Submitted:
02 September 2025
Posted:
03 September 2025
You are already at the latest version
Abstract
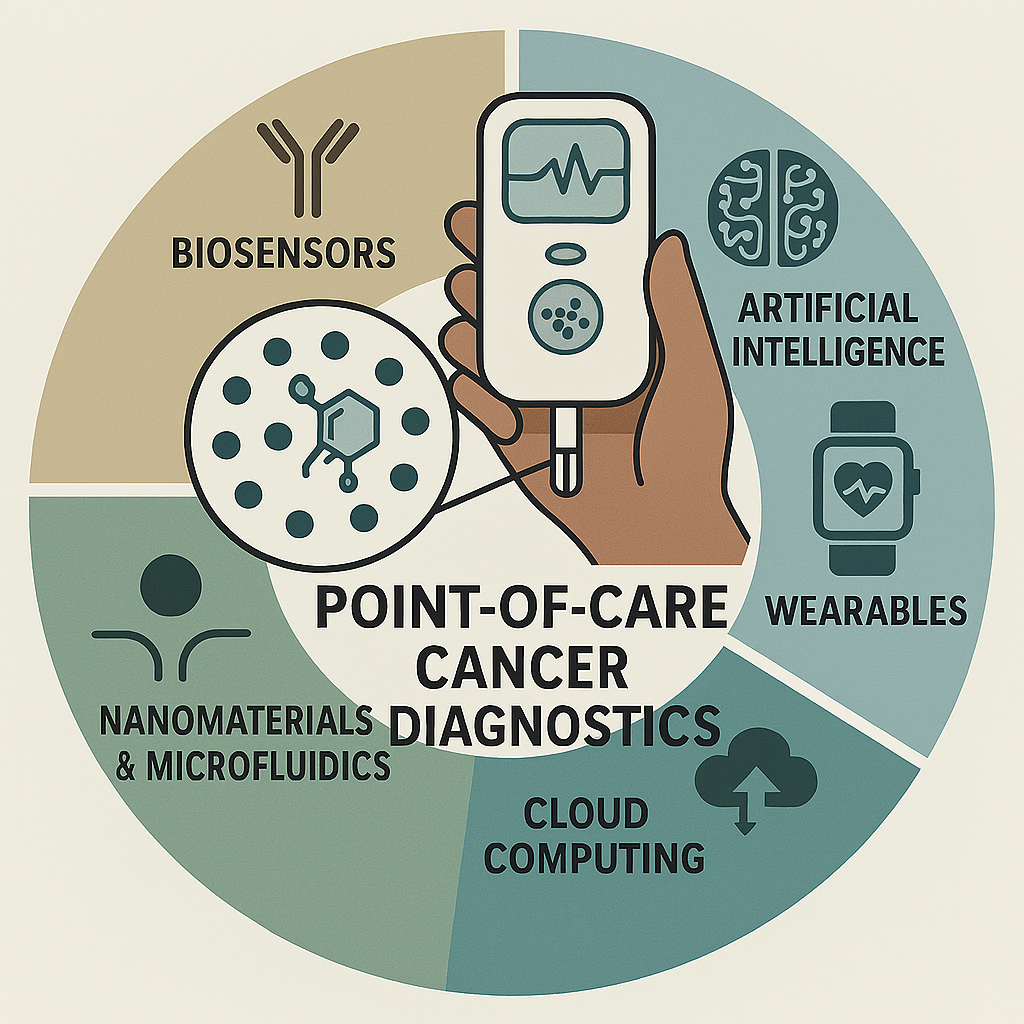
Keywords:
INTRODUCTION
DIGITAL HEALTH INTEGRATION AND DATA-DRIVEN ONCOLOGY DIAGMOSTICS
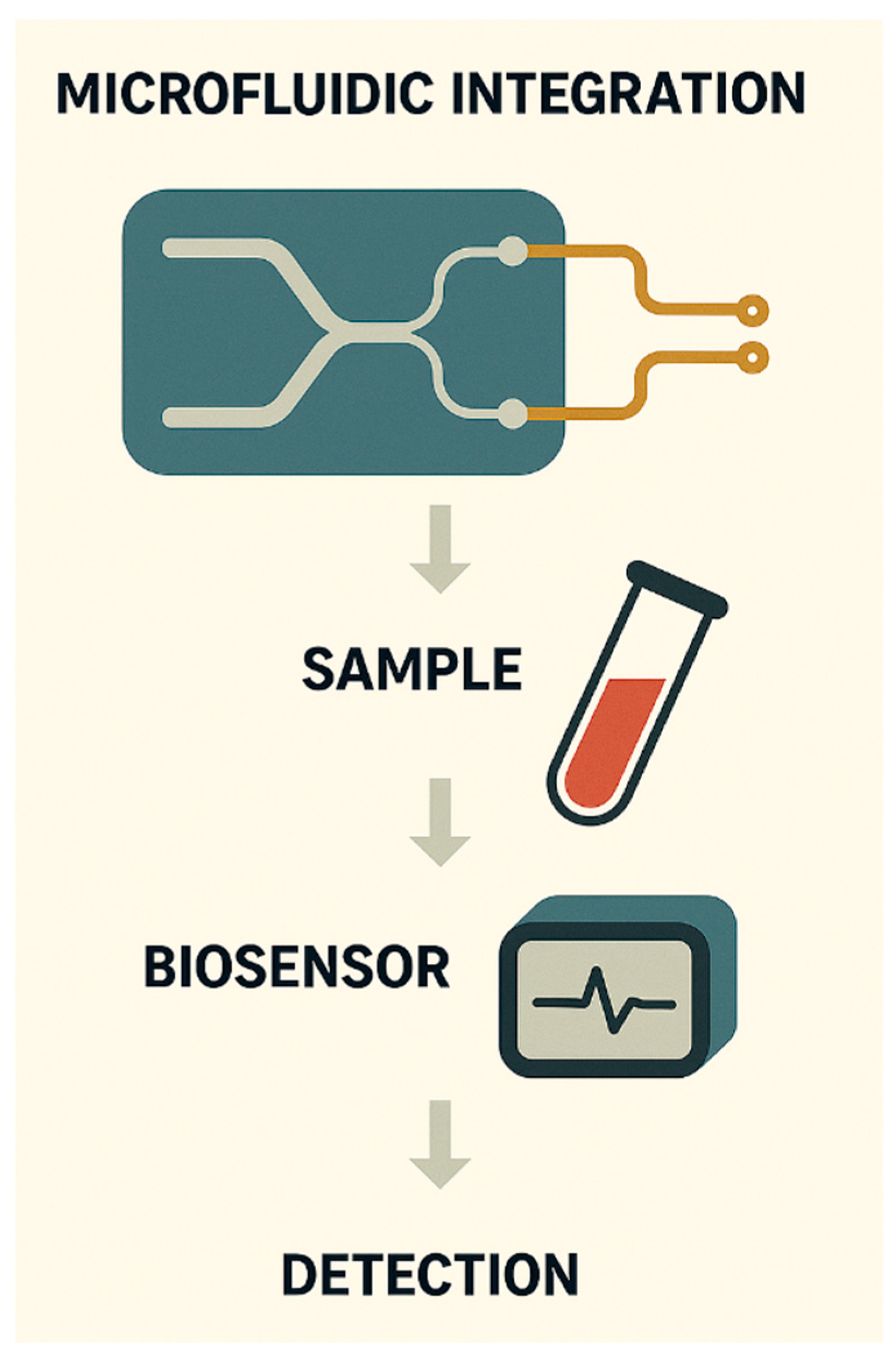
MICROFABRICATION AND DEVICE MINIATURISATION FOR DEPLOYABLE CANCER DIAGNOSTICS
CONNECTED DIAGNOSTICS AND INTELLIGENT DECISION SUPPORT IN PRECISION ONCOLOGY
PATHWAYS TO SCALABLE IMPLEMENTATION AND GLOBAL HEALTH INTEGRATION
IMPLEMENTATION STRATEGIES FOR LOW-RESOURCE AND DECENTRALISED SETTINGS
ETHICAL, LEGAL, AND DATA COVERNANCE CONSIDERATIONS IN DECEMTRALISED CANCER DIAGMOSTICS
ETHICAL IMPERATIVES: EQUITY, AUTONOMY, AND CONSENT
LEGAL AND REGULATORY FRAMEWORKS
DATA GOVERNANCE AND CYBERSECURITY
ETHICAL AI and ALGORITHMETIC TRANSPARENCY
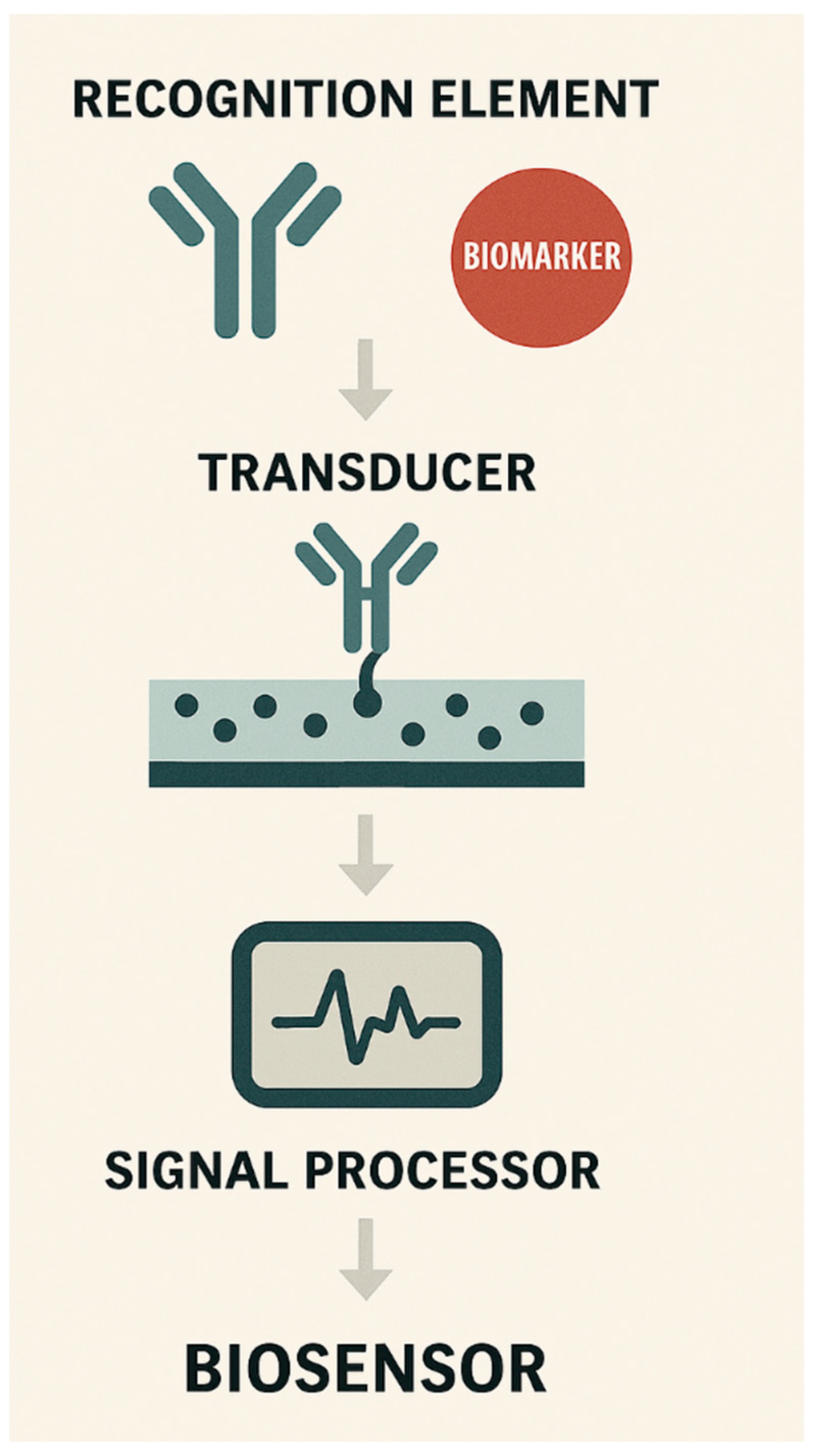
INNOVATION TRAJECTORIES: AI-ENHANCED, WEARABLE, and MULIPLEXED BIOSENSING PLATFORMS
AI-ENHANCED BIOSENSING AND PREDICTIVE ANALYTICS
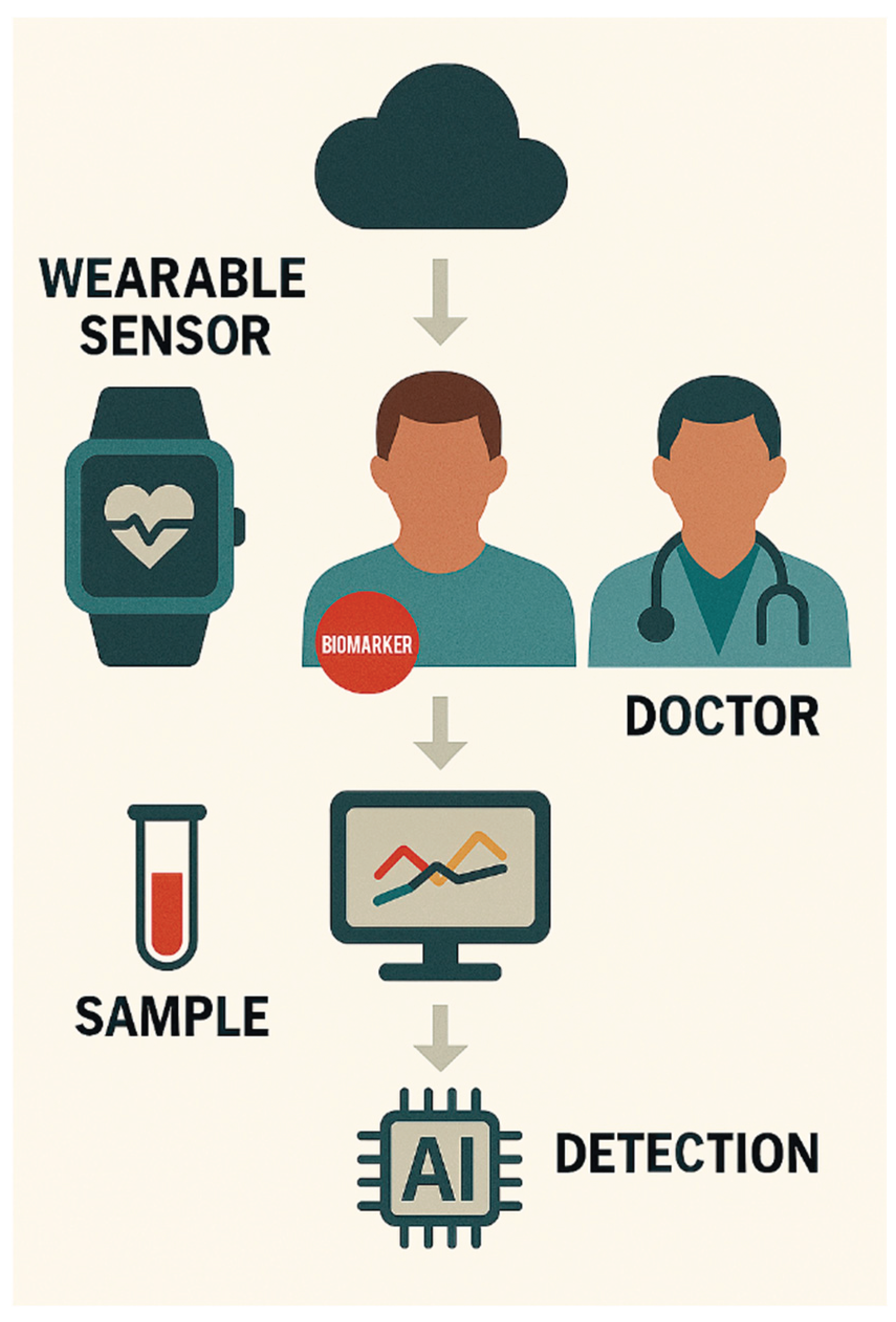
WEARABLE BIOSENSORS FOR CONTINOUS CANCER MONITORING
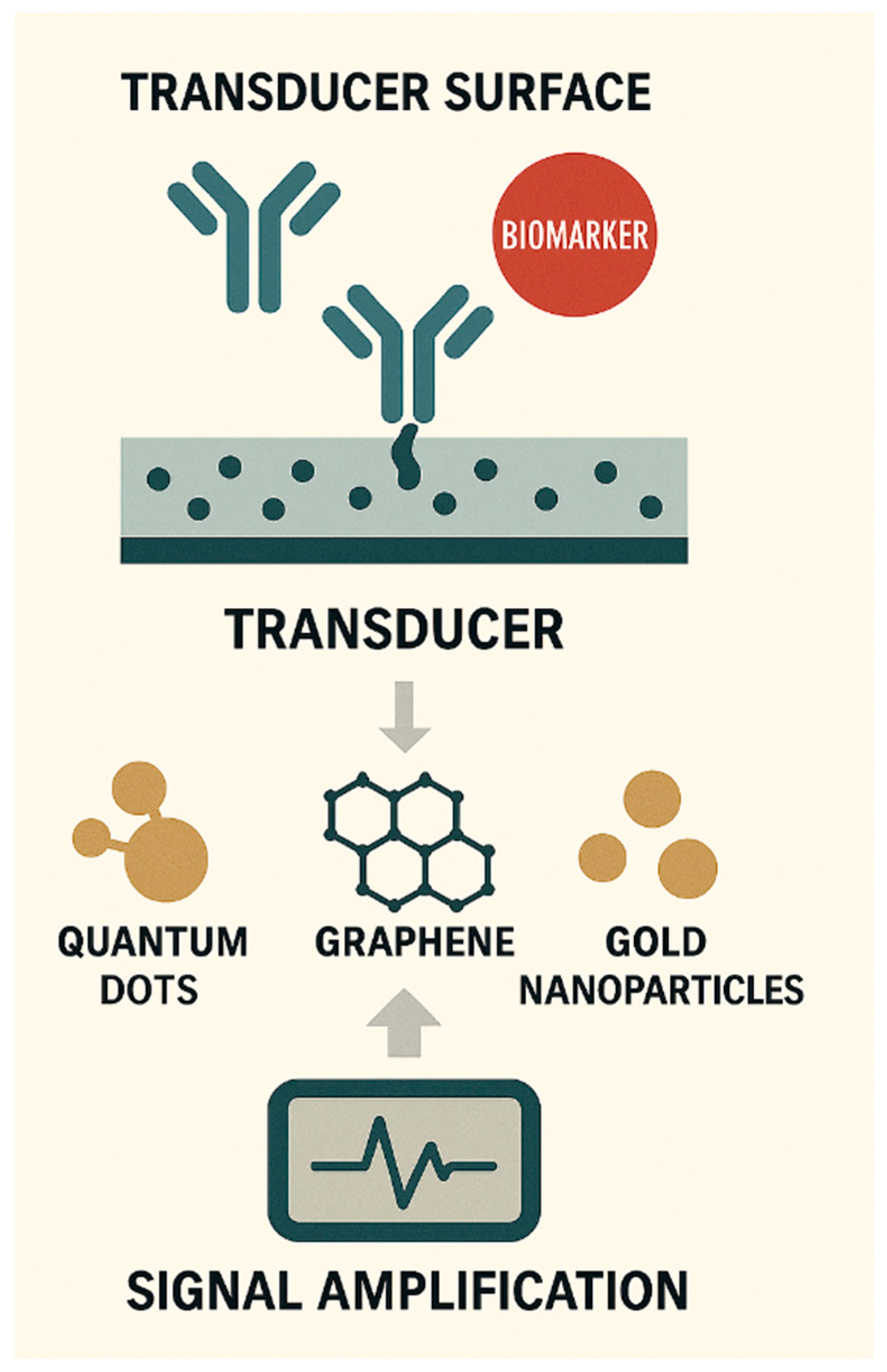
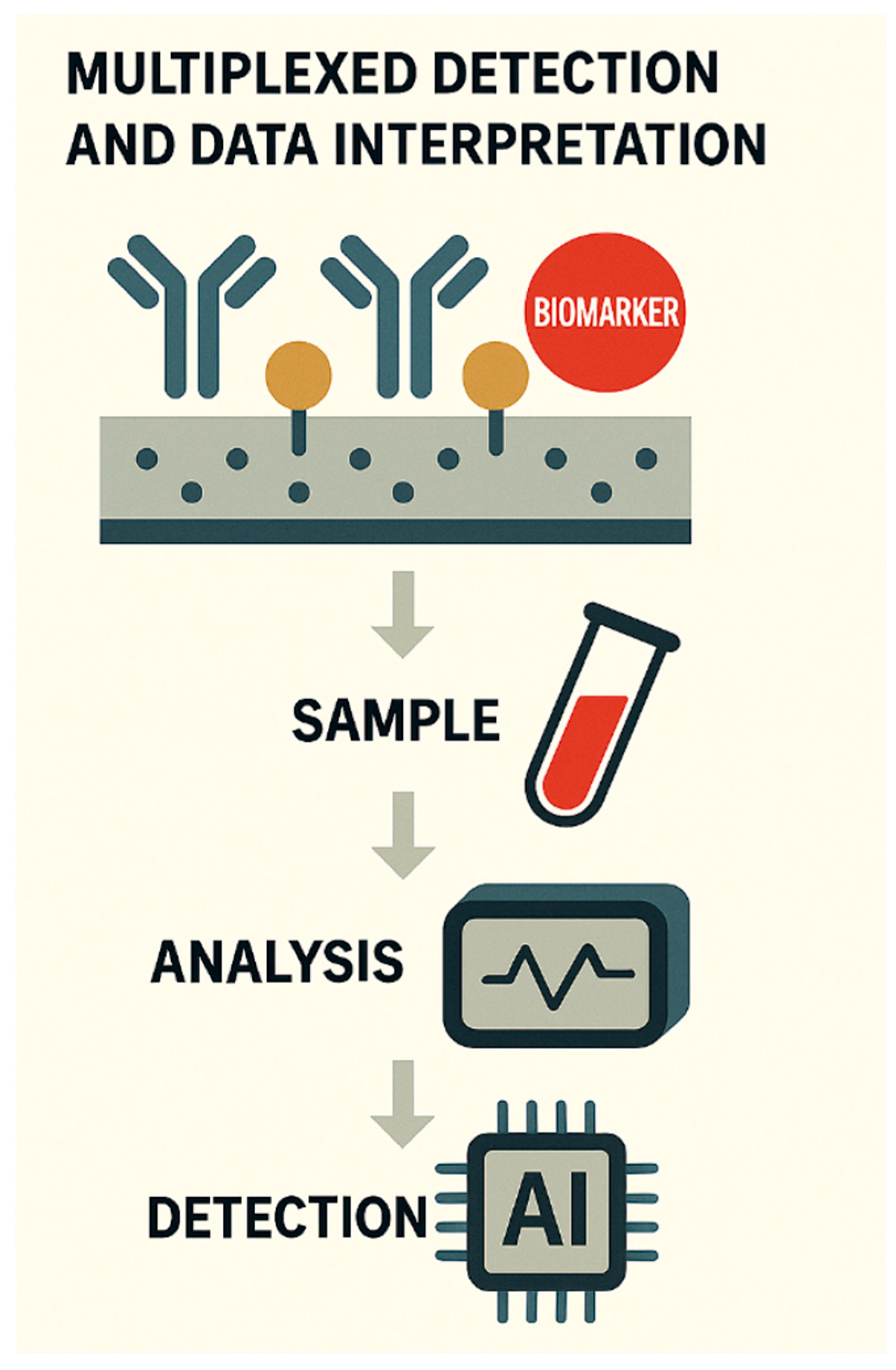
MULTIPLEXED DETECTION AND INTEGRATED ASSAY PLATFORMS
MODULAR AND RECONFIGURABLE BIOSENSOR ARCHITECTURES
CLINICAL VALIDATION AND TRANSLATIONAL READINESS OF BIOSENSOR TECHNOLOGIES
MULTI-PHASE VALIDATION FRAMEWORKS
BIOLOGICAL MATRIX CHALLENGES AND SAMPLE DIVERSITY
CLINICAL WORKFLOW INTEGRATION AND USER ACCEPTANCE
REGULATORY ALIGNMENT AND POST-MARKET SURVEILLANCE
GLOBAL HEALTH EQUITY AND POLICY ALIGNMENT IN BIOSENSOR DEPLOYMENT
BRIDGING THE URBAN-RURAL DIAGNOSTIC DIVIDE
POLICY FRAMEWORKS AND HEALTH SYSTEM INTEGRATION
COMMUNITY ENGAGEMENT AND PATIENT-CENTRIC DESIGN
FINANCING MODELS AND SUSTAINABLE ACCESS
PATIENT-CENTRIC DESIGN AND DIGITAL HEALTH INTEGRATION
USER EXPERIENCE (UX) AND HUMAN-CENTRED DESIGN
- Minimal sample requirements (e.g., finger-prick blood, saliva swabs)
- Real-time feedback with visual cues or haptic alerts
- Multilingual interfaces and accessibility features for vision or motor impairments
- Privacy-first architecture to protect sensitive health data
DIGITAL CONNECTIVITY AND CLOUD-BASED ANALYTICS
- Automated result interpretation using embedded AI algorithms
- Longitudinal tracking of biomarker trends and treatment response
- Teleconsultation triggers based on abnormal readings
- Integration with wearables for multimodal health insights
AI-DRIVEN DECISION SUPPORT AND PERSONALISATION
- Distinguish signal from noise in complex biological matrices
- Predict disease progression using machine learning models trained on historical data
- Recommend tailored interventions based on patient phenotype and biomarker profile
- Flag anomalies for clinician review, reducing diagnostic burden
CONTINUITY OF CARE AND BEHAVIOURAL ENGAGEMENT
- Integration with EMRs for seamless documentation
- Alerts and reminders to promote adherence and follow-up
- Gamification elements to encourage regular use and health engagement
- Peer support features to foster community and reduce isolation
FUTURE DIRECTIONS AND CONVERGENCE TRENDS IN BIOSENSOR-ENABLED ONCOLOGY
NANOTECHNOLOGY AND MULTIMODAL BIOSENSING
- Multiplexed detection of cancer biomarkers within a single assay
- Enhanced signal amplification for ultra-low concentration targets
- Targeted delivery and theranostics, where diagnostic and therapeutic functions are combined
ORGANOID-BASED VALIDATION AND PERSONALISED MODELS
- Functional testing of biosensor performance in physiologically relevant conditions
- Personalised calibration based on individual tumour biology
- Drug response prediction and biomarker discovery in tandem with biosensor readouts
PRECISION ONCOLOGY AND OMICS INTEGRATION
- Dynamic risk stratification and early detection of aggressive subtypes
- Real-time monitoring of treatment efficacy and resistance mechanisms
- Adaptive treatment algorithms powered by AI and systems biology
INTEROPERABLE ECOSYSTEMS AND GLOBAL COLLABORATION
- Open-source diagnostic frameworks for rapid deployment in LMICs
- Federated learning models that protect patient privacy while enabling global AI training
- Cross-border regulatory harmonisation to accelerate approval and distribution
DISCUSSION: TRANSLATIONAL BIOSENSOR PLATFORMS FOR CANCER DIAGNOSTICS AND GLOBAL HEALTH EQUITY
FUTURE DIRECTIONS AND REFERENCE FRAMEWORK
- Example: CRISPR-based biosensors have been developed for rapid detection of EGFR mutations in lung cancer, enabling real-time monitoring of therapy resistance [152]. This study demonstrates the use of CRISPR/Cas9 to engineer lung cancer cell lines with the EGFR T790M resistance mutation. It validates CRISPR as a precise tool for modeling therapy resistance and lays the groundwork for biosensor platforms capable of real-time mutation tracking and therapeutic response monitoring.
- Published Work: Biju et al. [153] reviewed the superiority of 3D culture models in drug screening and biosensor validation, highlighting their role in mimicking in vivo tumor architecture.
- Example: Patient-derived breast cancer spheroids have been used to test biosensor sensitivity to HER2 expression gradients [154].
- Example: The FDA-approved wearable biosensor for cardiac arrhythmia detection (e.g., Zio Patch) sets a precedent for oncology-focused devices [155].
- Example: Deep learning models trained on biosensor outputs have predicted chemotherapy response in colorectal cancer with >85% accuracy [160].
CONCLUSION AND OUTLOOK
Author Information
Acknowledgments
Conflict of Interest
References
- Aguiar-Ibáñez, R.; Mbous, Y.P.V.; Sharma, S.; Chawla, E. Assessing the Clinical, Humanistic, and Economic Impact of Early Cancer Diagnosis: A Systematic Literature Review. Front. Oncol. 2025, 15, 1546447. [CrossRef]
- Zafar, S.; Hafeez, A.; Shah, H.; Mutiullah, I.; Ali, A.; Khan, K.; et al. Emerging Biomarkers for Early Cancer Detection and Diagnosis: Challenges, Innovations, and Clinical Perspectives.Eur. J. Med. Res. 2025, 30, 760. [CrossRef]
- Hajjar, A.; Ergun, M.A.; Alagoz, O.; Rampurwala, M. Cost-effectiveness of adjuvant paclitaxel and trastuzumab for early-stage node-negative, HER2-positive breast cancer.PLOS ONE 2019, 14(6): e0217778. [CrossRef]
- Siravegna, G.; Marsoni, S.; Siena, S.; Bardelli, A. Integrating liquid biopsies into the management of cancer. Nature Reviews Clinical Oncology 2019, 16, 409–418.
- Savas, I.N.; Coskun, A.The Future of Tumor Markers: Advancing Early Malignancy Detection Through Omics Technologies, Continuous Monitoring, and Personalized Reference Intervals.Biomolecules 2025, 15(7), 1011. [CrossRef]
- Bamodu, O.A.; Chung, C.-C.Cancer Care Disparities: Overcoming Barriers to Cancer Control in Low- and Middle-Income Countries.JCO Glob. Oncol. 2024, 10, e2300439. [CrossRef]
- Weigl, B.H.; Neogi, T.; McGuire, H. Point-of-Care Diagnostics in Low-Resource Settings and Their Impact on Care in the Age of the Noncommunicable and Chronic Disease Epidemic.J. Lab. Autom. 2014, 19(3), 248–257. [CrossRef]
- Uddin, M.K.S.; Hossain, M.D.; Hassan, K.M.R.; Rahman, M.H.Breaking Boundaries: Revolutionizing Healthcare with Agile Methodology.World Journal of Advanced Engineering Technology and Sciences 2024, 13(2), 346–354. [CrossRef]
- Moetlhoa, B.; Nxele, S.R.; Maluleke, K.; Mathebula, E.; Marange, M.; Chilufya, M.; et al.Barriers and Enablers for Implementation of Digital-Linked Diagnostics Models at Point-of-Care in South Africa: Stakeholder Engagement. BMC Health Serv. Res. 2024, 24, 216. [CrossRef]
- Gao, W.; Manning, J.C.; Devaraj, K.; Richards-Kortum, R.R.; McFall, S.M.; Murphy, R.L.; et al. Emerging Trends in Point-of-Care Technology Development for Oncology in Low- and Middle-Income Countries. JCO Glob. Oncol. 2025, 11, e2500142.
- Bayuomi, M.M.; Siraj, M.M.; AlHassani, A.T.; Abunar, A.A.H.; Alahmadi, R.F.; et al.Point-of-Care Testing (POCT) and Its Impact on Turnaround Time and Patient Care: A Comprehensive Literature Review. Biomed J Sci & Tech Res. 2025, 61(5), BJSTR.MS.ID.009665.
- Applegate, T.L.; Causer, L.M.; Gowa, I.; Alternetti, N.; Anderson, L.; et al. Paving the Way for Quality Assured, Decentralised Point-of-Care Testing for Infectious Disease in Primary Care: Real World Lessons from Remote Australia. Expert Rev. Mol. Diagn. 2024, 24(3), 2403091. [CrossRef]
- Yammouri, G.; Ait Lahcen, A.AI-Reinforced Wearable Sensors and Intelligent Point-of-Care Tests.J. Pers. Med. 2024, 14(11), 1088. [CrossRef]
- Parihar, M.; Niharika, W.N.; Sahana; Biswas, R.; Dehury, B.; Mazumder, N. Point-of-Care Biosensors for Infectious Disease Diagnosis: Recent Updates and Prospects.RSC Adv. 2025, 36, D5RA03897A.
- Shephard, M.; Shephard, A.; Matthews, S.; Andrewartha, K. The Benefits and Challenges of Point-of-Care Testing in Rural and Remote Primary Care Settings in Australia. Arch. Pathol. Lab. Med. 2020, 144(11), 1372–1380. [CrossRef]
- Ofori, B.; Twum, S.; Nkansah Yeboah, S.; Ansah, F.; Amofa Nketia Sarpong, K. Towards the Development of Cost-Effective Point-of-Care Diagnostic Tools for Poverty-Related Infectious Diseases in Sub-Saharan Africa. PeerJ 2024, 12, e17198. [CrossRef]
- Wang, X.; Li, F.; Guo, Y.; Xu, W. Recent Trends in Nanomaterial-Based Biosensors for Point-of-Care Testing. Front.Chem. 2020, 8, 586702. [CrossRef]
- Roy, S.; Kumar, R.; Acooli, A.; Chatterjee, A.; Tripathy, S.K.; et al. Transforming Nanomaterial Synthesis through Advanced Microfluidic Approaches: A Review on Accessing Unrestricted Possibilities. J. Compos. Sci. 2024, 8(10), 386. [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21(4), 1109. [CrossRef]
- Lin, J.; Chen, Y.; Liu, X.; Jiang, H.; Wang, X. Engineered Intelligent Electrochemical Biosensors for Portable Point-of-Care Diagnostics. Chemosensors 2025, 13(4), 146. [CrossRef]
- Saylan, Y.; Özgür, E.; Denizli, A. Recent Advances of Medical Biosensors for Clinical Applications. Med Devices Sens. 2021, 4, e10129. [CrossRef]
- Ghazizadeh, E.; Naseri, Z.; Deigner, H.-P.; Rahimi, H.; Altintas, Z. Approaches of Wearable and Implantable Biosensors Towards Developing Precision Medicine. Front. Med. 2024, 11, 1390634. [CrossRef]
- Islam, T.; Washington, P. Non-Invasive Biosensing for Healthcare Using Artificial Intelligence: A Semi-Systematic Review. Biosensors 2024, 14(4), 183. [CrossRef]
- Flynn, C.D.; Chang, D. Artificial Intelligence in Point-of-Care Biosensing: Challenges and Opportunities. Diagnostics 2024, 14, 1100. [CrossRef]
- Mishra, A.; Singh, P.K.; Chauhan, N.; Roy, S.; Tiwari, A.; Gupta, S.; et al. Emergence of Integrated Biosensing-Enabled Digital Healthcare Devices. Sensors & Diagnostics 2024, 3, D4SD00017J. [CrossRef]
- Parihar, M.; Niharika, W.N.; Sahana; Biswas, R.; Dehury, B.; Mazumder, N. Point-of-care biosensors for infectious disease diagnosis: recent updates and prospects. RSC Advances 2025, Issue 36, Article D5RA03897A. [CrossRef]
- Gruson, D.; Cobbaert, C.; Dabla, P.K.; Stankovic, S.; Homsak, E.; Kotani, K.; Assaad, R.S.; Nichols, J.H.; Gouget, B.Validation and Verification Framework and Data Integration of Biosensors and In Vitro Diagnostic Devices: A Position Statement of the IFCC Committee on Mobile Health and Bioengineering in Laboratory Medicine.Clin. Chem. Lab. Med. 2024, 62(5), e20231455. [CrossRef]
- Liu, M.; Yang, W.; Zhu, W.; Yu, D.Innovative Applications and Research Advances of Bacterial Biosensors in Medicine.Front. Microbiol. 2025, 16, 1507491. [CrossRef]
- Mpofu, K.T.; Mthunzi-Kufa, P. Recent Advances in Artificial Intelligence and Machine Learning Based Biosensing Technologies. IntechOpen 2025. [CrossRef]
- Al-Tashi, Q.; Saad, M.B.; Muneer, A.; et al. Machine Learning Models for the Identification of Prognostic and Predictive Cancer Biomarkers: A Systematic Review. Int. J. Mol. Sci. 2023, 24(9), 7781. [CrossRef]
- Rawlings, J.; Kufuor, J. Evaluating the Impact of Mobile Health Applications on Patient Engagement and Health Management. Int. J. Med. Info. & Knowledge Insights 2025, 1(1), 4. [CrossRef]
- Shah, N.M.; Meric-Bernstam, F. The Present and Future of Precision Oncology and Tumor-Agnostic Therapeutic Approaches. The Oncologist 2025, 30(6), oyaf152. [CrossRef]
- Kulkarni, M.B.; Ayachit, N.H.; Aminabhavi, T.M. Biosensors and Microfluidic Biosensors: From Fabrication to Application. Biosensors 2022, 12(7), 543. [CrossRef]
- Pandey, Y.; Singh, S.P. Recent Advances in Bio-MEMS and Future Possibilities: An Overview. J. Inst. Eng. India Ser. B 2023, 104, 1377–1388. [CrossRef]
- Bai, H.; Hu, J.; Liu, T.; et al. A Sample-to-Answer Digital Microfluidic Multiplexed PCR System for Syndromic Pathogen Detection. Lab Chip 2025, 25(6), D4LC00704B. [CrossRef]
- Liu, D.; Wang, J.; Wu, L.; et al. Trends in Miniaturized Biosensors for Point-of-Care Testing. TrAC Trends Anal. Chem. 2020, 122, 115701. [CrossRef]
- Xu, Z.; Hao, Y.; Luo, A.; Jiang, Y. Technologies and Applications in Wireless Biosensors for Real-Time Health Monitoring. Med-X 2024, 2, 24. [CrossRef]
- Yuan, X.; Ouaskioud, O.; Yin, X.; et al. Epidermal Wearable Biosensors for Continuous Monitoring of Biomarkers in Interstitial Fluid. Micromachines 2023, 14(7), 1452. [CrossRef]
- Xu, J.; Fang, Y.; Chen, J. Wearable Biosensors for Non-Invasive Sweat Diagnostics. Biosensors 2021, 11(8), 245. [CrossRef]
- Docherty, N.; Macdonald, D.; Gordon, A.; et al. Maximising the Translation Potential of Electrochemical Biosensors. Chem. Commun. 2025, DOI: . [CrossRef]
- Panigrahi, C.R.; Sarkar, J.L.; Pati, B.; Buyya, R.; Mohapatra, P.; Majumder, A. Mobile Cloud Computing and Wireless Sensor Networks: A Review, Integration Architecture, and Future Directions. IET Netw. 2021, 10, 141–161. [CrossRef]
- Dinh-Le, C.; Chuang, R.; Chokshi, S.; Mann, D. Wearable Health Technology and Electronic Health Record Integration: Scoping Review and Future Directions. JMIR Mhealth Uhealth 2019, 7(9), e12861. [CrossRef]
- Alzghaibi, H. Nurses’ Perspectives on AI-Enabled Wearable Health Technologies: Opportunities and Challenges in Clinical Practice. BMC Nurs. 2025, 24, 799. [CrossRef]
- Liu, Y.; Wang, B. Advanced Applications in Chronic Disease Monitoring Using IoT Mobile Sensing Device Data, Machine Learning Algorithms and Frame Theory: A Systematic Review. Front. Public Health 2025, 13, 1510456. [CrossRef]
- Madrid, R.E.; Ramallo, F.A.; Barraza, D.E.; Chaile, R.E. Smartphone-Based Biosensor Devices for Healthcare: Technologies, Trends, and Adoption by End-Users. Bioengineering 2022, 9(3), 101. [CrossRef]
- Munos, B.; Baker, P.C.; Bot, B.M.; et al. Mobile Health: The Power of Wearables, Sensors, and Apps to Transform Clinical Trials. Ann. N.Y. Acad. Sci. 2025, doi: . [CrossRef]
- Wasilewski, T.; Kamysz, W.; Gębicki, J. AI-Assisted Detection of Biomarkers by Sensors and Biosensors for Early Diagnosis and Monitoring. Biosensors 2024, 14(7), 356. [CrossRef]
- Raji, H.; Tayyab, M.; Sui, J.; Mahmoodi, S.R.; Javanmard, M. Biosensors and Machine Learning for Enhanced Detection, Stratification, and Classification of Cells. Biomed. Microdevices 2022, 24, 26. [CrossRef]
- Liu, B.; Liu, S.; Cao, Z.; Zhang, J.; Pu, X.; Yu, J. Accurate Classification of Benign and Malignant Breast Tumors in Ultrasound Imaging with an Enhanced Deep Learning Model.Front. Bioeng. Biotechnol. 2025, 13, 1526260. [CrossRef]
- Wang, T.; Zang, B.; Kong, C.; Li, Y.; Yang, X.; Yu, Y. Intelligent and Precise Auxiliary Diagnosis of Breast Tumors Using Deep Learning and Radiomics. PLOS ONE 2025, 20(6), e0320732. [CrossRef]
- Idrees, A.K.; Alhussein, D.A.; Harb, H. Energy-Efficient Multisensor Adaptive Sampling and Aggregation for Patient Monitoring in Edge Computing-Based IoHT Networks. Appl. Inform. Sci. 2023, 13(4), 610. [CrossRef]
- Magnano San Lio, R.; Barchitta, M.; Maugeri, A.; et al. Updates on Developing and Applying Biosensors for the Detection of Microorganisms, Antimicrobial Resistance Genes and Antibiotics.Front. Public Health 2023, 11, 1240584. [CrossRef]
- Song, Y.; Min, J.; Gao, W. Wearable and Implantable Electronics: Moving toward Precision Therapy. ACS Nano 2019, 13(12), 12280–12286. [CrossRef]
- Tellechea-Luzardo, J.; Stiebritz, M.T.; Carbonell, P. Transcription Factor-Based Biosensors for Screening and Dynamic Regulation. Front. Bioeng. Biotechnol. 2023, 11, 1118702. [CrossRef]
- Tellechea-Luzardo, J.; Martín Lázaro, H.; Moreno López, R.; Carbonell, P. Sensbio: An Online Server for Biosensor Design. BMC Bioinformatics 2023, 24, 71. [CrossRef]
- Zheng, Z.; Zhu, R.; Peng, I.; Xu, Z.; Jiang, Y. Wearable and Implantable Biosensors: Mechanisms and Applications in Closed-Loop Therapeutic Systems. J. Mater. Chem. B 2024, 12, 8577–8604. [CrossRef]
- Jaime, F.J.; Muñoz, A.; Rodríguez-Gómez, F.; Jerez-Calero, A. Strengthening Privacy and Data Security in Biomedical Microelectromechanical Systems.Sensors 2023, 23(21), 8944. [CrossRef]
- Karim, M.E. Biosensors: Ethical, Regulatory, and Legal Issues. Handbook of Cell Biosensors 2020, Springer. [CrossRef]
- Khan, A.R.; Hussain, W.L.; Shum, H.C.; Hassan, S.U. Point-of-Care Testing: A Critical Analysis of the Market and Future Trends. Front. Lab Chip Technol. 2024, 3, 1394752. [CrossRef]
- Prestedge, J.; Kaufman, C.; Williamson, D.A. Regulation and Governance for the Implementation and Management of Point-of-Care Testing in Australia: A Scoping Review. BMC Public Health 2025, 25, 758. [CrossRef]
- Liu, L.X.; Clegg, S.; Pollack, J. The Effect of Public–Private Partnerships on Innovation in Infrastructure Delivery. Project Manag. J. 2024, 55, 31–49. [CrossRef]
- Srivastav, A.K.; Das, P.; Srivastava, A.K. Future Trends, Innovations, and Global Collaboration. In Biotech and IoT; Springer: Singapore, 2024; pp. 309–398. [CrossRef]
- Tumuhimbise, W.; Theuring, S.; Kaggwa, F.; Musiimenta, A. Enhancing the Implementation and Integration of mHealth Interventions in Resource-Limited Settings: A Scoping Review. Implement. Sci. 2024, 19, 72. [CrossRef]
- Hussain, A.; Abbas, N.; Ali, A. Inkjet Printing: A Viable Technology for Biosensor Fabrication. Chemosensors 2022, 10, 103. [CrossRef]
- Cagnani, G.R.; Ibáñez-Redín, G. Application of Large-Scale Fabrication Techniques for Development of Electrochemical Biosensors. In Advances in Bioelectrochemistry; Springer: Cham, 2022; pp. 91–111. [CrossRef]
- Schmidt Batista, A. Empowering Remote Healthcare with On-Premises Solar-Powered AI Units: Design and Implementation. J. Next-Gen Res. 2025, 1, 4. [CrossRef]
- Power, R.; Ussher, J.M.; Hawkey, A.; et al. Co-Designed, Culturally Tailored Cervical Screening Education with Migrant and Refugee Women in Australia: A Feasibility Study. BMC Women's Health 2022, 22, 353. [CrossRef]
- Mills, D.K.; Nestorova, G.G. Biosensor Development and Innovation in Healthcare and Medical Applications. Sensors 2023, 23, 2717. [CrossRef]
- Mahmoodpour, M.; Kiasari, B.A.; Karimi, M.; et al. Paper-Based Biosensors as Point-of-Care Diagnostic Devices for the Detection of Cancers: A Review of Innovative Techniques and Clinical Applications. Front. Oncol. 2023, 13, 1131435. [CrossRef]
- Beks, H.; Ewing, G.; Charles, J.A.; et al. Mobile Primary Health Care Clinics for Indigenous Populations: A Systematic Scoping Review. Int. J. Equity Health 2020, 19, 201. [CrossRef]
- Justino, C.I.L.; Duarte, A.C.; Rocha-Santos, T.A.P. Recent Progress in Biosensors for Environmental Monitoring: A Review. Sensors 2017, 17, 2918. [CrossRef]
- Toffner, G.; Koff, D.A.; Drossos, A.; et al. A Community-Based Task Shifting Program in 25 Remote Indigenous Communities in Nunavut, Canada. Int. J. Circumpolar Health 2025, 84, 2439119. [CrossRef]
- Farsangi, S.N.; Shahraki, S.K.; Cruz, J.P.; Farokhzadian, J. Designing, Implementing, and Evaluating a Mobile App-Based Cultural Care Training Program.BMC Med. Educ. 2023, 23, 979. [CrossRef]
- Iqbal, M.J.; Javed, Z.; Herrera-Bravo, J.; et al. Biosensing Chips for Cancer Diagnosis and Treatment: A New Wave Towards Clinical Innovation. Cancer Cell Int. 2022, 22, 354. [CrossRef]
- Warfade, T.S.; Dhoke, A.P.; Kitukale, M.D. Biosensors in Healthcare: Overcoming Challenges and Pioneering Innovations for Disease Management. World J. Biol. Pharm. Health Sci. 2025, 21, 350–358. [CrossRef]
- Brunton, G.; Thomas, J.; O’Mara-Eves, A.; et al. Narratives of Community Engagement: A Systematic Review-Derived Conceptual Framework for Public Health Interventions. BMC Public Health 2017, 17, 944. [CrossRef]
- Akintobi, T.H.; Bailey, R.E.; Michener, J.L. Harnessing the Power of Community Engagement for Population Health.Prev. Chronic Dis. 2025, 22, 250189. [CrossRef]
- Feldhaus, I.; Mathauer, I.Effects of Mixed Provider Payment Systems and Aligned Cost Sharing Practices: A Structured Review. BMC Health Serv. Res. 2018, 18, 996. [CrossRef]
- Cohen, J.L. The Enduring Debate Over Cost Sharing for Essential Public Health Tools. JAMA Netw. Open 2019, 2, e199810. [CrossRef]
- Bourgeois, A.; Horrill, T.; Mollison, A.; et al. Barriers to Cancer Treatment for People Experiencing Socioeconomic Disadvantage in High-Income Countries: A Scoping Review. BMC Health Serv. Res. 2024, 24, 670. [CrossRef]
- Dankwa-Mullan, I.; Ndoh, K.; Akogo, D.; et al. Artificial Intelligence and Cancer Health Equity: Bridging the Divide or Widening the Gap. Curr. Oncol. Rep. 2025, 27, 95–111. [CrossRef]
- Adesoye, T.; Katz, M.H.G.; Offodile II, A.C. Meeting Trial Participants Where They Are: Decentralized Clinical Trials as a Patient-Centered Paradigm. JCO Oncol. Pract. 2023, 19, 317. [CrossRef]
- Goldschmitt, M.; Gleim, P.; Mandelartz, S.; et al. Digitalizing Informed Consent in Healthcare: A Scoping Review. BMC Health Serv. Res. 2025, 25, 893. [CrossRef]
- Tsyganova, I.Medical Devices: Pre-market Authorisation. TGA Medical Devices Branch, Presentation, March 2018. TGA Pre-market Guidelines.
- Bassan, S. Data Privacy Considerations for Telehealth Consumers Amid COVID-19. J. Law Biosci. 2020, 7(1), lsaa075. [CrossRef]
- Bernasconi, L.; Şen, S.; Angerame, L.; et al. Legal and Ethical Framework for Global Health Information and Biospecimen Exchange: An International Perspective. BMC Med. Ethics 2020, 21, 8. [CrossRef]
- Piasecki, J.; Dranseika, V.; Vermeulen, E. Ownership of Individual-Level Health Data, Data Sharing, and Data Governance. BMC Med. Ethics 2022, 23, 8. [CrossRef]
- Kim, J.; Kim, H.; Bell, E.; et al. Patient Perspectives About Decisions to Share Medical Data and Biospecimens for Research. JAMA Netw. Open 2019, 2(8), e199550. [CrossRef]
- Ewoh, P.; Vartiainen, T. Vulnerability to Cyberattacks and Sociotechnical Solutions for Health Care Systems: Systematic Review. J. Med. Internet Res. 2024, 26, e46904. [CrossRef]
- Yang, W.; Wang, S.; Cui, H.; et al. A Review of Homomorphic Encryption for Privacy-Preserving Biometrics. Sensors 2023, 23(7), 3566. [CrossRef]
- Afrihyiav, E.; Chianumba, E.C.; Forkuo, A.Y.; et al. Explainable AI in Healthcare: Visualizing Black-Box Models for Better Decision-Making. Multidiscip. Glob. Eng. 2025, 3(1), 1113–1125.
- Bouderhem, R. A Comprehensive Framework for Transparent and Explainable AI Sensors in Healthcare. Eng. Proc. 2024, 82(1), 49. [CrossRef]
- Andreoletti, M.; Haller, L.; Vayena, E.; Blasimme, A. Mapping the Ethical Landscape of Digital Biomarkers: A Scoping Review. PLOS Digit. Health 2024, 3(5), e0000519. [CrossRef]
- Yammouri, G.; Lahcen, A. A. AI-Reinforced Wearable Sensors and Intelligent Point-of-Care Tests. J. Pers. Med. 2024, 14(11), 1088. [CrossRef]
- Al-Tashi, Q.; Saad, M.B.; Muneer, A.; et al. Machine Learning Models for the Identification of Prognostic and Predictive Cancer Biomarkers: A Systematic Review. Int. J. Mol. Sci. 2023, 24(9), 7781. [CrossRef]
- Porr, B.; Daryanavard, S.; Bohollo, L.M.; et al. Real-Time Noise Cancellation with Deep Learning. PLoS ONE 2022, 17(11), e0277974. [CrossRef]
- Mikhail, T.J.; El Shamy, R.; Swillam, M.A.; Li, X. Enhanced Performance of On-Chip Integrated Biosensor Using Deep Learning. Opt. Quant. Electron. 2023, 55, 967. [CrossRef]
- Heikenfeld, J.; Jajack, A.; Rogers, J.; et al. Wearable sensors: modalities, challenges, and prospects. Lab on a Chip 2019, 19(2), 217–248.
- Rabiee, N.; Ahmadi, S.; Fatahi, Y.; et al. Revolutionizing Biosensing with Wearable Microneedle Patches: Innovations and Applications. J. Mater. Chem. B 2025, 13(18), 251–268. [CrossRef]
- Niu, S.; Sun, T.; Wang, M.; et al. Multiple Time Points for Detecting Circulating Tumor DNA to Monitor the Response to Neoadjuvant Therapy in Breast Cancer: A Meta-Analysis. BMC Cancer 2025, 25, 115. [CrossRef]
- Liu, Z.; Yu, B.; Su, M.; et al. Construction of a Risk Stratification Model Integrating ctDNA to Predict Response and Survival in Neoadjuvant-Treated Breast Cancer. BMC Med. 2023, 21, 493. [CrossRef]
- Nan, X.; Wang, X.; Kang, T.; et al. Review of Flexible Wearable Sensor Devices for Biomedical Application. Micromachines 2022, 13(9), 1395. [CrossRef]
- Low, C.A. Harnessing Consumer Smartphone and Wearable Sensors for Clinical Cancer Research. npj Digit. Med. 2020, 3, 140. [CrossRef]
- Jarockyte, G.; Karabanovas, V.; Rotomskis, R.; Mobasheri, A. Multiplexed Nanobiosensors: Current Trends in Early Diagnostics. Sensors 2020, 20(23), 6890. [CrossRef]
- Kumari, S.; Islam, M.; Gupta, A. Paper-Based Multiplex Biosensors for Inexpensive Healthcare Diagnostics: A Comprehensive Review. Biomed. Microdevices 2023, 25, 17. [CrossRef]
- Shanmugasundaram, K. B.; Li, J.; Sina, A. I.; Wuethrich, A.; Trau, M. Toward Precision Oncology: SERS Microfluidic Systems for Multiplex Biomarker Analysis in Liquid Biopsy. Mater. Adv. 2022, 3(4), 1459–1471. [CrossRef]
- Shirgir, B.; Dimililer, K.; Asir, S. Applications of Artificial Intelligence in Biosensors. In Intelligent Informatics, Springer, 2024, pp. 299–315. [CrossRef]
- Lee, H.; Xie, T.; Kang, B.; Yu, X.; Schaffter, S. W.; Schulman, R. Plug-and-Play Protein Biosensors Using Aptamer-Regulated In Vitro Transcription. Nat. Commun. 2024, 15, 51907. [CrossRef]
- Syed, Z. Q.; Samaraweera, S.; Wang, Z.; Krishnan, S. Colorimetric Nano-Biosensor for Low-Resource Settings: Insulin as a Model Biomarker. Sens. Diagn. 2024, 3(8), 1659–1671. [CrossRef]
- D'Alton, L.; Souto, D. E. P.; Punyadeera, C.; Abbey, B.; Voelcker, N. H.; Hogan, C.; Silva, S. M. A holistic pathway to biosensor translation. Sens. Diagn. 2024, 3, 1234–1246. https://pubs.rsc.org/en/content/articlehtml/2024/sd/d4sd00088a.
- Karadurmus, L.; Kaya, S. I.; Ozcelikay, G.; Gumustas, M.; Uslu, B.; Ozkan, S. A. Validation Requirements in Biosensors. In Biosensors, 1st ed.; CRC Press, 2022; pp. 353–382. [CrossRef]
- Gagnier, J. J.; Morgenstern, H.; Altman, D. G.; Berlin, J.; Chang, S.; McCulloch, P.; Sun, X.; Moher, D. Consensus-Based Recommendations for Investigating Clinical Heterogeneity in Systematic Reviews. BMC Med. Res. Methodol. 2013, 13, 106. [CrossRef]
- Rodger, M.; Ramsay, T.; Fergusson, D. Diagnostic Randomized Controlled Trials: The Final Frontier. Trials 2012, 13, 137. [CrossRef]
- Chicea, D.; Nicolae-Maranciuc, A. Metal Nanocomposites as Biosensors for Biological Fluids Analysis. Materials 2025, 18(8), 1809. [CrossRef]
- Salvo, P.; Tedeschi, L. pH Sensors, Biosensors and Systems. Chemosensors 2025, 13(3), 90. [CrossRef]
- Lyu, N.; Hassanzadeh-Barforoushi, A.; Rey Gomez, L. M.; Zhang, W.; Wang, Y. SERS Biosensors for Liquid Biopsy Towards Cancer Diagnosis. Nano Convergence 2024, 11, 22. [CrossRef]
- Samson, S. Biosensors and Bioelectronics: Integrating Biomedical Technologies for Point-of-Care Diagnostics. J. Biomed. Syst. Emerg. Technol. 2023, 10(3), 170.
- Mills, D. K.; Nestorova, G. G. Biosensor Development and Innovation in Healthcare and Medical Applications. Sensors 2023, 23(5), 2717. [CrossRef]
- Or, C. K.; Holden, R. J.; Valdez, R. S. Human Factors Engineering and User-Centered Design for Mobile Health Technology. In Human-Automation Interaction, Springer, 2022, pp. 97–118. [CrossRef]
- Aw, R.; Polizzi, K. Enhancing the Performance of an In Vitro RNA Biosensor Through Iterative Design of Experiments. Biotechnol. Prog. 2025, e70005. [CrossRef]
- Makrani, M. F.; Goyal, A.; Sayeed, S. Y. A Comparative Overview of Global Regulatory Authorities: Ensuring Quality, Safety, and Efficacy in Medicines. Int. Res. J. Pharm. Med. Sci. 2025, 8(2), 150. http://irjpms.com/wp-content/uploads/2025/03/IRJPMS-V8N2P150Y25.pdf.
- Williams, T.; Duffy, T. Regulatory Requirements for Software, AI and Medical Devices. Flinders Digital Health Research Centre, 2024. https://blogs.flinders.edu.au/mdri-news/wp-content/uploads/sites/35/2024/12/click-here-to-view-the-slides.pdf.
- Beltran, N. Real-World Evidence in Post-Marketing Drug Surveillance. Walsh Med. Media 2025, 13(4), 508. https://www.walshmedicalmedia.com/open-access/realworld-evidence-in-postmarketing-drug-surveillance.pdf.
- Brand, N. R.; Qu, L. G.; Chao, A.; Ilbawi, A. M. Delays and Barriers to Cancer Care in Low- and Middle-Income Countries: A Systematic Review. The Oncologist 2019, 24(12), e1371–e1380. [CrossRef]
- Martínez-Pérez, G. Z.; Adetunji, T. A.; Salas Noriega, F. J. L.; et al. Point-of-Care Biochemistry for Primary Healthcare in Low-Middle Income Countries: A Qualitative Inquiry. BMC Prim. Care 2024, 25, 362. [CrossRef]
- PATH Diagnostics Program. Boosting the Local Supply Security of Diagnostics in Low- and Middle-Income Countries. PATH Diagnostics Initiative, 2023. https://www.path.org/who-we-are/programs/diagnostics/boosting-local-supply-security-diagnostics.
- World Health Organization (WHO). Controlling Cancer: A Global Strategy for National Cancer Control Programmes. WHO Technical Report Series, 2023. https://www.who.int/activities/controlling-cancer.
- Davis, A. M.; Engkvist, O.; Fairclough, R. J.; Feierberg, I.; Freeman, A.; Iyer, P. Public-Private Partnerships: Compound and Data Sharing in Drug Discovery and Development. SLAS Discov. 2021, 26(5), 604–619. [CrossRef]
- Conderino, S.; Bendik, S.; Richards, T. B.; et al. The Use of Electronic Health Records to Inform Cancer Surveillance Efforts: A Scoping Review. BMC Med. Inform. Decis. Mak. 2022, 22, 91. [CrossRef]
- Loscalzo, J.Biosensors and Privacy: Navigating Ethical Challenges in Health Monitoring. J. Biosens. Bioelectron. 2024, 15(5), 464.
- Tembo, D.; et al. Effective Engagement and Involvement with Community Stakeholders in the Co-Production of Global Health Research. BMJ 2021, 372, n178. https://www.bmj.com/content/bmj/372/bmj.n178.full.pdf.
- Whitehead, L.; Kirk, D.; Chejor, P.; et al. Interventions, Programmes and Resources That Address Culturally and Linguistically Diverse Consumer and Carers’ Cancer Information Needs: A Mixed Methods Systematic Review. BMC Cancer 2025, 25, 599. [CrossRef]
- Wiil, U. K. Editorial: mHealth and Smartphone Apps in Patient Follow-Up. Front. Med. Technol. 2025, 7, 1644384. [CrossRef]
- Chow, Q.; Ng, J. M.; Biese, K.; McCord, M. J. Technology in Microinsurance: How New Developments Affect the Work of Actuaries. Society of Actuaries, 2019. https://www.soa.org/globalassets/assets/files/resources/research-report/2019/2019-technology-microinsurance.pdf.
- Ball, R. Beyond Capital: How Philanthropy Can Advance Impact Investing. Reichstein Foundation, 2024. https://reichstein.org.au/beyond-capital-how-philanthropy-can-advance-impact-investing.
- Hawkins, N.; Grieve, R. Extrapolation of Survival Data in Cost-Effectiveness Analyses: The Need for Causal Clarity. Med. Decis. Mak. 2017, 37(3), 337–339. [CrossRef]
- Mullaney, T.; Pettersson, H.; Nyholm, T.; Stolterman, E. Thinking Beyond the Cure: A Case for Human-Centered Design in Cancer Care. Int. J. Des. 2012, 6(3), 27–39. https://www.ijdesign.org/index.php/IJDesign/article/view/1076/525.
- Ly, S.; Runacres, F.; Poon, P. Journey Mapping as a Novel Approach to Healthcare: A Qualitative Mixed Methods Study in Palliative Care. BMC Health Serv. Res. 2021, 21, 915. [CrossRef]
- Sarani, M.; Jahangiri, K.; Karami, M.; Honarvar, M. Enhancing Epidemic Preparedness: A Data-Driven System for Managing Respiratory Infections. BMC Infect. Dis. 2025, 25, 159. [CrossRef]
- Eke, C. I.; Shuib, L. The Role of Explainability and Transparency in Fostering Trust in AI Healthcare Systems: A Systematic Literature Review, Open Issues and Potential Solutions. Neural Comput. Appl. 2025, 37, 1999–2034. [CrossRef]
- Venema, T. A. G.; Kroese, F. M.; Benjamins, J. S.; de Ridder, D. T. D. When in Doubt, Follow the Crowd? Responsiveness to Social Proof Nudges in the Absence of Clear Preferences. Front. Psychol. 2020, 11, 1385. [CrossRef]
- Son, M. H.; Park, S. W.; Sagong, H. Y.; Jung, Y. K. Recent Advances in Electrochemical and Optical Biosensors for Cancer Biomarker Detection. BioChip J. 2023, 17, 44–67. [CrossRef]
- Mögele, T.; Hildebrand, K.; Sultan, A.; Sommer, S.; Rentschler, L.; Kling, M.; Sax, I.; Schlesner, M.; Märkl, B.; Trepel, M.; Schmutz, M.; Claus, R. Dissecting Tumor Heterogeneity by Liquid Biopsy—A Comparative Analysis of Post-Mortem Tissue and Pre-Mortem Liquid Biopsies in Solid Neoplasias. Int. J. Mol. Sci. 2025, 26(15), 7614. [CrossRef]
- Bertagnolli, M. M.; Carnival, D.; Jaffee, E. M. Achieving the Goals of the Cancer Moonshot Requires Progress Against All Cancers. Cancer Discov. 2023, 13(5), 1049–1052. [CrossRef]
- Alix-Panabières, C.; Pantel, K. From Discovery to Diagnosis: A Perspective for Circulating Tumor Cells in Personalized Oncology. Cancer Discov. 2025, 15(8), OF1–OF17. [CrossRef]
- Nasiri, R.; Zhu, Y.; de Barros, N. R. Microfluidics and Organ-on-a-Chip for Disease Modeling and Drug Screening. Biosensors 2024, 14(2), 86. [CrossRef]
- Parent, C.; Melayil, K. R.; Zhou, Y.; Aubert, V.; Surdez, D.; Delattre, O.; Wilhelm, C.; Viovy, J.-L. Simple Droplet Microfluidics Platform for Drug Screening on Cancer Spheroids. Lab Chip 2023, 23(24), 4567–4579. [CrossRef]
- Althobiti, M.; Nhung, T. T. T.; Verma, S. K.; Albugami, R. R. Artificial Intelligence and Biosensors: Transforming Cancer Diagnostics. Med. Novel Technol. Devices 2025, 27, 100378. [CrossRef]
- Wei, L.; Niraula, D.; Gates, E. D. H.; Fu, J.; Luo, Y.; Nyflot, M. J.; Bowen, S. R.; El Naqa, I. M.; Cui, S. Artificial Intelligence and Machine Learning in Precision Oncology: Enhancing Discoverability Through Multiomics Integration. Br. J. Radiol. 2023, 96(1150), 20230211. [CrossRef]
- Joshi, A.; Vishnu, A. G. K.; Sakorikar, T.; Kamal, A. M.; Vaidya, J. S.; Pandya, H. J. Recent Advances in Biosensing Approaches for Point-of-Care Breast Cancer Diagnostics: Challenges and Future Prospects. Nanoscale Adv. 2021, 3(14), 5542–5564. [CrossRef]
- Wyatt, A.; Sartor, O.; Lozada, J.; Perrin, D. M. Circulating Tumor DNA Biomarkers in the TheraP Trial: Cabazitaxel vs Lutetium-PSMA Comparison. ASCO GU 2025 Presentation & Post Hoc Analysis. https://www.urotoday.com/video-lectures/mcrpc-treatment/video/mediaitem/4980-ctdna-biomarkers-in-therap-trial-cabazitaxel-vs-lutetium-psma-comparison-alexander-wyatt.html.
- Park, M.-Y.; Jung, M. H.; Eo, E. Y.; Kim, S.; Lee, S. H.; Lee, Y. J.; Park, J. S.; Cho, Y. J.; Chung, J. H.; Kim, C. H.; Yoon, H. I.; Lee, J. H.; Lee, C.-T. Generation of Lung Cancer Cell Lines Harboring EGFR T790M Mutation by CRISPR/Cas9-Mediated Genome Editing. Oncotarget 2017, 8(22), 36331–36338. [CrossRef]
- Biju, V.; Sreenivasan, S.; Nair, R. V.; Thomas, S. Three-Dimensional Cell Culture Models in Cancer Drug Screening and Biosensor Validation: Bridging the Gap Between In Vitro and In Vivo. Biomaterials Science 2024, 12(3), 678–695.
- Huang, Y.; Burns, D. J.; Rich, B. E.; MacNeil, I. A.; Dandapat, A.; Soltani, S. M.; Myhre, S.; Sullivan, B. F.; Lange, C. A.; Furcht, L. T.; Laing, L. G. Development of a Test That Measures Real-Time HER2 Signaling Function in Live Breast Cancer Cell Lines and Primary Cells. BMC Cancer 2017, 17, 199. [CrossRef]
- iRhythm Technologies, Inc. FDA 510(k) Clearance for Zio AT Wearable Cardiac Monitoring System. MedTech Dive, 2024. https://www.medtechdive.com/news/iRhythm-FDA-510k-clearance-Zio-cardiac-monitor/730680/.
- WHO Publishes New Essential Diagnostics List and Urges Countries to Prioritize Investments in Testing. WHO News Release, 2021. WHO Essential Diagnostics List Overview.
- Boosting the Local Supply Security of Diagnostics in Low- and Middle-Income Countries. PATH, 2023. PATH’s Diagnostic Innovation Initiative.
- Kadama-Makanga, P.; Semeere, A.; Laker-Oketta, M.; et al. Usability of a Smartphone-Compatible, Confocal Micro-Endoscope for Cervical Cancer Screening in Resource-Limited Settings. BMC Women's Health 2024, 24, 483. [CrossRef]
- Pradhan, A.; Indian Institute of Technology (IIT) Kanpur. Pilot-Scale Testing of Smartphone-Based Fluorescence Spectroscopic Device for Point-of-Care Diagnosis of Early Cervical Cancer. India Science, Technology & Innovation Portal, DST Project, 2022. https://www.indiascienceandtechnology.gov.in/research/pilot-scale-testing-smartphone-based-fluorescence-spectroscopic-device-point-care-diagnosis-early.
- Kumarganesh, S.; Murugesan, M.; Ganesh, C.; Santhiyakumari, N.; Thangavel, M.; Jayaram, K.; Sagayam, K. M.; Pandey, B. K.; Pandey, D. Hybrid Plasmonic Biosensors with Deep Learning for Colorectal Cancer Detection. Plasmonics 2025, 20, 4895–4909. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



