Submitted:
25 August 2025
Posted:
26 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Method
2.1. Materials
2.2. Micro γ-CD-MOF Preparation
2.3. Synthesis of ROS-Sensitive Crosslinked Cyclodextrin Framework
2.4. Ferulic Acid Incorporation
2.5. Folic Acid Functionalized ROS-Sensitive COF
2.6. Physicochemical Characterization
2.7. In Vitro Antioxidant Capability of COF-FA
2.8. In Vitro Release Study
2.9. Cell Viability Assay
2.10. In Vivo Study
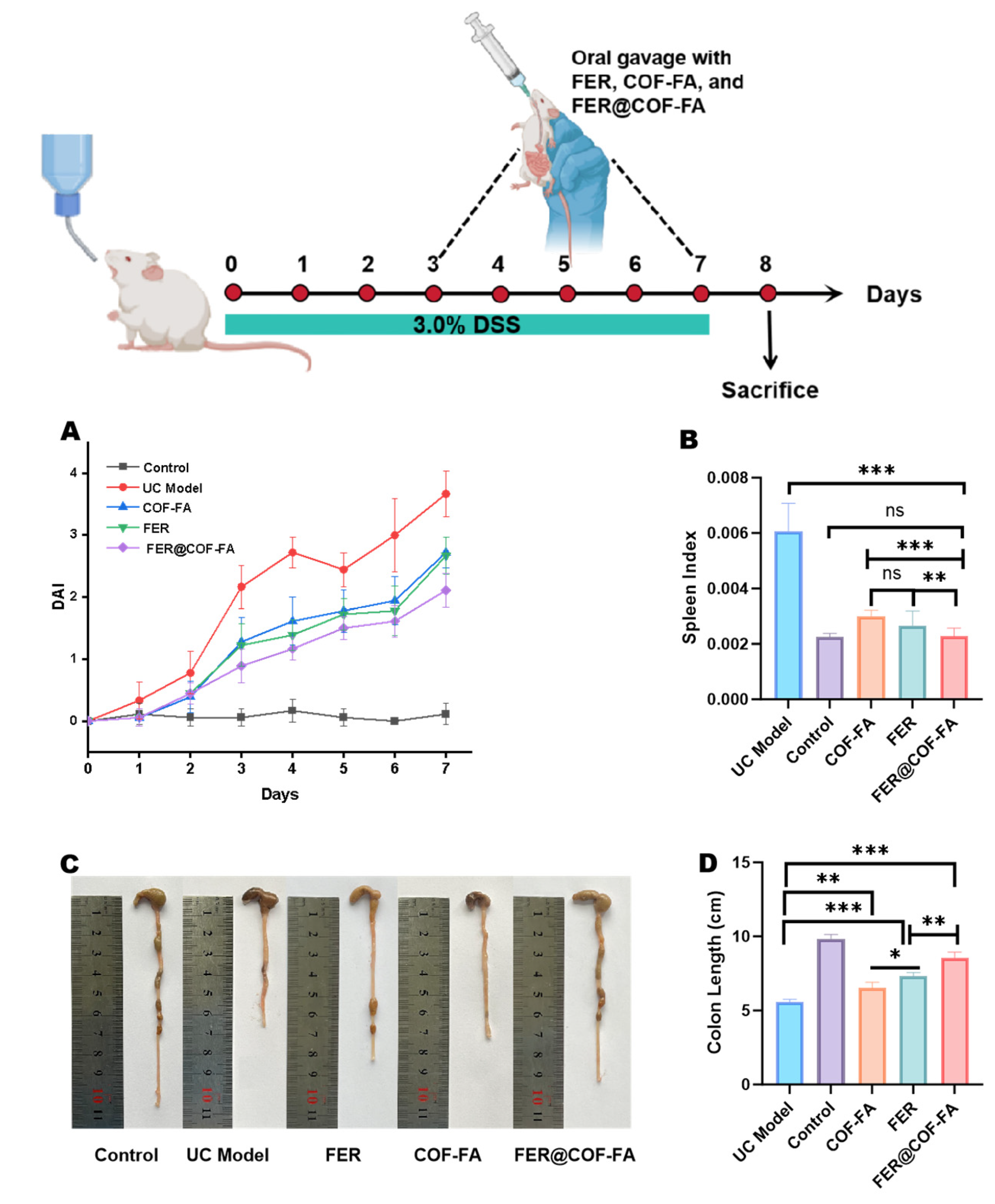
2.10.1. Animal Grouping and Study Design
2.10.2. Disease Activity Index (DAI) Evaluation
2.10.3. Determination of Colon Length and Spleen Index
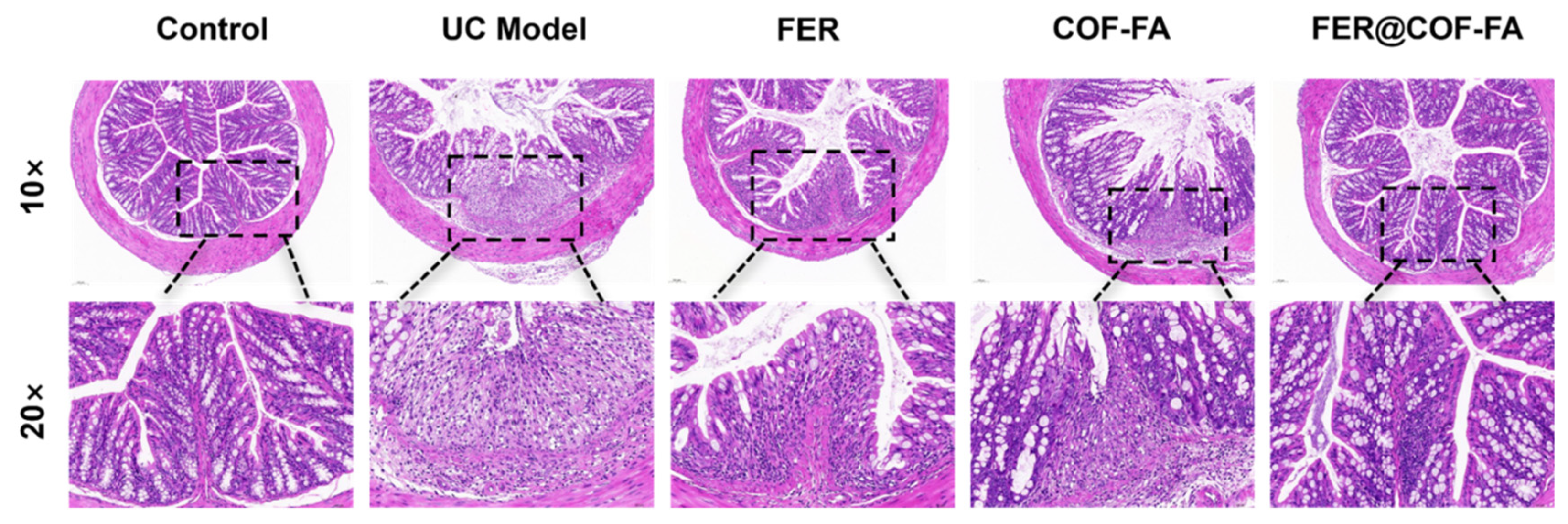
2.10.4. Analysis of Colon Tissue Pathology Sections
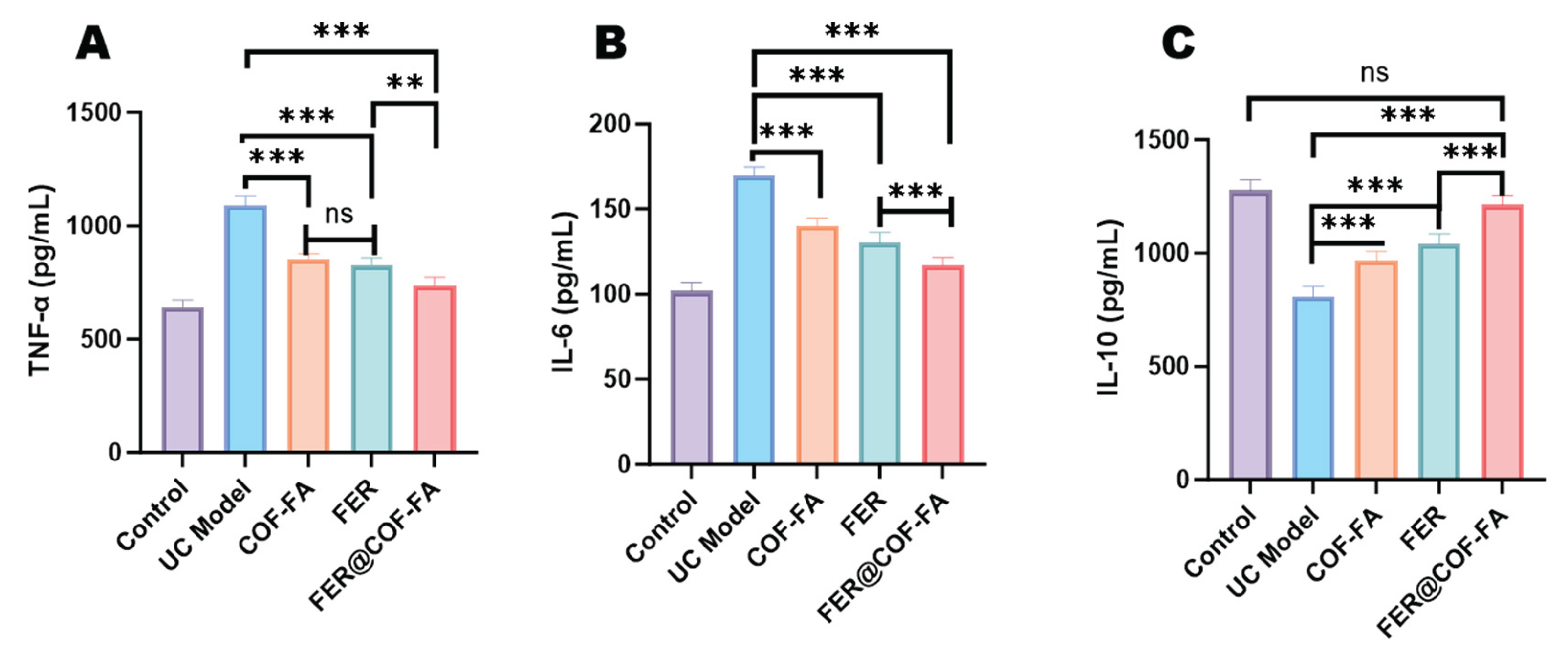
2.10.5. Serum Inflammatory Cytokine Measurement
2.11. Statistical Analysis
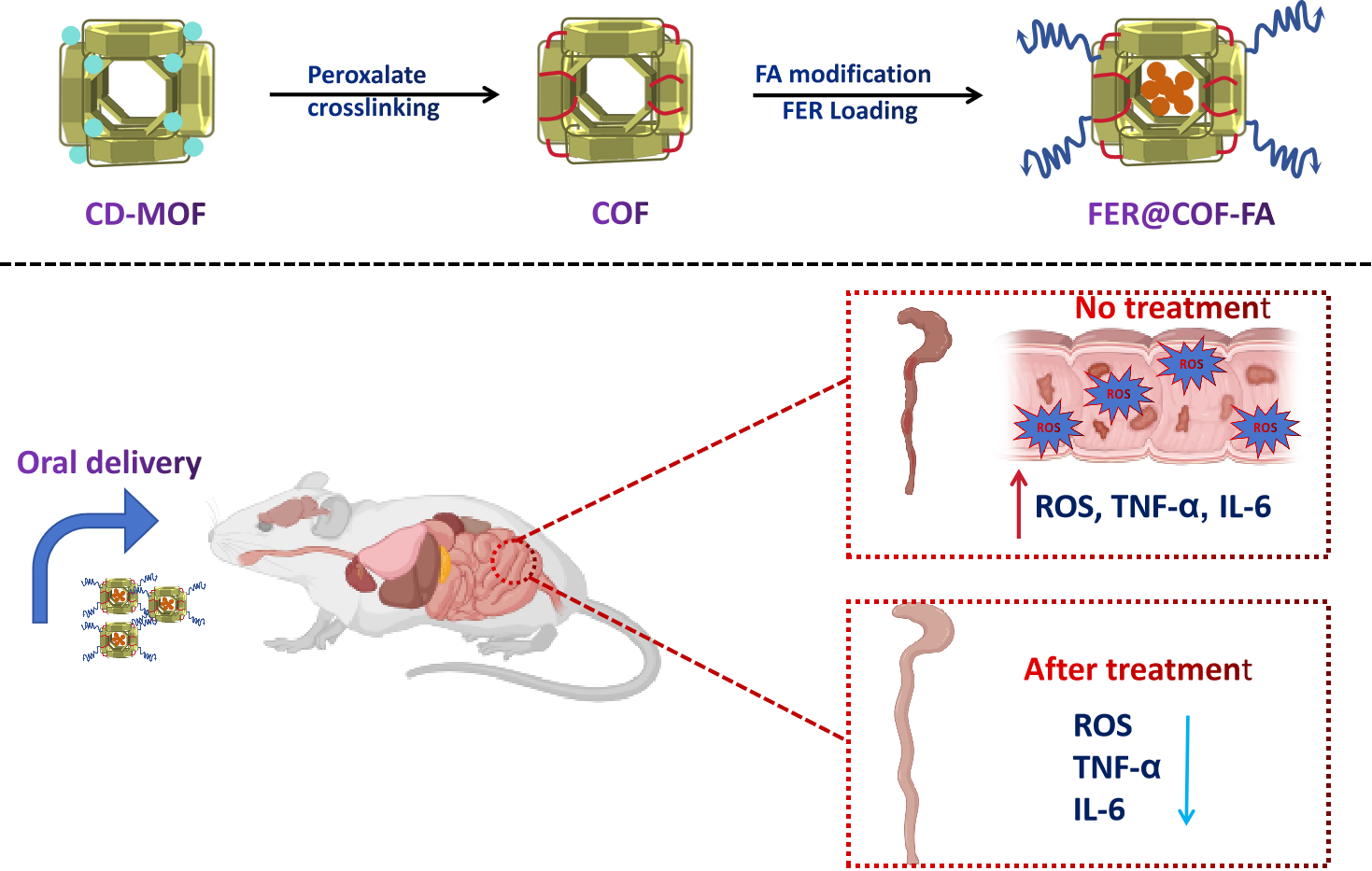
3. Results and Discussion
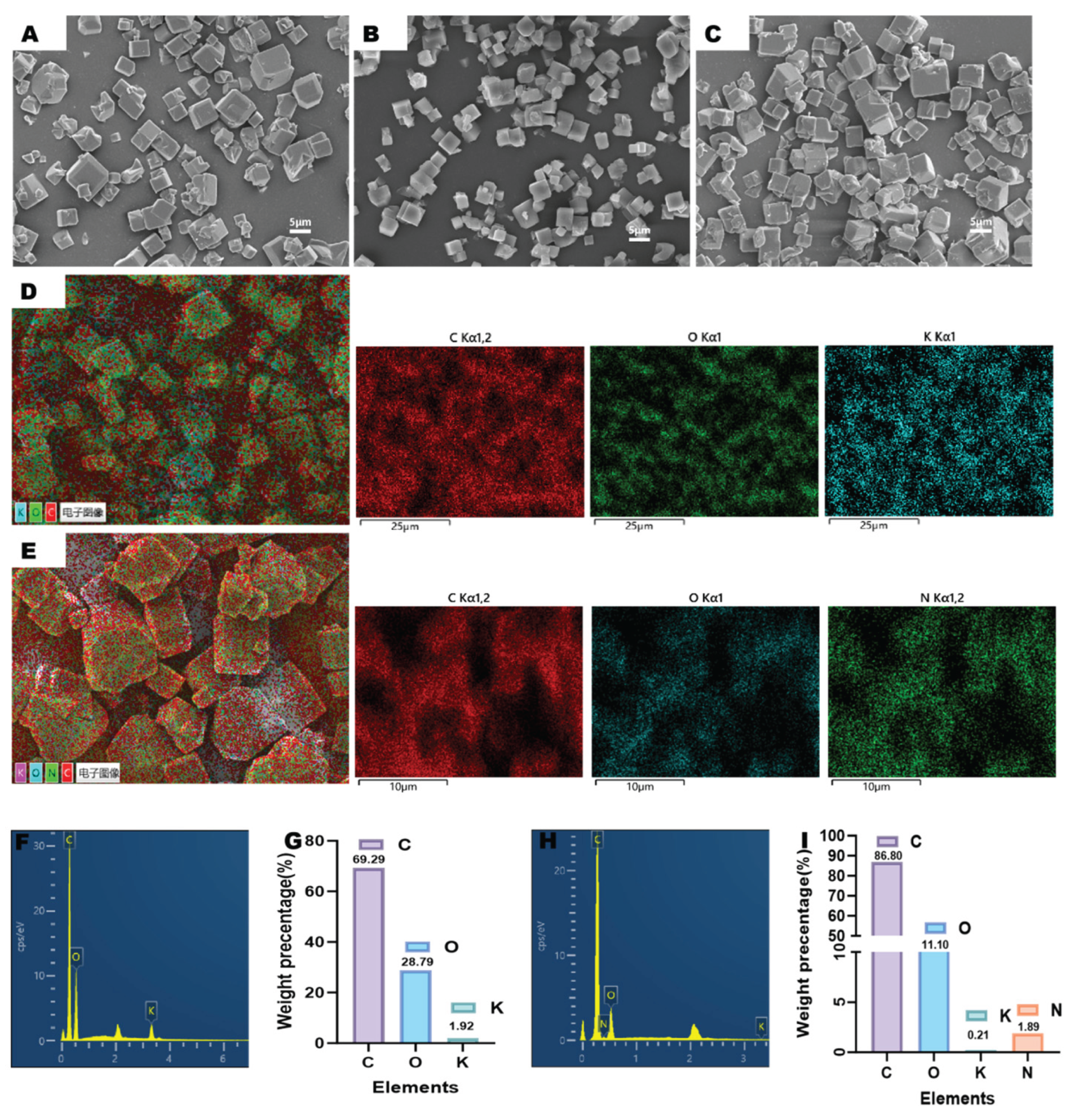
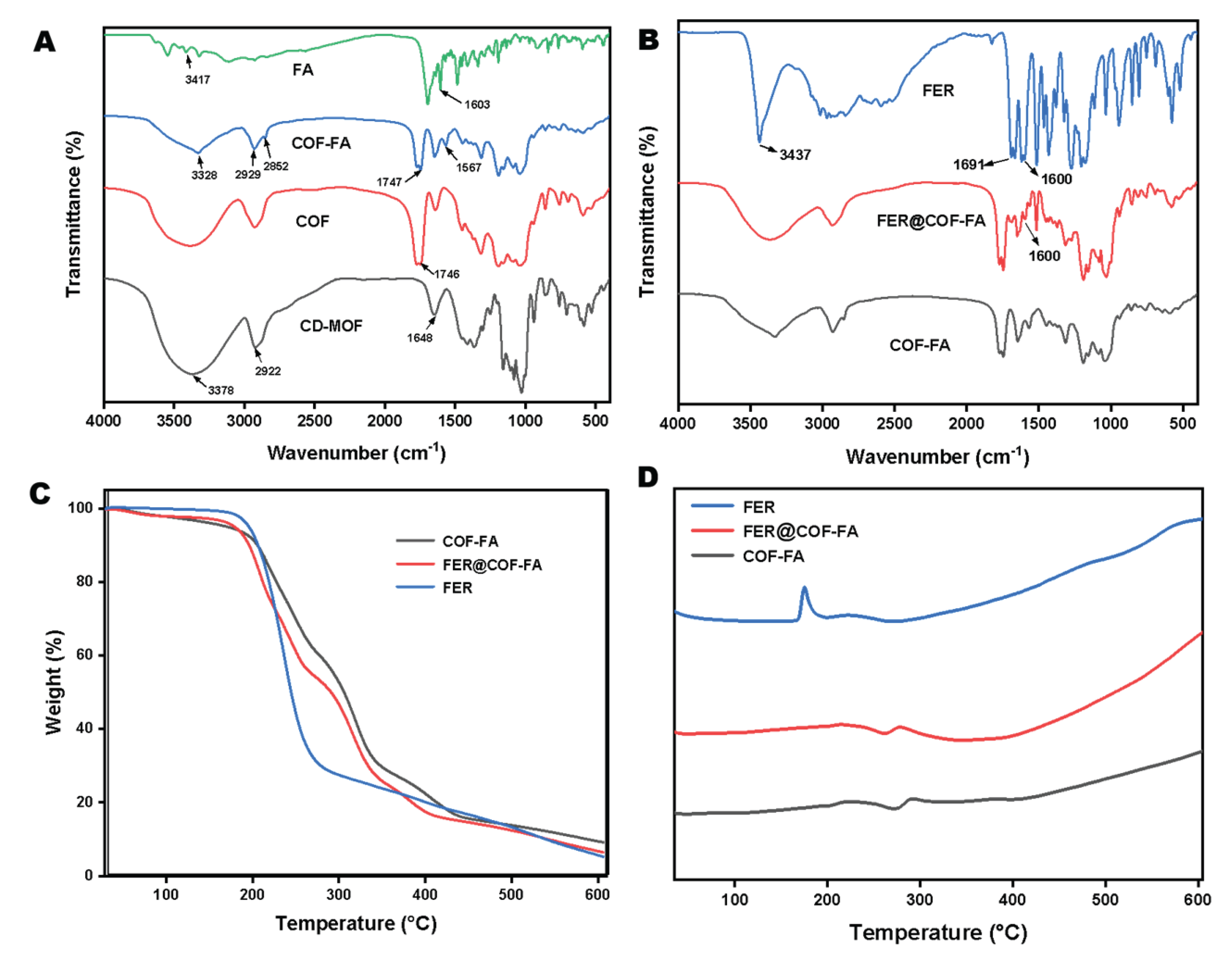
3.1. Characterization
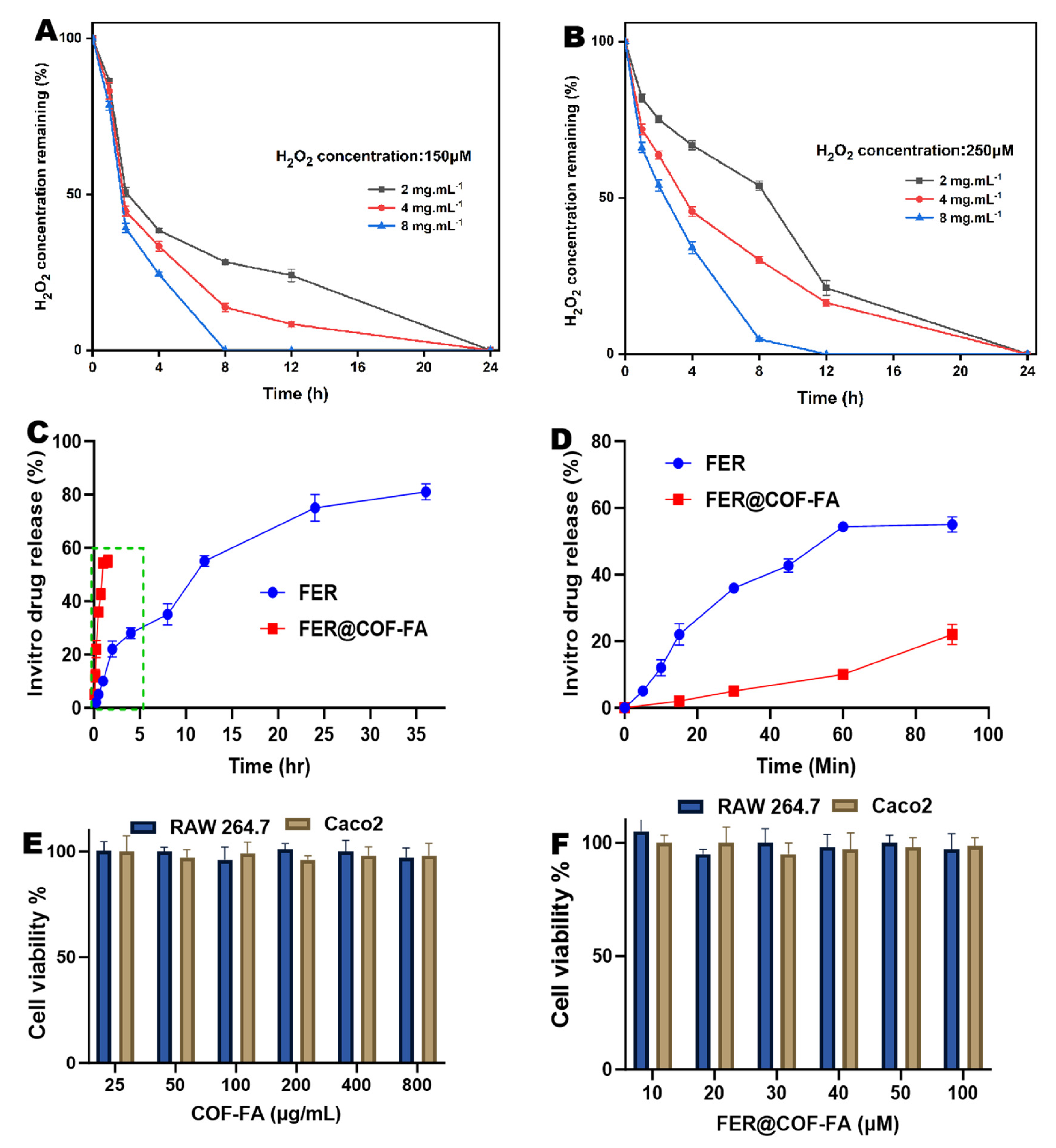
3.2. Evaluation of In Vitro H₂O₂ Scavenging Ability of COF-FA
3.3. In Vitro Release Study
3.4. Cytotoxicity of COF-FA and FER@COF-FA Carrier
3.5. In Vivo Study
3.5.1. Therapeutic Efficacy in DSS-Induced Colitis
3.5.2. Pathological Changes in the Colonic Tissue of Mice
3.5.3. Effect of Inflammatory Factors in Mice Serum
3.6. Limitations and Future Perspectives
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tatiya-aphiradee, N.; Chatuphonprasert, W.; Jarukamjorn, K. Immune Response and Inflammatory Pathway of Ulcerative Colitis. Journal of Basic and Clinical Physiology and Pharmacology 2018, 30, 1–10. [Google Scholar] [CrossRef]
- Saez, A.; Herrero-Fernandez, B.; Gomez-Bris, R.; Sánchez-Martinez, H.; Gonzalez-Granado, J.M. Pathophysiology of Inflammatory Bowel Disease: Innate Immune System. IJMS 2023, 24, 1526. [Google Scholar] [CrossRef]
- Kaur, A.; Goggolidou, P. Ulcerative Colitis: Understanding Its Cellular Pathology Could Provide Insights into Novel Therapies. J Inflamm 2020, 17, 15. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Jain, P. ; Ajazuddin Recent Advances in the Therapeutics and Modes of Action of a Range of Agents Used to Treat Ulcerative Colitis and Related Inflammatory Conditions. Inflammopharmacol 2025. [Google Scholar] [CrossRef]
- Vuyyuru, S.K.; Jairath, V. Unresolved Challenges in Acute Severe Ulcerative Colitis. Indian J Gastroenterol 2024, 43, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Sultan, K.; Becher, N. Ulcerative Colitis Diagnosis and Management: Past, Present, and Future Directions. In Inflammatory Bowel Disease; Rajapakse, R., Ed.; Clinical Gastroenterology; Springer International Publishing: Cham, 2021; ISBN 978-3-030-81779-4. [Google Scholar]
- Chaemsupaphan, T.; Arzivian, A.; Leong, R.W. Comprehensive Care of Ulcerative Colitis: New Treatment Strategies. Expert Review of Gastroenterology & Hepatology 2025, 1–17. [Google Scholar] [CrossRef]
- Wan, Y.; Yang, L.; Jiang, S.; Qian, D.; Duan, J. Excessive Apoptosis in Ulcerative Colitis: Crosstalk Between Apoptosis, ROS, ER Stress, and Intestinal Homeostasis. Inflammatory Bowel Diseases 2022, 28, 639–648. [Google Scholar] [CrossRef]
- Pravda, J. Radical Induction Theory of Ulcerative Colitis. WJG 2005, 11, 2371. [Google Scholar] [CrossRef]
- Du, J.; Zhang, J.; Wang, L.; Wang, X.; Zhao, Y.; Lu, J.; Fan, T.; Niu, M.; Zhang, J.; Cheng, F.; et al. Selective Oxidative Protection Leads to Tissue Topological Changes Orchestrated by Macrophage during Ulcerative Colitis. Nat Commun 2023, 14, 3675. [Google Scholar] [CrossRef]
- Yan, X.; Meng, L.; Zhang, X.; Deng, Z.; Gao, B.; Zhang, Y.; Yang, M.; Ma, Y.; Zhang, Y.; Tu, K.; et al. Reactive Oxygen Species-Responsive Nanocarrier Ameliorates Murine Colitis by Intervening Colonic Innate and Adaptive Immune Responses. Molecular Therapy 2023, 31, 1383–1401. [Google Scholar] [CrossRef]
- Poh, S.; Chelvam, V.; Low, P.S. Comparison of Nanoparticle Penetration into Solid Tumors and Sites of Inflammation: Studies Using Targeted and Nontargeted Liposomes. Nanomedicine (Lond.) 2015, 10, 1439–1449. [Google Scholar] [CrossRef]
- Song, S.; Xia, H.; Guo, M.; Wang, S.; Zhang, S.; Ma, P.; Jin, Y. Role of Macrophage in Nanomedicine-Based Disease Treatment. Drug Delivery 2021, 28, 752–766. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, F.; Yang, C.; Wang, L.; Sung, J.; Garg, P.; Zhang, M.; Merlin, D. Oral Targeted Delivery by Nanoparticles Enhances Efficacy of an Hsp90 Inhibitor by Reducing Systemic Exposure in Murine Models of Colitis and Colitis-Associated Cancer. Journal of Crohn’s and Colitis 2020, 14, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Zhang, L.; Zhang, Y.; Lu, R. Plant-Derived Exosomes as a Drug-Delivery Approach for the Treatment of Inflammatory Bowel Disease and Colitis-Associated Cancer. Pharmaceutics 2022, 14, 822. [Google Scholar] [CrossRef]
- Lin, Z.; Zhao, Z.; Lin, X.; Yang, Z.; Wang, L.; Xi, R.; Long, D. Advances in Oral Treatment of Inflammatory Bowel Disease Using Protein-Based Nanoparticle Drug Delivery Systems. Drug Delivery 2025, 32, 2544689. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, G.; Kim, J.; Kim, W.J. Reactive-Oxygen-Species-Responsive Drug Delivery Systems: Promises and Challenges. Advanced Science 2017, 4, 1600124. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Gambhir, K.; Kumar, S.; Gangenahalli, G.; Verma, Y.K. Interplay of Reactive Oxygen Species (ROS) and Tissue Engineering: A Review on Clinical Aspects of ROS-Responsive Biomaterials. J Mater Sci 2021, 56, 16790–16823. [Google Scholar] [CrossRef]
- Tao, W.; He, Z. ROS-Responsive Drug Delivery Systems for Biomedical Applications. Asian Journal of Pharmaceutical Sciences 2018, 13, 101–112. [Google Scholar] [CrossRef]
- Nidhi; Rashid, M. ; Kaur, V.; Hallan, S.S.; Sharma, S.; Mishra, N. Microparticles as Controlled Drug Delivery Carrier for the Treatment of Ulcerative Colitis: A Brief Review. Saudi Pharmaceutical Journal 2016, 24, 458–472. [Google Scholar] [CrossRef]
- Gvozdeva, Y.; Staynova, R. pH-Dependent Drug Delivery Systems for Ulcerative Colitis Treatment. Pharmaceutics 2025, 17, 226. [Google Scholar] [CrossRef]
- Chen, M.; Lan, H.; Jin, K.; Chen, Y. Responsive Nanosystems for Targeted Therapy of Ulcerative Colitis: Current Practices and Future Perspectives. Drug Delivery 2023, 30, 2219427. [Google Scholar] [CrossRef]
- Xiao, B.; Si, X.; Zhang, M.; Merlin, D. Oral Administration of pH-Sensitive Curcumin-Loaded Microparticles for Ulcerative Colitis Therapy. Colloids and Surfaces B: Biointerfaces 2015, 135, 379–385. [Google Scholar] [CrossRef]
- Ekhtiar, M.; Ghasemi-Dehnoo, M.; Azadegan-Dehkordi, F.; Bagheri, N. Evaluation of Anti-Inflammatory and Antioxidant Effects of Ferulic Acid and Quinic Acid on Acetic Acid-Induced Ulcerative Colitis in Rats. J Biochem & Molecular Tox 2025, 39, e70169. [Google Scholar] [CrossRef]
- Liu, J.; Guan, Y.; Yang, L.; Fang, H.; Sun, H.; Sun, Y.; Yan, G.; Kong, L.; Wang, X. Ferulic Acid as an Anti-Inflammatory Agent: Insights into Molecular Mechanisms, Pharmacokinetics and Applications. Pharmaceuticals 2025, 18, 912. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhu, H.; Luo, Y. Chitosan-Based Oral Colon-Specific Delivery Systems for Polyphenols: Recent Advances and Emerging Trends. J. Mater. Chem. B 2022, 10, 7328–7348. [Google Scholar] [CrossRef]
- Raj, N.D.; Singh, D. A Critical Appraisal on Ferulic Acid: Biological Profile, Biopharmaceutical Challenges and Nano Formulations. Health Sciences Review 2022, 5, 100063. [Google Scholar] [CrossRef]
- Purushothaman, J.R.; Rizwanullah, Md. Ferulic Acid: A Comprehensive Review. Cureus 2024. [Google Scholar] [CrossRef]
- He, S.; Wu, L.; Sun, H.; Wu, D.; Wang, C.; Ren, X.; Shao, Q.; York, P.; Tong, J.; Zhu, J.; et al. Antioxidant Biodegradable Covalent Cyclodextrin Frameworks as Particulate Carriers for Inhalation Therapy against Acute Lung Injury. ACS Appl. Mater. Interfaces 2022, 14, 38421–38435. [Google Scholar] [CrossRef] [PubMed]
- Yue, Q.; Yu, J.; Zhu, Q.; Xu, D.; Wang, M.; Bai, J.; Wang, N.; Bian, W.; Zhou, B. Polyrotaxanated Covalent Organic Frameworks Based on β-Cyclodextrin towards High-Efficiency Synergistic Inactivation of Bacterial Pathogens. Chemical Engineering Journal 2024, 486, 150345. [Google Scholar] [CrossRef]
- Bello, M.G.; Zhang, J.; Chen, L. Cyclodextrin Metal-Organic Framework Design Principles and Functionalization for Biomedical Application. Carbohydrate Polymers 2025, 364, 123684. [Google Scholar] [CrossRef] [PubMed]
- Bello, M.G.; Huang, S.; Qiao, Z.; Chen, Z.; Chen, L. Luteolin Stabilized in Nanosheet and Cubic γ-Cyclodextrin-Based Metal Organic Framework for Enhanced Bioavailability and Anti-Inflammatory Therapy. Carbohydrate Polymer Technologies and Applications 2025, 10, 100833. [Google Scholar] [CrossRef]
- Zhao, R.; Hu, S.; Yang, Z.; Chen, T.; Chi, X.; Wu, D.; Wang, W.; Liu, D.; Zhu, B.; Hu, J. Targeted Quercetin Delivery Nanoplatform via Folic Acid-Functionalized Metal-Organic Framework for Alleviating Ethanol-Induced Gastric Ulcer. Chemical Engineering Journal 2024, 498, 155700. [Google Scholar] [CrossRef]
- Chen, Y.; Feng, T.; Zhu, X.; Tang, Y.; Xiao, Y.; Zhang, X.; Wang, S.-F.; Wang, D.; Wen, W.; Liang, J.; et al. Ambient Synthesis of Porphyrin-Based Fe-Covalent Organic Frameworks for Efficient Infected Skin Wound Healing. Biomacromolecules 2024, 25, 3671–3684. [Google Scholar] [CrossRef] [PubMed]
- Kishi, M.; Hirai, F.; Takatsu, N.; Hisabe, T.; Takada, Y.; Beppu, T.; Takeuchi, K.; Naganuma, M.; Ohtsuka, K.; Watanabe, K.; et al. A Review on the Current Status and Definitions of Activity Indices in Inflammatory Bowel Disease: How to Use Indices for Precise Evaluation. J Gastroenterol 2022, 57, 246–266. [Google Scholar] [CrossRef]
- Caron, B.; Jairath, V.; D’Amico, F.; Al Awadhi, S.; Dignass, A.; Hart, A.L.; Kobayashi, T.; Kotze, P.G.; Magro, F.; Siegmund, B.; et al. International Consensus on Definition of Mild-to-Moderate Ulcerative Colitis Disease Activity in Adult Patients. Medicina 2023, 59, 183. [Google Scholar] [CrossRef]
- Bello, M.G.; Yang, Y.; Wang, C.; Wu, L.; Zhou, P.; Ding, H.; Ge, X.; Guo, T.; Wei, L.; Zhang, J. Facile Synthesis and Size Control of 2D Cyclodextrin-Based Metal–Organic Frameworks Nanosheet for Topical Drug Delivery. Part & Part Syst Charact 2020, 37, 2000147. [Google Scholar] [CrossRef]
- Ding, H.; Wu, L.; Guo, T.; Zhang, Z.; Garba, B.M.; Gao, G.; He, S.; Zhang, W.; Chen, Y.; Lin, Y.; et al. CD-MOFs Crystal Transformation from Dense to Highly Porous Form for Efficient Drug Loading. Crystal Growth & Design 2019, 19, 3888–3894. [Google Scholar] [CrossRef]
- Xiong, T.; Xu, H.; Nie, Q.; Jia, B.; Bao, H.; Zhang, H.; Li, J.; Cao, Z.; Wang, S.; Wu, L.; et al. Reactive Oxygen Species Triggered Cleavage of Thioketal-Containing Supramolecular Nanoparticles for Inflammation-Targeted Oral Therapy in Ulcerative Colitis. Adv Funct Materials 2025, 35, 2411979. [Google Scholar] [CrossRef]
- Karamipour, Sh.; Sadjadi, M.S.; Farhadyar, N. Fabrication and Spectroscopic Studies of Folic Acid-Conjugated Fe3O4@Au Core–Shell for Targeted Drug Delivery Application. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 2015, 148, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Wu, D.; Liu, L.; Chen, J.; Xu, Y. Preparation, Characterization, and in Vitro Release of Folic Acid-Conjugated Chitosan Nanoparticles Loaded with Methotrexate for Targeted Delivery. Polym. Bull. 2012, 68, 1707–1720. [Google Scholar] [CrossRef]
- Dummert, S.V.; Saini, H.; Hussain, M.Z.; Yadava, K.; Jayaramulu, K.; Casini, A.; Fischer, R.A. Cyclodextrin Metal–Organic Frameworks and Derivatives: Recent Developments and Applications. Chem. Soc. Rev. 2022, 51, 5175–5213. [Google Scholar] [CrossRef]
- Bezerra, G.S.N.; Pereira, M.A.V.; Ostrosky, E.A.; Barbosa, E.G.; De Moura, M.D.F.V.; Ferrari, M.; Aragão, C.F.S.; Gomes, A.P.B. Compatibility Study between Ferulic Acid and Excipients Used in Cosmetic Formulations by TG/DTG, DSC and FTIR. J Therm Anal Calorim 2017, 127, 1683–1691. [Google Scholar] [CrossRef]
- Lee, D.; Khaja, S.; Velasquez-Castano, J.C.; Dasari, M.; Sun, C.; Petros, J.; Taylor, W.R.; Murthy, N. In Vivo Imaging of Hydrogen Peroxide with Chemiluminescent Nanoparticles. Nature Mater 2007, 6, 765–769. [Google Scholar] [CrossRef]
- Chen, Y.; Teng, W.; Wang, J.; Wang, Y.; Zhang, Y.; Cao, J. The Intestinal Delivery Systems of Ferulic Acid: Absorption, Metabolism, Influencing Factors, and Potential Applications. Food Frontiers 2024, 5, 1126–1144. [Google Scholar] [CrossRef]
- Yu, Y.R.; Rodriguez, J.R. Clinical Presentation of Crohn’s, Ulcerative Colitis, and Indeterminate Colitis: Symptoms, Extraintestinal Manifestations, and Disease Phenotypes. Seminars in Pediatric Surgery 2017, 26, 349–355. [Google Scholar] [CrossRef]
- Okayasu, I.; Hatakeyama, S.; Yamada, M.; Ohkusa, T.; Inagaki, Y.; Nakaya, R. A Novel Method in the Induction of Reliable Experimental Acute and Chronic Ulcerative Colitis in Mice. Gastroenterology 1990, 98, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhang, F.; Yang, C.; Wang, L.; Sung, J.; Garg, P.; Zhang, M.; Merlin, D. Oral Targeted Delivery by Nanoparticles Enhances Efficacy of an Hsp90 Inhibitor by Reducing Systemic Exposure in Murine Models of Colitis and Colitis-Associated Cancer. Journal of Crohn’s and Colitis 2020, 14, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Poh, S.; Putt, K.S.; Low, P.S. Folate-Targeted Dendrimers Selectively Accumulate at Sites of Inflammation in Mouse Models of Ulcerative Colitis and Atherosclerosis. Biomacromolecules 2017, 18, 3082–3088. [Google Scholar] [CrossRef] [PubMed]
- Poh, S.; Chelvam, V.; Ayala-López, W.; Putt, K.S.; Low, P.S. Selective Liposome Targeting of Folate Receptor Positive Immune Cells in Inflammatory Diseases. Nanomedicine: Nanotechnology, Biology and Medicine 2018, 14, 1033–1043. [Google Scholar] [CrossRef]
- Yang, S.; Li, Y.; Zheng, X.; Zheng, X.; Lin, Y.; Guo, S.; Liu, C. Effects of Folate-Chicory Acid Liposome on Macrophage Polarization and TLR4/NF-κB Signaling Pathway in Ulcerative Colitis Mouse. Phytomedicine 2024, 128, 155415. [Google Scholar] [CrossRef]
- Kelderhouse, L.E.; Mahalingam, S.; Low, P.S. Predicting Response to Therapy for Autoimmune and Inflammatory Diseases Using a Folate Receptor-Targeted Near-Infrared Fluorescent Imaging Agent. Mol Imaging Biol 2016, 18, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Shajib, Md.S.; Manocha, M.M.; Khan, W.I. Investigating Intestinal Inflammation in DSS-Induced Model of IBD. JoVE 2012, 3678. [Google Scholar] [CrossRef]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran Sulfate Sodium (DSS)-Induced Colitis in Mice. CP in Immunology 2014, 104. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



