Submitted:
31 March 2026
Posted:
01 April 2026
You are already at the latest version
Abstract
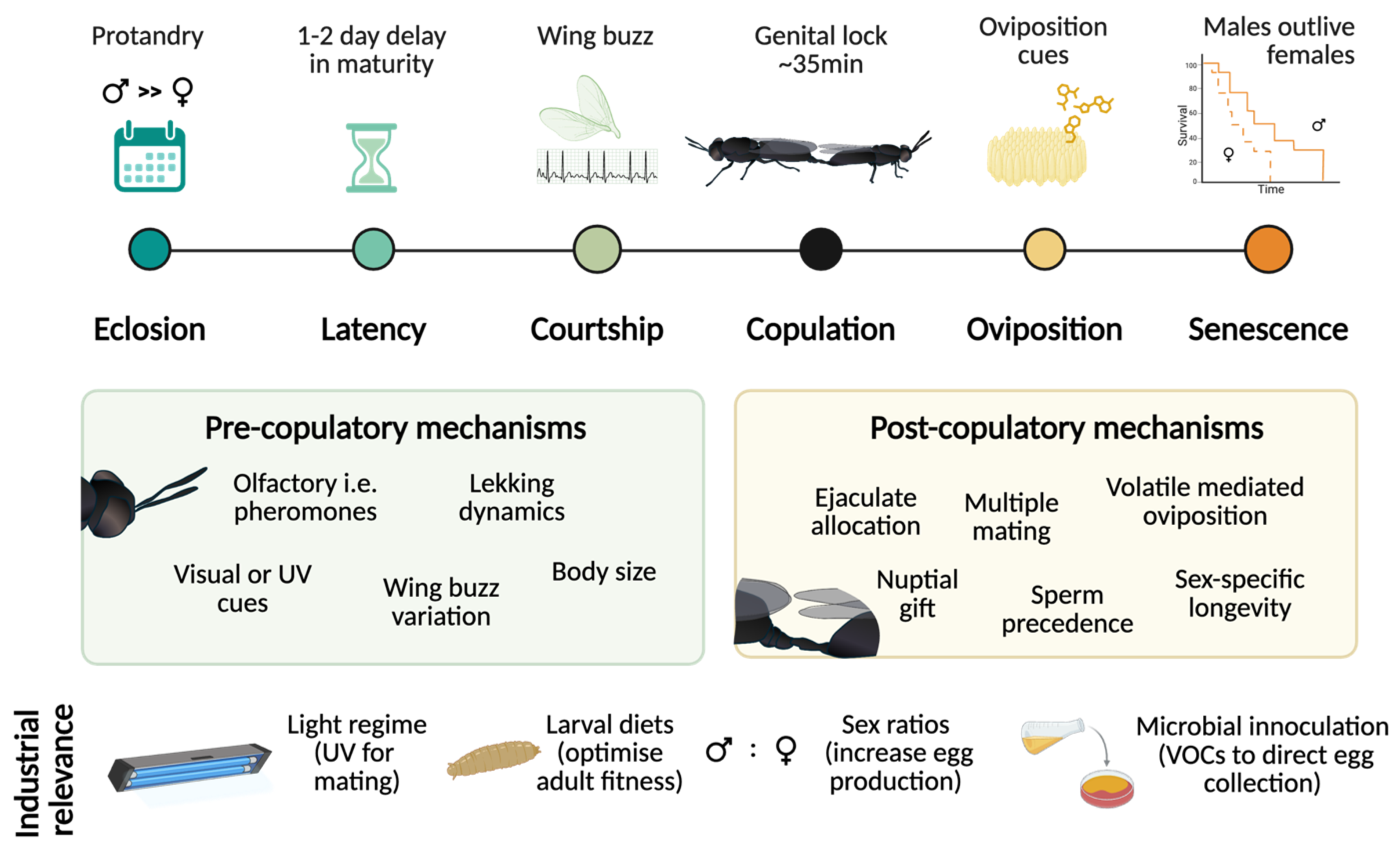
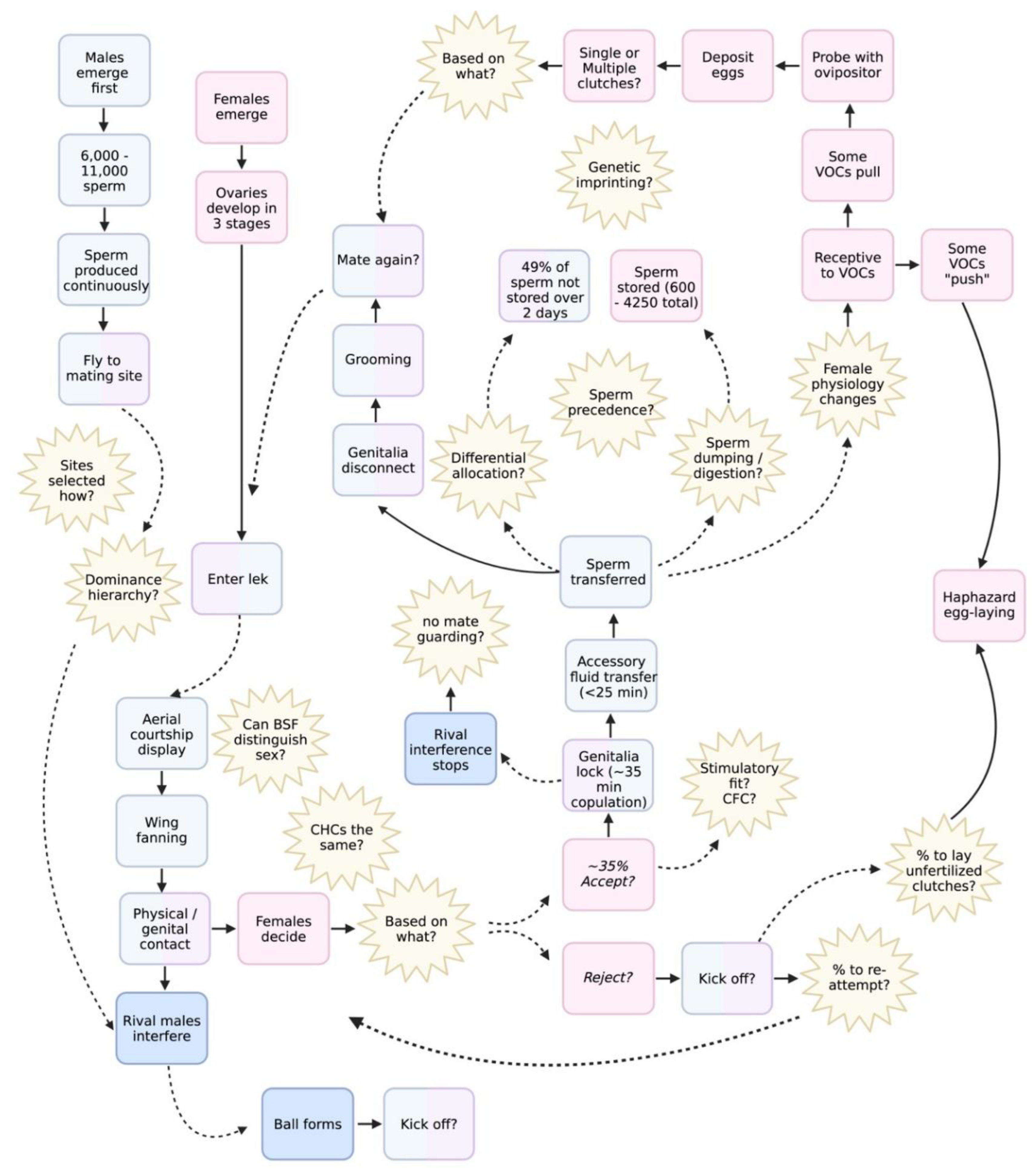
The black soldier fly (BSF; Hermetia illucens) is rapidly emerging as a model for evolutionary biology and insect biotechnology. Although larval biology has been extensively characterised, the reproductive biology of adults remains comparatively understudied. In this review, we synthesise the most recent empirical work on physiology, behavioural and chemical ecology to open the “black box” of BSF reproduction, focusing on processes that span eclosion to senescence. We highlight pre- and post-mating mechanisms that determine overall reproductive fitness: from mating latency, lekking dynamics, courtship and copulation, to sperm transfer, storage and oviposition. We discuss these processes within the framework of sexual selection theory. Several notable characteristics of BSF reproduction differ from traditional insect models. These include a hybrid capital-income breeding strategy (adults do not need to feed but can benefit from supplemental nutrition), protandry (early male emergence), sex-specific longevity that varies with mating status and a lek-like mating system. In addition, females possess morphologically complex sperm‑storage organs, providing ample opportunity for intense post‑copulatory sexual selection. Recent work shows that environmental factors such as light, humidity, temperature, substrate volatiles and rearing design strongly influence reproductive output in industrial settings, highlighting the potential for BSF to bridge fundamental and applied research. We propose a novel conceptual framework that integrates these elements and outline key unresolved questions (e.g., mechanisms of sperm precedence, female control of fertilization, reproductive barriers, drivers of speciation etc.). This interdisciplinary model supports both fundamental insights into the evolution of reproductive traits and provides practical improvements for optimizing industrial mass-rearing.
Keywords:
I. Introduction
- How nutritional legacies acquired during development impact adult fitness and reproductive strategies.
- How light exposure, humidity, temperature and other abiotic conditions, structure the dynamics of a mating system.
- How mating systems evolve and shift under the intense pressures of domestication and artificial selection.
- The relative strengths of pre- and post-copulatory sexual selection in a capital breeder.
- The mechanisms by which reproductive barriers form and contribute to isolation.
II. Mechanisms and Dynamics of Reproduction
- (3)
- Post-copulatory phase
- (a)
- Oviposition
- (b) Aging and senescence
- → BOX 6: Dynamics of oviposition site selection
III. Implications for Evolutionary Biology
- (1)
- Cryptic female choice, sperm competition and sexual conflict
- (2)
- Reproductive barriers and potential drivers of speciation
IV. Implications for Industrial Applications
- (1)
- Genetics and population structure
- (2)
- Nutritional management
- (3)
- Sex ratios and habitat design
- (4)
- Chemical ecology and oviposition control
V. Conceptual Model
VI. Conclusions and Future Directions
- (1)
- BSF is well-poised to be used as a research model to investigate questions in both fundamental evolutionary and applied sciences such as sustainable agriculture, waste management, biotechnology and insect welfare.
- (2)
- As a non-pest, lekking species that is easily culturable, meaning BSF provides an avenue to study reproductive processes that are happening in a wide range of species with similar life history and reproductive traits.
- (3)
- BSF displays a capital-income breeding hybrid strategy, allowing the study of reproductive outcomes that are mediated strictly by larval nutrition, as well as enhanced or otherwise affected by adult nutrition.
- (4)
- Future research priorities include: (i) Identifying the cues used for mate recognition and lek formation in wild and captive context; (ii) Quantifying sperm precedence patterns and female control over fertilization; (iii) Investigating the potential trade-offs between production efficiency (larval size and nutrition), supplemental adult nutrition and breed stock resilience (adult reproduction); (iv) Documenting the variation in BSF phenome with respect to differing genetic provenances and nutritional legacies; (v) Developing management strategies to balance genetic diversity with targeted selection; and (vi) Quantifying the relative strength of pre- and post-copulatory selection.
- (5)
- Achieving these will require the integration of controlled experiments, comparative fieldwork, molecular tools and machine-learning assisted computer vision. Standardizing methodologies such as population densities, light regimes and cage designs, will also improve reproducibility and comparisons across studies.
References
- Addeo, N.F., Li, C., Rusch, T.W., Dickerson, A.J., Tarone, A.M., Bovera, F. & Tomberlin, J.K. (2022) Impact of age, size, and sex on adult black soldier fly Hermetia illucens L. (Diptera: Stratiomyidae) thermal preference. In Journal of Insects as Food and Feed; Wageningen Academic Publishers; Volume 8, pp. 129–139.
- Alcock, J. Convergent evolution in perching and patrolling site preferences of some hilltopping insects of the sonoran desert. In The Southwestern Naturalist; Southwestern Association of Naturalists, 1984; Volume 29, pp. 475–480. [Google Scholar]
- Alcock, J. Leks and hilltopping in insects. In Journal of Natural History; Taylor & Francis, 1987; Volume 21, pp. 319–328. [Google Scholar]
- Alcock, J. A large male competitive advantage in a lekking fly, Hermetia comstocki Williston (Diptera: Stratiomyidae). Psyche: A Journal of Entomology 1990, 97, 267–279. [Google Scholar] [CrossRef]
- Aldersley, A. & Cator, L.J. (2019) Female resistance and harmonic convergence influence male mating success in Aedes aegypti. In Scientific Reports; Nature Publishing Group; Volume 9, p. 2145.
- Ankeny, R.A. & Leonelli, S. What’s so special about model organisms? Studies in History and Philosophy of Science Part A 42, 313–323.
- Athanassiou, C.G., Coudron, C.L., Deruytter, D., Rumbos, C.I., Gasco, L., Gai, F., Sandrock, C., De Smet, J., Tettamanti, G., Francis, A., Petrusan, J.-I. & Smetana, S. (2024) A decade of advances in black soldier fly research: from genetics to sustainability; Brill.
- Awal, Md.R., Rahman, Md.M., Choudhury, Md.A.R., Hasan, Md.M., Rahman, Md.T. & Mondal, Md.F. (2022) Influences of artificial light on mating of black soldier fly (Hermetia illucens)—a review. International Journal of Tropical Insect Science 1–5.
- Baker, C.A., Clemens, J. & Murthy, M. (2019) Acoustic pattern recognition and courtship songs: insights from insects. In Annual Review of Neuroscience; Annual Reviews; Volume 42, pp. 129–147.
- Barbosa, F. Cryptic female choice by female control of oviposition timing in a soldier fly. Behavioral Ecology 2009, 20, 957–960. [Google Scholar] [CrossRef]
- Barbosa, F. Copulation duration in the soldier fly: the roles of cryptic male choice and sperm competition risk. Behavioral Ecology 22, 1332–1336. [CrossRef]
- Barbosa, F. Males responding to sperm competition cues have higher fertilization success in a soldier fly. Behavioral Ecology 23, 815–819. [CrossRef]
- BARBOSA, F. (2015) An integrative view of postcopulatory sexual selection in a soldier fly: Interplay between cryptic male choice and sperm competition. In Cryptic Female Choice in Arthropods: Patterns, Mechanisms and Prospects (eds A.V. PERETTI & A. AISENBERG), pp. 385–401. Springer International Publishing, Cham.
- Barrett, M., Chia, S.Y., Fischer, B. & Tomberlin, J.K. (2022a) Welfare considerations for farming black soldier flies, Hermetia illucens (Diptera: Stratiomyidae): a model for the insects as food and feed industry. Journal of Insects as Food and Feed 1–30.
- Barrett, M., Godfrey, R.K., Sterner, E.J. & Waddell, E.A. (2022b) Impacts of development and adult sex on brain cell numbers in the Black Soldier Fly, Hermetia illucens L. (Diptera: Stratiomyidae). Arthropod Structure & Development 70, 101174.
- Barrett, M., Patel, N., McCarry, B., Shellenberger, G., Schwartz, E., Fiocca, K. & Waddell, E. (2025) Dietary preferences and impacts of feeding on behavior, longevity, and reproduction in adult black soldier flies (Diptera: Stratiomyidae; Hermetia illucens). Journal of Insects as Food and Feed Online 1–12.
- Bateman, A.J. Intra-sexual selection in Drosophila. Heredity 2, 349–368. [CrossRef]
- Beach, F.A. Coital behavior in dogs. IX. Sequelae to “coitus interruptus” in males and females. Physiology & Behavior 5, 263–268.
- Bedhomme, S., Bernasconi, G., Koene, J.M., Lankinen, Å., Arathi, H.S., Michiels, N.K. & Anthes, N. (2009) How does breeding system variation modulate sexual antagonism? In Biology Letters; Royal Society; Volume 5, pp. 717–720.
- Bertinetti, C., Samayoa, A.C. & Hwang, S.-Y. Effects of feeding adults of Hermetia illucens (Diptera: Stratiomyidae) on longevity, oviposition, and egg hatchability: Insights into optimizing egg production. Journal of Insect Science 19, 1–7.
- Birrell, N. Sensory morphology, investment and plasticity in the black soldier fly (Hermetia illucens). Thesis, ResearchSpace@Auckland.
- Boafo, H.A., Gbemavo, D.S.J.C., Timpong-Jones, E.C., Eziah, V., Billah, M., Chia, S.Y., Aidoo, O.F., Clottey, V.A. & Kenis, M. (2023) Substrates most preferred for black soldier fly Hermetia illucens (L.) oviposition are not the most suitable for their larval development; Brill.
- Boller, E. Behavioral aspects of mass-rearing of insects. Entomophaga 17, 9–25. [CrossRef]
- Briceño, R.D., Ramos, D. & Eberhard, W.G. (1999) Aggressive behavior in Medflies (Ceratitis capitata) and its modification by mass rearing (Diptera: Tephritidae). In Journal of the Kansas Entomological Society; Kansas (Central States) Entomological Society; Volume 72, pp. 17–27.
- Bruno, D., Manas, F., Bonelli, M., Gold, M., Marzari, M., Roma, D., Valoroso, M.C., Montali, A., Guillaume, J.B., Rebora, M., Bressac, C., Herman, N., Caccia, S., Casartelli, M. & Tettamanti, G. (2025) BugBook: Life cycle, reproduction, and morphofunctional characterisation of the gut, fat body, and haemocytes in the black soldier fly. Journal of Insects as Food and Feed 1–28.
- Butterworth, N.J., White, T.E., Byrne, P.G. & Wallman, J.F. (2021) Love at first flight: Wing interference patterns are species-specific and sexually dimorphic in blow flies (Diptera: Calliphoridae). Journal of Evolutionary Biology 34, 558–570.
- Buzatto, B.A. & Machado, G. (2008) Resource defense polygyny shifts to female defense polygyny over the course of the reproductive season of a Neotropical harvestman. Behavioral Ecology and Sociobiology 63, 85–94.
- Cai, M., Li, L., Zhao, Z., Zhang, K., Li, F., Yu, C., Yuan, R., Zhou, B., Ren, Z., Yu, Z. & Zhang, J. (2022) Morphometric characteristic of black soldier fly (Hermetia illucens) · Wuhan strain and Its egg production improved by selectively inbreeding. In Life; Multidisciplinary Digital Publishing Institute; Volume 12.
- Cammack, J.A. & Tomberlin, J.K. (2023) Environmental conditions influencing growth and development of the black soldier fly (Hermetia illucens (L.)) (Diptera: Stratiomyidae). In Journal of Insects as Food and Feed; Wageningen Academic; Volume 1, pp. 1–9.
- Candolin, U. Sexual selection and sexual conflict. In Encyclopedia of Ecology pp. 310–318. Elsevier.
- Chapman, T., Bangham, J., Vinti, G., Seifried, B., Lung, O., Wolfner, M.F., Smith, H.K. & Partridge, L. (2003) The sex peptide of Drosophila melanogaster: Female post-mating responses analyzed by using RNA interference. Proceedings of the National Academy of Sciences 100, 9923–9928.
- Chapman, T. & Partridge, L. (1996) Sexual conflict as fuel for evolution. In Nature; Nature Publishing Group; Volume 381, pp. 189–190.
- Chia, S.Y., Tanga, C.M., Khamis, F.M., Mohamed, S.A., Salifu, D., Sevgan, S., Fiaboe, K.K.M. & Niassy, S. (2018) Threshold temperatures and thermal requirements of black soldier fly Hermetia illucens: Implications for mass production. PLOS ONE 13, 1–26.
- Chiabotto, C., Grosso, F., Doretto, A. & Meneguz, M. (2024) Observation of mating behavior using marked flies of black soldier fly (Hermetia illucens) under sunlight condition. In Journal of Insects as Food and Feed; Brill; Volume 10, pp. 2017–2029.
- Chimeno, C., Hausmann, A., Schmidt, S., Raupach, M.J., Doczkal, D., Baranov, V., Hübner, J., Höcherl, A., Albrecht, R., Jaschhof, M., Haszprunar, G. & Hebert, P.D.N. (2022) Peering into the darkness: DNA barcoding reveals surprisingly high diversity of unknown species of diptera (insecta) in germany. In Insects; Multidisciplinary Digital Publishing Institute; Volume 13.
- Coudron, C.L., Adamaki-Sotiraki, C., Yakti, W., Pascual, J.J., Wiklicky, V., Sandrock, C., Van Peer, M., Athanassiou, C., Peguero, D.A., Rumbos, C., Naser El Deen, S., Veldkamp, T., Deruytter, D. & Cambra-López, M. (2025) Bugbook: Basic information and good practices on how to maintain stock populations for Tenebrio molitor and Hermetia illucens for research; Brill.
- Coudron, C.L., Berrens, S., Van Peer, M., Deruytter, D., Claeys, J. & Van Miert, S. (2024) Ammonia emissions related to black soldier fly larvae during growth on different diets. In Journal of Insects as Food and Feed; Wageningen Academic; Volume 10, pp. 1469–1483.
- Cui, Z., Si, P., Liu, L., Chen, S., Wang, Y., Li, X., Zhou, J.-J. & Zhou, Q. (2022) Push-pull strategy for integrated control of Bactrocera minax (Diptera, Tephritidae) based on olfaction and vision. Journal of Applied Entomology 146, 1243–1251.
- Davis, R.B., Javoiš, J., Kaasik, A., Õunap, E. & Tammaru, T. (2016) An ordination of life histories using morphological proxies: capital vs. income breeding in insects. Ecology 97, 2112–2124.
- Dearlove, E.; Van Gestel, C.A.M.; Loureiro, S.; Svendsen, C.; Lloyd, M.; Mugo-Kamiri, L.; Petersen, J.M.; Bessette, E.; Edwards, S.; Lim, F.S.; Herren, P.; Hernández Pelegrı́n, L.; Pienaar, R.D.; Huditz, H.; Mostafaie, A. BugBook: Determining multiple stressor interactions in mass-reared insects based on principles of ecotoxicology. Journal of Insects as Food and Feed 1–18.
- Dickerson, A.J., Lemke, N.B., Li, C. & Tomberlin, J.K. (2024) Impact of age on the reproductive output of Hermetia illucens (Diptera: Stratiomyidae). Journal of Economic Entomology toae107.
- Dortmans, B., Diener, S., Verstappen, B. & Zurbrügg, C. (2017) Black soldier fly biowaste processing: A step-by-step guide; Swiss Fedearl Institute of Aquatic Science and Technology (EAWAG): Department of Sanitation, Water and Solid Waste for Development (Sandec): Dübendorf, Switzerland.
- Dufresne, G., Bolduc, C., Warburton, C., Vandenberg, G., Deschamps, M.-H. & Alnahhas, N. (2025) Parentage assignment in black soldier fly (Hermetia illucens) using genotyping-by-sequencing. In Frontiers in Genetics; Frontiers.
- Eberhard, W.G. Copulatory courtship and cryptic female choice in insects. Biological Reviews 1991, 66, 1–31. [Google Scholar] [CrossRef]
- Eberhard, W.G. & Gelhaus, J.K. Genitalic stridulation during copulation in a species of crane fly, Tipula (Bellardina) sp. (Diptera: Tipulidae). Rev. Biol. Trop. 57, 6.
- Eberhard, W.G., Huber, B.A., S., R.L.R., Briceño, R.D., Salas, I. & Rodriguez, V. (1998) One size fits all? Relationships between the size and degree of variation in genitalia and other body parts in twenty species of insects and spiders. Evolution 52, 415–431.
- Emlen, S.T. & Oring, L.W. (1977) Ecology, sexual selection, and the evolution of mating systems. In Science; American Association for the Advancement of Science; Volume 197, pp. 215–223.
- Ewusie, E.A., Kwapong, P.K., Ofosu-Budu, G., Sandrock, C., Akumah, A.M., Nartey, E.K., Tetegaga, C. & Agyakwah, S.K. (2019) The black soldier fly, Hermetia illucens (Diptera: Stratiomyidae): Trapping and culturing of wild colonies in Ghana. Scientific African 5, e00134.
- Fagerström, T. & Wiklund, C. Why do males emerge before females? Protandry as a mating strategy in male and female butterflies. Oecologia 52, 164–166.
- Ferdousi, L. & Sultana, N. (2021) Impact of daylight duration on pupation and adult emergence of black soldier fly. Hermetia illucens 18–19.
- Ferdousi, L., Sultana, N., Bithi, U.H., Lisa, S.A., Hasan, Md.R. & Siddique, Md.A.B. (2022) Nutrient profile of wild black soldier fly (Hermetia illucens) prepupae reared on municipal dustbin’s organic waste substrate. Proceedings of the National Academy of Sciences, India Section B: Biological Sciences 92, 351–357.
- Fowler, K. & Partridge, L. (1989) A cost of mating in female fruit flies. In Nature; Nature Publishing Group; Volume 338, pp. 760–761.
- Fowles, T.M. & Nansen, C. (2019) Artificial selection of insects to bioconvert pre-consumer organic wastes. A review. Agronomy for Sustainable Development 39, 31.
- Free, D. & Wolfensohn, S. (2023) Assessing the welfare of captive group-housed cockroaches, Gromphadorhina oblongonota. In Animals; Multidisciplinary Digital Publishing Institute; Volume 13.
- Freitas Spindola, A. Morphological and physiological reproductive aspects of black soldier fly: applications for optimal industrial scale production. Thesis, Texas A&M University, College Station, Texas, USA.
- García-Castillo, P., Peralta, C., Hernández-Pelegrín, L. & Herrero, S. (2025) Shortening the Black Soldier Fly Life Cycle by Temperature-Driven Genetic Selection to Enhance Alternative Protein Production. eFood 6.
- Generalovic, T.N. Population genomics and domestication of the Black Soldier Fly (Hermetia illucens L.). PhD Thesis, University of Cambridge, Cambridge, United Kingdom.
- Generalovic, T.N., McCarthy, S.A., Warren, I.A., Wood, J.M.D., Torrance, J., Sims, Y., Quail, M., Howe, K., Pipan, M., Durbin, R. & Jiggins, C.D. (2021) A high-quality, chromosome-level genome assembly of the black soldier fly (Hermetia illucens L.). G3 Genes|Genomes|Genetics 11.
- Generalovic, T.N., Sandrock, C., Roberts, B.J., Meier, J.I., Hauser, M., Warren, I.A., Pipan, M., Durbin, R. & Jiggins, C.D. (2023) Cryptic diversity and signatures of domestication in the black soldier fly (Hermetia illucens). bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2023.10.21.563413v2 (accessed on 11 August 2025).
- Generalovic, T.N., Zhou, W., Zhao, L.C., Leonard, S., Warren, I.A., Pipan, M. & Jiggins, C.D. (2025) Repeatable phenotypic but not genetic response to selection on body size in the black soldier fly. bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2025.02.25.640052v1 (accessed on 29 May 2025).
- Giannetti, D., Schifani, E., Reggiani, R., Mazzoni, E., Reguzzi, M.C., Castracani, C., Spotti, F.A., Giardina, B., Mori, A. & Grasso, D.A. (2022) Do it by yourself: Larval locomotion in the black soldier fly Hermetia illucens, with a novel “self-harvesting” method to separate prepupae. In Insects; Multidisciplinary Digital Publishing Institute; Volume 13.
- Giunti, G., Campolo, O., Laudani, F. & Palmeri, V. (2018) Male courtship behaviour and potential for female mate choice in the black soldier fly Hermetia illucens L. (Diptera: Stratiomyidae). Entomologia Generalis 38, 29–46.
- Gobbi, P., Martínez-Sánchez, A. & Rojo, S. (2013) The effects of larval diet on adult life-history traits of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). European Journal of Entomology 110, 461–468.
- Grosso, F., Lattarulo, A., Meneguz, M. & Padula, C. (2025) Biomimicry in love-cage design: boosting black soldier fly mass production. In Journal of Insects as Food and Feed; Brill: Wageningen; p. 11.
- Hansen, L.S., Laursen, S.F., Bahrndorff, S., Sørensen, J.G., Sahana, G., Kristensen, T.N. & Nielsen, H.M. (2025) The unpaved road towards efficient selective breeding in insects for food and feed—A review. Entomologia Experimentalis et Applicata 173, 498–521.
- Harjoko, D.N., Hua, Q.Q.H., Toh, E.M.C., Goh, C.Y.J. & Puniamoorthy, N. (2023) A window into fly sex: Mating increases female but reduces male longevity in black soldier flies. Animal Behaviour 200, 25–36.
- Hebert, P.D.N., Ratnasingham, S. & de Waard, J.R. (2003) Barcoding animal life: Cytochrome c oxidase subunit 1 divergences among closely related species. Proceedings of the Royal Society B: Biological Sciences 270, S96–S99.
- Herberstein, M.E., Painting, C.J. & Holwell, G.I. (2017) Scramble competition polygyny in terrestrial arthropods. In Advances in the study of behavior (eds M. Naguib, J. Podos, L.W. Simmons, L. Barrett, S.D. Healy & M. Zuk); Elsevier: Cambridge, Massachusetts; pp. 237–295.
- Heussler, C.D., Klammsteiner, T., Stonig, K.T., Insam, H., Schlick-Steiner, B.C. & Steiner, F.M. (2023) Microbial influences on black soldier fly reproduction: A focus on egg surface colonization. Research Square. Available online: Https://www.researchsquare.com/article/rs-3314156/v1 (accessed on 29 May 2025).
- Hoc, B., Noël, G., Carpentier, J., Francis, F. & Megido, R.C. (2019) Optimization of black soldier fly (Hermetia illucens) artificial reproduction. In PLOS ONE; Public Library of Science; Volume 14, pp. 1–13.
- Hoffmann, L., Hull, K.L., Bierman, A., Badenhorst, R., Bester-van der Merwe, A.E. & Rhode, C. (2021) Patterns of genetic diversity and mating systems in a mass-reared black soldier fly colony. In Insects; Multidisciplinary Digital Publishing Institute; Volume 12.
- Höglund, J. & Alatalo, R.V. (1995) Leks; Princeton University Press: United Kingdom.
- Holze, H., Schrader, L. & Buellesbach, J. (2021) Advances in deciphering the genetic basis of insect cuticular hydrocarbon biosynthesis and variation. In Heredity; Nature Publishing Group; Volume 126, pp. 219–234.
- Hutchinson, G.E. Concluding remarks. Cold Spring Harbor Symposia on Quantitative Biology 22, 415–427. Cold Spring Harbor Lab, Yale University, Connecticut, USA.
- Ingleby, F.C. Insect cuticular hydrocarbons as dynamic traits in sexual communication. Insects 6, 732–742. Multidisciplinary Digital Publishing Institute.
- James, A., Seth, A., Marcireau, A., Mukhopadhyay, S., Hu, T. & Atayde, R. (2024) FlyCount: High-speed counting of black soldier flies using neuromorphic sensors. IEEE Sensors Journal 1–1.
- James, M.T. The genus Hermetia in the United States (Diptera: Stratiomyidae). Bulletin of the Brooklyn Entomological Society 30, 165–170.
- Jang, E.B., McInnis, D.O., Kurashima, R. & Carvalho, L.A. (1999) Behavioural switch of female Mediterranean fruit fly, Ceratitis capitata: Mating and oviposition activity in outdoor field cages in Hawaii. In Agricultural and Forest Entomology; John Wiley & Sons, Ltd; Volume 1, pp. 179–184.
- Jensen, K., Thormose, S.F., Noer, N.K., Schou, T.M., Kargo, M., Gligorescu, A., Nørgaard, J.V., Hansen, L.S., Zaalberg, R.M., Nielsen, H.M. & Kristensen, T.N. (2025) Controlled and polygynous mating in the black soldier fly: Advancing breeding programs utilizing quantitative genetic designs; Brill.
- Jiggins, C.D. Genetic diversity and improvement of the black soldier fly. Theatre, . 2024. [Google Scholar] [CrossRef]
- Jones, B.M. & Tomberlin, J.K. (2021) Effects of adult body size on mating success of the black soldier fly, Hermetia illucens (L.) (Diptera: Stratiomyidae). Journal of Insects as Food and Feed 7, 5–20.
- Jones, T.M. & Widemo, F. (2005) Survival and reproduction when food is scarce: implications for a lekking Hawaiian Drosophila. Ecological Entomology 30, 397–405.
- Julita, U., Fitri, L., Putra, R. & Permana, A. (2020) Mating success and reproductive behavior of black soldier fly Hermetia illucens L. (Diptera, Stratiomyidae) in tropics. Journal of Entomology 17, 117–127.
- Julita, U., Supriatna, A., Hambali, A., Fitri, L.L. & Permana, A. (2025) Impact of male and female body size on mating performance and reproductive success in black soldier fly, Hermetia illucens L. (Diptera: stratiomyidae). Journal of the Entomological Research Society 27, 53–64.
- Kaya, C.; Generalovic, T.N.; Ståhls, G.; Hauser, M.; Samayoa, A.C.; Nunes-Silva, C.G.; Roxburgh, H.; Wohlfahrt, J.; Ewusie, E.A.; Kenis, M.; Hanboonsong, Y.; Orozco, J.; Carrejo, N.; Nakamura, S.; Gasco, L.; et al. Global population genetic structure and demographic trajectories of the black soldier fly, Hermetia illucens. BMC Biology 19, 94.
- Klammsteiner, T., Heussler, C.D., Insam, H., Tomberlin, J.K., Schlick-Steiner, B.C. & Steiner, F.M. (2025) Exploring interkingdom communication: The case of black soldier fly mass-rearing. Current Opinion in Insect Science 72, 101427.
- Klammsteiner, T., Stonig, K., Insam, H., Schlick-Steiner, B. & Steiner, F. (2026) Stage-specific microbiota transitions throughout black soldier fly ontogeny. Microbial Ecology 89.
- Klüber, P., Arous, E., Jerschow, J., Fraatz, M., Bakonyi, D., Rühl, M. & Zorn, H. (2024) Fatty acids derived from oviposition systems guide female black soldier flies (Hermetia illucens) toward egg deposition sites. Insect Science 31, 1231–1248.
- Klüber, P., Arous, E., Zorn, H. & Rühl, M. (2023) Protein- and carbohydrate-rich supplements in feeding adult black soldier flies (Hermetia illucens) affect life history traits and egg productivity. In Life; Multidisciplinary Digital Publishing Institute; Volume 13.
- Kohler, R.E. Lords of the fly: Drosophila genetics and the experimental life. Chicago: University of Chicago Press. 1994. [Google Scholar]
- Kortsmit, Y., Sklavounou, M., Dicke, M. & van Loon, J.J.A. (2025) Influence of diet on initial mating behaviour in the black soldier fly (Hermetia illucens). In Journal of Insects as Food and Feed; Wageningen Academic; Volume 1, pp. 1–17.
- Kortsmit, Y., Van Der Bruggen, M., Wertheim, B., Dicke, M., Beukeboom, L.W. & Van Loon, J.J.A. (2023) Behaviour of two fly species reared for livestock feed: Optimising production and insect welfare. Journal of Insects as Food and Feed 9, 149–169.
- Kotiaho, J.S., Simmons, L.W. & Tomkins, J.L. (2001) Towards a resolution of the lek paradox. Nature 410, 684–686.
- Kotzé, Z. & Tomberlin, J.K. Influence of substrate age and interspecific colonization on oviposition behavior of a generalist feeder, black soldier fly (Diptera: Straiomyidae), on carrion. Journal of Medical Entomology 57, 987–993.
- Kubli, E. My favorite molecule. The sex-peptide. BioEssays 14, 779–784. [CrossRef] [PubMed]
- Laksanawimol, P., Singsa, S. & Thancharoen, A. (2023) Behavioral responses of different reproductive statuses and sexes in Hermetia illucens (L.) adults to different attractants [PeerJ]. PeerJ 11.
- Lalander, C., Ermolaev, E., Wiklicky, V. & Vinnerås, B. (2020) Process efficiency and ventilation requirement in black soldier fly larvae composting of substrates with high water content. Science of The Total Environment 729, 138968.
- Lambkin, C.L., Sinclair, B.J., Pape, T., Courtney, G.W., Skevington, J.H., Meier, R., Yeates, D.K., Blagoderov, V. & Wiegmann, B.M. (2013) The phylogenetic relationships among infraorders and superfamilies of Diptera based on morphological evidence. Systematic Entomology 38, 164–179.
- Laturney, M. & Billeter, J.-C. Drosophila melanogaster females restore their attractiveness after mating by removing male anti-aphrodisiac pheromones. In Nature Communications; Nature Publishing Group; Volume 7, pp. 1–11.
- Laturney, M., van Eijk, R. & Billeter, J.-C. (2018) Last male sperm precedence is modulated by female remating rate in Drosophila melanogaster. Evolution Letters 2, 180–189.
- Laudani, F., Campolo, O., Latella, I., Modafferi, A., Palmeri, V. & Giunti, G. (2024) Does Hermetia illucens recognize sibling mates to avoid inbreeding depression? Entomologia Generalis 44, 1225–1232.
- Laursen, S.F., Flint, C.A., Bahrndorff, S., Tomberlin, J.K. & Kristensen, T.N. (2024) Reproductive output and other adult life-history traits of black soldier flies grown on different organic waste and by-products. Waste Management 181, 136–144.
- Lemke, N.B., Dickerson, A.J. & Tomberlin, J.K. (2023) No neonates without adults. BioEssays 45, 2200162.
- Lemke, N.B., Li, C. & De Smet, J. (2025a) Temporal trends: Phase-shifted time-series analysis reveals highly correlated reproductive behaviors in the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2025.08.26.672371v2 (accessed on 23 September 2025).
- Lemke, N.B., Li, C., Dickerson, A.J., Salazar, D.A., Rollinson, L.N., Mendoza, J.E., Miranda, C.D., Crawford, S. & Tomberlin, J.K. (2025b) Heterogeny in cages: Age-structure and attractant availability impacts fertile egg production in the black soldier fly, Hermetia illucens. In Journal of Insects as Food and Feed Online; Brill.
- Lemke, N.B., Rollison, L.N. & Tomberlin, J.K. (2024) Sex-specific perching: Monitoring of artificial plants reveals dynamic female-biased perching behavior in the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). Insects 15, 770.
- Lemke, N.B., Smith, M.B., Smink, J.A., Jordan, H.R., Picard, C.J. & Tomberlin, J.K. (2025c) Wild flies: Mating behavior, adult foraging, habitat use, and gut microbiota of Hermetia illucens. in Costa Rica. Preprints. Available online: Https://www.preprints.org/manuscript/202512.0942 (accessed on 6 February 2026).
- Li, C., Addeo, N.F., Rusch, T.W., Tarone, A.M. & Tomberlin, J.K. (2023) Black soldier fly (Diptera: Stratiomyidae) larval heat generation and management. Insect Science 30, 964–974.
- Liedo, P., Salgado, S., Oropeza, A. & Toledo, J. (2007) Improving mating performance of mass-reared sterile Mediterranean fruit flies (Diptera: Tephritidae) through changes in adult holding conditions: Demography and mating competitiveness. In Florida Entomologist; Florida Entomological Society; Volume 90, pp. 33–40.
- Luck, N., Dejonghe, B., Fruchard, S., Huguenin, S. & Joly, D. (2007) Male and female effects on sperm precedence in the giant sperm species Drosophila bifurca. Genetica 130, 257–265.
- Lüpold, S., Manier, M.K., Puniamoorthy, N., Schoff, C., Starmer, W.T., Luepold, S.H.B., Belote, J.M. & Pitnick, S. (2016) How sexual selection can drive the evolution of costly sperm ornamentation. In Nature; Nature Publishing Group; Volume 533, pp. 535–538.
- Macavei, L.I., Benassi, G., Stoian, V. & Maistrello, L. (2020) Optimization of Hermetia illucens (L.) egg laying under different nutrition and light conditions. PLOS ONE 15, 1–18.
- Mack, P.D., Priest, N.K. & Promislow, D.E.L. (2003) Female age and sperm competition: last-male precedence declines as female age increases. In Proceedings of the Royal Society of London. Series B: Biological Sciences; Royal Society; Volume 270, pp. 159–165.
- Malawey, A., Tarone, A., Tomberlin, J. & Walsh, E. (2020) Interaction of age and temperature on heat shock protein expression, sperm count, and sperm viability of the adult black soldier fly (Diptera: Stratiomyidae). Journal of Insects as Food and Feed 7.
- Malawey, A.S., Mercati, D., Love, C.C. & Tomberlin, J.K. (2019) Adult reproductive tract morphology and spermatogenesis in the black soldier fly (Diptera: Stratiomyidae). Annals of the Entomological Society of America 112, 576–586.
- Manas, F., Labrousse, C. & Bressac, C. (2025a) Plastic responses in sperm expenditure to sperm competition risk in black soldier fly (Hermetia illucens, Diptera) males. Journal of Insect Physiology 161, 104751.
- Manas, F., Piterois, H., Labrousse, C., Beaugeard, L., Uzbekov, R. & Bressac, C. (2024) Gone but not forgotten: dynamics of sperm storage and potential ejaculate digestion in the black soldier fly Hermetia illucens. In Royal Society Open Science; Royal Society; Volume 11, p. 241205.
- Manas, F., Venon, P., Yang, L., Labrousse, C. & Bressac, C. (2025b) Multiple mating is not driven by size and sperm management in black soldier fly (Hermetia illucens). Entomologia Experimentalis et Applicata 173, 815–825.
- Markow, T.A. The secret lives of Drosophila flies. eLife 2015, 4. [Google Scholar]
- Meneguz, M., Miranda, C.D., Cammack, J.A. & Tomberlin, J.K. (2023) Adult behaviour as the next frontier for optimising industrial production of the black soldier fly Hermetia illucens (L.) (Diptera: Stratiomyidae). In Journal of Insects as Food and Feed; Wageningen Academic Publishers; Volume 9, pp. 399–414.
- Menger, D.J., Omusula, P., Holdinga, M., Homan, T., Carreira, A.S., Vandendaele, P., Derycke, J.-L., Mweresa, C.K., Mukabana, W.R., Loon, J.J.A. van & Takken, W. (2015) Field evaluation of a push-pull system to reduce Malaria transmission. PLOS ONE 10, e0123415.
- Merle, M., Gueddes, T., Gueye, M.M., Foughar, M., Jiogue-Lacdo, J., Maquart, P.-O., Marion-Poll, F. & Filée, J. (2026) The hidden sweet tooth of the black soldier fly (Hermetia illucens) Online. Journal of Experimental Biology.
- Meyermans, R., Broeckx, L., Mondelaers, J., Gorssen, W., Frooninckx, L., Janssens, S., Van Miert, S. & Buys, N. (2025) Exploring the potential of crossbreeding to enhance black soldier fly (Hermetia illucens) production; Brill.
- Meza, J.S., Ibañez-Palacios, J., Cardenas-Enriquez, D.P., Luis-Alvares, J.H. & Liedo, P. (2025) Bi-environmental cage for colony management in the mass rearing of Anastrepha ludens (Diptera: Tephritidae). In Insect Science; John Wiley & Sons, Ltd.
- Michiels, N.K. & Dhondt, A.A. Differences in male and female activity patterns in the dragonfly Sympetrum danae (Sulzer) and their relation to mate-finding (Anisoptera: Libellulidae). Odonatologica 18, 349–364.
- Miller, W.E. Reproductive bulk in capital-breeding Lepidoptera. Journal of the Lepidopterists’ Society 59, 228–232.
- MOORE, P. (2014) Reproductive physiology and behaviour. In Evoltuion of Insect Mating Systems (eds D.M. SHUKER & L.W. SIMMONS), pp. 78–91Illustrated. Oxford University Press, United Kingdom.
- Morrow, E.H. & Gage, M.J. (2001) Sperm competition experiments between lines of crickets producing different sperm lengths. Proceedings of the Royal Society B: Biological Sciences 268, 2281–2286.
- Muchina, P., Kinyua, J., Khamis, F., Tanga, C., Satti, M., Gebreyesus, G., Sahana, G. & Cai, Z. (2026) A haplotype reference panel and genotype imputation framework for the black soldier fly (Hermetia illucens). Genomics 118, 111206.
- Munsch-Masset, P., Labrousse, C., Beaugeard, L. & Bressac, C. (2023) The reproductive tract of the black soldier fly (Hermetia illucens) is highly differentiated and suggests adaptations to sexual selection. Entomologia Experimentalis et Applicata n/a.
- Muraro, T., Lalanne, L., Pelozuelo, L. & Calas-List, D. (2024) Mating and oviposition of a breeding strain of black soldier fly Hermetia illucens (Diptera: Stratiomyidae): polygynandry and multiple egg-laying. In Journal of Insects as Food and Feed; Wageningen Academic; Volume 10, pp. 1–13.
- Nakamura, S., Ichiki, R.T., Shimoda, M. & Morioka, S. (2016) Small-scale rearing of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae), in the laboratory: Low-cost and year-round rearing. Applied Entomology and Zoology 51, 161–166.
- Nawoya, S., Ssemakula, F., Akol, R., Geissmann, Q., Karstoft, H., Bjerge, K., Mwikirize, C., Katumba, A. & Gebreyesus, G. (2024) Computer vision and deep learning in insects for food and feed production: A review. Computers and Electronics in Agriculture 216, 108503.
- Nyakeri, E.M., Ogola, H.J.O., Ayieko, M.A. & Amimo, F.A. (2017) Valorisation of organic waste material: growth performance of wild black soldier fly larvae (Hermetia illucens) reared on different organic wastes. Journal of Insects as Food and Feed 3, 193–202.
- Oonincx, D.G.A.B., Volk, N., Diehl, J.J.E., van Loon, J.J.A. & Belušič, G. (2016) Photoreceptor spectral sensitivity of the compound eyes of black soldier fly (Hermetia illucens) informing the design of LED-based illumination to enhance indoor reproduction. Journal of Insect Physiology 95, 133–139.
- Papanastasiou, S.A., Diamantidis, A.D., Nakas, C.T., Carey, J.R. & Papadopoulous, N.R. (2011) Dual reproductive cost of aging in male medflies: Dramatic decrease in mating competitiveness and gradual reduction in mating performance. In Journal of Insect Physiology; Pergamon; Volume 57, pp. 1368–1374.
- Park, K.-H., Han, M.-H., Lee, S., Kim, E.-S., Song, M.-H., Kim, W.-T., Choi, J.-Y. & Kim, H.G. (2017) Oviposition activity of black soldier fly (Hermetia illucens) under artificial illumination. International Journal of Industrial Entomology 35, 100–105.
- Parker, G.A. Sperm competition and its evolutionary consequences in the insects. Biological Reviews 45, 525–567. [CrossRef]
- Parker, G.A. Evolution of competitive mate searching. In Annual Review of Entomology; Annual Reviews; Volume 23, pp. 173–196.
- Pélisson, P.-F., Bel-Venner, M.-C., Rey, B., Burgevin, L., Martineau, F., Fourel, F., Lecuyer, C., Menu, F. & Venner, S. (2012) Contrasted breeding strategies in four sympatric sibling insect species: when a proovigenic and capital breeder copes with a stochastic environment. Functional Ecology 26, 198–206.
- Peñalver, E., Matamales-Andreu, R., Nel, A. & Pérez-de la Fuente, R. (2022) Early adaptations of true flies (Diptera) to moist and aquatic continental environments. Papers in Palaeontology 8, e1472.
- Permana, A.D., Fitri, L.L. & Julita, U. (2020) Influence of mates virginity on black soldier fly, Hermetia illucens L. (Diptera: Stratiomyidae) mating performance. Jurnal Biodjati 5, 174–181.
- Pezzi, M., Scapoli, C., Bharti, M., Faucheux, M.J., Chicca, M., Leis, M., Marchetti, M.G., Mamolini, E., Salvia, R., Falabella, P. & Bonacci, T. (2021) Fine structure of maxillary palps in adults of Hermetia illucens (Diptera: Stratiomyidae). Journal of Medical Entomology 58, 658–665.
- Piersanti, S., Rebora, M., Marri, G.C. & Salerno, G. (2024) Antennal olfactory responses in the black soldier fly Hermetia illucens. Journal of Insect Physiology 159, 104722.
- Pitnick, S. & García–González, F. (2002) Harm to females increases with male body size in Drosophila melanogaster. In Proceedings of the Royal Society of London. Series B: Biological Sciences; Royal Society; Volume 269, pp. 1821–1828.
- Pitnick, S., Markow, T.A. & Spicer, G.S. (1995) Delayed male maturity is a cost of producing large sperm in Drosophila. In Proceedings of the National Academy of Sciences; Proceedings of the National Academy of Sciences; Volume 92, pp. 10614–10618.
- Plesnar-Bielak, A. & Łukasiewicz, A. (2021) Sexual conflict in a changing environment. Biological Reviews 96, 1854–1867.
- Pomonis, J.G. Cuticular hydrocarbons of the screwworm, Cochliomyia hominivorox (Diptera: Calliphoridae). Journal of Chemical Ecology 1989, 15, 2301–2317. [Google Scholar] [CrossRef] [PubMed]
- Purkayastha, D. & Sarkar, S. (2023) Performance evaluation of black soldier fly larvae fed on human faeces, food waste and their mixture. Journal of Environmental Management 326, 116727.
- Rathore, A., Isvaran, K. & Guttal, V. (2023) Lekking as collective behaviour. In Philosophical Transactions of the Royal Society B: Biological Sciences; Royal Society; Volume 378, p. 20220066.
- RATTE, H.T. (1985) Temperature and insect development. In Environmental Physiology and Biochemistry of Insects (ed K.H. HOFFMANN), pp. 33–66. Springer, Berlin, Heidelberg.
- Rebora, M., Piersanti, S., Romani, A., Kovalev, A., Gorb, S. & Salerno, G. (2024) Sexual dimorphism in the structural colours of the wings of the black soldier fly (BSF) Hermetia illucens (Diptera: Stratiomyidae). In Scientific Reports; Nature Publishing Group; Volume 14, p. 19655.
- Rhode, C., Badenhorst, R., Hull, K.L., Greenwood, M.P., Merwe, A.E.B. der, Andere, A.A., Picard, C.J. & Richards, C. (2020) Genetic and phenotypic consequences of early domestication in black soldier flies (Hermetia illucens). Animal Genetics 51, 752–762.
- Rollinson, L.N., Lemke, N.B., Crawford, S., Woolley, J.B. & Tomberlin, J.K. (2025) Size-stabilized, hypoallometric, genitalia determined for male black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2025.08.07.666420v1 (accessed on 12 August 2025).
- Romano, N., Fischer, H. & Egnew, N. (2020) Color and sugar preferences of adult black soldier fly (Hermetia illucens) (Diptera: Stratiomyidae) for feeding and oviposition. Journal of Environmental Biology 41, 1132–1137.
- Sable, A. & Chavan, K. (2024) Natural occurrence and biology of black soldier fly, Hermetia illucens L. (Diptera: stratiomyidae) under local conditions of Maharashtra. UTTAR PRADESH JOURNAL OF ZOOLOGY 45, 10–25.
- Salari, S.P. & De Goede, M.L. (2024) Study into Hermetia illucens behaviour in large artificial mating chambers and possible optimizations. Theatre, .
- Sandrock, C., Kaya, C., Kapun, M., Wohlfahrt, J. & Blanckenhorn, W.U. (2021) Deciphering the evolutionary history of the black soldier fly, Hermetia illucens, on a global scale. Book of Abstracts of the 72nd Annual Meeting of the European Federation of Animal Sciences, Davos, Switzerland, 30 August - 3 September 2021; Wageningen Academic Publishers, The Netherlands; p. 295.
- Scharf, I. & Martin, O.Y. Same-sex sexual behavior in insects and arachnids: prevalence, causes, and consequences. Behavioral Ecology and Sociobiology 67, 1719–1730.
- Schnakenberg, S.L., Siegal, M.L. & Bloch Qazi, M.C. Oh, the places they’ll go: Female sperm storage and sperm precedence in Drosophila melanogaster. In Spermatogenesis; Taylor & Francis; Volume 2, pp. 224–235.
- Shelly, T.E. Sexual selection on leks: A fruit fly primer. Journal of Insect Science 2018, 18. [Google Scholar] [CrossRef]
- Shelly, T.E., Whittier, T.S. & Kaneshiro, K.Y. Behavioral responses of Mediterranean fruit flies (Diptera: Tephritidae) to trimedlure baits: Can leks be created artificially? Annals of the Entomological Society of America 86, 341–351.
- Sheppard, C., Tomberlin, J., Joyce, J., Kiser, B. & Sumner, S. (2002) Rearing methods for the black soldier fly (Diptera: Stratiomyidae). Journal of Medical Entomology 39, 695–698.
- Shrestha, K., Junes, P., van den Boer, E., Christianen, I., Jacobse, R. & Schmitt, E. (2025) Correlated response to selection for increased body weight on fecundity in Hermetia illucens. Entomologia Experimentalis et Applicata 173, 532–544.
- Shuker, D.M. & Simmons, L.W.<The evolution of insect mating systems; Oxford University Press, University of Oxford, 2014.
- Sibonje, J.N. Determination of the gross nutrient composition of larvae and substrate during black soldier fly treatment of faecal and kitchen waste. Thesis, Meru University of Science & Technology. 2024. [Google Scholar]
- Silvaraju, S., Loh, R.K., Kittelmann, S. & Puniamoorthy, N. (2026) Genetic differentiation in domesticated black soldier fly (Hermetia illucens) populations. BMC Biology 24, 7.
- Silvaraju, S., Zhang, Q., Kittelmann, S. & Puniamoorthy, N. (2024) Genetics, age, and diet influence gut bacterial communities and performance of black soldier fly larvae (Hermetia illucens). Animal Microbiome 6, 56.
- Smetana, S., Coudron, C., Deruytter, D., Francis, A., Pascual, J.J., Klammsteiner, T., Lemke, N., Sandrock, C. & Zanoli, R. (2025) BugBook: Data analysis methods in studies of insects for food and feed; Brill.
- Spranghers, T., Noyez, A., Schildermans, K. & De Clercq, P. (2017) Cold hardiness of the black soldier fly (Diptera: Stratiomyidae). Journal of Economic Entomology 110, 1501–1507.
- Sripontan, Y., Juntavimon, T., Songin, S. & Chiu, C.-I. (2017) Egg-trapping of black soldier fly, Hermetia illucens (L.) (Diptera: Stratiomyidae) with various wastes and the effects of environmental factors on egg-laying. Khon Kaen Agricultural Journal 45, 179–184.
- Ståhls, G.; Meier, R.; Sandrock, C.; Hauser, M.; Šašić Zorić, L.; Laiho, E.; Aracil, A.; Doderović, J.; Badenhorst, R.; Unadirekkul, P.; Mohd Adom, N.A.B.; Wein, L.; Richards, C.; Tomberlin, J.K.; Rojo, S. puzzling mitochondrial phylogeography of the black soldier fly (Hermetia illucens), the commercially most important insect protein species. BMC Evolutionary Biology 2020, 20, 60. [Google Scholar] [CrossRef] [PubMed]
- Stephens, P.A., Boyd, I.L., McNamara, J.M. & Houston, A.I. (2009) Capital breeding and income breeding: their meaning, measurement, and worth. Ecology 90, 2057–2067.
- Sui, Z., Wu, Q., Geng, J., Xiao, J. & Huang, D. (2024) CRISPR/Cas9-mediated efficient white genome editing in the black soldier fly Hermetia illucens. Molecular Genetics and Genomics 299, 5.
- Surendra, K.C., Tomberlin, J.K., van Huis, A., Cammack, J.A., Heckmann, L.-H.L. & Khanal, S.K. (2020) Rethinking organic wastes bioconversion: Evaluating the potential of the black soldier fly (Hermetia illucens (L.)) (Diptera: Stratiomyidae) (BSF). Waste Management 117, 58–80.
- Tammaru, T. & Haukioja, E. (1996) Capital Breeders and Income Breeders among Lepidoptera: Consequences to Population Dynamics. In Oikos; Nordic Society Oikos, Wiley; Volume 77, pp. 561–564.
- Teder, T., Kaasik, A., Taits, K. & Tammaru, T. (2021) Why do males emerge before females? Sexual size dimorphism drives sexual bimaturism in insects. Biological Reviews 96, 2461–2475.
- Tekaat, L. Maternal oviposition instincts vs. their influence on developmental outcomes: life-history analyses of the black soldier fly. M.Sc., Universität Innsbruck, Innsbruck, Austria. 2024. [Google Scholar]
- Thinn, A.A. & Kainoh, Y. (2022) Effect of diet on the longevity and oviposition performance of black soldier flies, Hermetia illucens (Diptera: Stratiomyidae). Japan Agricultural Research Quarterly: JARQ 56 211–217.
- Thomas, N.K., Karpati, Z., Schmitt, T. & Riabinina, O. (2024a) A chemically defined oviposition attractant and repellent of black soldier flies (Hermetia illucens). bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2024.06.04.597456v1 (accessed on 29 May 2025).
- Thomas, N.K., Karpati, Z., Schmitt, T. & Riabinina, O. (2024b) A chemically defined oviposition attractant and repellent of Black Soldier Flies (Hermetia illucens). bioRxiv. Available online: Https://www.biorxiv.org/content/10.1101/2024.06.04.597456v1 (accessed on 15 July 2024).
- Thornhill, R.; Alcock, J. The evolution of insect mating systems. Harvard University Press, Cambridge, Massachusetts. 1983. [Google Scholar]
- Tomberlin, J.K. & van Huis, A. (2020) Black soldier fly from pest to ‘crown jewel’ of the insects as feed industry: an historical perspective. Journal of Insects as Food and Feed 6, 1–4.
- Tomberlin, J.K., Klammsteiner, T., Lemke, N., Yadav, P. & Sandrock, C. (2025) BugBook: Black soldier fly as a model to assess behaviour of insects mass produced as food and feed; Brill.
- Tomberlin, J.K.; Picard, C.J.; Jordan, H.R.; Preyer, C.; Warburton, C.; Crowley, P.; Zheng, R.; Boulanger, F.X.; Banks, I.; Lefranc, M.; Zorrilla, M.J.; Olson, G.; Aid, G.; Fluker, D.; Bench, B.J.; et al. Government and industry investment plays crucial role in further establishment, evolution, and diversification of insect agriculture: a case example from the United States. Journal of Insects as Food and Feed 8, 109–111. [CrossRef]
- Tomberlin, J.K. & Sheppard, D.C. (2001) Lekking behavior of the black soldier fly (Diptera: Stratiomyidae). Florida Entomologist 84, 729–730.
- Tomberlin, J.K. & Sheppard, D.C. (2002) Factors influencing mating and oviposition of black soldier flies (Diptera: Straiomyidae) in a colony. Journal of Entomological Science 37, 345–352.
- Tomberlin, J.K., Sheppard, D.C. & Joyce, J.A. (2002) Selected life-history traits of black soldier flies (Diptera: Stratiomyidae) reared on three artificial diets. Annals of the Entomological Society of America 95, 379–386.
- Vahed, K. The function of nuptial feeding in insects: a review of empirical studies. Biological Reviews 73, 43–78. Cambridge University Press.
- West-Eberhard, M.J. Phenotypic plasticity and the origins of diversity. Annual Review of Ecology and Systematics 20, 249–278. [CrossRef]
- Wicker-Thomas, C. Pheromonal communication involved in courtship behavior in Diptera. Journal of Insect Physiology 53, 1089–1100. [CrossRef]
- Wigby, S. & Chapman, T. (2005) Sex peptide causes mating costs in female Drosophila melanogaster. Current Biology 15, 316–321.
- Wigglesworth, V.B. & Beament, J.W.L. (1960) The respiratory structures in the eggs of higher Diptera. Journal of Insect Physiology 4, 184–189.
- Wilson, E.O. The Diversity of Life. W. W. Norton & Company, New York, NY, USA.
- Yaseen, Y., Ullah, A., Khan, I., Murad, W., Taj, R., Ahmad, M., Fahad Alrefaei, A., Ali, S. & Hamayun, M. (2025) An investigation and trapping wild coloniesof the black soldier fly, Morphological identification of larval stages undera controlled environment in Pakistan. Polish Journal of Environmental Studies 34, 2461–2472.
- Yeates, D.K. & Wiegmann, B.M The Evolutionary Biology of Flies. Columbia University Press.; 2005.
- Zaalberg, R.M., Nielsen, H.M., Noer, N.K., Schou, T.M., Jensen, K., Thormose, S., Kargo, M. & Slagboom, M. (2024) A bio-economic model for estimating economic values of important production traits in the black soldier fly (Hermetia illucens); Brill.
- Zayed, O., Hewedy, O.A., Abdelmoteleb, A., Ali, M., Youssef, M.S., Roumia, A.F., Seymour, D. & Yuan, Z.-C. (2023) Nitrogen journey in plants: From uptake to metabolism, stress response, and microbe interaction. Biomolecules 13, 1443.
- Zeh, J.A. & Zeh, D.W. (1997) Last-male sperm precedence breaks down when females mate with three males. In Proceedings of the Royal Society of London. Series B: Biological Sciences; Royal Society; Volume 257, pp. 287–292.
- Zeni, V., Benelli, G., Campolo, O., Giunti, G., Palmeri, V., Maggi, F., Rizzo, R., Lo Verde, G., Lucchi, A. & Canale, A. (2021) Toxics or lures? Biological and behavioral effects of plant essential oils on tephritidae fruit flies. In Molecules; Volume 26, Multidisciplinary Digital Publishing Institute.
- Zhan, S.; Fang, G.; Cai, M.; Kou, Z.; Xu, J.; Cao, Y.; Bai, L.; Zhang, Y.; Jiang, Y.; Luo, X.; Xu, J.; Xu, X.; Zheng, L.; Yu, Z.; Yang, H. Genomic landscape and genetic manipulation of the black soldier fly Hermetia illucens, a natural waste recycler. Cell Research 30, 50–60. Nature Publishing Group.
- Zhang, J., Huang, L., He, J., Tomberlin, J.K., Li, J., Lei, C., Sun, M., Liu, Z. & Yu, Z. (2010) An artificial light source influences mating and oviposition of black soldier flies, Hermetia illucens. Journal of Insect Science 10, 1–7.
- Zhang, Q.-H., Ng, K.H., Chin, W.S., Tang, Y.J., Lin, J. & Puniamoorthy, N. (2025a) What you eat affects how you mate: sex-specific plasticity in black soldier fly reproduction driven by larval nutrition. Animal Behaviour 227, 123299.
- Zhang, Q.-H. & Puniamoorthy, N. (2025) Impact of rearing substrates on black soldier fly growth and fertility: A semi-industrial scale study to optimize egg collection. In Insects; Multidisciplinary Digital Publishing Institute; Volume 16.
- Zhang, Y., Henawy, A.R., Rehman, K.U., van Huis, A., Cai, M., Zheng, L., Huang, F., Ding, X., Lei, H. & Zhang, J. (2025b) Artificial light source combined with functional microorganism improves reproductive performance of black soldier fly. Insect Science.
- Zheng, L., Crippen, T.L., Holmes, L., Singh, B., Pimsler, M.L., Benbow, M.E., Tarone, A.M., Dowd, S., Yu, Z., Vanlaerhoven, S.L., Wood, T.K. & Tomberlin, J.K. (2013) Bacteria mediate oviposition by the black soldier fly, Hermetia illucens (L.), (Diptera: Stratiomyidae). In Scientific Reports; Nature Publishing Group; Volume 3, pp. 1–8.
- Zim, J., Chkih, H., Bouharroud, R., Sarehane, M. & Lhomme, P. (2023) Effect of various odour attractants on egg-laying activity of black soldier flies (Hermetia illucens). In Journal of Insects as Food and Feed; Brill: Wageningen; Volume 9, pp. 1461–1472.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



