Submitted:
01 August 2025
Posted:
01 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Phylogenetic Relationships within Noccaea (Formerly Thlaspi) Species
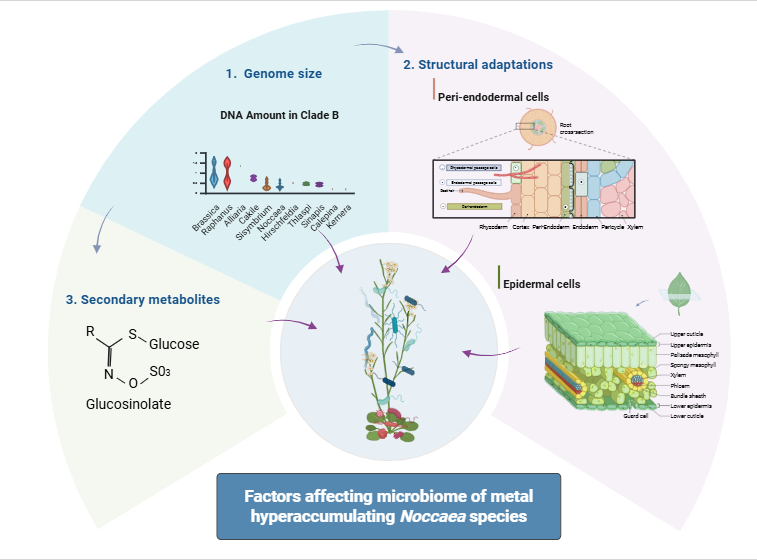
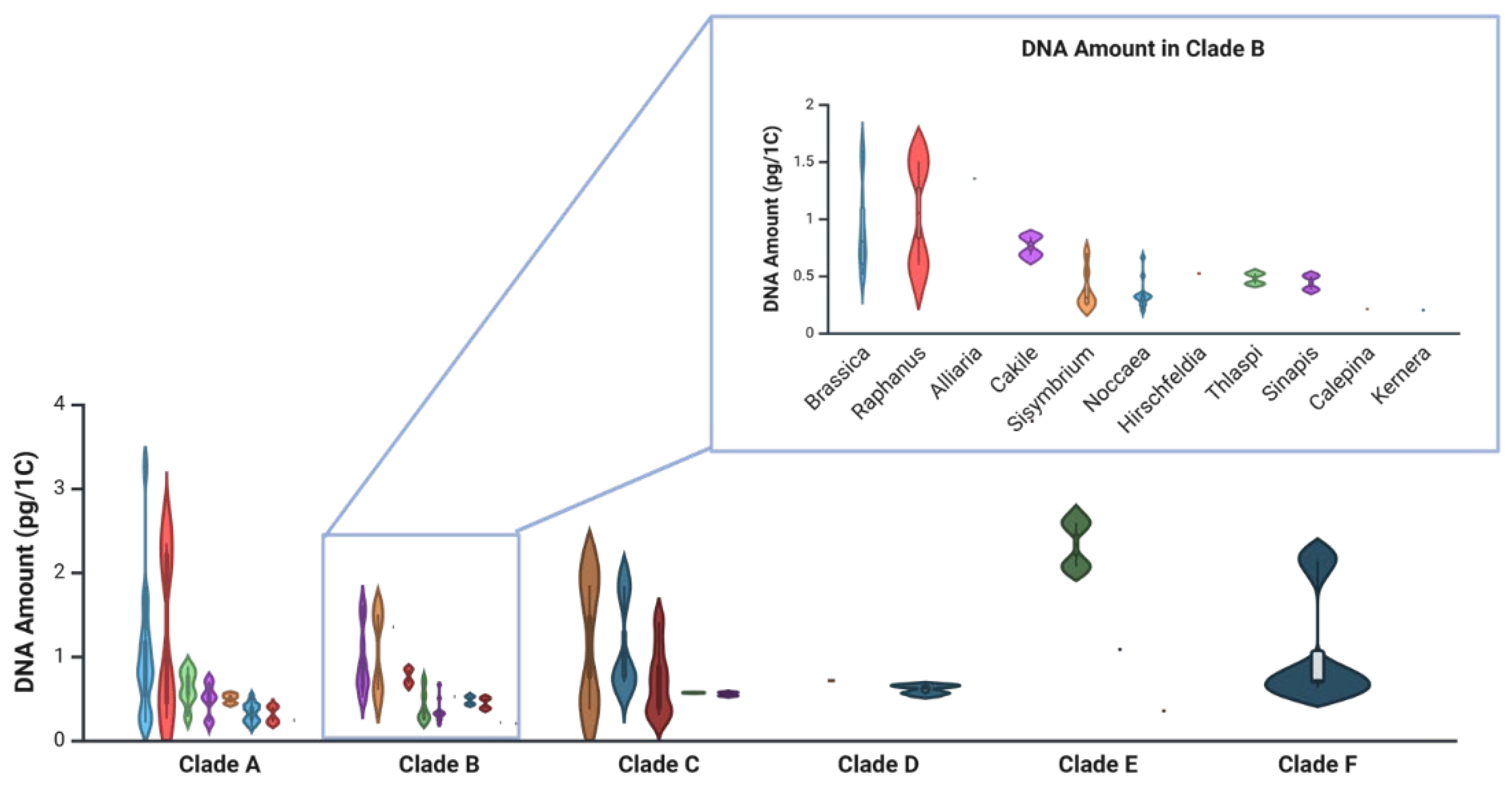
3. The Large Genome Size Constraint Hypothesis
4. Genetic Requirements of Metal Hyperaccumulation and Impacts on the Microbiome in Noccaea Species
5. Bioimaging Techniques Reveal Adaptation Mechanisms in the Leaves of Noccaea Species
6. Specific Features in the Roots of Noccaea Species Affect Microbial Composition and Metal Uptake
7. Glucosinolates
8. Conclusions and Further Directions
Author Contributions
Funding
Data Availability Statement
Acknowledgements
Conflicts of Interest
Abbreviations
| AM | Arbuscular mycorrhiza |
| DNA | Deoxyribonucleic Acid |
| FTIR | Fourier Transform Infrared Spectroscopy |
| HMA4 | Heavy Metal ATPase 4 |
| LA-ICP-MS | Laser Ablation Inductively Coupled Plasma Mass Spectrometry |
| LEXRF | Low-Energy X-Ray-Fluorescence |
| NAS2 | Nicotianamine Synthase 2 |
| NcHMA4 | Noccaea caerulescens Heavy Metal ATPase 4 |
| NcMTP1 | Noccaea caerulescens Metal Tolerance Protein 1 |
| PIXE | Particle-Induced X-ray Emission |
| SR-μ-XRF | Synchrotron micro X-Ray-Fluorescence |
| YSL3 | Yellow Stripe-Like 3 |
| XCT | X-ray computed tomography |
| WDG | whole-genome duplication |
| ZIFL1 | Zinc-Induced Facilitator-Like 1 |
Appendix A
Appendix A.1
| No | Clade | Genus | Species | DNA Amount |
Original Reference See https://ww2.bgbm.org/EuroPlusMed/query.asp |
Clade average DNA Amount |
| 1C (pg) | 1C (pg) | |||||
| 1 | Clade A | Capsella | rubella | 0.22 | Lysák et al.,2009 | |
| 2 | Clade A | Capsella | bursa-pastoris | 0.4 | Lysák et al.,2009 | |
| 3 | Clade A | Pachycladon | exilis | 0.44 | Lysák et al.,2009 | |
| 4 | Clade A | Pachycladon | fastigiata | 0.51 | Lysák et al.,2009 | |
| 5 | Clade A | Pachycladon | novae-zelandiae | 0.55 | Lysák et al.,2009 | |
| 6 | Clade A | Turritis | glabra | 0.24 | Lysák et al.,2009 | |
| 7 | Clade A | Arabidopsis | thaliana | 0.16 | Bennett et al.,2003 | |
| 8 | Clade A | Arabidopsis | arenosa | 0.2 | Lysák et al.,2009 | |
| 9 | Clade A | Arabidopsis | neglecta | 0.2 | Lysák et al.,2009 | |
| 10 | Clade A | Arabidopsis | halleri | 0.24 | Lysák et al.,2009 | |
| 11 | Clade A | Arabidopsis | korshynskyi | 0.25 | Nagl et al.,1983 | |
| 12 | Clade A | Arabidopsis | lyrata | 0.25 | Lysák et al.,2009 | |
| 13 | Clade A | Arabidopsis | cebennensis | 0.29 | Lysák et al.,2009 | |
| 14 | Clade A | Arabidopsis | pumila | 0.34 | Houben et al.,2003 | |
| 15 | Clade A | Arabidopsis | suecica | 0.35 | Lysák et al.,2009 | |
| 16 | Clade A | Arabidopsis | arenosa | 0.39 | Lysák et al.,2009 | |
| 17 | Clade A | Arabidopsis | wallichii | 0.4 | Houben et al.,2003 | |
| 18 | Clade A | Arabidopsis | neglecta | 0.4 | Lysák et al.,2009 | |
| 19 | Clade A | Arabidopsis | thaliana | 0.44 | Schmuths et al.,2004 | |
| 20 | Clade A | Arabidopsis | lyrata | 0.45 | Dart et al.,2004 | |
| 21 | Clade A | Arabidopsis | kamchatica | 0.52 | Wolf et al.,2014 | |
| 22 | Clade A | Physaria | gracilis | 0.26 | Lysák et al.,2009 | |
| 23 | Clade A | Physaria | ovalifolia | 0.43 | Lysák et al.,2009 | |
| 24 | Clade A | Physaria | arctica | 0.69 | Lysák et al.,2009 | |
| 25 | Clade A | Physaria | didymocarpa | 2.23 | Lysák et al.,2009 | |
| 26 | Clade A | Physaria | bellii | 2.34 | Lysák et al.,2009 | |
| 27 | Clade A | Erysimum | sylvestre | 0.28 | Vidic et al.,2009 | |
| 28 | Clade A | Erysimum | duriaei | 0.47 | Loureiro et al.,2013 | |
| 29 | Clade A | Erysimum | scoparium | 0.54 | Suda et al.,2003 | |
| 30 | Clade A | Erysimum | bicolor | 0.58 | Suda et al.,2003 | |
| 31 | Clade A | Erysimum | goniocaulon | 0.69 | Bou Dagher-Kharrat et al.,2013 | |
| 32 | Clade A | Erysimum | bicolor | 0.76 | Lysák et al.,2009 | |
| 33 | Clade A | Erysimum | cheiranthoides | 0.83 | Lysák et al.,2009 | |
| 34 | Clade A | Erysimum | diffusum | 0.88 | Pustahija et al.,2013 | |
| 35 | Clade A | Rorippa | lipizensis | 0.22 | Vallès et al.,2014 | |
| 36 | Clade A | Rorippa | sylvestris | 0.48 | Pustahija et al.,2013 | |
| 37 | Clade A | Rorippa | palustris | 0.54 | Lysák et al.,2009 | |
| 38 | Clade A | Rorippa | nasturtium-aquaticum | 0.7 | Kenton and Owens,1988 | |
| 39 | Clade A | Cardamine | impatiens | 0.21 | Johnston et al.,2005 | |
| 40 | Clade A | Cardamine | hirsuta | 0.23 | Johnston et al.,2005 | |
| 41 | Clade A | Cardamine | amara | 0.24 | Hanson et al.,2002 | |
| 42 | Clade A | Cardamine | glauca | 0.28 | Pustahija et al.,2013 | |
| 43 | Clade A | Cardamine | chelidonia | 0.36 | Kubešová et al.,2010 | |
| 44 | Clade A | Cardamine | schinziana | 0.68 | Marhold et al.,2010 | |
| 45 | Clade A | Cardamine | yezoensis | 0.7 | Marhold et al.,2010 | |
| 46 | Clade A | Cardamine | amaraeiformis | 0.71 | Marhold et al.,2010 | |
| 47 | Clade A | Cardamine | valida | 0.71 | Marhold et al.,2010 | |
| 48 | Clade A | Cardamine | yezoensis | 0.87 | Marhold et al.,2010 | |
| 49 | Clade A | Cardamine | flexuosa | 0.9 | Mowforth and Grime,1989 | |
| 50 | Clade A | Cardamine | yezoensis | 0.99 | Marhold et al.,2010 | |
| 51 | Clade A | Cardamine | schinziana | 1 | Marhold et al.,2010 | |
| 52 | Clade A | Cardamine | torrentis | 1.13 | Marhold et al.,2010 | |
| 53 | Clade A | Cardamine | yezoensis | 1.24 | Marhold et al.,2010 | |
| 54 | Clade A | Cardamine | asarifolia | 1.34 | Lihova et al.,2006 | |
| 55 | Clade A | Cardamine | diphylla | 1.62 | Sonnier,2016 | |
| 56 | Clade A | Cardamine | pratensis | 1.70 | Band,1984 | |
| 57 | Clade A | Cardamine | concatenata | 3.25 | Bai et al.,2012 | 0.67 |
| 1 | Clade B | Raphanus | sativus | 0.6 | Dolezel et al.,1992 | |
| 2 | Clade B | Raphanus | sativus | 1.50 | Olszewska and Osiecka,1983 | |
| 3 | Clade B | Hirschfeldia | incana | 0.52 | Lysák et al.,2009 | |
| 4 | Clade B | Brassica | hirta | 0.51 | Arumuganathan and Earle,1991 | |
| 5 | Clade B | Brassica | tournefortii | 0.6 | Nagpal et al.,1996 | |
| 6 | Clade B | Brassica | campestris | 0.6 | Bennett et al.,1976 | |
| 7 | Clade B | Brassica | nigra | 0.8 | Verma and Rees,1974 | |
| 8 | Clade B | Brassica | oleracea | 0.8 | Olszewska and Osiecka,1983 | |
| 9 | Clade B | Brassica | rapa | 0.8 | Ingle et al.,1975 | |
| 10 | Clade B | Brassica | napus | 1.10 | Greilhuber,1988 | |
| 11 | Clade B | Brassica | juncea | 1.50 | Verma and Rees,1974 | |
| 12 | Clade B | Brassica | carinata | 1.60 | Verma and Rees,1974 | |
| 13 | Clade B | Sinapis | arvensis | 0.38 | Arumuganathan and Earle,1991 | |
| 14 | Clade B | Sinapis | alba | 0.50 | Bennett MD, Smith JB, Heslop-Harrison,1982 | |
| 15 | Clade B | Cakile | maritima | 0.68 | Lysák et al.,2009 | |
| 16 | Clade B | Cakile | edentula | 0.84 | Bai et al.,2012 | |
| 17 | Clade B | Sisymbrium | officinale | 0.24 | Lysák et al.,2009 | |
| 18 | Clade B | Sisymbrium | loeselii | 0.24 | Kubešová et al.,2010 | |
| 19 | Clade B | Sisymbrium | altissimum | 0.26 | Kubešová et al.,2010 | |
| 20 | Clade B | Sisymbrium | orientale | 0.31 | Johnston et al.,2005 | |
| 21 | Clade B | Sisymbrium | austriacum | 0.36 | Lysák et al.,2009 | |
| 22 | Clade B | Sisymbrium | irio | 0.53 | Johnston et al.,2005 | |
| 23 | Clade B | Sisymbrium | strictissimum | 0.7 | Kubešová et al.,2010 | |
| 24 | Clade B | Thlaspi | ceratocarpum | 0.43 | Lysák et al.,2009 | |
| 25 | Clade B | Thlaspi | arvense | 0.52 | Lysák et al.,2009 | |
| 26 | Clade B | Alliaria | petiolata | 1.35 | Barow & Meister,2003 | |
| 27 | Clade B | Calepina | irregularis | 0.21 | Lysák et al.,2009 | |
| 28 | Clade B | Noccaea | alpestris | 0.24 | Lysák et al.,2009 | |
| 29 | Clade B | Noccaea | alpestris | 0.2 | Band,1984 | |
| 30 | Clade B | Noccaea | caerulescens | 0.34 | Peer et al.,2006 | |
| 31 | Clade B | Noccaea | montana | 0.29 | Peer et al.,2006 | |
| 32 | Clade B | Noccaea | goesingensis | 0.5 | Siljak-Yakovlev et al.,2010 | |
| 33 | Clade B | Noccaea | oxyceras | 0.34 | Peer et al.,2003 | |
| 34 | Clade B | Noccaea | praecox | 0.26 | Temsch et al.,2010 | |
| 35 | Clade B | Noccaea | perfoliata | 0.31 | Peer et al.,2006 | |
| 36 | Clade B | Noccaea | rosularis | 0.32 | Peer et al.,2003 | |
| 37 | Clade B | Noccaea | tymphaea | 0.32 | Peer et al.,2006 | |
| 38 | Clade B | Noccaea | tymphaea | 0.66 | Peer et al.,2006 | |
| 39 | Clade B | Noccaea | violascens | 0.31 | Peer et al.,2003 | |
| 40 | Clade B | Kernera | saxatilis | 0.2 | Lysák et al.,2009 | 0.57 |
| 1 | Clade C | Iberis | sempervirens | 0.56 | Lysák et al.,2009 | |
| 2 | Clade C | Iberis | gibraltarica | 0.57 | Lysák et al.,2009 | |
| 3 | Clade C | Lobularia | libyaca | 0.53 | Lysák et al.,2009 | |
| 4 | Clade C | Lobularia | canariensis | 0.57 | Suda et al.,2003 | |
| 5 | Clade C | Cochlearia | aucheri | 0.3 | Peer et al.,2003 | |
| 6 | Clade C | Cochlearia | sempervivum | 0.33 | Peer et al.,2003 | |
| 7 | Clade C | Cochlearia | pyrenaica | 0.4 | Krisai and Greilhuber,1997 | |
| 8 | Clade C | Cochlearia | danica | 0.7 | Lysák et al.,2009 | |
| 9 | Clade C | Cochlearia | officinalis | 0.75 | Lysák et al.,2009 | |
| 10 | Clade C | Cochlearia | tatrae | 1.04 | Kochjarova et al.,2006 | |
| 11 | Clade C | Cochlearia | borzaeana | 1.41 | Kochjarova et al.,2006 | |
| 12 | Clade C | Lunaria | rediviva | 0.37 | Pustahija et al.,2013 | |
| 13 | Clade C | Lunaria | biennis | 1.85 | Zonneveld et al.,2005 | |
| 14 | Clade C | Biscutella | auriculata | 0.69 | Lysák et al.,2009 | |
| 15 | Clade C | Biscutella | didyma | 0.79 | Peer et al.,2003 | |
| 16 | Clade C | Biscutella | laevigata | 1.83 | Temsch et al.,2010 | 0.79 |
| 1 | Clade D | Alyssum | saxatile | 0.65 | Lysák et al.,2009 | |
| 2 | Clade D | Alyssum | markgrafii | 0.54 | Pustahija et al.,2013 | |
| 3 | Clade D | Alyssum | murale | 0.58 | Siljak-Yakovlev et al.,2010 | |
| 4 | Clade D | Alyssum | saxatile | 0.65 | Lysák et al.,2009 | |
| 5 | Clade D | Berteroa | incana | 0.71 | Lysák et al.,2009 | 0.63 |
| 1 | Clade E | Parrya | nudicaulis | 1.08 | Lysák et al.,2009 | |
| 2 | Clade E | Chorispora | tenella | 0.35 | Lysák et al.,2009 | |
| 3 | Clade E | Bunias | erucago | 2.07 | Greilhuber and Obermayer,1999 | |
| 4 | Clade E | Bunias | orientalis | 2.59 | Greilhuber and Obermayer,1999 | |
| 5 | Clade E | Hesperis | matronalis | 3.8 | Kubešová et al.,2010 | 1.98 |
| 1 | Clade F | Aethionema | saxatile | 0.62 | Pustahija et al.,2013 | |
| 2 | Clade F | Aethionema | grandiflorum | 0.71 | Lysák et al.,2009 | |
| 3 | Clade F | Aethionema | schistosum | 0.71 | Lysák et al.,2009 | |
| 4 | Clade F | Aethionema | cordifolium | 2.14 | Bou Dagher-Kharrat et al.,2013 | 1.05 |
References
- Turner, T.R.; James, E.K.; Poole, P.S. The Plant Microbiome. Genome Biol 2013, 14, 1–10. [CrossRef]
- Trivedi, P.; Leach, J.E.; Tringe, S.G.; Sa, T.; Singh, B.K. Plant–Microbiome Interactions: From Community Assembly to Plant Health. Nature Reviews Microbiology 2020 18:11 2020, 18, 607–621. [CrossRef]
- Fitzpatrick, C.R.; Salas-González, I.; Conway, J.M.; Finkel, O.M.; Gilbert, S.; Russ, D.; Teixeira, P.J.P.L.; Dangl, J.L. The Plant Microbiome: From Ecology to Reductionism and Beyond. Annu Rev Microbiol 2020, 74, 81–100. [CrossRef]
- Reeves, R.D.; Baker, A.J.M.; Jaffré, T.; Erskine, P.D.; Echevarria, G.; van der Ent, A. A Global Database for Plants That Hyperaccumulate Metal and Metalloid Trace Elements. New Phytologist 2018, 218, 407–411. [CrossRef]
- Al-Shehbaz, I.A.; Beilstein, M.A.; Kellogg, E.A. Systematics and Phylogeny of the Brassicaceae (Cruciferae): An Overview. Plant Systematics and Evolution 2006, 259, 89–120. [CrossRef]
- Al-Shehbaz, I.A. A Synopsis of the Genus Noccaea (Coluteocarpeae, Brassicaceae). Harv Pap Bot 2014, 19, 25–51. [CrossRef]
- Franzke, A.; German, D.; Al-Shehbaz, I.A.; Mummenhoff, K. Arabidopsis Family Ties: Molecular Phylogeny and Age Estimates in Brassicaceae. Taxon 2009, 58, 425–437. [CrossRef]
- Hohmann, N.; Wolf, E.M.; Lysak, M.A.; Koch, M.A. A Time-Calibrated Road Map of Brassicaceae Species Radiation and Evolutionary History. Plant Cell 2015, 27, 2770–2784. [CrossRef]
- Mandáková, T.; Joly, S.; Krzywinski, M.; Mummenhoff, K.; Lysaka, M.A. Fast Diploidization in Close Mesopolyploid Relatives of Arabidopsis. Plant Cell 2010, 22, 2277–2290. [CrossRef]
- Kagale, S.; Robinson, S.J.; Nixon, J.; Xiao, R.; Huebert, T.; Condie, J.; Kessler, D.; Clarke, W.E.; Edger, P.P.; Links, M.G.; et al. Polyploid Evolution of the Brassicaceae during the Cenozoic Era. Plant Cell 2014, 26, 2777–2791. [CrossRef]
- Van De Peer, Y.; Mizrachi, E.; Marchal, K. The Evolutionary Significance of Polyploidy. Nature Reviews Genetics 2017 18:7 2017, 18, 411–424. [CrossRef]
- Huang, C.H.; Sun, R.; Hu, Y.; Zeng, L.; Zhang, N.; Cai, L.; Zhang, Q.; Koch, M.A.; Al-Shehbaz, I.; Edger, P.P.; et al. Resolution of Brassicaceae Phylogeny Using Nuclear Genes Uncovers Nested Radiations and Supports Convergent Morphological Evolution. Mol Biol Evol 2016, 33, 394–412. [CrossRef]
- Zhang, H.; Liu, H.; Han, X. Traits-Based Approach: Leveraging Genome Size in Plant–Microbe Interactions. Trends Microbiol 2024, 32, 333–341. [CrossRef]
- Sotelo, T.; Lema, M.; Soengas, P.; Cartea, M.E.; Velasco, P. In Vitro Activity of Glucosinolates and Their Degradation Products against Brassica-Pathogenic Bacteria and Fungi. Appl Environ Microbiol 2015, 81, 432–440. [CrossRef]
- Abdel-Massih, R.M.; Debs, E.; Othman, L.; Attieh, J.; Cabrerizo, F.M. Glucosinolates, a Natural Chemical Arsenal: More to Tell than the Myrosinase Story. Front Microbiol 2023, 14, 1130208. [CrossRef]
- Kováč, J.; Lux, A.; Soukup, M.; Weidinger, M.; Gruber, D.; Lichtscheidl, I.; Vaculík, M. A New Insight on Structural and Some Functional Aspects of Peri-Endodermal Thickenings, a Specific Layer in Noccaea Caerulescens Roots. Ann Bot 2020, 126, 423–434. [CrossRef]
- Trautwig, A.N.; Jackson, M.R.; Kivlin, S.N.; Stinson, K.A. Reviewing Ecological Implications of Mycorrhizal Fungal Interactions in the Brassicaceae. Front Plant Sci 2023, 14, 1269815. [CrossRef]
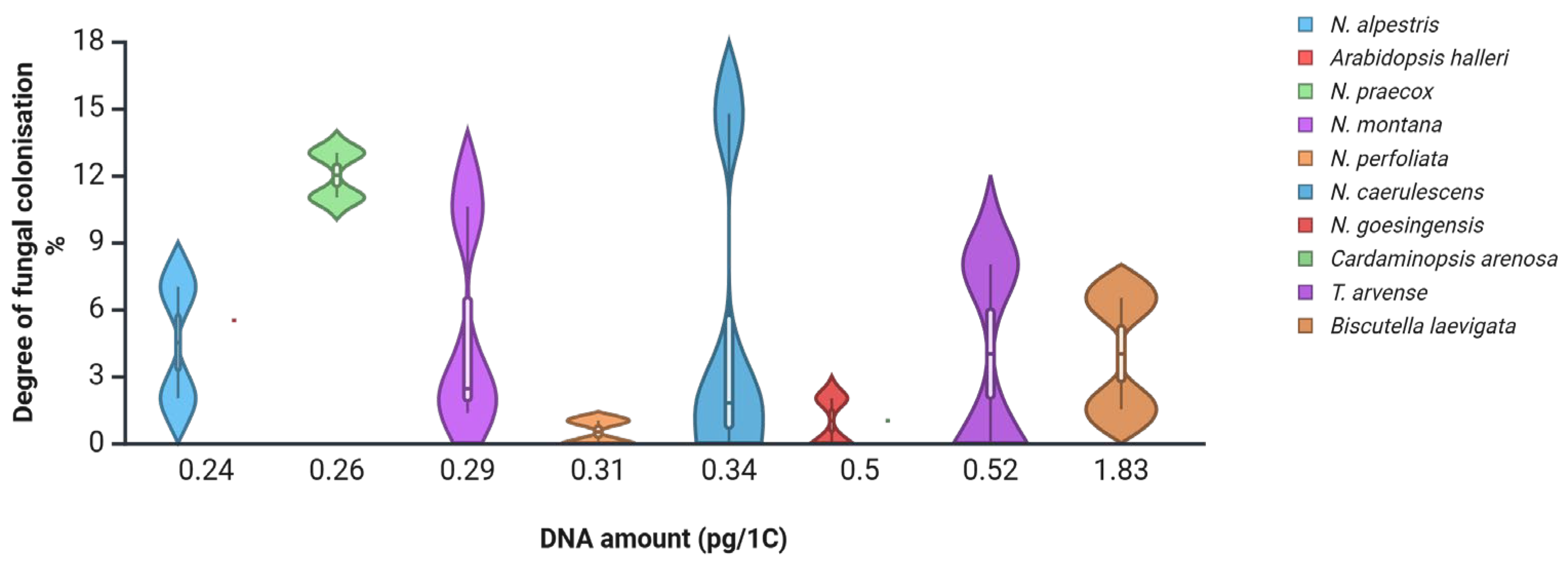
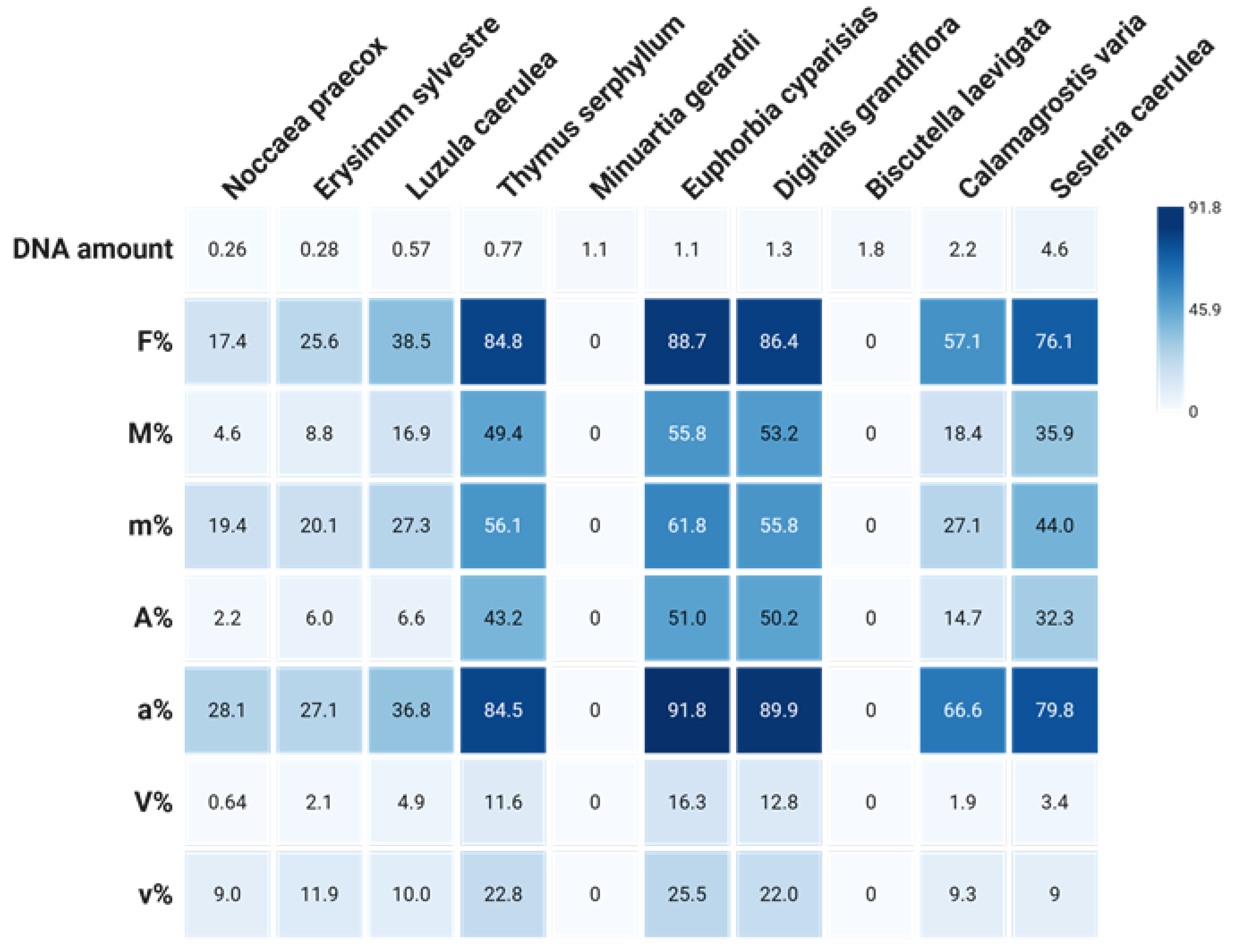
- Pongrac, P.; Sonjak, S.; Vogel-Mikuš, K.; Kump, P.; Nečemer, M.; Regvar, M. Roots of Metal Hyperaccumulating Population of Thlaspi Praecox (Brassicaceae) Harbour Arbuscular Mycorrhizal and Other Fungi under Experimental Conditions. Int J Phytoremediation 2009, 11, 347–359. [CrossRef]
- Vogel-Mikuš, K.; Drobne, D.; Regvar, M. Zn, Cd and Pb Accumulation and Arbuscular Mycorrhizal Colonisation of Pennycress Thlaspi Praecox Wulf. (Brassicaceae) from the Vicinity of a Lead Mine and Smelter in Slovenia. Environmental Pollution 2005, 133, 233–242. [CrossRef]
- Regvar, M.; Vogel-Mikuš, K.; Kugonič, N.; Turk, B.; Batič, F. Vegetational and Mycorrhizal Successions at a Metal Polluted Site: Indications for the Direction of Phytostabilisation? Environmental Pollution 2006, 144, 976–984. [CrossRef]
- Vogel-Mikuš, K.; Pongrac, P.; Kump, P.; Nečemer, M.; Regvar, M. Colonisation of a Zn, Cd and Pb Hyperaccumulator Thlaspi Praecox Wulfen with Indigenous Arbuscular Mycorrhizal Fungal Mixture Induces Changes in Heavy Metal and Nutrient Uptake. Environmental Pollution 2006, 139, 362–371. [CrossRef]
- Zunk, K.; Mummenhoff, K.; Koch, M.; Hurka, H. Phylogenetic Relationships of Thlaspi s.I. (Subtribe Thlaspidinae, Lepidieae) and Allied Genera Based on Chloroplast DNA Restriction- Site Variation. Theoretical and Applied Genetics 1996, 92, 375–381. [CrossRef]
- Koch, M.; Mummenhoff, K. Thlaspi s.Str. (Brassicaceae) versus Thlaspi s.l.: Morphological and Anatomical Characters in the Light of ITS NrDNA Sequence Data. Plant Systematics and Evolution 2001, 227, 209–225. [CrossRef]
- Krämer, U. Metal Hyperaccumulation in Plants. Annu Rev Plant Biol 2010, 61, 517–534. [CrossRef]
- Fasani, E.; Zamboni, A.; Sorio, D.; Furini, A.; DalCorso, G. Metal Interactions in the Ni Hyperaccumulating Population of Noccaea Caerulescens Monte Prinzera. Biology (Basel) 2023, 12, 1537. [CrossRef]
- Vogel-Mikuš, K.; Regvar, M.; Mesjasz-Przybyłowicz, J.; Przybyłowicz, W.J.; Simčič, J.; Pelicon, P.; Budnar, M. Spatial Distribution of Cadmium in Leaves of Metal Hyperaccumulating Thlaspi Praecox Using Micro-PIXE. New Phytologist 2008, 179, 712–721. [CrossRef]
- Koren, Š.; Arčon, I.; Kump, P.; Nečemer, M.; Vogel-Mikuš, K. Influence of CdCl2 and CdSO4 Supplementation on Cd Distribution and Ligand Environment in Leaves of the Cd Hyperaccumulator Noccaea (Thlaspi) Praecox. Plant Soil 2013, 370, 125–148. [CrossRef]
- Likar, M.; Pongrac, P.; Vogel-Mikuš, K.; Regvar, M. Molecular Diversity and Metal Accumulation of Different Thlaspi Praecox Populations from Slovenia. Plant Soil 2010, 330, 195–205. [CrossRef]
- Koch, M.; Al-Shehbaz, I.A. Taxonomic and Phylogenetic Evaluation of the American “Thlaspi” Species: Identity and Relationship to the Eurasian Genus Noccaea (Brassicaceae). Syst Bot 2004, 29, 375–384. [CrossRef]
- Mandáková, T.; Li, Z.; Barker, M.S.; Lysak, M.A. Diverse Genome Organization Following 13 Independent Mesopolyploid Events in Brassicaceae Contrasts with Convergent Patterns of Gene Retention. The Plant Journal 2017, 91, 3–21. [CrossRef]
- Lysak, M.A.; Koch, M.A.; Pecinka, A.; Schubert, I. Chromosome Triplication Found across the Tribe Brassiceae. Genome Res 2005, 15, 516–525. [CrossRef]
- Vision, T.J.; Brown, D.G.; Tanksley, S.D. The Origins of Genomic Duplications in Arabidopsis. Science (1979) 2000, 290, 2114–2117. [CrossRef]
- Johnston, J.S.; Pepper, A.E.; Hall, A.E.; Chen, Z.J.; Hodnett, G.; Drabek, J.; Lopez, R.; Price, H.J. Evolution of Genome Size in Brassicaceae. Ann Bot 2005, 95, 229–235. [CrossRef]
- Mandáková, T.; Singh, V.; Krämer, U.; Lysak, M.A. Genome Structure of the Heavy Metal Hyperaccumulator Noccaea Caerulescens and Its Stability on Metalliferous and Nonmetalliferous Soils. Plant Physiol 2015, 169, 674–689. [CrossRef]
- Knight, C.A.; Molinari, N.A.; Petrov, D.A. The Large Genome Constraint Hypothesis: Evolution, Ecology and Phenotype. Ann Bot 2005, 95, 177–190. [CrossRef]
- Temsch, E.M.; Temsch, W.; Ehrendorfer-Schratt, L.; Greilhuber, J. Heavy Metal Pollution, Selection, and Genome Size: The Species of the Žerjav Study Revisited with Flow Cytometry. J Bot 2010, 2010, 596542. [CrossRef]
- Vidic, T.; Greilhuber, J.; Vilhar, B.; Dermastia, M. Selective Significance of Genome Size in a Plant Community with Heavy Metal Pollution. Ecological Applications 2009, 19, 1515–1521. [CrossRef]
- Leitch IJ, J.E.P.J.H.O.B.M. release 7. 1, A. 2019 Plant DNA C-Values Database | Royal Botanic Gardens, Kew Available online: https://cvalues.science.kew.org/ (accessed on 29 July 2025).
- Rodríguez-Gijón, A.; Nuy, J.K.; Mehrshad, M.; Buck, M.; Schulz, F.; Woyke, T.; Garcia, S.L. A Genomic Perspective Across Earth’s Microbiomes Reveals That Genome Size in Archaea and Bacteria Is Linked to Ecosystem Type and Trophic Strategy. Front Microbiol 2022, 12, 761869. [CrossRef]
- Greilhuber, J.; Borsch, T.; Müller, K.; Worberg, A.; Porembski, S.; Barthlott, W. Smallest Angiosperm Genomes Found in Lentibulariaceae, with Chromosomes of Bacterial Size. Plant Biol (Stuttg) 2006, 8, 770–777. [CrossRef]
- Pellicer, J.; Fay, M.F.; Leitch, I.J. The Largest Eukaryotic Genome of Them All? Botanical Journal of the Linnean Society 2010, 164, 10–15. [CrossRef]
- Francis, D.; Davies, M.S.; Barlow, P.W. A Strong Nucleotypic Effect on the Cell Cycle Regardless of Ploidy Level. Ann Bot 2008, 101, 747–757. [CrossRef]
- D’Ario, M.; Tavares, R.; Schiessl, K.; Desvoyes, B.; Gutierrez, C.; Howard, M.; Sablowski, R. Cell Size Controlled in Plants Using DNA Content as an Internal Scale. Science 2021, 372, 1176–1181. [CrossRef]
- Segraves, K.A. The Effects of Genome Duplications in a Community Context. New Phytologist 2017, 215, 57–69. [CrossRef]
- Regvar, M.; Vogel, K.; Irgel, N.; Hildebrandt, U.; Wilde, P.; Bothe, H. Colonization of Pennycresses (Thlaspi Spp.) of the Brassicaceae by Arbuscular Mycorrhizal Fungi; 2003; Vol. 160;.
- Orłowska, E.; Zubek, S.; Jurkiewicz, A.; Szarek-Łukaszewska, G.; Turnau, K. Influence of Restoration on Arbuscular Mycorrhiza of Biscutella Laevigata L. (Brassicaceae) and Plantago Lanceolata L. (Plantaginaceae) from Calamine Spoil Mounds. Mycorrhiza 2002, 12, 153–159. [CrossRef]
- Trouvelot, A.; Kough, J.L.; Gianinazzi-Pearson, V. Du Taux de Mycorhization VA d’un Système Radiculaire. Recherche de Méthodes d’estimation Ayant Une Signification Fonctionnelle. Mycorhizes: Physiologie et génétique 1986.
- Ancousture, J.; Durand, A.; Blaudez, D.; Benizri, E. A Reduced but Stable Core Microbiome Found in Seeds of Hyperaccumulators. Science of The Total Environment 2023, 887, 164131. [CrossRef]
- Levy, A.; Salas Gonzalez, I.; Mittelviefhaus, M.; Clingenpeel, S.; Herrera Paredes, S.; Miao, J.; Wang, K.; Devescovi, G.; Stillman, K.; Monteiro, F.; et al. Genomic Features of Bacterial Adaptation to Plants. Nature Genetics 2017 50:1 2017, 50, 138–150. [CrossRef]
- van der Ent, A.; Spiers, K.M.; Brueckner, D.; Echevarria, G.; Aarts, M.G.M.; Montargès-Pelletier, E. Spatially-Resolved Localization and Chemical Speciation of Nickel and Zinc in Noccaea Tymphaea and Bornmuellera Emarginata. Metallomics 2019. [CrossRef]
- Hanikenne, M.; Talke, I.N.; Haydon, M.J.; Lanz, C.; Nolte, A.; Motte, P.; Kroymann, J.; Weigel, D.; Krämer, U. Evolution of Metal Hyperaccumulation Required Cis-Regulatory Changes and Triplication of HMA4. Nature 2008 453:7193 2008, 453, 391–395. [CrossRef]
- Roux, C.; Castric, V.; Pauwels, M.; Wright, S.I.; Saumitou-Laprade, P.; Vekemans, X. Does Speciation between Arabidopsis Halleri and Arabidopsis Lyrata Coincide with Major Changes in a Molecular Target of Adaptation? PLoS ONE 2011, 6, e26872. [CrossRef]
- Ó Lochlainn, S.; Bowen, H.C.; Fray, R.G.; Hammond, J.P.; King, G.J.; White, P.J.; Graham, N.S.; Broadley, M.R. Tandem Quadruplication of HMA4 in the Zinc (Zn) and Cadmium (Cd) Hyperaccumulator Noccaea Caerulescens. PLoS ONE 2011, 6, e17814. [CrossRef]
- Craciun, A.R.; Meyer, C.L.; Chen, J.; Roosens, N.; De Groodt, R.; Hilson, P.; Verbruggen, N. Variation in HMA4 Gene Copy Number and Expression among Noccaea Caerulescens Populations Presenting Different Levels of Cd Tolerance and Accumulation. J Exp Bot 2012, 63, 4179–4189. [CrossRef]
- Clemens, S.; Palmgren, M.G.; Krämer, U. A Long Way Ahead: Understanding and Engineering Plant Metal Accumulation. Trends Plant Sci 2002, 7, 309–315. [CrossRef]
- Hanikenne, M.; Nouet, C. Metal Hyperaccumulation and Hypertolerance: A Model for Plant Evolutionary Genomics. Curr Opin Plant Biol 2011, 14, 252–259. [CrossRef]
- Milner, M.J.; Mitani-Ueno, N.; Yamaji, N.; Yokosho, K.; Craft, E.; Fei, Z.; Ebbs, S.; Clemencia Zambrano, M.; Ma, J.F.; Kochian, L. V. Root and Shoot Transcriptome Analysis of Two Ecotypes of Noccaea Caerulescens Uncovers the Role of NcNramp1 in Cd Hyperaccumulation. The Plant Journal 2014, 78, 398–410. [CrossRef]
- Martos, S.; Gallego, B.; Sáez, L.; López-Alvarado, J.; Cabot, C.; Poschenrieder, C. Characterization of Zinc and Cadmium Hyperaccumulation in Three Noccaea (Brassicaceae) Populations from Non-Metalliferous Sites in the Eastern Pyrenees. Front Plant Sci 2016, 7, 173623. [CrossRef]
- Lin, Y.F.; Severing, E.I.; te Lintel Hekkert, B.; Schijlen, E.; Aarts, M.G.M. A Comprehensive Set of Transcript Sequences of the Heavy Metal Hyperaccumulator Noccaea Caerulescens. Front Plant Sci 2014, 5, 78115. [CrossRef]
- Ważny, R.; Rozpądek, P.; Domka, A.; Jędrzejczyk, R.J.; Nosek, M.; Hubalewska-Mazgaj, M.; Lichtscheidl, I.; Kidd, P.; Turnau, K. The Effect of Endophytic Fungi on Growth and Nickel Accumulation in Noccaea Hyperaccumulators. Science of The Total Environment 2021, 768, 144666. [CrossRef]
- Bočaj, V.; Pongrac, P.; Grčman, H.; Šala, M.; Likar, M. Rhizobiome Diversity of Field-Collected Hyperaccumulating Noccaea Sp. BMC Plant Biol 2024, 24, 1–14. [CrossRef]
- Durand, A.; Sterckeman, T.; Gonnelli, C.; Coppi, A.; Bacci, G.; Leglize, P.; Benizri, E. A Core Seed Endophytic Bacterial Community in the Hyperaccumulator Noccaea Caerulescens across 14 Sites in France. Plant Soil 2021, 459, 203–216. [CrossRef]
- Wolters, H.; Jürgens, G. Survival of the Flexible: Hormonal Growth Control and Adaptation in Plant Development. Nature Reviews Genetics 2009 10:5 2009, 10, 305–317. [CrossRef]
- Callahan, D.L.; Hare, D.J.; Bishop, D.P.; Doble, P.A.; Roessner, U. Elemental Imaging of Leaves from the Metal Hyperaccumulating Plant Noccaea Caerulescens Shows Different Spatial Distribution of Ni, Zn and Cd. RSC Adv 2015, 6, 2337–2344. [CrossRef]
- Vázquez, M.D.; Barceló, J.; Poschenrieder, Ch.; Mádico, J.; Hatton, P.; Baker, A.J.M.; Cope, G.H. Localization of Zinc and Cadmium in Thlaspi Caerulescens (Brassicaceae), a Metallophyte That Can Hyperaccumulate Both Metals. J Plant Physiol 1992, 140, 350–355. [CrossRef]
- Regvar, M.; Eichert, D.; Kaulich, B.; Gianoncelli, A.; Pongrac, P.; Vogel-Mikuš, K. Biochemical Characterization of Cell Types within Leaves of Metal-Hyperaccumulating Noccaea Praecox (Brassicaceae). Plant Soil 2013, 373, 157–171. [CrossRef]
- Braccini, I.; Pérez, S. Molecular Basis of Ca2+-Induced Gelation in Alginates and Pectins: The Egg-Box Model Revisited. Biomacromolecules 2001, 2, 1089–1096. [CrossRef]
- Pongrac, P.; Vogel-Mikuš, K.; Vavpetič, P.; Tratnik, J.; Regvar, M.; Simčič, J.; Grlj, N.; Pelicon, P. Cd Induced Redistribution of Elements within Leaves of the Cd/Zn Hyperaccumulator Thlaspi Praecox as Revealed by Micro-PIXE. Nucl Instrum Methods Phys Res B 2010, 268, 2205–2210. [CrossRef]
- Smets, W.; Chock, M.K.; Walsh, C.M.; Vanderburgh, C.Q.; Kau, E.; Lindow, S.E.; Fierer, N.; Koskella, B. Leaf Side Determines the Relative Importance of Dispersal versus Host Filtering in the Phyllosphere Microbiome. mBio 2023, 14, e0111123. [CrossRef]
- Fones, H.N.; McCurrach, H.; Mithani, A.; Smith, J.A.C.; Preston, G.M. Local Adaptation Is Associated with Zinc Tolerance in Pseudomonas Endophytes of the Metal-Hyperaccumulator Plant Noccaea Caerulescens. Proceedings of the Royal Society B: Biological Sciences 2016, 283. [CrossRef]
- Zelko, I.; Lux, A.; Czibula, K. Difference in the Root Structure of Hyperaccumulator Thlaspi Caerulescens and Non-Hyperaccumulator Thlaspi Arvense. Int J Environ Pollut 2008, 33, 123–132. [CrossRef]
- Hiruma, K.; Gerlach, N.; Sacristán, S.; Nakano, R.T.; Hacquard, S.; Kracher, B.; Neumann, U.; Ramírez, D.; Bucher, M.; O’Connell, R.J.; et al. Root Endophyte Colletotrichum Tofieldiae Confers Plant Fitness Benefits That Are Phosphate Status Dependent. Cell 2016, 165, 464–474. [CrossRef]
- Andersen, T.G.; Naseer, S.; Ursache, R.; Wybouw, B.; Smet, W.; De Rybel, B.; Vermeer, J.E.M.; Geldner, N. Diffusible Repression of Cytokinin Signalling Produces Endodermal Symmetry and Passage Cells. Nature 2018 555:7697 2018, 555, 529–533. [CrossRef]
- Holbein, J.; Shen, D.; Andersen, T.G. The Endodermal Passage Cell – Just Another Brick in the Wall? New Phytologist 2021, 230, 1321–1328. [CrossRef]
- Paungfoo-Lonhienne, C.; Rentsch, D.; Robatzek, S.; Webb, R.I.; Sagulenko, E.; Näsholm, T.; Schmidt, S.; Lonhienne, T.G.A. Turning the Table: Plants Consume Microbes as a Source of Nutrients. PLoS ONE 2010, 5, e11915. [CrossRef]
- Durr, J.; Reyt, G.; Spaepen, S.; Hilton, S.; Meehan, C.; Qi, W.; Kamiya, T.; Flis, P.; Dickinson, H.G.; Feher, A.; et al. A Novel Signaling Pathway Required for Arabidopsis Endodermal Root Organization Shapes the Rhizosphere Microbiome. Plant Cell Physiol 2021, 62, 248–261. [CrossRef]
- Salas-González, I.; Reyt, G.; Flis, P.; Custódio, V.; Gopaulchan, D.; Bakhoum, N.; Dew, T.P.; Suresh, K.; Franke, R.B.; Dangl, J.L.; et al. Coordination between Microbiota and Root Endodermis Supports Plant Mineral Nutrient Homeostasis. Science 2021, 371. [CrossRef]
- Maciá-Vicente, J.G.; Nam, B.; Thines, M. Root Filtering, Rather than Host Identity or Age, Determines the Composition of Root-Associated Fungi and Oomycetes in Three Naturally Co-Occurring Brassicaceae. Soil Biol Biochem 2020, 146, 107806. [CrossRef]
- Sharma, A.; Sinharoy, S.; Bisht, N.C. The Mysterious Non-Arbuscular Mycorrhizal Status of Brassicaceae Species. Environ Microbiol 2023, 25, 917–930. [CrossRef]
- Delaux, P.M.; Radhakrishnan, G. V.; Jayaraman, D.; Cheema, J.; Malbreil, M.; Volkening, J.D.; Sekimoto, H.; Nishiyama, T.; Melkonian, M.; Pokorny, L.; et al. Algal Ancestor of Land Plants Was Preadapted for Symbiosis. Proc Natl Acad Sci U S A 2015, 112, 13390–13395. [CrossRef]
- Pongrac, P.; Vogel-Mikuš, K.; Poschenrieder, C.; Barceló, J.; Tolrà, R.; Regvar, M. Arbuscular Mycorrhiza in Glucosinolate-Containing Plants: The Story of the Metal Hyperaccumulator Noccaea (Thlaspi) Praecox (Brassicaceae); 2013;.
- Pongrac, P.; Vogel-Mikuš, K.; Regvar, M.; Tolrà, R.; Poschenrieder, C.; Barceló, J. Glucosinolate Profiles Change during the Life Cycle and Mycorrhizal Colonization in a Cd/Zn Hyperaccumulator Thlaspi Praecox (Brassicaceae). J Chem Ecol 2008, 34, 1038–1044. [CrossRef]
- Bočaj, V.; Regvar, M.; Pongrac, P. Linking Microbiome and Hyperaccumulation in Plants. 2025. [CrossRef]
- Yang, J.; Song, N.; Zhao, X.; Qi, X.; Hu, Z.; Zhang, M. Genome Survey Sequencing Provides Clues into Glucosinolate Biosynthesis and Flowering Pathway Evolution in Allotetrapolyploid Brassica Juncea. BMC Genomics 2014, 15, 1–9. [CrossRef]
- Grubb, C.D.; Abel, S. Glucosinolate Metabolism and Its Control. Trends Plant Sci 2006, 11, 89–100. [CrossRef]
- Agerbirk, N.; Olsen, C.E. Glucosinolate Structures in Evolution. Phytochemistry 2012, 77, 16–45. [CrossRef]
- De Graaf, R.M.; Krosse, S.; Swolfs, A.E.M.; Te Brinke, E.T.; Prill, N. Te; Leimu, R.; Van Galen, P.M.; Wang, Y.; Aarts, M.G.M.; Van Dam, N.M. Isolation and Identification of 4-α-Rhamnosyloxy Benzyl Glucosinolate in Noccaea Caerulescens Showing Intraspecific Variation. Phytochemistry 2015, 110, 166–171. [CrossRef]
- Jeon, B.W.; Oh, M.H.; Kim, H.S.; Kim, E.O.; Chae, W.B. Glucosinolate Variation among Organs, Growth Stages and Seasons Suggests Its Dominant Accumulation in Sexual over Asexual-Reproductive Organs in White Radish. Sci Hortic 2022, 291, 110617. [CrossRef]
- Tolrà, R.; Pongrac, P.; Poschenrieder, C.; Vogel-Mikuš, K.; Regvar, M.; Barceló, J. Distinctive Effects of Cadmium on Glucosinolate Profiles in Cd Hyperaccumulator Thlaspi Praecox and Non-Hyperaccumulator Thlaspi Arvense. Plant Soil 2006, 288, 333–341. [CrossRef]
- Unger, K.; Raza, S.A.K.; Mayer, T.; Reichelt, M.; Stuttmann, J.; Hielscher, A.; Wittstock, U.; Gershenzon, J.; Agler, M.T. Glucosinolate Structural Diversity Shapes Recruitment of a Metabolic Network of Leaf-Associated Bacteria. Nature Communications 2024, 15. [CrossRef]
- Anthony, M.A.; Celenza, J.L.; Armstrong, A.; Frey, S.D. Indolic Glucosinolate Pathway Provides Resistance to Mycorrhizal Fungal Colonization in a Non-Host Brassicaceae. Ecosphere 2020, 11, e03100. [CrossRef]
- Szucs, Z.; Plaszkó, T.; Cziáky, Z.; Kiss-Szikszai, A.; Emri, T.; Bertóti, R.; Sinka, L.T.; Vasas, G.; Gonda, S. Endophytic Fungi from the Roots of Horseradish (Armoracia Rusticana) and Their Interactions with the Defensive Metabolites of the Glucosinolate - Myrosinase - Isothiocyanate System. BMC Plant Biol 2018, 18, 1–15. [CrossRef]
- Chen, J.; Ullah, C.; Reichelt, M.; Beran, F.; Yang, Z.L.; Gershenzon, J.; Hammerbacher, A.; Vassão, D.G. The Phytopathogenic Fungus Sclerotinia Sclerotiorum Detoxifies Plant Glucosinolate Hydrolysis Products via an Isothiocyanate Hydrolase. Nature Communications 2020 11:1 2020, 11, 1–12. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




