Submitted:
15 August 2025
Posted:
18 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Protein Expression and Purification
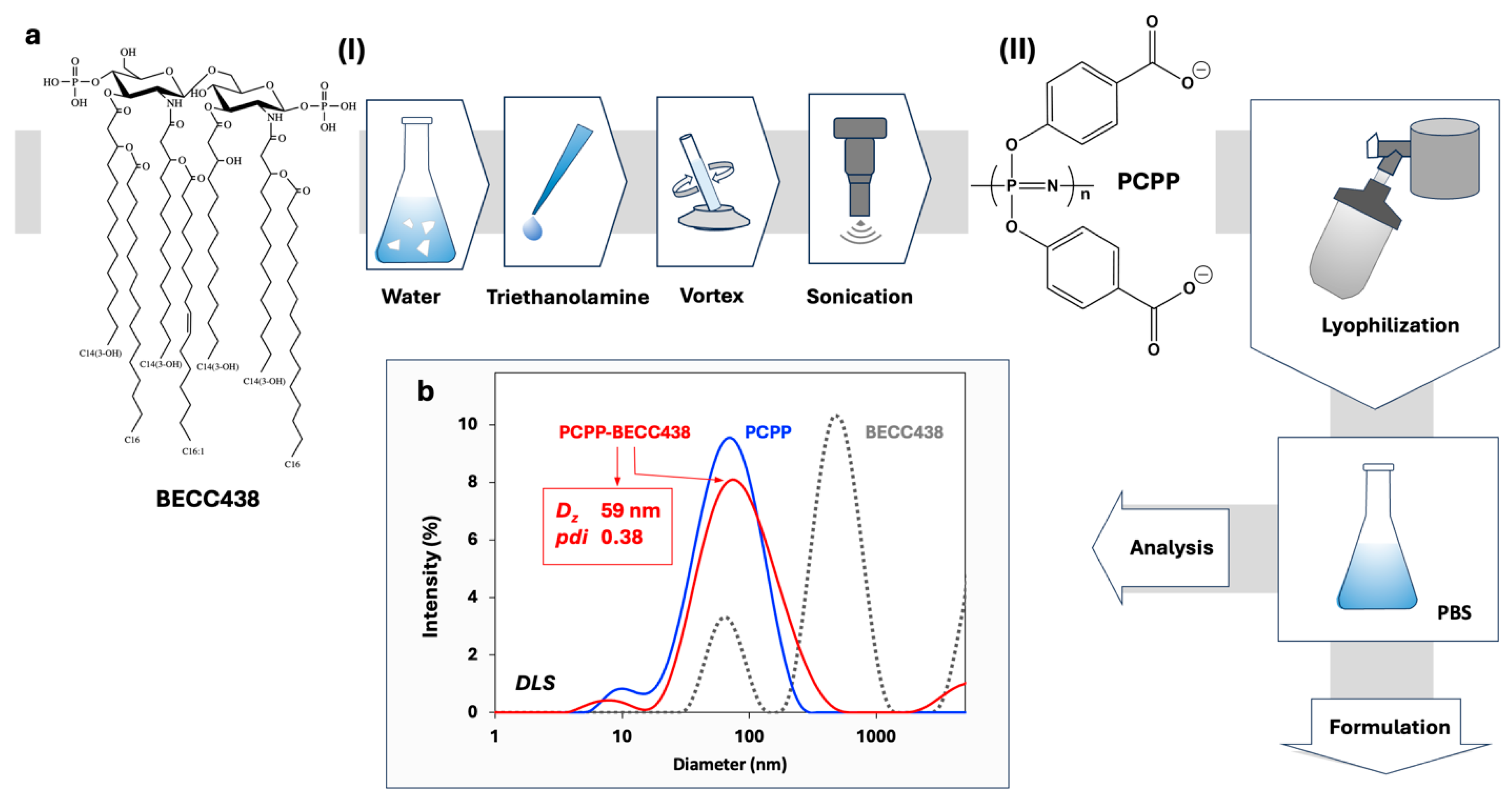
2.2. Preparation of BECC438
2.3. Preparation of PCPP-BECC438 Formulations
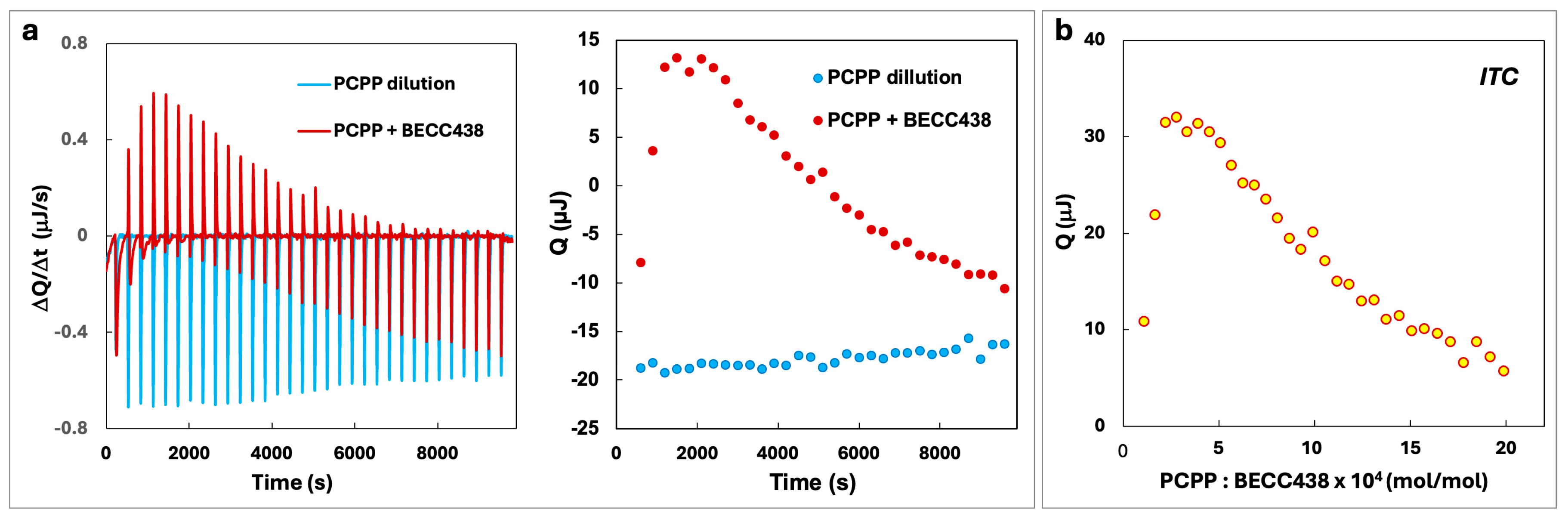
2.3. Dynamic Light Scattering (DLS) and Isothermal Titration Calorimetry (ITC) Analysis
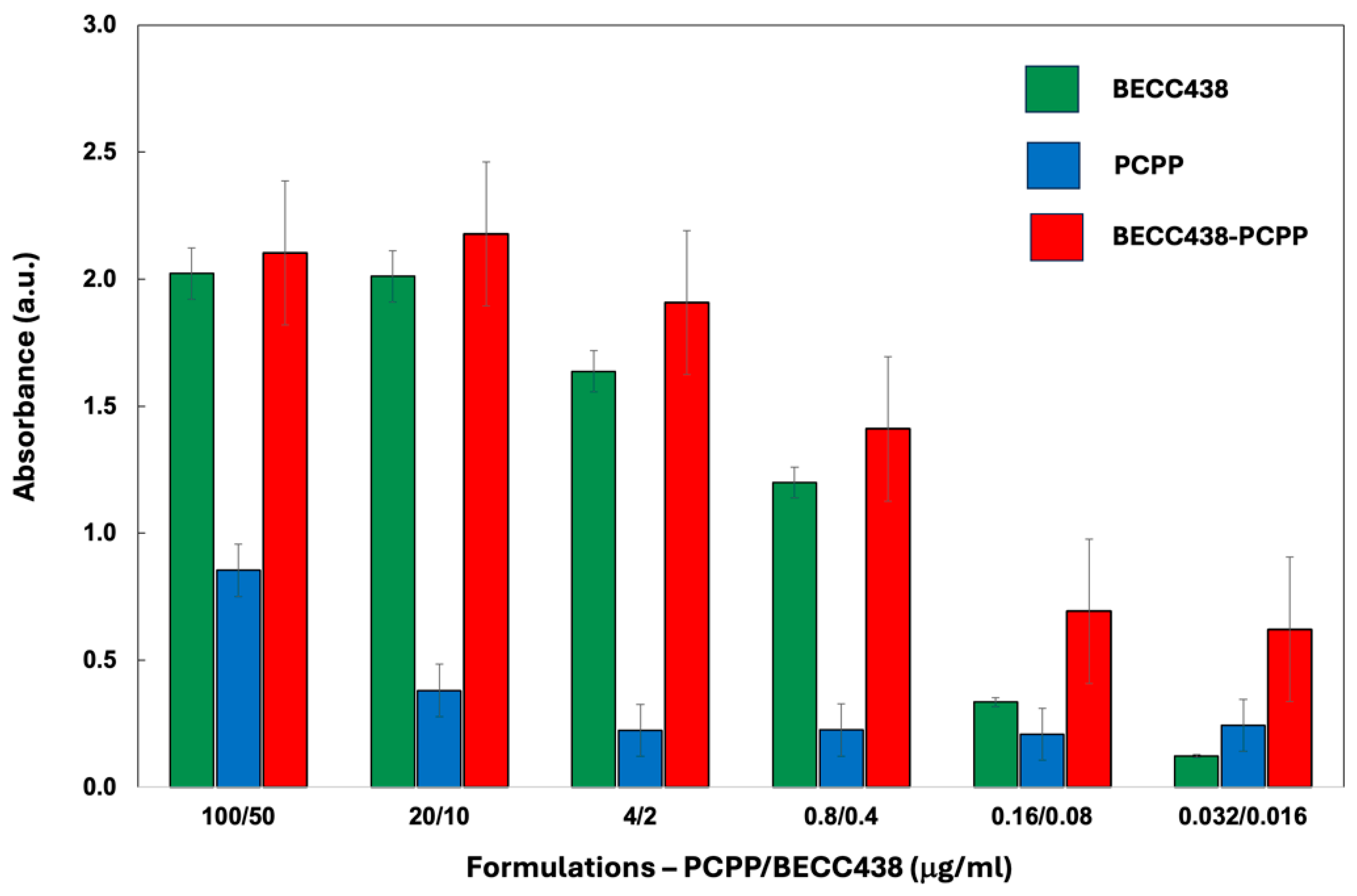
2.4. In Vitro Evaluation of Immunostimulating Activity
2.5. In Vivo Studies
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. HCV Pseudoparticles (HCVpp) Neutralization Assay
2.8. Statistical Analysis
3. Results
3.1. PCPP Enables Water-Soluble Formulations of BECC438
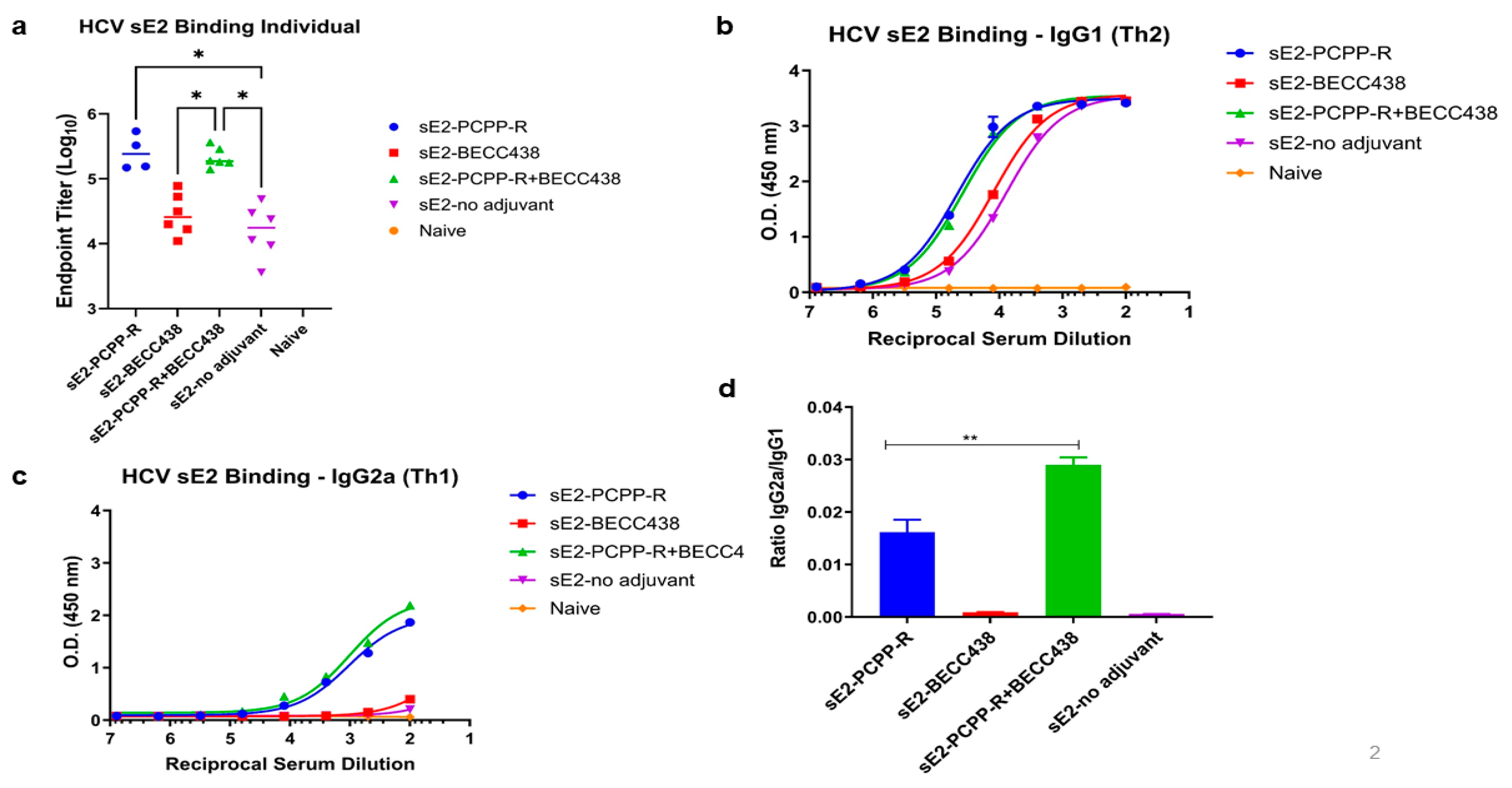
3.2. In Vivo Studies
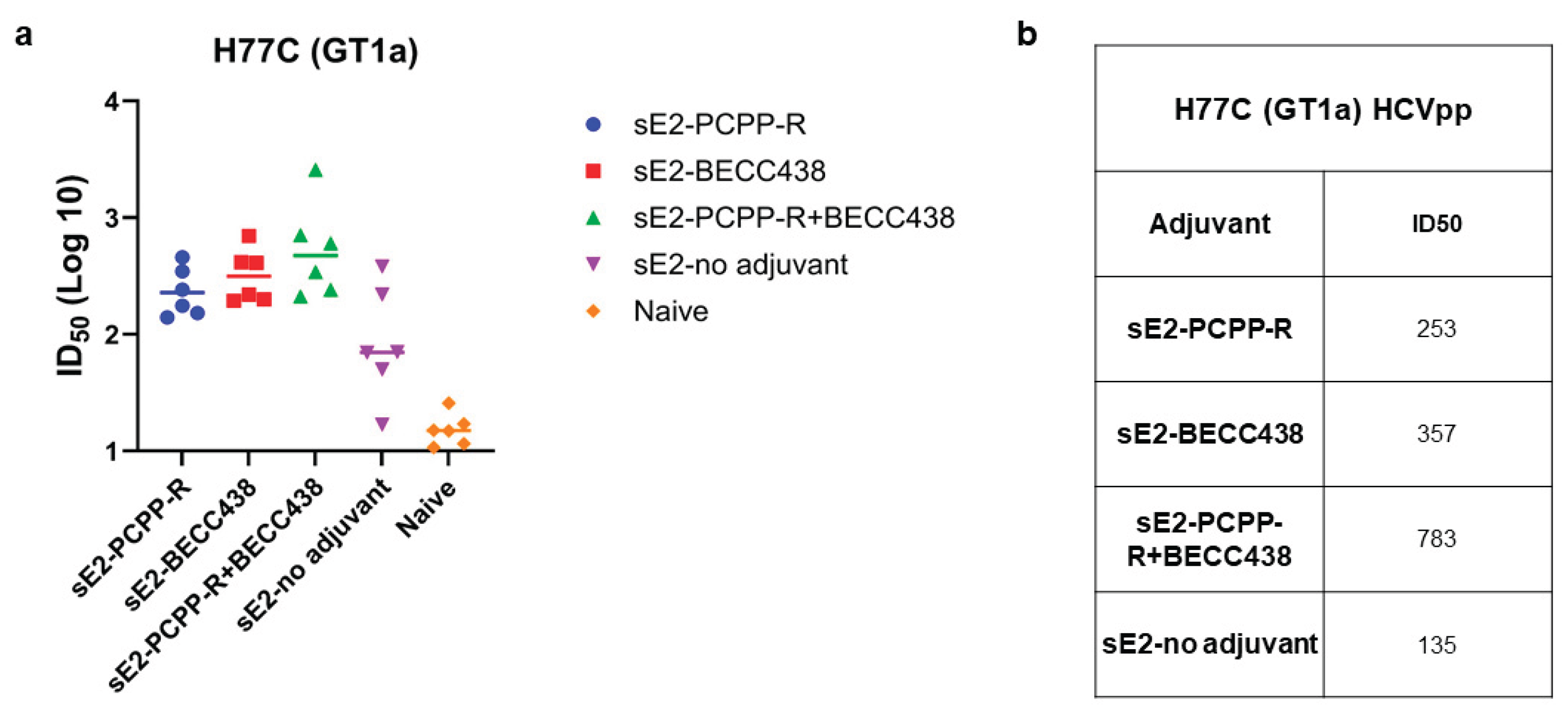
3.3. Evaluation of Serological Responses and Neutralization
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PCPP | Poly[di(carboxylatophenoxy)phosphazene] |
| BECC | Bacterial Enzymatic Combinatorial Chemistry |
| DLS | Dynamic light scattering |
| ITC…. | Isothermal titration calorimetry |
| Dz | z-average hydrodynamic diameter |
| pdi | polydispersity index |
| TLR | Toll-like receptor |
| R848 | Resiquimod |
| PCPP-R | PCPP containing resiquimod as a counterion |
| API | Active pharmaceutical ingredient |
| SEAP | Secreted embryonic alkaline phosphatase |
| ELISA | Enzyme-linked immunosorbent assay |
References
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Brito, L.A.; Malyala, P.; O’Hagan, D.T. Vaccine adjuvant formulations: A pharmaceutical perspective. Semin. Immunol. 2013, 25, 130–145. [Google Scholar] [CrossRef]
- Petrovsky, N.; Heinzel, S.; Honda, Y.; Lyons, A.B. New-Age Vaccine Adjuvants: Friend or Foe? New-Age 2007. [Google Scholar]
- Pulendran, B.; S. Arunachalam, P.; O’Hagan, D.T. Emerging concepts in the science of vaccine adjuvants. Nat. Rev. Drug. Discov. 2021, 20, 454–475. [Google Scholar] [CrossRef]
- Nanishi, E.; Dowling, D.J.; Levy, O. Toward precision adjuvants: optimizing science and safety. Curr. Opin. Pediatr. 2020, 32, 125–138. [Google Scholar] [CrossRef]
- Persing, D.H.; Coler, R.N.; Lacy, M.J.; Johnson, D.A.; Baldridge, J.R.; Hershberg, R.M.; Reed, S.G. Taking toll: lipid A mimetics as adjuvants and immunomodulators. Trends Microbiol. 2002, 10, s32–s37. [Google Scholar] [CrossRef]
- Garçon, N.; Chomez, P.; Van Mechelen, M. GlaxoSmithKline Adjuvant Systems in vaccines: concepts, achievements and perspectives. Expert review of vaccines 2007, 6, 723–739. [Google Scholar] [CrossRef]
- Catanese, M.T.; Ansuini, H.; Graziani, R.; Huby, T.; Moreau, M.; Ball, J.K.; Paonessa, G.; Rice, C.M.; Cortese, R.; Vitelli, A.; et al. Role of scavenger receptor class B type I in hepatitis C virus entry: kinetics and molecular determinants. J Virol 2010, 84, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Gregg, K.A.; Harberts, E.; Gardner, F.M.; Pelletier, M.R.; Cayatte, C.; Yu, L.; McCarthy, M.P.; Marshall, J.D.; Ernst, R.K. Rationally designed TLR4 ligands for vaccine adjuvant discovery. MBio 2017, 8, e00492–00417. [Google Scholar] [CrossRef]
- Gregg, K.A.; Harberts, E.; Gardner, F.M.; Pelletier, M.R.; Cayatte, C.; Yu, L.; McCarthy, M.P.; Marshall, J.D.; Ernst, R.K. A lipid A-based TLR4 mimetic effectively adjuvants a Yersinia pestis rF-V1 subunit vaccine in a murine challenge model. Vaccine 2018, 36, 4023–4031. [Google Scholar] [CrossRef] [PubMed]
- Andrianov, A.K.; Langer, R. Polyphosphazene immunoadjuvants: Historical perspective and recent advances. J. Controlled Release 2021, 329, 299–315. [Google Scholar] [CrossRef]
- Palmer, C.D.; Ninković, J.; Prokopowicz, Z.M.; Mancuso, C.J.; Marin, A.; Andrianov, A.K.; Dowling, D.J.; Levy, O. The effect of stable macromolecular complexes of ionic polyphosphazene on HIV Gag antigen and on activation of human dendritic cells and presentation to T-cells. Biomaterials 2014, 35, 8876–8886. [Google Scholar] [CrossRef]
- Awate, S.; Wilson, H.L.; Singh, B.; Babiuk, L.A.; Mutwiri, G. The adjuvant PCEP induces recruitment of myeloid and lymphoid cells at the injection site and draining lymph node. Vaccine 2014, 32, 2420–2427. [Google Scholar] [CrossRef]
- Awate, S.; Wilson, H.L.; Lai, K.; Babiuk, L.A.; Mutwiri, G. Activation of adjuvant core response genes by the novel adjuvant PCEP. Mol. Immunol. 2012, 51, 292–303. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Marin, A.; Fuerst, T.R. Molecular-Level Interactions of Polyphosphazene Immunoadjuvants and Their Potential Role in Antigen Presentation and Cell Stimulation. Biomacromolecules 2016, 17, 3732–3742. [Google Scholar] [CrossRef] [PubMed]
- Andrianov, A.K.; Marin, A.; Wang, R.; Chowdhury, A.; Agnihotri, P.; Yunus, A.S.; Pierce, B.G.; Mariuzza, R.A.; Fuerst, T.R. In Vivo and In Vitro Potency of Polyphosphazene Immunoadjuvants with Hepatitis C Virus Antigen and the Role of Their Supramolecular Assembly. Mol. Pharm. 2021, 18, 726–734. [Google Scholar] [CrossRef]
- Fuerst, T.R.; Marin, A.; Jeong, S.; Kulakova, L.; Hlushko, R.; Gorga, K.; Toth, E.A.; Singh, N.J.; Andrianov, A.K. Virus-Mimicking Polymer Nanocomplexes Co-Assembling HCV E1E2 and Core Proteins with TLR 7/8 Agonist—Synthesis, Characterization, and In Vivo Activity. J. Funct. Biomater. 2025, 16, 34. [Google Scholar] [CrossRef]
- Bouveret Le Cam, N.N.; Ronco, J.; Francon, A.; Blondeau, C.; Fanget, B. Adjuvants for influenza vaccine. Res. Immunol. 1998, 149, 19–23. [Google Scholar] [CrossRef]
- Ison, M.G.; Mills, J.; Openshaw, P.; Zambon, M.; Osterhaus, A.; Hayden, F. Current research on respiratory viral infections: Fourth International Symposium. Antiviral Res. 2002, 55, 227–278. [Google Scholar] [CrossRef] [PubMed]
- Thongcharoen, P.; Suriyanon, V.; Paris, R.M.; Khamboonruang, C.; de Souza, M.S.; Ratto-Kim, S.; Karnasuta, C.; Polonis, V.R.; Baglyos, L.; El Habib, R. A Phase 1/2 Comparative Vaccine Trial of the Safety and Immunogenicity of a CRF01_AE (Subtype E) Candidate Vaccine: ALVAC-HIV (vCP1521) Prime With Oligomeric gp160 (92TH023/LAI-DID) or Bivalent gp120 (CM235/SF2) Boost. J. Acquired Immune Defic. Syndr. 2007, 46, 48–55. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, R.J.; Excler, J.-L.; Polonis, V.R.; Ratto-Kim, S.; Cox, J.; Jagodzinski, L.L.; Liu, M.; Wieczorek, L.; McNeil, J.G.; El-Habib, R. Safety and Immunogenicity of a randomized Phase I prime-boost trial with ALVAC-HIV (vCP205) and Oligomeric gp160 MN/LAI-2 Adjuvanted in Alum or Polyphosphazene. J. Infect. Dis. 2016, 213, 1946–1954. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, R.; Polonis, V.; Ratto-Kim, S.; Cox, J.; Jagodzinski, L.; Malia, J.; Michael, N.; Excler, J.; Robb, M.; Kim, J. Safety and immunogenicity of a randomized phase I prime-boost trial with ALVAC-HIV (vCP205) and gp160 MN/LAI-2 adjuvanted in alum or polyphosphazene. Retrovirology 2012, 9, O50. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Marin, A.; Wang, R.; Karauzum, H.; Chowdhury, A.; Agnihotri, P.; Yunus, A.S.; Mariuzza, R.A.; Fuerst, T.R. Supramolecular assembly of Toll-like receptor 7/8 agonist into multimeric water-soluble constructs enables superior immune stimulation in vitro and in vivo. ACS Appl. Bio Mater. 2020, 3, 3187–3195. [Google Scholar] [CrossRef]
- WHO Hepatitis C Key Facts. Available online: https://www/who.int/en/news-room/fact-sheets/detail/hepatitis-c (accessed on 9 April 2025).
- Fiehn, F.; Beisel, C.; Binder, M. Hepatitis C virus and hepatocellular carcinoma: carcinogenesis in the era of direct-acting antivirals. Curr Opin Virol 2024, 67, 101423. [Google Scholar] [CrossRef]
- Caniglia, E.C.; Cain, L.E.; Sabin, C.A.; Robins, J.M.; Logan, R.; Abgrall, S.; Mugavero, M.J.; Hernandez-Diaz, S.; Meyer, L.; Seng, R.; et al. Comparison of dynamic monitoring strategies based on CD4 cell counts in virally suppressed, HIV-positive individuals on combination antiretroviral therapy in high-income countries: a prospective, observational study. Lancet HIV 2017, 4, e251–e259. [Google Scholar] [CrossRef]
- Hepatitis - The Silent Epidemic. May 20, 2020. 20 May.
- Yukawa, Y.; Tamori, A.; Iio, E.; Ogawa, S.; Yoshida, K.; Uchida-Kobayashi, S.; Enomoto, M.; Tanaka, Y.; Kawada, N. Hepatitis C virus recurrence in two patients who achieved sustained viral response with interferon-free direct-acting antiviral therapy: reinfection or relapse? Clinical journal of gastroenterology 2019, 12, 598–602. [Google Scholar] [CrossRef]
- Berenguer, J.; Gil-Martin, A.; Jarrin, I.; Montes, M.L.; Dominguez, L.; Aldamiz-Echevarria, T.; Tellez, M.J.; Santos, I.; Troya, J.; Losa, J.E.; et al. Reinfection by hepatitis C virus following effective all-oral direct-acting antiviral drug therapy in HIV/hepatitis C virus coinfected individuals. AIDS 2019, 33, 685–689. [Google Scholar] [CrossRef]
- Rossi, C.; Butt, Z.A.; Wong, S.; Buxton, J.A.; Islam, N.; Yu, A.; Darvishian, M.; Gilbert, M.; Wong, J.; Chapinal, N.; et al. Hepatitis C virus reinfection after successful treatment with direct-acting antiviral therapy in a large population-based cohort. Journal of hepatology 2018, 69, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Hepatitis Report 2024; World Health Organization, 2024. [Google Scholar]
- Borgia, S.M.; Hedskog, C.; Parhy, B.; Hyland, R.H.; Stamm, L.M.; Brainard, D.M.; Subramanian, M.G.; McHutchison, J.G.; Mo, H.; Svarovskaia, E.; et al. Identification of a novel hepatitis C virus genotype from Punjab, India: Expanding classification of hepatitis C virus into 8 genotypes. Journal of Infectious Diseases 2018, 218, 1722–1729. [Google Scholar] [CrossRef]
- Fuerst, T.R.; Pierce, B.G.; Keck, Z.Y.; Foung, S.K.H. Designing a B Cell-Based Vaccine against a Highly Variable Hepatitis C Virus. Frontiers in microbiology 2017, 8, 2692. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.L. Challenges and Promise of a Hepatitis C Virus Vaccine. Cold Spring Harb Perspect Med 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Duggal, P.; Thio, C.L.; Wojcik, G.L.; Goedert, J.J.; Mangia, A.; Latanich, R.; Kim, A.Y.; Lauer, G.M.; Chung, R.T.; Peters, M.G.; et al. Genome-wide association study of spontaneous resolution of hepatitis C virus infection: data from multiple cohorts. Annals of internal medicine 2013, 158, 235–245. [Google Scholar] [CrossRef]
- Urbanowicz, R.A.; Wang, R.; Schiel, J.E.; Keck, Z.-y.; Kerzic, M.C.; Lau, P.; Rangarajan, S.; Garagusi, K.J.; Tan, L.; Guest, J.D.; et al. Antigenicity and Immunogenicity of Differentially Glycosylated Hepatitis C Virus E2 Envelope Proteins Expressed in Mammalian and Insect Cells. J. Virol. 2019, 93, e01403–01418. [Google Scholar] [CrossRef] [PubMed]
- Pierce, B.G.; Keck, Z.-Y.; Wang, R.; Lau, P.; Garagusi, K.; Elkholy, K.; Toth, E.A.; Urbanowicz, R.A.; Guest, J.D.; Agnihotri, P.; et al. Structure-based design of hepatitis C virus E2 glycoprotein improves serum binding and cross-neutralization. J. Virol. 2020, 94, e00704–00720. [Google Scholar] [CrossRef]
- Zacharia, A.; Harberts, E.; Valencia, S.M.; Myers, B.; Sanders, C.; Jain, A.; Larson, N.R.; Middaugh, C.R.; Picking, W.D.; Difilippantonio, S.; et al. Optimization of RG1-VLP vaccine performance in mice with novel TLR4 agonists. Vaccine 2021, 39, 292–302. [Google Scholar] [CrossRef]
- Siepmann, J.; Faham, A.; Clas, S.-D.; Boyd, B.J.; Jannin, V.; Bernkop-Schnürch, A.; Zhao, H.; Lecommandoux, S.; Evans, J.C.; Allen, C.; et al. Lipids and polymers in pharmaceutical technology: Lifelong companions. Int. J. Pharm. 2019, 558, 128–142. [Google Scholar] [CrossRef]
- Kesisoglou, F.; Panmai, S.; Wu, Y. Nanosizing — Oral formulation development and biopharmaceutical evaluation. Adv. Drug Delivery Rev. 2007, 59, 631–644. [Google Scholar] [CrossRef]
- Heurtault, B.; Saulnier, P.; Pech, B.; Proust, J.-E.; Benoit, J.-P. Physico-chemical stability of colloidal lipid particles. Biomaterials 2003, 24, 4283–4300. [Google Scholar] [CrossRef]
- Andrianov, A.K.; Fuerst, T.R. Immunopotentiating and Delivery Systems for HCV Vaccines. Viruses 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Landi, A.; Law, J.; Hockman, D.; Logan, M.; Crawford, K.; Chen, C.; Kundu, J.; Ebensen, T.; Guzman, C.A.; Deschatelets, L.; et al. Superior immunogenicity of HCV envelope glycoproteins when adjuvanted with cyclic-di-AMP, a STING activator or archaeosomes. Vaccine 2017, 35, 6949–6956. [Google Scholar] [CrossRef]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat Med 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Kayesh, M.E.H.; Kohara, M.; Tsukiyama-Kohara, K. TLR agonists as vaccine adjuvants in the prevention of viral infections: an overview. Frontiers in microbiology 2023, 14, 1249718. [Google Scholar] [CrossRef] [PubMed]
- Weissenberger, G.; Henderikx, R.J.M.; Peters, P.J. Understanding the invisible hands of sample preparation for cryo-EM. Nat Methods 2021, 18, 463–471. [Google Scholar] [CrossRef]
- Yip, K.M.; Fischer, N.; Paknia, E.; Chari, A.; Stark, H. Atomic-resolution protein structure determination by cryo-EM. Nature 2020, 587, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y. Single-particle cryo-EM-How did it get here and where will it go. Science 2018, 361, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Petrovsky, N. Comparative Safety of Vaccine Adjuvants: A Summary of Current Evidence and Future Needs. Drug Saf 2015, 38, 1059–1074. [Google Scholar] [CrossRef]
- Iyer, V.; Cayatte, C.; Guzman, B.; Schneider-Ohrum, K.; Matuszak, R.; Snell, A.; Rajani, G.M.; McCarthy, M.P.; Muralidhara, B. Impact of formulation and particle size on stability and immunogenicity of oil-in-water emulsion adjuvants. Hum Vaccin Immunother 2015, 11, 1853–1864. [Google Scholar] [CrossRef]
- Iyer, V.; Cayatte, C.; Marshall, J.D.; Sun, J.; Schneider-Ohrum, K.; Maynard, S.K.; Rajani, G.M.; Bennett, A.S.; Remmele, R.L., Jr.; Bishop, S.M.; et al. Feasibility of Freeze-Drying Oil-in-Water Emulsion Adjuvants and Subunit Proteins to Enable Single-Vial Vaccine Drug Products. J Pharm Sci 2017, 106, 1490–1498. [Google Scholar] [CrossRef]
| Group | Label | sE2 FL (µg) | PCPP-R (µg) | BECC438 (µg) |
|---|---|---|---|---|
| 1 | sE2-PCPP-R | 50 | 50 | 0 |
| 2 | sE2-BECC438 | 50 | 0 | 25 |
| 3 | sE2-PCPP-R+BECC438 | 50 | 50 | 25 |
| 4 | sE2-no adjuvant | 50 | 0 | 0 |
| 5 | Naive | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




