Submitted:
07 August 2025
Posted:
08 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
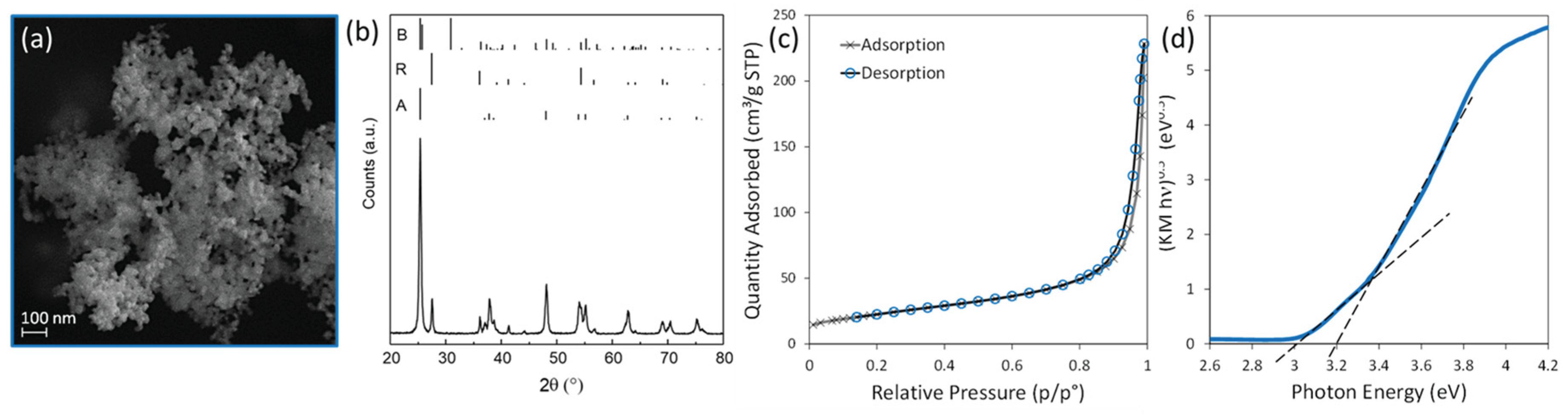
2.1. TiO2 Powder Characterization
2.2. Preparation of TiO2 Suspensions
2.3. Cells and virus
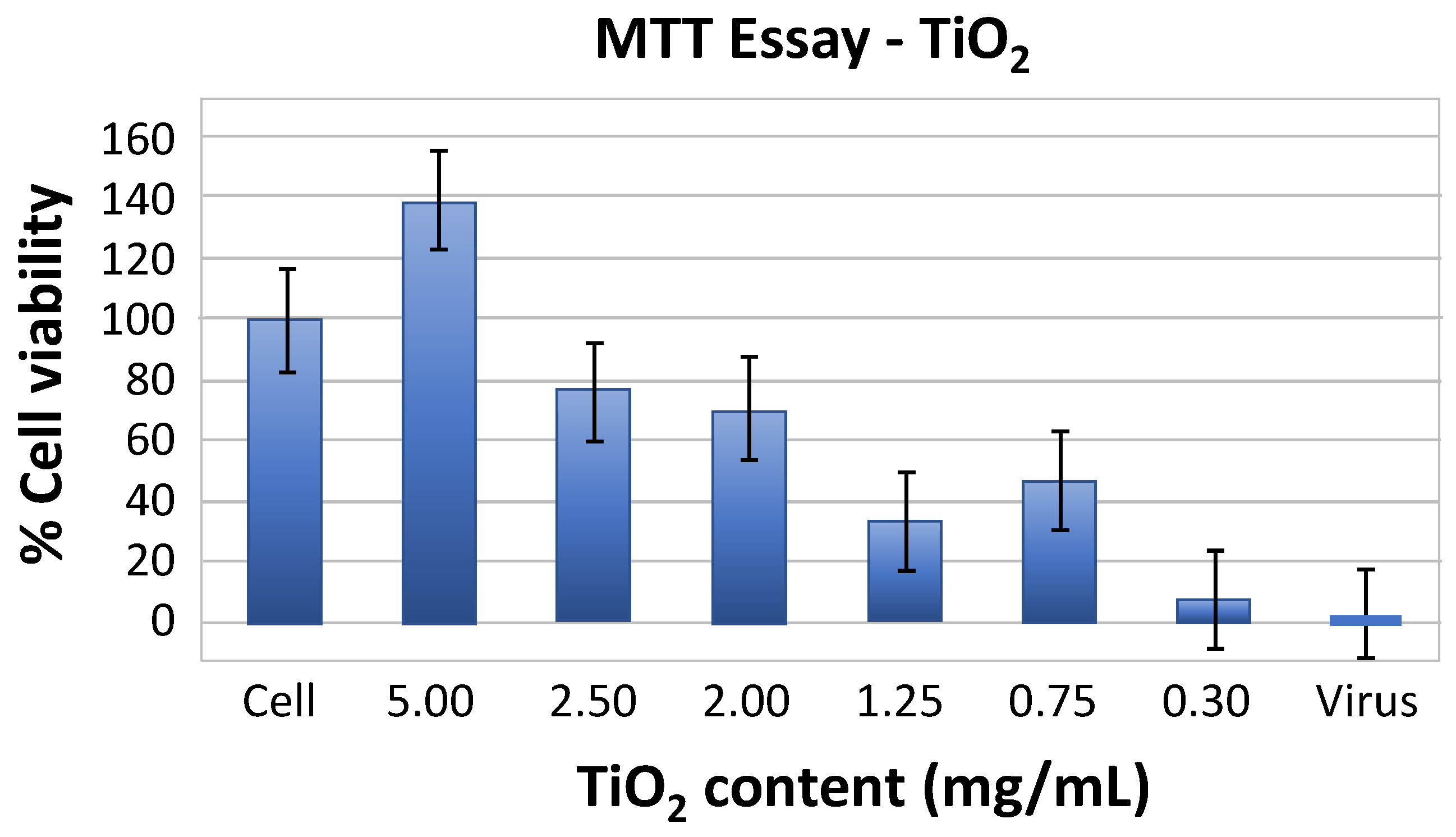
2.4. Investigation of TiO2 Cytotoxic Properties on the E-11 Cell Line
2.5. TiO2 Inhibitory Properties on Nodaviruses Replication
2.6. RNA Extraction and Real-Time qPCR Analysis
3. Results
3.1. TiO2 Powder Characterization
3.2. Investigation of TiO2 Cytotoxic Properties on the E-11 Cell Line
3.3. In Vitro Investigation of the Inhibitory Properties of TiO2 on Nodavirus Replication
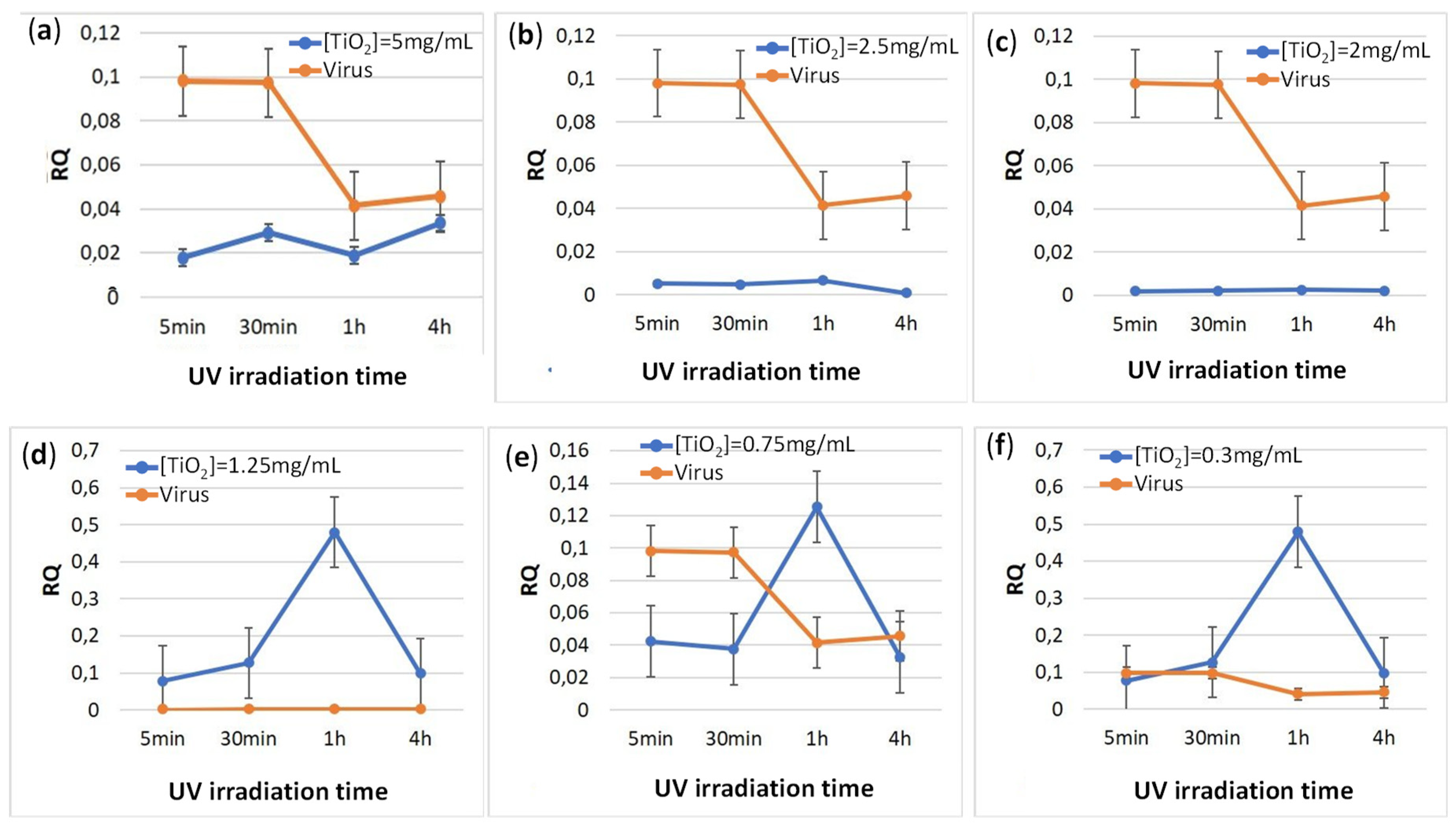
3.4. Relative Expression of the Viral Capsid Gene (CP) in E-11 Cells
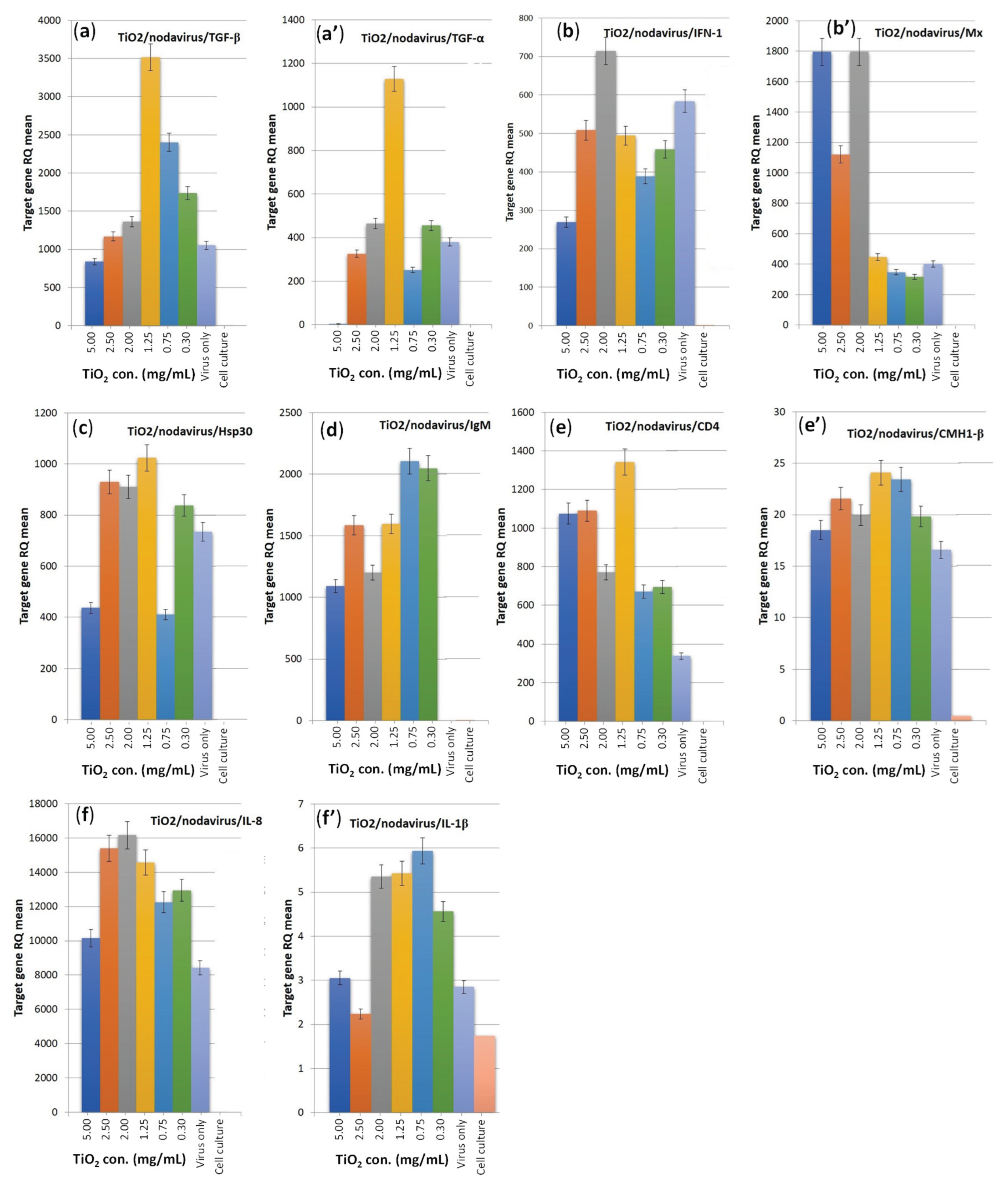
3.5. Immune Genes Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Ethical Statements
Conflicts of Interest
References
- F. Pascoli et al., “Evaluation of oxidative stress biomarkers in Zosterisessor ophiocephalus from the Venice Lagoon, Italy,” Aquatic Toxicology, vol. 101, no. 3–4, pp. 512–520, 2011. [CrossRef]
- A. L. Ahmad, J. Y. Chin, M. H. Z. Mohd Harun, and S. C. Low, “Environmental impacts and imperative technologies towards sustainable treatment of aquaculture wastewater: A review,” Journal of Water Process Engineering, vol. 46, p. 102553, Apr. 2022. [CrossRef]
- G. Ren et al., “Recent Advances of Photocatalytic Application in Water Treatment: A Review,” Nanomaterials, vol. 11, no. 7, p. 1804, Jul. 2021. [CrossRef]
- V. Zorzi, A. Bertini, A. Robertson, A. Berardinelli, L. Palmisano, and F. Parrino, “The application of advanced oxidation processes including photocatalysis-based ones for the off-flavours removal (GSM and MIB) in recirculating aquaculture systems,” Molecular Catalysis, vol. 551, p. 113616, Dec. 2023. [CrossRef]
- S. Livolsi et al., “Innovative photoelectrocatalytic water remediation system for ammonia abatement,” Catal Today, vol. 413–415, p. 113996, Mar. 2023. [CrossRef]
- E. Buoio et al., “From Photocatalysis to Photo-Electrocatalysis: An Innovative Water Remediation System for Sustainable Fish Farming,” Sustainability, vol. 14, no. 15, p. 9067, Jul. 2022. [CrossRef]
- B. Randazzo et al., “A novel photocatalytic purification system for fish culture,” Zebrafish, vol. 14, no. 5, 2017. [CrossRef]
- M. Altomare, G. L. Chiarello, A. Costa, M. Guarino, and E. Selli, “Photocatalytic abatement of ammonia in nitrogen-containing effluents,” Chemical Engineering Journal, vol. 191, pp. 394–401, 2012. [CrossRef]
- G. L. Chiarello, A. Zuliani, D. Ceresoli, R. Martinazzo, and E. Selli, “Exploiting the Photonic Crystal Properties of TiO2 Nanotube Arrays to Enhance Photocatalytic Hydrogen Production,” ACS Catalysis, vol. 6, no. 2, pp. 1345–1353, 2016. [CrossRef]
- G. L. Chiarello, C. Tealdi, P. Mustarelli, and E. Selli, “Fabrication of Pt/Ti/TiO2 photoelectrodes by RF-Magnetron sputtering for separate hydrogen and oxygen production,” Materials, vol. 9, no. 4, 2016. [CrossRef]
- S. M. Murgia, A. Poletti, and R. Selvaggi, “Photocatalytic degradation of high ammonia concentration water solutions by TiO2,” Annali di Chimica, vol. 95, no. 5, pp. 335–343, 2005. [CrossRef]
- Y. Wu et al., “Preparation of photocatalytic anatase nanowire films by in situ oxidation of titanium plate,” Nanotechnology, vol. 20, no. 18, 2009. [CrossRef]
- N. Moreira et al., “Fast mineralization and detoxification of amoxicillin and diclofenac by photocatalytic ozonation and application to an urban wastewater,” Water research, vol. 87, pp. 87–96, 2015.
- T. Do, D. Nguyen, K. Nguyen, P. L.- Materials, and U. 2019, “TiO2 and Au-TiO2 Nanomaterials for Rapid Photocatalytic Degradation of Antibiotic Residues in Aquaculture Wastewater,” mdpi.com, vol. 12, no. 15, 2019.
- R. C. Gilson, K. C. L. Black, D. D. Lane, and S. Achilefu, “Hybrid TiO2 -Ruthenium Nano-photosensitizer Synergistically Produces Reactive Oxygen Species in both Hypoxic and Normoxic Conditions ,” Angewandte Chemie, vol. 129, no. 36, pp. 10857–10860, 2017. [CrossRef]
- P. Hajkova, P. Spatenka, Jan Horsky, I. Horska, A. Kolouch. “Photocatalytic Effect of TiO2 Films on Viruses and Bacteria,” Plasma Processes and Polymers, 4(S1), S397–S401, 2007. [CrossRef]
- N. A. Mazurkova, Y. E. Spitsyna, N. V. Shikina, Z. R. Ismagilov, S. N. Zagrebel’nyi, and E. I. Ryabchikova, “Interaction of titanium dioxide nanoparticles with influenza virus,” Nanotechnologies in Russia, vol. 5, no. 5–6, pp. 417–420, 2010. [CrossRef]
- P. Hajkova, P. Spatenka, J. Horsky, I. Horska, and A. Kolouch, “Photocatalytic effect of TiO2 films on viruses and bacteria,” Plasma Processes and Polymers, vol. 4, no. SUPPL.1, 2007. [CrossRef]
- N. Bono, F. Ponti, C. Punta, and G. Candiani, “Effect of UV Irradiation and TiO2-Photocatalysis on Airborne Bacteria and Viruses: An Overview,” Materials, vol. 14, no. 1075, 2021. [CrossRef]
- M. M. A. Hussein et al., “Amelioration of titanium dioxide nanoparticle reprotoxicity by the antioxidants morin and rutin,” Environmental Science and Pollution Research, vol. 26, no. 28, pp. 29074–29084, 2019. [CrossRef]
- H. Cheng et al., “Adverse reproductive performance in zebrafish with increased bioconcentration of microcystin-LR in the presence of titanium dioxide nanoparticles,” Environmental Science: Nano, no. 5, pp. 1208–1217, 2018.
- K. Khosravi-Katuli, E. Prato, G. Lofrano, M. Guida, G. Vale, and G. Libralato, “Effects of nanoparticles in species of aquaculture interest,” Environmental Science and Pollution Research, vol. 24, no. 21, pp. 17326–17346, 2017. [CrossRef]
- L. Lu, R. Sun, R. Chen, C. Hui, C.M. Ho, J.M. Luk, G.K. Lau, C.M. Che, “Silver nanoparticles inhibit hepatitis B virus replication,” Antivir Ther. 2008;13(2):253-62. [CrossRef]
- P. Orłowski, A. Kowalczyk, et al., “Antiviral activity of tannic acid modified silver nanoparticles: potential to activate immune response in herpes genitalis,” Viruses 2018, 10(10), 524. [CrossRef]
- J. L. Elechiguerra et al., “Interaction of silver nanoparticles with HIV-1,” Journal of Nanobiotechnology, vol. 3, 6, 2005. [CrossRef]
- R. W. Y. Sun, R. Chen, N. P. Y. Chung, C. M. Ho, C. L. S. Lin, and C. M. Che, “Silver nanoparticles fabricated in Hepes buffer exhibit cytoprotective activities toward HIV-1 infected cells,” Chemical Communications, no. 40, pp. 5059–5061, 2005. [CrossRef]
- K. Kandasamy, N. M. Alikunhi, G. Manickaswami, A. Nabikhan, and G. Ayyavu, “Synthesis of silver nanoparticles by coastal plant Prosopis chilensis (L.) and their efficacy in controlling vibriosis in shrimp Penaeus monodon,” Applied Nanoscience (Switzerland), vol. 3, no. 1, pp. 65–73, 2013. [CrossRef]
- A. R. Ochoa-Meza et al., “Silver nanoparticles enhance survival of white spot syndrome virus infected Penaeus vannamei shrimps by activation of its immunological system,” Fish and Shellfish Immunology, vol. 84, pp. 1083–1089, 2019. [CrossRef]
- E. Sivaramasamy and W. Zhiwei, “Enhancement of Vibriosis Resistance in Litopenaeus vannamei by Supplementation of Biomastered Silver Nanoparticles by Bacillus subtilis,” Journal of Nanomedicine & Nanotechnology, vol. 07, no. 01, 2016. [CrossRef]
- J. C. M. Márquez, A. H. Partida, M. del Carmen, M. Dosta, J. C. Mejía, and J. A. B. Martínez, “Silver nanoparticles applications (AgNPS) in aquaculture,” International Journal of Fisheries and Aquatic Studies, vol. 6, no. 2, pp. 5–11, 2018.
- V. Panzarin , A. Toffan , E. Vetrini et al. Development of a universal real-time RT-PCR assay targeting RNA2 of betanodaviruses, capable of detecting all genotypes of fish nodavirus. Journal of Virological Methods, 167(2), 110–118, 2010.
- G.K Purohit, S.K Nayak, B.K. Mishra . Evaluation of β-actin gene as a reference for RT-qPCR analysis in Puntius sophore under thermal stress. BMC Genomics, 18:617, 2017.
- G. Scapigliati, F. Buonocore , G. Marino. Functional aspects of fish lymphocytes: markers, cytokines and gene expression. Fish & Shellfish Immunology, 29(2), 229–236, 2010.
- L. Poisa-Beiro, Q.K. Doan, M.C. Piazzon et al. Virus–host interaction in European sea bass: induction of Mx, IL-1β and TNF-α expression by betanodavirus. Fish & Shellfish Immunology, 25(3), 462–470, 2008.
- M.P. Sepulcre , V. Mulero , J.V. Planas . Interleukin-8 gene in sea bass: molecular cloning and expression pattern in response to infection. Developmental & Comparative Immunology, 31(1), 413–425, 2007.
- T. Angsujinda , S. Auewarakul , S. Pongsiri. Tumor necrosis factor-α and heat shock protein expression in fish under stress conditions. Fish & Shellfish Immunology, 103, 229–236, 2020.
- A. Santos, M.E. Marrero, M. Sánchez et al. Characterization of IgM heavy chain gene expression during fish development. Developmental & Comparative Immunology, 25(7), 549–556, 2001.
- S; Picchietti , G. Scapigliati , E. Randelli et al. Molecular cloning and expression of MHC class I-β gene in European sea bass. Developmental & Comparative Immunology, 48(2), 234–242, 2015.
- S. Rafiei, S. E. Rezatofighi, M. R. Ardakani, and S. Rastegarzadeh, “Gold Nanoparticles Impair Foot-and-Mouth Disease Virus Replication,” IEEE Transactions on Nanobioscience, vol. 15, no. 1, pp. 34–40, 2016. [CrossRef]
- M. Tello-Olea et al., “Gold nanoparticles (AuNP) exert immunostimulatory and protective effects in shrimp (Litopenaeus vannamei) against Vibrio parahaemolyticus,” Fish and Shellfish Immunology, vol. 84, pp. 756–767, 2019. [CrossRef]
- X. Sang et al., “Photocatalytic inactivation of diarrheal viruses by visible-light-catalytic titanium dioxide,” Clinical Laboratory, vol. 53, no. 7–8, pp. 413–421, 2007.
- Kibenge frederick SB, “Emerging viruses in aquaculture,” Current Opinion in Virology, vol. 34, pp. 97–103, 2019.
- P. J. Walker and C. V. Mohan, “Viral disease emergence in shrimp aquaculture: origins, impact and the effectiveness of health management strategies,” Reviews in Aquaculture, vol. 1, no. 2, pp. 125–154, Jun. 2009. [CrossRef]
- H. D. Rodger, “Fish disease causing economic impact in global aquaculture” Birkhauser Advances in Infectious Diseases, no. 9783034809788, pp. 1–34, 2016. [CrossRef]
- N. Cherif, H. Attia El Hili, N. Mzoughi, L. Chouba, M. El Bour, D. El-Amri, A. Hamza, K. Maatoug, S. Zaafrane, S. Hammami, (2011): “Tunisian aquaculture: present situation and potentialities” in: Gavin L. Andrews and Lauren A. Vexton (Eds), ”Fish Farms: Management, Disease Control and the Environment”. Nova Science Publishers N.Y., USA. Editors:, Chapter 4, pp.113-132. ISBN: 978-1-61209-538-7.
- A. Altomare et al., “Applied Crystallography Quanto: a Rietveld program for quantitative phase analysis of polycrystalline mixtures,” J. Appl. Cryst, vol. 34, 392-397, 2001. [CrossRef]
- T. Iwamoto, T. Nakai, K. Mori, M. Arimoto, and I. Furusawa, “Cloning of the fish cell line SSN-1 for piscine nodaviruses,” vol. 43, no. 1997, pp. 81–89, 2000.
- C. A. Suttle, “Marine viruses - Major players in the global ecosystem,” Nature Reviews Microbiology, vol. 5, no. 10, pp. 801–812, 2007. [CrossRef]
- H. Su and J. Su, “Cyprinid viral diseases and vaccine development,” Fish and Shellfish Immunology, vol. 83, pp. 84–95, 2018. [CrossRef]
- Rahaman, M.M.; Sharma, B.; Talukder, S.; Uddin, M.J.; Siddik, M.A.B.; Sarker, S. Viral Threats to Australian Fish and Prawns: Economic Impacts and Biosecurity Solutions—A Systematic Review. Viruses 2025, 17, 692. [CrossRef]
- S. Wang, H. Yu, and J. K. Wickliffe, “Limitation of the MTT and XTT assays for measuring cell viability due to superoxide formation induced by nano-scale TiO2,” Toxicology in Vitro, vol. 25, no. 8, pp. 2147–2151, Dec. 2011. [CrossRef]
- L. Canesi, C. Ciacci, R. Fabbri, A. Marcomini, G. Pojana, and G. Gallo, “Bivalve molluscs as a unique target group for nanoparticle toxicity,” Marine Environmental Research, vol. 76, pp. 16–21, 2012.
- C. Ciacci et al., “Immunomodulation by different types of N-oxides in the hemocytes of the marine bivalve Mytilus galloprovincialis,” PLoS ONE, vol. 7, no. 5, May 2012. [CrossRef]
- C. Barmo, C. Ciacci, B. Canonico, R. Fabbri, K. C.-A. Toxicology, and U. 2013, “In vivo effects of n-TiO2 on digestive gland and immune function of the marine bivalve Mytilus galloprovincialis,” Elsevier, 20AD.
- B. N. Mueller NC, “Exposure modeling of engineered nanoparticles in the environment,” ACS Publications, vol. 42, no. 12, pp. 4447–4453, Jun. 2008. [CrossRef]
- S. Pérez and D. B. M la Farré, “Analysis, behavior and ecotoxicity of carbon-based nanomaterials in the aquatic environment,” Elsevier, 1990.

| Gene | Forward | Reverse | Reference |
|---|---|---|---|
| Nodavirus | CAACTGACARCGAHCACAC | CCCACCAYTTGGCVAC | [31] |
| β-actine | GCC TTC CTT CCT TGG TAT GG | GTG TTG GCG TAC AGG TCC TT | [32] |
| IFN-I | GGCTCTACTGGATACGATGG | CT GCGTCCAAAGCATCAGCT | [33] |
| Mx | ATTCTGAGTTCTTGCTGAAGG | CCTCTAGAACTCCACCAGG | [34] |
| IL-1β | CAGGACTCCGGTTTGAACAT | GTCCATTCAAAAGGGGACAA | [34] |
| IL-8 | GTGCTCCTGGCGTTC | CTTCACCCAGGGAGC | [35] |
| TNF-α | AGA CAA GGT GGA GTG GAA GA | CCT GGC TGT AGA CGA AGT AGA | [36] |
| TGF-β | GACCTGGGATGGAAGTGG | CAGCTGCTCCACCTTGTG | [33] |
| IgM | GAGCTGCAGAAGGACAGTG | TCAGACTGGCCTCACAGCT | [37] |
| CD4 | GTGATAACGCTGAAGATCGAGCC | GAGGTGTGTCATCTTCCGTTG | [33] |
| CMH1-β | CAGAGACGGACAGGAAG | CAAGATCAGACCCAGGA | [38] |
| Hsp30 | CAG GTG GGC AGG AAG CTG | ACC CCT TCA GGC AGA TCA AAC TC | [36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




