Submitted:
30 July 2025
Posted:
30 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
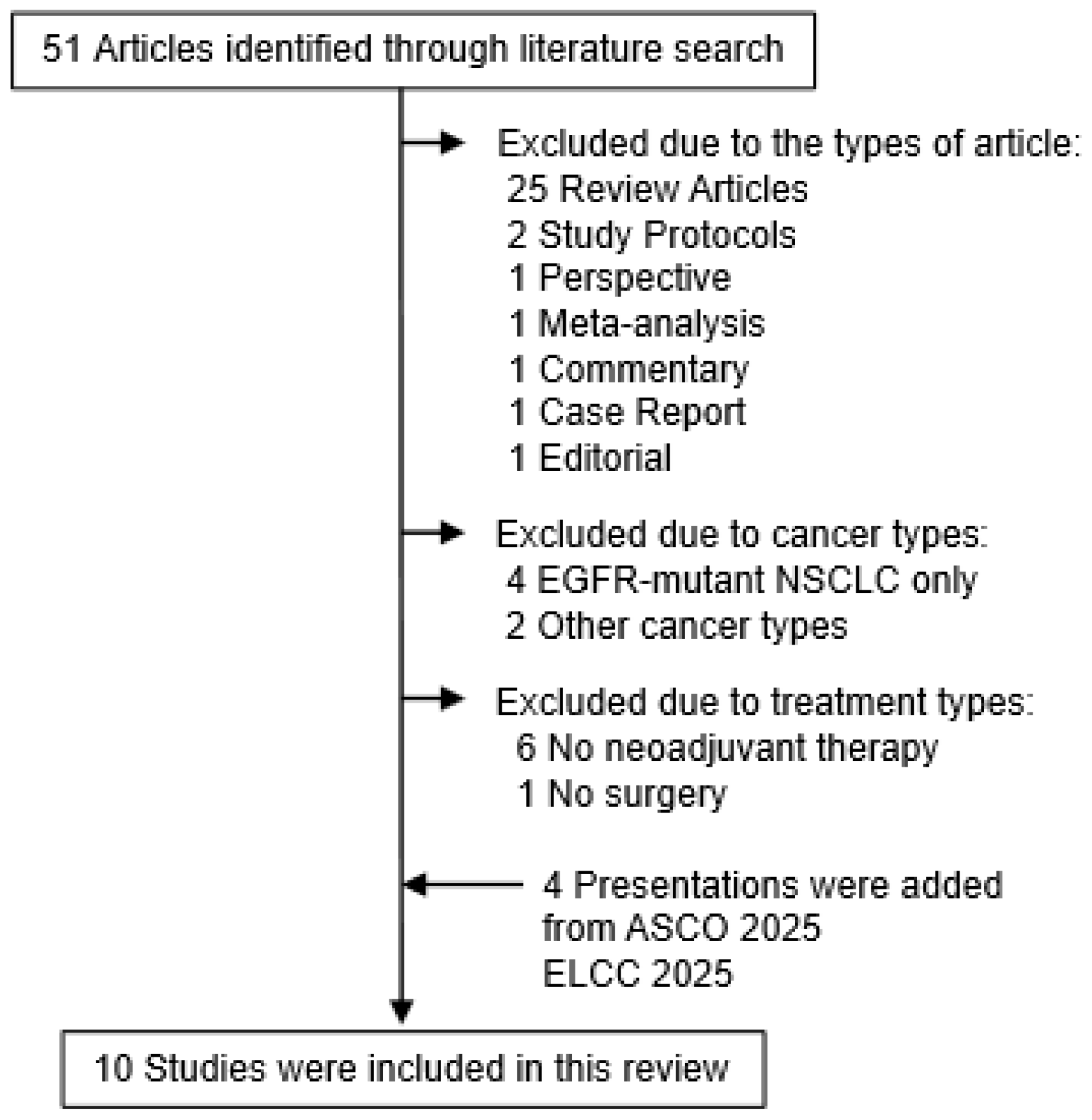
2. Literature Search
3. Current Clinical Challenges of Perioperative/Neoadjuvant Chemo-Immunotherapy
3.1. Need for Neoadjuvant Chemo-Immunotherapy
3.2. Adequate Treatment Courses of Neoadjuvant Chemo-Immunotherapy
3.3. Monitoring Tumor Response to Neoadjuvant Treatment
3.4. Recurrence Risk Stratification After Pulmonary Resection
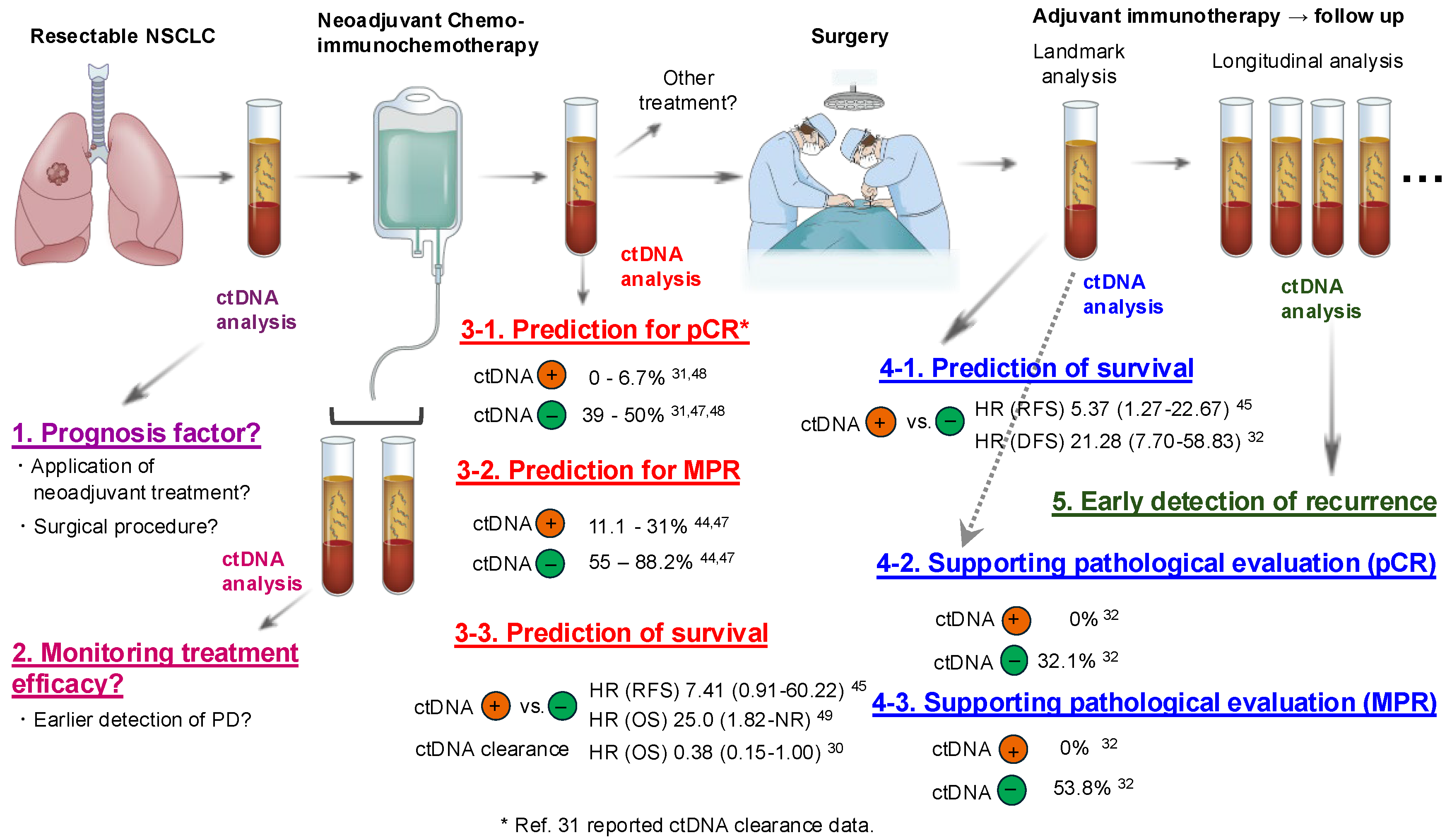
4. Utility of ctDNA Analysis as a Tool to Solve the Above Clinical Questions
4.1. Utility of ctDNA Analysis Before and During Neoadjuvant Treatment
4.2. Utility of ctDNA Analysis Before Surgical Resection
4.3. Landmark ctDNA Status as a Tool for MRD Detection
4.4. Longitudinal ctDNA Analysis for Early Detection of Disease Recurrence
5. Ongoing Clinical Trials Involving ctDNA During Perioperative Chemo-Immunotherapy
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Felip, E.; Altorki, N.; Zhou, C.; Vallières, E.; Martínez-Martí, A.; Rittmeyer, A.; Chella, A.; Reck, M.; Goloborodko, O.; Huang, M.; et al. Overall survival with adjuvant atezolizumab after chemotherapy in resected stage II-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase III trial. Ann. Oncol. 2023, 34, 907–919. [CrossRef]
- O’bRien, M.; Paz-Ares, L.; Marreaud, S.; Dafni, U.; Oselin, K.; Havel, L.; Esteban, E.; Isla, D.; Martinez-Marti, A.; Faehling, M.; et al. Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial. Lancet Oncol. 2022, 23, 1274–1286. [CrossRef]
- Forde PM, Spicer J, Lu S, Provencio M, Mitsudomi T, Awad MM, Felip E, Broderick SR, Brahmer JR, Swanson SJ et al. Neoadjuvant Nivolumab plus Chemotherapy in Resectable Lung Cancer. N Engl J Med 2022; 386: 1973-1985.
- Spicer, J.D.; Garassino, M.C.; Wakelee, H.; Liberman, M.; Kato, T.; Tsuboi, M.; Lee, S.-H.; Chen, K.-N.; Dooms, C.; Majem, M.; et al. Neoadjuvant pembrolizumab plus chemotherapy followed by adjuvant pembrolizumab compared with neoadjuvant chemotherapy alone in patients with early-stage non-small-cell lung cancer (KEYNOTE-671): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2024, 404, 1240–1252. [CrossRef]
- Heymach, J.V.; Harpole, D.; Mitsudomi, T.; Taube, J.M.; Galffy, G.; Hochmair, M.; Winder, T.; Zukov, R.; Garbaos, G.; Gao, S.; et al. Perioperative Durvalumab for Resectable Non–Small-Cell Lung Cancer. New Engl. J. Med. 2023, 389, 1672–1684. [CrossRef]
- Abbosh, C.; Birkbak, N.J.; Wilson, G.A.; Jamal-Hanjani, M.; Constantin, T.; Salari, R.; Le Quesne, J.; Moore, D.A.; Veeriah, S.; Rosenthal, R.; et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 2017, 545, 446–451. [CrossRef]
- Abbosh, C.; Hodgson, D.; Doherty, G.J.; Gale, D.; Black, J.R.; Horn, L.; Reis-Filho, J.S.; Swanton, C. Implementing circulating tumor DNA as a prognostic biomarker in resectable non-small cell lung cancer. Trends Cancer 2024, 10, 643–654. [CrossRef]
- Peng, Y.; Mei, W.; Ma, K.; Zeng, C. Circulating Tumor DNA and Minimal Residual Disease (MRD) in Solid Tumors: Current Horizons and Future Perspectives. Front. Oncol. 2021, 11. [CrossRef]
- Pellini, B.; Chaudhuri, A.A. Circulating Tumor DNA Minimal Residual Disease Detection of Non–Small-Cell Lung Cancer Treated With Curative Intent. J. Clin. Oncol. 2022, 40, 567–575. [CrossRef]
- Diehl, F.; Schmidt, K.; Choti, M.A.; Romans, K.; Goodman, S.; Li, M.; Thornton, K.; Agrawal, N.; Sokoll, L.; Szabo, S.A.; et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 2008, 14, 985–990. [CrossRef]
- Qin, Z.; Ljubimov, V.A.; Zhou, C.; Tong, Y.; Liang, J. Cell-free circulating tumor DNA in cancer. Chin. J. Cancer 2016, 35, 1–9. [CrossRef]
- Burgener, J.M.; Rostami, A.; De Carvalho, D.D.; Bratman, S.V. Cell-free DNA as a post-treatment surveillance strategy: current status. Semin. Oncol. 2017, 44, 330–346. [CrossRef]
- Chakrabarti, S.; Kasi, A.K.; Parikh, A.R.; Mahipal, A. Finding Waldo: The Evolving Paradigm of Circulating Tumor DNA (ctDNA)—Guided Minimal Residual Disease (MRD) Assessment in Colorectal Cancer (CRC). Cancers 2022, 14, 3078. [CrossRef]
- Ohara, S.; Suda, K.; Sudhaman, S.; Hamada, A.; Chiba, M.; Shimoji, M.; Takemoto, T.; Kalashnikova, E.; Cheung, S.K.; Krainock, M.; et al. Clinical Significance of Perioperative Minimal Residual Disease Detected by Circulating Tumor DNA in Patients With Lung Cancer With a Long Follow-up Data: An Exploratory Study. JTO Clin. Res. Rep. 2024, 6, 100762. [CrossRef]
- Gale, D.; Heider, K.; Ruiz-Valdepenas, A.; Hackinger, S.; Perry, M.; Marsico, G.; Rundell, V.; Wulff, J.; Sharma, G.; Knock, H.; et al. Residual ctDNA after treatment predicts early relapse in patients with early-stage non-small cell lung cancer. Ann. Oncol. 2022, 33, 500–510. [CrossRef]
- Chen, K.; Zhao, H.; Shi, Y.; Yang, F.; Wang, L.T.; Kang, G.; Nie, Y.; Wang, J. Perioperative Dynamic Changes in Circulating Tumor DNA in Patients with Lung Cancer (DYNAMIC). Clin. Cancer Res. 2019, 25, 7058–7067. [CrossRef]
- Chen, K.; Yang, F.; Shen, H.; Wang, C.; Li, X.; Chervova, O.; Wu, S.; Qiu, F.; Peng, D.; Zhu, X.; et al. Individualized tumor-informed circulating tumor DNA analysis for postoperative monitoring of non-small cell lung cancer. Cancer Cell 2023, 41, 1749–1762.e6. [CrossRef]
- Chaudhuri, A.A.; Chabon, J.J.; Lovejoy, A.F.; Newman, A.M.; Stehr, H.; Azad, T.D.; Khodadoust, M.S.; Esfahani, M.S.; Liu, C.L.; Zhou, L.; et al. Early Detection of Molecular Residual Disease in Localized Lung Cancer by Circulating Tumor DNA Profiling. Cancer Discov. 2017, 7, 1394–1403. [CrossRef]
- Xia, L.; Pu, Q.; Kang, R.; Mei, J.; Li, L.; Yang, Y.; Deng, S.; Feng, G.; Ma, L.; Lin, F.; et al. Dynamic ctDNA to inform the precise management of resected NSCLC: LUNGCA-2 study.. J. Clin. Oncol. 2023, 41, 8528–8528. [CrossRef]
- Chin, R.-I.; Chen, K.; Usmani, A.; Chua, C.; Harris, P.K.; Binkley, M.S.; Azad, T.D.; Dudley, J.C.; Chaudhuri, A.A. Detection of Solid Tumor Molecular Residual Disease (MRD) Using Circulating Tumor DNA (ctDNA). Mol. Diagn. Ther. 2019, 23, 311–331. [CrossRef]
- Fu, R.; Huang, J.; Tian, X.; Liang, C.; Xiong, Y.; Zhang, J.; Jiang, B.; Dong, S.; Gong, Y.; Gao, W.; et al. Postoperative circulating tumor DNA can refine risk stratification in resectable lung cancer: results from a multicenter study. Mol. Oncol. 2023, 17, 825–838. [CrossRef]
- Li, N.; Wang, B.; Shao, Y.; Li, M.; Li, J.; Kuang, P.; Liu, Z.; Sun, T.; Wu, H.; Ou, W.; et al. Perioperative circulating tumor DNA as a potential prognostic marker for operable stage I to IIIA non–small cell lung cancer. Cancer 2021, 128, 708–718. [CrossRef]
- Tian, X.; Liu, X.; Wang, K.; Wang, R.; Li, Y.; Qian, K.; Wang, T.; Zhao, X.; Liu, L.; Zhang, P.L.; et al. Postoperative ctDNA in indicating the recurrence risk and monitoring the effect of adjuvant therapy in surgical non-small cell lung cancer. Thorac. Cancer 2024, 15, 797–807. [CrossRef]
- Tran, H.; Heeke, S.; Sujit, S.; Vokes, N.; Zhang, J.; Aminu, M.; Lam, V.; Vaporciyan, A.; Swisher, S.; Godoy, M.; et al. Circulating tumor DNA and radiological tumor volume identify patients at risk for relapse with resected, early-stage non-small-cell lung cancer. Ann. Oncol. 2023, 35, 183–189. [CrossRef]
- Waldeck, S.; Mitschke, J.; Wiesemann, S.; Rassner, M.; Andrieux, G.; Deuter, M.; Mutter, J.; Lüchtenborg, A.; Kottmann, D.; Titze, L.; et al. Early assessment of circulating tumor DNA after curative-intent resection predicts tumor recurrence in early-stage and locally advanced non-small-cell lung cancer. Mol. Oncol. 2021, 16, 527–537. [CrossRef]
- Wang, S.; Li, M.; Zhang, J.; Xing, P.; Wu, M.; Meng, F.; Jiang, F.; Wang, J.; Bao, H.; Huang, J.; et al. Circulating tumor DNA integrating tissue clonality detects minimal residual disease in resectable non-small-cell lung cancer. J. Hematol. Oncol. 2022, 15, 1–6. [CrossRef]
- Wakelee, H.; Reck, M.; Felip, E.; Altorki, N.; Vallieres, E.; Liersch, R.; Oizumi, S.; Tanaka, H.; Hamm, J.; McCune, S.; et al. 1211P IMpower010: ctDNA status and 5y DFS follow up in patients (pts) with resected NSCLC who received adjuvant chemotherapy (chemo) followed by atezolizumab (atezo) or best supportive care (BSC). Ann. Oncol. 2024, 35, S779–S780. [CrossRef]
- Qiu, B.; Guo, W.; Zhang, F.; Lv, F.; Ji, Y.; Peng, Y.; Chen, X.; Bao, H.; Xu, Y.; Shao, Y.; et al. Dynamic recurrence risk and adjuvant chemotherapy benefit prediction by ctDNA in resected NSCLC. Nat. Commun. 2021, 12, 1–11. [CrossRef]
- Wang, B.; Zou, B.; Xu, S.; Zhao, C.; Pei, J.; Wang, S.; Zhao, K.; Yu, J.; Liu, J. Postoperative ctDNA detection predicts relapse but has limited effects in guiding adjuvant therapy in resectable stage I NSCLC. Front. Oncol. 2023, 13, 1083417. [CrossRef]
- Forde, P.M.; Spicer, J.; Provencio, M.; Mitsudomi, T.; Awad, M.M.; Wang, C.; Lu, S.; Felip, E.; Broderick, S.; Swanson, S.; et al. Overall survival with neoadjuvant nivolumab (NIVO) + chemotherapy (chemo) in patients with resectable NSCLC in CheckMate 816.. J. Clin. Oncol. 2025, 43. [CrossRef]
- Cascone, T.; Awad, M.M.; Spicer, J.; He, J.; Lu, S.; Tanaka, F.; Cornelissen, R.; Petruzelka, L.B.; Ito, H.; Koch, L.D.O.; et al. Perioperative nivolumab (NIVO) vs placebo (PBO) in patients (pts) with resectable NSCLC: Updated survival and biomarker analyses from CheckMate 77T.. J. Clin. Oncol. 2025, 43. [CrossRef]
- Reck, M.; Gale, D.; Zhu, Z.; Harpole, D.H.; Taube, J.M.; Mitsudomi, T.; Van Luong, D.; Hochmair, M.; Lee, K.-Y.; Horio, Y.; et al. Association of post-surgical MRD status with neoadjuvant ctDNA dynamics, genomic mutations, and clinical outcomes in patients with resectable NSCLC (R-NSCLC) from the phase 3 AEGEAN trial.. J. Clin. Oncol. 2025, 43, 8009–8009. [CrossRef]
- Serna-Blasco, R.; Nadal, E.; Gonzalez-Larriba, J.; Martinez-Marti, A.; Caro, R.B.; Bosch-Barrera, J.; De Juan, V.C.; Molla, M.I.; Reguart, N.; Carpeño, J.d.C.; et al. 210P: Prognostic Value of minimal residual disease in the NADIM II trial. J. Thorac. Oncol. 2025, 20, S138–S139. [CrossRef]
- Forde PM, Spicer JD, Provencio M, Mitsudomi T, Awad MM, Wang C, Lu S, Felip E, Swanson SJ, Brahmer JR et al. Overall Survival with Neoadjuvant Nivolumab plus Chemotherapy in Lung Cancer. N Engl J Med 2025.
- Zhang, B.; Guo, X.; Jia, R.; Wang, Z.; Wu, J.; Chen, X.; Li, J.; Yang, D.; Li, X.; Wang, W.; et al. Neoadjuvant chemoimmunotherapy cycle number selection for non-small cell lung cancer and clinical outcomes: a real-world analysis. Front. Oncol. 2023, 13, 1200625. [CrossRef]
- Shao, M.; Yao, J.; Wang, Y.; Zhao, L.; Li, B.; Li, L.; Wu, Z.; Chen, Z.; Fan, J.; Qiu, F. Two vs three cycles of neoadjuvant sintilimab plus chemotherapy for resectable non-small-cell lung cancer: neoSCORE trial. Signal Transduct. Target. Ther. 2023, 8, 1–3. [CrossRef]
- Reck, M.; Gale, D.; Harpole, D.; Taube, J.; Mitsudomi, T.; Hochmair, M.; Winder, T.; Zhu, Z.; Lai, Z.; Stewart, R.; et al. LBA59 Associations of ctDNA clearance and pathological response with neoadjuvant treatment in patients with resectable NSCLC from the phase III AEGEAN trial. Ann. Oncol. 2023, 34, S1300–S1300. [CrossRef]
- Suda K. DNA shedding in non-small-cell lung cancer: useful to assess? Lancet Respir Med 2018; 6: 77-78.
- Zheng, J.; Qin, C.; Wang, Q.; Tian, D.; Chen, Z. Circulating tumour DNA-Based molecular residual disease detection in resectable cancers: a systematic review and meta-analysis. EBioMedicine 2024, 103, 105109. [CrossRef]
- Saw, S.P.L.; Ong, B.-H.; Chua, K.L.M.; Takano, A.; Tan, D.S.W. Revisiting neoadjuvant therapy in non-small-cell lung cancer. Lancet Oncol. 2021, 22, e501–e516. [CrossRef]
- Pataer, A.; Kalhor, N.; Correa, A.M.; Raso, M.G.; Erasmus, J.J.; Kim, E.S.; Behrens, C.; Lee, J.J.; Roth, J.A.; Stewart, D.J.; et al. Histopathologic Response Criteria Predict Survival of Patients with Resected Lung Cancer After Neoadjuvant Chemotherapy. J. Thorac. Oncol. 2012, 7, 825–832. [CrossRef]
- Cottrell, T.; Thompson, E.; Forde, P.; Stein, J.; Duffield, A.; Anagnostou, V.; Rekhtman, N.; Anders, R.; Cuda, J.; Illei, P.; et al. Pathologic features of response to neoadjuvant anti-PD-1 in resected non-small-cell lung carcinoma: a proposal for quantitative immune-related pathologic response criteria (irPRC). Ann. Oncol. 2018, 29, 1853–1860. [CrossRef]
- Travis, W.D.; Dacic, S.; Wistuba, I.; Sholl, L.; Adusumilli, P.; Bubendorf, L.; Bunn, P.; Cascone, T.; Chaft, J.; Chen, G.; et al. IASLC Multidisciplinary Recommendations for Pathologic Assessment of Lung Cancer Resection Specimens After Neoadjuvant Therapy. J. Thorac. Oncol. 2020, 15, 709–740. [CrossRef]
- Xu, L.; Si, H.; Zhuang, F.; Li, C.; Zhang, L.; Zhao, Y.; Chen, T.; Dong, Y.; Wang, T.; Hou, L.; et al. Predicting therapeutic response to neoadjuvant immunotherapy based on an integration model in resectable stage IIIA (N2) non–small cell lung cancer. J. Thorac. Cardiovasc. Surg. 2024, 169, 242–253.e4. [CrossRef]
- Yue, D.; Liu, W.; Chen, C.; Zhang, T.; Ma, Y.; Cui, L.; Gu, Y.; Bei, T.; Zhao, X.; Zhang, B.; et al. Circulating tumor DNA predicts neoadjuvant immunotherapy efficacy and recurrence-free survival in surgical non-small cell lung cancer patients. Transl. Lung Cancer Res. 2022, 11, 263–276. [CrossRef]
- Deutsch, J.S.; Cimino-Mathews, A.; Thompson, E.; Provencio, M.; Forde, P.M.; Spicer, J.; Girard, N.; Wang, D.; Anders, R.A.; Gabrielson, E.; et al. Association between pathologic response and survival after neoadjuvant therapy in lung cancer. Nat. Med. 2023, 30, 218–228. [CrossRef]
- Zhao, J.; Zhao, L.; Guo, W.; Wang, S.; Tao, X.; Li, L.; Mao, Y.; Tan, F.; Gao, Y.; Wu, N.; et al. Efficacy, Safety, and Biomarker Analysis of Neoadjuvant Camrelizumab and Apatinib in Patients With Resectable NSCLC: A Phase 2 Clinical Trial. J. Thorac. Oncol. 2023, 18, 780–791. [CrossRef]
- Dong, S.; Yang, X.-N.; Liao, R.-Q.; Jiang, B.-Y.; Wang, Q.; Ben, X.-S.; Qiao, G.-B.; Lin, J.-T.; Yan, H.-H.; Yan, L.-X.; et al. Neoadjuvant nivolumab with or without platinum-doublet chemotherapy based on PD-L1 expression in resectable NSCLC (CTONG1804): a multicenter open-label phase II study. Signal Transduct. Target. Ther. 2023, 8, 1–10. [CrossRef]
- Provencio, M.; Serna-Blasco, R.; Nadal, E.; Insa, A.; García-Campelo, M.R.; Rubio, J.C.; Dómine, M.; Majem, M.; Rodríguez-Abreu, D.; Martínez-Martí, A.; et al. Overall Survival and Biomarker Analysis of Neoadjuvant Nivolumab Plus Chemotherapy in Operable Stage IIIA Non–Small-Cell Lung Cancer (NADIM phase II trial). J. Clin. Oncol. 2022, 40, 2924–2933. [CrossRef]
- Abbosh, C.; Frankell, A.M.; Harrison, T.; Kisistok, J.; Garnett, A.; Johnson, L.; Veeriah, S.; Moreau, M.; Chesh, A.; Chaunzwa, T.L.; et al. Tracking early lung cancer metastatic dissemination in TRACERx using ctDNA. Nature 2023, 616, 553–562. [CrossRef]
| Ref. # | Year | N | Neoadjuvant regimen | Adjuvant regimen |
| 45 | 2022 | 22 | ICI + CTx vs. ICI combo vs. CTx | - |
| 49 | 2022 | 46 | Nivo + CTx | Nivo vs. placebo |
| 46 | 2023 | 358 | Nivo + CTx vs. CTx | - |
| 48 | 2023 | 52 | Nivo + CTx vs. Nivo | - |
| 47 | 2023 | 78 | Camrelizumab + apatinib | - |
| 44 | 2025 | 45 | Sintilimab + CTx | - |
| 30 | 2025 | 358 | Nivo + CTx vs. CTx | - |
| 31 | 2025 | 461 | Nivo + CTx vs. CTx | Nivo vs. placebo |
| 32 | 2025 | 802 | Durval + CTx vs. CTx | Durval vs. placebo |
| 33 | 2025 | 86 | Nivo + CTx vs. CTx | Nivo vs. placebo |
| NCT # | Primary endpoints | Secondary endpoints | Exploratory analysis |
| NCT04638582 | ctDNA clearance (pre/post-surgery) |
MPR, pCR, radiologic response |
Correlation of ctDNA dynamics with DFS/OS |
| NCT05382052 | Association of ctDNA clearance with PFS |
DFS, OS | Longitudinal ctDNA monitoring feasibility |
| NCT05778253 | ctDNA clearance, AI-based prediction of pCR | MPR, ORR, DFS, OS, QoL, surgical outcomes | Correlation of AI pathology with clinical outcomes |
| NCT06111807 | Sensitivity/specificity of ctDNA assays | Association of ctDNA with recurrence/DFS | ctDNA-guided risk stratification |
| NCT06123754 | MPR (pre-op), EFS (post-op) |
pCR, DFS, OS, safety | Biomarker analysis, ctDNA dynamics |
| NCT06221462 | pCR/MPR, safety, surgical feasibility |
DFS, OS, AEs | ctDNA clearance, radiologic-pathologic correlation |
| NCT06284317 | DFS in non-pCR patients | DFS/OS in pCR group, safety, ctDNA analysis | Correlation of ctDNA with recurrence and survival |
| NCT06743555 | Feasibility and safety of surgery omission |
EFS, OS, recurrence rate | ctDNA monitoring for recurrence detection |
| NCT06902272 | Correlation between ctDNA and pCR/MPR |
DFS, OS, ctDNA kinetics | ctDNA MRD detection, longitudinal profiling |
| NCT06977074 | pCR, MPR, surgical eligibility |
DFS, OS, AEs | ctDNA-guided treatment optimization |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




