Submitted:
21 August 2025
Posted:
22 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
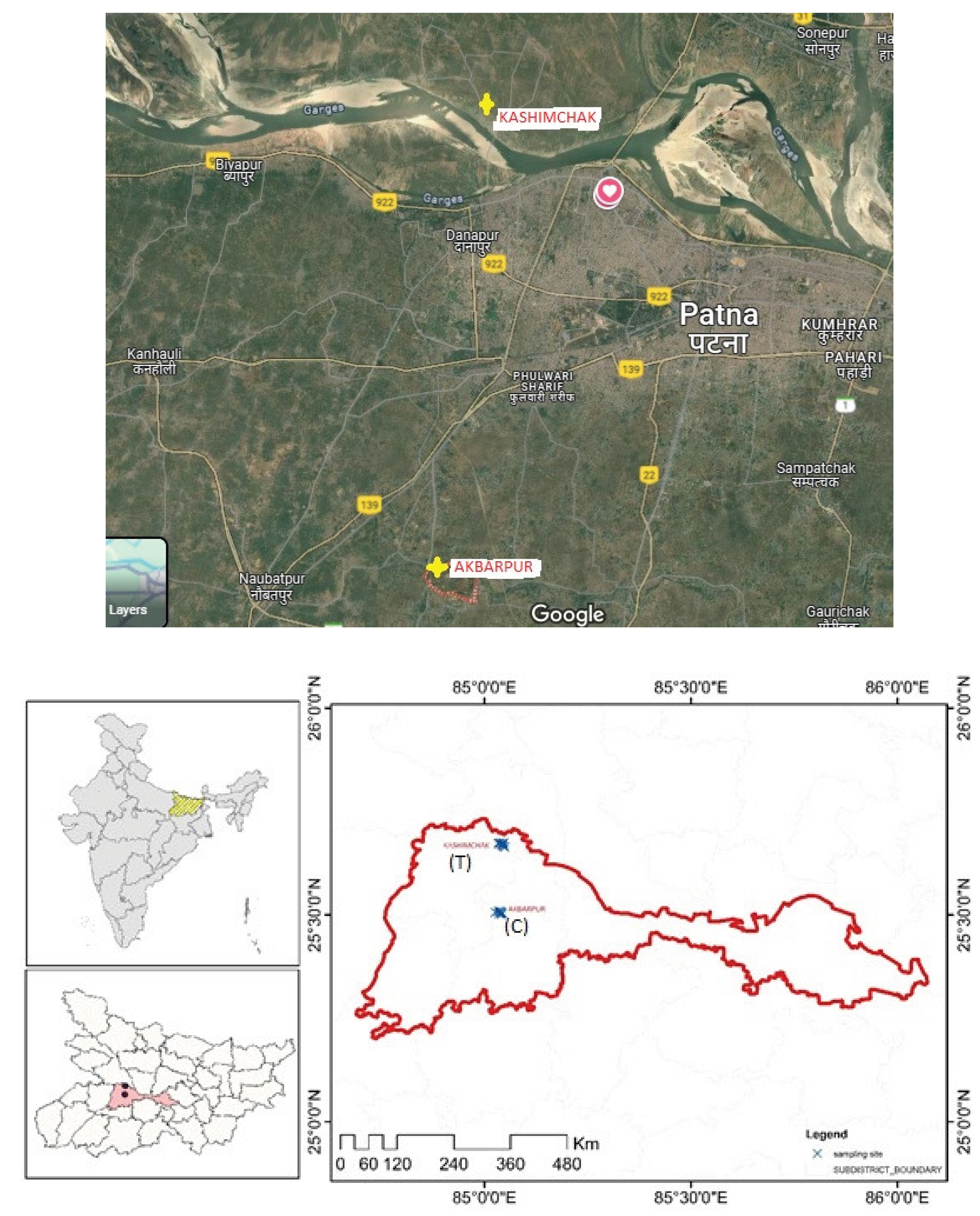
Materials and Methods
- a)
- Lactating cattle were born, reared, and maintained on fodder and water from the selected villages.
- b)
- The water source for drinking was groundwater from shallow tubewells burrowed about 40-100 meters.
- c)
- Cattle have a proper history of routine vaccination and deworming, and no clinical signs of infectious diseases.
Result and Discussion
Total Arsenic in Water and Feed
Total Arsenic in Lactating Cattle
Conclusion
Acknowledgment
Conflict of Interest
References
- Abhinav, S., Navin, S., Verma, S. K., Kumar, R., Ali, M., Kumar, A. and Ghosh, A. K. (2016, June). Groundwater and blood samples assessment for arsenic toxicity in rural population of Darbhanga district of Bihar, India. In Arsenic research and global sustainability–proceedings of the 6th international congress on arsenic in the environment. CRC Press, Taylor & Francis, London (pp. 419-420).
- Bjørklund, G., Oliinyk, P., Lysiuk, R., Rahaman, M. S., Antonyak, H., Lozynska, I., Lenchyk, L. and Peana, M. (2020). Arsenic intoxication: general aspects and chelating agents. Archives of toxicology, 94(6): 1879-1897.
- CCME (Canadian Council of Ministers of the Environment). (1993). Appendix XV—Protocols for deriving water quality guidelines for the protection of agricultural water uses (October 1993). In: Canadian water quality guidelines, Canadian Council of Resource and Environment Ministers. Available online: https://ccme.ca/en/res/arsenic-canadian-water-quality-guidelines-for-the-protection-of-agricultural-water-uses-en.pdf (accessed on 17 July 2025).
- Chakraborti, D., Rahman, M. M., Ahamed, S., Dutta, R. N., Pati, S. and Mukherjee, S.C. (2016). Arsenic contamination of groundwater and its induced health effects in Shahpur block, Bhojpur district, Bihar state, India: risk evaluation. Environmental Science and Pollution Research, 23(10): 9492-9504.
- Datta, B. K., Mishra, A., Singh, A., Sar, T. K., Sarkar, S., Bhatacharya, A., Chakraborty, A.K. and Mandal, T. K. (2010). Chronic arsenicosis in cattle with special reference to its metabolism in arsenic endemic village of Nadia district West Bengal, India. Science of the total environment, 409(2): 284-288.
- Dey, T. K., Banerjee, P., Bakshi, M., Kar, A. and Ghosh, S. (2014). Groundwater arsenic contamination in West Bengal: current scenario, effects and probable ways of mitigation. International Letters of Natural Sciences, 13: 45–58. https://doi.org/10.18052/www.scipress.com/ILNS.13.45. [CrossRef]
- Dube R.S, Singh S, Gupte A, Modi A. (2024) Arsenic Contamination in Eastern India: Exploring the Impact, Mitigation, and Bioremediation Strategies. Curr Agri Res, 12(2). [CrossRef]
- Ghosh, A., Singh, S. K., Bose, N., Roy, N. P., Singh, S. K., Upadhyaya, A. K., ... and Singh, A. (2009). Arsenic Hot Spots Detected in Patna District, Bihar (India) A Serious Health Hazard for Estimated Human Population of 5.5 Lakhs. Assessment of groundwater resources and management, 62-70.
- Joint F (2011). Organization WH, Additives WECoF. Evaluation of certain contaminants in food: seventy-second [72nd] report of the Joint FAO/WHO Expert Committee on Food Additives: World Health Organization.s.
- Jones, J.S. and Hatch, M.B. (1945). Spray residues and crop assimilation of Arsenic and lead. Soil Sci., 60:277–88.
- Klaassen, C. D. (2006). Heavy metals and heavy-metal antagonists. In: Goodman & Gilman’s the pharmacological basis of therapeutics, 1753-1775.
- Kumar, A., Ali, M., Rahman, S. M., Iqubal, A. M., Anand, G., Niraj, P. K., Shankar, P. and Kumar, R. (2015). Groundwater arsenic poisoning in “Tilak Rai Ka Hatta” village of Buxar district, Bihar, India causing severe health hazards and hormonal imbalance. J Environ Anal Toxicol, 5(4): 1-7.
- Kumar, A., Kumar, R., Ali, M., Gahlot, V. and Ghosh, A. (2016). Groundwater arsenic poisoning in Buxar District, Bihar, India: health hazards. In Arsenic Research and Global Sustainability: Proceedings of the Sixth International Congress on Arsenic in the Environment (As2016), June 19-23, 2016, Stockholm, Sweden (Vol. 378). CRC Press.
- Lakso, A. N. and Kliewer, W. M. (1975). The influence of temperature on malic acid metabolism in grape berries: I. Enzyme responses. Plant physiology, 56(3): 370-372.
- Maji, C., Sarkar, S., Biswas, S., Patra, P. H., Datta, B. K., Bandyopadhyay, S., Biswas, T.K., Jana, C. and Mandal, T. K. (2016). Experimental assessment of arsenic toxicity in garole sheep in India. Emerging Contaminants, 2(3): 128-134.
- Rahman, M. S., Kumar, A., Kumar, R., Ali, M., Ghosh, A. K. and Singh, S. K. (2019). Comparative quantification study of arsenic in the groundwater and biological samples of Simri village of Buxar District, Bihar, India. Indian journal of occupational and environmental medicine, 23(3): 126-132. [CrossRef]
- Nandi, D., Patra, R.C. and Swarup, D. (2005). Arsenic residues in hair samples from cattle in some Arsenic affected areas of west Bengal, India. Environ. Contamin. Toxicol. 75 (2): 251-256.
- Nath, A., Vendan, P.S., Kumar, S., Kumar, A. and Singh, J.K. (2013). Toxicity due to Arsenic in Gangetic Zone of Patna, India, and its linkage with cancer. J. Environ. Anal. Toxicol. 3: 192.
- National Research Council (NRC) (1991). Arsenic in Drinking Water. (1999). Arsenic in drinking water. National Academy Press, Washington, DC, pp 1-310.
- Pal, A., Nayak, B., Das, B., Hossain, M. A., Ahamed, S., and Chakraborti, D. (2007). Additional danger of arsenic exposure through inhalation from burning of cow dung cakes laced with Arsenic as a fuel in Arsenic affected villages in Ganga–Meghna–Brahmaputra plain. J. environ. Monit, 9(10), 1067-1070.
- Paramita Mandal (2017) An insight of environmental contamination of Arsenic on animal health Emerging Contaminants 3 (1), 17-22.
- Prakash, C., Soni, M. and Kumar, V. (2015). Biochemical and molecular alterations following arsenic-induced oxidative stress and mitochondrial dysfunction in rat brain. Biological trace element research, 167(1): 121-129.
- Radostits. O.M. Gay, C.C. Blood, D.C. Incheliff, K.W. (2000). Veterinary medicine: A textbook of the diseases of cattle. Sheep, Pigs, goats and horses. 9th ed. W.B. Saunders Company. London, Philadelphia. 1585-1589.
- Rana, T., Bera, A. K., Das, S., Bhattacharya, D., Bandyopadhyay, S., Pan, D., & Das, S. K. (2010). Effect of chronic intake of arsenic-contaminated water on blood oxidative stress indices in cattle in an arsenic-affected zone. Ecotoxicology and environmental safety, 73(6), 1327-1332.
- Rana, T., Sarkar, S., Mandal, T., Bhattacharyya, K., Roy, A., & Kol, L. (2008). Contribution of Arsenic from agricultural food chain to cow milk in highly arsenic prone zone in Nadia District of West Bengal in India. The Int J Vet Med, 4(2).
- Roy P, and Saha A. (2002). Review article of Metabolism and toxicity of Arsenic a human carcinogen. Curr Sci. 82:38–45.
- Saha, U. S., Prajapati, J. B., Sompurkar, M. S., & Mohapatra, S. S. (2022). Water Footprint of Milk Production in India: A case of Anand District of Gujarat. IRMA Working Paper 336, Pp 1-29 ISSN 2454-7115. Retrieved on 19.08.2025 https://www.academia.edu/86222336/Water_Footprint_of_Milk_Production_in_India_A_case_of_Anand_District_of_Gujarat.
- Shrivastava BK. (2016) Policy intervention for arsenic mitigation in drinking water in rural habitations in India: achievements and challenges. J Water Health, 14:827-838.
- Singh, A. and Choudhary, S.K. (2010). Arsenic in groundwater in five villages under Nathnagar Block of Bhagalpur District, Bihar. Ecoscan, 4(2&3): 213-216.
- Singh, S.K. and Vedwan, N., (2015). Mapping composite vulnerability to groundwater arsenic contamination: an analytical framework and a case study in India. Natural Hazards, 75(2), pp.1883-1908.
- Singh, S.K., (2015). Groundwater arsenic contamination in the Middle-Gangetic plain, Bihar (India): the danger arrived. Int. Res. J. Environ. Sc, 4(2):70-76.
- Singh, S.K., Ghosh, A.K., Kumar, A., Kislay, K., Kumar, C., Tiwari, R.R., Parwez, R., Kumar, N. and Imam, M.D., 2014. Groundwater arsenic contamination and associated health risks in Bihar, India. Int. J. Environ. Res., 8(1), pp.49-60 2161-0525.
- WHO (2003). Environmental Health Criteria-224. Arsenic and Arsenic Compounds, second ed., World Health Organization, Geneva,.
- WHO (2005). Water sanitation and health. Guidelines for drinking water quality. 3rd ed. Geneva.
| Kashimchak (T) As-affected village | Akbarpur (C) As-safe village | |
| Water (mg/L) |
0.079±0.0045a (0.06-0.1) |
0.007±0.0012b (0.00-0.01) |
| Straw (mg/Kg) |
1.046±0.077a (0.55-1.35) |
0.058±0.016b (0.1-0.17) |
| Sample | Arsenic (ppm) concentration | |
| Kashimchak (T) | Akbarpur (C) | |
| Blood (mg/L) | 0.287±0.008a (0.24-0.32) |
0.011±0.003b (0.00-0.04) |
| Hair (mg/kg) | 1.692±0.173a (0.88-2.24) |
0.103±0.0191b (0.03-0.19) |
| Milk (mg/L) | 0.080±0.003a (0.07-0.10) |
0.013±0.006b (0.00-0.07) |
| Urine (mg/L) | 0.083±0.004a (0.06-0.10) |
0.032±0.003b (0.02-0.05) |
| Faeces (mg/kg) | 0.440±0.011a (0.37-0.49) |
0.073±0.009b (0.03-0.49) |
| Feed | Water | Blood | Milk | Hair | Urine | Dung | ||
| Feed | Pearson Correlation | 1 | 0.391 | 0.948** | 0.897** | 0.892** | 0.837** | 0.804** |
| Water | Pearson Correlation | 0.391 | 1 | 0.207 | 0.247 | 0.303 | 0.153 | 0.191 |
| Blood | Pearson Correlation | 0.948** | 0.207 | 1 | 0.934** | 0.787** | 0.773** | 0.742* |
| Milk | Pearson Correlation | 0.897** | 0.247 | 0.934** | 1 | 0.727* | 0.701* | 0.610 |
| Hair | Pearson Correlation | 0.892** | 0.303 | 0.787** | 0.727* | 1 | 0.857** | 0.792** |
| Urine | Pearson Correlation | 0.837** | 0.153 | 0.773** | 0.701* | 0.857** | 1 | 0.815** |
| Dung | Pearson Correlation | 0.804** | 0.191 | 0.742* | 0.610 | 0.792** | 0.815** | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




