Submitted:
01 September 2025
Posted:
25 September 2025
You are already at the latest version
Abstract
Objectives: This prospective, monocentric, randomized study examined data from 5500 women underwent transcervical (TC-CVS), transabdominal (TA-CVS) and transvaginal (TV-CVS), by a single operator. We assessed the efficacy of transabdominal CVS compared with transcervical and transvaginal CVS, and examined factors implicated in causing spontaneous abortion. Methods: Over a fifteen year’s period 5500 women underwent chorionic villus sampling at 10-14 weeks of gestation, by transcervical, (850 patients) transabdominal (4500 patients) and transvaginal technique (150 patients).Results: In total 850 (15.5 per cent) patients underwent transcervical CVS (TC-CVS) using a flexible catheter and spontaneous abortion rate after TC-CVS occurred following the procedure in five cases (0.6 per cent). Additionaly,4500 (81.8 per cent) patients underwent transabdominal CVS (TA-CVS) using a 20-gauge spinal needle after which spontaneous abortion occurred in eight women (0.18 per cent). Transvaginal CVS (TV-CVS) was performed in 150 (2.7 per cent) patients using a 20 gauge needle and spontaneous abortion after TV-CVS occurred in two cases (1.3 per cent). In eighty (4.0 per cent) patients’ chromosomal aberration was detected. The rate of spontaneous abortion rate was lower among women who underwent TA-CVS after 13 weeks of gestation. There were no significant differences found in mean pulsatility indices (PIs) between maternal end fetal circulation, before and after CVS procedures, using transvaginal color doppler. Conclusions Transabdominal CVS is associated with a lower fetal loss rate than transcervical and transvaginal CVS.
Keywords:
1. Introduction
2. Material and Methods
3. Results
| N=400 | PI 10 MIN | PI 10 MIN | ||
|---|---|---|---|---|
| BEFORE CVS | AFTER CVS | |||
| Mean | 1SD | Mean | 1 SD | |
| UTERINE ARTERY | 2,10 | 0,55 | 2,07 | 0,58 |
| SPIRAL ARTERY | 0,60 | 0,55 | 0,59 | 0,18 |
|
INTRAPLACENT. ARTERY |
0.48 | 0.15 | 0.50 |
0.16 P>0.01 |
|
UMBILICAL ARTERY |
2.96 | 1.42 | 2.86 |
1.55 P>0.01 |
|
MIDDLE CEREBRAL ARTERY |
1.88 | 0.55 | 1,75 |
0,60 P>0,01 |
| FETAL MALFORMATION N |
AMOUNT OF CHORIONIC TISSUE(MG) |
|---|---|
|
ANENCEPHALY 2 |
20 |
|
HYDROCEPHALUS 2 |
25 |
|
SPINA BIFIDA 2 |
30 |
|
CLEFT-LIP PALATE 1 |
20 |
|
CARDIAC ANOMALY 10 |
30 |
|
OESOPHAGAL ATRESIA 1 |
25 |
|
DIAFRAGMATIC HERNIA 2 |
20 |
|
HYGROMA COLLI MULTILOCULARAE 11 |
30 |
|
OMPHALOCOELE 5 |
25 |
|
RENAL AGENESIS 3 |
15 |
|
CYSTIC KIDNEYS 6 |
20 |
|
HYDROPS UNIVERSALIS 6 |
25 |
|
TANATOTROFYC DYSPLASIA 1 |
15 |
|
POLYDACTILIA 3 |
20 |
|
LIMB REDUCTION 0 |
20 |
|
TOTAL 55(1%) |
20,3 +-9,7 mg |
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Canadian Collaborative CVS, Amniocentesis Clinical Trial Group. Multicenter randomized clinical trial of chorionic villus sampling and amniocentesis. Lancet.1989,1,1-6.
- Jackson and the U. S. NICHD Chorion Villus Sampling and Amniocentesis Study Group. A randomized comparison of transcervical and transabdominal chorionic villus sampling. N Engl J Med. 1992, 327, 594-598.
- Canadian Early and Midtrimester Amniocentesis Trial Group. Randomized trial to assess safety and fetal outcome of early and midtrimester amniocentesis. Lancet. 1998, 351: 242-247.
- American College of Obstetricians and Gynecologists Committae on Practice Bulletins-Obstetrics Committae on Genetics;Society for Maternal-fetal Medicine. Screening for Fetal Chromosomal Abnormaluries-ACGO Practice Bulletin,Number 226,Obstet Gynecol.2020,Oc,136(4):e48-e69.
- Jackson and the U. S. NICHD Chorion Villus Sampling and Amniocentesis Study Group. A randomized comparison of transcervical and transabdominal chorionic villus sampling. N Engl J Med. 1992, 327, 594-598.
- Buijtendijk, M.F.; Bet, B.B.; Leeflang, M.M.; Shah, H.; Reuvekamp, T.; Goring, T.; Docter, D.; Timmerman, M.G.; Dawood, Y.; A Lugthart, M.; et al. et al. Diagnostic accuracy of ultrasound screening for fetal structural abnormalities during the first and second trimester of pregnancy in low-risk and unselected populations. Cochrane Database Syst. Rev. 2024, 5, CD014715.
- International Society of Ultrasound in Obstetrics and Gynecology; Bilardo CM, Chaoui R, Hyett Kagan KO, Karim JN, Papageorghiou AT, Poon LC, Salomon LJ, Syngelaki A, Nicolaides KH.ISUOG Practice Guidelines (up- dated): performance of 11-14-week ultrasound scan. Ultrasound Obstet Gynecol. 2023,61(1),127-143.
- Alamri,L; Ludwingston, A; Kaizer, LK; Hamilton,S; Mary, H; Sammel M; Putra, M. Clinical factors that infuence chorionic villus sampling sample size. Arhives of Gynecology and Obstetrics. 2025, 311, 213-221.
- Liu, S.; Yang, F.; Chang, Q.; Liu, S.; Yang, F.; Chang, Q; Lyan, L; 1,Chen, W; 1,Yin, A;1, Huang F; et al. Positive predictive value estimates for noninvasive prenatal testing from data of a prenatal diagnosis laboratory and literature review. Mol. Cytogenet. 2022, 15, 29–36.9. [CrossRef]
- Podobnik, P; Meštrović,T; Đorđević A; Kurdija,K; Jelčić, Đ; Ogrin,N; Bertović-Žunec,I;Gebauer-Vuković,B;Hoćevar,G;Lončar,I; et al. A Decade of Non-Invasive Prenatal.
- Testing (NIPT) for Chromosomal Abnormalities in Croatia: First National Monocentric Study to Inform Country’s Future Prenatal Care Strategy.Genes.2024,Dec 11,15(12),.
- 1590. [CrossRef]
- Giovannopolou, E; Tsakiridis, I; Mamopoulos,A; Populidis,I; Anastanidis, A; T. Dagklis,T.Invasive Prenatal Diagnostic Testing for Aneuploidies in Singleton Pregnancies:A Cimparative Review of Major Gidlines.Medicina(Kaunas).2022,10, 58-68.
- Crane, JP; Beaver, HA; Cheung, SW. First trimester chorionic villus sampling versus mid-trimester genetic amniocentesis: preliminary results of a controlled prospective trial. Prenat Diagn. 1988, 8, 355-366.
- Navratan,K; Alfirevic, Z; on behaf of the Royal College Of Obstetrician and Gynecologist. Amniocentesis and chorionic villus sampling.Green-top Guidlance No.8 BJOG,2022,129,pp e-1 3-15. [CrossRef]
- Salomon, L.J.; Sotiriadis, A.; Wulff, C.B.; Odibo, A.; Alolekar, R. Risk of miscarriage following amniocentesis or chorionic villus saqmpling:Systematic review of literature and updated meta analysis. Ultrasound Obstet. Gynecol. 2019, 54, 442–451. [CrossRef]
- Alfirevic, Z.; Navaratnam, K.; Mujezinovic, F. Amniocentesis and chorionic villus sampling for prenatal diagnosis. Cochrane Database Syst. Rev. 2017, 9, CD003252. [CrossRef]
- Hui, L; Ellis, D; MayenD; Mark, D; Pertile, M; Reiner, R; Sun, L; Vora N; Lyn, S; Chitty, L. “Position Statement From the International Society for Prenatal Diagnosis on the Use of Non-Invasive Prenatal Testing for the Detection of Fetal Chromosomal Conditions in Singleton Pregnancies”.Penatal Diagnosis. 2023, no. 7 (06 2023): 814– 828. [CrossRef]
- Yeon, LI; Young, K; Sunghun, N; et al.Practise guidelines for prenatal aneuploidy screening and diagnostic testing from Korean Society of maternal-fetal medicine. Invasive diagnostic testing for fetal chromosomal abnormalities. J Korean Med Sci. 2021, 25, 36 (4),326-88.
- Holzgreve, W; Ginsberg, N; Ammala, P; Dumez, Y. Risk of CVS. Prenat. Diagn 1993, 13, 197-209.
- MCR Working Party on the Evaluation of Chorionic Villus Sampling. Medical Research Council European Trial of chorionic villus sampling. Lancet. 1991, 337, 1491-1499. [CrossRef]
- Singer, Z; Profeta, K. Chorionic frondosum cultivation technique in antenatal detection of genetic disease. Period. Biol. 1989, 91, 77-81.
- Podobnik, M; Ciglar, S; Singer Z; Podobnik-Šarkanji, S; Duic, Z; Skalak, D. Transabdominal chorionic villus sampling in the second and third trimester of high-risk pregnancies. Prenatal Diagnosis. 1997,17,125-133.
- Smidt-Jensen, S; Lundsten, C; Lind, AM; Dinesen, K; Philip, J. Transabdominal chorionic villus sampling in the second and third trimester of pregnancy: chromosome quality, reporting time, and feto-maternal bleeding. Prenat Diagn. 1993. 13, 957-969. [CrossRef]
- Tzela,P; Antsaklis,P; Kanellopolus,D; Antonakopoluos, N;Gourounti K. Factor Influencing the Decision-Makinhg Process for Undergoing Invasive Prenatal Testing. Cureus. 2024 Apr;16(4)358803.
- Petousis, S; Sotiriadis, A; Margioula-Siarkou, C;Tsakiridis, I;Christidis P, Kyriakakis M. Detection of structural abnormalities in fetuses with normal karyotype at 11-13 weeks using the anatomic examination protocol of the International Society of Ultrasound in Obstetrics and Gynecology (ISUOG). Journal of Maternal-Fetal & Neonatal Medicine. 2020;33(15):2581-7. [CrossRef]
- Wang, BB; Rubin, CH; Williams II, J. Mosaicism in chorionic villus sampling: an analysis of incidence and chromosomes involved in 2612 consecutive cases. Prenat. Diagn. 1993,13:179-190. [CrossRef]
- Reilly, S. Doyle, S. J. Hamilton, M. D. Kilby, and F. Mone, “Pitfalls of Prenatal Diagnosis Associated With Mosaicism,” Obstetrician and Gynaecologist 25, no. 1 (January 2023): 28–37.
- Liao, Y; Wen, H; Ouyang, S; Guan,Y;,Bi J; Fu, Q; Yang,X; Guo, W; Huang,Y;Zeng,Q;et al. Routine firsttrimester ultrasound screening using a standardized anatomical protocol. American Journal of Obstetrics & Gynecology 2021;224(4):396.e1-396.e15.
- Kagan,KI; Sonek,J;Kozlowski, P.Antenatal screening for chromosomal abnormalities. Arch Gynecol Obstet.2022,13:305(4),825-835. [CrossRef]
- Khalil, A; Sotiriadis, A; D’Antonio, F; Da Silva Costa, F; Odibo, A; Prefumo, F; Papageorghiou, AT; Salomon, LJ. ISUOG Practice Guidelines: perform- ance of third-trimester obstetric ultrasound scan. Ultrasound Obstet Gynecol. 2024Jan; 63(1):131-147. [CrossRef]
- Spingler, J; Sonek, M; Hoopmann, N; Prodan, H; Abele; Kagan,KI. “Complication Rate After Termination of Pregnancy for Fetal Defects,” Ultrasound in Obstetrics and Gynes and Gynecology 62, no. 1 (July 2023): 88–93, . [CrossRef]
- Cohen-Overbeek, TK; Jahoda, MGJ; Wladimiroff, JW. Uterine blood flow velocity waveforms before and after transcervical chorionic villus sampling. Ultrasound in Med.& Biol. 1990, 16, 129-1323.
- 31 Arduini, D; Rizzo, G. Umbilical artery velocity waveforms in early pregnancy: A transvaginal color Doppler study. J. Clin. Ultrasound. 1991,19, 335-339. [CrossRef]
- Podobnik, M.; Ciglar, S.; Singer, Z.; Gebauer, B., Podgajski, M. Doppler assessment of fetal circulation during late chorionic villus sampling in high risk pregnancies. Prenat. Neonatal Med. 1998, 3 (Suppl. S1), 9.
- Maines,J; Montero,FJ. Chorionic villus sampling. InStatPearlsd.Publishing in Theasure Island,FL,USA 2025.
- Akolekar, R; Beta, JL; Piccarelli, G; Olive, C; Dantonio, F. Predictive related risk of miscarriage following amniocentesis and chorionic villus sampling. A systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2015, 45, 16-26. [CrossRef]
- Bakker, M; Birnie, E; Robles de Medina, P; Sollie, KM; Pajkrt, EL; Bilardo, CM. Total pregnancy loss after chorionic villus sampling and amniocentesis: a cohort stud. Ulrasound Obstet gynecol. 2017, 49, 599-606. [CrossRef]
- Ching-Hua, H; Jia-Shing, C; Yu-Ming, S; Yann-Jang,L; Chen Yi-Cheng, Wu. Prenatal diagnosis using chromosomal microarray analysis in high- risk pregnancies. J Clin Med. 2022, 11, 3624-3644.
- Brambati, B; Tului, L; Lislaghi, C; Alberti, E; First 10 000 chorionic villus sampling pereformed on singleton pregnancies by a single operator. Prenatal Diagn. 1998, 3, 255-266.
- Firth, HV; Body, PA; Chamberlain, P; Mackenzie, IZ; Lindenbaum, RH; Huson, SM. Severe limb abnormalities after chorionic villus sampling at 56-66 days gestation. Lancet.1991, 337,127-135. [CrossRef]
- Saura, R; Longy, M; Horovitz, J; Grison, O; Vergaud, A; Taine, LN; et al. Risk of transabdominal chorionic villus sampling before 12 week of amenorrhea. Prenatal diagnosis. 1990, 10, 461-467. [CrossRef]
- Holzgreve, W; Ginsberg, N; Ammala, P; Dumez, Y. Risk of CVS. Prenat. Diagn 1993, 13, 197-209.
- Saura, R; Longy, M; Horovitz, J; Grison, O; Vergaud, A; Taine, LN; et al. Risk of transabdominal chorionic villus sampling before 12 week of amenorrhea. Prenatal diagnosis. 1990, 10, 461-467. [CrossRef]
- Nicolaides, K; Soothil, PW; Rodeck, CH;, Waren, RC; Gosden, CM. Why confine chorionic villus (placental) biopsy to the first trimester. Lancet. 1986b, 1,543-544. [CrossRef]
- Smidt-Jensen, S; Lundsten, C; Lind, AM; Dinesen, K; Philip, J. Transabdominal chorionic villus sampling in the second and third trimester of pregnancy: chromosome quality, reporting time, and feto-maternal bleeding. Prenat Diagn. 1993. 13, 957-969. [CrossRef]
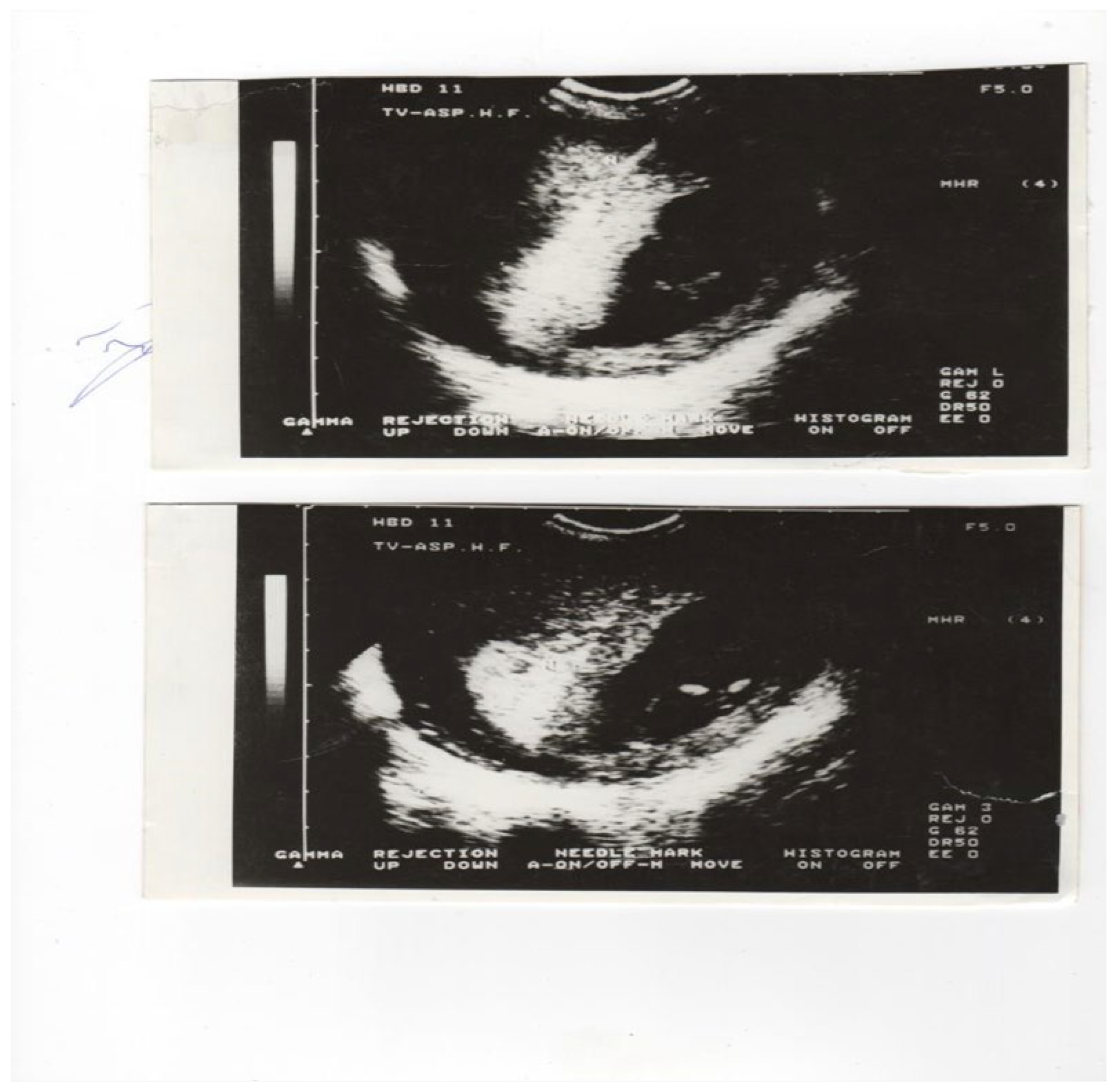
| N | % | |
|---|---|---|
|
Maternal age 37> Susp. UZV findings of chromosomal aberration or susp susp. NIPTI testes |
3575 1375 |
65 25 |
| Previous child with chromosome anomaly | 110 | 2.0 |
| Parental balanced translocations | 110 | 2.0 |
| Risk for metabolic disease (monogenic) | 80 | 1.5 |
| Risk for thalassemia or haemoglobinopathy | 80 | 1.5 |
| Sex determination (including 5 for X-linked disease) | 40 | 0.7 |
| Drug or radiation exposure | 45 | 0.8 |
| Family history of intellectual disability | 55 | 1,0 |
| Unspecified or other concerns (e. g. anxiety) | 30 | 0.5 |
| TC-CVS | TA-CVS | TV-CVS | TOTAL | |||||
|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | N | % | |
| TOTAL NUMBER | 850 | 15,5 | 4500 | 81,8 | 150 | 2,7 | 5500 | 100,0 |
|
EARLY COMPLICATIONS |
||||||||
| BLEEDING | 34 | 4,0 | 45 | 1,0 | 15 | 10,0 | 94 | 1,7 |
| INFECTION | 4 | 0,4 | 2 | 0,4 | 2 | 3,0 | 0,2 | 0.145 |
|
SPONTANEOUS ABORTION |
5 | 0,6 | 8 | 0,18 | 2 | 1,3 | 14 | 0,66 |
|
ABNORMAL CYTOGENETIC RESULTS |
61 | 7,1 | 154 | 3,4 | 5 | 3.3 | 220 | 4,0 |
|
SELECTIVE ABORTION |
58 | 6,8 | 150 | 3,3 | 5 | 3,3 | 213 | 3,9 |
|
TOTAL FETAL LOSS |
66 | 7,7 | 162 | 3,6 | 8 | 5,3 | 236 | 4,3 |
| CSV METHOD | <9 mg | 10-20 mg | >20 mg | |||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| TRANSABDOMINAL CVS | 500 | 11,1 | 3000 | 66,7 | 1000 | 22,2 |
| TRANSCERVICAL CVS | 150 | 17,6 | 300 | 35,3 | 400 |
47,1 |
| TRANSVAGINAL CVS | 20 | 13,3 | 55 | 36,7 | 75 | 50,0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




