Submitted:
28 July 2025
Posted:
30 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Etiology
3. Epidemiology
4. Clinical Presentation
4.1. Asymptomatic Patient
4.2. Symptomatic Patient
4.3. Pathologic Fracture
4.4. Pathologic Fracture
5. Differential Diagnosis
| Lesion | Typical Age | Location | Radiographic Features | Histology Key Feature |
|---|---|---|---|---|
| Non-ossifying Fibroma | 5-20 yrs | Metaphysis (eccentric) | Lytic, scalloped, sclerotic rim, bubbly appearance | Storiform spindle cells, hemosiderin |
| Fibrous Dysplasia | <30 yrs | Metaphysis/Diaphysis (central) | Ground-glass, "Rind" sign, Shepherd’s Crook deformity | "Chinese character" woven bone |
| Aneurysmal Bone Cyst | <20 yrs | Metaphysis (central) | Expansile, lytic, fluid-fluid levels on MRI | Blood-filled spaces, giant cells |
| Simple Bone Cyst (UBC) | <20 yrs | Metaphysis (central) | Lytic, well-defined, "fallen leaf" sign after fracture | Thin fibrous lining, clear fluid |
| Giant Cell Tumor | 20-40 yrs | Epiphysis (abuts joint) | Lytic, expansile, non-sclerotic margin | Numerous osteoclast-like giant cells |
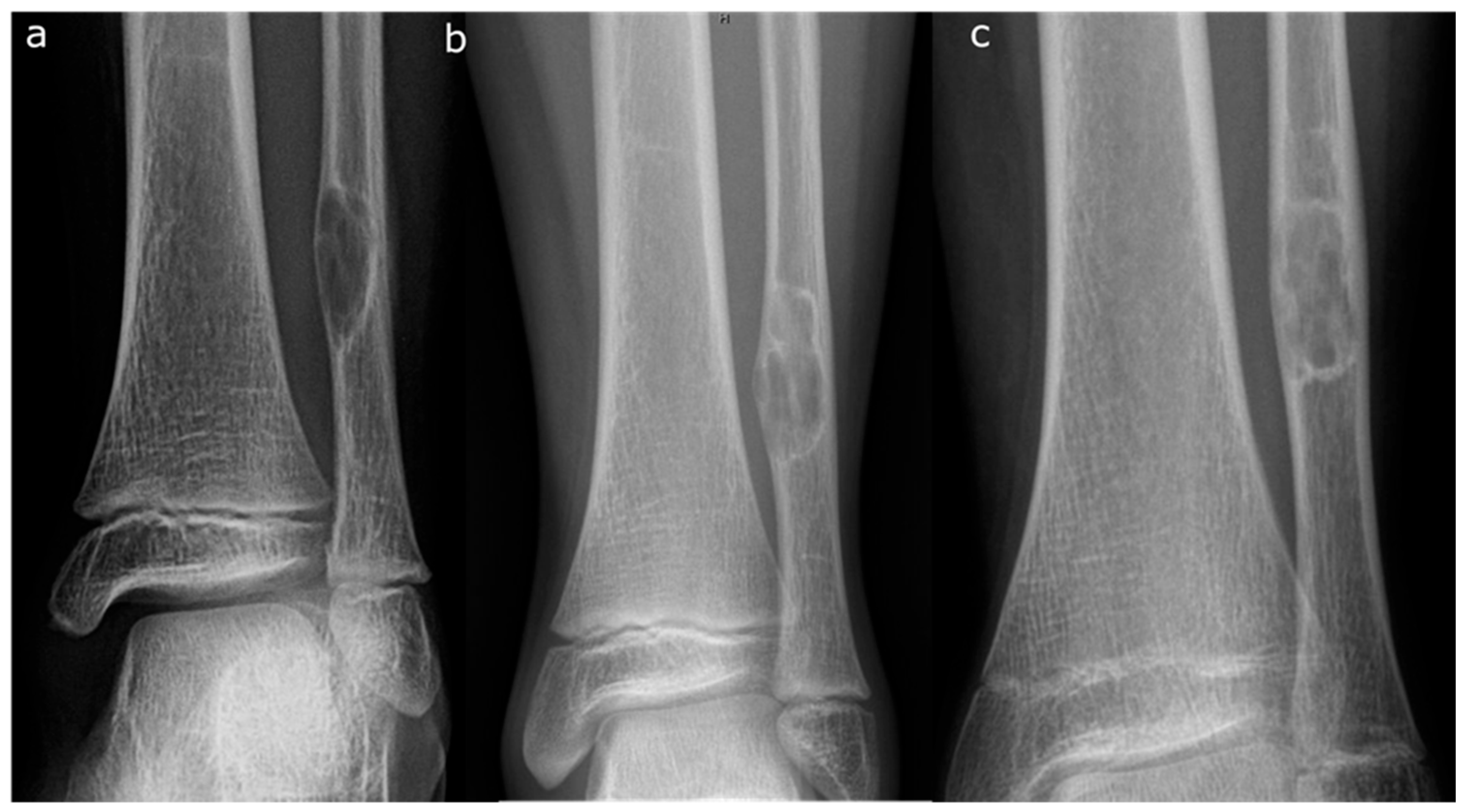
6. Radiographic and Advanced Imaging
6.1. Radiographic Features
6.2. Staging
- Stage A: Small, eccentric, cortically based lesions near the epiphysis with smooth, round borders.
- Stage B: Lesions become polycystic with clear but thin sclerotic borders, increasing in size and exhibiting variable distances from the epiphysis. The most significant growth typically occurs during this stage, transitioning from Stage A or within Stage B.
- Stage C: Lesions demonstrate increased sclerosis and reduced growth potential.
- Stage D: Complete and homogeneous sclerosis is observed, with no further growth.
6.3. Advanced Imaging
-
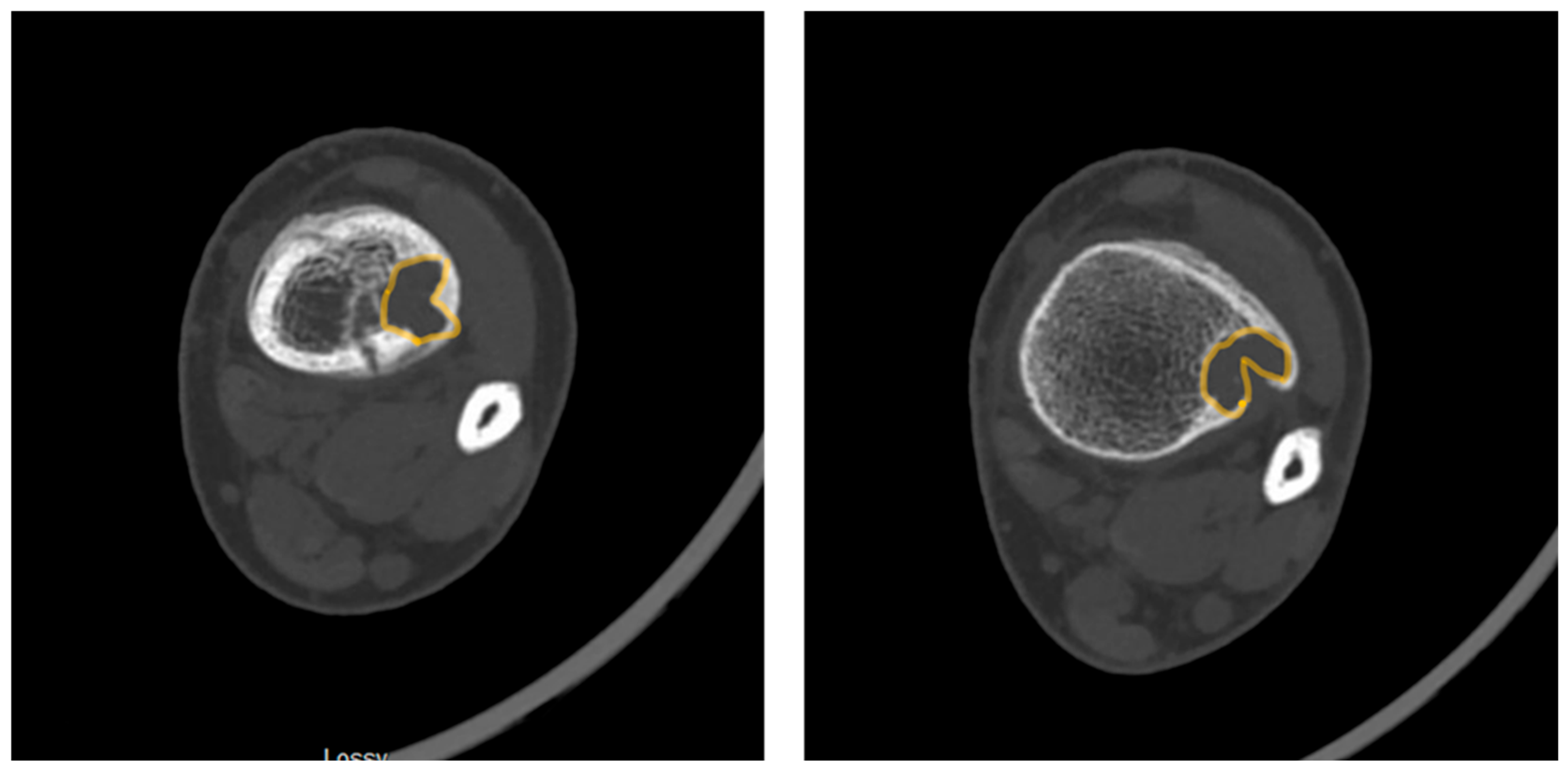
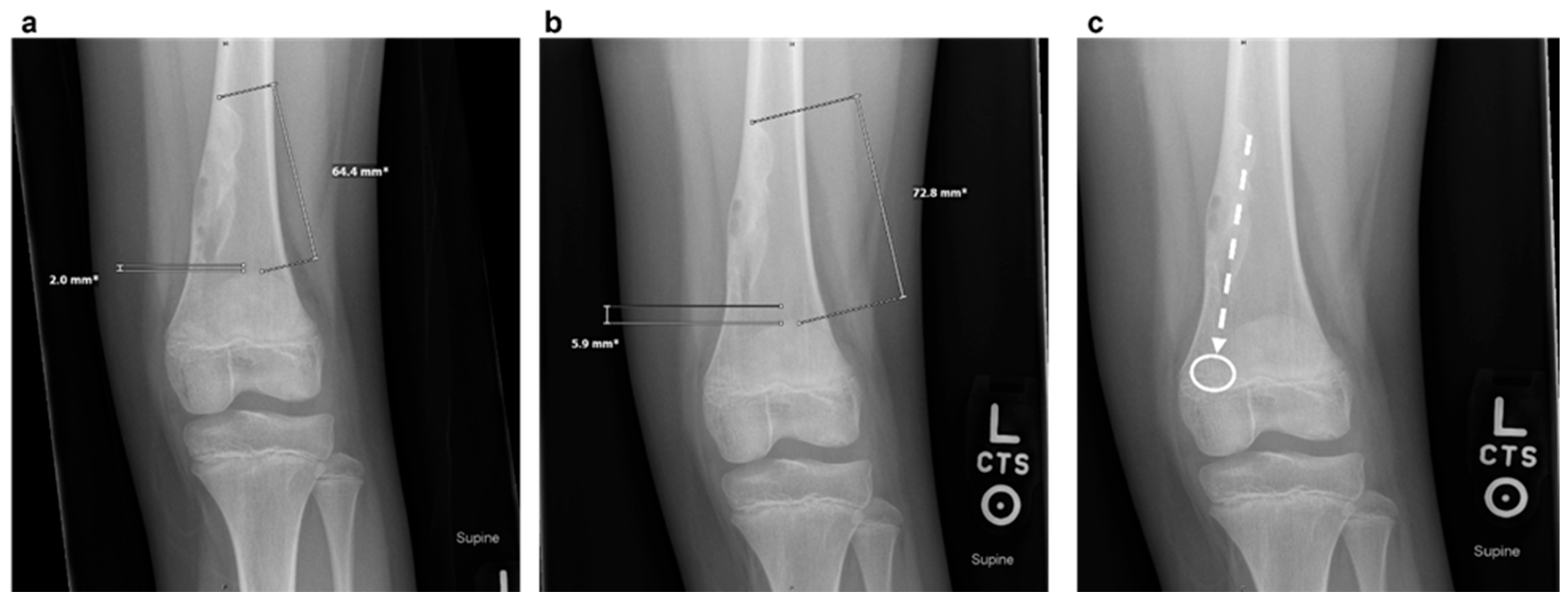
Magnetic Resonance Imaging (MRI): MRI can help differentiate NOFs from other lesions such as fibrous dysplasia or malignancies [7]. On MRI, NOFs are typically T1-hypointense and T2-hyperintense. A peripheral, low-signal rim on all sequences corresponds to the sclerotic border seen on radiographs. Post-contrast sequences usually show minimal, peripheral enhancement, whereas more aggressive lesions often demonstrate avid, diffuse enhancement. In 2021, Baghdadi et al. identified advanced imaging features, such as the "Pac-Man sign” and the “syndesmosis sign” that may indicate increased fracture risk in distal tibia NOFs [8].
- o
- Pac-Man Sign - Proliferation of bone anterior and posterior to the syndesmosis results in a shape which resembles the video game character “Pac-Man”. This sign was found to be highly specific (95%) but not very sensitive (47%) for predicting pathologic fracture. (Figure 3)
- o
- Syndesmosis Sign - Advanced imaging shows the syndesmosis inserting into the distal tibia lesion. This sign was found to be highly sensitive (94%) but less specific (48%) for predicting fracture risk.
- Computed Tomography (CT): CT can be useful for evaluating degree of cortical thinning, which is important when assessing fracture risk [9]. CT is superior for delineating the precise cortical integrity. It allows for quantitative measurement of the cross-sectional area occupied by the lesion, which is a key factor in biomechanical models predicting fracture risk. The signs described above can also be visualized on CT scan. Figure 3 demonstrates a “Pac-Man Sign” on CT of a 13-year-old boy who did have a fracture through an NOF.
- Bone Scintigraphy: Demonstrates mild uptake, reflecting the lesion’s low metabolic activity.
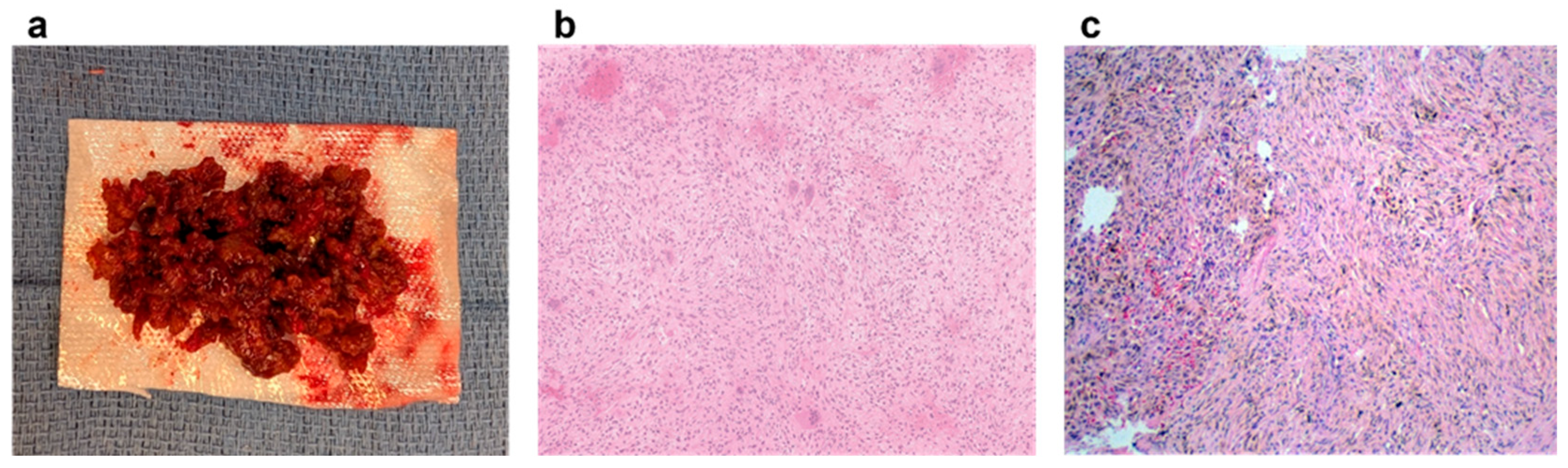
7. Pathology
8. Non-Operative Management
8.1. Observation and Monitoring
8.2. Activity Modification
8.3. Closed Reduction and Casting of Pathologic Fractures
9. Operative Intervention
9.1. Indications
9.2. Techniques
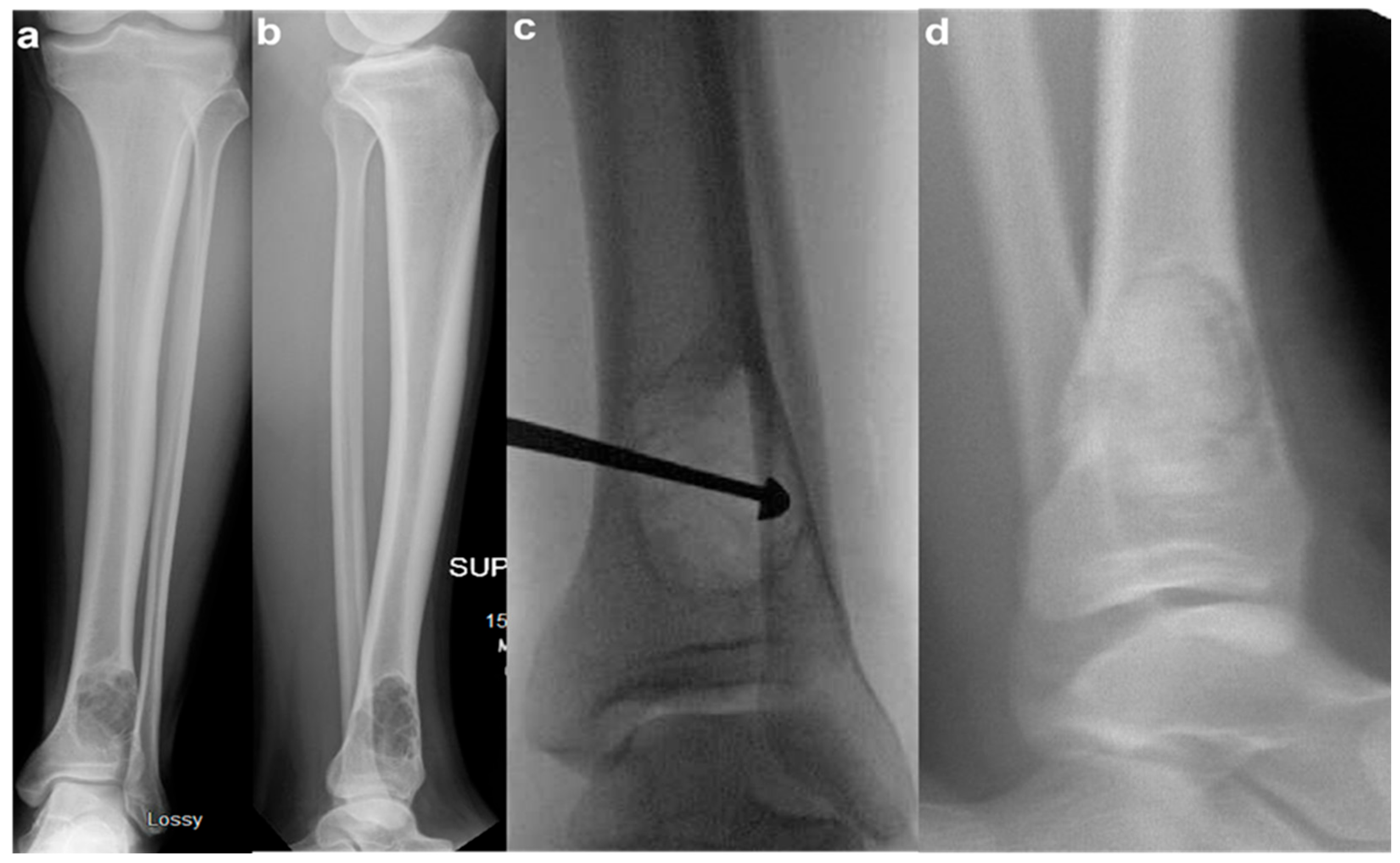
- Curettage and Bone Grafting: This is by far the most performed procedure. After curettage, the cavity is filled with autograft, allograft, or synthetic bone substitute [12]. While autograft is theoretically biologically superior, the donor site morbidity makes allograft the most used graft choice. In addition, the relatively high success rate and low risk of recurrence, make the risks of an additional incision with autograft prohibitive. Synthetic substitutes such as calcium sulfate and calcium phosphate can provide structural support to allow earlier weight bearing. Again, the risks of cementation, in the setting of a benign disease with good operative results, makes this a less common choice. Adjuvants are typically not used in the curettage stage for NOFs as they are in giant cell tumors of bone or aneurysmal bone cysts. NOFs are not locally aggressive, and the use of phenol or argon beam coagulation are not typically indicated to reduce recurrence. Phenol and argon have risk of local tissue damage, making them uncommonly used in the setting of NOFs. Figure 5 demonstrates pre-, intra-, and post-operative radiographic images of a 17-year-old boy with a large symptomatic distal tibia NOF who underwent a curettage and bone grafting procedure.
- Internal Fixation: For structural support in large lesions or fractures, internal fixation with plates or intramedullary nails may be necessary.
9. Future Directions
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sontag L, Pyle S. The appearance and nature of cyst-like areas in the distal femoral metaphyses of children. Am J Roentgenol. 1941;46(185).
- Jaffe HL, Lichtenstein L. Non-osteogenic fibroma of bone. Am J Pathol. 1942;18(2):205-221.
- Collier CD, Nelson GB, Conry KT, Kosmas C, Getty PJ, Liu RW. The Natural History of Benign Bone Tumors of the Extremities in Asymptomatic Children: A Longitudinal Radiographic Study. JBJS. 2021;103(7):575. [CrossRef]
- Baumhoer D, Kovac M, Sperveslage J, et al. Activating mutations in the MAP-kinase pathway define non-ossifying fibroma of bone. J Pathol. 2019;248(1):116-122. [CrossRef]
- Bovée JV, Hogendoorn PC. Non-ossifying fibroma: A RAS-MAPK driven benign bone neoplasm. J Pathol. 2019;248(2):127-130. [CrossRef]
- Ritschl P, Karnel F, Hajek P. Fibrous metaphyseal defects — determination of their origin and natural history using a radiomorphological study. Skeletal Radiol. 1988;17(1):8-15. [CrossRef]
- Rammanohar J, Zhang C, Thahir A, Krkovic M. Imaging of Non-ossifying Fibromas: A Case Series. Cureus. 13(3):e14102. [CrossRef]
- Baghdadi S, Nguyen JC, Arkader A. Nonossifying Fibroma of the Distal Tibia: Predictors of Fracture and Management Algorithm. J Pediatr Orthop. 2021;41(8):e671. [CrossRef]
- Goldin AN, Muzykewicz DA, Mubarak SJ. Nonossifying Fibromas: A Computed Tomography–based Criteria to Predict Fracture Risk. J Pediatr Orthop. 2020;40(2):e149-e154. [CrossRef]
- Patel RR, Damron TA. The Role of Surveillance in Predicting Fracture in Pediatric Patients With Incidentally Discovered Nonossifying Fibromas and Fibrous Cortical Defects: Is It Worth It? J Pediatr Orthop. 2024;44(6):395. [CrossRef]
- Herget GW, Mauer D, Krauß T, et al. Non-ossifying fibroma: natural history with an emphasis on a stage-related growth, fracture risk and the need for follow-up. BMC Musculoskelet Disord. 2016;17:147. [CrossRef]
- Arata MA, Peterson HA, Dahlin DC. Pathological fractures through non-ossifying fibromas. Review of the Mayo Clinic experience. JBJS. 1981;63(6):980.
- Andreacchio A, Alberghina F, Testa G, Canavese F. Surgical treatment for symptomatic non-ossifying fibromas of the lower extremity with calcium sulfate grafts in skeletally immature patients. Eur J Orthop Surg Traumatol. 2018;28(2):291-297. [CrossRef]
- Phemister DB. CHRONIC FIBROUS OSTEOMYELITIS. Ann Surg. 1929;90(4):756-764.
- Goldin A, Muzykewicz DA, Dwek J, Mubarak SJ. The aetiology of the non-ossifying fibroma of the distal femur and its relationship to the surrounding soft tissues. J Child Orthop. 2017;11(5):373-379. [CrossRef]
- Schindeler A, Little DG. Ras-MAPK signaling in osteogenic differentiation: friend or foe? J Bone Miner Res. 2006 Sep;21(9):1331-8. PMID: 16939391. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




