Submitted:
28 July 2025
Posted:
28 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
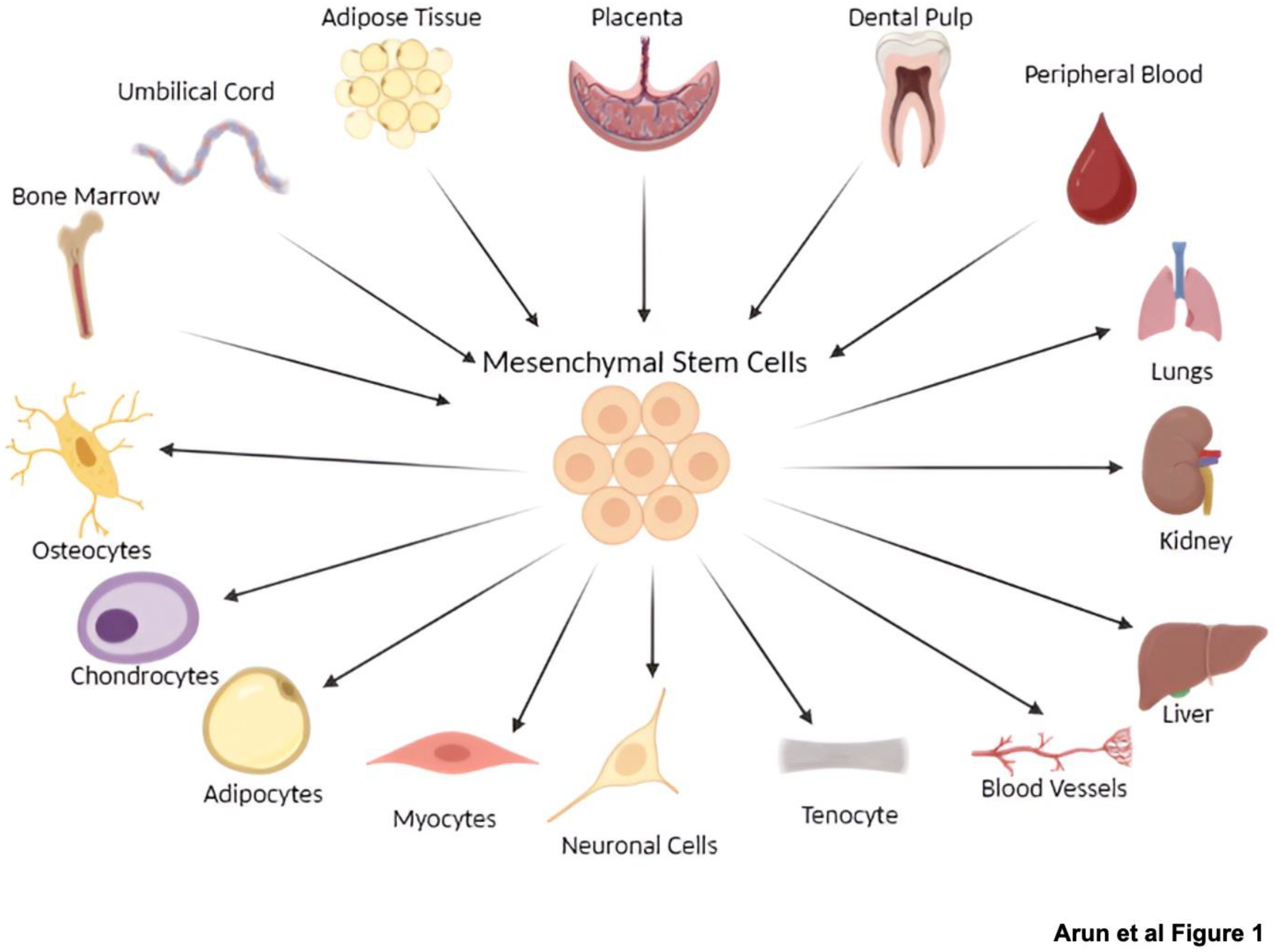
2. Mesenchymal Stem Cells and Exosomes: An Overview
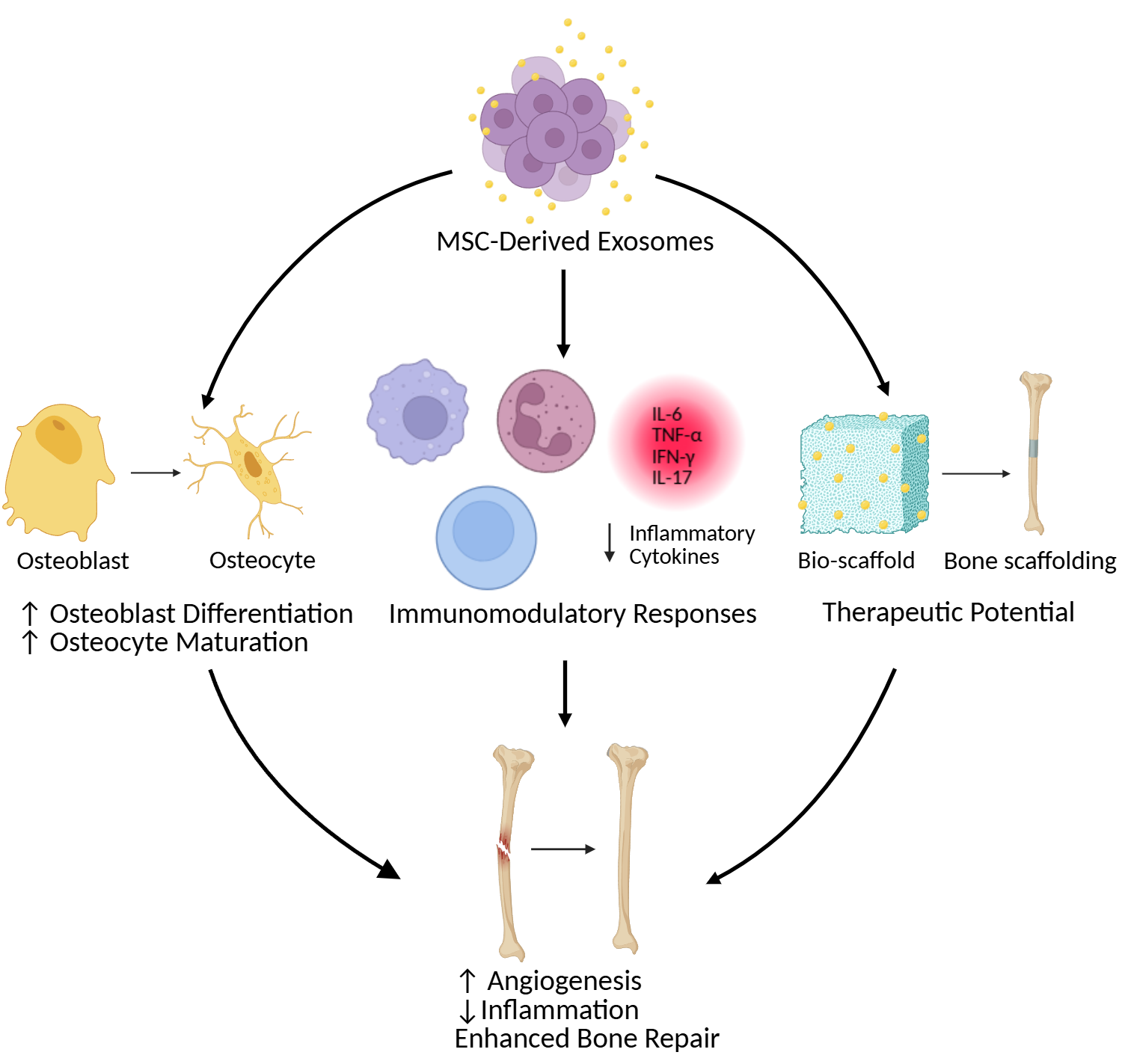
2. MSC-Derived Exosomes in Bone Regeneration
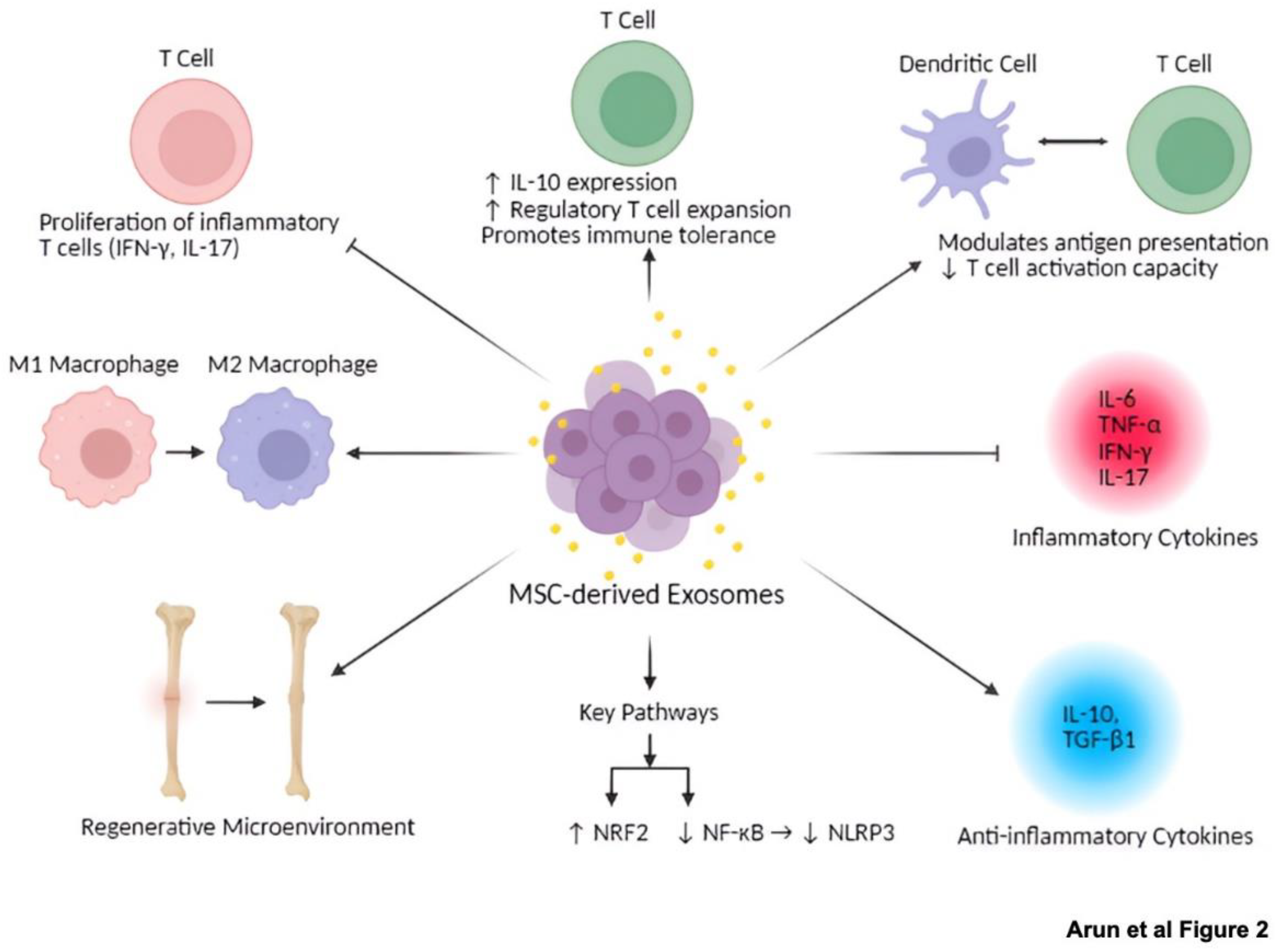
3. Immunomodulatory Properties of MSC-Derived Exosomes
4. Evidence of MSC-Derived Exosomes Promoting Osteocyte Differentiation
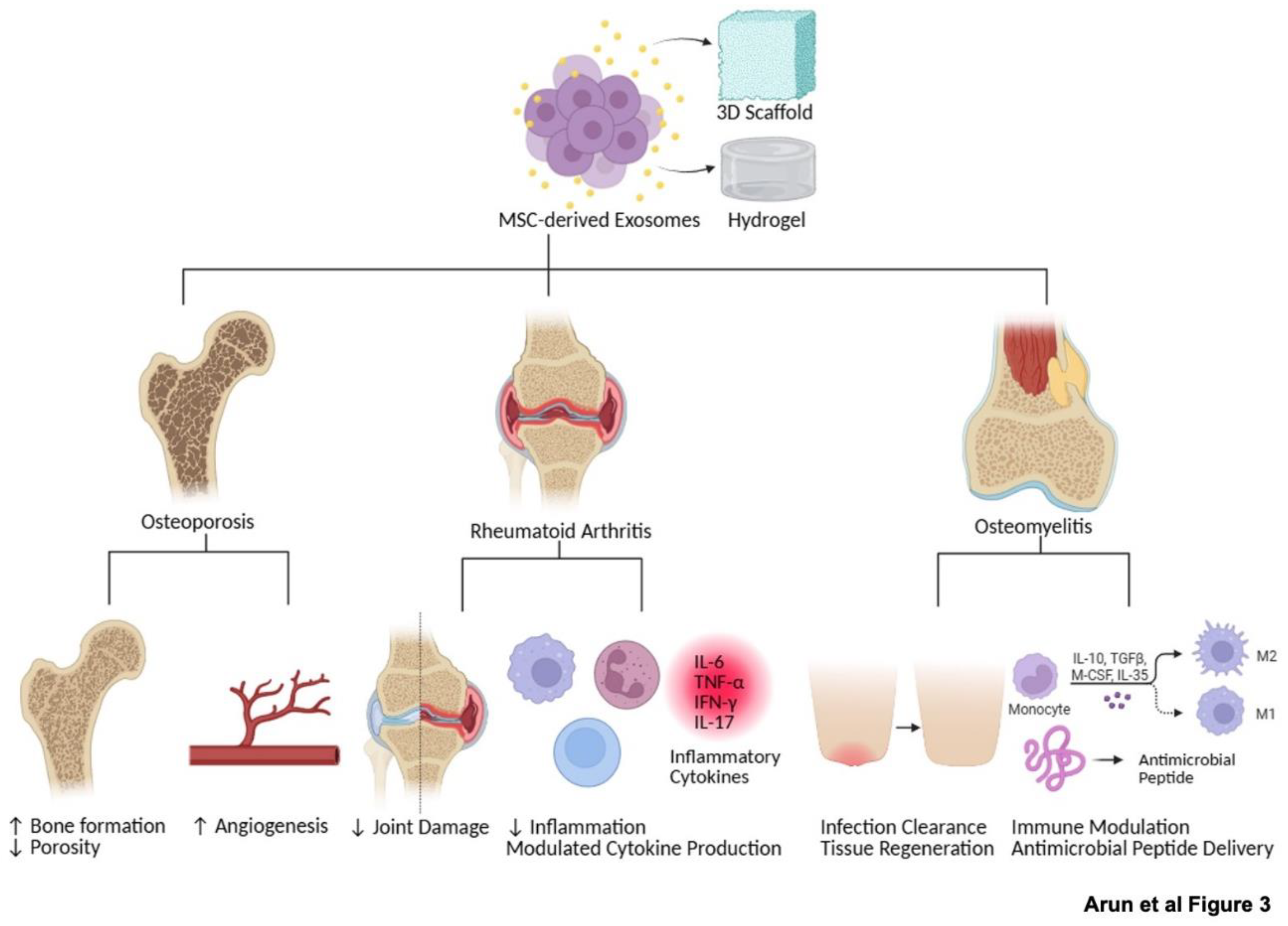
5. Therapeutic Potential and Clinical Implications
6. Challenges and Future Directions
7. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Florencio-Silva, R.; Sasso, G.R.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed Res Int 2015, 2015, 421746. [CrossRef]
- Raggatt, L.J.; Partridge, N.C. Cellular and molecular mechanisms of bone remodeling. J Biol Chem 2010, 285, 25103-25108. [CrossRef]
- Bonewald, L.F. The amazing osteocyte. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 2011, 26, 229-238. [CrossRef]
- Cao, W.; Helder, M.N.; Bravenboer, N.; Wu, G.; Jin, J.; Ten Bruggenkate, C.M.; Klein-Nulend, J.; Schulten, E. Is There a Governing Role of Osteocytes in Bone Tissue Regeneration? Curr Osteoporos Rep 2020, 18, 541-550. [CrossRef]
- Tresguerres, F.G.F.; Torres, J.; López-Quiles, J.; Hernández, G.; Vega, J.A.; Tresguerres, I.F. The osteocyte: A multifunctional cell within the bone. Ann Anat 2020, 227, 151422. [CrossRef]
- Marahleh, A.; Kitaura, H.; Ohori, F.; Noguchi, T.; Mizoguchi, I. The osteocyte and its osteoclastogenic potential. Front Endocrinol (Lausanne) 2023, 14, 1121727. [CrossRef]
- Du, J.H.; Lin, S.X.; Wu, X.L.; Yang, S.M.; Cao, L.Y.; Zheng, A.; Wu, J.N.; Jiang, X.Q. The Function of Wnt Ligands on Osteocyte and Bone Remodeling. J Dent Res 2019, 98, 930-938. [CrossRef]
- Niedźwiedzki, T.; Filipowska, J. Bone remodeling in the context of cellular and systemic regulation: the role of osteocytes and the nervous system. J Mol Endocrinol 2015, 55, R23-36. [CrossRef]
- Forsberg, M.H.; Kink, J.A.; Hematti, P.; Capitini, C.M. Mesenchymal Stromal Cells and Exosomes: Progress and Challenges. Front Cell Dev Biol 2020, 8, 665. [CrossRef]
- Akbar, N.; Razzaq, S.S.; Salim, A.; Haneef, K. Mesenchymal Stem Cell-Derived Exosomes and Their MicroRNAs in Heart Repair and Regeneration. J Cardiovasc Transl Res 2024, 17, 505-522. [CrossRef]
- Hassanzadeh, A.; Rahman, H.S.; Markov, A.; Endjun, J.J.; Zekiy, A.O.; Chartrand, M.S.; Beheshtkhoo, N.; Kouhbanani, M.A.J.; Marofi, F.; Nikoo, M.; et al. Mesenchymal stem/stromal cell-derived exosomes in regenerative medicine and cancer; overview of development, challenges, and opportunities. Stem Cell Res Ther 2021, 12, 297. [CrossRef]
- Wang, Z.; Zhao, Z.; Gao, B.; Zhang, L. Exosome mediated biological functions within skeletal microenvironment. Front Bioeng Biotechnol 2022, 10, 953916. [CrossRef]
- Torrecillas-Baena, B.; Pulido-Escribano, V.; Dorado, G.; Gálvez-Moreno, M.; Camacho-Cardenosa, M.; Casado-Díaz, A. Clinical Potential of Mesenchymal Stem Cell-Derived Exosomes in Bone Regeneration. J Clin Med 2023, 12. [CrossRef]
- Baharlooi, H.; Salehi, Z.; Minbashi Moeini, M.; Rezaei, N.; Azimi, M. Immunomodulatory Potential of Human Mesenchymal Stem Cells and their Exosomes on Multiple Sclerosis. Adv Pharm Bull 2022, 12, 389-397. [CrossRef]
- Che, J.; Wang, H.; Dong, J.; Wu, Y.; Zhang, H.; Fu, L.; Zhang, J. Human umbilical cord mesenchymal stem cell-derived exosomes attenuate neuroinflammation and oxidative stress through the NRF2/NF-κB/NLRP3 pathway. CNS Neurosci Ther 2024, 30, e14454. [CrossRef]
- He, J.G.; Wu, X.X.; Li, S.; Yan, D.; Xiao, G.P.; Mao, F.G. Exosomes derived from microRNA-540-3p overexpressing mesenchymal stem cells promote immune tolerance via the CD74/nuclear factor-kappaB pathway in cardiac allograft. World J Stem Cells 2024, 16, 1022-1046. [CrossRef]
- Guo, G.; Tan, Z.; Liu, Y.; Shi, F.; She, J. The therapeutic potential of stem cell-derived exosomes in the ulcerative colitis and colorectal cancer. Stem Cell Res Ther 2022, 13, 138. [CrossRef]
- Liu, W.; Huang, J.; Chen, F.; Xie, D.; Wang, L.; Ye, C.; Zhu, Q.; Li, X.; Li, X.; Yang, L. MSC-derived small extracellular vesicles overexpressing miR-20a promoted the osteointegration of porous titanium alloy by enhancing osteogenesis via targeting BAMBI. Stem Cell Res Ther 2021, 12, 348. [CrossRef]
- Ponzetti, M.; Rucci, N. Updates on Osteoimmunology: What's New on the Cross-Talk Between Bone and Immune System. Front Endocrinol (Lausanne) 2019, 10, 236. [CrossRef]
- Ignatius, A.; Sobacchi, C. Editorial: Innate Immunity in the Context of Osteoimmunology. Front Immunol 2020, 11, 603. [CrossRef]
- Yang, M.; Zhu, L. Osteoimmunology: The Crosstalk between T Cells, B Cells, and Osteoclasts in Rheumatoid Arthritis. Int J Mol Sci 2024, 25. [CrossRef]
- Lin, T.; Pajarinen, J.; Nabeshima, A.; Lu, L.; Nathan, K.; Jämsen, E.; Yao, Z.; Goodman, S.B. Preconditioning of murine mesenchymal stem cells synergistically enhanced immunomodulation and osteogenesis. Stem Cell Res Ther 2017, 8, 277. [CrossRef]
- Wang, C.; Shou, Z.; Xu, C.; Huo, K.; Liu, W.; Liu, H.; Zan, X.; Wang, Q.; Li, L. Enhancing the Implant Osteointegration via Supramolecular Co-Assembly Coating with Early Immunomodulation and Cell Colonization. Adv Sci (Weinh) 2025, 12, e2410595. [CrossRef]
- Lee, S.C.; Shin, M.K.; Jang, B.Y.; Lee, S.H.; Kim, M.; Sung, J.S. Immunomodulatory Effect and Bone Homeostasis Regulation in Osteoblasts Differentiated from hADMSCs via the PD-1/PD-L1 Axis. Cells 2022, 11. [CrossRef]
- Nitkin, C.R.; Rajasingh, J.; Pisano, C.; Besner, G.E.; Thébaud, B.; Sampath, V. Stem cell therapy for preventing neonatal diseases in the 21st century: Current understanding and challenges. Pediatr Res 2020, 87, 265-276. [CrossRef]
- Rajasingh, S.; Sigamani, V.; Selvam, V.; Gurusamy, N.; Kirankumar, S.; Vasanthan, J.; Rajasingh, J. Comparative analysis of human induced pluripotent stem cell-derived mesenchymal stem cells and umbilical cord mesenchymal stem cells. Journal of cellular and molecular medicine 2021, 25, 8904-8919. [CrossRef]
- Vasanthan, J.; Gurusamy, N.; Rajasingh, S.; Sigamani, V.; Kirankumar, S.; Thomas, E.L.; Rajasingh, J. Role of Human Mesenchymal Stem Cells in Regenerative Therapy. Cells 2020, 10. [CrossRef]
- Shah, P.; Aghazadeh, M.; Rajasingh, S.; Dixon, D.; Jain, V.; Rajasingh, J. Stem cells in regenerative dentistry: Current understanding and future directions. J Oral Biosci 2024, 66, 288-299. [CrossRef]
- Vembuli, H.; Rajasingh, S.; Nabholz, P.; Guenther, J.; Morrow, B.R.; Taylor, M.M.; Aghazadeh, M.; Sigamani, V.; Rajasingh, J. Induced mesenchymal stem cells generated from periodontal ligament fibroblast for regenerative therapy. Exp Biol Med (Maywood) 2025, 250, 10342. [CrossRef]
- Mani, S.; Gurusamy, N.; Ulaganathan, T.; Paluck, A.J.; Ramalingam, S.; Rajasingh, J. Therapeutic potentials of stem cell-derived exosomes in cardiovascular diseases. Exp Biol Med (Maywood) 2023, 248, 434-444. [CrossRef]
- Samanta, S.; Rajasingh, S.; Drosos, N.; Zhou, Z.; Dawn, B.; Rajasingh, J. Exosomes: new molecular targets of diseases. Acta pharmacologica Sinica 2017. [CrossRef]
- Sun, S.J.; Wei, R.; Li, F.; Liao, S.Y.; Tse, H.F. Mesenchymal stromal cell-derived exosomes in cardiac regeneration and repair. Stem Cell Reports 2021, 16, 1662-1673. [CrossRef]
- Lee, B.C.; Kang, I.; Yu, K.R. Therapeutic Features and Updated Clinical Trials of Mesenchymal Stem Cell (MSC)-Derived Exosomes. J Clin Med 2021, 10. [CrossRef]
- Nakamura, Y.; Niho, S.; Shimizu, Y. Cell-Based Therapy for Fibrosing Interstitial Lung Diseases, Current Status, and Potential Applications of iPSC-Derived Cells. Cells 2024, 13. [CrossRef]
- Zhang, J.; Guan, J.; Niu, X.; Hu, G.; Guo, S.; Li, Q.; Xie, Z.; Zhang, C.; Wang, Y. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. Journal of translational medicine 2015, 13, 49. [CrossRef]
- Levy, D.; Abadchi, S.N.; Shababi, N.; Ravari, M.R.; Pirolli, N.H.; Bergeron, C.; Obiorah, A.; Mokhtari-Esbuie, F.; Gheshlaghi, S.; Abraham, J.M.; et al. Induced Pluripotent Stem Cell-Derived Extracellular Vesicles Promote Wound Repair in a Diabetic Mouse Model via an Anti-Inflammatory Immunomodulatory Mechanism. Advanced healthcare materials 2023, 12, e2300879. [CrossRef]
- Li, Z.; Zhang, B.; Shang, J.; Wang, Y.; Jia, L.; She, X.; Xu, X.; Zhang, D.; Guo, J.; Zhang, F. Diabetic and nondiabetic BMSC-derived exosomes affect bone regeneration via regulating miR-17-5p/SMAD7 axis. Int Immunopharmacol 2023, 125, 111190. [CrossRef]
- Wei, F.; Li, Z.; Crawford, R.; Xiao, Y.; Zhou, Y. Immunoregulatory role of exosomes derived from differentiating mesenchymal stromal cells on inflammation and osteogenesis. J Tissue Eng Regen Med 2019, 13, 1978-1991. [CrossRef]
- Wang, D.; Cao, H.; Hua, W.; Gao, L.; Yuan, Y.; Zhou, X.; Zeng, Z. Mesenchymal Stem Cell-Derived Extracellular Vesicles for Bone Defect Repair. Membranes (Basel) 2022, 12. [CrossRef]
- Huang, D.; Shen, H.; Xie, F.; Hu, D.; Jin, Q.; Hu, Y.; Zhong, T. Role of mesenchymal stem cell-derived exosomes in the regeneration of different tissues. J Biol Eng 2024, 18, 36. [CrossRef]
- Ren, S.; Lin, Y.; Liu, W.; Yang, L.; Zhao, M. MSC-Exos: Important active factor of bone regeneration. Front Bioeng Biotechnol 2023, 11, 1136453. [CrossRef]
- Huo, K.L.; Yang, T.Y.; Zhang, W.W.; Shao, J. Mesenchymal stem/stromal cells-derived exosomes for osteoporosis treatment. World J Stem Cells 2023, 15, 83-89. [CrossRef]
- Zhang, X.; Liang, Y.; Luo, D.; Li, P.; Chen, Y.; Fu, X.; Yue, Y.; Hou, R.; Liu, J.; Wang, X. Advantages and disadvantages of various hydrogel scaffold types: A research to improve the clinical conversion rate of loaded MSCs-Exos hydrogel scaffolds. Biomed Pharmacother 2024, 179, 117386. [CrossRef]
- Silva, A.M.; Teixeira, J.H.; Almeida, M.I.; Gonçalves, R.M.; Barbosa, M.A.; Santos, S.G. Extracellular Vesicles: Immunomodulatory messengers in the context of tissue repair/regeneration. European journal of pharmaceutical sciences : official journal of the European Federation for Pharmaceutical Sciences 2017, 98, 86-95. [CrossRef]
- Phinney, D.G.; Pittenger, M.F. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells 2017, 35, 851-858. [CrossRef]
- Long, R.; Wang, S. Exosomes from preconditioned mesenchymal stem cells: Tissue repair and regeneration. Regen Ther 2024, 25, 355-366. [CrossRef]
- Liu, W.; Li, L.; Rong, Y.; Qian, D.; Chen, J.; Zhou, Z.; Luo, Y.; Jiang, D.; Cheng, L.; Zhao, S.; et al. Hypoxic mesenchymal stem cell-derived exosomes promote bone fracture healing by the transfer of miR-126. Acta biomaterialia 2020, 103, 196-212. [CrossRef]
- Chu, C.H.; Lee, R.P.; Wu, W.T.; Chen, I.H.; Yeh, K.T.; Wang, C.C. Advancing Osteoarthritis Treatment: The Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes and Biomaterial Integration. Biomedicines 2024, 12. [CrossRef]
- Zhu, M.; Li, S.; Li, S.; Wang, H.; Xu, J.; Wang, Y.; Liang, G. Strategies for Engineering Exosomes and Their Applications in Drug Delivery. Journal of biomedical nanotechnology 2021, 17, 2271-2297. [CrossRef]
- Elsharkasy, O.M.; Nordin, J.Z.; Hagey, D.W.; de Jong, O.G.; Schiffelers, R.M.; Andaloussi, S.E.; Vader, P. Extracellular vesicles as drug delivery systems: Why and how? Advanced drug delivery reviews 2020, 159, 332-343. [CrossRef]
- Amiri, A.; Bagherifar, R.; Ansari Dezfouli, E.; Kiaie, S.H.; Jafari, R.; Ramezani, R. Exosomes as bio-inspired nanocarriers for RNA delivery: preparation and applications. Journal of translational medicine 2022, 20, 125. [CrossRef]
- Lener, T.; Gimona, M.; Aigner, L.; Börger, V.; Buzas, E.; Camussi, G.; Chaput, N.; Chatterjee, D.; Court, F.A.; Del Portillo, H.A.; et al. Applying extracellular vesicles based therapeutics in clinical trials - an ISEV position paper. J Extracell Vesicles 2015, 4, 30087. [CrossRef]
- Wang, X.; Thomsen, P. Mesenchymal stem cell–derived small extracellular vesicles and bone regeneration. Basic & Clinical Pharmacology & Toxicology 2021, 128, 18-36. [CrossRef]
- Liang, W.; Han, B.; Hai, Y.; Sun, D.; Yin, P. Mechanism of Action of Mesenchymal Stem Cell-Derived Exosomes in the Intervertebral Disc Degeneration Treatment and Bone Repair and Regeneration. Front Cell Dev Biol 2021, 9, 833840. [CrossRef]
- Wa, Q.; Luo, Y.; Tang, Y.; Song, J.; Zhang, P.; Linghu, X.; Lin, S.; Li, G.; Wang, Y.; Wen, Z.; et al. Mesoporous bioactive glass-enhanced MSC-derived exosomes promote bone regeneration and immunomodulation in vitro and in vivo. J Orthop Translat 2024, 49, 264-282. [CrossRef]
- Wang, X.; Fu, L.; Sun, R.; Zhang, C.; Zhang, Y. Bone Marrow Mesenchymal Stem Cell-Exosomes (BMSC-ExO) Promote Osteogenic Differentiation In Vitro and Osteogenesis In Vivo by Regulating miR-318/Runt-Related Transcription Factor 2 (RUNX2). Journal of Biomaterials and Tissue Engineering 2022, 12, 1266-1271. [CrossRef]
- Soriano-Cruz, M.; Vázquez-González, W.G.; Molina-Vargas, P.; Faustino-Trejo, A.; Chávez-Rueda, A.K.; Legorreta-Haquet, M.V.; Aguilar-Ruíz, S.R.; Chávez-Sánchez, L. Exosomes as Regulators of Macrophages in Cardiovascular Diseases. Biomedicines 2024, 12. [CrossRef]
- Zhang, Y.; Bai, J.; Xiao, B.; Li, C. BMSC-derived exosomes promote osteoporosis alleviation via M2 macrophage polarization. Molecular medicine (Cambridge, Mass.) 2024, 30, 220. [CrossRef]
- Agrawal, M.; Rasiah, P.K.; Bajwa, A.; Rajasingh, J.; Gangaraju, R. Mesenchymal Stem Cell Induced Foxp3(+) Tregs Suppress Effector T Cells and Protect against Retinal Ischemic Injury. Cells 2021, 10. [CrossRef]
- Xie, Q.H.; Zheng, J.Q.; Ding, J.Y.; Wu, Y.F.; Liu, L.; Yu, Z.L.; Chen, G. Exosome-Mediated Immunosuppression in Tumor Microenvironments. Cells 2022, 11. [CrossRef]
- Zhao, Y.; Liu, T.; Zhou, M. Immune-Cell-Derived Exosomes for Cancer Therapy. Mol Pharm 2022, 19, 3042-3056. [CrossRef]
- Othman, N.; Jamal, R.; Abu, N. Cancer-Derived Exosomes as Effectors of Key Inflammation-Related Players. Front Immunol 2019, 10, 2103. [CrossRef]
- Kugeratski, F.G.; Kalluri, R. Exosomes as mediators of immune regulation and immunotherapy in cancer. Febs j 2021, 288, 10-35. [CrossRef]
- Madel, M.B.; Ibáñez, L.; Wakkach, A.; de Vries, T.J.; Teti, A.; Apparailly, F.; Blin-Wakkach, C. Immune Function and Diversity of Osteoclasts in Normal and Pathological Conditions. Front Immunol 2019, 10, 1408. [CrossRef]
- Zheng, Q.; Zhang, S.; Guo, W.Z.; Li, X.K. The Unique Immunomodulatory Properties of MSC-Derived Exosomes in Organ Transplantation. Front Immunol 2021, 12, 659621. [CrossRef]
- Tian, C.M.; Yang, M.F.; Xu, H.M.; Zhu, M.Z.; Zhang, Y.; Yao, J.; Wang, L.S.; Liang, Y.J.; Li, D.F. Mesenchymal Stem Cell-derived Exosomes: Novel Therapeutic Approach for Inflammatory Bowel Diseases. Stem Cells Int 2023, 2023, 4245704. [CrossRef]
- Tian, J.; Zhu, Q.; Zhang, Y.; Bian, Q.; Hong, Y.; Shen, Z.; Xu, H.; Rui, K.; Yin, K.; Wang, S. Olfactory Ecto-Mesenchymal Stem Cell-Derived Exosomes Ameliorate Experimental Colitis via Modulating Th1/Th17 and Treg Cell Responses. Front Immunol 2020, 11, 598322. [CrossRef]
- Shahir, M.; Mahmoud Hashemi, S.; Asadirad, A.; Varahram, M.; Kazempour-Dizaji, M.; Folkerts, G.; Garssen, J.; Adcock, I.; Mortaz, E. Effect of mesenchymal stem cell-derived exosomes on the induction of mouse tolerogenic dendritic cells. Journal of cellular physiology 2020, 235, 7043-7055. [CrossRef]
- Fathollahi, A.; Hashemi, S.M.; Haji Molla Hoseini, M.; Yeganeh, F. In vitro analysis of immunomodulatory effects of mesenchymal stem cell- and tumor cell -derived exosomes on recall antigen-specific responses. International immunopharmacology 2019, 67, 302-310. [CrossRef]
- Alshahrani, M.Y.; Jasim, S.A.; Altalbawy, F.M.A.; Bansal, P.; Kaur, H.; Al-Hamdani, M.M.; Deorari, M.; Abosaoda, M.K.; Hamzah, H.F.; B, A.M. A comprehensive insight into the immunomodulatory role of MSCs-derived exosomes (MSC-Exos) through modulating pattern-recognition receptors (PRRs). Cell biochemistry and function 2024, 42, e4029. [CrossRef]
- Nikfarjam, S.; Rezaie, J.; Zolbanin, N.M.; Jafari, R. Mesenchymal stem cell derived-exosomes: a modern approach in translational medicine. J Transl Med 2020, 18, 449. [CrossRef]
- Harrell, C.R.; Djonov, V.; Volarevic, A.; Arsenijevic, A.; Volarevic, V. Molecular Mechanisms Responsible for the Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes in the Treatment of Lung Fibrosis. Int J Mol Sci 2024, 25. [CrossRef]
- Ti, D.; Hao, H.; Fu, X.; Han, W. Mesenchymal stem cells-derived exosomal microRNAs contribute to wound inflammation. Sci China Life Sci 2016, 59, 1305-1312. [CrossRef]
- Qin, Y.; Wang, L.; Gao, Z.; Chen, G.; Zhang, C. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Sci Rep 2016, 6, 21961. [CrossRef]
- Narayanan, R.; Huang, C.C.; Ravindran, S. Hijacking the Cellular Mail: Exosome Mediated Differentiation of Mesenchymal Stem Cells. Stem Cells Int 2016, 2016, 3808674. [CrossRef]
- Zhu, Y.; Li, Y.; Cao, Z.; Xue, J.; Wang, X.; Hu, T.; Han, B.; Guo, Y. Mechanically strained osteocyte-derived exosomes contained miR-3110-5p and miR-3058-3p and promoted osteoblastic differentiation. Biomed Eng Online 2024, 23, 44. [CrossRef]
- Wang, B.; Lyu, F.J.; Deng, Z.; Zheng, Q.; Ma, Y.; Peng, Y.; Guo, S.; Lei, G.; Lai, Y.; Li, Q. Therapeutic potential of stem cell-derived exosomes for bone tissue regeneration around prostheses. J Orthop Translat 2025, 52, 85-96. [CrossRef]
- Wu, F.; Song, C.; Zhen, G.; Jin, Q.; Li, W.; Liang, X.; Xu, W.; Guo, W.; Yang, Y.; Dong, W.; et al. Exosomes derived from BMSCs in osteogenic differentiation promote type H blood vessel angiogenesis through miR-150-5p mediated metabolic reprogramming of endothelial cells. Cellular and molecular life sciences : CMLS 2024, 81, 344. [CrossRef]
- Liang, W.; Li, Y.; Ji, Y.; Kang, R.; Zhang, K.; Su, X.; Li, J.; Ji, M.; Wu, T.; Cao, X.; et al. Exosomes derived from bone marrow mesenchymal stem cells induce the proliferation and osteogenic differentiation and regulate the inflammatory state in osteomyelitis in vitro model. Naunyn-Schmiedeberg's archives of pharmacology 2025, 398, 1695-1705. [CrossRef]
- Chen, Y.; Huang, Y.; Li, J.; Jiao, T.; Yang, L. Enhancing osteoporosis treatment with engineered mesenchymal stem cell-derived extracellular vesicles: mechanisms and advances. Cell Death Dis 2024, 15, 119. [CrossRef]
- Wang, X.; Omar, O.; Vazirisani, F.; Thomsen, P.; Ekström, K. Mesenchymal stem cell-derived exosomes have altered microRNA profiles and induce osteogenic differentiation depending on the stage of differentiation. PLoS One 2018, 13, e0193059. [CrossRef]
- Zhai, M.; Zhu, Y.; Yang, M.; Mao, C. Human Mesenchymal Stem Cell Derived Exosomes Enhance Cell-Free Bone Regeneration by Altering Their miRNAs Profiles. Adv Sci (Weinh) 2020, 7, 2001334. [CrossRef]
- Al-Sharabi, N.; Mohamed-Ahmed, S.; Shanbhag, S.; Kampleitner, C.; Elnour, R.; Yamada, S.; Rana, N.; Birkeland, E.; Tangl, S.; Gruber, R.; et al. Osteogenic human MSC-derived extracellular vesicles regulate MSC activity and osteogenic differentiation and promote bone regeneration in a rat calvarial defect model. Stem Cell Res Ther 2024, 15, 33. [CrossRef]
- Meng, F.; Wang, G.; Zhou, F.; Li, G.; Wang, M.; Zhou, Z.; Han, Y.; Chen, X.; Hu, Y.; Zhang, Y.; et al. Exosomes from young plasma alleviate osteoporosis through miR-217-5p-regulated osteogenesis of bone marrow mesenchymal stem cell. Composites Part B: Engineering 2024, 276, 111358. [CrossRef]
- Wu, Z.; Su, Y.; Li, J.; Liu, X.; Liu, Y.; Zhao, L.; Li, L.; Zhang, L. Induced pluripotent stem cell-derived mesenchymal stem cells: whether they can become new stars of cell therapy. Stem Cell Res Ther 2024, 15, 367. [CrossRef]
- Yang, X.; Zhang, S.; Lu, J.; Chen, X.; Zheng, T.; He, R.; Ye, C.; Xu, J. Therapeutic potential of mesenchymal stem cell-derived exosomes in skeletal diseases. Front Mol Biosci 2024, 11, 1268019. [CrossRef]
- Janockova, J.; Slovinska, L.; Harvanova, D.; Spakova, T.; Rosocha, J. New therapeutic approaches of mesenchymal stem cells-derived exosomes. J Biomed Sci 2021, 28, 39. [CrossRef]
- Lian, M.; Qiao, Z.; Qiao, S.; Zhang, X.; Lin, J.; Xu, R.; Zhu, N.; Tang, T.; Huang, Z.; Jiang, W.; et al. Nerve Growth Factor-Preconditioned Mesenchymal Stem Cell-Derived Exosome-Functionalized 3D-Printed Hierarchical Porous Scaffolds with Neuro-Promotive Properties for Enhancing Innervated Bone Regeneration. ACS Nano 2024, 18, 7504-7520. [CrossRef]
- Bi, Y.; Qiao, X.; Liu, Q.; Song, S.; Zhu, K.; Qiu, X.; Zhang, X.; Jia, C.; Wang, H.; Yang, Z.; et al. Systemic proteomics and miRNA profile analysis of exosomes derived from human pluripotent stem cells. Stem Cell Res Ther 2022, 13, 449. [CrossRef]
- González-Cubero, E.; González-Fernández, M.L.; Gutiérrez-Velasco, L.; Navarro-Ramírez, E.; Villar-Suárez, V. Isolation and characterization of exosomes from adipose tissue-derived mesenchymal stem cells. Journal of Anatomy 2021, 238, 1203-1217. [CrossRef]
- Currey, J.D.; Dean, M.N.; Shahar, R. Revisiting the links between bone remodelling and osteocytes: insights from across phyla. Biol Rev Camb Philos Soc 2017, 92, 1702-1719. [CrossRef]
- Altıntaş, Ö.; Saylan, Y. Exploring the Versatility of Exosomes: A Review on Isolation, Characterization, Detection Methods, and Diverse Applications. Anal Chem 2023, 95, 16029-16048. [CrossRef]
- Hade, M.D.; Suire, C.N.; Suo, Z. Mesenchymal Stem Cell-Derived Exosomes: Applications in Regenerative Medicine. Cells 2021, 10. [CrossRef]
| Bioactive Cargo | Function | Citation |
|---|---|---|
| miR-3110-5p | Promote osteoblast differentiation | [76] |
| miR-3058-3p | ||
| miR-21 | Enhance osteoblast proliferation and differentiation by targeting the TGF-β signaling pathway | [39] |
| miR-29a | Promotes osteoblast differentiation and extracellular matrix mineralization | [39] |
| miR-20a | Contributes to osteogenic differentiation by targeting BAMBI | [18] |
| miR-540-3p | Modulates dendritic cells and T cells via the CD74/NF-κB pathway, enhancing immune tolerance and reducing graft rejection | [16] |
| miR-3940-5p | Can inhibit tumour proliferation and migration by targeting oncogenic pathways | [17] |
| miR-22-3p | ||
| miR-16-5p | ||
| miR-21 | Involved in regulating inflammatory signalling and cell survival | [72,73] |
| miR-146a | ||
| miR-181 | ||
| Pro-inflammatory cytokines (e.g., IL-1β, IFN-γ) | Suppressed by MSC-derived exosomes | [16] |
| Anti-inflammatory cytokines (e.g., IL-10, TGF-β1) | Promoted by MSC-derived exosomes, contributing to immune tolerance | [16] |
| Growth factors (VEGF, HGF) | Support tissue repair and angiogenesis | [71] |
| Disease Model | Key Findings | Citations |
|---|---|---|
| Osteoporosis | Promoted osteointegration of porous titanium alloy by enhancing osteogenesis | [18] |
| Improve bone mass and formation | [39] | |
| Enhance bone density and reduce fracture risk likely through stimulation of osteoblast activity and inhibition of osteoclastogenesis | [42] | |
| Rheumatoid arthritis (RA) | Alleviate joint damage and improve function | [21] |
| Immune Cell Type | Observed Immunomodulatory Effects | Citation |
|---|---|---|
| Macrophages | Induce either pro-inflammatory or anti-inflammatory phenotypes depending on the microenvironment | [57] |
| Promote M2 macrophage phenotype in neuroinflammatory models | [15] | |
| Macrophages (DPSC-Exo specific) | Facilitate the conversion of macrophages from a pro-inflammatory to an anti-inflammatory phenotype, thereby suppressing periodontal inflammation | [28] |
| T cells | Suppress proliferation of inflammatory T cells (producing interferon-γ and IL-17) | [14] |
| T cells | Enhance IL-10 expression | [65] |
| T cells | Encourage regulatory T cell expansion and immune tolerance | [66] |
| Dendritic cells | Alter antigen presentation and T cell activation capacity | [62] |
| Dendritic cells | miR-540-3p modulates dendritic cells via CD74/NF-κB pathway | [16] |
| Effect | Specifications | Citation | |
|---|---|---|---|
| Promotion of Osteogenesis | Enhance osteoblast proliferation and differentiation | Deliver specific microRNAs (miRNAs) that promote osteogenic markers and inhibit negative regulators of bone formation. For instance, miR-21 enhances osteoblast proliferation and differentiation by targeting the TGF-β signaling pathway. miR-29a promotes osteoblast differentiation and extracellular matrix mineralization. | [37] |
| [38] | |||
| [39] | |||
| Influence osteoblast maturation into osteocytes | Exosomes deliver specific microRNAs (miRNAs) that influence the maturation of osteoblasts into osteocytes. | [37] | |
| [38] | |||
| Stimulation of Angiogenesis | Promote formation of new blood vessels | Crucial for supplying nutrients and oxygen to regenerating bone tissue and developing osteocytes within that tissue | [40] |
| In Vivo Bone Regeneration | Accelerate bone healing, increase bone volume, and improve mechanical strength | Animal models of bone defects have demonstrated this in response to local or systemic administration. Crucial for restoring structural integrity and long-term functionality of bone tissue | [18] |
| [42] | |||
| Modulation of Inflammatory Response | Create a favorable environment for tissue regeneration | MSC-derived exosomes can modulate the inflammatory response in the bone defect site | [58] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




