Submitted:
17 August 2025
Posted:
27 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
3. Results
3.1. Randomized trial included in the analysis
3.2. Reconstruction of individual patient data of OS from Kaplan Meier curves by application of the IPDfrom KM method.
3.3. Estimation of between-trial heterogeneity from individual patient data of the 5 trials reconstructed from Kaplan-Meier curves: description of the previous method
| First author and reference | Experimental group | Control group | Maintenance therapy | Standard treatment | |
| Ai et al. [4] | 48/125 | 22/60 | Xxxx | Xxxx | |
| Byers et al. [5] §§ | 50/61 | 41/61 | xxxx | xxxx | |
| Owonikoko et al. [6] | 52/64 | 54/64 | veliparib+CE | Xxxx | |
| Pietanza et al. [7] | 46/55 | 39/49 | veliparib | xxxx | |
| Woll et al. [8] §§§ | 64§§§/73 olaparib TDS |
59§§§/73 olaparib BD |
60§/74 placebo |
olaparib | placebo |
| . | ||
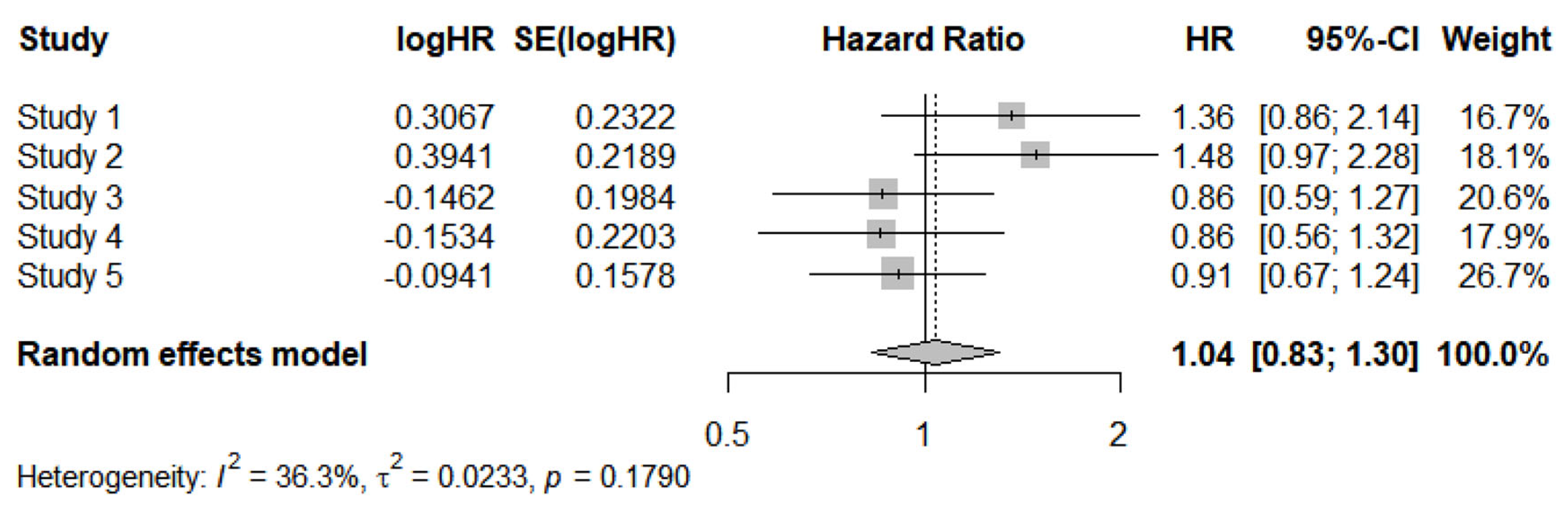
| Original RCT | Adjusted values of HR reported in the original trial§ | HR estimated from “reconstructed patients”§. |
| Ai et al. [4] | 1.03 (95%CI, 0.62 to 1.73), p=0.90 | 1.359 (95%CI, 0.8623 to 2.143), p=0.186 |
| Byers et al. [5] | 1.460 (80% CI, 1.104 to 1.931†), p=0.083 | 1.483 (95%CI, 0.9657 to 2.278†), p=0.072) |
| Owonikoko et al. [6] | 0.83 (80% CI, 0.64 to 1.07†), p=0.34 | 0.864 (95%CI, 0.5857 to 1.275†), p=0.461 |
| Pietanza et al. [7] | NR | 0.8578 (95%CI, 0.557 to 1.321), p=0.487 |
| Woll et al. [8] |
-Split HR:§§ 1) 0.85 (90%CI, 0.63, 1.15; p=0.376) 2) 1.03 (90%CI, 0.77, 1.39; p=0.85) -Pooled HR: NR |
- Split HR:§§ 1) 0.8587 (95%CI, 0.603 1.222), p=0.398 2) 1.036 (95%CI, 0.7228 to 1.484), p=0.849 -Pooled HR: 0.9102 (95%CI, 0.668 to 1.2399), p=0.551 |
| Overall effect | 1.03, 95%CI, 0.92 to 1.15, test for overall effect: Z=0.53, P=0.60. | 1.04, 95%CI: 0.83 to 1.30, P=0.74 |
| . | ||
3.4. Estimation of between-trial heterogeneity from individual patient data of the 5 trials reconstructed from Kaplan-Meier curves: description of the I-squared method
| Comparison | Results of the heterogeneity assessment | |
| Previous method | Method proposed herein | |
|
1) Comparison between the five treatment arms pooled together versus the five control arms pooled together: |
Concordance= 0.521 (se = 0.012 ) Likelihood ratio test= 0.67 on 1 df, p=0.4 Wald test = 0.67 on 1 df, p=0.4 |
The reconstructed curves are shown in Figure 1, panel A; the heterogeneity assessment based on the I-square is shown in |
| 2) Comparison between the five treatment arms plotted individually: | Concordance= 0.565 (se = 0.02 ); Likelihood ratio test= 26.7 on 4 df, p=2e-05; Wald test = 24.82 on 4 df, p=5e-05 |
The reconstructed curves are shown in Figure 1, panel B. |
| 3) Comparison between the five control arms plotted individually: | Concordance= 0.59 (se = 0.02 ); Likelihood ratio test= 14.72 on 4 df, p=0.005; Wald test = 14.76 on 4 df, p=0.005 |
The reconstructed curves are shown in Figure 1, panel C. |
| Parameter | Does the parameter measure the overall effect of A vs B? | Does the parameter measure the between-trial heterogeneity ? | Is the parameter influenced by the overall effect? |
| I² | No | Yes | No |
| Wald test | Yes | No | Yes |
| Log-likelihood ratio§ | No | Yes | No |
| Concordance or C-index | Yes | No | Yes |
4. Discussion
Appendix A. Script in R-language that executes the estimation of between trial heterogeneity based on the worked example shown in Table 2.
|
install.packages("meta") library(meta) # Input of HRs with their respective 95%CI: studi <- c("Studio 1", "Studio 2", "Studio 3", "Studio 4", "Studio 5") HR <- c(1.359, 1.483, 0.864, 0.8578, 0.9102) lower_CI <- c(0.8623, 0.9657, 0.5857, 0.557, 0.668) upper_CI <- c(2.143, 2.278, 1.275, 1.321, 1.2399) # Running the meta-analysis meta_HR <- metagen( TE = log(HR), # log(HR) lower = log(lower_CI), # log(Lower CI) upper = log(upper_CI), # log(Upper CI) studlab = studi, sm = "HR", # hazard ratio comb.fixed = FALSE, # random-effects model comb.random = TRUE, method.tau = "DL" # DerSimonian-Laird method for estimating tau-squared ) # Main results print(meta_HR) forest(meta_HR) |
References
- Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002 Jun 15;21(11):1539-58. PMID: 12111919. [CrossRef]
- Liu N, Zhou Y, Lee JJ. IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med Res Methodol. 2021 Jun 1;21(1):111. PMID: 34074267; PMCID: PMC8168323. [CrossRef]
- Pratama S, Wiyono L, Setiawan MS, Lauren BC. PARP inhibitors as therapy for small cell lung carcinoma: A systematic review and meta-analysis of clinical trials. Cancer Treat Res Commun. 2024;42:100874. Epub 2025 Jan 27. PMID: 39892078. [CrossRef]
- Ai X, Y Pan, J Shi, et al., Efficacy and safety of niraparib as maintenance treatment in patients with extensive-stage SCLC after first-line chemotherapy: a randomized, double-blind, phase 3 study, J. Thorac. Oncol. 16 (8) (2021) 1403–1414. [CrossRef]
- Byers LA, D Bentsion, S Gans, et al., Veliparib in combination with carboplatin and etoposide in patients with treatment-naïve extensive-stage small cell lung cancer: a phase 2 randomized study, Clin. Cancer Res. Off J. Am. Assoc. Cancer Res 27 (14) (2021) 3884–3895. [CrossRef]
- Owonikoko TK, SE Dahlberg, GL Sica, et al., Randomized phase II trial of cisplatin and etoposide in combination with veliparib or placebo for extensive-stage small-cell lung cancer: ECOG-ACRIN 2511 study, J. Clin. Oncol. 37 (3) (2019) 222–229. [CrossRef]
- Pietanza MC, SN Waqar, LM Krug, et al. Randomized, double-blind, phase II study of temozolomide in combination with either veliparib or placebo in patients with relapsed-sensitive or refractory small-cell lung cancer, J. Clin. Oncol. 36 (23) (2018) 2386–2394. [CrossRef]
- Woll P, P Gaunt, S Danson, et al., Olaparib as maintenance treatment in patients with chemosensitive small cell lung cancer (STOMP): A randomised, double-blind, placebo-controlled phase II trial, Lung Cancer Amst. Neth 171 (2022) 26–33. [CrossRef]
- Hemming K, Hughes JP, McKenzie JE, Forbes AB. Extending the I-squared statistic to describe treatment effect heterogeneity in cluster, multi-centre randomized trials and individual patient data meta-analysis. Stat Methods Med Res. 2021 Feb;30(2):376-395. Epub 2020 Sep 21. PMID: 32955403; PMCID: PMC8173367. [CrossRef]

| First author and reference | Experimental group(s) | Control group | Maintenance therapy | Standard treatment | |
| Ai et al. [4] | 125 | 60 | Niraparib | ||
| Byers et al. [5] | 61 throughout |
59 Veliparib combination |
61 | Veliparib | |
| Owonikoko et al. [6] | 64 | 64 | Veliparib | CE | |
| Pietanza et al. [7] | 55 | 49 | veliparib | ||
| Woll et al. [8] | 73 Olaparib TDS |
73 Olaparib BD |
74 placebo |
Olaparib TDS or Olaparib BD | |
| Study | PARPI | Placebo | Risk ratio | ||||
| Events | Total | Events | Total | Risk ratio | Lower 95%CI | Upper 95%CI | |
| Ai 2021 | 48 | 125 | 22 | 60 | 1.05 | 0.70 | 1.96 |
| Byers 2021 § | 50 | 61 | 41 | 61 | 1.22 | 0.99 | 1.51 |
| Pietanza 2018 | 49 | 55 | 44 | 49 | 0.99 | 0.87 | 1.13 |
| Owonikoko 2019 | 51 | 64 | 54 | 64 | 0.94 | 0.80 | 1.11 |
| Woll 2022 §§ | 48 | 146 | 25 | 74 | 0.97 | 0.66 | 1.44 |
| Metanalysis | 1.03 | 0.92 | 1.15 | ||||
| Total events | 246 | 451 | 186 | 308 | 237 | ||
| Heterogeneity: | Chi-square=3.95, df=4, P=0.41, I-square=0%, Z=0.53, P=0.60 | ||||||
| Test for overall effect | Z = t 1.96, p = 0.05 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




