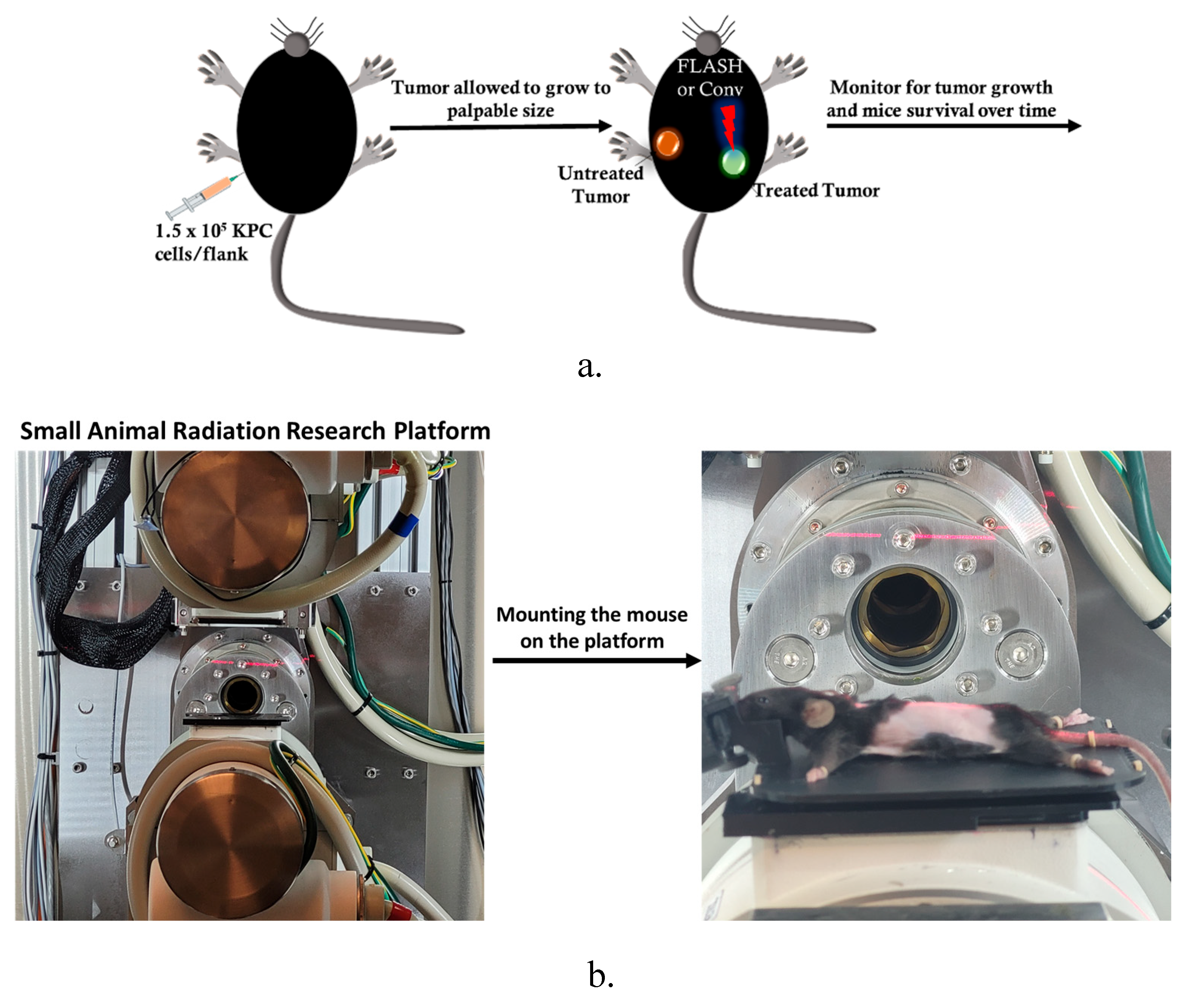
2.5. Small Animal FLASH Radiation Research Platform to Deliver Conventional and Flash Radiotherapy
2.5.1. FLASH-SARRP
2.5.2. Animal Setup and Irradiation
2.6. Histology Staining
2.7. Immunohistochemistry Staining
2.8. Multiplex Immunofluorescence
2.9. Statistical Analysis
3. Results
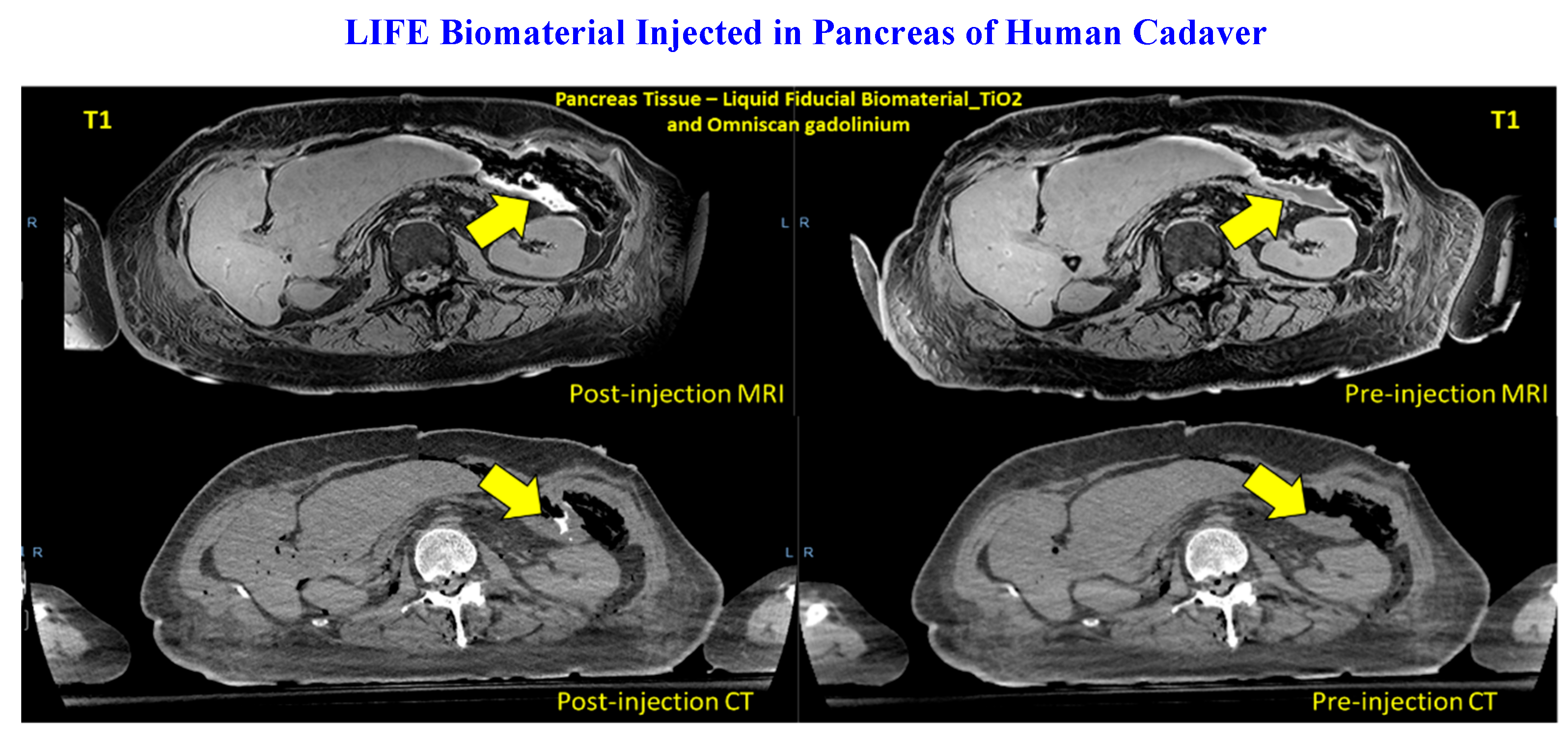
3.1. LIFE Biomaterial Providing Image-Guidance During RT
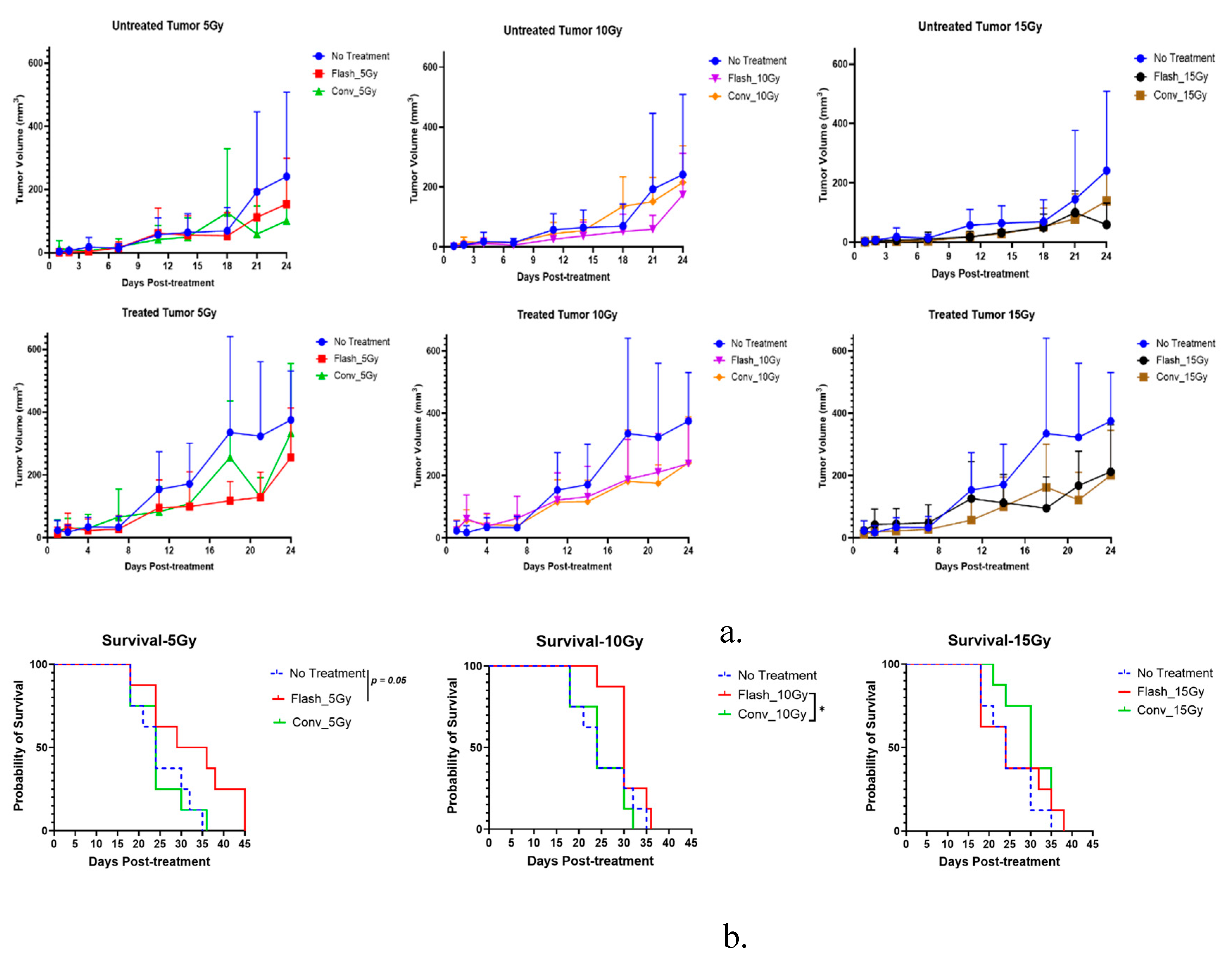
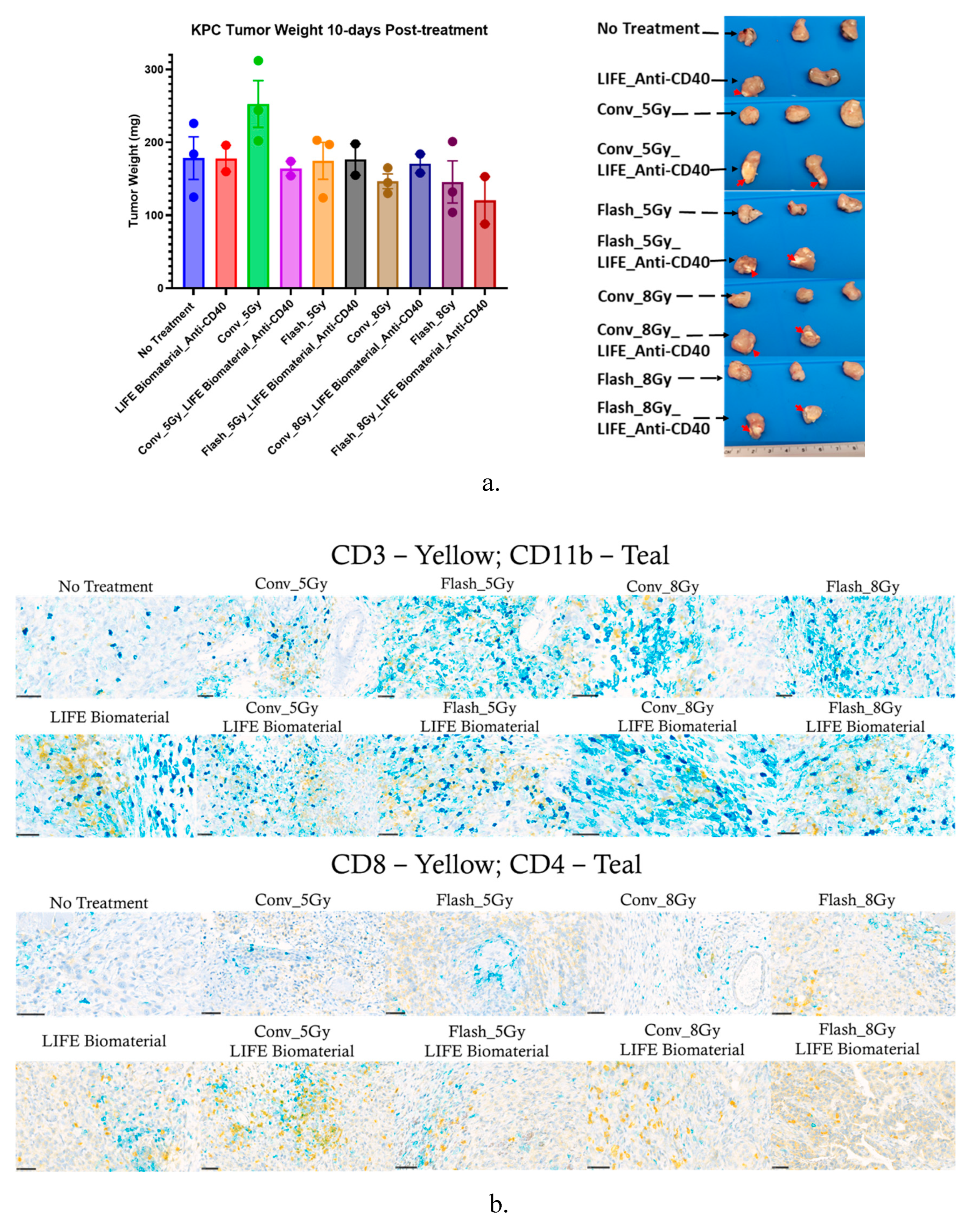
3.2. Effect of FLASH-RT Versus CONV RT on Pancreatic Cancer
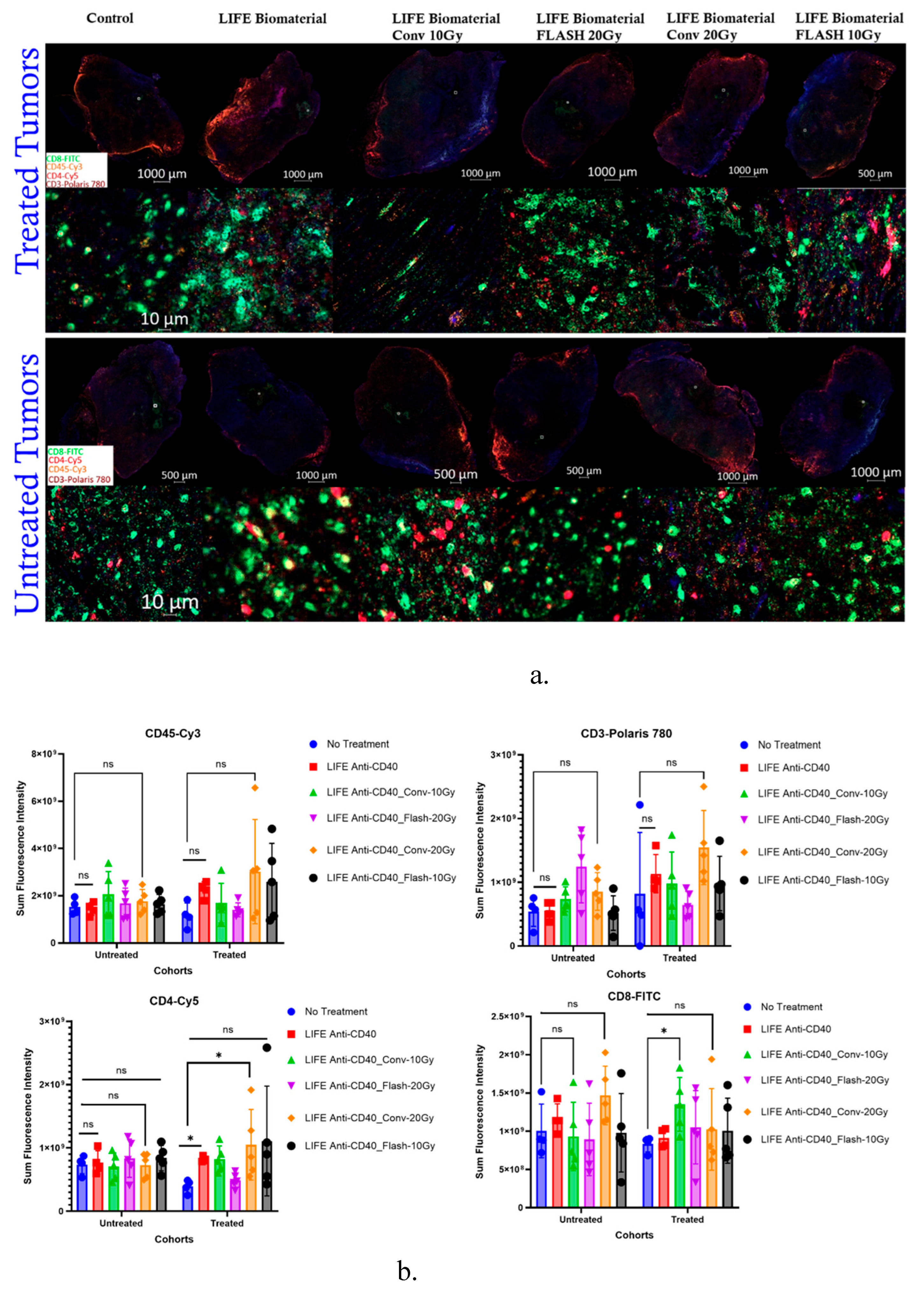
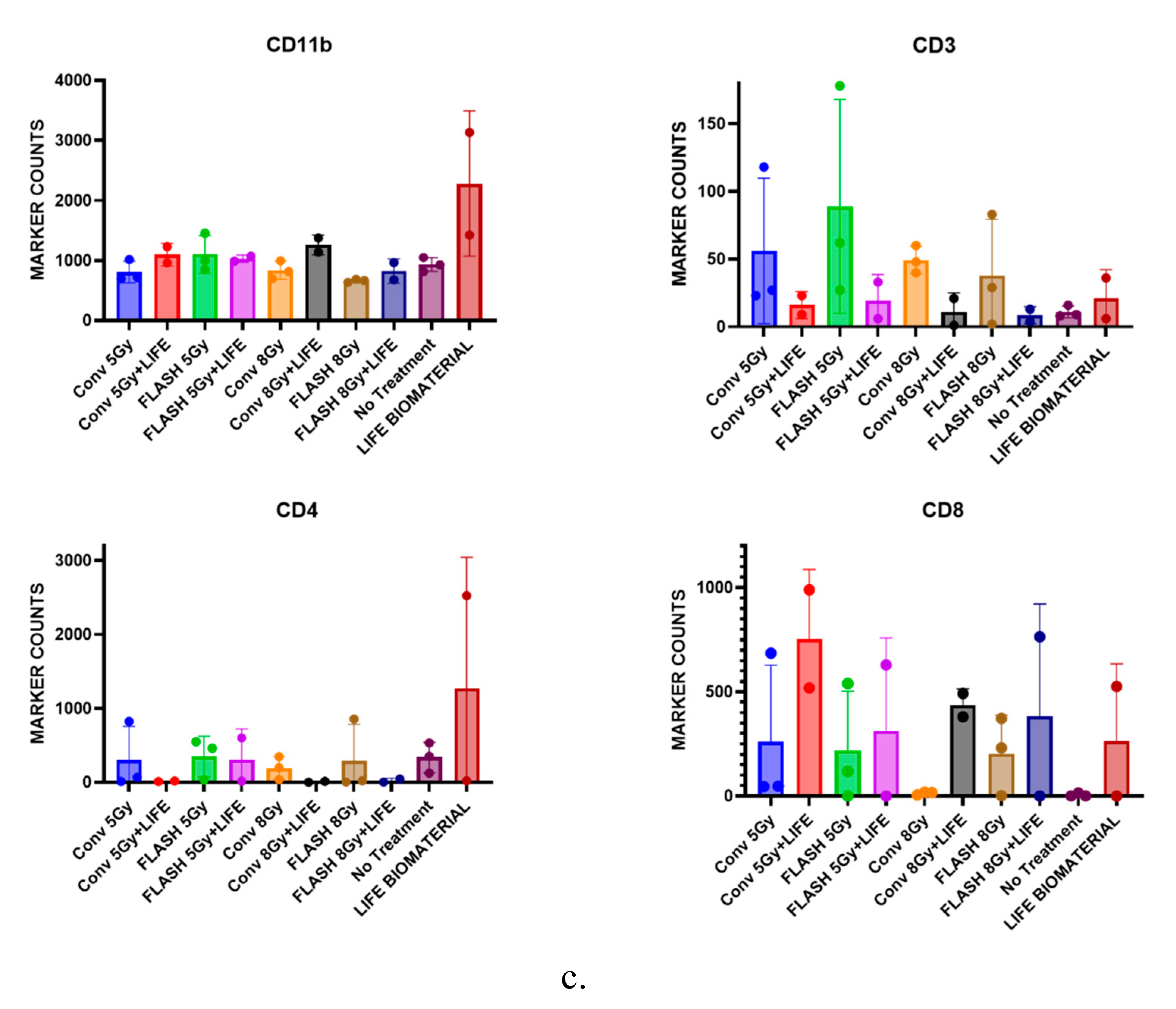
3.3. Assessment of T-Cell Infiltrations in the Pancreatic Tumor Following the Combination of FLASH-RT and LIFE Biomaterial.
3.3.1. Multiplex Immunofluorescence
3.3.2. Immunohistochemistry
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barton, M.B.; Frommer, M.; Shafiq, J. Role of Radiotherapy in Cancer Control in Low-Income and Middle-Income Countries. Lancet Oncol. 2006, 7, 584–595. [Google Scholar] [CrossRef]
- McGarrigle, J.M.; Long, K.R.; Prezado, Y. The FLASH Effect—an Evaluation of Preclinical Studies of Ultra-High Dose Rate Radiotherapy. Front. Oncol. 2024, 14. [Google Scholar] [CrossRef]
- Ezzell, G.A.; Galvin, J.M.; Low, D.; Palta, J.R.; Rosen, I.; Sharpe, M.B.; Xia, P.; Xiao, Y.; Xing, L.; Yu, C.X. Guidance Document on Delivery, Treatment Planning, and Clinical Implementation of IMRT: Report of the IMRT Subcommittee of the AAPM Radiation Therapy Committee. Med. Phys. 2003, 30, 2089–2115. [Google Scholar] [CrossRef] [PubMed]
- Garibaldi, C.; Jereczek-Fossa, B.A.; Marvaso, G.; Dicuonzo, S.; Rojas, D.P.; Cattani, F.; Starzyńska, A.; Ciardo, D.; Surgo, A.; Leonardi, M.C.; et al. Recent Advances in Radiation Oncology. Ecancermedicalscience 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Erickson, D.P.J.; Ford, E.C.; Emery, R.C.; Kranz, M.; Goff, P.; Schwarz, M.; Meyer, J.; Wong, T.; Saini, J.; et al. Preclinical Ultra-High Dose Rate (FLASH) Proton Radiation Therapy System for Small Animal Studies. Adv. Radiat. Oncol. 2024, 9, 101425. [Google Scholar] [CrossRef]
- Lin, B.; Gao, F.; Yang, Y.; Wu, D.; Zhang, Y.; Feng, G.; Dai, T.; Du, X. FLASH Radiotherapy: History and Future. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Lancellotta, V.; Chierchini, S.; Perrucci, E.; Saldi, S.; Falcinelli, L.; Iacco, M.; Zucchetti, C.; Palumbo, I.; Bini, V.; Aristei, C. Skin Toxicity after Chest Wall/Breast plus Level III-IV Lymph Nodes Treatment with Helical Tomotherapy. Cancer Invest. 2018, 36, 504–511. [Google Scholar] [CrossRef]
- Hughes, J.R.; Parsons, J.L. FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy. Int. J. Mol. Sci. 2020, 21, 6492. [Google Scholar] [CrossRef] [PubMed]
- Miles, D.; Sforza, D.; Wong, J.W.; Gabrielson, K.; Aziz, K.; Mahesh, M.; Coulter, J.B.; Siddiqui, I.; Tran, P.T.; Viswanathan, A.N.; et al. FLASH Effects Induced by Orthovoltage X-Rays. Int. J. Radiat. Oncol. 2023, 117, 1018–1027. [Google Scholar] [CrossRef]
- Miles, D.; Sforza, D.; Cano, M.; Peterson, C.; Gabrielson, K.; Wong, J.W.; Handa, J.; Rezaee, M. A Feasibility Study of Preclinical Ocular X-Ray FLASH Radiation Therapy. Int. J. Radiat. Oncol. 2025. [Google Scholar] [CrossRef] [PubMed]
- Favaudon, V.; Caplier, L.; Monceau, V.; Pouzoulet, F.; Sayarath, M.; Fouillade, C.; Poupon, M.-F.; Brito, I.; Hupé, P.; Bourhis, J.; et al. Ultrahigh Dose-Rate FLASH Irradiation Increases the Differential Response between Normal and Tumor Tissue in Mice. Sci. Transl. Med. 2014, 6. [Google Scholar] [CrossRef]
- Zhang, Y.; Ding, Z.; Perentesis, J.P.; Khuntia, D.; Pfister, S.X.; Sharma, R.A. Can Rational Combination of Ultra-High Dose Rate FLASH Radiotherapy with Immunotherapy Provide a Novel Approach to Cancer Treatment? Clin. Oncol. 2021, 33, 713–722. [Google Scholar] [CrossRef]
- Vozenin, M.-C.; De Fornel, P.; Petersson, K.; Favaudon, V.; Jaccard, M.; Germond, J.-F.; Petit, B.; Burki, M.; Ferrand, G.; Patin, D.; et al. The Advantage of FLASH Radiotherapy Confirmed in Mini-Pig and Cat-Cancer Patients. Clin. Cancer Res. 2019, 25, 35–42. [Google Scholar] [CrossRef]
- Yilmaz, M.T.; Hurmuz, P.; Yazici, G. FLASH-Radiotherapy: A New Perspective in Immunotherapy Era? Radiother. Oncol. 2020, 145, 137. [Google Scholar] [CrossRef]
- Moon, E.J.; Petersson, K.; Olcina, M.M. The Importance of Hypoxia in Radiotherapy for the Immune Response, Metastatic Potential and FLASH-RT. Int. J. Radiat. Biol. 2022, 98, 439–451. [Google Scholar] [CrossRef]
- Padilla, O.; Minns, H.E.; Wei, H.-J.; Fan, W.; Webster-Carrion, A.; Tazhibi, M.; McQuillan, N.M.; Zhang, X.; Gallitto, M.; Yeh, R.; et al. Immune Response Following FLASH and Conventional Radiation in Diffuse Midline Glioma. Int. J. Radiat. Oncol. 2024, 119, 1248–1260. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.M.; Schoenfeld, J.D. Radiotherapy and Immunotherapy for Head and Neck Cancer: Current Evidence and Challenges. Front. Oncol. 2021, 10. [Google Scholar] [CrossRef]
- Guevara, M.L.; Persano, F.; Persano, S. Nano-Immunotherapy: Overcoming Tumour Immune Evasion. Semin. Cancer Biol. 2021, 69, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Pardoll, D.M. The Blockade of Immune Checkpoints in Cancer Immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Mardiana, S.; Solomon, B.J.; Darcy, P.K.; Beavis, P.A. Supercharging Adoptive T Cell Therapy to Overcome Solid Tumor–Induced Immunosuppression. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- DeMuth, P.C.; Min, Y.; Irvine, D.J.; Hammond, P.T. Implantable Silk Composite Microneedles for Programmable Vaccine Release Kinetics and Enhanced Immunogenicity in Transcutaneous Immunization. Adv. Healthc. Mater. 2014, 3, 47–58. [Google Scholar] [CrossRef]
- Moreau, M.; Keno, L.S.; China, D.; Mao, S.; Acter, S.; Sy, G.; Hooshangnejad, H.; Chow, K.F.; Sajo, E.; Walker, J.; et al. Investigating the Use of a Liquid Immunogenic Fiducial Eluter Biomaterial in Cervical Cancer Treatment. Cancers (Basel). 2024, 16, 1212. [Google Scholar] [CrossRef]
- Moreau, M.; Richards, G.; Yasmin-Karim, S.; Narang, A.; Deville, C.; Ngwa, W. A Liquid Immunogenic Fiducial Eluter for Image-Guided Radiotherapy. Front. Oncol. 2022, 12. [Google Scholar] [CrossRef]
- Moreau, M.; Mao, S.; Ngwa, U.; Yasmin-Karim, S.; China, D.; Hooshangnejad, H.; Sforza, D.; Ding, K.; Li, H.; Rezaee, M.; et al. Democratizing FLASH Radiotherapy. Semin. Radiat. Oncol. 2024, 34, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Tajik Mansoury, M.-A.; Sforza, D.; Wong, J.; Iordachita, I.; Rezaee, M. Dosimetric Commissioning of Small Animal FLASH Radiation Research Platform. Phys. Med. Biol. 2025, 70, 115015. [Google Scholar] [CrossRef] [PubMed]
- Wirsdörfer, F.; Jendrossek, V. The Role of Lymphocytes in Radiotherapy-Induced Adverse Late Effects in the Lung. Front. Immunol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



