Submitted:
29 August 2025
Posted:
29 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
The Physiological Roles of Prions
An Origin in Antagonistic Pleiotropy
Network Insufficiencies
Connection Mode vs. Protection Mode
Translational Implications of the Theory: Pathophysiology
Translational Implications of the Theory: Therapeutics
The Failure of Simple Models of Alzheimer’s Disease
Clinical Outcomes of the Pr2 Theory
Implications of the Pr2 Theory
Summary
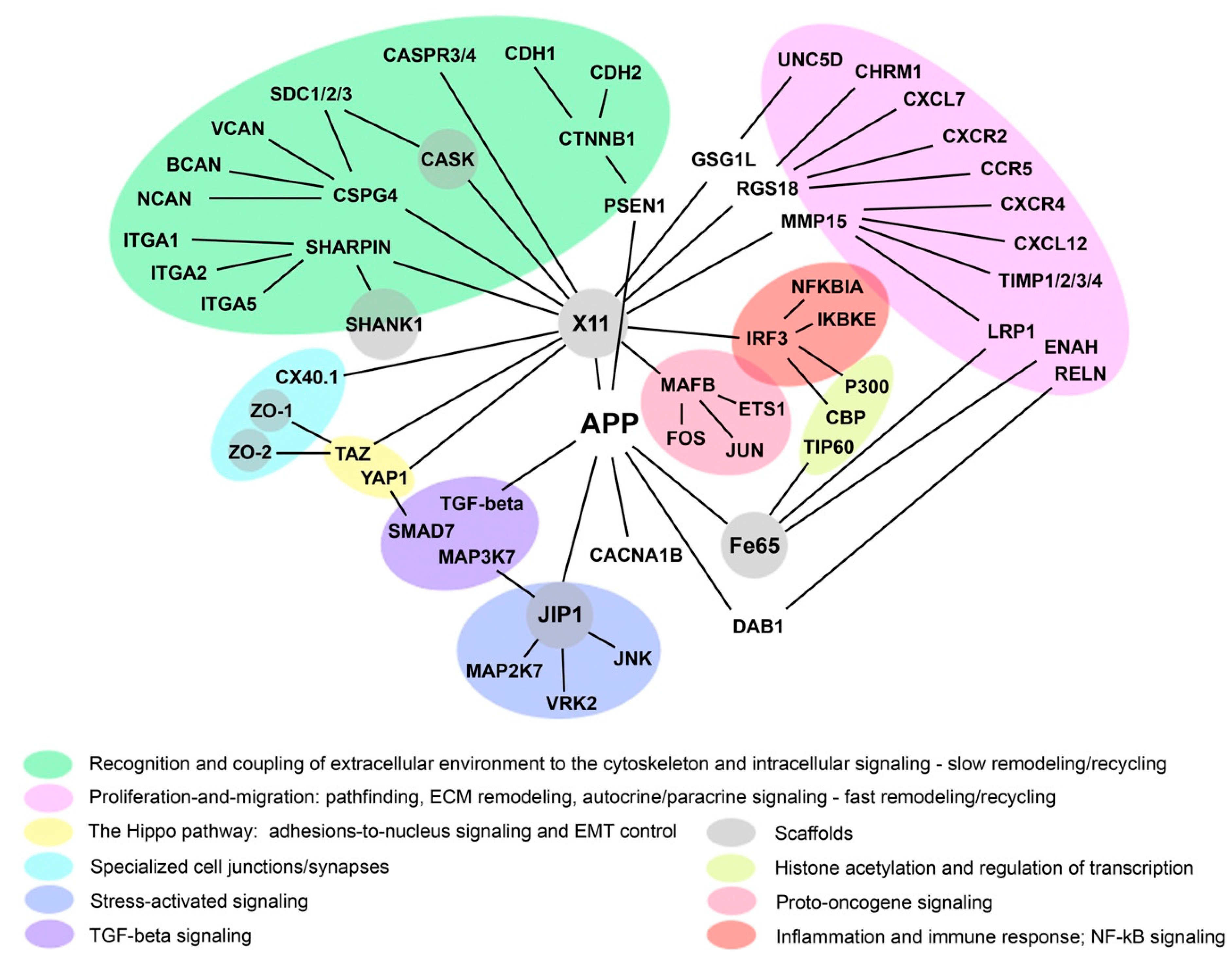
Acknowledgments
References
- Hardy JA, Higgins GA Alzheimer’s disease: the amyloid cascade hypothesis. Science 1992, 256, 184–185. [CrossRef] [PubMed]
- Sweeney P, Park H, Baumann M, Dunlop J, Frydman J, Kopito R, McCampbell A, Leblanc G, Venkateswaran A, Nurmi A, Hodgson R Protein misfolding in neurodegenerative diseases: implications and strategies. Transl Neurodegener 2017, 6, 6.
- Arnsten AFT, Datta D, Del Tredici K, Braak H Hypothesis: Tau pathology is an initiating factor in sporadic Alzheimer’s disease. Alzheimers Dement 2021, 17, 115–124. [CrossRef]
- Prusiner SB Novel proteinaceous infectious particles cause scrapie. Science 1982, 216, 136–144. [CrossRef]
- Bhatt S, Puli L, Patil CR Role of reactive oxygen species in the progression of Alzheimer’s disease. Drug Discov Today 2021, 26, 794–803. [CrossRef]
- Seaks CE, Wilcock DM Infectious hypothesis of Alzheimer disease. PLoS Pathog 2020, 16, e1008596.
- Kciuk M, Kruczkowska W, Gałęziewska J, Wanke K, Kałuzińska-Kołat Ż, Aleksandrowicz M, Kontek R Alzheimer’s Disease as Type 3 Diabetes: Understanding the Link and Implications. International Journal of Molecular Sciences 2024, 25, 11955. [CrossRef] [PubMed]
- Mehta D, Jackson R, Paul G, Shi J, Sabbagh M Why do trials for Alzheimer’s disease drugs keep failing? A discontinued drug perspective for 2010-2015. Expert Opin Investig Drugs 2017, 26, 735–739. [CrossRef]
- Nelson PT, Dickson DW, Trojanowski JQ, Jack CR, Boyle PA, Arfanakis K, Rademakers R, Alafuzoff I, Attems J, Brayne C, Coyle-Gilchrist ITS, Chui HC, Fardo DW, Flanagan ME, Halliday G, Hokkanen SRK, Hunter S, Jicha GA, Katsumata Y, Kawas CH, Keene CD, Kovacs GG, Kukull WA, Levey AI, Makkinejad N, Montine TJ, Murayama S, Murray ME, Nag S, Rissman RA, Seeley WW, Sperling RA, White III CL, Yu L, Schneider JA Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain 2019, 142, 1503–1527.
- Le Guen Y, Belloy ME, Grenier-Boley B, de Rojas I, Castillo-Morales A, Jansen I, Nicolas A, Bellenguez C, Dalmasso C, Küçükali F, Eger SJ, Rasmussen KL, Thomassen JQ, Deleuze JF, He Z, Napolioni V, Amouyel P, Jessen F, Kehoe PG, van Duijn C, Tsolaki M, Sánchez-Juan P, Sleegers K, Ingelsson M, Rossi G, Hiltunen M, Sims R, van der Flier WM, Ramirez A, Andreassen OA, Frikke-Schmidt R, Williams J, Ruiz A, Lambert JC, Greicius MD, Arosio B, Benussi L, Boland A, Borroni B, Caffarra P, Daian D, Daniele A, Debette S, Dufouil C, Düzel E, Galimberti D, Giedraitis V, Grimmer T, Graff C, Grünblatt E, Hanon O, Hausner L, Heilmann-Heimbach S, Holstege H, Hort J, Jürgen D, Kuulasmaa T, van der Lugt A, Masullo C, Mecocci P, Mehrabian S, de Mendonça A, Moebus S, Nacmias B, Nicolas G, Olaso R, Papenberg G, Parnetti L, Pasquier F, Peters O, Pijnenburg YAL, Popp J, Rainero I, Ramakers I, Riedel-Heller S, Scarmeas N, Scheltens P, Scherbaum N, Schneider A, Seripa D, Soininen H, Solfrizzi V, Spalletta G, Squassina A, van Swieten J, Tegos TJ, Tremolizzo L, Verhey F, Vyhnalek M, Wiltfang J, Boada M, García-González P, Puerta R, Real LM, Álvarez V, Bullido MJ, Clarimon J, García-Alberca JM, Mir P, Moreno F, Pastor P, Piñol-Ripoll G, Molina-Porcel L, Pérez-Tur J, Rodríguez-Rodríguez E, Royo JL, Sánchez-Valle R, Dichgans M, Rujescu D () Association of Rare APOE Missense Variants V236E and R251G With Risk of Alzheimer Disease. JAMA Neurol 2022, 79, 652–663.
- Aizenstein HJ, Nebes RD, Saxton JA, Price JC, Mathis CA, Tsopelas ND, Ziolko SK, James JA, Snitz BE, Houck PR, Bi W, Cohen AD, Lopresti BJ, DeKosky ST, Halligan EM, Klunk WE (2008) Frequent amyloid deposition without significant cognitive impairment among the elderly. Arch Neurol, 1517.
- Jones MK, Nair A, Gupta M (2019) Mast Cells in Neurodegenerative Disease. Front Cell Neurosci.
- van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, Kanekiyo M, Li D, Reyderman L, Cohen S, Froelich L, Katayama S, Sabbagh M, Vellas B, Watson D, Dhadda S, Irizarry M, Kramer LD, Iwatsubo T (2023) Lecanemab in Early Alzheimer’s Disease. N Engl J Med.
- Soscia SJ, Kirby JE, Washicosky KJ, Tucker SM, Ingelsson M, Hyman B, Burton MA, Goldstein LE, Duong S, Tanzi RE, Moir RD (2010) The Alzheimer’s disease-associated amyloid beta-protein is an antimicrobial peptide. PLoS One.
- Eimer W, Vijaya Kumar DK, Tanzi RE (2019) Cure Alzheimer’s Fund.
- Toups K, Hathaway A, Gordon D, Chung H, Raji C, Boyd A, Hill BD, Hausman-Cohen S, Attarha M, Chwa WJ, Jarrett M, Bredesen DE (2022) Precision Medicine Approach to Alzheimer’s Disease: Successful Pilot Project. Journal of Alzheimer’s Disease.
- Sandison H, Callan NGL, Rao RV, Phipps J, Bradley R (2023) Observed Improvement in Cognition During a Personalized Lifestyle Intervention in People with Cognitive Decline. J Alzheimers Dis.
- Bredesen DE (2014) Reversal of cognitive decline: a novel therapeutic program. Aging (Albany NY).
- Bredesen DE, Amos EC, Canick J, Ackerley M, Raji C, Fiala M, Ahdidan J (2016) Reversal of cognitive decline in Alzheimer’s disease. Aging (Albany NY).
- Bredesen DE, Sharlin K, Jenkins K, Okuno M, Youngberg W, al. e (2018) Reversal of Cognitive Decline: 100 Patients. J Alzheimers Dis Parkinsonism.
- Choi CJ, Kanthasamy A, Anantharam V, Kanthasamy AG (2006) Interaction of metals with prion protein: possible role of divalent cations in the pathogenesis of prion diseases. Neurotoxicology.
- Ezpeleta J, Boudet-Devaud F, Pietri M, Baudry A, Baudouin V, Alleaume-Butaux A, Dagoneau N, Kellermann O, Launay J-M, Schneider B (2017) Protective role of cellular prion protein against TNFα-mediated inflammation through TACE α-secretase. Scientific Reports.
- Spilman PR, Corset V, Gorostiza O, Poksay KS, Galvan V, Zhang J, Rao R, Peters-Libeu C, Vincelette J, McGeehan A, Dvorak-Ewell M, Beyer J, Campagna J, Bankiewicz K, Mehlen P, John V, Bredesen DE (2016) Netrin-1 Interrupts Amyloid-beta Amplification, Increases sAbetaPPalpha in vitro and in vivo, and Improves Cognition in a Mouse Model of Alzheimer’s Disease. J Alzheimers Dis.
- Chakrabortee S, Byers JS, Jones S, Garcia DM, Bhullar B, Chang A, She R, Lee L, Fremin B, Lindquist S, Jarosz DF (2016) Intrinsically Disordered Proteins Drive Emergence and Inheritance of Biological Traits. Cell.
- Fioriti L, Myers C, Huang YY, Li X, Stephan JS, Trifilieff P, Colnaghi L, Kosmidis S, Drisaldi B, Pavlopoulos E, Kandel ER (2015) The Persistence of Hippocampal-Based Memory Requires Protein Synthesis Mediated by the Prion-like Protein CPEB3. Neuron.
- Jansen WJ, Ossenkoppele R, Knol DL, Tijms BM, Scheltens P, Verhey FR, Visser PJ, Aalten P, Aarsland D, Alcolea D, Alexander M, Almdahl IS, Arnold SE, Baldeiras I, Barthel H, van Berckel BN, Bibeau K, Blennow K, Brooks DJ, van Buchem MA, Camus V, Cavedo E, Chen K, Chetelat G, Cohen AD, Drzezga A, Engelborghs S, Fagan AM, Fladby T, Fleisher AS, van der Flier WM, Ford L, Förster S, Fortea J, Foskett N, Frederiksen KS, Freund-Levi Y, Frisoni GB, Froelich L, Gabryelewicz T, Gill KD, Gkatzima O, Gómez-Tortosa E, Gordon MF, Grimmer T, Hampel H, Hausner L, Hellwig S, Herukka SK, Hildebrandt H, Ishihara L, Ivanoiu A, Jagust WJ, Johannsen P, Kandimalla R, Kapaki E, Klimkowicz-Mrowiec A, Klunk WE, Köhler S, Koglin N, Kornhuber J, Kramberger MG, Van Laere K, Landau SM, Lee DY, de Leon M, Lisetti V, Lleó A, Madsen K, Maier W, Marcusson J, Mattsson N, de Mendonça A, Meulenbroek O, Meyer PT, Mintun MA, Mok V, Molinuevo JL, Møllergård HM, Morris JC, Mroczko B, Van der Mussele S, Na DL, Newberg A, Nordberg A, Nordlund A, Novak GP, Paraskevas GP, Parnetti L, Perera G, Peters O, Popp J, Prabhakar S, Rabinovici GD, Ramakers IH, Rami L, Resende de Oliveira C, Rinne JO, Rodrigue KM, Rodríguez-Rodríguez E, Roe CM, Rot U, Rowe CC, Rüther E, Sabri O, Sanchez-Juan P, Santana I, Sarazin M, Schröder J, Schütte C, Seo SW, Soetewey F, Soininen H, Spiru L, Struyfs H, Teunissen CE, Tsolaki M, Vandenberghe R, Verbeek MM, Villemagne VL, Vos SJ, van Waalwijk van Doorn LJ, Waldemar G, Wallin A, Wallin Å K, Wiltfang J, Wolk DA, Zboch M, Zetterberg H Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. Jama 2015, 313, 1924–1938.
- Donohue MC, Sperling RA, Petersen R, Sun CK, Weiner MW, Aisen PS (2017) Association Between Elevated Brain Amyloid and Subsequent Cognitive Decline Among Cognitively Normal Persons. Jama, 317, 2305–2316.
- Williams GC (1957) Pleiotropy, Natural Selection, and the Evolution of Senescence. Evolution.
- Nicklas WJ, Youngster SK, Kindt MV, Heikkila RE (1987) MPTP, MPP+ and mitochondrial function. Life Sci.
- Vos M (2022) Mitochondrial Complex I deficiency: guilty in Parkinson’s disease. Signal Transduction and Targeted Therapy.
- Foran E, Trotti D Glutamate transporters and the excitotoxic path to motor neuron degeneration in amyotrophic lateral sclerosis. Antioxid Redox Signal 2009, 11, 1587–1602. [CrossRef] [PubMed]
- Escobar-Khondiker M, Höllerhage M, Muriel MP, Champy P, Bach A, Depienne C, Respondek G, Yamada ES, Lannuzel A, Yagi T, Hirsch EC, Oertel WH, Jacob R, Michel PP, Ruberg M, Höglinger GU (2007) Annonacin, a natural mitochondrial complex I inhibitor, causes tau pathology in cultured neurons. J Neurosci.
- Emery DC, Davies M, Cerajewska TL, Taylor J, Hazell M, Paterson A, Allen-Birt SJ, West NX (2022) High resolution 16S rRNA gene Next Generation Sequencing study of brain areas associated with Alzheimer’s and Parkinson’s disease. Frontiers in Aging Neuroscience.
- Cacciottolo M, Wang X, Driscoll I, Woodward N, Saffari A, Reyes J, Serre ML, Vizuete W, Sioutas C, Morgan TE, Gatz M, Chui HC, Shumaker SA, Resnick SM, Espeland MA, Finch CE, Chen JC (2017) Particulate air pollutants, APOE alleles and their contributions to cognitive impairment in older women and to amyloidogenesis in experimental models. Transl Psychiatry.
- Zhang J, You L, Wu W, Wang X, Chrienova Z, Nepovimova E, Wu Q, Kuca K The neurotoxicity of trichothecenes T-2 toxin and deoxynivalenol (DON): Current status and future perspectives. Food and Chemical Toxicology 2020, 145, 111676. [CrossRef]
- Bredesen DE (2016) Inhalational Alzheimer’s disease: an unrecognized - and treatable - epidemic. Aging (Albany NY).
- Tan ZS, Beiser A, Vasan RS, Au R, Auerbach S, Kiel DP, Wolf PA, Seshadri S (2008) Thyroid function and the risk of Alzheimer disease: the Framingham Study. Arch Intern Med.
- Capsoni S, Ugolini G, Comparini A, Ruberti F, Berardi N, Cattaneo A (2000) Alzheimer-like neurodegeneration in aged antinerve growth factor transgenic mice. Proceedings of the National Academy of Sciences.
- Ouanes S, Popp J (2019) High Cortisol and the Risk of Dementia and Alzheimer’s Disease: A Review of the Literature. Front Aging Neurosci.
- Zhang C, Kuo CC, Moghadam SH, Monte L, Campbell SN, Rice KC, Sawchenko PE, Masliah E, Rissman RA (2016) Corticotropin-releasing factor receptor-1 antagonism mitigates beta amyloid pathology and cognitive and synaptic deficits in a mouse model of Alzheimer’s disease. Alzheimers Dement.
- Li X, Zhang Y, Zhang C, Zheng Y, Liu R, Xiao S (2023) Education counteracts the genetic risk of Alzheimer’s disease without an interaction effect. Front Public Health.
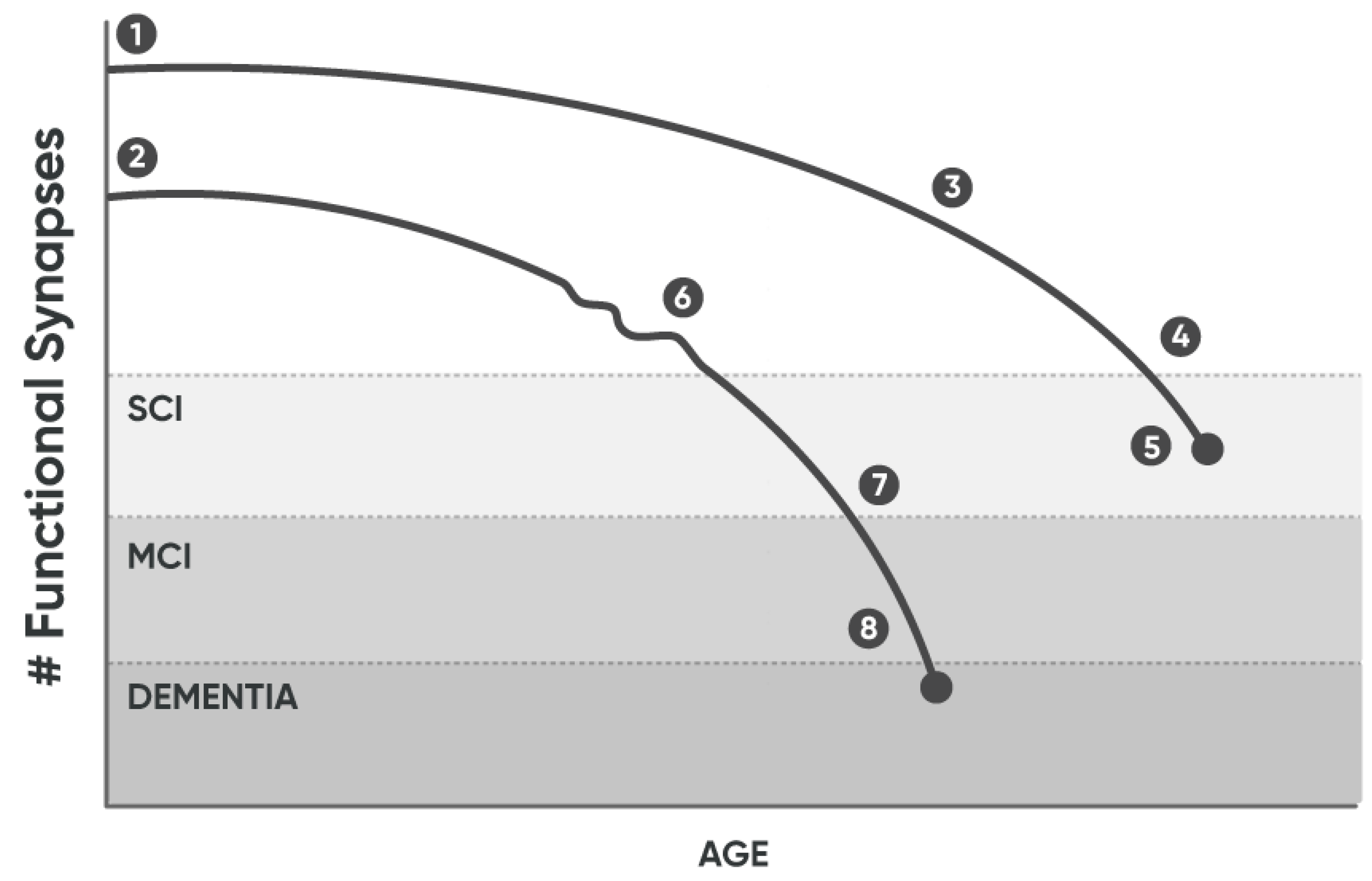
- Vermunt L, van Paasen AJL, Teunissen CE, Scheltens P, Visser PJ, Tijms BM, Initiative ftAsDN (2019) Alzheimer disease biomarkers may aid in the prognosis of MCI cases initially reverted to normal. Neurology.
- Fessel J (2023) Analysis of Why Alzheimer’s Dementia Never Spontaneously Reverses, Suggests the Basis for Curative Treatment. J Clin Med.
- Lourenco FC, Galvan V, Fombonne J, Corset V, Llambi F, Muller U, Bredesen DE, Mehlen P (2009) Netrin-1 interacts with amyloid precursor protein and regulates amyloid-beta production. Cell Death Differ.
- Livingstone RW, Elder MK, Barrett MC, Westlake CM, Peppercorn K, Tate WP, Abraham WC, Williams JM Secreted Amyloid Precursor Protein-Alpha Promotes Arc Protein Synthesis in Hippocampal Neurons. Front Mol Neurosci 2019, 12, 198. [CrossRef]
- Matrone C, Di Luzio A, Meli G, D’Aguanno S, Severini C, Ciotti MT, Cattaneo A, Calissano P (2008) Activation of the amyloidogenic route by NGF deprivation induces apoptotic death in PC12 cells. J Alzheimers Dis.
- Lu DC, Rabizadeh S, Chandra S, Shayya RF, Ellerby LM, Ye X, Salvesen GS, Koo EH, Bredesen DE (2000) A second cytotoxic proteolytic peptide derived from amyloid beta-protein precursor. Nat Med.
- Lu DC, Soriano S, Bredesen DE, Koo EH (2003) Caspase cleavage of the amyloid precursor protein modulates amyloid beta-protein toxicity. J Neurochem.
- Bredesen DE, Mehlen P, Rabizadeh S (2005) Receptors that mediate cellular dependence. Cell Death Differ, 12, 1031–1043.
- Obregon D, Hou H, Deng J, Giunta B, Tian J, Darlington D, Shahaduzzaman M, Zhu Y, Mori T, Mattson MP, Tan J (2012) Soluble amyloid precursor protein-α modulates β-secretase activity and amyloid-β generation. Nat Commun.
- Cottrell BA, Galvan V, Banwait S, Gorostiza O, Lombardo CR, Williams T, Schilling B, Peel A, Gibson B, Koo EH, Link CD, Bredesen DE (2005) A pilot proteomic study of amyloid precursor interactors in Alzheimer’s disease. Ann Neurol.
- Theendakara V, Peters-Libeu CA, Spilman P, Poksay KS, Bredesen DE, Rao RV (2016) Direct Transcriptional Effects of Apolipoprotein E. J Neurosci.
- Ideker T, Galitski T, Hood L (2001) A new approach to decoding life: systems biology. Annu Rev Genomics Hum Genet.
- Powell-Doherty RD, Abbott ARN, Nelson LA, Bertke AS (2020) Amyloid-β and p-Tau Anti-Threat Response to Herpes Simplex Virus 1 Infection in Primary Adult Murine Hippocampal Neurons. J Virol.
- Kurkinen M Lecanemab (Leqembi) is not the right drug for patients with Alzheimer’s disease. Adv Clin Exp Med 2023, 32, 943–947. [CrossRef]
- Dickson DW, Crystal HA, Mattiace LA, Masur DM, Blau AD, Davies P, Yen SH, Aronson MK (1992) Identification of normal and pathological aging in prospectively studied nondemented elderly humans. Neurobiol Aging.
- Kokjohn TA, Maarouf CL, Roher AE (2012) Is Alzheimer’s disease amyloidosis the result of a repair mechanism gone astray? Alzheimers Dement.
- Bredesen DE, Ross MK, Ross S (2024) Sustained Cognitive Improvement in Alzheimer’s Disease Patients Following a Precision Medicine Protocol: Case Series. Biomedicines.
- Twohig D, Nielsen HM (2019) α-synuclein in the pathophysiology of Alzheimer’s disease. Molecular Neurodegeneration.
- Rocca WA, Bower JH, Maraganore DM, Ahlskog JE, Grossardt BR, de Andrade M, Melton LJ, 3rd (2007) Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology, 69, 1074–1083.
- Yu J-T, Xu W, Tan C-C, Andrieu S, Suckling J, Evangelou E, Pan A, Zhang C, Jia J, Feng L, Kua E-H, Wang Y-J, Wang H-F, Tan M-S, Li J-Q, Hou X-H, Wan Y, Tan L, Mok V, Tan L, Dong Q, Touchon J, Gauthier S, Aisen PS, Vellas B Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. Journal of Neurology, Neurosurgery & Psychiatry 2020, 91, 1201–1209.
- Elder GA, Gama Sosa MA, De Gasperi R (2010) Transgenic Mouse Models of Alzheimer’s Disease. Mount Sinai Journal of Medicine: A Journal of Translational and Personalized Medicine.
- Marsh SE, Abud EM, Lakatos A, Karimzadeh A, Yeung ST, Davtyan H, Fote GM, Lau L, Weinger JG, Lane TE, Inlay MA, Poon WW, Blurton-Jones M (2016) The adaptive immune system restrains Alzheimer’s disease pathogenesis by modulating microglial function. Proc Natl Acad Sci U S A, 113, E1316–1325.
- Zhao H, Zhou X, Fu K, Duan Y, Wen Q, Wang S, Zhan S (2024) Prospective cohort study evaluating the association between influenza vaccination and neurodegenerative diseases. npj Vaccines.
- Eyting M, Xie M, Heß S, Geldsetzer P (2023) Causal evidence that herpes zoster vaccination prevents a proportion of dementia cases. medRxiv.
- Lehrer S, Rheinstein PH (2021) Herpes Zoster Vaccination Reduces Risk of Dementia. In Vivo.
- Greenblatt CL, Lathe R (2024) Vaccines and Dementia: Part II. Efficacy of BCG and Other Vaccines Against Dementia. J Alzheimers Dis.
- Chaurasia B, Chavda V, Lu B, Garg K, Montemurro N (2022) Cognitive deficits and memory impairments after COVID-19 (Covishield) vaccination. Brain Behav Immun Health.
- Brown P, Farrell M (2015) A practical approach to avoiding iatrogenic Creutzfeldt-Jakob disease (CJD) from invasive instruments. Infect Control Hosp Epidemiol.
- Marreiros R, Müller-Schiffmann A, Bader V, Selvarajah S, Dey D, Lingappa VR, Korth C Viral capsid assembly as a model for protein aggregation diseases: Active processes catalyzed by cellular assembly machines comprising novel drug targets. Virus Res 2015, 207, 155–164. [CrossRef] [PubMed]
- Jia J, Ning Y, Chen M, Wang S, Yang H, Li F, Ding J, Li Y, Zhao B, Lyu J, Yang S, Yan X, Wang Y, Qin W, Wang Q, Li Y, Zhang J, Liang F, Liao Z, Wang S (2024) Biomarker Changes during 20 Years Preceding Alzheimer’s Disease. New England Journal of Medicine.
- Bredesen DE, Toups K, Hathaway A, Gordon D, Chung H, Raji C, Boyd A, Hill BD, Hausman-Cohen S, Attarha M, Chwa WJ, Kurakin A, Jarrett M Precision Medicine Approach to Alzheimer’s Disease: Rationale and Implications. J Alzheimers Dis 2023, 96, 429–437.
- Sinclair E, Trivedi DK, Sarkar D, Walton-Doyle C, Milne J, Kunath T, Rijs AM, de Bie RMA, Goodacre R, Silverdale M, Barran P (2021) Metabolomics of sebum reveals lipid dysregulation in Parkinson’s disease. Nature Communications.
- Yu SF, Michon M, Lingappa AF, Paulvannan K, Solas D, Staats K, Ichida J, Dey D, Rosenfeld J, Lingappa VR (2025) An ALS assembly modulator signature in peripheral blood mononuclear cells: implications for ALS pathophysiology, therapeutics, and diagnostics. Clin Proteomics.
- Murros KE, Huynh VA, Takala TM, Saris PEJ (2021) Desulfovibrio Bacteria Are Associated With Parkinson’s Disease. Front Cell Infect Microbiol.
- Duan QQ, Jiang Z, Su WM, Gu XJ, Wang H, Cheng YF, Cao B, Gao X, Wang Y, Chen YP (2023) Risk factors of amyotrophic lateral sclerosis: a global meta-summary. Front Neurosci.
- Chakravarthy U, Wong TY, Fletcher A, Piault E, Evans C, Zlateva G, Buggage R, Pleil A, Mitchell P (2010) Clinical risk factors for age-related macular degeneration: a systematic review and meta-analysis. BMC Ophthalmology.
- Nakatsuji T, Kao MC, Zhang L, Zouboulis CC, Gallo RL, Huang CM (2010) Sebum free fatty acids enhance the innate immune defense of human sebocytes by upregulating beta-defensin-2 expression. J Invest Dermatol.
- Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, Challis C, Schretter CE, Rocha S, Gradinaru V, Chesselet MF, Keshavarzian A, Shannon KM, Krajmalnik-Brown R, Wittung-Stafshede P, Knight R, Mazmanian SK Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell 2016, 167, 1469–1480.
- Adlimoghaddam A, Benson T, Albensi BC (2022) Mitochondrial Transfusion Improves Mitochondrial Function Through Up-regulation of Mitochondrial Complex II Protein Subunit SDHB in the Hippocampus of Aged Mice. Mol Neurobiol.
- Ruiz-Pozo VA, Tamayo-Trujillo R, Cadena-Ullauri S, Frias-Toral E, Guevara-Ramírez P, Paz-Cruz E, Chapela S, Montalván M, Morales-López T, Simancas-Racines D, Zambrano AK (2023) The Molecular Mechanisms of the Relationship between Insulin Resistance and Parkinson’s Disease Pathogenesis. Nutrients.
- Rothstein JD, Tsai G, Kuncl RW, Clawson L, Cornblath DR, Drachman DB, Pestronk A, Stauch BL, Coyle JT (1990) Abnormal excitatory amino acid metabolism in amyotrophic lateral sclerosis. Ann Neurol.
- Bradley WG, Cox PA (2009) Beyond Guam: cyanobacteria, BMAA and sporadic amyotrophic lateral sclerosis. Amyotroph Lateral Scler.
- Wirsching I, Ort N, Üçeyler N ALS or ALS mimic by neuroborreliosis-A case report. Clin Case Rep 2020, 8, 86–91. [CrossRef]
- Caller TA, Doolin JW, Haney JF, Murby AJ, West KG, Farrar HE, Ball A, Harris BT, Stommel EW (2009) A cluster of amyotrophic lateral sclerosis in New Hampshire: a possible role for toxic cyanobacteria blooms. Amyotroph Lateral Scler.
- Johnson FO, Atchison WD (2009) The role of environmental mercury, lead and pesticide exposure in development of amyotrophic lateral sclerosis. Neurotoxicology.
- Chen QY, Wu P, Wen T, Qin X, Zhang R, Jia R, Jin J, Hu F, Xie X, Dang J (2022) Association of cerebral spinal fluid copper imbalance in amyotrophic lateral sclerosis. Frontiers in Aging Neuroscience, 2022.
- Alfahad T, Nath A (2013) Retroviruses and amyotrophic lateral sclerosis. Antiviral Res.
- Daneshvar DH, Mez J, Alosco ML, Baucom ZH, Mahar I, Baugh CM, Valle JP, Weuve J, Paganoni S, Cantu RC, Zafonte RD, Stern RA, Stein TD, Tripodis Y, Nowinski CJ, McKee AC (2021) Incidence of and Mortality From Amyotrophic Lateral Sclerosis in National Football League Athletes. JAMA Network Open, 8801.
- Harrison D, Mehta P, van Es MA, Stommel E, Drory VE, Nefussy B, van den Berg LH, Crayle J, Bedlack R “ALS reversals”: demographics, disease characteristics, treatments, and co-morbidities. Amyotroph Lateral Scler Frontotemporal Degener 2018, 19, 495–499.
- Isaacson RS, Ganzer CA, Hristov H, Hackett K, Caesar E, Cohen R, Kachko R, Meléndez-Cabrero J, Rahman A, Scheyer O, Hwang MJ, Berkowitz C, Hendrix S, Mureb M, Schelke MW, Mosconi L, Seifan A, Krikorian R (2018) The clinical practice of risk reduction for Alzheimer’s disease: A precision medicine approach. Alzheimers Dement, 14, 1663–1673.
- Itzhaki RF, Lathe R, Balin BJ, Ball MJ, Bearer EL, Braak H, Bullido MJ, Carter C, Clerici M, Cosby SL, Del Tredici K, Field H, Fulop T, Grassi C, Griffin WS, Haas J, Hudson AP, Kamer AR, Kell DB, Licastro F, Letenneur L, Lovheim H, Mancuso R, Miklossy J, Otth C, Palamara AT, Perry G, Preston C, Pretorius E, Strandberg T, Tabet N, Taylor-Robinson SD, Whittum-Hudson JA (2016) Microbes and Alzheimer’s Disease. J Alzheimers Dis.
- Kandimalla R, Thirumala V, Reddy PH Is Alzheimer’s disease a Type 3 Diabetes? A critical appraisal. Biochim Biophys Acta Mol Basis Dis 2017, 1863, 1078–1089. [CrossRef] [PubMed]
- Karran E, De Strooper B (2016) The amyloid cascade hypothesis: are we poised for success or failure? J Neurochem.
- Kametani F, Hasegawa M (2018) Reconsideration of Amyloid Hypothesis and Tau Hypothesis in Alzheimer’s Disease. Front Neurosci.
- Huang WJ, Zhang X, Chen WW (2016) Role of oxidative stress in Alzheimer’s disease. Biomed Rep.
- Bartzokis G, Lu PH, Mintz J Human brain myelination and amyloid beta deposition in Alzheimer’s disease. Alzheimers Dement 2007, 3, 122–125. [CrossRef] [PubMed]
- Alzheimer’s Prevention and Reversal Project I, Precision Medicine Approach for Early Dementia & Mild Cognitive Impairment (EVANTHEA), https://clinicaltrials.gov/study/NCT05894954?cond=Cognitive%20Decline&term=reversal&rank=5, Accessed 3/25/2025.
- Hashimoto M, Rockenstein E, Mante M, Mallory M, Masliah E (2001) beta-Synuclein inhibits alpha-synuclein aggregation: a possible role as an anti-parkinsonian factor. Neuron.
- Greene LE, Saba F, Silberman RE, Zhao X (2020) Mechanisms for Curing Yeast Prions. Int J Mol Sci.
- Holmes DL, Lancaster AK, Lindquist S, Halfmann R (2013) Heritable remodeling of yeast multicellularity by an environmentally responsive prion. Cell, 153, 153–165.
- Kurakin A, Bredesen DE (2015) Dynamic self-guiding analysis of Alzheimer’s disease. Oncotarget, 6, 14092–14122.
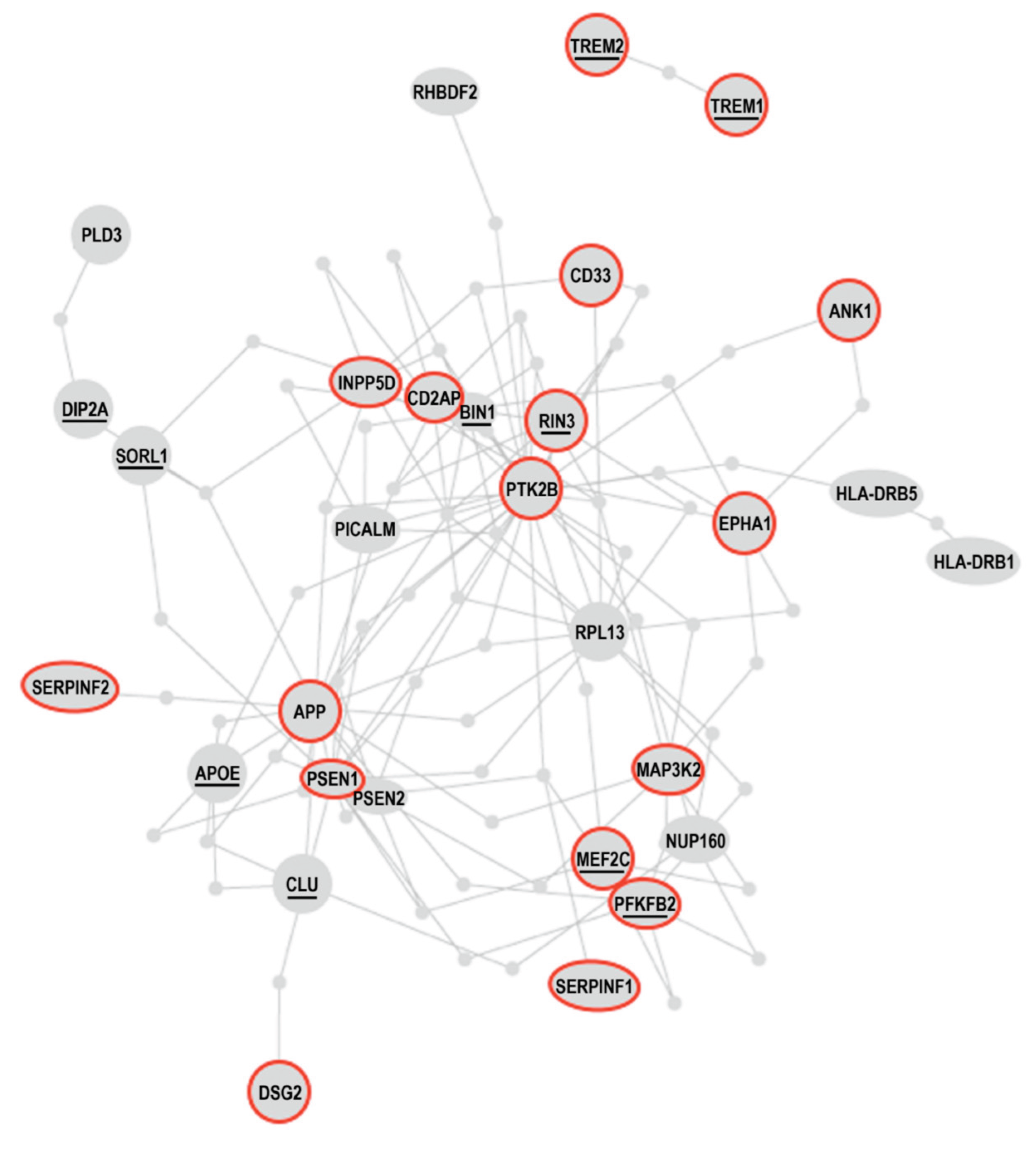
- De Jager PL, Srivastava G, Lunnon K, Burgess J, Schalkwyk LC, Yu L, Eaton ML, Keenan BT, Ernst J, McCabe C, Tang A, Raj T, Replogle J, Brodeur W, Gabriel S, Chai HS, Younkin C, Younkin SG, Zou F, Szyf M, Epstein CB, Schneider JA, Bernstein BE, Meissner A, Ertekin-Taner N, Chibnik LB, Kellis M, Mill J, Bennett DA Alzheimer’s disease: early alterations in brain DNA methylation at ANK1, BIN1, RHBDF2 and other loci. Nat Neurosci 2014, 17, 1156–1163.
| Effect | References |
|---|---|
| Proteinaceous infectious agents (prions) | [4] |
| Proteinaceous anti-infectious agents (“praion” effect of prions) | [14] |
| Metal-binding agents (anti-biofilm agents?) | [21] |
| Physiological response amplifiers of signals (“prason” effect of prions; i.e., switches) | [23] |
| Mediators of the proteinaceous inheritance of traits (“printon” effect of prions) | [24] |
| Pre-inflammatory immune mediators (“priion” effect of prions) | Proposed here |
| Feature | Amnestic presentation | Non-amnestic presentation | Comments |
|---|---|---|---|
| Typical presenting symptoms | Episodic memory loss | Executive dysfunction, prosopagnosia, dyscalculia, PCA, PPA (LV), or CBS | |
| Age of symptom onset | Usually >60, rarely 40s or 50s | Commonly 40s or 50s | Initial symptoms of the non-amnestic presentations often appear at peri-menopause or menopause. |
| Family history | Usually positive | Often negative | |
| Frequency | About 2/3 of patients | About 1/3 of patients | |
| ApoE alleles | Rarely ApoE4-negative | Commonly ApoE4-negative | With ApoE4, the predominantly non-amnestic presentations often include some memory loss with their other symptoms. |
| Preceding depression | Uncommon | Common | |
| Effect on job performance | Often mild initially | Often severe even early on | |
| Progression of symptoms | Tends to be slower | Tends to be more rapid | |
| Dominant neuroanatomical regions | Entorhinal cortex, hippocampus, locus coeruleus (temporal lobe > parietal) | Parietal lobe, HPA axis (parietal lobe > temporal) | |
| Typical contributors | Metabolic syndrome, inflammation | Toxins, toxicants, tick-borne infections | |
| Response pattern | Programmatic synaptoclasis due to endogenous metabolic or infectious insult? | Non-programmatic synaptoclasis due to exogenous toxic or infectious insult? | |
| Effect of stress | Mild exacerbation | Marked exacerbation | May be related to HPA axis effects. |
| Leukopenia | Uncommon | Common | May be due to immunosuppression from biotoxin. |
| Thrombocytopenia | Uncommon | Common | |
| Reduced glutathione | Uncommon | Common | May be due to chronic, ongoing detoxification. |
| Increased TGF-β1 and MMP-9 | Uncommon | Common | Common response to biotoxins and some chronic infections. |
| Physiological function | Connection mode | Protection mode | Comment |
|---|---|---|---|
| APP processing and signaling | sAPPα, αCTF | sAPPβ, Aβ, Jcasp, C31 | Amyloid-β is part of protection mode. |
| Tau structure and function | Tau binds, stabilizes microtubules | P-tau is antimicrobial protein, prion | [54] |
| EMT | Epithelial | Mesenchymal | Alters organization, respiration, energy |
| Thrombosis | Anti-thrombotic | Pro-thrombotic | Multiple mechanisms |
| Immune response | Anti-inflammatory | Pro-inflammatory | Aβ is part of innate immunity. |
| Autonomic nervous system | Parasympathetic dominant | Sympathetic dominant | |
| Microglial type | M2, anti-inflammatory | M1, pro-inflammatory | |
| Trp metabolism | 5-HIAA (5HT)--transmission | QA—anti-microbial, inflammatory | QA in CNS is microglial product. |
| ApoE4 signaling | ApoE4-//-DNA; lack of DNA binding prevents repression | ApoE4 binds DNA → transc. repression of SirT1 et al. | ApoE4 functions as intercellular (and intracellular) transcription factor. R251G removes transcriptional repression. |
| Blood-brain barrier | Tight | Leaky | |
| GSK-3β | Inhibited | Active, phosphorylates tau | Li+ inhibits |
| Aβ polymerization | Monomeric | Oligomeric, anti-microbial, prionic | Homotaurine and modified citrus pectin inhibit oligomerization. |
| Neurite outgrowth | High reelin | Low reelin | Migration of neurons and processes |
| Hippo signaling | Autophagy | Reduced autophagy, increasing Aβ |
| Neuroplasticity | Motor modulation | Power amplification | Color and fine vision | |
|---|---|---|---|---|
| Diseases | AD, LBD, LATE, FTD | PD, PSP, CBD | ALS | AMD |
| Network vulnerability | Energetics, inflammation | Mitochondrial complex I | Excitotoxicity | Metabolic demand, complement |
| Common contributors | Metabolic syndrome, chronic infections, OSA, air pollution | Organic toxicants | Head trauma, physical stress, heavy metals, pesticides | Cigarette smoking, high-energy light, OSA |
| Early detection | p-tau 217, Aβ42:40 (plasma or CSF), PET, speech patterns, EOMs |
18F-DOPA PET, LC-MS sebum, skin biopsy, pre-motor Sx (RBD, anosmia, constipation) |
[74] | OCT, DA, CAP |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




