Submitted:
28 July 2025
Posted:
30 July 2025
You are already at the latest version
Abstract
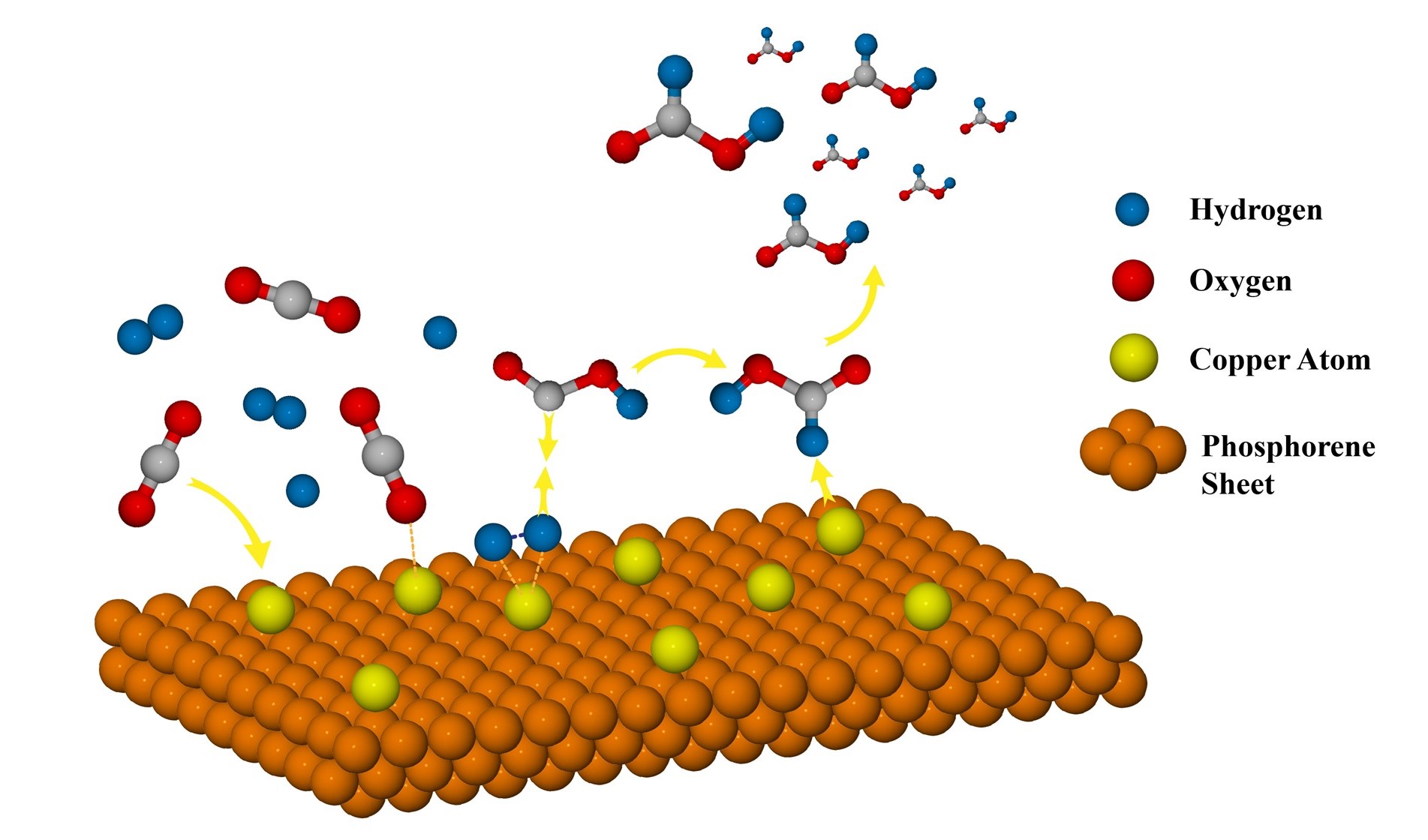
Keywords:
1. Introduction
2. Computational Methods
3. Results and Discussion
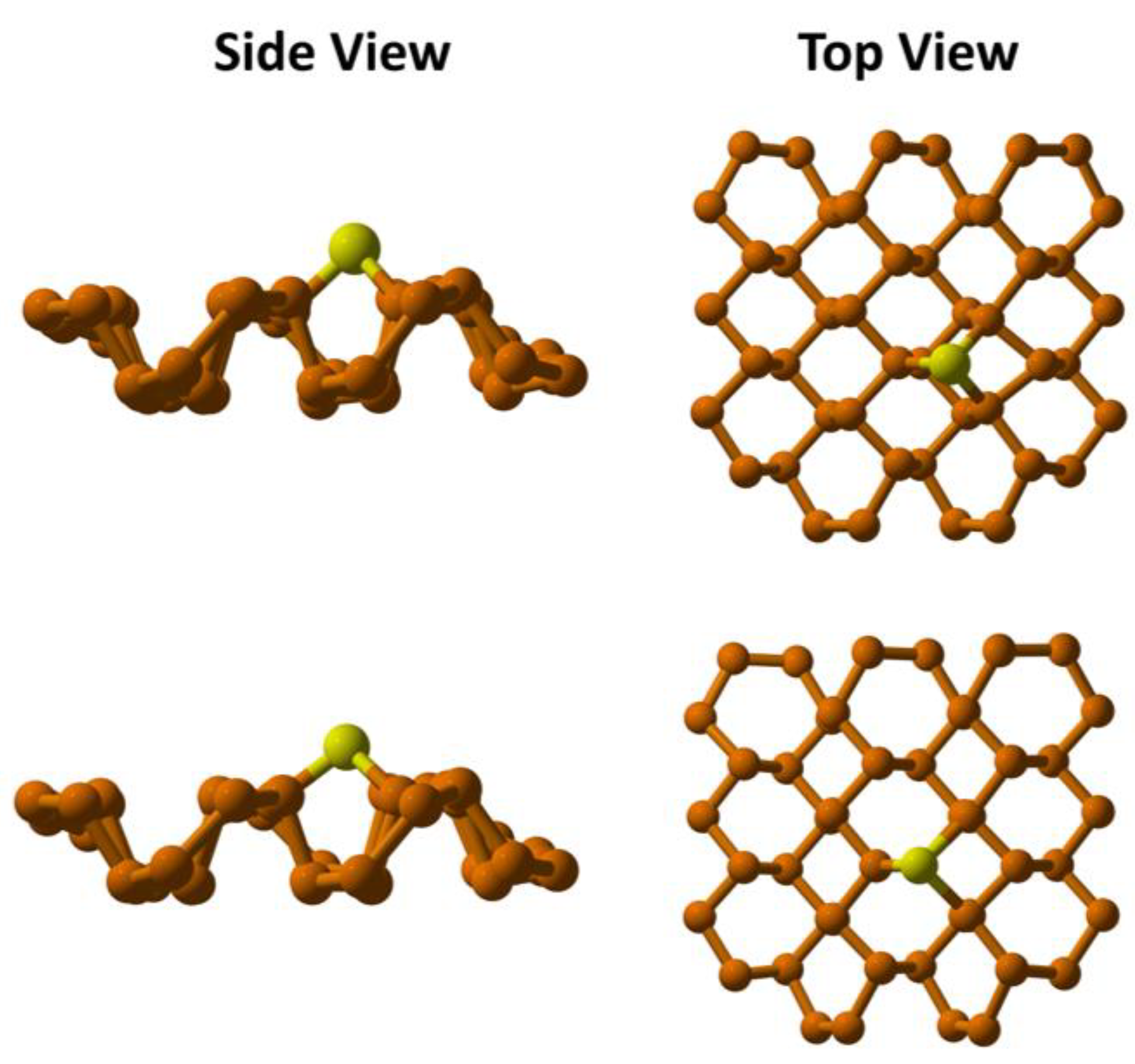
3.1. Geometry Optimization of Cu Embedded Phosphorene
- i.
- CP 88 (Cu--P27)
- ii.
- CP 95 (Cu--P10)
- iii.
- CP 101 (Cu--P23).
|
QTAIM analysis BCPs (Cu@Phosphorene) |
Density (ρ) | Laplacian (∇2ρ) | Lagrangian kinetic energy G(r) | Potential energy V(r) | Total energy density H(r) | Bond interaction energies |
| Cu-P27 | 0.06124 | 0.15367 | 0.05039 | −0.06237 | −0.01197 | −19.56 |
| Cu-P10 | 0.07704 | 0.2149 | 0.07078 | −0.08783 | −0.01705 | −27.56 |
| Cu-P23 | 0.05922 | 0.1468 | 0.048119 | −0.05952 | −0.01140 | −18.67 |
3.2. Adsorption of Different Species on Cu-Embedded Phosphorene
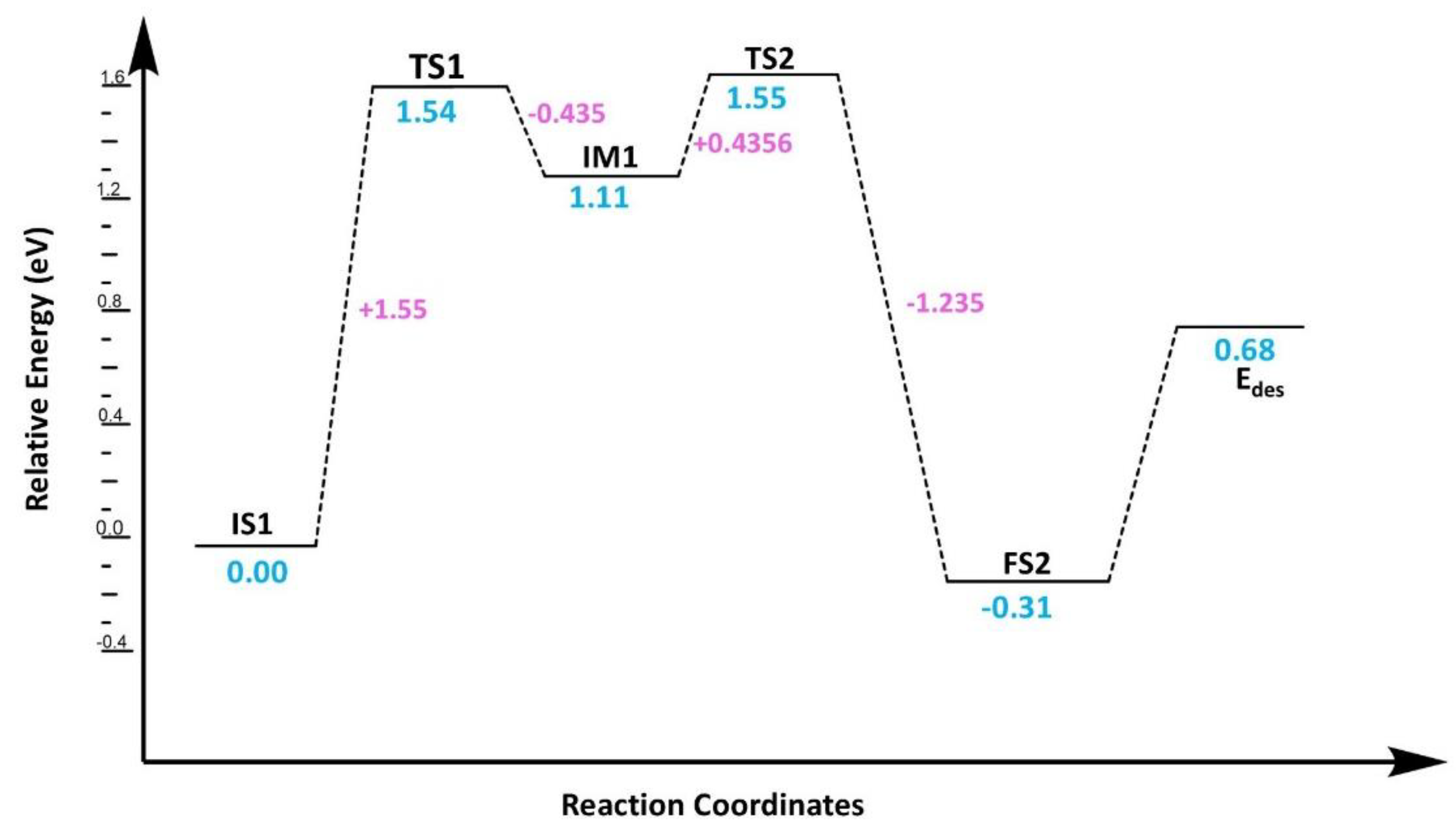
3.3. Reaction Mechanism for CO2 Reduction Using Cu@Phosphorene
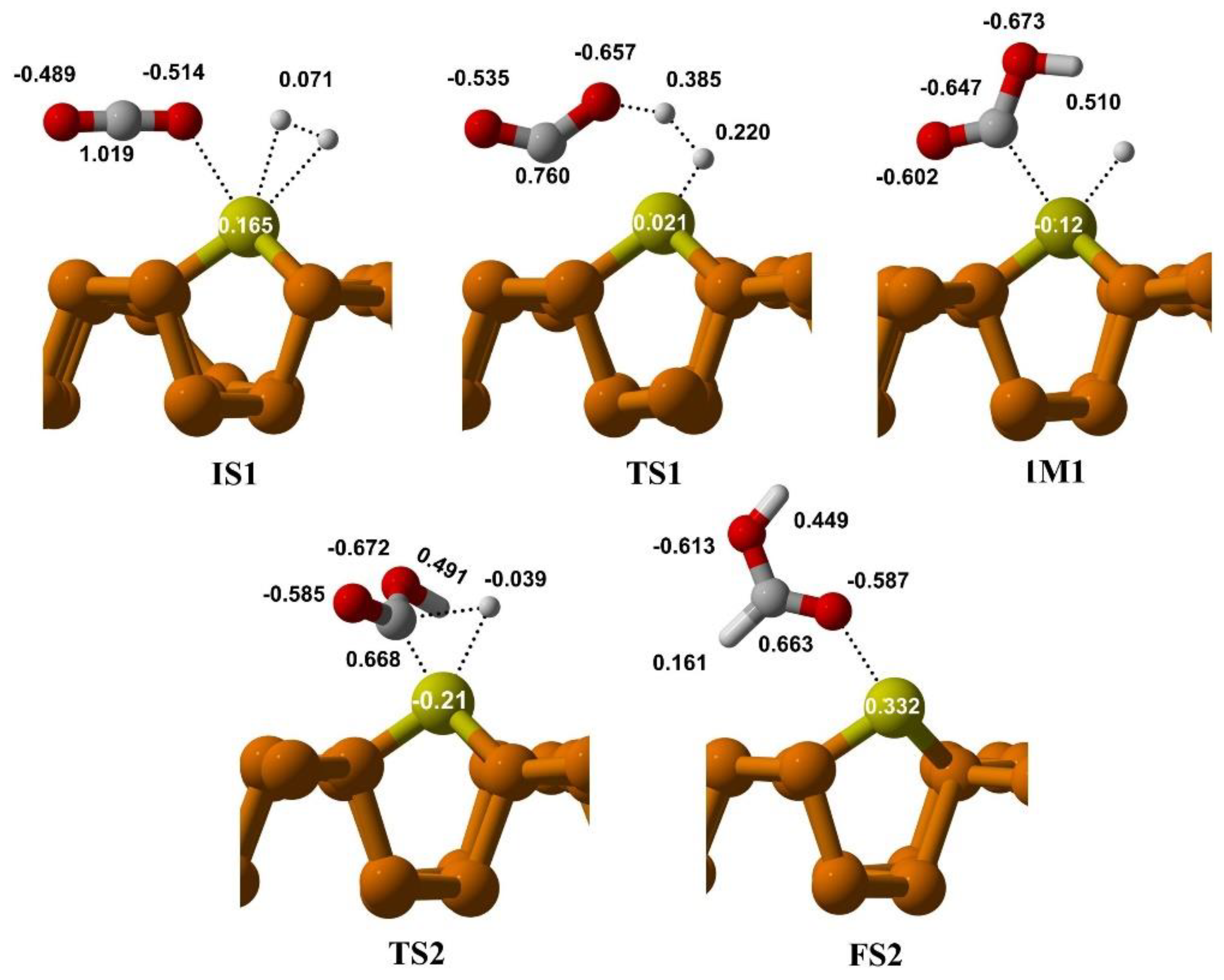
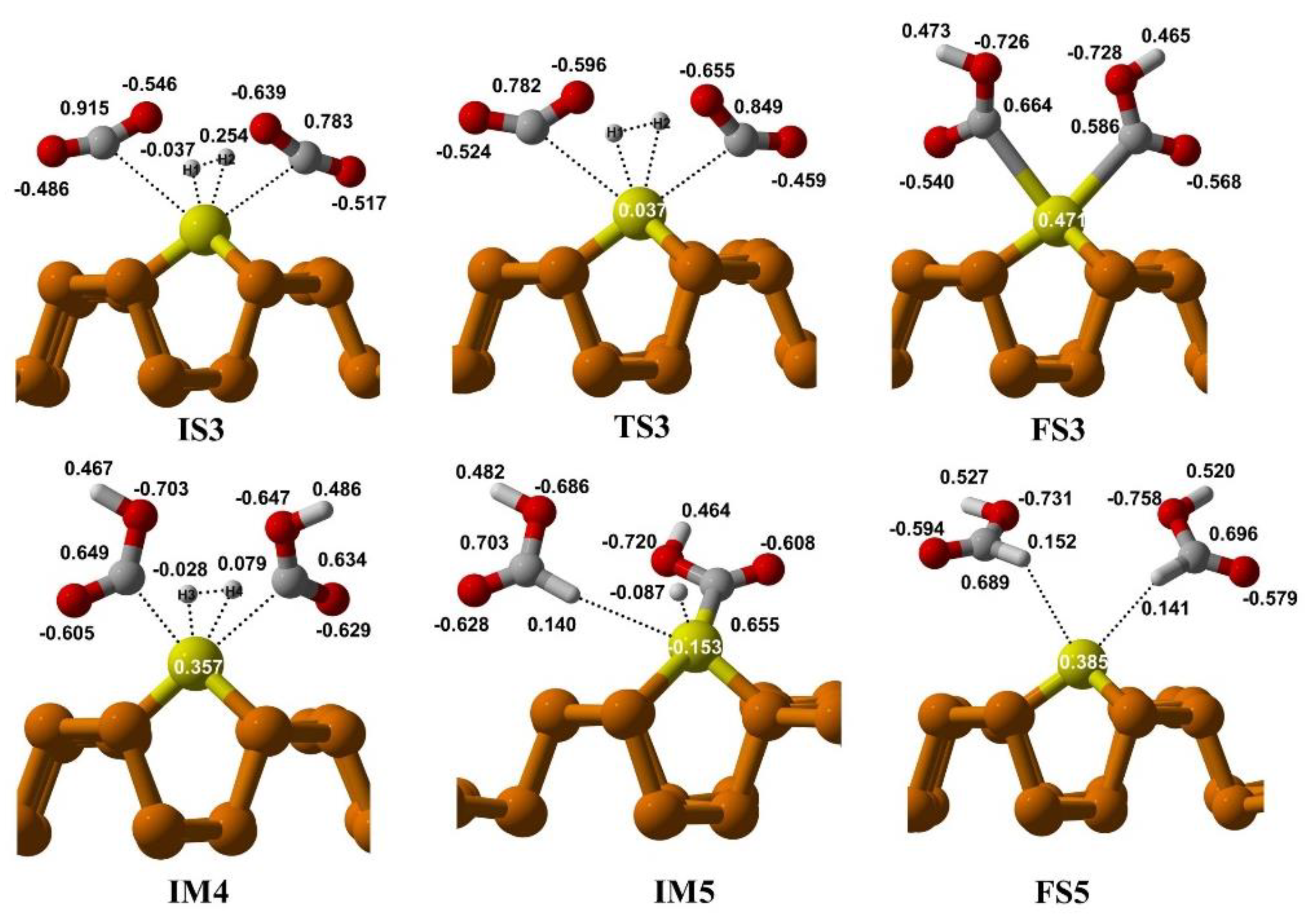
3.3.1. CO2 Reduction via Bi-Molecular (BM) Mechanism
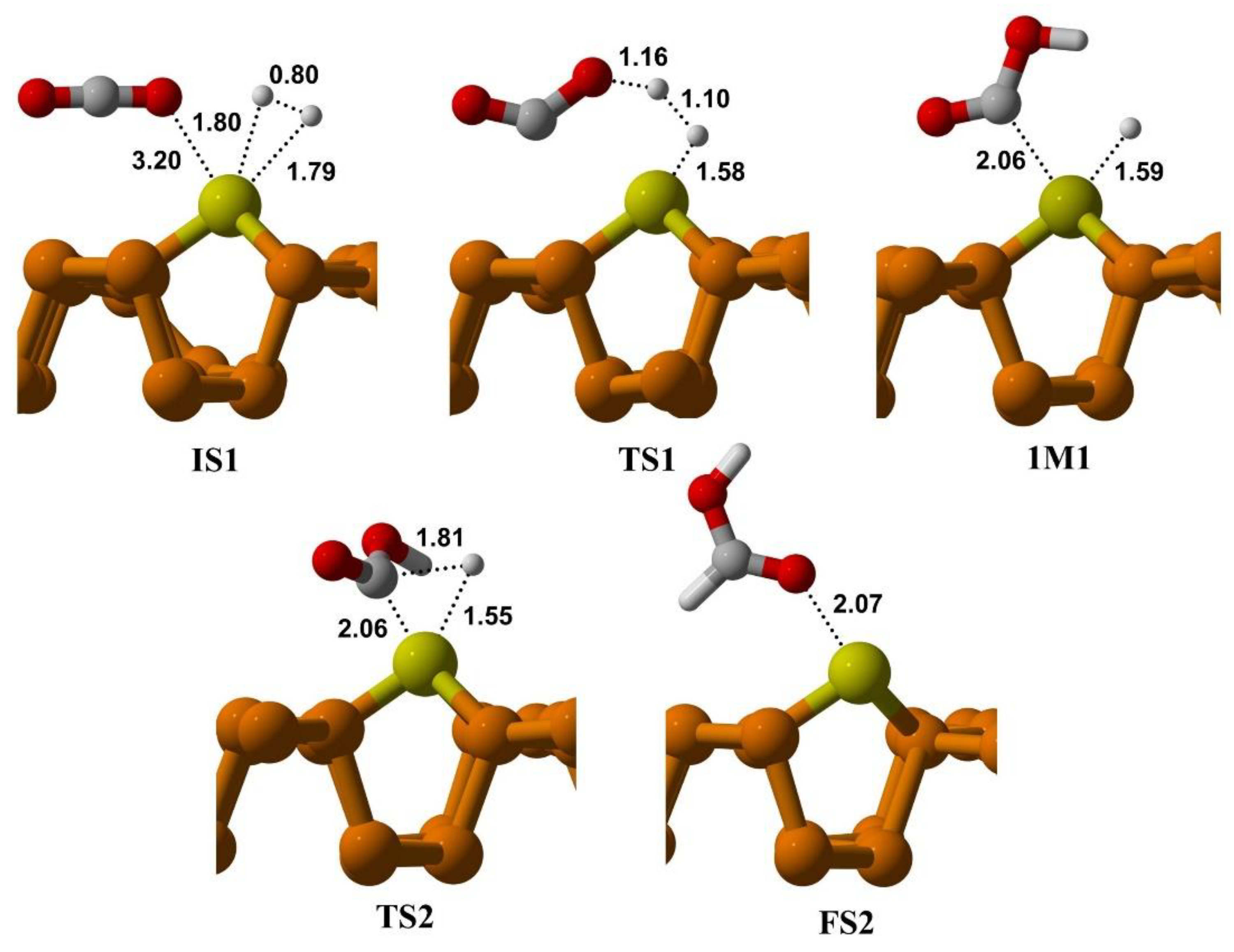
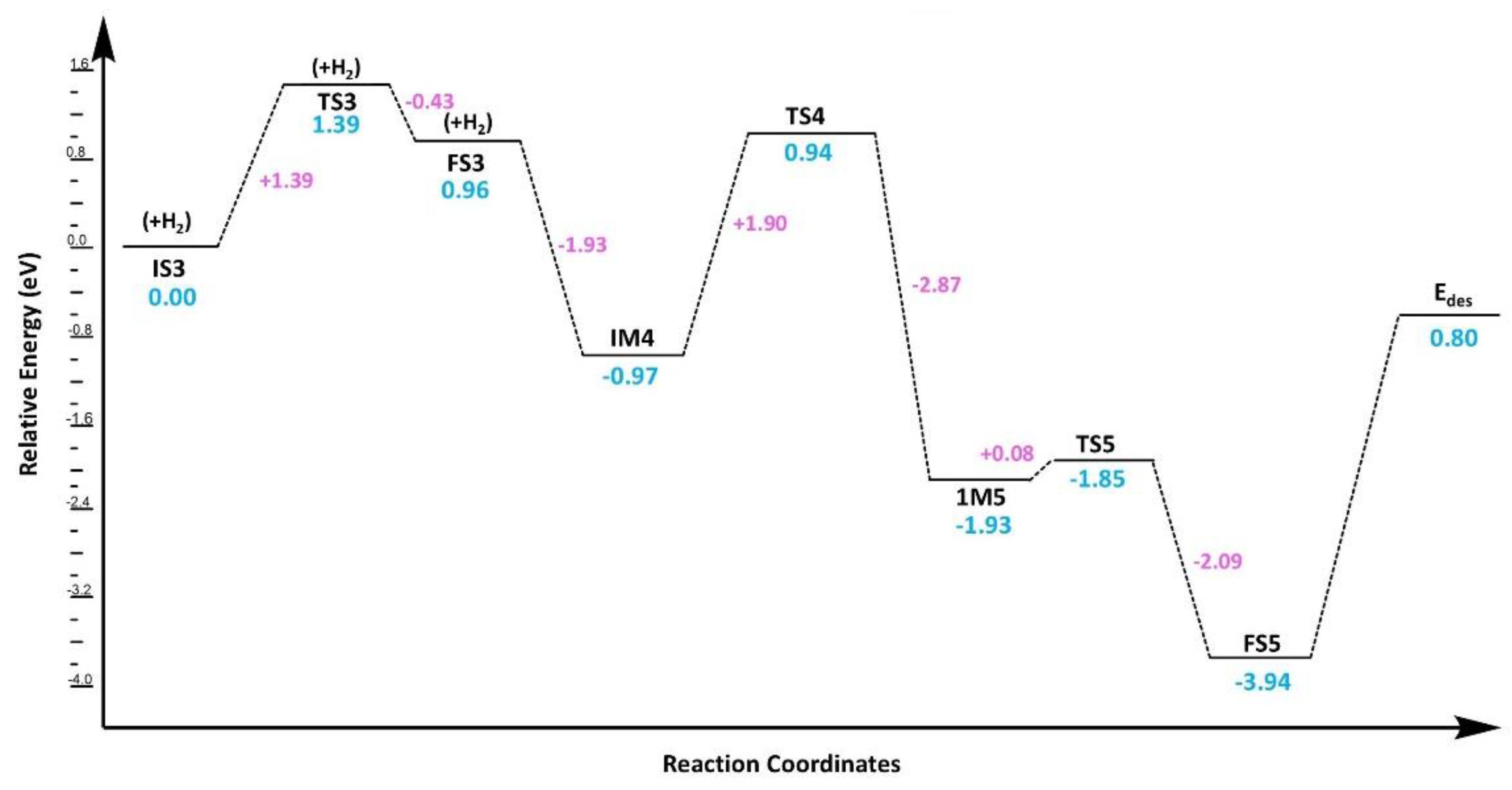
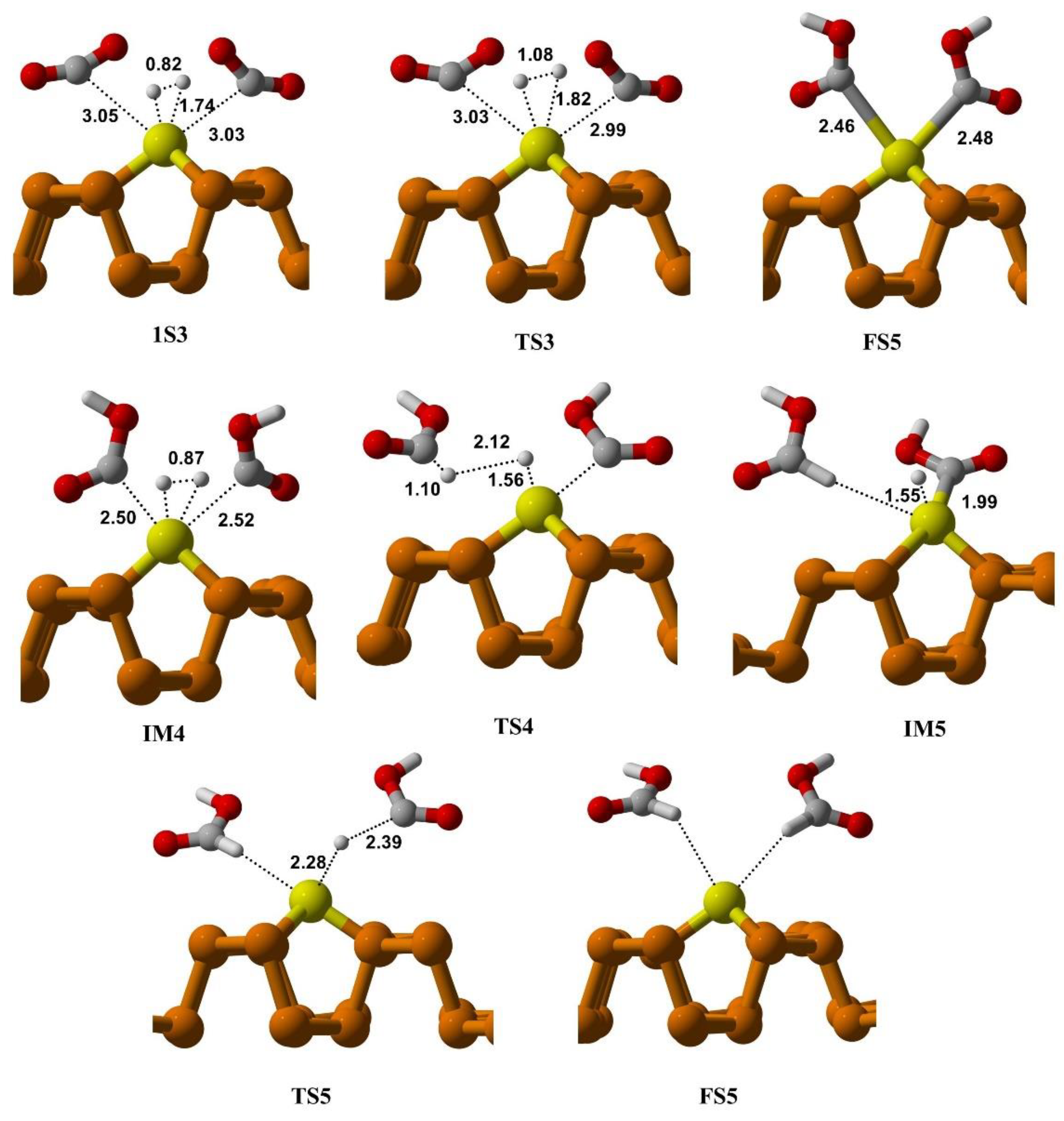
3.3.2. CO2 Reduction via Ter-Molecular (TER) Mechanism
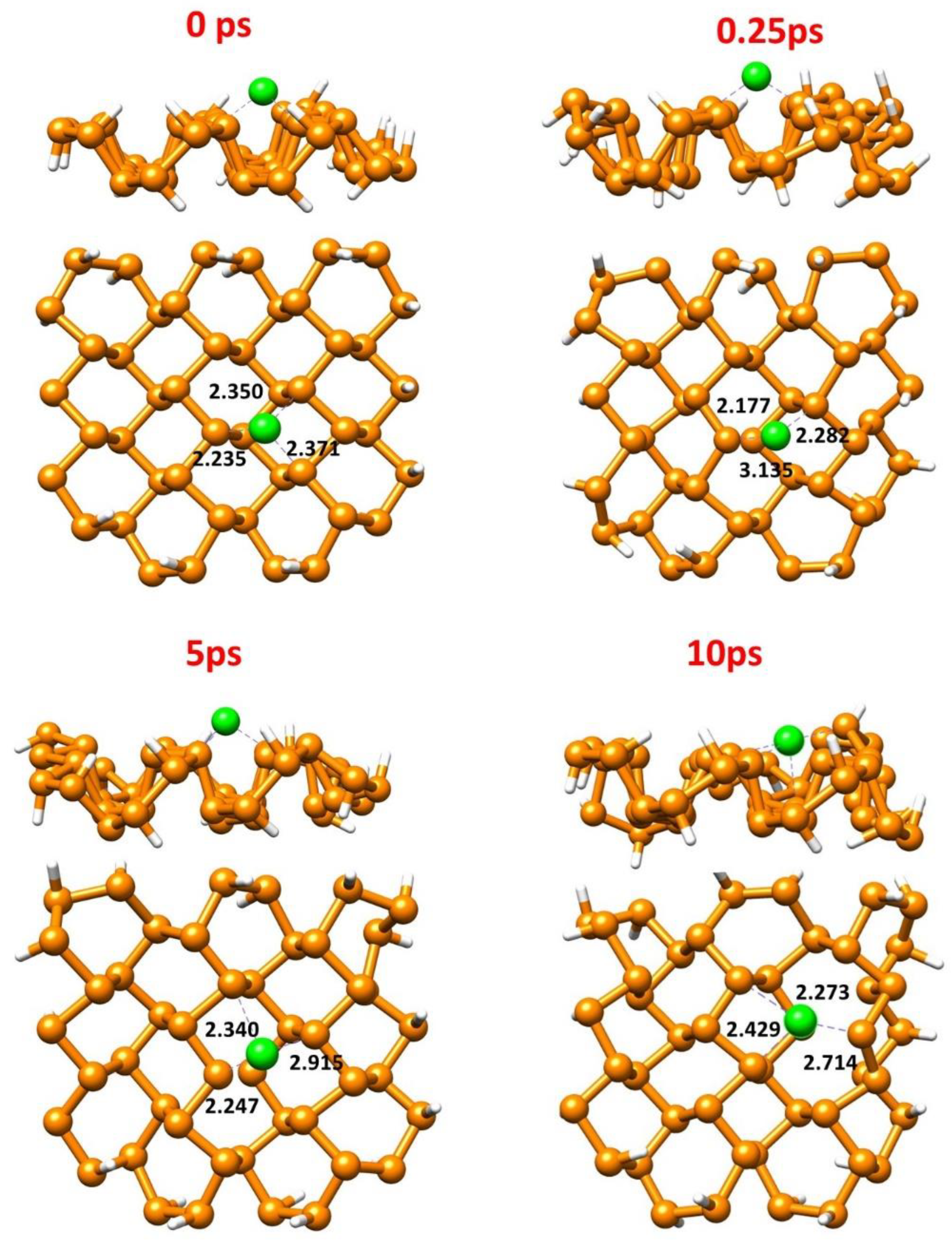
3.4. Ab Initio Molecular Dynamics (AIMD)
3.5. NBO Analysis
3.6. NCI Analysis
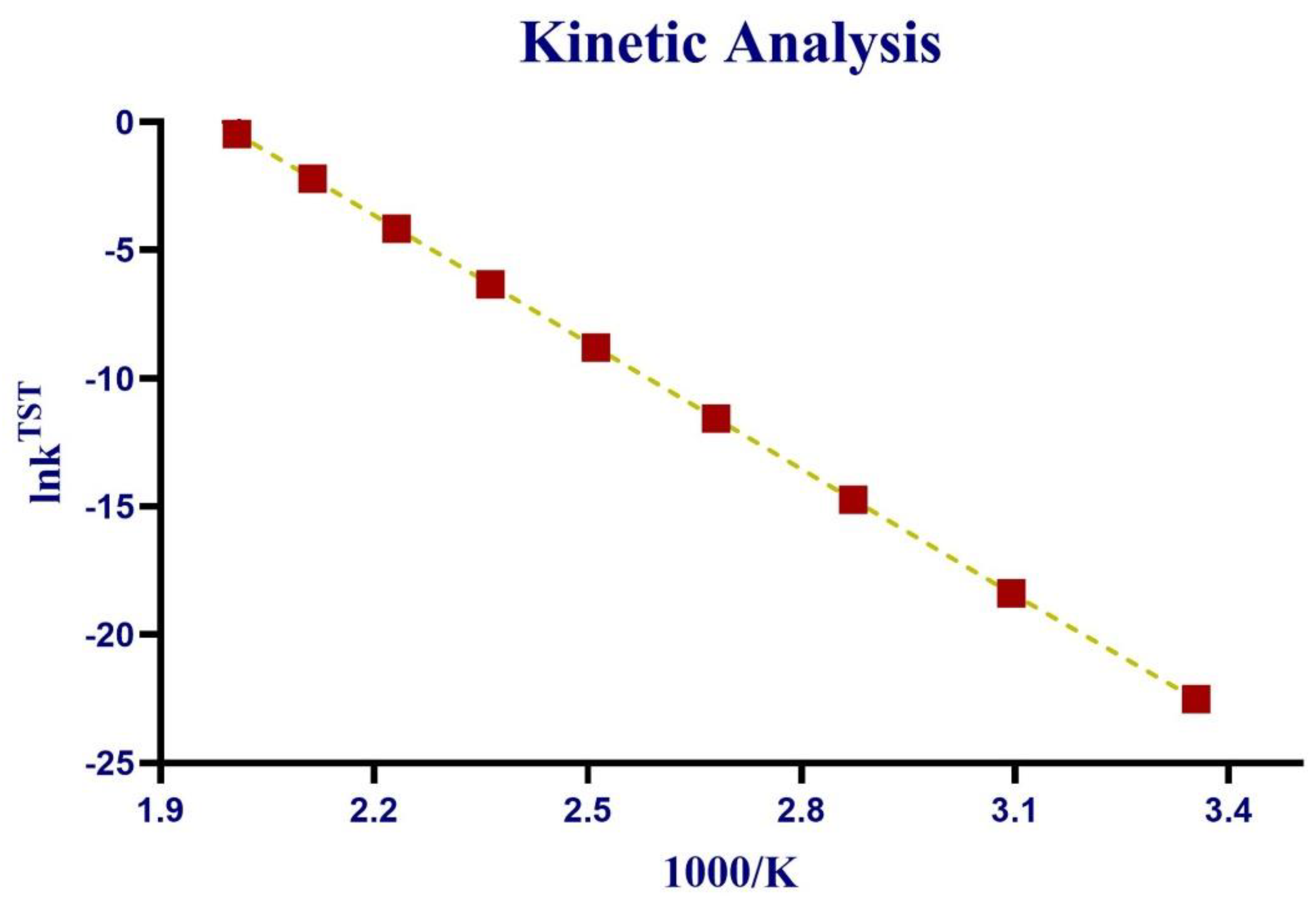
3.7. Kinetic Analysis
4. Conclusions
Supplementary Materials
Conflict of Interest
Acknowledgements
CRediT Author Statement
References
- Jing, L.; et al. Surface tuning for oxide-based nanomaterials as efficient photocatalysts. Chemical Society Reviews 2013, 42, 9509–9549. [Google Scholar] [CrossRef]
- Wang, Y.; et al. DFT Study on Sulfur-Doped g-C3N4 Nanosheets as a Photocatalyst for CO2 Reduction Reaction. The Journal of Physical Chemistry C 2018, 122, 7712–7719. [Google Scholar] [CrossRef]
- Esrafili, M.D.; Dinparast, L. , A DFT study on the catalytic hydrogenation of CO2 to formic acid over Ti-doped graphene nanoflake. Chemical Physics Letters 2017, 682, 49–54. [Google Scholar] [CrossRef]
- Usubharatana, P.; et al. Photocatalytic Process for CO2 Emission Reduction from Industrial Flue Gas Streams. Industrial & Engineering Chemistry Research 2006, 45, 2558–2568. [Google Scholar] [CrossRef]
- Esrafili, M.D.; Sharifi, F.; Dinparast, L. Catalytic hydrogenation of CO2 over Pt- and Ni-doped graphene: A comparative DFT study. Journal of Molecular Graphics and Modelling 2017, 77, 143–152. [Google Scholar] [CrossRef]
- He, M.; Sun, Y.; Han, B. Green Carbon Science: Scientific Basis for Integrating Carbon Resource Processing, Utilization, and Recycling. Angewandte Chemie International Edition 2013, 52, 9620–9633. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; et al. Recent advances in catalytic hydrogenation of carbon dioxide. Chemical Society Reviews 2011, 40, 3703–3727. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; et al. Single-atom catalysis: Bridging the homo- and heterogeneous catalysis. Chinese Journal of Catalysis 2018, 39, 893–898. [Google Scholar] [CrossRef]
- Wang, Y.; et al. TiC supported single-atom platinum catalyst for CO oxidation: A density functional theory study. Applied Surface Science 2018, 453, 159–165. [Google Scholar] [CrossRef]
- Butt, M.H.; et al. Cu-doped phosphorene as highly efficient single atom catalyst for CO oxidation: A DFT study. Molecular Catalysis 2021, 509, 111630. [Google Scholar] [CrossRef]
- Esrafili, M.D.; Rad, F.A. A DFT Study of Single-Atom Catalysis of CO Oxidation Using Carbon-Embedded Hexagonal Boron Nitride Monolayer. Chemistry Select 2018, 3, 7402–7409. [Google Scholar] [CrossRef]
- Esrafili, M.D.; Nejadebrahimi, B. Theoretical insights into hydrogenation of CO2 to formic acid over a single Co atom incorporated nitrogen-doped graphene: A DFT study. Applied Surface Science 2019, 475, 363–371. [Google Scholar] [CrossRef]
- He, T.; Zhang, C.; Du, A. Single-atom supported on graphene grain boundary as an efficient electrocatalyst for hydrogen evolution reaction. Chemical Engineering Science 2019, 194, 58–63. [Google Scholar] [CrossRef]
- Som, N.N.; Mankad, V.; Jha, P.K. Hydrogen evolution reaction: The role of arsenene nanosheet and dopant. International Journal of Hydrogen Energy, 2018, 43, 21634–21641. [Google Scholar] [CrossRef]
- Yaseen, F.; et al. The first row transition metal-corrole complexes as a single atom catalyst for electrochemical hydrogen evolution reaction: A DFT insight. International Journal of Hydrogen Energy 2024, 57, 1389–1397. [Google Scholar] [CrossRef]
- Liang, S.; Hao, C.; Shi, Y. The power of single--atom catalysis. ChemCatChem 2015, 7, 2559–2567. [Google Scholar] [CrossRef]
- Yuan, W.; et al. Single-atom catalysts for CO oxidation, CO2 reduction, and O2 electrochemistry. Journal of Energy Chemistry 2021. [Google Scholar] [CrossRef]
- Wang, A.; Li, J.; Zhang, T. Heterogeneous single-atom catalysis. Nature Reviews Chemistry 2018, 2, 65–81. [Google Scholar] [CrossRef]
- Wang, X.; et al. Rare--Earth Single--Atom Catalysts: A New Frontier in Photo/Electrocatalysis. Small Methods 2022, 6, 2200413. [Google Scholar] [CrossRef]
- Yang, X.-F.; et al. Single-Atom Catalysts: A New Frontier in Heterogeneous Catalysis. Accounts of Chemical Research 2013, 46, 1740–1748. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; et al. From double--atom catalysts to single--cluster catalysts: A new frontier in heterogeneous catalysis. Nano Select 2021, 2, 251–270. [Google Scholar] [CrossRef]
- Liu, J. Single-atom catalysis for a sustainable and greener future. Current Opinion in Green and Sustainable Chemistry 2020, 22, 54–64. [Google Scholar] [CrossRef]
- Zhang, B.; et al. Versatile Applications of Metal Single--Atom@ 2D Material Nanoplatforms. Advanced Science 2019, 6, 1901787. [Google Scholar] [CrossRef]
- Kwon, K.C.; et al. Electrocatalytic water splitting and CO2 reduction: sustainable solutions via single--atom catalysts supported on 2D materials. Small Methods 2019, 3, 1800492. [Google Scholar] [CrossRef]
- Luo, Z.; et al. Chemically activating MoS2 via spontaneous atomic palladium interfacial doping towards efficient hydrogen evolution. Nature communications 2018, 9, 2120. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; et al. Triggering the electrocatalytic hydrogen evolution activity of the inert two-dimensional MoS2 surface via single-atom metal doping. Energy & Environmental Science 2015, 8, 1594–1601. [Google Scholar]
- Deng, J.; et al. Multiscale structural and electronic control of molybdenum disulfide foam for highly efficient hydrogen production. Nature communications 2017, 8, 14430. [Google Scholar] [CrossRef]
- Wang, Y.; et al. Catalysis with Two-Dimensional Materials Confining Single Atoms: Concept, Design, and Applications. Chemical Reviews 2019, 119, 1806–1854. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.; et al. A single iron site confined in a graphene matrix for the catalytic oxidation of benzene at room temperature. Science Advances 2015, 1, e1500462. [Google Scholar] [CrossRef] [PubMed]
- Liu, J. Catalysis by Supported Single Metal Atoms. ACS Catalysis 2017, 7, 34–59. [Google Scholar] [CrossRef]
- Alarawi, A.; Ramalingam, V.; He, J.-H. Recent advances in emerging single atom confined two-dimensional materials for water splitting applications. Materials Today Energy 2019, 11, 1–23. [Google Scholar] [CrossRef]
- Yan, H.; et al. Single-Atom Pd1/Graphene Catalyst Achieved by Atomic Layer Deposition: Remarkable Performance in Selective Hydrogenation of 1,3-Butadiene. Journal of the American Chemical Society 2015, 137, 10484–10487. [Google Scholar] [CrossRef]
- Lu, S.; Lou, F.; Yu, Z. Recent Progress in Two-Dimensional Materials for Electrocatalytic CO2 Reduction. Catalysts 2022, 12. [Google Scholar] [CrossRef]
- Tao, L.; et al. Creating coordinatively unsaturated metal sites in metal-organic-frameworks as efficient electrocatalysts for the oxygen evolution reaction: Insights into the active centers. Nano Energy 2017, 41, 417–425. [Google Scholar] [CrossRef]
- Xue, Y.; et al. Anchoring zero valence single atoms of nickel and iron on graphdiyne for hydrogen evolution. Nature communications 2018, 9, 1460. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; et al. Identification of High--Performance Single--Atom Mxenes Catalysts for Low--Temperature CO Oxidation. Advanced Theory Simulations 2019, 2, 1900006. [Google Scholar] [CrossRef]
- Zhang, W.; et al. Recent Advances in Phosphorene: Structure, Synthesis, and Properties. Small 2024, 20, 2303115. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; et al. Black phosphorene as a hole extraction layer boosting solar water splitting of oxygen evolution catalysts. Nature Communications 2019, 10, 2001. [Google Scholar] [CrossRef]
- Nair, A.S.; Ahuja, R.; Pathak, B. Unraveling the single-atom electrocatalytic activity of transition metal-doped phosphorene. Nanoscale Advances 2020, 2, 2410–2421. [Google Scholar] [CrossRef]
- Yin, T.; et al. Advancing Applications of Black Phosphorus and BP-Analog Materials in Photo/Electrocatalysis through Structure Engineering and Surface Modulation. Advanced Science 2020, 7, 2001431. [Google Scholar] [CrossRef]
- Amrillah, T.; et al. MXene-Based Photocatalysts and Electrocatalysts for CO2 Conversion to Chemicals. Transactions of Tianjin University 2022, 28, 307–322. [Google Scholar] [CrossRef]
- Khaidar, D.M.; et al. Transition metal dichalcogenides-based catalysts for CO2 conversion: An updated review. International Journal of Hydrogen Energy 2024, 68, 35–50. [Google Scholar] [CrossRef]
- Ren, X.; et al. Properties, preparation and application of black phosphorus/phosphorene for energy storage: a review. Journal of Materials Science 2017, 52, 10364–10386. [Google Scholar] [CrossRef]
- Mu, X.; Wang, J.; Sun, M. Two-dimensional black phosphorus: physical properties and applications. Materials Today Physics 2019, 8, 92–111. [Google Scholar] [CrossRef]
- Wang, J.; et al. Recent progress, developing strategies, theoretical insights, and perspectives towards high-performance copper single atom electrocatalysts. Materials Today Energy 2021, 21, 100761. [Google Scholar] [CrossRef]
- Zhang, H.; et al. Atomic-Level Regulation of Cu-Based Electrocatalyst for Enhancing Oxygen Reduction Reaction: From Single Atoms to Polymetallic Active Sites. Small 2024, 20, 2307384. [Google Scholar] [CrossRef]
- Hao, C.; et al. Hydrogenation of CO2 to formic acid on supported ruthenium catalysts. Catalysis today 2011, 160, 184–190. [Google Scholar] [CrossRef]
- Mosconi, E. and F.D. Angelis, DFT Investigation of Ligand-Based Reduction of CO2 to CO on an Anionic Niobium Nitride Complex: Reaction Mechanism and Role of the Na+ Counterion. Organometallics 2011, 30, 4838–4846. [Google Scholar] [CrossRef]
- Peng, G.; et al. CO2 hydrogenation to formic acid on Ni(110). Surface Science 2012, 606, 1050–1055. [Google Scholar] [CrossRef]
- Song, H.; et al. Developments in stability and passivation strategies for black phosphorus. Nano Research 2021, 14, 4386–4397. [Google Scholar] [CrossRef]
- Ryder, C.R.; et al. Covalent functionalization and passivation of exfoliated black phosphorus via aryl diazonium chemistry. Nature Chemistry 2016, 8, 597–602. [Google Scholar] [CrossRef]
- Koenig, S.P.; et al. Electron Doping of Ultrathin Black Phosphorus with Cu Adatoms. Nano Letters 2016, 16, 2145–2151. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; et al. Te-Doped Black Phosphorus Field-Effect Transistors. Advanced materials (Deerfield Beach, Fla.) 2016, 28, 9408–9415. [Google Scholar] [CrossRef]
- Caporali, M.; et al. Enhanced ambient stability of exfoliated black phosphorus by passivation with nickel nanoparticles. Nanotechnology 2020, 31, 275708. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; et al. Te-Doped Black Phosphorus Field-Effect Transistors. Advanced Materials 2016, 28, 9408–9415. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; et al. Twin Heterostructure Engineering and Facet Effect Boosts Efficient Reduction CO2-to-Ethanol at Low Potential on Cu2O@Cu2S Catalysts. ACS Catalysis 2024, 14, 3266–3277. [Google Scholar] [CrossRef]
- Zhang, H.-p.; et al. CO2 reduction to CH4 on Cu-doped phosphorene: a first-principles study. Nanoscale 2021, 13, 20541–20549. [Google Scholar] [CrossRef]
- Frisch, M.; et al. Gaussian 16 Rev. B. 01 Release Notes. 2016, Wallingford, CT. [Google Scholar]
- Legault, C. CYLview, 1. 0 b. 2009, 436, 437. [Google Scholar]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys Rev Lett 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Wang, Y. Generalized gradient approximation for the exchange-correlation hole of a many-electron system. Phys Rev B Condens Matter 1996, 54, 16533–16539. [Google Scholar] [CrossRef] [PubMed]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys Chem Chem Phys 2005, 7, 3297–305. [Google Scholar] [CrossRef]
- Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J Comput Chem 2006, 27, 1787–99. [Google Scholar] [CrossRef]
- Grimme, S.; et al. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys 2010, 132, 154104. [Google Scholar] [CrossRef]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J Comput Chem 2011, 32, 1456–65. [Google Scholar] [CrossRef]
- Iqbal, S.; et al. Transition metals incorporated on phosphorene sheet as cost-effective single atom catalysts for hydrogen evolution reaction: A DFT study. Computational and Theoretical Chemistry 2023, 1220, 113998. [Google Scholar] [CrossRef]
- Zhang, Q.; et al. Theoretical Insights into the Favorable Functionalized Ti2C-Based MXenes for Lithium–Sulfur Batteries. ACS Omega 2020, 5, 29272–29283. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F. Multiwfn: a multifunctional wavefunction analyzer. Journal of computational chemistry 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K.J. VMD: visual molecular dynamics. Journal of molecular graphics 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Yan, G.; et al. CO2 hydrogenation to formic acid over platinum cluster doped defective graphene: A DFT study. Applied Surface Science 2020, 517, 146200. [Google Scholar] [CrossRef]
- Neese, F.; et al. The ORCA quantum chemistry program package. The Journal of Chemical Physics 2020, 152. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; et al. NO Oxidation Using H2O2 at a Single-Atom Iron Catalyst. The Journal of Physical Chemistry C 2023, 127, 13011–13020. [Google Scholar] [CrossRef]
- Yang, W.; et al. Single-atom iron as a promising low-temperature catalyst for selective catalytic reduction of NOx with NH3: A theoretical prediction. Fuel 2021, 302, 121041. [Google Scholar] [CrossRef]
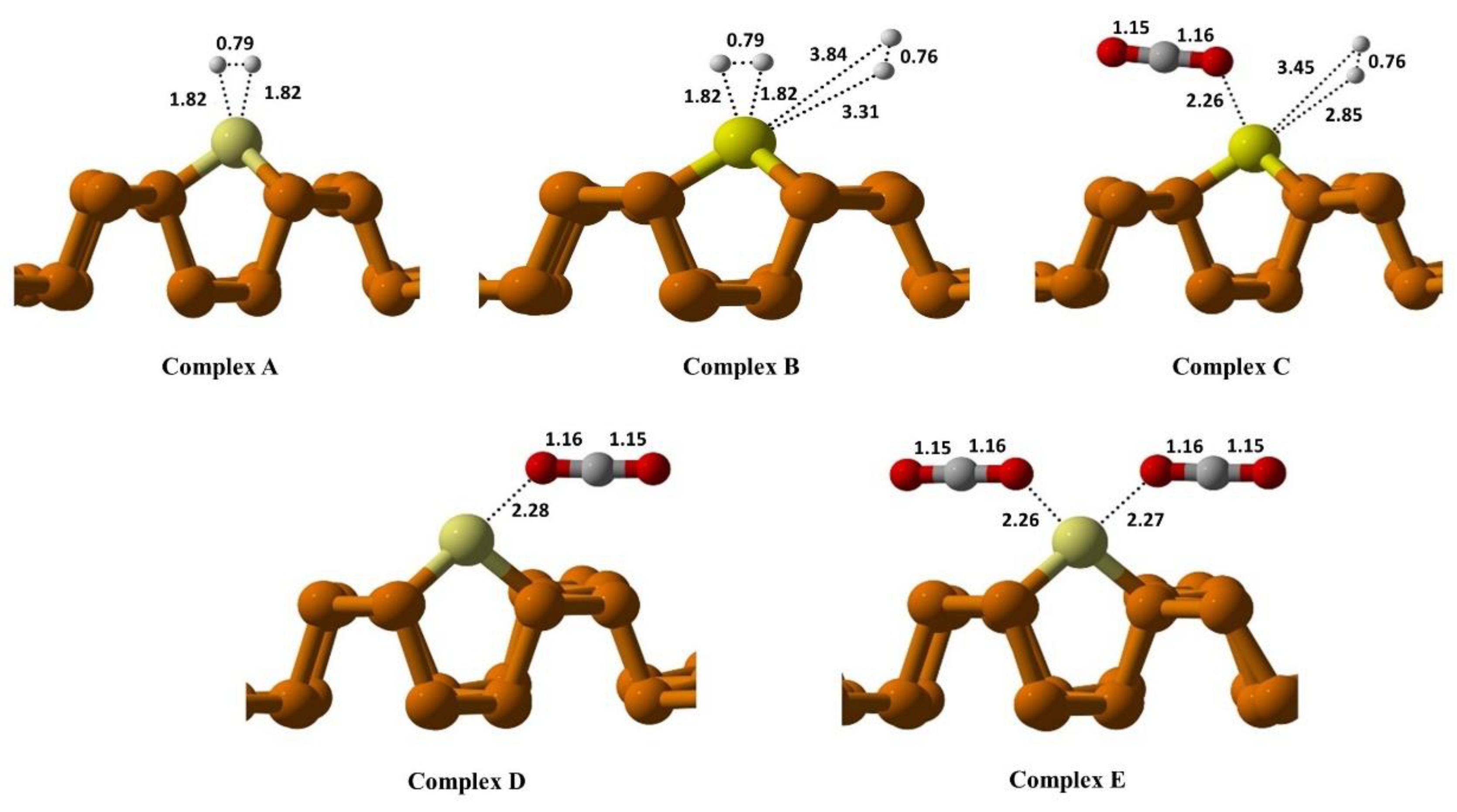
| Substrate | Species | Eads(eV) | Bond Lengths (Å) |
| Cu@Phosphorene | CO2 | −0.19 | O—C—O 1.152, 1.167 |
| H2 | −0.06 | H—H 0.79 | |
| 2H2 | −0.15 | H-H 0.79 H-H 0.79 |
|
| H2-CO2 | −0.26 | O—C—O 1.151, 1.169 H—H 0.76 |
|
| 2CO2 | −0.59 | O—C—O 1.153, 1.167 O—C—O 1.153, 1.166 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




