Submitted:
28 August 2025
Posted:
29 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Design and Recruitment
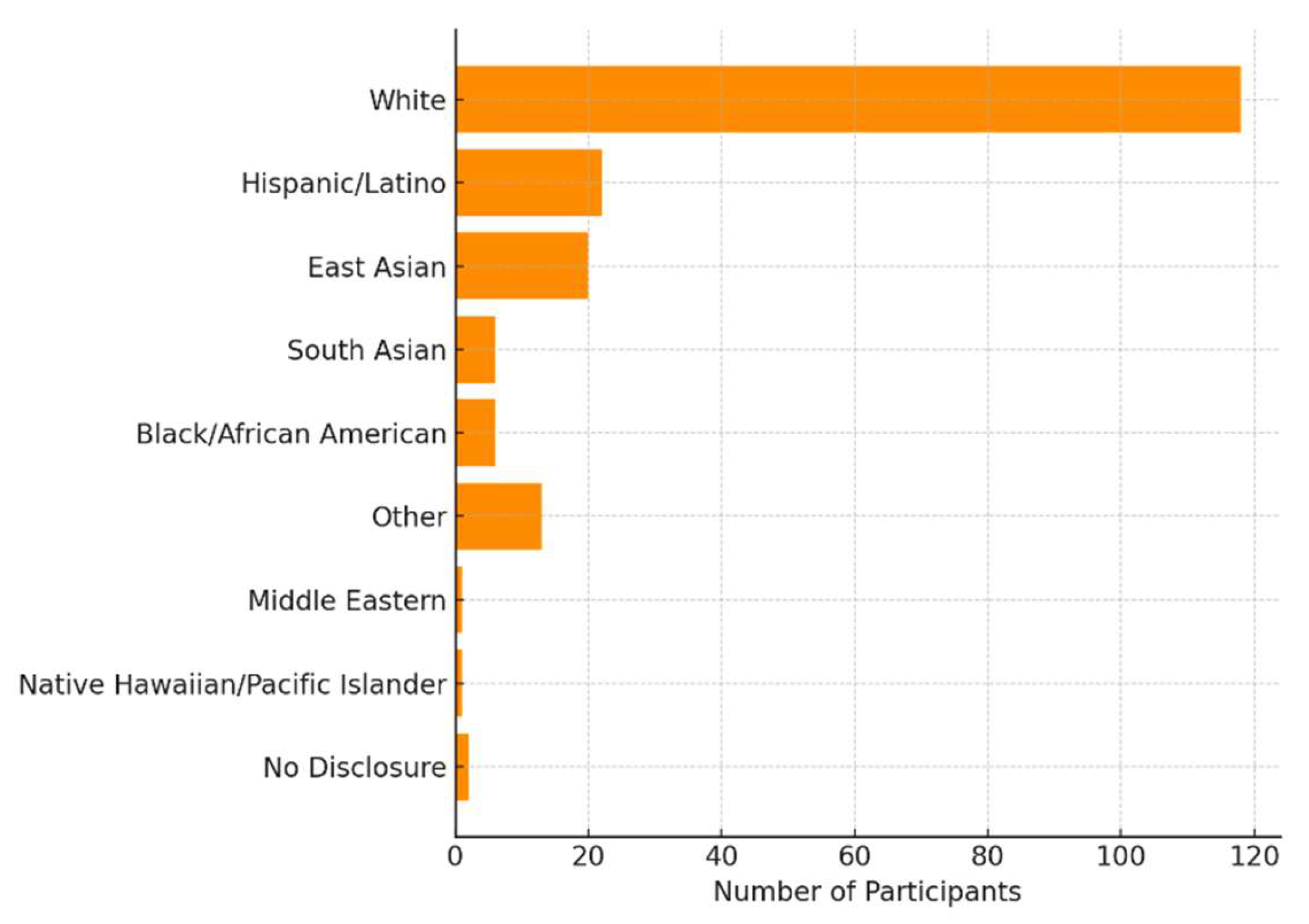
2.2. Participants
2.3. Ethical Considerations
2.4. Survey Materials
2.5. Data Analysis Strategy
3. Results
3.1. Descriptive Characteristics of the Sample
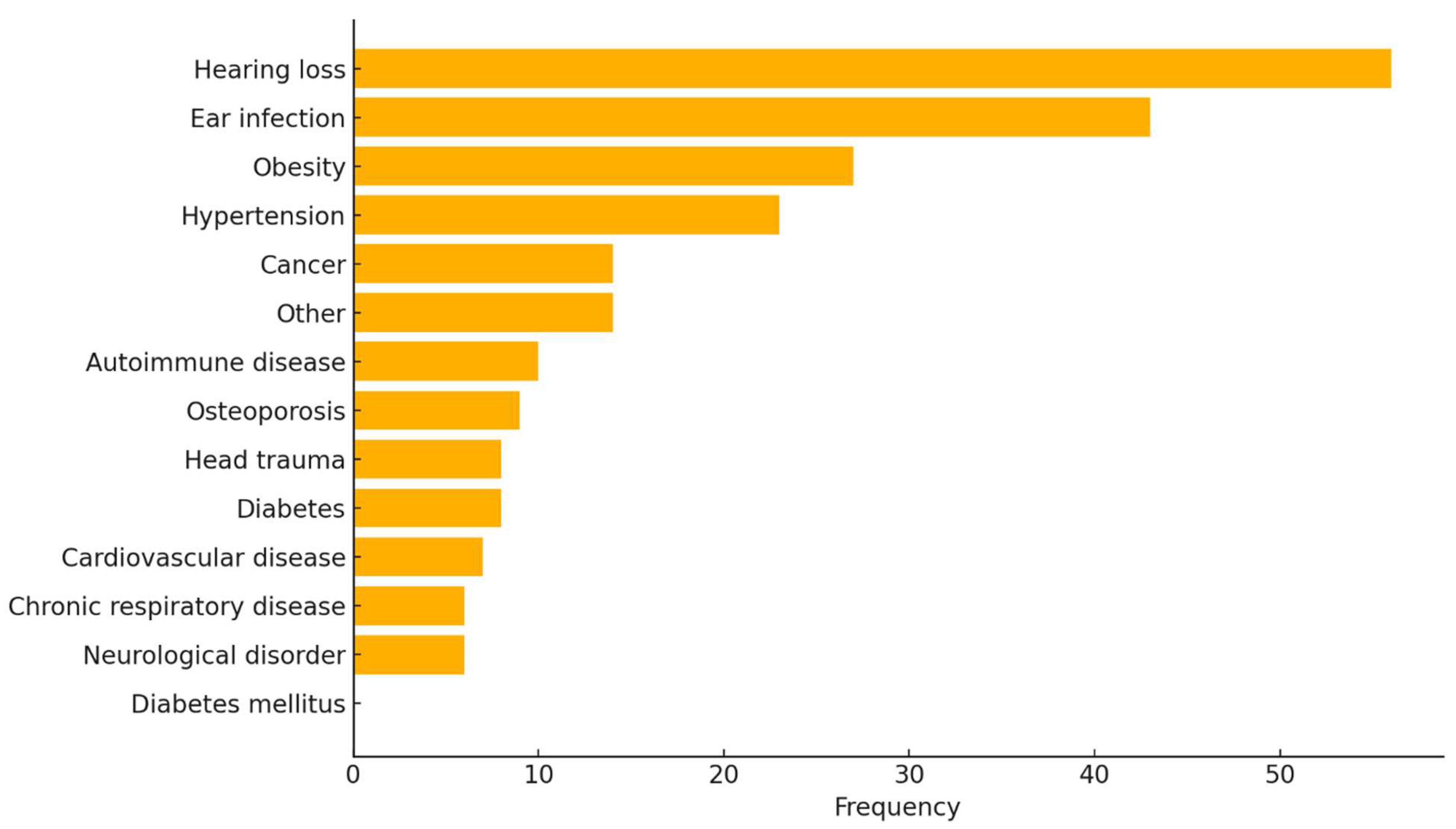
3.2. Medical History of Participants
3.3. COVID-19 Vaccination Status
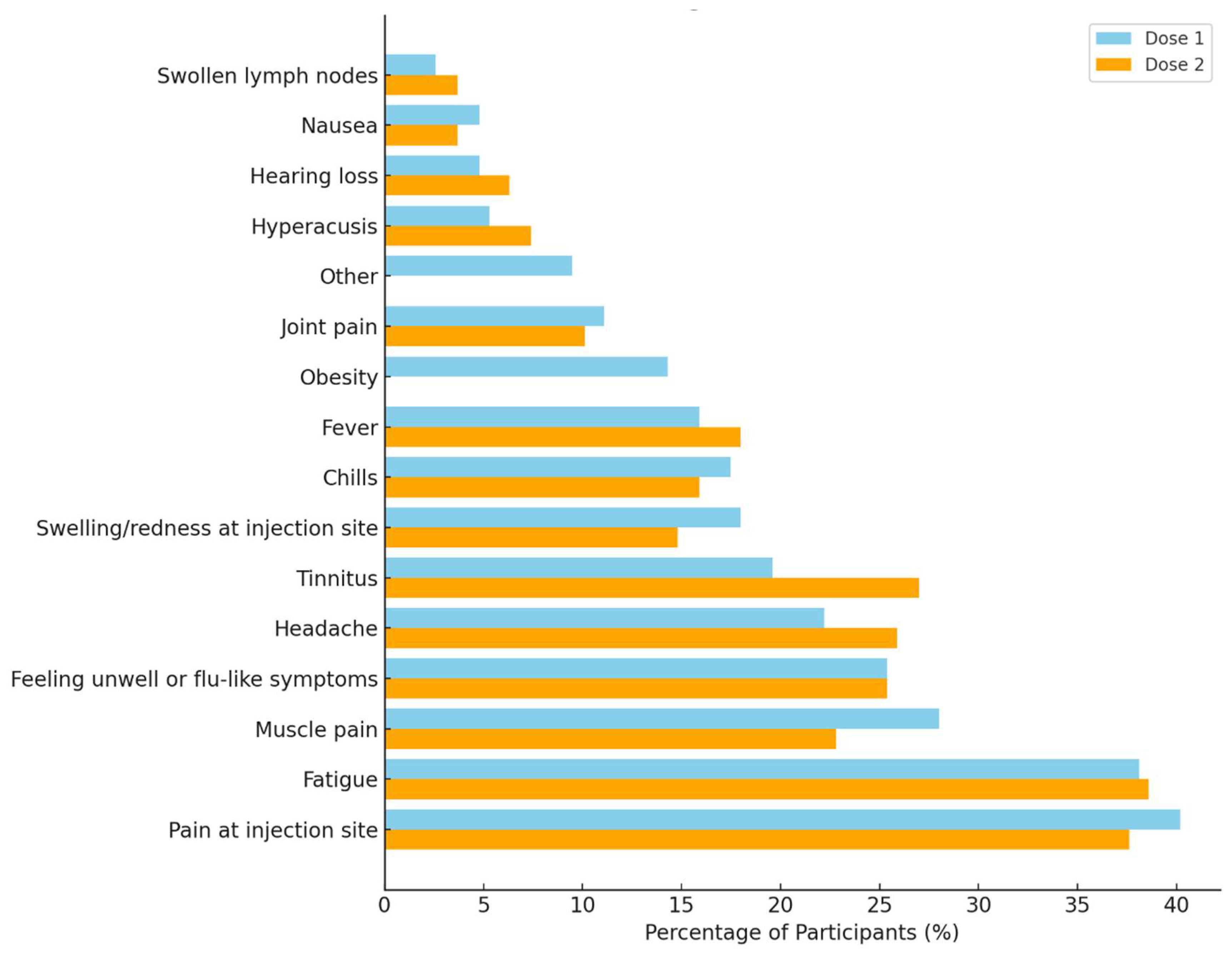
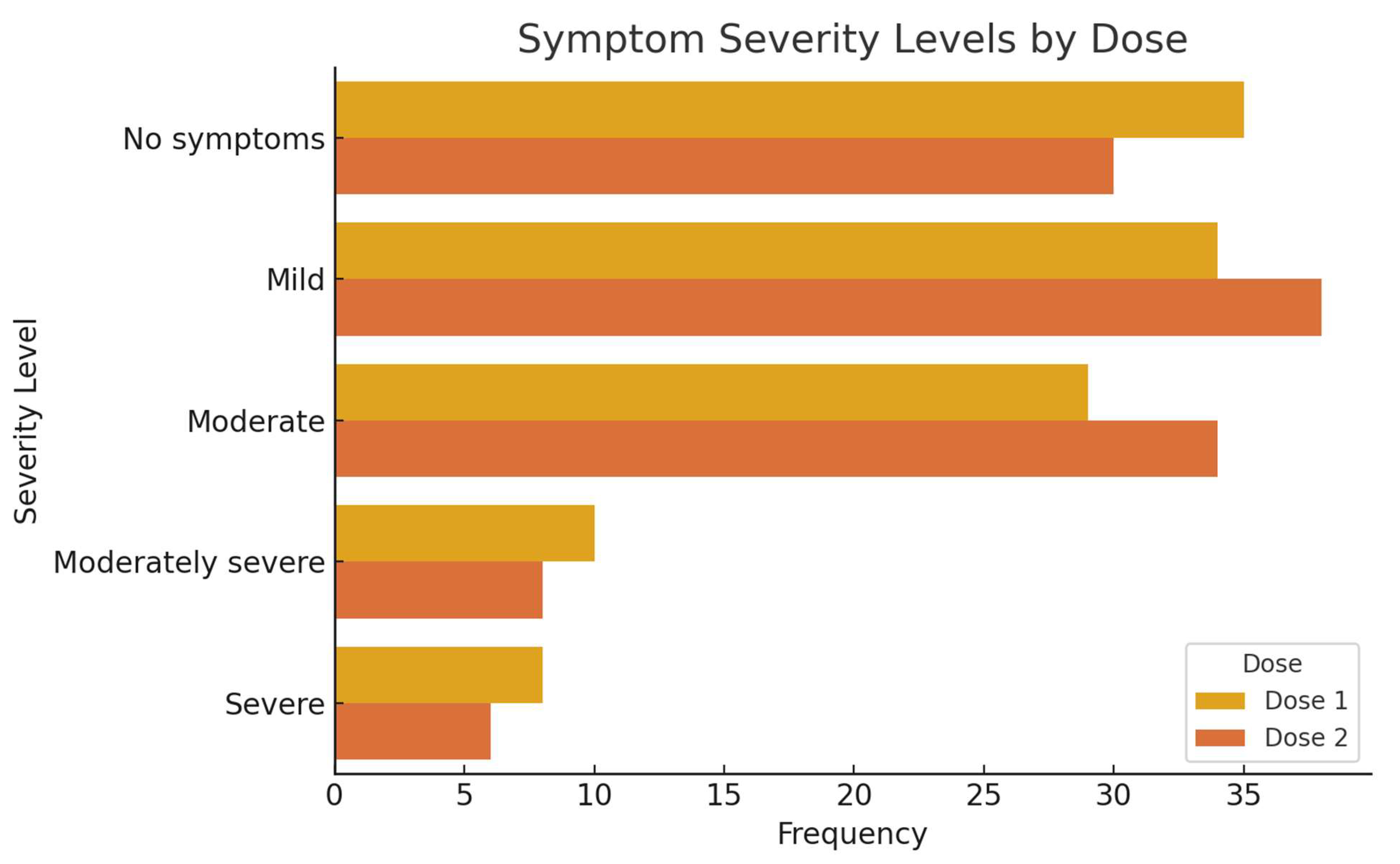
3.4. Adverse Reactions and Symptom Severity Following COVID-19 Vaccination
| Severity Level | Dose 1 Male | Dose 1 Female | Dose 2 Male | Dose 2 Female |
|---|---|---|---|---|
| No symptoms | 21 | 14 | 17 | 13 |
| Mild | 17 | 17 | 16 | 22 |
| Moderate | 11 | 18 | 13 | 21 |
| Moderately severe | 4 | 6 | 2 | 6 |
| Severe | 3 | 5 | 2 | 4 |
3.5. Tinnitus Onset Following COVID-19 Vaccination
3.6. COVID-19 Infection Status and Timing Relative to Vaccination
3.7. COVID-19-Related Hospitalization
| Variable | Yes (n, %) | No (n, %) |
|---|---|---|
| Tinnitus after first dose | 49 (25.9%) | 140 (74.1%) |
| Tinnitus after second dose | 37 (19.6%)* | 79 (41.8%)* |
| COVID-19 infection prior to vaccination | 34 (18.0%) | 155 (82.0%) |
| COVID-19 positive between vaccine doses | 43 (22.8%) | 146 (77.2%) |
3.8. Longitudinal Changes in Tinnitus Handicap Inventory (THI) Scores
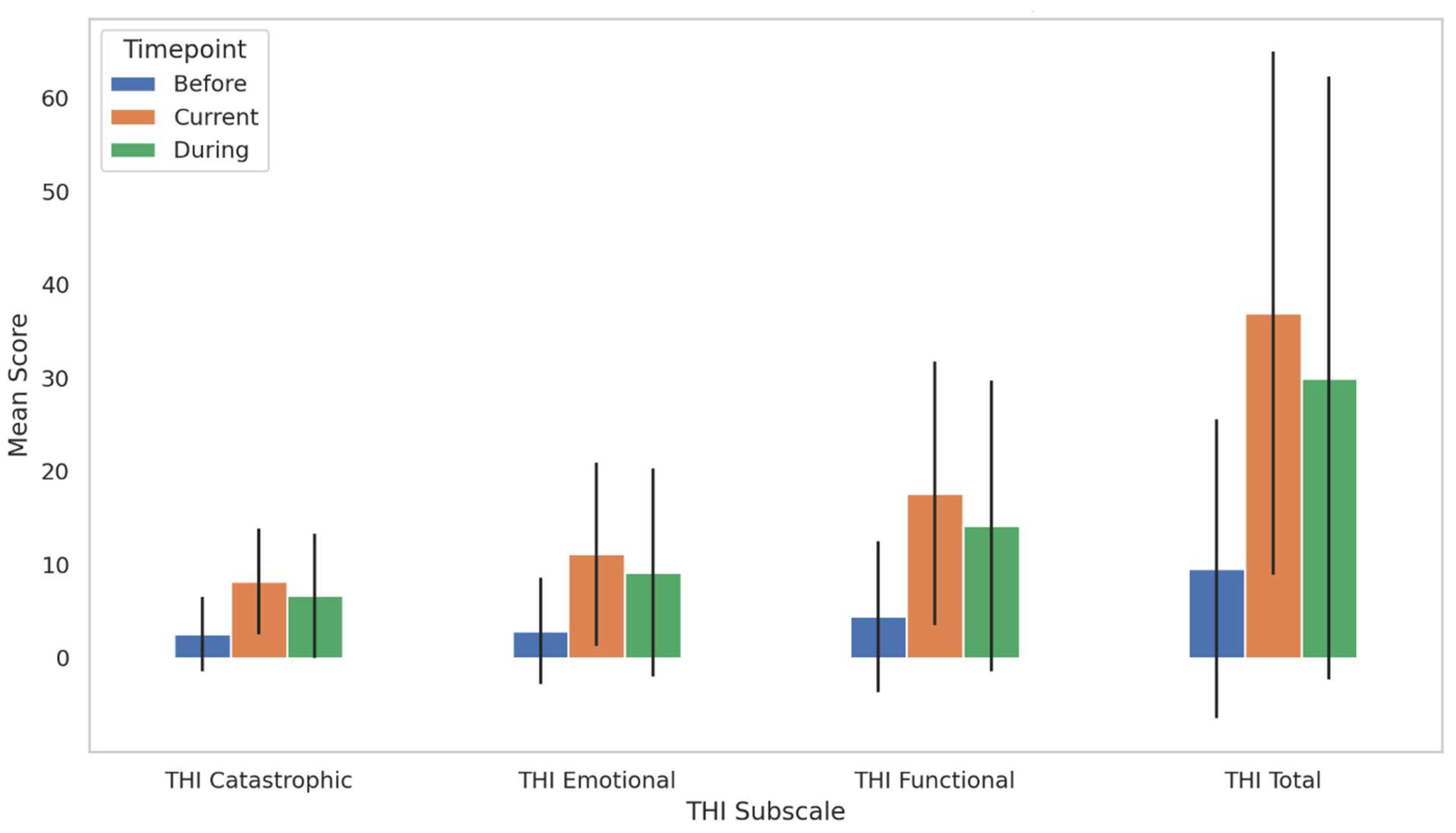
3.8.1. THI Functional Subscale
3.8.2. THI Emotional Subscale
3.8.3. THI Catastrophic Subscale
3.9. Tinnitus Reaction Questionnaire (TRQ)
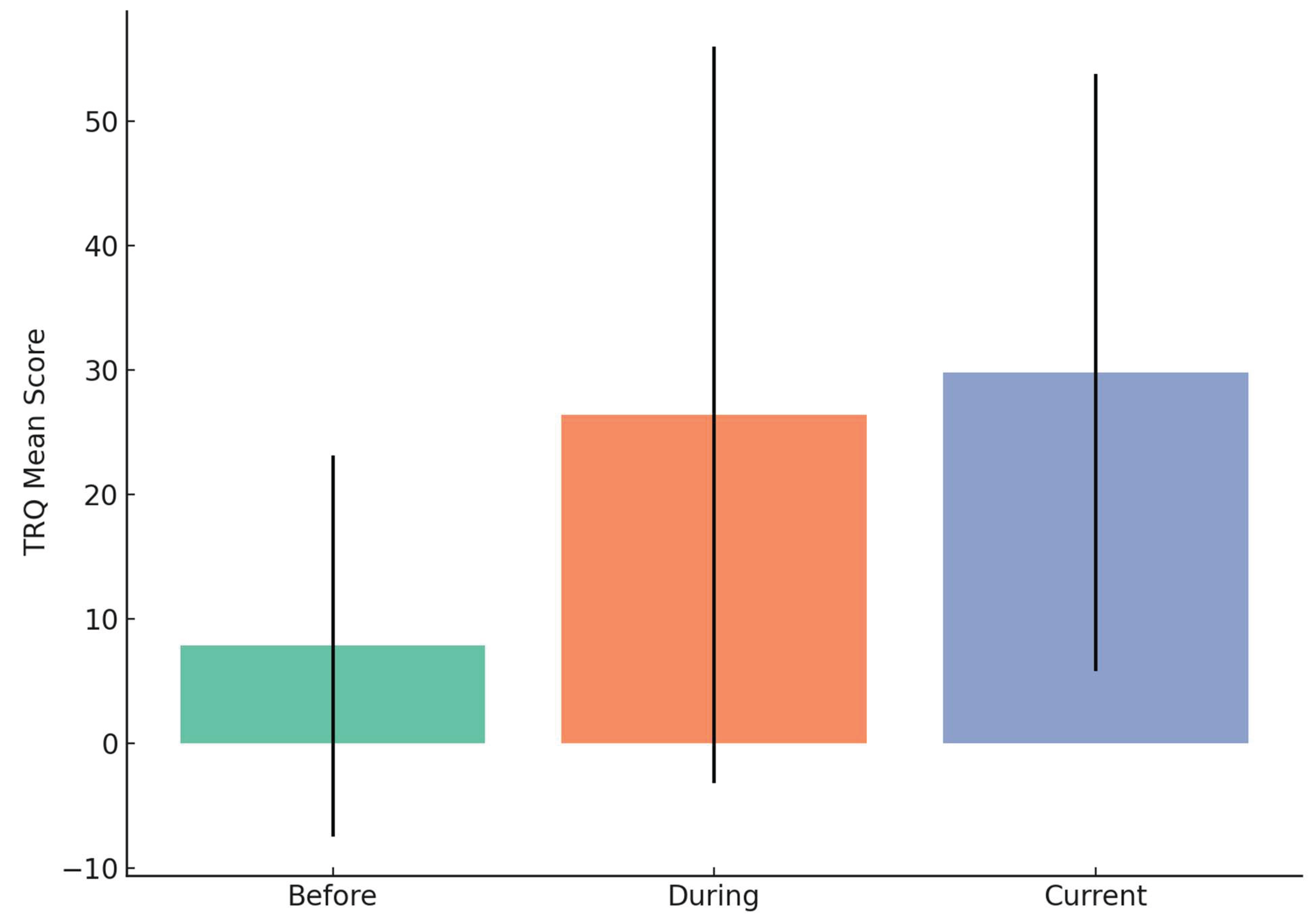
TRQ Severity Levels Across Timepoints
| TRQ severity levels | Before | During | Current |
|---|---|---|---|
| No/Slight | 87 | 55 | 36 |
| Mild | 14 | 21 | 36 |
| Moderate | 4 | 12 | 18 |
| Severe | 1 | 7 | 11 |
| Very Severe | 1 | 12 | 6 |
4. Discussion
5. Conclusions
6. Limitations
7. Future Directions
Ethics Statement
Declaration of the Use of Artificial Intelligence (AI) in Manuscript Preparation
Conflicts of Interest Statement
References
- Aydogan, Z., Can, M., Soylemez, E., Karakoc, K., Buyukatalay, Z. C., & Tokgoz Yilmaz, S. (2024). The effects of COVID-19 on tinnitus severity and quality of life in individuals with subjective tinnitus. Brain and Behavior, 14(3), e7031. [CrossRef]
- Wang, W., Yellamsetty, A., Edmonds, R. M., Barcavage, S. R., & Bao, S. (2024). COVID-19 vaccination-related tinnitus is associated with pre-vaccination metabolic disorders. Frontiers in Pharmacology, 15, 1374320. [CrossRef]
- Beukes, E. W., Baguley, D. M., Jacquemin, L., Lourenco, M. P., & Manchaiah, V. (2020). Changes in tinnitus experiences during the COVID-19 pandemic. Frontiers in Public Health, 8, 592878. [CrossRef]
- Beukes, E., Ulep, A. J., Eubank, T., & Manchaiah, V. (2021). The impact of COVID-19 and the pandemic on tinnitus: A systematic review. Journal of Clinical Medicine, 10(13), 2763. [CrossRef]
- Cullen, W., Gulati, G., & Kelly, B. D. (2020). Mental health in the COVID-19 pandemic. QJM: An International Journal of Medicine, 113(5), 311–312. [CrossRef]
- Wang, D., Li, P., Huang, X., Liu, Y., Mao, S., Yin, H., Wang, N., Luo, Y., & Sun, S. (2024). Exploring the prevalence of tinnitus and ear-related symptoms in China after the COVID-19 pandemic: Online cross-sectional survey. JMIR Formative Research, 8, e54326. [CrossRef]
- Kennedy, V., Wilson, C., & Stephens, D. (2004). Quality of life and tinnitus. Audiological Medicine, 2(1), 29–40.
- Mazurek, B., Szczepek, A. J., & Hébert, S. (2015). Stress and tinnitus. HNO, 63(4), 258–265. [CrossRef]
- Newman, C. W., Jacobson, G. P., & Spitzer, J. B. (1996). Development of the Tinnitus Handicap Inventory. Archives of Otolaryngology–Head & Neck Surgery, 122(2), 143–148. [CrossRef]
- Pfefferbaum, B., & North, C. S. (2020). Mental health and the Covid-19 pandemic. The New England Journal of Medicine, 383(6), 510–512. [CrossRef]
- Tai, Y., Jain, N., Kim, G., & Husain, F. T. (2024). Tinnitus and COVID-19: Effect of infection, vaccination, and the pandemic. Frontiers in Public Health, 12, 1508607. [CrossRef]
- Taylor, S. (2019). The psychology of pandemics: Preparing for the next global outbreak of infectious disease. Cambridge Scholars Publishing.
- Taylor, S., Landry, C. A., Paluszek, M. M., Fergus, T. A., McKay, D., & Asmundson, G. J. G. (2020). Development and initial validation of the COVID Stress Scales. Journal of Anxiety Disorders, 72, 102232. [CrossRef]
- Taylor, S. (2022). The psychology of pandemics. Annual Review of Clinical Psychology, 18, 581–609. [CrossRef]
- Wilson, P. H., Henry, J., Bowen, M., & Haralambous, G. (1991). Tinnitus Reaction Questionnaire: Psychometric properties of a measure of distress associated with tinnitus. Journal of Speech and Hearing Research, 34(1), 197–201.
- Xiong, J., Lipsitz, O., Nasri, F., Lui, L. M. W., Gill, H., Phan, L., Chen-Li, D., Iacobucci, M., Ho, R., Majeed, A., & McIntyre, R. S. (2020). Impact of COVID-19 pandemic on mental health in the general population: A systematic review. Journal of Affective Disorders, 277, 55–64. [CrossRef]
- Yellamsetty, A., Etu, E.-E., & Bao, S. (2025). Impact of COVID-19 vaccination on tinnitus onset and severity: A comprehensive survey study. Frontiers in Audiology and Otology, 3, Article 1509444. [CrossRef]
- Yellamsetty, A. (2024a). COVID-19 vaccination effects on tinnitus and hyperacusis: Longitudinal case study. The International Tinnitus Journal, 27(2), 253–258. [CrossRef]
- Yellamsetty, A., Fortuna, G., Egbe-Etu, E., & Bao, S. (Submitted). Functional and perceptual impacts of tinnitus following COVID-19 vaccination: Implications for public health surveillance and communication. Frontiers in Audiology and Otology.
- Nieminen TA, Kivekäs I, Artama M, Nohynek H, Kujansivu J, Hovi P. (2023). Sudden hearing loss following vaccination against COVID-19. JAMA Otolaryngol Head Neck Surg, 149(2), 133–140. [CrossRef]
- Yanir Y, Doweck I, Shibli R, Najjar-Debbiny R, Saliba W. (2022). Association between the BNT162b2 messenger RNA COVID-19 vaccine and the risk of sudden sensorineural hearing loss. JAMA Otolaryngol Head Neck Surg, 148(4), 299–306. [CrossRef]
- Leong, S., Teh, B., & Kim, A. H. (2023). Characterization of otologic symptoms appearing after COVID-19 vaccination. American Journal of Otolaryngology, 44(2), 103725. [CrossRef]
- Frontera, J. A., Tamborska, A. A., Doheim, M. F., Garcia-Azorin, D., Gezegen, H., Guekht, A., Khan Yusof Khan, A. H., Santacatterina, M., Sejvar, J., Thakur, K. T., Westenberg, E., Winkler, A. S., Beghi, E., & contributors from the Global COVID-19 Neuro Research Coalition. (2022). Neurological events reported after COVID-19 vaccines: An analysis of VAERS. Annals of Neurology, 91(6), 756–771. [CrossRef]
- Formeister EJ, Wu MJ, Chari DA, et al. (2022). Assessment of sudden sensorineural hearing loss after COVID-19 vaccination. JAMA Otolaryngol Head Neck Surg, 148(4), 307–315. [CrossRef]
- Whittaker HR, Gulea C, Koteci A, et al. (2021). GP consultation rates for sequelae after acute COVID-19 in patients managed in the community or hospital in the UK: Population-based study. BMJ, 375, e065834. [CrossRef]
- Kant A, Jansen J, van Balveren L, van Hunsel F. (2022). Description of frequencies of reported adverse events following immunization among four different COVID-19 vaccine brands. Drug Safety, 45(4), 319–331. [CrossRef]
- Dorney I, Bobak L, Otteson T, Kaelber DC. (2023). Prevalence of new-onset tinnitus after COVID-19 vaccination with comparison to other vaccinations. Laryngoscope, 133(7), 1722–1725. [CrossRef]
| Dose | Pfizer-BioNTech | Moderna | Johnson & Johnson | Other |
|---|---|---|---|---|
| First Dose | 100 | 72 | 14 | 2 |
| Second Dose | 91 | 74 | 2 | 2 |
| 1st Booster | 70 | 59 | 1 | 2 |
| 2nd Booster | 41 | 35 | 2 | 3 |
| 3rd Booster | 29 | 20 | 0 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




