Submitted:
29 August 2025
Posted:
03 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction

2. Biochar for Crop Production: Multifaceted Functionality
2.1. Soil Fertility Improvement
2.2. Plant Rooting Promotion
2.3. Plant Productivity Boost
2.4. Plant Biodefense Manipulation
- (1)
- The liming effect. Biochar is typically alkaline and has been widely reported to elevate soil pH. As pH plays a critical role in shaping microbial community development, diversity, structure, and pathogen virulence, such alterations can have profound ecological implications. Given that many soil pathogens are adapted to narrow pH ranges [46], biochar-mediated shifts in rhizosphere pH may significantly influence pathogen survival and activity.
- (2)
- The supply of organic compounds varies with the type of biochar feedstock, as different raw materials lead to differences in elemental composition and ash content [47]. These variations affect the concentrations of active components—such as soluble organic compounds, silicon, and calcium, in the resulting biochars, which likely explains their divergent effectiveness in suppressing plant diseases [48]. For example, one study confirmed that wood vinegar contains diverse volatile organic compounds that have long been used as pesticides [49]. However, it should be noted that the introduction of certain toxic compounds through biochar amendment may also increase the incidence and soil persistence of pathogens such as Plasmodiophora brassicae in Brassicaceae cropping systems [50].
- (3)
- Biochar can deactivate toxic compounds released by roots through its strong adsorption capacity, attributed to its high surface area and microporous structure [51]. For example, activated charcoal has been shown to effectively adsorb various root exudates, such as lactic acid, benzoic acid, vanillic acid, and succinic acid, significantly improving the yield of taro under continuous cropping conditions [52]. Growing evidence supports the role of biochar in immobilizing allelochemicals derived from root exudates [45] and suppressing soil-borne pathogens [44]. In our study on biochar amendment in replanted ginseng (Panax ginseng), biochar markedly reduced the accumulation of root-derived phenolic allelochemicals, thereby inhibiting soil-borne pathogenic fungi, while simultaneously enhancing microbial diversity and network complexity [11].
- (4)
- Soil microbial manipulation is a critical approach to addressing the challenges posed by continuous cropping, which adversely affects soil health and promotes soil-borne diseases. These conditions further disrupt soil properties, alter microbial community structure, and lead to pathogen accumulation in the rhizosphere [53,54]. Biochar amendment has been shown to promote beneficial microorganisms while reducing the abundance and pathogenicity of pathotrophic fungi. Notably, maize biochar outperforms wood biochar in enhancing the abundance of arbuscular mycorrhizal fungi (AMF) and beneficial bacteria [11]. Additionally, biochar application increases the complexity of microbial co-occurrence networks, particularly within fungal communities [11,54]. As a result, the core microbial networks exhibit enhanced resistance even as pathogenic fungi proliferate in biochar-amended soils. Biochar also promotes the enrichment of plant growth-promoting rhizobacteria (PGPR) in the rhizosphere via host-mediated recruitment. For instance, Jin et al. [55] demonstrated that biochar stimulates tomato roots to assemble a protective bacterial community that confers resistance to Fusarium wilt.
- (5)
- Biochar induced plant resistance. Biochar soil amendment can directly influence the physiological status of plants, particularly by modifying root exudation, which facilitates the recruitment of plant growth-promoting rhizobacteria. Previous studies have indicated that biochar exerts direct effects on plant growth and physiological processes [56,57]. Moreover, biochar shows considerable potential in activating immunity-related gene expression. Transcriptomic analyses in tomato have revealed that biochar primes defense-related pathways, upregulating genes and hormones associated with plant immunity and development—including jasmonic acid, brassinosteroids, cytokinins, auxin, as well as the synthesis of flavonoids, phenylpropanoids, and cell wall components [58]. In a study by Kong et al. [59], exogenous application of nanoscale biochar was shown to enhance plant defense responses and confer resistance against the pathogen Phytophthora nicotianae. These findings suggest that biochar, when applied at levels that optimally stimulate plant immunity, could serve as an effective plant protection agent in future agricultural practices.
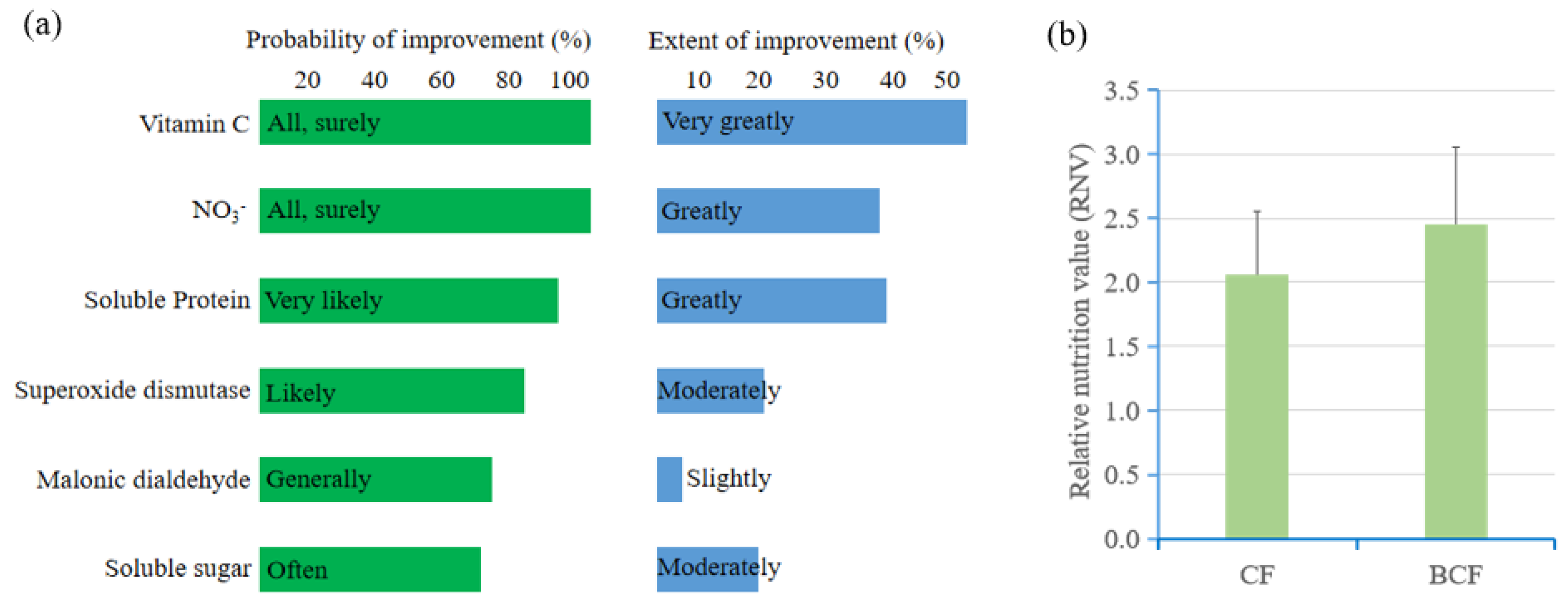
2.5. Food Quality Enhancement
3. Prospectives
- (1)
- To what extent plant process will be affected by soil changes: abiotic versus biotic?
- (2)
- How plant roots respond to biochar material input versus to biochar: habituating versus signaling?
- (3)
- What mediate the interplay among plant growth, resistance and biosynthesis following biochar soil application?
- (4)
- How will be the legacy of soil-plant system following application of biochar?
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lehmann, J., Bossio, D. A., Kögel- Knabner, I., Rillig, M. C. 2020. The concept and future prospects of soil health. Nat. Rev. Earth Environ. 1, 544–553. [CrossRef]
- Zhao, Z., Liu, C., Yan, M., Pan, G. 2023. Understanding and enhancing soil conservation of water and life. Soil Science and Environment 2:9. [CrossRef]
- IPBES. 2019. Summary for policymakers of the global assessment report on biodiversity and ecosystem services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. IPBES secretariat, Bonn, Germany. 56 pp.
- United Nations Environment Programme. 2022. Nature-based Solutions: Opportunities and Challenges for Scaling Up. Nairobi.
- Glaser, B., Lehmann, J., Zech, W. (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal - a review. Biology and Fertility of Soils 35, 219-230. [CrossRef]
- Lehmann, J. (2007) A handful of carbon. Nature 447, 143-144. [CrossRef]
- Lehmann, J., Gaunt, J., Rondon, M. (2006) Bio-char Sequestration in Terrestrial Ecosystems – A Review. Mitigation and Adaptation Strategies for Global Change 11, 403-427. [CrossRef]
- Sohi, S.P. (2012) Carbon Storage with Benefits. Science 338, 1034-1035. [CrossRef]
- Xiang Y, Deng Q, Duan H, Guo Y (2017) Effects of biochar application on root traits: a meta-analysis. GCB Bioenergy 9:1563–1572. [CrossRef]
- Yang, Y.H., Chen, T.T., Xiao, R., Chen, X.P., Zhang, T. (2022) A quantitative evaluation of the biochar's influence on plant disease suppress: a global meta-analysis. Biochar 4. [CrossRef]
- Liu, C., Xia, R., Tang, M., Chen, X., Zhong, B., Liu, X.Y., Bian, R.J., Yang, L., Zheng, J.F., Cheng, K., Zhang, X.H., Drosos, M., Li, L.Q., Shan, S.D., Joseph, S., Pan, G.X. (2022a) Improved ginseng production under continuous cropping through soil health reinforcement and rhizosphere microbial manipulation with biochar: a field study of Panax ginseng from Northeast China. Horticulture Research 9. [CrossRef]
- Liu C, Tian J, ..., Pan G. Biochar boosted high oleic peanut production with enhanced root development and biological N fixation by diazotrophs in an alluvic Primisol. Science of the Total Environment, 2024, 932: 173061. [CrossRef]
- Liu C, Shang S, Wang C, ..., Pan G. Biochar amendment increases peanut production through improvement of the extracellular enzyme activities and microbial community composition in replanted field. Plants, 2025, 14, 922. [CrossRef]
- Omondi, M.O., Xia, X., Nahayo, A., Liu, X.Y., Korai, P.K., Pan, G.X. (2016) Quantification of biochar effects on soil hydrological properties using meta-analysis of literature data. Geoderma 274, 28-34. [CrossRef]
- Razzaghi, F., Obour, P.B., Arthur, E. (2020) Does biochar improve soil water retention? A systematic review and meta-analysis. Geoderma 361. [CrossRef]
- Singh, H., Northup, B.K., Rice, C.W., Prasad, P.V.V. (2022) Biochar applications influence soil physical and chemical properties, microbial diversity, and crop productivity: a meta-analysis. Biochar 4. [CrossRef]
- Wu, W.A., Han, J.Y., Gu, Y.N., Li, T., Xu, X.R., Jiang, Y.H., Li, Y.P., Sun, J.F., Pan, G.X., Cheng, K. (2022) Impact of biochar amendment on soil hydrological properties and crop water use efficiency: A global meta-analysis and structural equation model. Global Change Biology Bioenergy 14, 657-668. [CrossRef]
- Edeh, I.G., Masek, O., Buss, W. (2020) A meta-analysis on biochar's effects on soil water properties - New insights and future research challenges. Science of The Total Environment 714. [CrossRef]
- Sun, Z.H., Hu, Y., Shi, L., Li, G., Pang, Z., Liu, S.Q., Chen, Y.M., Jia, B.B. (2022) Effects of biochar on soil chemical properties: A global meta-analysis of agricultural soil. Plant Soil and Environment 68, 272-289. [CrossRef]
- Biederman, L.A., Harpole, W.S. (2013) Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. GCB Bioenergy 5, 202-214. [CrossRef]
- Chan, K.Y., Van Zwieten, L., Meszaros, I., Downie, A., Joseph, S. (2007) Agronomic values of greenwaste biochar as a soil amendment. Australian Journal of Soil Research 45, 629-634. [CrossRef]
- Gao, S., DeLuca, T.H., Cleveland, C.C. (2019) Biochar additions alter phosphorus and nitrogen availability in agricultural ecosystems: A meta-analysis. Science of The Total Environment 654, 463-472. [CrossRef]
- Tesfaye, F., Liu, X.Y., Zheng, J.F., Cheng, K., Bian, R.J., Zhang, X.H., Li, L.Q., Drosos, M., Joseph, S., Pan, G.X. (2021) Could biochar amendment be a tool to improve soil availability and plant uptake of phosphorus? A meta-analysis of published experiments. Environmental Science and Pollution Research 28, 34108-34120. [CrossRef]
- Nguyen, T.T.N., Xu, C.Y., Tahmasbian, I., Che, R.X., Xu, Z.H., Zhou, X.H., Wallace, H.M., Bai, S.H. (2017) Effects of biochar on soil available inorganic nitrogen: A review and meta-analysis. Geoderma 288, 79-96. [CrossRef]
- Liu, Q., Zhang, Y.H., Liu, B.J., Amonette, J.E., Lin, Z.B., Liu, G., Ambus, P., Xie, Z.B. (2018) How does biochar influence soil N cycle? A meta-analysis. Plant and Soil 426, 211-225. [CrossRef]
- Borchard, N., Schirrmann, M., Cayuela, M.L., Kammann, C., Wrage-Monnig, N., Estavillo, J.M., Fuertes-Mendizabal, T., Sigua, G., Spokas, K., Ippolito, J.A., Novak, J. (2019) Biochar, soil and land-use interactions that reduce nitrate leaching and N2O emissions: A meta-analysis. Science of The Total Environment 651, 2354-2364. [CrossRef]
- Sha, Z.P., Li, Q.Q., Lv, T.T., Misselbrook, T., Liu, X.J. (2019) Response of ammonia volatilization to biochar addition: A meta-analysis. Science of The Total Environment 655, 1387-1396. [CrossRef]
- Major, J., Rondon, M., Molina, D., Riha, S.J., Lehmann, J. (2010) Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant and Soil 333, 117-128. [CrossRef]
- Liu, X., Li, L.Q., Bian, R.J., Chen, D., Qu, J.J., Kibue, G.W., Pan, G.X., Zhang, X.H., Zheng, J.W., Zheng, J.F. (2014) Effect of biochar amendment on soil- silicon availability and rice uptake. Journal of Plant Nutrition and Soil Science 177, 91-96. [CrossRef]
- Liu C, Sun B, Zhang X, ..., Pan G. The Water-soluble pool in biochar dominates maize plant growth promotion under biochar amendment. J Plant Growth Regul, 2021a, 40: 1466-1476. [CrossRef]
- Liu, X., Wang, H.D., Liu, C., Sun, B.B., Zheng, J.F., Bian, R.J., Drosos, M., Zhang, X.H., Li, L.Q., Pan, G.X. (2021b) Biochar increases maize yield by promoting root growth in the rainfed region. Archives of Agronomy and Soil Science 67, 1411-1424. [CrossRef]
- Liu X., Liu C., Gao W., et al. Impact of biochar amendment on the abundance and structure of diazotrophic community in an alkaline soil. The Science of the Total Environment 2019, 688: 944-951. [CrossRef]
- Bai, S.H., Omidvar, N., Gallart, M., Kamper, W., Tahmasbian, I., Farrar, M.B., Singh, K., Zhou, G., Muqadass, B., Xu, C.Y., Koech, R., Li, Y., Nguyen, T.T.N., van Zwieten, L. (2022) Combined effects of biochar and fertilizer applications on yield: A review and meta-analysis. Sci Total Environ 808, 152073. [CrossRef]
- Farhangi-Abriz, S., Torabian, S., Qin, R., Noulas, C., Lu, Y., Gao, S. (2021) Biochar effects on yield of cereal and legume crops using meta-analysis. Science of The Total Environment 775, 145869. [CrossRef]
- Liu, X., Zhang, A., Ji, C., Joseph, S., Bian, R., Li, L., Pan, G., Paz-Ferreiro, J. (2013) Biochar’s effect on crop productivity and the dependence on experimental conditions—a meta-analysis of literature data. Plant and Soil 373, 583-594. [CrossRef]
- Ye, L., Camps-Arbestain, M., Shen, Q., Lehmann, J., Singh, B., Sabir, M., Condron, L.M. (2019) Biochar effects on crop yields with and without fertilizer: A meta-analysis of field studies using separate controls. Soil Use and Management 36, 2-18. [CrossRef]
- Zhang, L., Wu, Z., Zhou, J., Zhou, L., Lu, Y., Xiang, Y., Zhang, R., Deng, Q., Wu, W. (2022a) Meta-Analysis of the Response of the Productivity of Different Crops to Parameters and Processes in Soil Nitrogen Cycle under Biochar Addition. Agronomy 12, 1857. [CrossRef]
- Dai, Y., Zheng, H., Jiang, Z., Xing, B. (2020) Combined effects of biochar properties and soil conditions on plant growth: A meta-analysis. Sci Total Environ 713, 136635. [CrossRef]
- Jeffery, S., Abalos, D., Prodana, M., Bastos, A.C., van Groenigen, J.W., Hungate, B.A., Verheijen, F. (2017) Biochar boosts tropical but not temperate crop yields. Environmental Research Letters 12, 053001. [CrossRef]
- Liu, Y., Li, H., Hu, T., Mahmoud, A., Li, J., Zhu, R., Jiao, X., Jing, P. (2022b) A quantitative review of the effects of biochar application on rice yield and nitrogen use efficiency in paddy fields: A meta-analysis. Sci Total Environ 830, 154792. [CrossRef]
- Zhu, X.M., Chen, B.L., Zhu, L.Z., Xing, B.S. (2017) Effects and mechanisms of biochar-microbe interactions in soil improvement and pollution remediation: A review. Environ Pollut 227, 98-115. [CrossRef]
- Graber, E.R., Harel, Y.M., Kolton, M., Cytryn, E., Silber, A., David, D.R., Tsechansky, L., Borenshtein, M., Elad, Y. (2010) Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant and Soil 337, 481-496. [CrossRef]
- Yang, Y.H., Chen, T.T., Xiao, R., Chen, X.P., Zhang, T. (2022) A quantitative evaluation of the biochar's influence on plant disease suppress: a global meta-analysis. Biochar 4. [CrossRef]
- Jaiswal, A.K., Elad, Y., Paudel, I., Graber, E.R., Cytryn, E., Frenkel, O. (2017) Linking the Belowground Microbial Composition, Diversity and Activity to Soilborne Disease Suppression and Growth Promotion of Tomato Amended with Biochar. Sci Rep 7,44382. [CrossRef]
- Gu, Y., Hou, Y.G., Huang, D.P., Hao, Z.X., Wang, X.F., Wei, Z., Jousset, A., Tan, S.Y., Xu, D.B., Shen, Q.R., Xu, Y.C., Friman, V.P. (2017) Application of biochar reduces Ralstonia solanacearum infection via effects on pathogen chemotaxis, swarming motility, and root exudate adsorption. Plant and Soil 415, 269-281. [CrossRef]
- Husson, O. (2013) Redox potential (Eh) and pH as drivers of soil/plant/microorganism systems: a transdisciplinary overview pointing to integrative opportunities for agronomy. Plant and Soil 362, 389-417. [CrossRef]
- Zhou, Y.W., Qin, S.Y., Verma, S., Sar, T., Sarsaiya, S., Ravindran, B., Liu, T., Sindhu, R., Patel, A.K., Binod, P., Varjani, S., Singhnia, R.R., Zhang, Z.Q., Awasthi, M.K. (2021) Production and beneficial impact of biochar for environmental application: A comprehensive review. Bioresource Technology 337. [CrossRef]
- Bian, R., Liu, X., Zheng, J., Cheng, K., Zhang, X., Li, L., et al. (2022) Chemical composition and bioactivity of dissolvable organic matter in biochars. Sci. Agric. Sin. 55, 2174–2186. (In Chinese with English abstract).
- Liu, X., Wang, J.A., Feng, X.H., Yu, J.L. (2021c) Wood vinegar resulting from the pyrolysis of apple tree branches for annual bluegrass control. Industrial Crops and Products 174. [CrossRef]
- Liu, L., Chen, Z., Su, Z. et al. (2023) Soil pH indirectly determines Ralstonia solanacearum colonization through its impacts on microbial networks and specific microbial groups. Plant Soil 482, 73–88. [CrossRef]
- Ahmad, M., Rajapaksha, A.U., Lim, J.E., Zhang, M., Bolan, N., Mohan, D., Vithanage, M., Lee, S.S., Ok, Y.S. (2014) Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 99, 19-33. [CrossRef]
- Asao, T., Hasegawa, K., Sueda, Y., Tomita, K., Taniguchi, K., Hosoki, T., Pramanik, M.H.R., Matsui, Y. (2003) Autotoxicity of root exudates from taro. Scientia Horticulturae 97, 389-396. [CrossRef]
- Wang, J., Chen, H.L., Gao, J., Guo, J.X., Zhao, X.S., Zhou, Y.F. (2018) Ginsenosides and ginsenosidases in the pathobiology of ginseng-Cylindrocarpon destructans (Zinss) Scholten. Plant Physiology and Biochemistry 123, 406-413. [CrossRef]
- Wu, H.M., Qin, X.J., Wu, H.M., Li, F., Wu, J.C., Zheng, L., Wang, J.Y., Chen, J., Zhao, Y.L., Lin, S., Lin, W.X. (2020) Biochar mediates microbial communities and their metabolic characteristics under continuous monoculture. Chemosphere 246. [CrossRef]
- Jin, X., Bai, Y., Rahman, M.K., Kang, X., Pan, K., Wu, F., Pommier, T., Zhou, X., Wei, Z. 2022. Biochar stimulates tomato roots to recruit a bacterial assemblage contributing to disease resistance against Fusarium wilt. iMeta e37. [CrossRef]
- Graber, E.R., Tsechansky, L., Mayzlish-Gati, E., Shema, R., Koltai, H. (2015) A humic substances product extracted from biochar reduces Arabidopsis root hair density and length under P-sufficient and P-starvation conditions. Plant and Soil 395, 21-30. [CrossRef]
- Sun, J.L., Drosos, M., Mazzei, P., Savy, D., Todisco, D., Vinci, G., Pan, G.X., Piccolo, A. (2017) The molecular properties of biochar carbon released in dilute acidic solution and its effects on maize seed germination. Science of The Total Environment 576, 858-867. [CrossRef]
- Jaiswal, A.K., Alkan, N., Elad, Y., Sela, N., Philosoph, A.M., Graber, E.R., Frenkel, O. (2020) Molecular insights into biochar-mediated plant growth promotion and systemic resistance in tomato against Fusarium crown and root rot disease. Sci Rep 10, 13934. [CrossRef]
- Kong, M.M., Liang, J., White, J.C., Elmer, W.H., Wang, Y., Xu, H.L., He, W.X., Shen, Y., Gao, X.W. (2022) Biochar nanoparticle-induced plant immunity and its application with the elicitor methoxyindole in Nicotiana benthamiana. Environmental Science-Nano 9, 3514-3524. [CrossRef]
- Burkey, K.O., Booker, F.L., Pursley, W.A., Heagle, A.S., 2007. Elevated carbon dioxide and ozone effects on peanut: II. Seed yield and quality. Crop Sci. 47 (4), 1488–1497. [CrossRef]
- Ghannadzadeh, A.M., 2015. The Effect of Irrigation Regime and Nitrogen Fertilizer on Seed Yield and Qualitative Characteristics of Peanut (Arachis hypogaea L) (Case Study of Gilan Province, Iran) (Ph D thesis).
- Ibrahim, M.M., Hu, K., Tong, C., Xing, S., Zou, S., Mao, Y., 2020. De-ashed biochar enhances nitrogen retention in manured soil and changes soil microbial dynamics. Geoderma 378, 114589. [CrossRef]
- Barbour, J.A., Howe, P.R.C., Buckley, J.D., Bryan, J., Coates, A.M., 2017. Cerebrovascular and cognitive benefits of high-oleic peanut consumption in healthy overweight middle-aged adults. Nutr. Neurosci. 20 (10), 555–562. [CrossRef]
- Ministry of Agriculture and Rural Affairs, 2018. Industry Standard of High Oleic Acid Peanut.
- Seleiman, M.F., Refay, Y., Al-Suhaibani, N., AI-Ashkar, I., EI-Hendawy, S., Hafez, E.M., 2019. Integrative effects of rice-straw biochar and silicon on oil and seed quality, yield and physiological traits of Helianthus annuus L. grown under water deficit stress. Agronomy 2019 (10), 637. [CrossRef]
- Khan, Z., Khan, M.N., Zhang, K., Luo, T., Zhu, K., Hu, L., 2021. The application of biochar alleviated the adverse effects of drought on the growth, physiology, yield and quality of rapeseed through regulation of soil status and nutrients availability. Ind. Crop. Prod. 171, 113878. [CrossRef]
- Vigar, M., Hancock, R.D., Miglietta, F., Taylor, G., 2015. More plant growth but less plant defence? First global gene expression data for plants grown in soil amended with biochar. Glob. Chang. Biol. Bioenergy 7, 658–672. [CrossRef]
- Moradi, S., Sajedi, N.A., Madani, H., Gomarian, M., Chavoshi, S., 2023. Integrated effects of nitrogen fertilizer, biochar, and salicylic acid on yield and fatty acid profile of six rapeseed cultivars. J. Soil Sci. Plant Nutr. 23 (1), 380–397. [CrossRef]



| Authors & Year | Change of crop productivity | Observation | Experiment |
|---|---|---|---|
| Amirahmadi et al. (2025) | Yield: 16.2% | 1166 | Field |
| Singh et al. (2022) | Crop yield: 32% | 110 | Pot and field |
| Zhang et al. (2022a) | Crop productivity: 13% | 691 | Field |
| Liu et al. (2022b) | Yield: 10.7% | 378 | Pot and field |
| Farhangi-Abriz et al. (2021) | Grain yield: maize 28%; wheat 13% | 296 | Field |
| Dai et al. (2020) | Plant productivity: 16% | 1254 | Pot and field |
| Liu et al. (2019) | Yield: 15.4% | 605 | Field |
| Ye et al. (2019) | Yield: 9.9% | 232 | Field |
| Jeffery et al. (2017) | Yield: 13% | 1125 | Pot and field |
| Biederman & Harpole (2013) | Biomassag:29.7%; Biomassbg: 39.9%; Yield: 18.7% | 317 | Pot and field |
| Liu et al. (2013) | Biomass: 12.5%; Yield: 8.4% | 880 | Pot and field |
| Jeffery et al. (2011) | Biomassag and grain yield: 10% | 177 | Pot and field |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




