Device Architecture and Functional Design
The baseline module uses three honeycomb layers in a 12 × 12 cm footprint. Fifty-six hexagonal wells (17 mm across flats, 4 mm depth) provide approximately 0.75 mL working volume each, with peripheral through-holes for alignment and stacking. Adjacent layers are separated by 0.2-µm hydrophilic microfiltration membranes of polytetrafluoroethylene (PTFE), polyethersulfone (PES), or polyvinylidene fluoride (PVDF) with 80–150 µm thickness. Where finer selectivity is needed, affinity or molecularly imprinted membranes can replace or complement resin capture without altering the overall stack [
15,
16].
Metabolites are captured in a dedicated layer containing 10–25 mg of HP-20/Amberlite XAD-type resin per well (up to 40 mg when yield permits), spanning a broad polarity range [
20,
21,
22]. Below each capture well, a silicone duckbill check-valve (elastomeric, not a filter membrane) sits in a shallow conical seat (slit 2.0–2.5 mm, wall 0.25–0.35 mm, opening angle 8–12°, cracking pressure 0.2–0.5 kPa), favoring unidirectional flow toward a lateral vacuum manifold. An inline non-return valve at the outlet adds redundancy.
If negative pressure is employed, downstream placement relative to the capture layer is suggested, limited to a pressure differential (ΔP) of approximately 5–10 mbar (0.5–1.0 kPa). Oil-free diaphragm pumps (for example, KNF Neuberger) are suggested at 0.1–1.0 L·min⁻¹ with programmable duty-cycling—illustratively 1–5 minutes ON per 30–60 minutes OFF—to support retrieval while keeping the culture interface near ambient partial pressures of oxygen.
Materials are specified for chemical inertness and field robustness: poly(ether ether ketone) (PEEK), polycarbonate (PC), and polypropylene (PP) for structure; PEEK or titanium grade 2 (Ti-2) for fasteners (with stainless steel 316 [SS-316] only if passivated and kept away from high-salinity or acidic environments); and expanded polytetrafluoroethylene (ePTFE) or medical-grade silicone gaskets (0.5–1.0 mm) under torque-controlled compression. Adhesive-backed or thermally laminated membranes can improve uniformity and handling.
Device Loading and Environmental Deployment
To favor isolation, NP-TRAP employs limiting-dilution loading under a Poisson regime with λ ≈ 0.1–0.2 cells·well⁻¹, yielding P(≥2) < 2%. For 0.5 mL fills, a titer of 0.2–0.4 cells·mL⁻¹ is targeted and verified by droplet plating. Physical compartmentalization maintains clonal enrichment during extended in situ incubation. Slow-growing or low-abundance taxa are supported by low-nutrient gellan/agar (1.5–2%), matrix-matched osmolarity, and longer deployments (21–56 days), followed by single-well subculture and standard taxonomic workflows (morphology plus 16S rRNA gene sequencing [16S], internal transcribed spacer [ITS], and whole-genome sequencing [WGS]) [
1,
2,
3,
9,
10,
11].
When both faces of a culture well are membrane-bound, two practical fills are used. Fill-then-seal: the bottom membrane is sealed; warm soft-gel medium (40–45 °C) plus inoculum is dispensed (0.5–0.75 mL), allowed to gel, and the top membrane is laminated by low-temperature pressure lamination or a pressure-sensitive adhesive (PSA) membrane using a rigid stencil. Micro-port injection: both membranes are pre-laminated; a 100–200 µm port is laser-punched and medium is injected through a 30–32 G needle; the puncture is sealed with an ultraviolet (UV)-curable biocompatible dot or PSA micro-patch. Suggested acceptance: no dye leakage or mass loss greater than 1% under ±20 mbar for 10 minutes; visual integrity at 40–45 °C.
Reproducibility Package
Per-well dimensions: across-flats 17 ± 0.2 mm, depth 4.0 ± 0.1 mm, wall 2.0 ± 0.1 mm, seat chamfer 30° × 0.5 mm.
Membranes: 0.2 µm hydrophilic PTFE/PES/PVDF (80–150 µm), adhesive-backed or thermally laminated.
Resins: HP-20/XAD-18; 10–25 mg per well (up to 40 mg).
Valves: silicone duckbill; slit 2.0–2.5 mm, wall 0.25–0.35 mm, cracking 0.2–0.5 kPa, seat outer diameter (OD) 4.0–4.5 mm.
Pump and control: suggested oil-free diaphragm pump; ΔP 5–10 mbar; duty 1–5 min·h⁻¹; outlet non-return valve.
Materials: structural PEEK/PC/PP; fasteners PEEK/Ti-2; gaskets ePTFE/silicone 0.5–1.0 mm.
Suggested Bench Pre-Validation
A compact benchtop verification set is outlined to make the concept actionable once a basic lab setup is available. Airtightness and sealing: pressure-decay at +20 mbar and –10 mbar for 10 minutes with less than 5% ΔP as a practical target. Valve unidirectionality: bubble-point and dye-backflow per well with no reverse flow at +0.5 kPa; confirm redundancy via the inline non-return valve. Resin adsorption/desorption: spike extracts across the octanol/water partition coefficient (logP) range −1 to 5; compare static versus duty-cycled flow; elute with methanol/ethyl acetate; use liquid chromatography–mass spectrometry (LC-MS) recovery to tune resin mass [
20,
21,
22]. Vacuum perturbation: monitor partial pressure of oxygen (pO₂) at the culture interface with a fiber-optic micro-sensor during cycling; aim for change in partial pressure of oxygen (ΔpO₂) less than 5% of ambient per cycle. Membrane sealing of culture holes: dye penetration and leak-rate checks under ±20 mbar; target no visible ingress/egress and less than 1% mass transfer in 10 minutes. Fouling evaluation (contingent): challenge with particles under 63 µm to induce about 30% flow drop, add a clip-on 5–10 µm polyamide/PP mesh prefilter, and verify at least 80% flow recovery without backflush. Monoclonality verification: seed a tracer strain at λ = 0.1/0.2/0.3; plate-out to estimate P(≥2) versus Poisson predictions and refine standard operating procedures (SOPs).
Figure 1.
Illustrative exploded schematic of the NP-TRAP device highlighting its main conceptual components. The top layer allows passive influx of nutrients and substrate-derived molecules from the environment and remains unsealed by membrane. The second layer contains 56 hexagonal cultivation chambers arranged in a honeycomb pattern and is separated from the top layer by a 0.2 µm microfiltration (cell-excluding) membrane, enabling molecular diffusion while excluding cells. The third layer, designed for metabolite capture, is also separated by a 0.2 µm microfiltration membrane and houses adsorbent resins or compound-selective membranes. A unidirectional diaphragm layer with duckbill-type silicone check-valves, beneath an additional 0.2 µm microfiltration membrane, prevents reflux. All layers are aligned through peripheral orifices and secured using stainless steel rods and nuts. The bottom cap includes a lateral vacuum outlet fitted with a non-return valve for mild suction.
Figure 1.
Illustrative exploded schematic of the NP-TRAP device highlighting its main conceptual components. The top layer allows passive influx of nutrients and substrate-derived molecules from the environment and remains unsealed by membrane. The second layer contains 56 hexagonal cultivation chambers arranged in a honeycomb pattern and is separated from the top layer by a 0.2 µm microfiltration (cell-excluding) membrane, enabling molecular diffusion while excluding cells. The third layer, designed for metabolite capture, is also separated by a 0.2 µm microfiltration membrane and houses adsorbent resins or compound-selective membranes. A unidirectional diaphragm layer with duckbill-type silicone check-valves, beneath an additional 0.2 µm microfiltration membrane, prevents reflux. All layers are aligned through peripheral orifices and secured using stainless steel rods and nuts. The bottom cap includes a lateral vacuum outlet fitted with a non-return valve for mild suction.
Figure 2.
Middle honeycomb-patterned layer of the NP-TRAP device (12 × 12 cm). This component contains 56 uniform hexagonal through-holes (each ~17 mm across and 4 mm deep), with an internal volume of approximately 0.75 mL per well. These orifices are arranged in a honeycomb pattern and support either microbial cultivation or metabolite capture, accommodating solid or semisolid growth media, adsorbent resins, or imprinted membranes. The hexagonal geometry improves surface-to-volume ratio, molecular diffusion efficiency, and spatial compartmentalization. Peripheral alignment holes facilitate precise stacking and sealing of layers.
Figure 2.
Middle honeycomb-patterned layer of the NP-TRAP device (12 × 12 cm). This component contains 56 uniform hexagonal through-holes (each ~17 mm across and 4 mm deep), with an internal volume of approximately 0.75 mL per well. These orifices are arranged in a honeycomb pattern and support either microbial cultivation or metabolite capture, accommodating solid or semisolid growth media, adsorbent resins, or imprinted membranes. The hexagonal geometry improves surface-to-volume ratio, molecular diffusion efficiency, and spatial compartmentalization. Peripheral alignment holes facilitate precise stacking and sealing of layers.
Figure 3.
Diaphragm interface integrated into the top of the bottom cap. Each hexagonal compartment is fitted with a central duckbill-type silicone valve to ensure unidirectional flow and prevent backflow or cross-contamination. This configuration enables passive or suggested mild vacuum-assisted metabolite transport from the cultivation chambers into the capture layer.
Figure 3.
Diaphragm interface integrated into the top of the bottom cap. Each hexagonal compartment is fitted with a central duckbill-type silicone valve to ensure unidirectional flow and prevent backflow or cross-contamination. This configuration enables passive or suggested mild vacuum-assisted metabolite transport from the cultivation chambers into the capture layer.
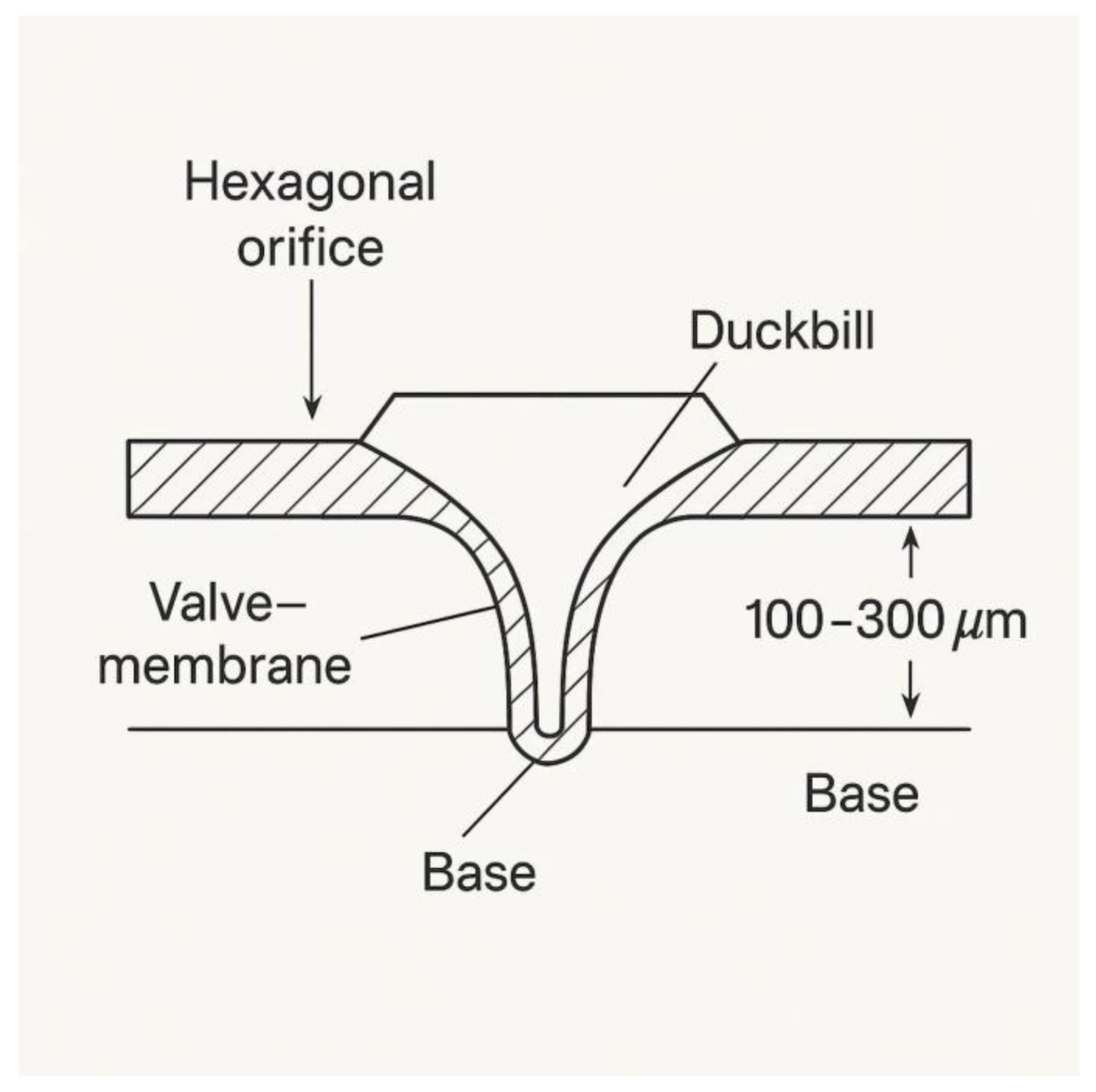
Figure 4.
Cross-sectional schematic of a duckbill silicone valve integrated into each hexagonal orifice on the upper surface of the vacuum outlet cap of the NP-TRAP device. This valve layer is positioned beneath the microfiltration membrane adhered to the bottom of the metabolite capture chamber. The elastomeric element (~100–300 µm thick) is molded into a unidirectional duckbill shape, allowing metabolite flow toward the bottom cap and connected vacuum line, while preventing backflow into the capture zone.
Figure 4.
Cross-sectional schematic of a duckbill silicone valve integrated into each hexagonal orifice on the upper surface of the vacuum outlet cap of the NP-TRAP device. This valve layer is positioned beneath the microfiltration membrane adhered to the bottom of the metabolite capture chamber. The elastomeric element (~100–300 µm thick) is molded into a unidirectional duckbill shape, allowing metabolite flow toward the bottom cap and connected vacuum line, while preventing backflow into the capture zone.