Submitted:
11 August 2025
Posted:
12 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Image Analysis
- The .tif files were loaded into MATLAB and binarized
- Objects were defined using the bwlabeln function with 26 connectivity
- Object volumes were measured with the regionprops3 function
- Small objects (<100 voxels) were removed with the function bwareaopen
- Outliers deriving from segmentation artefacts were removed with rmoutlier
- Normality of data was checked with chi2gof at 5% significance
- The presence of multiple populations was checked with fitgmdist, choosing the number of populations (between 1 and 10) that minimized Akaike information criterion. Best numbers were three for total population, and two for reuniting and ED populations.
- Sum of gaussians fit was performed using the nlsLM function in R. The parameters A, µ, and σ indicated amplitude, mean and spread for each gaussian component.
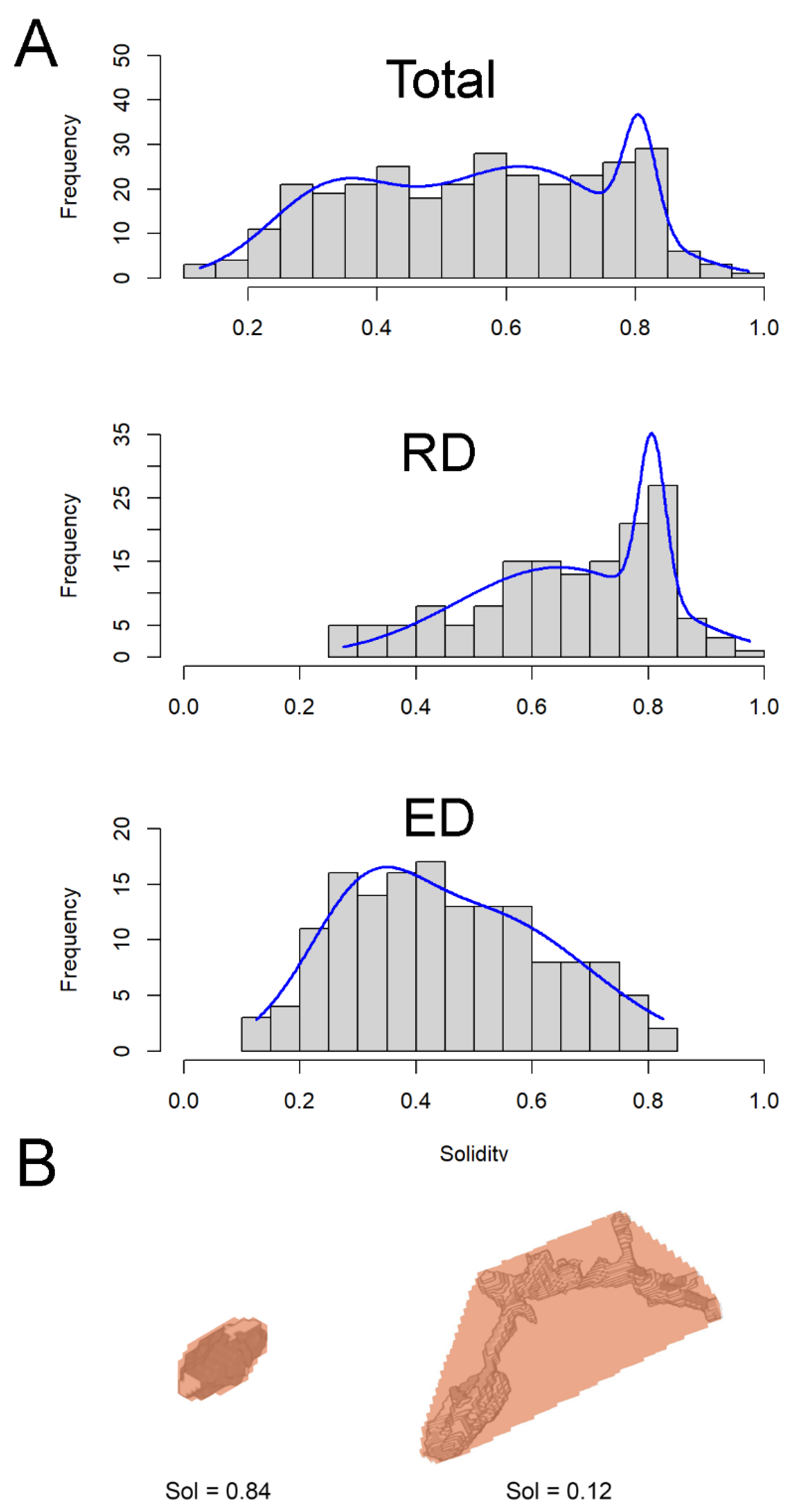
3. Results
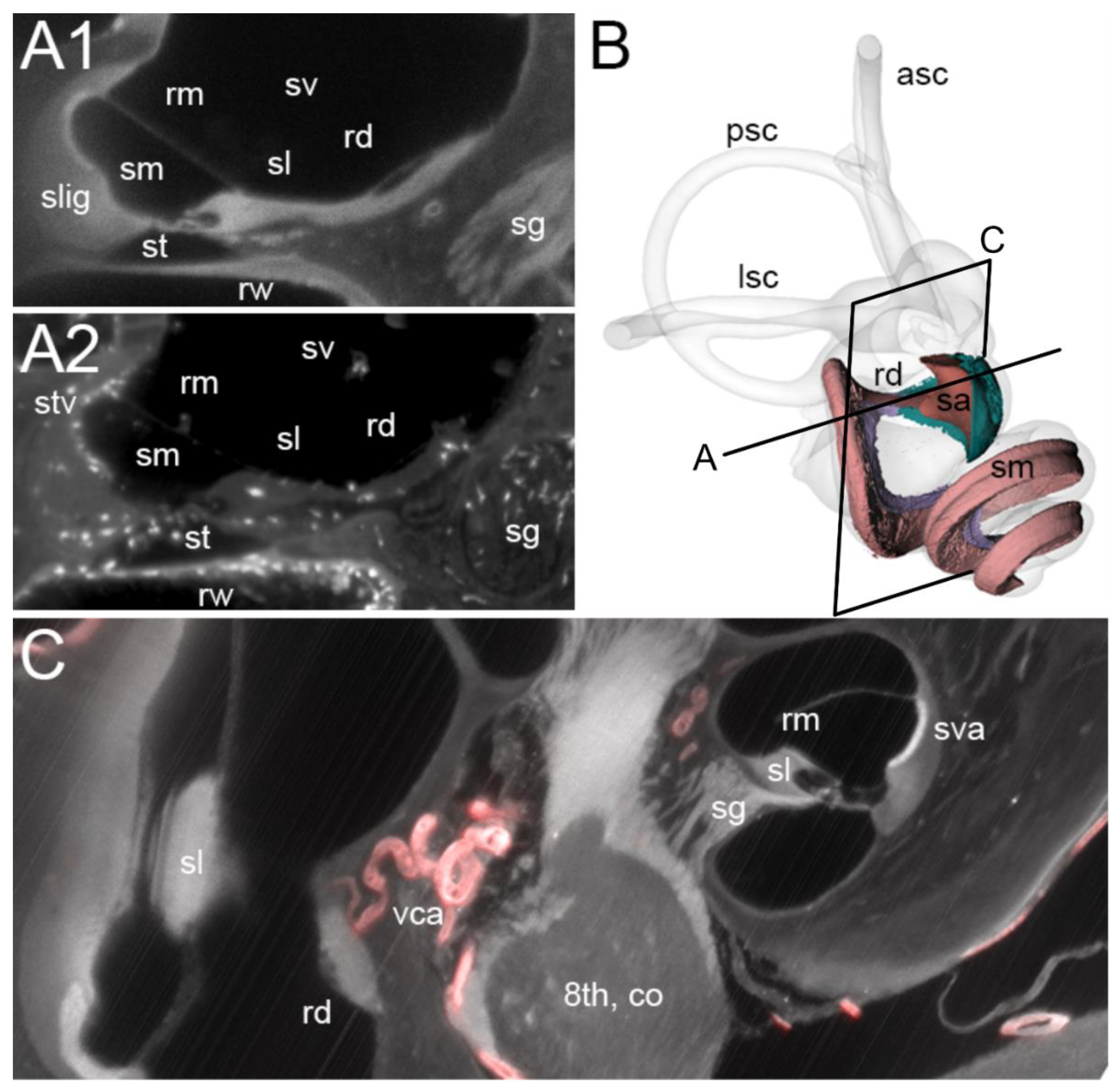
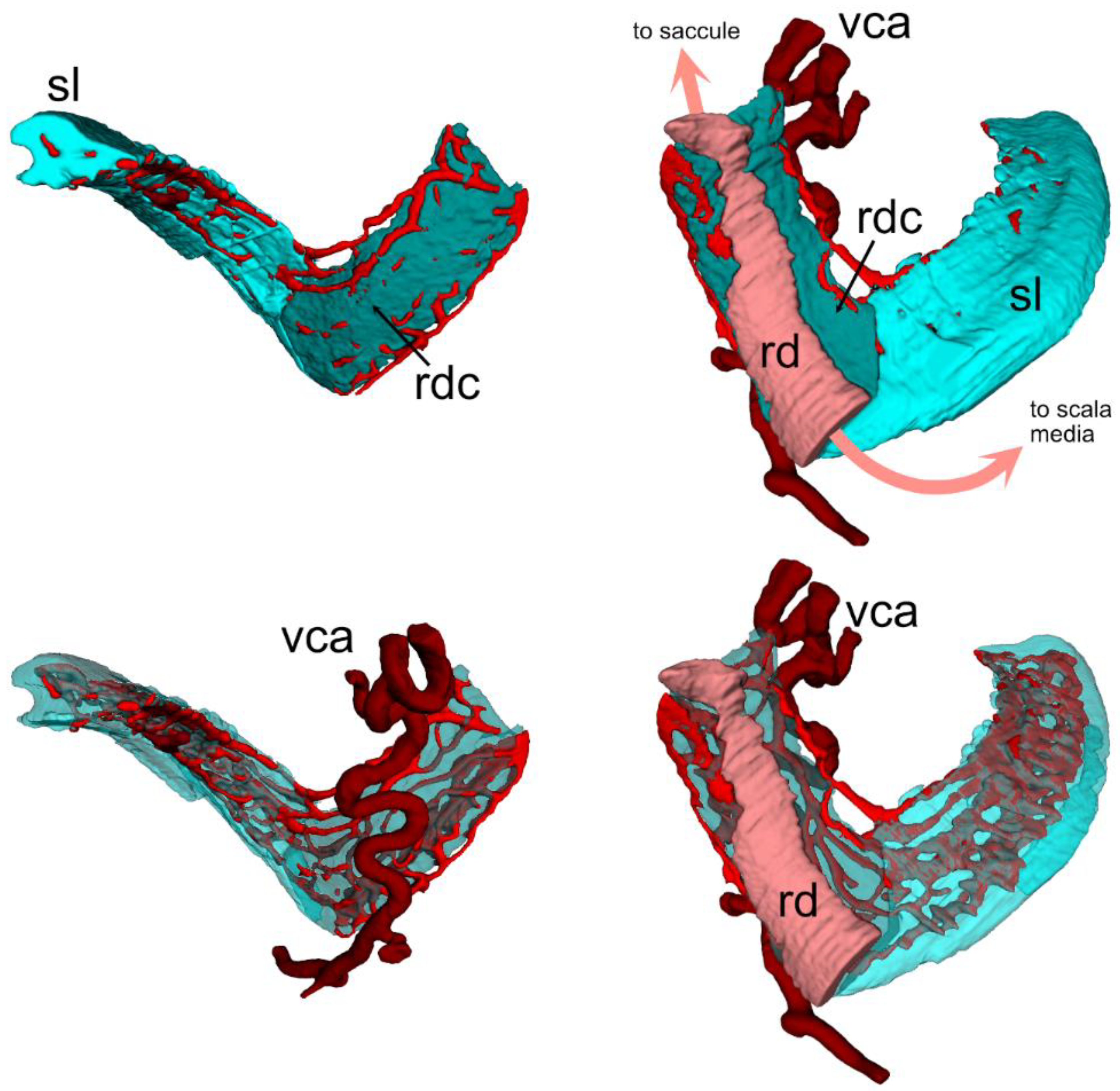
3.1. Reuniting Duct
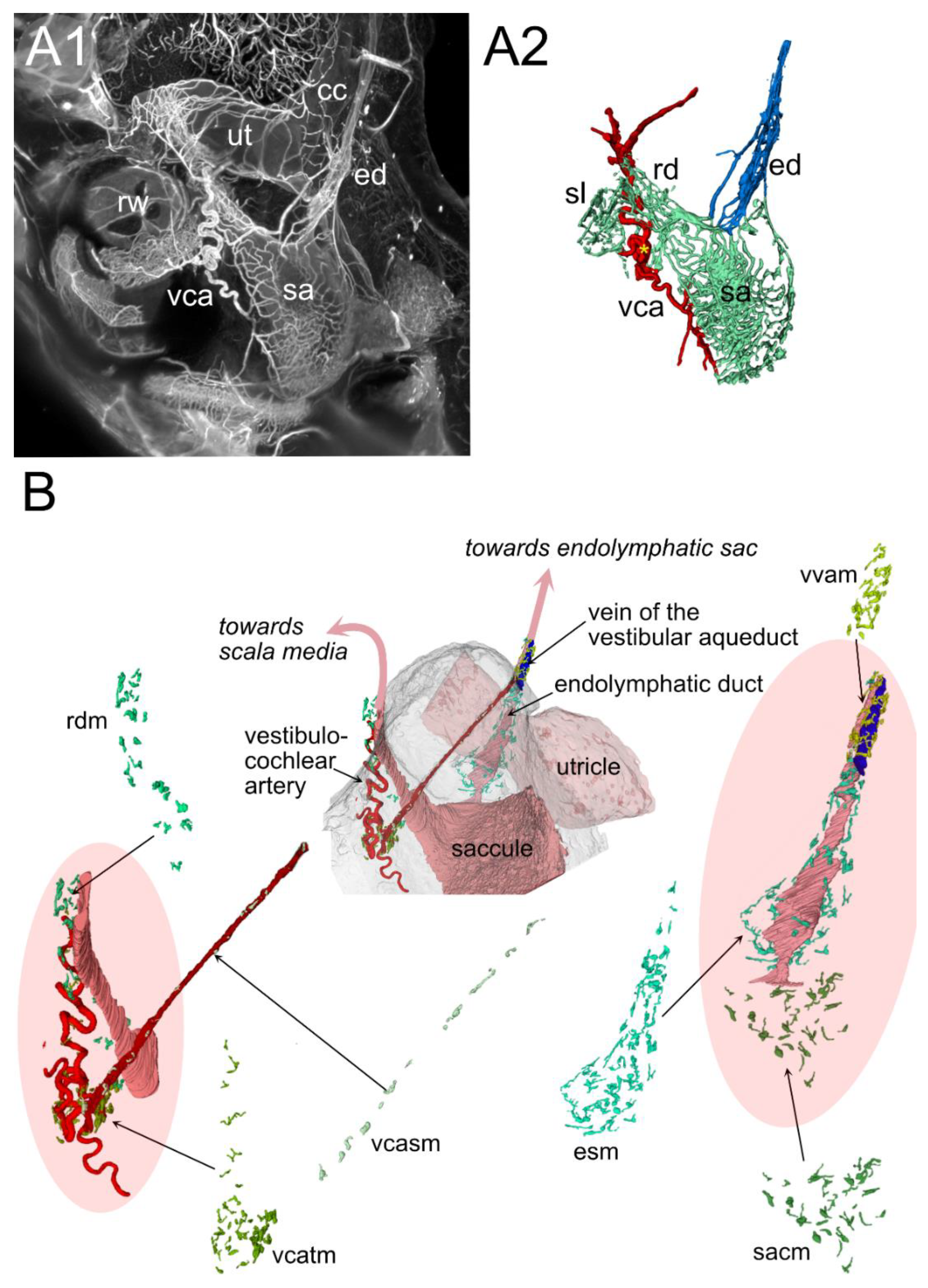
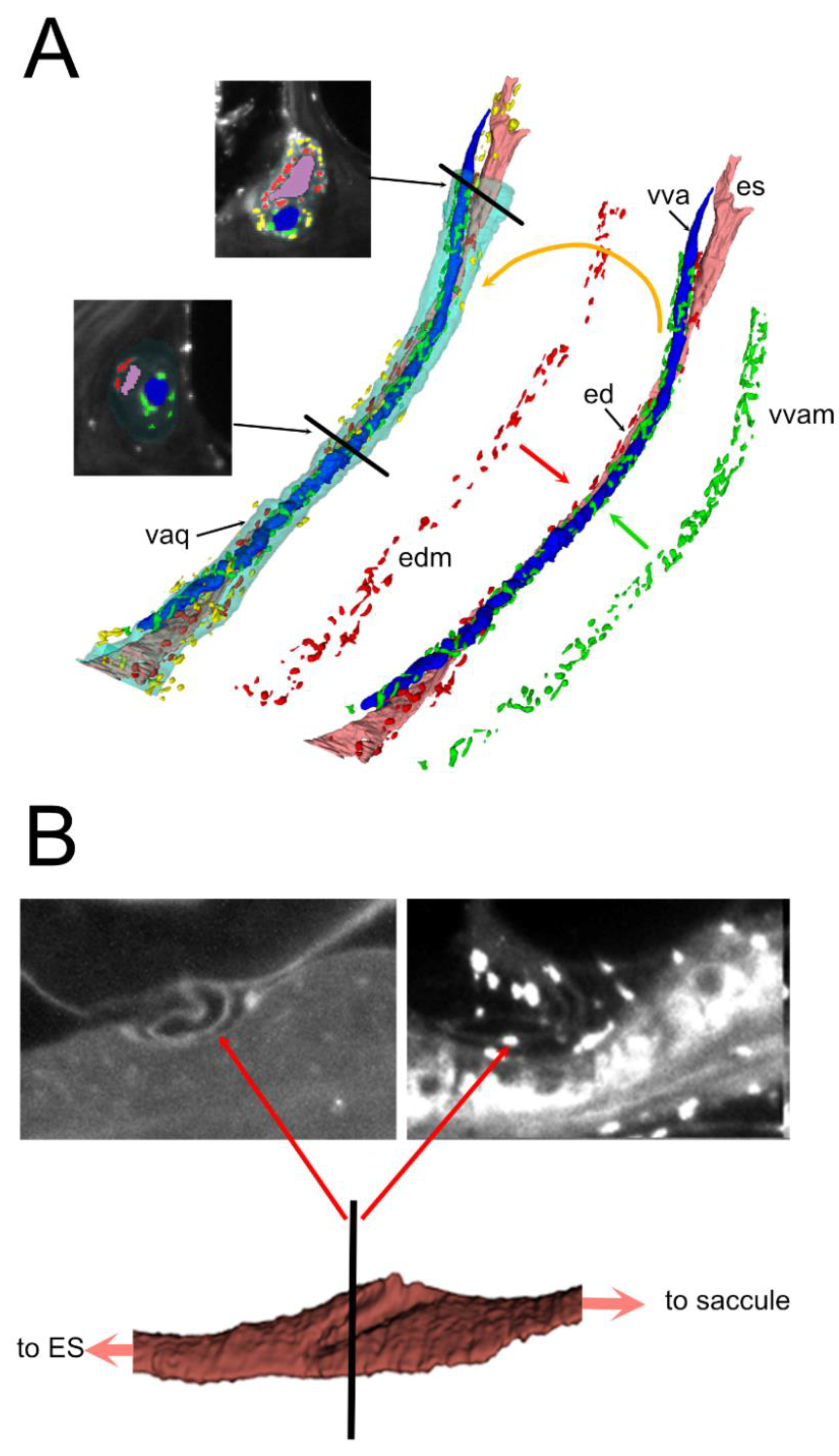
3.2. Endolymphatic Duct
3.3. Macrophage Features
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BPPV | Benign paroxysmal positional vertigo |
| ColIV | Collagen IV |
| ED | Endolymphatic duct |
| MMP14 | Matrix metalloprotease 14 |
| RD | Reuniting duct |
| ROI | Region of interest |
| SMA | Smooth muscle actin |
| VCA | Vestibulocochlear artery |
References
- Hornibrook, J. Saccular otoconia as a cause of Ménière’s disease: hypothesis based on two theories. J Laryngol Otol 2018, 132, 771–774. [Google Scholar] [CrossRef]
- Álvarez De Linera-Alperi M, Dominguez P, Blanco-Pareja M, Menéndez Fernández-Miranda P, Manrique-Huarte R, Liaño G, et al. Is endolymphatic hydrops, as detected in MRI, a truly cochleocentric finding? Front Neurol 2024, 15, 1477282. [CrossRef]
- Yamane H, Sunami K, Iguchi H, Sakamoto H, Imoto T, Rask-Andersen H. Assessment of Meniere’s disease from a radiological aspect - saccular otoconia as a cause of Meniere’s disease? Acta Otolaryngol (Stockh) 2012, 132, 1054–1060. [Google Scholar] [CrossRef]
- Hornibrook J, Bird P. A New Theory for Ménière’s Disease: Detached Saccular Otoconia. Otolaryngol--Head Neck Surg Off J Am Acad Otolaryngol-Head Neck Surg 2017, 156, 350–352. [Google Scholar] [CrossRef]
- Smith CM, Curthoys IS, Plontke SK, Menzel M, Mukherjee P, Wong C, et al. Insights into Inner Ear Function and Disease Through Novel Visualization of the Ductus Reuniens, a Seminal Communication Between Hearing and Balance Mechanisms. J Assoc Res Otolaryngol JARO 2022, 23, 633–645. [Google Scholar] [CrossRef]
- Kimura RS, Schuknecht HF, Ota CY, Jones DD. Obliteration of the ductus reuniens. Acta Otolaryngol (Stockh) 1980, 89, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Konishi, S. The ductus reuniens and utriculo-endolymphatic valve in the presence of endolymphatic hydrops in guinea-pigs. J Laryngol Otol 1977, 91, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Li H, Rajan GP, Shaw J, Rohani SA, Ladak HM, Agrawal S, et al. A Synchrotron and Micro-CT Study of the Human Endolymphatic Duct System: Is Meniere’s Disease Caused by an Acute Endolymph Backflow? Front Surg 2021, 8, 662530. [CrossRef]
- Yamane H, Takayama M, Sunami K, Sakamoto H, Imoto T, Anniko M. Visualization and assessment of saccular duct and endolymphatic sinus. Acta Otolaryngol (Stockh) 2011, 131, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Susaki, EA. Unlocking the potential of large-scale 3D imaging with tissue clearing techniques. Microsc Oxf Engl 2025, 74, 179–188. [Google Scholar] [CrossRef]
- Cossellu D, Vivado E, Batti L, Gantar I, Pizzala R, Perin P. Volumetric atlas of the rat inner ear from microCT and iDISCO+ cleared temporal bones. PeerJ 2025, 13, e19512. [Google Scholar] [CrossRef]
- Frejo L, Lopez-Escamez JA. Cytokines and Inflammation in Meniere Disease. Clin Exp Otorhinolaryngol 2022, 15, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Wilkerson BA, Zebroski HL, Finkbeiner CR, Chitsazan AD, Beach KE, Sen N, et al. Novel cell types and developmental lineages revealed by single-cell RNA-seq analysis of the mouse crista ampullaris. eLife 2021, 10, e60108. [Google Scholar] [CrossRef]
- Jansson B, Rask-Andersen H. Osmotically induced macrophage activity in the endolymphatic sac. On the possible interaction between periaqueductal bone marrow cells and the endolymphatic sac. ORL J Oto-Rhino-Laryngol Its Relat Spec 1992, 54, 191–197. [Google Scholar] [CrossRef]
- Ito T, Kurata N, Fukunaga Y. Tissue-Resident Macrophages in the Stria Vascularis. Front Neurol 2022, 13, 818395. [Google Scholar] [CrossRef]
- Imoto T, Rask-Andersen H, Bagger-Sjöbäck D. The role of the endolymphatic sac in statoconial formation and degradation. Acta Otolaryngol (Stockh) 1983, 96, 227–235. [Google Scholar] [CrossRef]
- Perin P, Cossellu D, Vivado E, Batti L, Gantar I, Voigt FF, et al. Temporal bone marrow of the rat and its connections to the inner ear. Front Neurol 2024, 15, 1386654. [Google Scholar] [CrossRef]
- Renier N, Wu Z, Simon DJ, Yang J, Ariel P, Tessier-Lavigne M. iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 2014, 159, 896–910. [Google Scholar] [CrossRef] [PubMed]
- Perin P, Voigt FF, Bethge P, Helmchen F, Pizzala R. iDISCO+ for the Study of Neuroimmune Architecture of the Rat Auditory Brainstem. Front Neuroanat 2019, 13, 15. [Google Scholar] [CrossRef]
- Barozzi S, Soi D, Intieri E, Giani M, Aldè M, Tonon E, et al. Vestibular and audiological findings in the Alport syndrome. Am J Med Genet A 2020, 182, 2345–2358. [Google Scholar] [CrossRef] [PubMed]
- Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Yushkevich PA, Yang Gao null, Gerig G. ITK-SNAP: An interactive tool for semi-automatic segmentation of multi-modality biomedical images. Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Int Conf 2016, 2016, 3342–3345. [Google Scholar] [CrossRef]
- Schnitzerlein M, Greto E, Wegner A, Möller A, Aust O, Ben Brahim O, et al. Cellular morphodynamics as quantifiers for functional states of resident tissue macrophages in vivo. PLoS Comput Biol 2025, 21, e1011859. [Google Scholar] [CrossRef]
- Gulya, AJ. Anatomy of the Temporal Bone with Surgical Implications. 3rd ed. Boca Raton: CRC Press; 2007. [CrossRef]
- Dahlmann A, von Düring M. The endolymphatic duct and sac of the rat: a histological, ultrastructural, and immunocytochemical investigation. Cell Tissue Res 1995, 282, 277–289. [Google Scholar] [CrossRef]
- Keithley, EM. Inner ear immunity. Hear Res 2022, 419, 108518. [Google Scholar] [CrossRef]
- Perin P, Marino F, Varela-Nieto I, Szczepek AJ. Editorial: Neuroimmunology of the Inner Ear. Front Neurol 2021, 12, 635359. [Google Scholar] [CrossRef] [PubMed]
- Manickam V, Gawande DY, Stothert AR, Clayman AC, Batalkina L, Warchol ME, et al. Macrophages Promote Repair of Inner Hair Cell Ribbon Synapses following Noise-Induced Cochlear Synaptopathy. J Neurosci Off J Soc Neurosci 2023, 43, 2075–2089. [Google Scholar] [CrossRef]
- Pan J, Wang K, Qu J, Chen D, Chen A, You Y, et al. Activated tissue-resident macrophages contribute to hair cell insults in noise-induced hearing loss in mice. Commun Biol 2024, 7, 1078. [Google Scholar] [CrossRef]
- Jansson B, Rask-Andersen H. Erythrocyte removal and blood clearance in the endolymphatic sac. An experimental and TEM study. Acta Otolaryngol (Stockh) 1996, 116, 429–434. [CrossRef]
- Chiot A, Felgner MJ, Brownell D, Rott KH, Bogachuk A, Rosmus D-D, et al. Single-cell, spatial, and fate-mapping analyses uncover niche dependent diversity of cochlear myeloid cells. BioRxiv Prepr Serv Biol 2024. [CrossRef]
- Dieterich M, Hergenroeder T, Boegle R, Gerb J, Kierig E, Stöcklein S, et al. Endolymphatic space is age-dependent. J Neurol 2023, 270, 71–81. [Google Scholar] [CrossRef]
- Tang Y, Zhao C, Zhuang Y, Zhong A, Wang M, Zhang W, et al. Mechanosensitive Piezo1 protein as a novel regulator in macrophages and macrophage-mediated inflammatory diseases. Front Immunol 2023, 14, 1149336. [Google Scholar] [CrossRef]
- Xie Y, Hang L. Mechanical gated ion channel Piezo1: Function, and role in macrophage inflammatory response. Innate Immun 2024, 30, 32–39. [Google Scholar] [CrossRef]
- Mei X, Schart-Morén N, Li H, Ladak HM, Agrawal S, Behr R, et al. Three-dimensional imaging of the human internal acoustic canal and arachnoid cistern: a synchrotron study with clinical implications. J Anat 2019, 234, 316–326. [Google Scholar] [CrossRef]
- Webster KE, George B, Galbraith K, Harrington-Benton NA, Judd O, Kaski D, et al. Positive pressure therapy for Ménière’s disease. Cochrane Database Syst Rev 2023, 2, CD015248. [Google Scholar] [CrossRef]
- Lundberg YW, Zhao X, Yamoah EN. Assembly of the otoconia complex to the macular sensory epithelium of the vestibule. Brain Res 2006, 1091, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Kao WTK, Parnes LS, Chole RA. Otoconia and otolithic membrane fragments within the posterior semicircular canal in benign paroxysmal positional vertigo. The Laryngoscope 2017, 127, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Lim, DJ. Formation and fate of the otoconia. Scanning and transmission electron microscopy. Ann Otol Rhinol Laryngol 1973, 82, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Dickman JD, Huss D, Lowe M. Morphometry of otoconia in the utricle and saccule of developing Japanese quail. Hear Res 2004, 188, 89–103. [Google Scholar] [CrossRef]
- Lins U, Farina M, Kurc M, Riordan G, Thalmann R, Thalmann I, et al. The otoconia of the guinea pig utricle: internal structure, surface exposure, and interactions with the filament matrix. J Struct Biol 2000, 131, 67–78. [Google Scholar] [CrossRef]
- Quick ME, Bird PA. A review of Meniere’s disease - reflection of published MRI, ECochG and newer otoconial theory from Aotearoa New Zealand. J R Soc N Z 2025, 55, 441–450. [Google Scholar] [CrossRef]
- Yamane H, Takayama M, Sunami K, Sakamoto H, Imoto T, Anniko M. Blockage of reuniting duct in Meniere’s disease. Acta Otolaryngol (Stockh) 2010, 130, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Zucca G, Valli S, Valli P, Perin P, Mira E. Why do benign paroxysmal positional vertigo episodes recover spontaneously? J Vestib Res Equilib Orientat 1998, 8, 325–329. [Google Scholar] [CrossRef]
- Lund H, Hunt MA, Kurtović Z, Sandor K, Kägy PB, Fereydouni N, et al. CD163+ macrophages monitor enhanced permeability at the blood-dorsal root ganglion barrier. J Exp Med 2024, 221, e20230675. [Google Scholar] [CrossRef] [PubMed]
| Sample | Age(Days) | Sex | Signal | Voxel Size (µm) |
| R1a,b | 111 | F | Auto, Iba-1 | 2 |
| R2 | 95 | F | Auto, Iba-1 | 2 |
| R3 | 494 | M | Auto, SMA | 3.26 |
| R4 | 79 | M | ColIV | 4.08 |
| Region | Macrophage Number |
| All | 311 |
| Endolymphatic sinus | 56 |
| Endolymphatic duct | 54 |
| Reuniting duct | 18 |
| Vestibulocochlear artery, tortuous | 33 |
| Vestibulocochlear artery, straight | 15 |
| Vestibular aqueduct vein | 68 |
| Vestibular aqueduct | 67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




