Submitted:
04 August 2025
Posted:
05 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Basics of Terpene Biosynthesis in Medicinal Plants: Pathways and Regulation
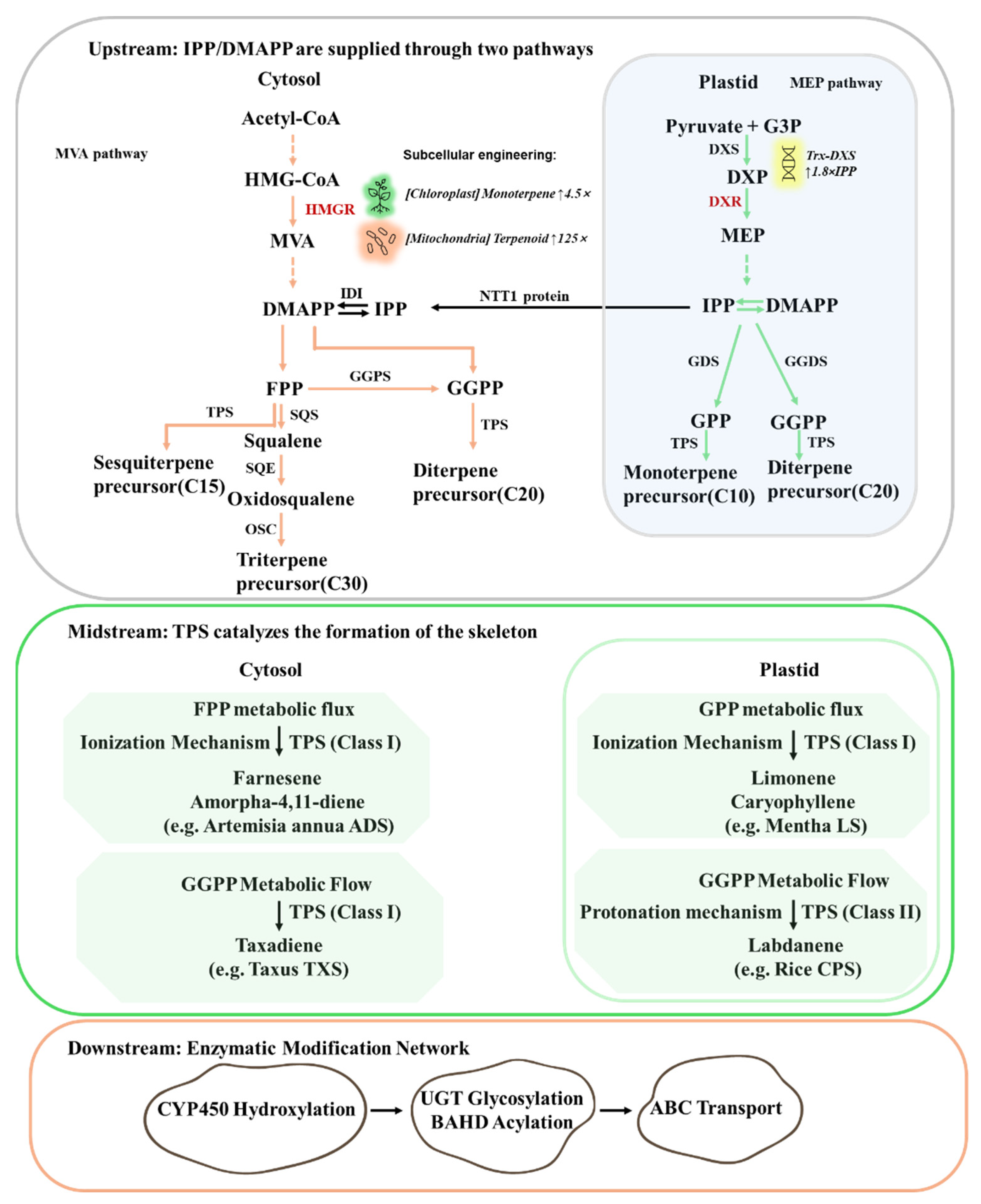
2.1. Core Pathways: From Precursors to Structural Diversification
2.2. Medicinal Plant-Specific Adaptations
2.3. Multilayer Regulatory Networks
2.4. Emerging Insights and Challenges
3. Genomics and Multi-Omics: Unveiling the Blueprint and Targets
3.1. Genome Sequencing: Foundation for Gene Discovery
3.2. Transcriptomics: Spatial-Temporal Dynamics of Terpenoid Pathways
3.3. Metabolomics: Bridging Genotype to Chemotype
3.4. Proteomics and Post-Translational Regulation
3.5. Epigenomics: Chromatin-Level Control
3.6. Integrative Multi-Omics: From Description to Prediction
4. Metabolic Engineering Strategies: From Targets to Phenotypes
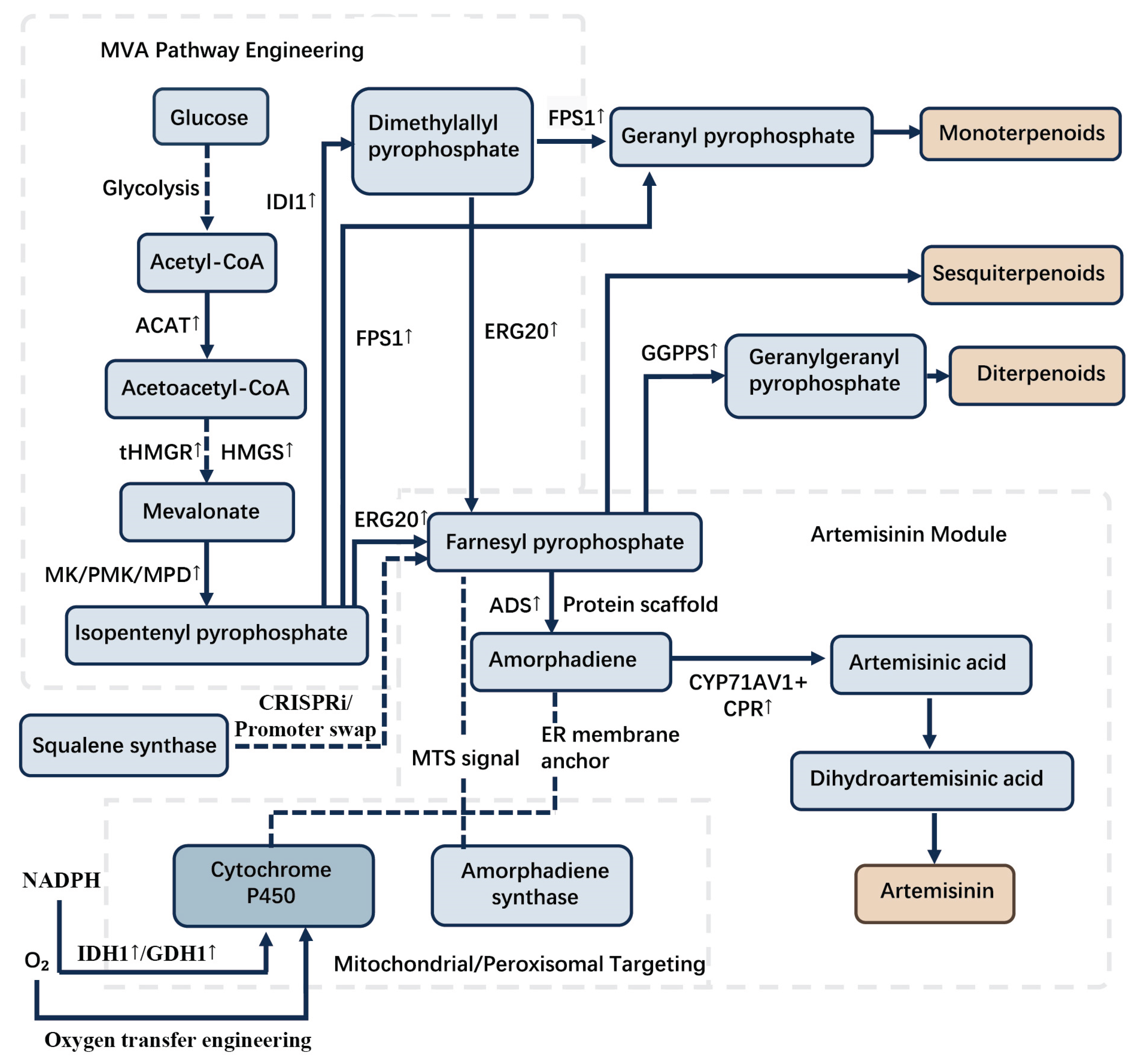
4.1. Rate-Limiting Enzyme Overexpression and Pathway Enhancement
4.2. Precise Suppression of Competing Pathways
4.3. Transcriptional Factor (TF) Hierarchical Regulation
4.4. Heterologous Pathway Reconstruction and Enzyme Engineering
4.5. Directed Subcellular Metabolic Channeling
4.6. Cofactor Balancing and Dynamic Regulation
4.7. Synthetic Biology Tool Advancements
5. Biotechnological Applications: From Laboratory to Potential Industrialization
5.1. High-Yielding Medicinal Plant/Cell Line Cultivation
5.2. Production of Rare or Complex Terpenoids
5.3. Biosynthesis of Novel Terpenoid Derivatives
5.4. Enhanced Plant Stress Resistance
5.5. Plant Cell/Tissue Culture & Scale-Up Challenges
5.6. Plant Systems as Green Cell Factories
6. Current Challenges and Bottlenecks
6.1. Complex Pathways and Incompletely Understood Regulation
6.2. Limitations in Genetic Transformation and Regeneration
6.3. Metabolic Imbalance, Growth Penalties, and Toxicity
6.4. Compartmentalization and Transport Barriers
6.5. Suboptimal Enzyme Properties
6.6. Lack of Universal Chassis Plants
6.7. Scale-Up Challenges and Economic Viability
6.8. Regulatory and Societal Hurdles
7. Prospects and Frontier Directions
7.1. Deep Integration of Multi-Omics and Systems Biology for Predictive Modeling
7.2. Innovation in Gene Editing Technologies
7.3. Synthetic Biology and Modular Design
7.4. Enzyme Engineering and Directed Evolution
7.5. Organelle Engineering
7.6. Development of Efficient Universal Chassis
7.7. Cell-Free Synthetic Biology Systems
7.8. Integration of Artificial Intelligence and Machine Learning
7.9. Focus on Non-Model Medicinal Plants
7.10. End-to-End Integration and Collaborative Innovation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| Abbreviation | Full Name |
| ABC | ATP-Binding Cassette |
| ADS | Amorpha-4,11-Diene Synthase |
| AI | Artificial Intelligence |
| ALDH1 | Aldehyde Dehydrogenase 1 |
| ATAC-seq | Assay for Transposase-Accessible Chromatin with high-throughput sequencing |
| ATP | Adenosine Triphosphate |
| BE | Base Editing |
| ChIP-seq | Chromatin Immunoprecipitation sequencing |
| COX-2 | Cyclooxygenase-2 |
| CPR | Cytochrome P450 Reductase |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| CRISPR-Cas9 | CRISPR-associated protein 9 |
| CRISPRi | CRISPR interference |
| CYP | Cytochrome P450 |
| DMAPP | Dimethylallyl Diphosphate |
| DXS | 1-Deoxy-D-Xylulose-5-Phosphate Synthase |
| EFSA | European Food Safety Authority |
| ER | Endoplasmic Reticulum |
| FAD3 | Fatty Acid Desaturase 3 |
| FPP | Farnesyl Diphosphate |
| G6PDH | Glucose-6-Phosphate Dehydrogenase |
| GA3 | Gibberellin A3 |
| GC-MS | Gas Chromatography-Mass Spectrometry |
| GEMs | Genome-scale Metabolic Models |
| GGPP | Geranylgeranyl Diphosphate |
| GMO | Genetically Modified Organism |
| GPP | Geranyl Diphosphate |
| GRNs | Gene Regulatory Networks |
| HMG-CoA | 3-Hydroxy-3-Methylglutaryl-CoA |
| HMGR | 3-Hydroxy-3-Methylglutaryl-CoA Reductase |
| HRMS | High-Resolution Mass Spectrometry |
| IDI | Isopentenyl Diphosphate Isomerase |
| IMS | Ion Mobility Spectrometry |
| IPK | Isopentenyl Phosphate Kinase |
| IPP | Isopentenyl Diphosphate |
| iNOS | Inducible Nitric Oxide Synthase |
| IUP | Isopentenol Utilization Pathway |
| JA | Jasmonate |
| JAZ | Jasmonate ZIM-domain |
| LC-MS/MS | Liquid Chromatography-Tandem Mass Spectrometry |
| MAFF | Ministry of Agriculture, Forestry and Fisheries (Japan) |
| MD | Molecular Dynamics |
| MeJA | Methyl Jasmonate |
| MEP | Methylerythritol Phosphate pathway |
| ML | Machine Learning |
| MVA | Mevalonate pathway |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate (reduced form) |
| NF-κB | Nuclear Factor kappa B |
| NMR | Nuclear Magnetic Resonance |
| oxPPP | Oxidative Pentose Phosphate Pathway |
| PAL | Phenylalanine Ammonia-Lyase |
| PDH | Pyruvate Dehydrogenase |
| PE | Prime Editing |
| PEPC | Phosphoenolpyruvate Carboxylase |
| POR | Protochlorophyllide Oxidoreductase |
| PSY | Phytoene Synthase |
| PTMs | Post-Translational Modifications |
| RNAi | RNA Interference |
| RNA-seq | RNA Sequencing |
| RNP | Ribonucleoprotein |
| ROS | Reactive Oxygen Species |
| SA | Salicylic Acid |
| scRNA-seq | Single-cell RNA Sequencing |
| SDN-1 | Site-Directed Nuclease 1 |
| SEM | Structural Equation Modeling |
| SQS | Squalene Synthase |
| SRM | Selected Reaction Monitoring |
| SWATH-MS | Sequential Window Acquisition of All Theoretical Mass Spectra |
| TF | Transcription Factor |
| TLA | Three Letter Acronym (included as per example) |
| TMT | Tandem Mass Tag |
| TPS | Terpene Synthase |
| UGT | UDP-Glycosyltransferase |
| USDA | United States Department of Agriculture |
| VIGS | Virus-Induced Gene Silencing |
| WGCNA | Weighted Gene Co-expression Network Analysis |
References
- Abdullah, Jiang, Z., Hong, X., Zhang, S., Yao, R., and Xiao, Y. (2020). CRISPR base editing and prime editing: DSB and template-free editing systems for bacteria and plants. Synth Syst Biotechnol 5, 277-292. [CrossRef]
- Agapakis, C. M., Niederholtmeyer, H., Noche, R. R., Lieberman, T. D., Megason, S. G., Way, J. C., and Silver, P. A. (2011). Towards a synthetic chloroplast. PLoS One 6, e18877. [CrossRef]
- Aharoni, A., Giri, A. P., Deuerlein, S., Griepink, F., de Kogel, W.-J., Verstappen, F. W. A., Verhoeven, H. A., Jongsma, M. A., Schwab, W., and Bouwmeester, H. J. (2003). Terpenoid metabolism in wild-type and transgenic Arabidopsis plants. Plant Cell 15, 2866-2884. [CrossRef]
- Aharoni, A., Jongsma, M. A., Kim, T.-Y., Ri, M.-B., Giri, A. P., Verstappen, F. W. A., Schwab, W., and Bouwmeester, H. J. (2006). Metabolic engineering of terpenoid biosynthesis in plants. Phytochem. Rev 5, 49-58. [CrossRef]
- Ajikumar, P. K., Xiao, W. H., Tyo, K. E., Wang, Y., Simeon, F., Leonard, E., Mucha, O., Phon, T. H., Pfeifer, B., and Stephanopoulos, G. (2010). Isoprenoid pathway optimization for Taxol precursor overproduction in Escherichia coli. Science 330, 70-74. [CrossRef]
- Al-Khayri, J. M., Sudheer, W. N., Lakshmaiah, V. V., Mukherjee, E., Nizam, A., Thiruvengadam, M., Nagella, P., Alessa, F. M., Al-Mssallem, M. Q., and Rezk, A. A. (2022). Biotechnological approaches for production of artemisinin, an anti-malarial drug from Artemisia annua L. Molecules 27, 3040. [CrossRef]
- Amini, H., Naghavi, M. R., Shen, T., Wang, Y., Nasiri, J., Khan, I. A., Fiehn, O., Zerbe, P., and Maloof, J. N. (2019). Tissue-specific transcriptome analysis reveals candidate genes for terpenoid and phenylpropanoid metabolism in the medicinal plant Ferula assafoetida. G3: Genes, Genomes, Genetics 9, 807-816. [CrossRef]
- Atanasov, A. G., Waltenberger, B., Pferschy-Wenzig, E.-M., Linder, T., Wawrosch, C., Uhrin, P., Temml, V., Wang, L., Schwaiger, S., and Heiss, E. H. (2015). Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv 33, 1582-1614. [CrossRef]
- Atanasov, A. G., Zotchev, S. B., Dirsch, V. M., and Supuran, C. T. (2021). Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov 20, 200-216. [CrossRef]
- Bai, W., Li, C., Li, W., Wang, H., Han, X., Wang, P., and Wang, L. (2024). Machine learning assists prediction of genes responsible for plant specialized metabolite biosynthesis by integrating multi-omics data. BMC Genom 25, 418. [CrossRef]
- Baloglu, M. C., Celik Altunoglu, Y., Baloglu, P., Yildiz, A. B., Türkölmez, N., and Özden Çiftçi, Y. (2022). Gene-Editing Technologies and Applications in Legumes: Progress, Evolution, and Future Prospects. Front Genet 13. [CrossRef]
- Bat-Erdene, U., Billingsley, J. M., Turner, W. C., Lichman, B. R., Ippoliti, F. M., Garg, N. K., O’Connor, S. E., and Tang, Y. (2021). Cell-Free Total Biosynthesis of Plant Terpene Natural Products using an Orthogonal Cofactor Regeneration System. ACS Catal 11, 9898-9903. [CrossRef]
- Bathe, U., Frolov, A., Porzel, A., and Tissier, A. (2019). CYP76 Oxidation Network of Abietane Diterpenes in Lamiaceae Reconstituted in Yeast. J Agric Food Chem 67, 13437-13450. [CrossRef]
- Bellucci, M., De Marchis, F., and Pompa, A. (2018). The endoplasmic reticulum is a hub to sort proteins toward unconventional traffic pathways and endosymbiotic organelles. J Exp Bot 69, 7-20. [CrossRef]
- Bhuyan, S. J., Kumar, M., Ramrao Devde, P., Rai, A. C., Mishra, A. K., Singh, P. K., and Siddique, K. H. M. (2023). Progress in gene editing tools, implications and success in plants: a review. Frontiers in Genome Editing 5, 1272678. [CrossRef]
- Booth, J. K., Yuen, M. M. S., Jancsik, S., Madilao, L. L., Page, J. E., and Bohlmann, J. (2020). Terpene synthases and terpene variation in Cannabis sativa. Plant Physiol 184, 130-147. [CrossRef]
- Boutanaev, A. M., Moses, T., Zi, J., Nelson, D. R., Mugford, S. T., Peters, R. J., and Osbourn, A. (2015). Investigation of terpene diversification across multiple sequenced plant genomes. Proc Natl Acad Sci USA 112, E81-E88. [CrossRef]
- Boynton, J. E., Gillham, N. W., Harris, E. H., Hosler, J. P., Johnson, A. M., Jones, A. R., Randolph-Anderson, B. L., Robertson, D., Klein, T. M., Shark, K. B., and et al. (1988). Chloroplast transformation in Chlamydomonas with high velocity microprojectiles. Science 240, 1534-1538. [CrossRef]
- Cao, K., Cui, Y., Sun, F., Zhang, H., Fan, J., Ge, B., Cao, Y., Wang, X., Zhu, X., and Wei, Z. (2023). Metabolic engineering and synthetic biology strategies for producing high-value natural pigments in Microalgae. Biotechnol Adv 68, 108236. [CrossRef]
- Chen, B.-S., and Wu, C.-C. (2013). Systems biology as an integrated platform for bioinformatics, systems synthetic biology, and systems metabolic engineering. Cells 2, 635-688. [CrossRef]
- Chen, C., Zheng, Y., Zhong, Y., Wu, Y., Li, Z., Xu, L.-A., and Xu, M. (2018). Transcriptome analysis and identification of genes related to terpenoid biosynthesis in Cinnamomum camphora. BMC genomics 19, 550. [CrossRef]
- Chen, F., Tholl, D., D’Auria, J. C., Farooq, A., Pichersky, E., and Gershenzon, J. (2003). Biosynthesis and emission of terpenoid volatiles from Arabidopsis flowers. Plant Cell 15, 481-494. [CrossRef]
- Chen, K. H., Boettiger, A. N., Moffitt, J. R., Wang, S., and Zhuang, X. (2015). Spatially resolved, highly multiplexed RNA profiling in single cells. Science 348, aaa6090. [CrossRef]
- Chung, C. H., Lin, D.-W., Eames, A., and Chandrasekaran, S. (2021). Next-generation genome-scale metabolic modeling through integration of regulatory mechanisms. Metabolites 11, 606. [CrossRef]
- Clough, S. J., and Bent, A. F. (1998). Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16, 735-743. [CrossRef]
- Cong, L., Ran, F. A., Cox, D., Lin, S., Barretto, R., Habib, N., Hsu, P. D., Wu, X., Jiang, W., and Marraffini, L. A. (2013). Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819-823. [CrossRef]
- Cong, L., Ran, F. A., Cox, D., Lin, S., Barretto, R., Habib, N., Hsu, P. D., Wu, X., Jiang, W., Marraffini, L. A., and Zhang, F. (2013). Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819-823. [CrossRef]
- Courdavault, V., O’Connor, S. E., Jensen, M. K., and Papon, N. (2021). Metabolic engineering for plant natural products biosynthesis: new procedures, concrete achievements and remaining limits. Nat Prod Rep 38, 2145-2153. [CrossRef]
- Czechowski, T., Li, Y., Gilday, A. D., Harvey, D., Swamidatta, S. H., Lichman, B. R., Ward, J. L., and Graham, I. A. (2025). Evolution of linear triterpenoid biosynthesis within the Euphorbia genus. Nat Commun 16, 5602. [CrossRef]
- Dahal, S., Yurkovich, J. T., Xu, H., Palsson, B. O., and Yang, L. (2020). Synthesizing systems biology knowledge from omics using genome-scale models. Proteomics 20, 1900282. [CrossRef]
- Dai, Z., Liu, Y., Zhang, X., Shi, M., Wang, B., Wang, D., Huang, L., and Zhang, X. (2013). Metabolic engineering of Saccharomyces cerevisiae for production of ginsenosides. Metabolic engineering 20, 146-156. [CrossRef]
- Daliri, E. B.-M., Ofosu, F. K., Chelliah, R., Kim, J.-H., Kim, J.-R., Yoo, D., and Oh, D.-H. (2020). Untargeted metabolomics of fermented rice using UHPLC Q-TOF MS/MS reveals an abundance of potential antihypertensive compounds. Foods 9, 1007. [CrossRef]
- Das, S., Kwon, M., and Kim, J.-Y. (2024). Enhancement of specialized metabolites using CRISPR/Cas gene editing technology in medicinal plants. Frontiers in plant science 15, 1279738. [CrossRef]
- De Paola, C. (2024). Enhancing Nicotiana benthamiana as chassis for molecular farming: Targeting flowering time for increased biomass and recombinant protein production.
- Domon, B., and Aebersold, R. (2010). Options and considerations when selecting a quantitative proteomics strategy. Nat Biotechnol 28, 710-721. [CrossRef]
- Dong, Q., Zou, Q.-C., Mao, L.-H., Tian, D.-Q., Hu, W., Cao, X.-R., and Ding, H.-Q. (2022). The chromosome-scale assembly of the Curcuma alismatifolia genome provides insight into anthocyanin and terpenoid biosynthesis. Front Plant Sci 13, 899588. [CrossRef]
- Du, Y., Song, W., Yin, Z., Wu, S., Liu, J., Wang, N., Jin, H., Qiao, J., and Huo, Y.-X. (2022). Genomic analysis based on chromosome-level genome assembly reveals an expansion of terpene biosynthesis of Azadirachta indica. Front Plant Sci 13, 853861. [CrossRef]
- Faktor, J., Michalová, E., and Bouchal, P. (2014). p-SRM, SWATH a HRM-cílené proteomické přístupy na hmotnostním spektrometru TripleTOF 5600+ a jejich aplikace v onkologickém výzkumu. Klinická onkologie 27, 110-115. [CrossRef]
- Feng, Z., Zhang, Z., Hua, K., Gao, X., Mao, Y., Botella, J. R., and Zhu, J.-K. (2018). A highly efficient cell division-specific CRISPR/Cas9 system generates homozygous mutants for multiple genes in Arabidopsis. Int J Mol Sci 19, 3925. [CrossRef]
- Flügge, U. I., and Gao, W. (2005). Transport of isoprenoid intermediates across chloroplast envelope membranes. Plant Biol 7, 91-97. [CrossRef]
- Fu, J., Xu, W., Huang, W., Wang, B., Li, S., Zhang, J., and Chang, L. (2021). Importation of taxadiene synthase into chloroplast improves taxadiene production in tobacco. Planta 253, 107. [CrossRef]
- Fu, X., Liu, H., Hassani, D., Peng, B., Yan, X., Wang, Y., Wang, C., Li, L., Liu, P., and Pan, Q. (2020). AaABCG40 enhances artemisinin content and modulates drought tolerance in Artemisia annua. Front Plant Sci 11, 950. [CrossRef]
- Gillet, L. C., Navarro, P., Tate, S., Röst, H., Selevsek, N., Reiter, L., Bonner, R., and Aebersold, R. (2012). Targeted data extraction of the MS/MS spectra generated by data-independent acquisition: a new concept for consistent and accurate proteome analysis. Mol Cell Proteomics 11, O111-016717. [CrossRef]
- Graham, M. W., Craig, S., and Waterhouse, P. M. (1997). Expression patterns of vascular-specific promoters RolC and Sh in transgenic potatoes and their use in engineering PLRV-resistant plants. Plant Mol Biol 33, 729-735. [CrossRef]
- Gruchattka, E., and Kayser, O. (2015). In vivo validation of in silico predicted metabolic engineering strategies in yeast: Disruption of α-ketoglutarate dehydrogenase and expression of ATP-citrate lyase for terpenoid production. PloS one 10, e0144981. [CrossRef]
- Guerriero, G., Berni, R., Muñoz-Sanchez, J. A., Apone, F., Abdel-Salam, E. M., Qahtan, A. A., Alatar, A. A., Cantini, C., Cai, G., and Hausman, J.-F. (2018). Production of plant secondary metabolites: Examples, tips and suggestions for biotechnologists. Genes 9, 309. [CrossRef]
- Gwak, Y. S., Han, J. Y., and Choi, Y. E. (2019). Production of ginsenoside aglycone (protopanaxatriol) and male sterility of transgenic tobacco co-overexpressing three Panax ginseng genes: PgDDS, CYP716A47, and CYP716A53v2. J Ginseng Res 43, 261-271. [CrossRef]
- Hamdan, M. F., Mohd Noor, S. N., Abd-Aziz, N., Pua, T.-L., and Tan, B. C. (2022). Green revolution to gene revolution: technological advances in agriculture to feed the world. Plants 11, 1297. [CrossRef]
- Han, J., Wu, Y., Zhou, Y., and Li, S. (2021). Engineering Saccharomyces cerevisiae to produce plant benzylisoquinoline alkaloids. Abiotech 2, 264-275. [CrossRef]
- Hao, G., Ji, H., Li, Y., Shi, R., Wang, J., Feng, L., and Huang, L. (2012). Exogenous ABA and polyamines enhanced salvianolic acids contents in hairy root cultures of Salvia miltiorrhiza Bge. f. alba. Plant Omics 5, 446-452.
- Hao, X., Pu, Z., Cao, G., You, D., Zhou, Y., Deng, C., Shi, M., Nile, S. H., Wang, Y., and Zhou, W. (2020). Tanshinone and salvianolic acid biosynthesis are regulated by SmMYB98 in Salvia miltiorrhiza hairy roots. J Adv Res 23, 1-12. [CrossRef]
- Hassani, D., Taheri, A., Fu, X., Qin, W., Hang, L., Ma, Y., and Tang, K. (2023). Elevation of artemisinin content by co-transformation of artemisinin biosynthetic pathway genes and trichome-specific transcription factors in Artemisia annua. Front Plant Sci 14, 1118082. [CrossRef]
- He, C., Zhang, C., Bian, T., Jiao, K., Su, W., Wu, K.-J., and Su, A. (2023). A review on artificial intelligence enabled design, synthesis, and process optimization of chemical products for industry 4.0. Processes 11, 330. [CrossRef]
- He, J., Liu, X., and Li, C. (2024). Engineering Electron Transfer Pathway of Cytochrome P450s. Molecules 29. [CrossRef]
- Heinig, U., Scholz, S., and Jennewein, S. (2013). Getting to the bottom of Taxol biosynthesis by fungi. Fungal Divers 60, 161-170. [CrossRef]
- Hiei, Y., Ohta, S., Komari, T., and Kumashiro, T. (1994). Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6, 271-282. [CrossRef]
- Hong, G.-J., Hu, W.-L., Li, J.-X., Chen, X.-Y., and Wang, L.-J. (2009). Increased accumulation of artemisinin and anthocyanins in Artemisia annua expressing the Arabidopsis blue light receptor CRY1. Plant Mol Biol Rep 27, 334-341. [CrossRef]
- Hong, J., Yang, L., Zhang, D., and Shi, J. (2016). Plant metabolomics: an indispensable system biology tool for plant science. Int J Mol Sci 17, 767. [CrossRef]
- Hsu, C. T., Yuan, Y. H., Lin, Y. C., Lin, S., Cheng, Q. W., Wu, F. H., Sheen, J., Shih, M. C., and Lin, C. S. (2021). Efficient and Economical Targeted Insertion in Plant Genomes via Protoplast Regeneration. The CRISPR journal 4, 752-760. [CrossRef]
- Ignea, C., Pontini, M., Maffei, M. E., Makris, A. M., and Kampranis, S. C. (2014). Engineering monoterpene production in yeast using a synthetic dominant negative geranyl diphosphate synthase. ACS Synth Biol 3, 298-306. [CrossRef]
- Ishii, Y., Takeda, S., Yamada, H., and Oguri, K. (2005). Functional protein-protein interaction of drug metabolizing enzymes. Front Biosci 10, 887-895. [CrossRef]
- Jackson, A. R. (2009). Predicting flavonoid UGT regioselectivity with graphical residue models and machine learning.
- Jackson, H. O., Taunt, H. N., Mordaka, P. M., Smith, A. G., and Purton, S. (2021). The algal chloroplast as a testbed for synthetic biology designs aimed at radically rewiring plant metabolism. Front Plant Sci 12, 708370. [CrossRef]
- Jennewein, S., Wildung, M. R., Chau, M., Walker, K., and Croteau, R. (2004). Random sequencing of an induced Taxus cell cDNA library for identification of clones involved in Taxol biosynthesis. Proc Natl Acad Sci USA 101, 9149-9154. [CrossRef]
- Jiang, W., Fu, X., Pan, Q., Tang, Y., Shen, Q., Lv, Z., Yan, T., Shi, P., Li, L., Zhang, L., Wang, G., Sun, X., and Tang, K. (2016). Overexpression of AaWRKY1 Leads to an Enhanced Content of Artemisinin in Artemisia annua. BioMed Res Int 2016, 7314971. [CrossRef]
- Jiang, Z., Hong, X., Zhang, S., Yao, R., and Xiao, Y. I. (2020). CRISPR base editing and prime editing: DSB and template-free editing systems for bacteria and plants. Synth Syst Biotechnol 5, 277-292. [CrossRef]
- Jinek, M., Chylinski, K., Fonfara, I., Hauer, M., Doudna, J. A., and Charpentier, E. (2012). A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816-821. [CrossRef]
- Jung, S.-C., Kim, W., Park, S. C., Jeong, J., Park, M. K., Lim, S., Lee, Y., Im, W.-T., Lee, J. H., and Choi, G. (2014). Two ginseng UDP-glycosyltransferases synthesize ginsenoside Rg3 and Rd. Plant Cell Physiol 55, 2177-2188. [CrossRef]
- Kanagarajan, S., Muthusamy, S., Gliszczyńska, A., Lundgren, A., and Brodelius, P. E. (2012). Functional expression and characterization of sesquiterpene synthases from Artemisia annua L. using transient expression system in Nicotiana benthamiana. Plant Cell Rep 31, 1309-1319. [CrossRef]
- Karkute, S. G., Singh, A. K., Gupta, O. P., Singh, P. M., and Singh, B. (2017). CRISPR/Cas9 mediated genome engineering for improvement of horticultural crops. Front Plant Sci 8, 1635. [CrossRef]
- Kautsar, S. A., Suarez Duran, H. G., Blin, K., Osbourn, A., and Medema, M. H. (2017). plantiSMASH: automated identification, annotation and expression analysis of plant biosynthetic gene clusters. Nucleic Acids Res 45, W55-W63. [CrossRef]
- Kochan, E., Caban, S., Szymańska, G., Szymczyk, P., Lipert, A., Kwiatkowski, P., and Sienkiewicz, M. (2017). Ginsenoside content in suspension cultures of Panax quinquefolium L. cultivated in shake flasks and stirred-tank bioreactor. [CrossRef]
- Köllner, T. G., Degenhardt, J., and Gershenzon, J. (2020). The product specificities of maize terpene synthases TPS4 and TPS10 are determined both by active site amino acids and residues adjacent to the active site. Plants 9, 552. [CrossRef]
- Kowalczyk, T., Wieczfinska, J., Skała, E., Śliwiński, T., and Sitarek, P. (2020). Transgenesis as a tool for the efficient production of selected secondary metabolites from plant in vitro cultures. Plants 9, 132. [CrossRef]
- Kumar, S. (2010). Engineering cytochrome P450 biocatalysts for biotechnology, medicine and bioremediation. Expert Opin. Drug Metab Toxicol 6, 115-131. [CrossRef]
- Lange, B. M., and Croteau, R. (1999). Isopentenyl diphosphate biosynthesis via a mevalonate-independent pathway: isopentenyl monophosphate kinase catalyzes the terminal enzymatic step. Proc Natl Acad Sci USA 96, 13714-13719. [CrossRef]
- Lange, V., Picotti, P., Domon, B., and Aebersold, R. (2008). Selected reaction monitoring for quantitative proteomics: a tutorial. Mol Syst Biol 4, 222. [CrossRef]
- Lanier, E. R., Andersen, T. B., and Hamberger, B. (2023). Plant terpene specialized metabolism: complex networks or simple linear pathways? Plant J 114, 1178-1201. [CrossRef]
- Lawson, C. E., Martí, J. M., Radivojevic, T., Jonnalagadda, S. V. R., Gentz, R., Hillson, N. J., Peisert, S., Kim, J., Simmons, B. A., Petzold, C. J., Singer, S. W., Mukhopadhyay, A., Tanjore, D., Dunn, J. G., and Garcia Martin, H. (2021). Machine learning for metabolic engineering: A review. Metab Eng 63, 34-60. [CrossRef]
- Lee, K. W., Lee, W. H., Han, B.-S., Lee, J. H., Doo, E. K., and Kim, J.-H. (2020). Molecular drug Discovery of single Ginsenoside compounds as a potent Bruton’s tyrosine kinase inhibitor. Int J Mol Sci 21, 3065. [CrossRef]
- Leivar, P., Antolín-Llovera, M., Ferrero, S., Closa, M., Arro, M., Ferrer, A., Boronat, A., and Campos, N. (2011). Multilevel control of Arabidopsis 3-hydroxy-3-methylglutaryl coenzyme A reductase by protein phosphatase 2A. Plant Cell 23, 1494-1511. [CrossRef]
- Li-Beisson, Y., Shorrosh, B., Beisson, F., Andersson, M. X., Arondel, V., Bates, P. D., Baud, S., Bird, D., DeBono, A., and Durrett, T. P. (2013). Acyl-lipid metabolism. The Arabidopsis book/American Society of Plant Biologists 11, e0161. [CrossRef]
- Li, B., Cui, G., Shen, G., Zhan, Z., Huang, L., Chen, J., and Qi, X. (2017). Targeted mutagenesis in the medicinal plant Salvia miltiorrhiza. Sci Rep 7, 43320. [CrossRef]
- Li, C., Yan, X., Xu, Z., Wang, Y., Shen, X., Zhang, L., Zhou, Z., and Wang, P. (2022). Pathway elucidation of bioactive rhamnosylated ginsenosides in Panax ginseng and their de novo high-level production by engineered Saccharomyces cerevisiae. Commun Biol 5, 775. [CrossRef]
- Li, F., Wu, C., Dewer, Y., Liang, Y., Zhou, J. J., Luo, C., and Wang, G. Tandem duplication of P450 genes is involved in homoterpene biosynthesis in lima bean (Phaseolus lunatus). Pest Manag Sci. [CrossRef]
- Li, J.-F., Norville, J. E., Aach, J., McCormack, M., Zhang, D., Bush, J., Church, G. M., and Sheen, J. (2013). Multiplex and homologous recombination-mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat Biotechnol 31, 688-691. [CrossRef]
- Li, L., Fu, J., and Liu, N. (2024). Advances in the Structures, Pharmacological Activities, and Biosynthesis of Plant Diterpenoids. J Microbiol Biotechnol 34, 1563-1579. [CrossRef]
- Li, L., Hao, X., Liu, H., Wang, W., Fu, X., Ma, Y., Shen, Q., Chen, M., and Tang, K. (2019). Jasmonic acid-responsive AabHLH1 positively regulates artemisinin biosynthesis in Artemisia annua. Biotechnol Appl Biochem 66, 369-375. [CrossRef]
- Li, Y.-C., Tao, W.-Y., and Cheng, L. (2009). Paclitaxel production using co-culture of Taxus suspension cells and paclitaxel-producing endophytic fungi in a co-bioreactor. Appl Microbiol Biotechnol 83, 233-239. [CrossRef]
- Li, Y., Chen, J., Zhi, J., Huang, D., Zhang, Y., Zhang, L., Duan, X., Zhang, P., Qiu, S., and Geng, J. (2025). The ABC transporter SmABCG1 mediates tanshinones export from the peridermic cells of Salvia miltiorrhiza root. J. Integr Plant Biol 67, 135-149. [CrossRef]
- Li, Y., Huang, C., Liu, Y., Zeng, J., Yu, H., Tong, Z., Yuan, X., Sui, X., Fang, D., and Xiao, B. (2023). CRISPR/Cas9-mediated seamless gene replacement in protoplasts expands the resistance spectrum to TMV-U1 strain in regenerated Nicotiana tabacum. Plant Biotechnol J 21, 2641-2653. [CrossRef]
- Li, Y., Yang, Y., Li, L., Tang, K., Hao, X., and Kai, G. (2024a). Advanced metabolic engineering strategies for increasing artemisinin yield in Artemisia annua L. Hortic Res 11, uhad292. [CrossRef]
- Li, Y., Yang, Y., Li, L., Tang, K., Hao, X., and Kai, G. (2024b). Advanced metabolic engineering strategies for increasing artemisinin yield in Artemisia annua L. Hortic Res 11. [CrossRef]
- Liao, X., Guo, S., Liao, B., Shen, X., He, W., Meng, Y., Liang, C., Pei, J., Liu, J., and Zhang, Y. (2023). Chromatin architecture of two different strains of Artemisia annua reveals the alterations in interaction and gene expression. Planta 258, 74. [CrossRef]
- Liu, C.-L., Xue, K., Yang, Y., Liu, X., Li, Y., Lee, T. S., Bai, Z., and Tan, T. (2022). Metabolic engineering strategies for sesquiterpene production in microorganism. Crit Rev Biotechnol 42, 73-92. [CrossRef]
- Liu, N., Li, T., Reid, W. R., Yang, T., and Zhang, L. (2011). Multiple cytochrome P450 genes: their constitutive overexpression and permethrin induction in insecticide resistant mosquitoes, Culex quinquefasciatus. PLoS ONE 6, e23403. [CrossRef]
- Liu, X., Wu, S., Xu, J., Sui, C., and Wei, J. (2017). Application of CRISPR/Cas9 in plant biology. Acta Pharm Sin B 7, 292-302. [CrossRef]
- Lowe, K., Wu, E., Wang, N., Hoerster, G., Hastings, C., Cho, M.-J., Scelonge, C., Lenderts, B., Chamberlin, M., and Cushatt, J. (2016). Morphogenic regulators Baby boom and Wuschel improve monocot transformation. Plant Cell 28, 1998-2015. [CrossRef]
- Lu, X., Tang, K., and Li, P. (2016). Plant metabolic engineering strategies for the production of pharmaceutical terpenoids. Front Plant Sci 7, 1647. [CrossRef]
- Ma, D., Pu, G., Lei, C., Ma, L., Wang, H., Guo, Y., Chen, J., Du, Z., Wang, H., and Li, G. (2009). Isolation and characterization of AaWRKY1, an Artemisia annua transcription factor that regulates the amorpha-4, 11-diene synthase gene, a key gene of artemisinin biosynthesis. Plant Cell Physiol 50, 2146-2161. [CrossRef]
- Ma, R., Su, P., Ma, Q., Guo, J., Chen, S., Jin, B., Zhang, H., Tang, J., Zhou, T., and Xiao, C. (2022). Identification of (-)-bornyl diphosphate synthase from Blumea balsamifera and its application for (-)-borneol biosynthesis in Saccharomyces cerevisiae. Syst Biotechnol 7, 490-497. [CrossRef]
- Ma, Y.-N., Xu, D.-B., Li, L., Zhang, F., Fu, X.-Q., Shen, Q., Lyu, X.-Y., Wu, Z.-K., Pan, Q.-F., and Shi, P. (2018). Jasmonate promotes artemisinin biosynthesis by activating the TCP14-ORA complex in Artemisia annua. Sci Adv 4, eaas9357. [CrossRef]
- Ma, Y., Yuan, L., Wu, B., Li, X. e., Chen, S., and Lu, S. (2012). Genome-wide identification and characterization of novel genes involved in terpenoid biosynthesis in Salvia miltiorrhiza. J Exp Bot 63, 2809-2823. [CrossRef]
- Ma, Y. N., Xu, D. B., Yan, X., Wu, Z. K., Kayani, S. I., Shen, Q., Fu, X. Q., Xie, L. H., Hao, X. L., and Hassani, D. (2021). Jasmonate-and abscisic acid-activated AaGSW1-AaTCP15/AaORA transcriptional cascade promotes artemisinin biosynthesis in Artemisia annua. Plant Biotechnol J 19, 1412-1428. [CrossRef]
- Madhavan, A., Arun, K. B., Sindhu, R., Binod, P., Kim, S. H., and Pandey, A. (2019). Tailoring of microbes for the production of high value plant-derived compounds: From pathway engineering to fermentative production. Biochimica et biophysica acta. Proteins and proteomics 1867, 140262. [CrossRef]
- Mani, V., Park, S., Kim, J. A., Lee, S. I., and Lee, K. (2021). Metabolic Perturbation and Synthetic Biology Strategies for Plant Terpenoid Production-An Updated Overview. Plants (Basel, Switzerland) 10. [CrossRef]
- Mao, J.-p., Huang, L.-w., Hao, J., Liu, T.-y., and Huang, S.-w. (2020). The evolutionary rate variation among genes of MVA and MEP pathways in plant terpenoid biosynthesis. [CrossRef]
- Masakapalli, S. (2011). Network flux analysis of central metabolism in plants.
- Masakapalli, S. K., Bryant, F. M., Kruger, N. J., and Ratcliffe, R. G. (2014). The metabolic flux phenotype of heterotrophic Arabidopsis cells reveals a flexible balance between the cytosolic and plastidic contributions to carbohydrate oxidation in response to phosphate limitation. Plant J 78, 964-977. [CrossRef]
- Matías-Hernández, L., Jiang, W., Yang, K., Tang, K., Brodelius, P. E., and Pelaz, S. (2017). AaMYB1 and its orthologue AtMYB61 affect terpene metabolism and trichome development in Artemisia annua and Arabidopsis thaliana. Plant J 90, 520-534. [CrossRef]
- Meng, J., Qiu, Y., and Shi, S. (2020). CRISPR/Cas9 systems for the development of Saccharomyces cerevisiae cell factories. Front Bioeng Biotechnol 8, 594347. [CrossRef]
- Mitra, S., Anand, U., Ghorai, M., Kant, N., Kumar, M., Radha, Jha, N. K., Swamy, M. K., Proćków, J., and de la Lastra, J. M. P. (2023). Genome editing technologies, mechanisms and improved production of therapeutic phytochemicals: Opportunities and prospects. Biotechnol Bioeng 120, 82-94. [CrossRef]
- Moses, T., Pollier, J., Almagro, L., Buyst, D., Van Montagu, M., Pedreño, M. A., Martins, J. C., Thevelein, J. M., and Goossens, A. (2014). Combinatorial biosynthesis of sapogenins and saponins in Saccharomyces cerevisiae using a C-16α hydroxylase from Bupleurum falcatum. Proc Natl Acad Sci USA 111, 1634-1639. [CrossRef]
- Motolinía-Alcántara, E. A., Castillo-Araiza, C. O., Rodríguez-Monroy, M., Román-Guerrero, A., and Cruz-Sosa, F. (2021). Engineering considerations to produce bioactive compounds from plant cell suspension culture in bioreactors. Plants 10, 2762. [CrossRef]
- Noushahi, H. A., Khan, A. H., Noushahi, U. F., Hussain, M., Javed, T., Zafar, M., Batool, M., Ahmed, U., Liu, K., and Harrison, M. T. (2022). Biosynthetic pathways of triterpenoids and strategies to improve their biosynthetic efficiency. Plant Growth Regul 97, 439-454. [CrossRef]
- Obata, T., and Fernie, A. R. (2012). The use of metabolomics to dissect plant responses to abiotic stresses. Cell Mol Life Sci 69, 3225-3243. [CrossRef]
- Oey, M., Lohse, M., Kreikemeyer, B., and Bock, R. (2009). Exhaustion of the chloroplast protein synthesis capacity by massive expression of a highly stable protein antibiotic. Plant J: for cell and molecular biology 57, 436-445. [CrossRef]
- Oh, S.-W., Imran, M., Kim, E.-H., Park, S.-Y., Lee, S.-G., Park, H.-M., Jung, J.-W., and Ryu, T.-H. (2023). Approach strategies and application of metabolomics to biotechnology in plants. Front Plant Sci 14, 1192235. [CrossRef]
- Otey, C. R., Bandara, G., Lalonde, J., Takahashi, K., and Arnold, F. H. (2006). Preparation of human metabolites of propranolol using laboratory-evolved bacterial cytochromes P450. Biotechnol Bioeng 93, 494-499. [CrossRef]
- Ouyang, X., Cha, Y., Li, W., Zhu, C., Zhu, M., Li, S., Zhuo, M., Huang, S., and Li, J. (2019). Stepwise engineering of Saccharomyces cerevisiae to produce (+)-valencene and its related sesquiterpenes. RSC advances 9, 30171-30181. [CrossRef]
- Paddon, C. J., Westfall, P. J., Pitera, D. J., Benjamin, K., Fisher, K., McPhee, D., Leavell, M. D., Tai, A., Main, A., Eng, D., Polichuk, D. R., Teoh, K. H., Reed, D. W., Treynor, T., Lenihan, J., Fleck, M., Bajad, S., Dang, G., Dengrove, D., Diola, D., Dorin, G., Ellens, K. W., Fickes, S., Galazzo, J., Gaucher, S. P., Geistlinger, T., Henry, R., Hepp, M., Horning, T., Iqbal, T., Jiang, H., Kizer, L., Lieu, B., Melis, D., Moss, N., Regentin, R., Secrest, S., Tsuruta, H., Vazquez, R., Westblade, L. F., Xu, L., Yu, M., Zhang, Y., Zhao, L., Lievense, J., Covello, P. S., Keasling, J. D., Reiling, K. K., Renninger, N. S., and Newman, J. D. (2013). High-level semi-synthetic production of the potent antimalarial artemisinin. Nature 496, 528-532. [CrossRef]
- Patil, R. A., Kolewe, M. E., Normanly, J., Walker, E. L., and Roberts, S. C. (2012). Contribution of taxane biosynthetic pathway gene expression to observed variability in paclitaxel accumulation in Taxus suspension cultures. Biotechnol J 7, 418-427. [CrossRef]
- Payne, R. M. E., Xu, D., Foureau, E., Teto Carqueijeiro, M. I. S., Oudin, A., Bernonville, T. D. d., Novak, V., Burow, M., Olsen, C.-E., and Jones, D. M. (2017). An NPF transporter exports a central monoterpene indole alkaloid intermediate from the vacuole. Nat Plants 3, 1-9. [CrossRef]
- Peterson, A. C., Russell, J. D., Bailey, D. J., Westphall, M. S., and Coon, J. J. (2012). Parallel reaction monitoring for high resolution and high mass accuracy quantitative, targeted proteomics. MOL CELL PROTEOMICS 11, 1475-1488. [CrossRef]
- Pick, T. R., and Weber, A. P. M. (2014). Unknown components of the plastidial permeome. Front Plant Sci 5, 410. [CrossRef]
- Pinu, F. R., Beale, D. J., Paten, A. M., Kouremenos, K., Swarup, S., Schirra, H. J., and Wishart, D. (2019). Systems biology and multi-omics integration: viewpoints from the metabolomics research community. Metabolites 9, 76. [CrossRef]
- Promdonkoy, P., Sornlek, W., Preechakul, T., Tanapongpipat, S., and Runguphan, W. (2022). Metabolic engineering of Saccharomyces cerevisiae for production of fragrant terpenoids from agarwood and sandalwood. Fermentation 8, 429. [CrossRef]
- Qiu, C., Liu, Y., Wu, Y., Zhao, L., and Pei, J. (2022). Functional characterization and screening of promiscuous kinases and isopentenyl phosphate kinases for the synthesis of DMAPP via a one-pot enzymatic cascade. Int J Mol Sci 23, 12904. [CrossRef]
- Reed, J., and Osbourn, A. (2018). Engineering terpenoid production through transient expression in Nicotiana benthamiana. Plant Cell Rep 37, 1431-1441. [CrossRef]
- Reiter, L., Rinner, O., Picotti, P., Hüttenhain, R., Beck, M., Brusniak, M.-Y., Hengartner, M. O., and Aebersold, R. (2011). mProphet: automated data processing and statistical validation for large-scale SRM experiments. Nat Methods 8, 430-435. [CrossRef]
- Ren, L., Wan, W., Yin, D., Deng, X., Ma, Z., Gao, T., and Cao, X. (2023). Genome-wide analysis of WRKY transcription factor genes in Toona sinensis: An insight into evolutionary characteristics and terpene synthesis. Front Plant Sci 13, 1063850. [CrossRef]
- Rinaldi, M. A., Ferraz, C. A., and Scrutton, N. S. (2022). Alternative metabolic pathways and strategies to high-titre terpenoid production in Escherichia coli. Nat Prod Rep 39, 90-118. [CrossRef]
- Rizvi, N. F., Weaver, J. D., Cram, E. J., and Lee-Parsons, C. W. T. (2016). Silencing the transcriptional repressor, ZCT1, illustrates the tight regulation of terpenoid indole alkaloid biosynthesis in Catharanthus roseus hairy roots. PLoS One 11, e0159712. [CrossRef]
- Ro, D.-K., Paradise, E. M., Ouellet, M., Fisher, K. J., Newman, K. L., Ndungu, J. M., Ho, K. A., Eachus, R. A., Ham, T. S., and Kirby, J. (2006). Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940-943. [CrossRef]
- Ro, D. K., Paradise, E. M., Ouellet, M., Fisher, K. J., Newman, K. L., Ndungu, J. M., Ho, K. A., Eachus, R. A., Ham, T. S., Kirby, J., Chang, M. C., Withers, S. T., Shiba, Y., Sarpong, R., and Keasling, J. D. (2006). Production of the antimalarial drug precursor artemisinic acid in engineered yeast. Nature 440, 940-943. [CrossRef]
- Sakamoto, W., Miyagishima, S. Y., and Jarvis, P. (2008). Chloroplast biogenesis: control of plastid development, protein import, division and inheritance. The Arabidopsis Book 6, e0110. [CrossRef]
- Samusevich, R., Hebra, T., Bushuiev, R., Bushuiev, A., Chatpatanasiri, R., Kulhánek, J., Čalounová, T., Perković, M., Engst, M., and Tajovská, A. (2024). Discovery and characterization of terpene synthases powered by machine learning. bioRxiv 10, 29-577750. [CrossRef]
- Scossa, F., Benina, M., Alseekh, S., Zhang, Y., and Fernie, A. R. (2018). The integration of metabolomics and next-generation sequencing data to elucidate the pathways of natural product metabolism in medicinal plants. Planta Med 84, 855-873. [CrossRef]
- Shan, Q., Wang, Y., Li, J., Zhang, Y., Chen, K., Liang, Z., Zhang, K., Liu, J., Xi, J. J., and Qiu, J.-L. (2013). Targeted genome modification of crop plants using a CRISPR-Cas system. Nat Biotechnol 31, 686-688. [CrossRef]
- She, J., Yan, H., Yang, J., Xu, W., and Su, Z. (2019). croFGD: Catharanthus roseus functional genomics database. Front Genet 10, 238. [CrossRef]
- Shen, Q., Huang, H., Zhao, Y., Xie, L., He, Q., Zhong, Y., Wang, Y., Wang, Y., and Tang, K. (2019). The transcription factor Aabzip9 positively regulates the biosynthesis of artemisinin in Artemisia annua. Front Plant Sci 10, 1294. [CrossRef]
- Shen, Q., Zhang, L., Liao, Z., Wang, S., Yan, T., Shi, P. U., Liu, M., Fu, X., Pan, Q., and Wang, Y. (2018). The genome of Artemisia annua provides insight into the evolution of Asteraceae family and artemisinin biosynthesis. Mol Plant 11, 776-788. [CrossRef]
- Shi, P., Fu, X., Liu, M., Shen, Q., Jiang, W., Li, L., Sun, X., and Tang, K. (2017). Promotion of artemisinin content in Artemisia annua by overexpression of multiple artemisinin biosynthetic pathway genes. Plant Cell Tissue Organ Cult 129, 251-259. [CrossRef]
- Silva, T. N., Thomas, J. B., Dahlberg, J., Rhee, S. Y., and Mortimer, J. C. (2022). Progress and challenges in sorghum biotechnology, a multipurpose feedstock for the bioeconomy. J Exp Bot 73, 646-664. [CrossRef]
- Sirirungruang, S., Markel, K., and Shih, P. M. (2022). Plant-based engineering for production of high-valued natural products. Nat Prod Rep 39, 1492-1509. [CrossRef]
- Sood, H. (2020). Production of medicinal compounds from endangered and commercially important medicinal plants through cell and tissue culture technology for herbal industry. In Bioactive compounds in nutraceutical and functional food for good human health. IntechOpen.
- Spyropoulou, E. A., Haring, M. A., and Schuurink, R. C. (2014). RNA sequencing on Solanum lycopersicum trichomes identifies transcription factors that activate terpene synthase promoters. BMC genomics 15, 402. [CrossRef]
- Sykłowska-Baranek, K., Szala, K., Pilarek, M., Orzechowski, R., and Pietrosiuk, A. (2018). A cellulase-supported two-phase in situ system for enhanced biosynthesis of paclitaxel in Taxus× media hairy roots. Acta Physiol Plant 40, 201. [CrossRef]
- Szymczyk, P., Szymańska, G., Kuźma, Ł., Jeleń, A., and Balcerczak, E. (2022). Methyl jasmonate activates the 2C methyl-D-erithrytol 2, 4-cyclodiphosphate synthase gene and stimulates tanshinone accumulation in Salvia miltiorrhiza solid callus cultures. Molecules 27, 1772. [CrossRef]
- Tai, Y., Hou, X., Liu, C., Sun, J., Guo, C., Su, L., Jiang, W., Ling, C., Wang, C., and Wang, H. (2020). Phytochemical and comparative transcriptome analyses reveal different regulatory mechanisms in the terpenoid biosynthesis pathways between Matricaria recutita L. and Chamaemelum nobile L. BMC genomics 21, 169. [CrossRef]
- Tang, Y., and Fu, Y. (2018). Class 2 CRISPR/Cas: an expanding biotechnology toolbox for and beyond genome editing. Cell Biosci 8, 59. [CrossRef]
- Tang, Y., Xiang, L., Zhang, F., Tang, K., and Liao, Z. (2023). Metabolic regulation and engineering of artemisinin biosynthesis in A. annua. Medicinal Plant Biology 2. [CrossRef]
- Tao, S., Liu, P., Shi, Y., Feng, Y., Gao, J., Chen, L., Zhang, A., Cheng, X., Wei, H., and Zhang, T. (2022). Single-cell transcriptome and network analyses unveil key transcription factors regulating mesophyll cell development in maize. Genes 13, 374. [CrossRef]
- Tee, M. K., and Miller, W. L. (2013). Phosphorylation of human cytochrome P450c17 by p38α selectively increases 17, 20 lyase activity and androgen biosynthesis. J Biol Chem 288, 23903-23913. [CrossRef]
- Tharasirivat, V., and Jantaro, S. (2023). Increased biomass and polyhydroxybutyrate production by synechocystis sp. PCC 6803 overexpressing RuBisCO genes. Int J Mol Sci 24. [CrossRef]
- Tian, M., Luo, L., Jin, B., Liu, J., Chen, T., Tang, J., Shen, Y., Zhang, H., Guo, J., Zhang, H., Cui, G., and Huang, L. (2025). Highly efficient Agrobacterium rhizogenes-mediated gene editing system in Salvia miltiorrhiza inbred line bh2-7. Plant Biotechnol J 23, 2406-2417. [CrossRef]
- Tippmann, S., Chen, Y., Siewers, V., and Nielsen, J. (2013). From flavors and pharmaceuticals to advanced biofuels: production of isoprenoids in Saccharomyces cerevisiae. Biotechnol J 8, 1435-1444. [CrossRef]
- Trapp, S. C., and Croteau, R. B. (2001). Genomic organization of plant terpene synthases and molecular evolutionary implications. Genetics 158, 811-832. [CrossRef]
- Traverse, K. K. F., Breselge, S., Trautman, J. G., Dee, A., Wang, J., Childs, K. L., and Lee-Parsons, C. W. T. (2024). Characterization of the ZCTs, a subgroup of Cys2-His2 zinc finger transcription factors regulating alkaloid biosynthesis in Catharanthus roseus. Plant Cell Rep 43, 209. [CrossRef]
- Uetz, P., Melnik, S., Grünwald-Gruber, C., Strasser, R., and Stoger, E. (2022). CRISPR/Cas9-mediated knockout of a prolyl-4-hydroxylase subfamily in Nicotiana benthamiana using DsRed2 for plant selection. Biotechnol J 17, e2100698. [CrossRef]
- Van den Broeck, L., Gordon, M., Inzé, D., Williams, C., and Sozzani, R. (2020). Gene regulatory network inference: connecting plant biology and mathematical modeling. Front Genet 11, 457. [CrossRef]
- Verma, V., Kumar, A., Partap, M., Thakur, M., and Bhargava, B. (2023). CRISPR-Cas: A robust technology for enhancing consumer-preferred commercial traits in crops. Front Plant Sci 14, 1122940. [CrossRef]
- Volk, M. J., Tran, V. G., Tan, S.-I., Mishra, S., Fatma, Z., Boob, A., Li, H., Xue, P., Martin, T. A., and Zhao, H. (2022). Metabolic engineering: methodologies and applications. Chem Rev 123, 5521-5570. [CrossRef]
- Walker, K., and Croteau, R. (2000). Molecular cloning of a 10-deacetylbaccatin III-10-O-acetyl transferase cDNA from Taxus and functional expression in Escherichia coli. Proc Natl Acad Sci USA 97, 583-587. [CrossRef]
- Wan, L., Huang, Q., Li, C., Yu, H., Tan, G., Wei, S., El-Sappah, A. H., Sooranna, S., Zhang, K., and Pan, L. (2024). Integrated metabolome and transcriptome analysis identifies candidate genes involved in triterpenoid saponin biosynthesis in leaves of Centella asiatica (L.) Urban. Front Plant Sci 14, 1295186. [CrossRef]
- Wang, H.-m., and Zu, Y.-g. (2007). Agrobacterium-mediated genetic transformation of Camptotheca acuminata. J For Res 18, 316-318. [CrossRef]
- Wang, H., Han, J., Kanagarajan, S., Lundgren, A., and Brodelius, P. E. (2013). Studies on the expression of sesquiterpene synthases using promoter-β-glucuronidase fusions in transgenic Artemisia annua L. PLoS ONE 8, e80643. [CrossRef]
- Wang, H., Wang, P., Wang, F., Chen, H., Chen, L., Hu, Y., and Liu, Y. (2024). Integrated HS-GC–IMS and UPLC-Q-Orbitrap HRMS-based metabolomics revealed the characteristics and differential volatile and nonvolatile metabolites of different citrus peels. Curr Res Food Sci 8, 100755. [CrossRef]
- Wang, J., Gao, W.-Y., Zhang, J., Zuo, B.-M., Zhang, L.-M., and Huang, L.-Q. (2012). Production of ginsenoside and polysaccharide by two-stage cultivation of Panax quinquefolium L. cells. In Vitro Cellular & Developmental Biology-Plant 48, 107-112. [CrossRef]
- Wang, J. W., Tian, H., Yu, X., and Zheng, L. P. (2017). Glucose-6-phosphate dehydrogenase plays critical role in artemisinin production of Artemisia annua under salt stress. Biol Plant 61, 529-539. [CrossRef]
- Wani, K. I., Choudhary, S., Zehra, A., Naeem, M., Weathers, P., and Aftab, T. (2021). Enhancing artemisinin content in and delivery from Artemisia annua: a review of alternative, classical, and transgenic approaches. Planta 254, 29. [CrossRef]
- Wijma, H. J., Floor, R. J., Bjelic, S., Marrink, S. J., Baker, D., and Janssen, D. B. Enantioselective enzymes by computational design and screening. [CrossRef]
- Wu, S., Jiang, Z., Kempinski, C., Eric Nybo, S., Husodo, S., Williams, R., and Chappell, J. (2012). Engineering triterpene metabolism in tobacco. Planta 236, 867-877. [CrossRef]
- Xia, K., Sun, H.-X., Li, J., Li, J., Zhao, Y., Chen, R., Liu, G., Chen, Z., Yin, R., and Hao, S. (2021). Single-cell Stereo-seq enables cell type-specific spatial transcriptome characterization in Arabidopsis leaves. bioRxiv, 2021-2010. [CrossRef]
- Xu, J., and Zhang, N. (2014). On the way to commercializing plant cell culture platform for biopharmaceuticals: present status and prospect. Pharm Bioprocess 2, 499. [CrossRef]
- Yadav, A. K., Singh, S., Dhyani, D., and Ahuja, P. S. (2011). A review on the improvement of stevia [Stevia rebaudiana (Bertoni)]. Can J Plant Sci 91, 1-27. [CrossRef]
- Yang, Z., Li, X., Yang, L., Peng, S., Song, W., Lin, Y., Xiang, G., Li, Y., Ye, S., and Ma, C. (2023). Comparative genomics reveals the diversification of triterpenoid biosynthesis and origin of ocotillol-type triterpenes in Panax. Plant Commun 4. [CrossRef]
- Ye, X., Al-Babili, S., Kloti, A., Zhang, J., Lucca, P., Beyer, P., and Potrykus, I. (2000). Engineering the provitamin A (β-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 287, 303-305. [CrossRef]
- Yi, X., Wang, X., Wu, L., Wang, M., Yang, L., Liu, X., Chen, S., and Shi, Y. (2022). Integrated analysis of basic helix loop helix transcription factor family and targeted terpenoids reveals candidate AarbHLH genes involved in terpenoid biosynthesis in Artemisia argyi. Front Plant Sci 12. [CrossRef]
- Yıldırım, K., Miladinović, D., Sweet, J., Akin, M., Galović, V., Kavas, M., Zlatković, M., and de Andrade, E. (2023). Genome editing for healthy crops: traits, tools and impacts. Front Plant Sci 14, 1231013. [CrossRef]
- Yu, F., and De Luca, V. (2013). ATP-binding cassette transporter controls leaf surface secretion of anticancer drug components in Catharanthus roseus. Proc Natl Acad Sci USA 110, 15830-15835. [CrossRef]
- Zhang, C., and Hong, K. (2020). Production of terpenoids by synthetic biology approaches. Front Bioeng Biotechnol 8, 347. [CrossRef]
- Zhang, C., Li, M., Zhao, G.-R., and Lu, W. (2019). Alpha-Terpineol production from an engineered Saccharomyces cerevisiae cell factory. Microb Cell Fact 18, 160. [CrossRef]
- Zhang, L., Ding, R., Chai, Y., Bonfill, M., Moyano, E., Oksman-Caldentey, K.-M., Xu, T., Pi, Y., Wang, Z., and Zhang, H. (2004). Engineering tropane biosynthetic pathway in Hyoscyamus niger hairy root cultures. Proc Natl Acad Sci USA 101, 6786-6791. [CrossRef]
- Zhang, M., Jin, X., Chen, Y., Wei, M., Liao, W., Zhao, S., Fu, C., and Yu, L. (2018). TcMYC2a, a basic helix-loop-helix transcription factor, transduces JA-signals and regulates taxol biosynthesis in Taxus chinensis. Front Plant Sci 9, 863. [CrossRef]
- Zhang, P., Du, H., Wang, J., Pu, Y., Yang, C., Yan, R., Yang, H., Cheng, H., and Yu, D. (2020). Multiplex CRISPR/Cas9-mediated metabolic engineering increases soya bean isoflavone content and resistance to soya bean mosaic virus. Plant Biotechnol J 18, 1384-1395. [CrossRef]
- Zhang, Y., Cao, X., Wang, J., and Tang, F. (2022). Enhancement of linalool production in Saccharomyces cerevisiae by utilizing isopentenol utilization pathway. Microb Cell Fact 21, 212. [CrossRef]
- Zhang, Y., Cheng, Y., Fang, H., Roberts, N., Zhang, L., Vakulskas, C. A., Niedz, R. P., Culver, J. N., and Qi, Y. (2022). Highly Efficient Genome Editing in Plant Protoplasts by Ribonucleoprotein Delivery of CRISPR-Cas12a Nucleases. Front Genome Ed 4, 780238. [CrossRef]
- Zhang, Y., Gao, J., Ma, L., Tu, L., Hu, T., Wu, X., Su, P., Zhao, Y., Liu, Y., and Li, D. (2023). Tandemly duplicated CYP82Ds catalyze 14-hydroxylation in triptolide biosynthesis and precursor production in Saccharomyces cerevisiae. Nat Commun 14, 875. [CrossRef]
- Zhang, Y., Ni, X., Fu, X., Taheri, A., Zhang, W., Liu, P., Liu, H., Li, L., Wang, Y., and Tang, K. (2025). Comprehensive analysis of SPL gene family and miR156a/SPLs in the regulation of terpenoid indole alkaloid biosynthesis in Catharanthus roseus L. BMC Plant Biol 25, 817. [CrossRef]
- Zhang, Y., Zhang, F., Jin, L., Zhang, T., Pu, X., Qiu, B., and Li, G. (2023). A chromosome-level genome assembly of the Knoxia roxburghii (Rubiaceae). Sci Data 10, 803. [CrossRef]
- Zhang, Z., Wu, Q. Y., Ge, Y., Huang, Z. Y., Hong, R., Li, A., Xu, J. H., and Yu, H. L. (2023). Hydroxylases involved in terpenoid biosynthesis: a review. Bioresour Bioprocess 10, 39. [CrossRef]
- Zhao, E. M., Suek, N., Wilson, M. Z., Dine, E., Pannucci, N. L., Gitai, Z., Avalos, J. L., and Toettcher, J. E. (2019). Light-based control of metabolic flux through assembly of synthetic organelles. Nat Chem Biol 15, 589-597. [CrossRef]
- Zhao, X., Ge, W., and Miao, Z. (2024). Integrative metabolomic and transcriptomic analyses reveals the accumulation patterns of key metabolites associated with flavonoids and terpenoids of Gynostemma pentaphyllum (Thunb.) Makino. Sci Rep 14, 8644. [CrossRef]
- Zheng, S., Zeng, T., Li, C., Chen, B., Coley, C. W., Yang, Y., and Wu, R. (2022). Deep learning driven biosynthetic pathways navigation for natural products with BioNavi-NP. Nat Commun 13, 3342. [CrossRef]
- Zhou, T., Bai, G., Hu, Y., Ruhsam, M., Yang, Y., and Zhao, Y. (2022). De novo genome assembly of the medicinal plant Gentiana macrophylla provides insights into the genomic evolution and biosynthesis of iridoids. DNA Res 29, dsac034. [CrossRef]
- Zhu, F., Wen, W., Cheng, Y., Alseekh, S., and Fernie, A. R. (2023). Integrating multiomics data accelerates elucidation of plant primary and secondary metabolic pathways. Abiotech 4, 47-56. [CrossRef]
- Zi, J., Mafu, S., and Peters, R. J. (2014). To gibberellins and beyond! Surveying the evolution of (di) terpenoid metabolism. Annu Rev Plant Biol 65, 259-286. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



