Submitted:
25 July 2025
Posted:
30 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. In Vitro study
2.1.1. Inhibition of HSD11β Type 2 Enzyme
2.1.2. Melanin Synthesis Inhibition
2.2. Clinical Study
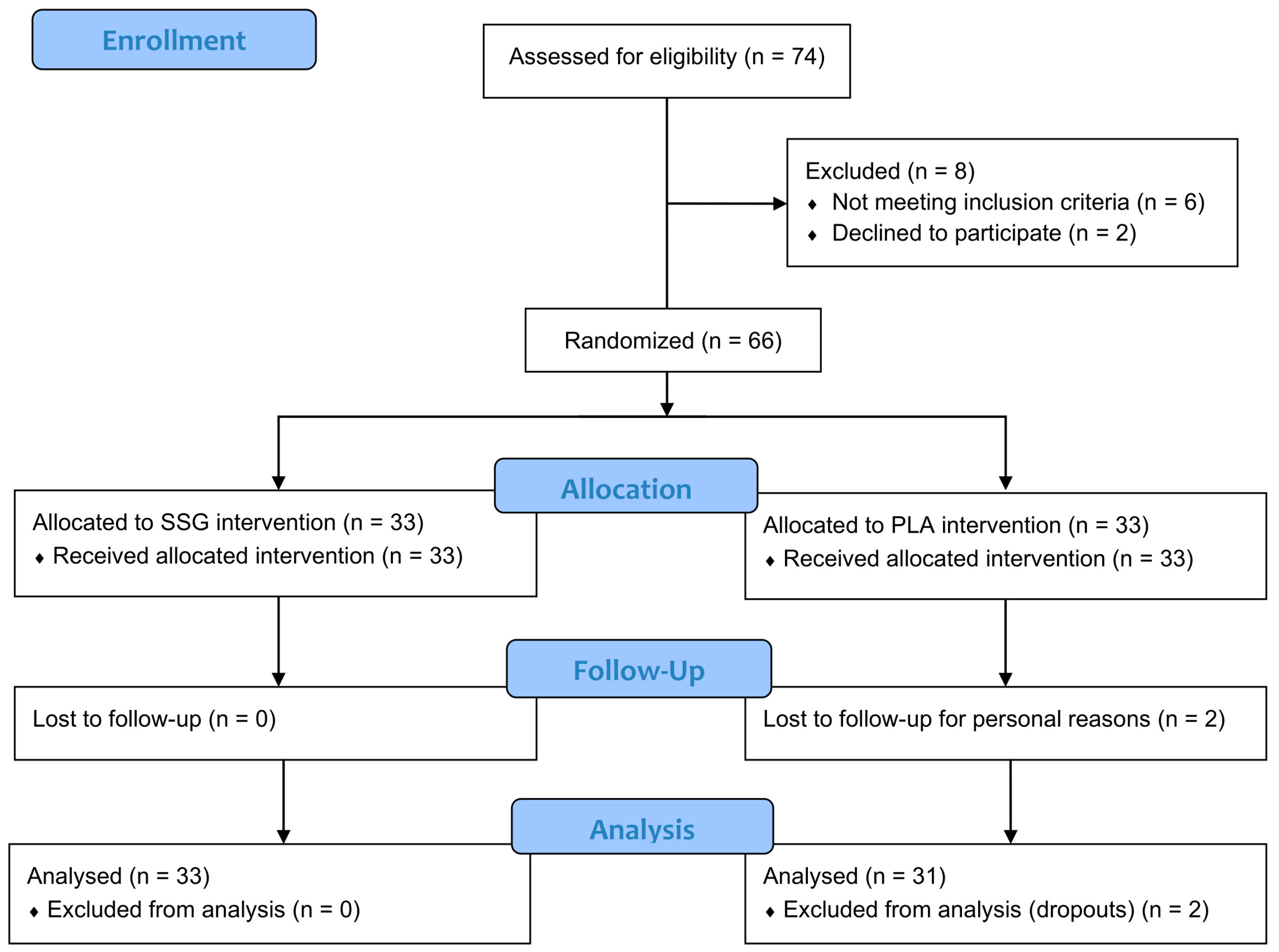
2.2.1. Trial Design and Ethics
2.2.2. Participants and Compliance with Treatment
2.2.3. Interventions and Randomization
2.2.4. Outcomes
2.2.4.1. Primary Outcomes
2.2.4.2. Secondary Outcomes
2.2.4.3. Self-Assessment Questionnaire
2.5. Randomization and Blinding
2.6. Statistical Analysis
2.6.1. In Vitro Studies
2.6.2. Clinical Study: Sample Size and Statistical Analysis
3. Results
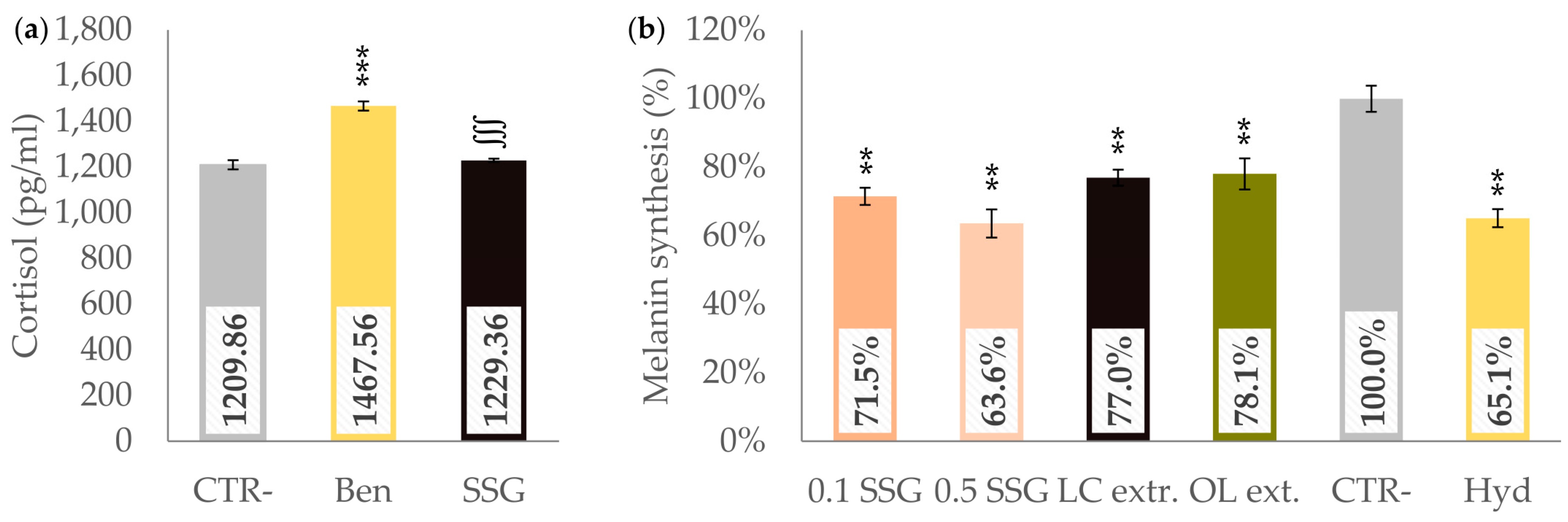
3.1. In Vitro Study
3.1.1. Inhibition of HSD11β Type 2 Enzyme
3.1.2. Melanin Synthesis
3.2. Participant Characteristics, Tolerability, and Compliance with Treatment
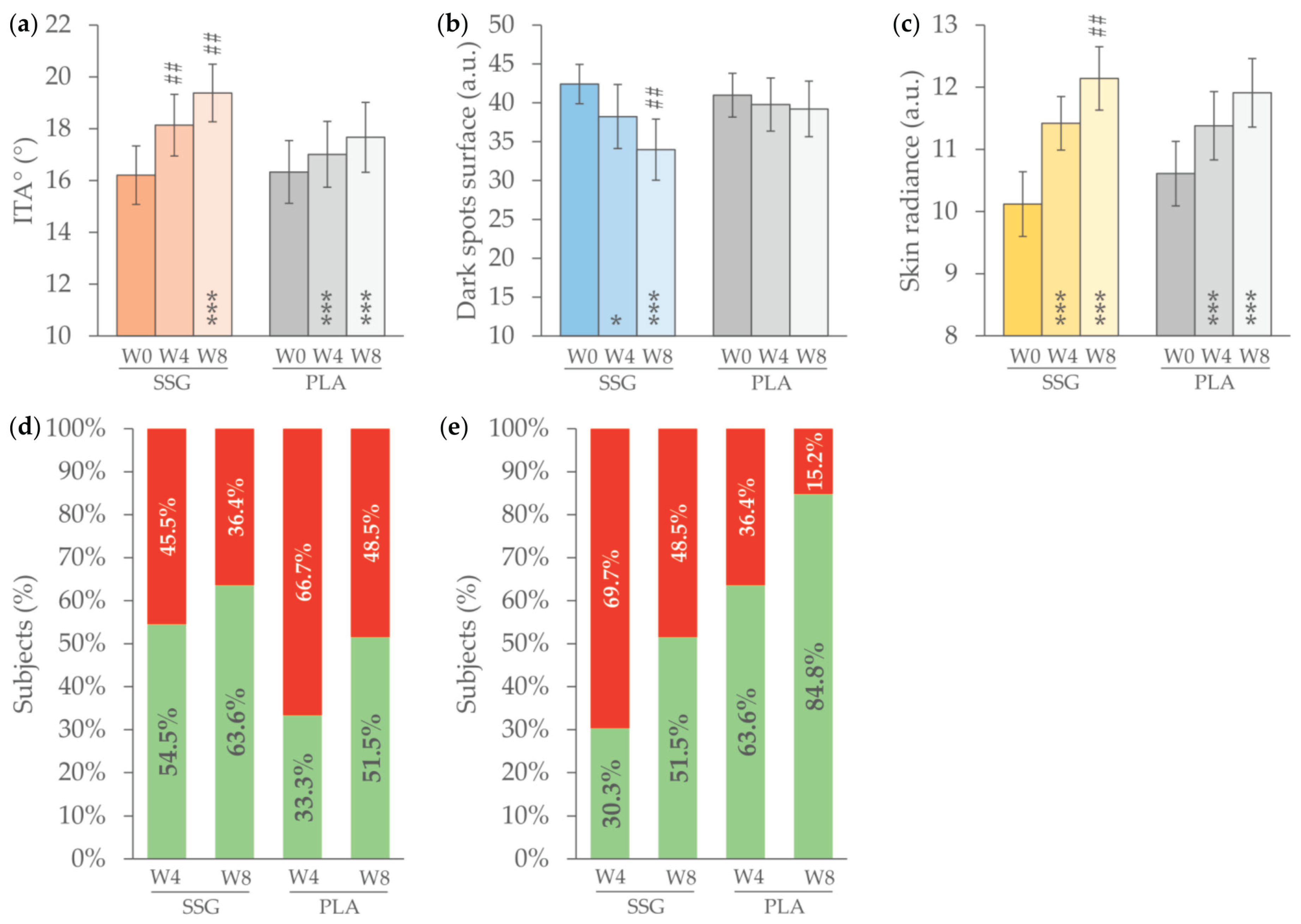
3.3. Primary Endpoints
3.4. Secondary Endpoints
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AGEs | Advanced Glycation End products |
| ITA° | Individual Typology Angle |
| PIH | PostInflammatory Hyperpigmentation |
| PLA | Placebo food supplement |
| RM-ANOVA | Repeated Measures Analysis of Variance |
| SSG | Active food supplement |
| W4 | Week 4 |
| W8 | Week 8 |
| AGEs | Advanced Glycation End products |
References
- Thawabteh, A. M.; Jibreen, A.; Karaman, D.; Thawabteh, A.; Karaman, R. Skin Pigmentation Types, Causes and Treatment—A Review. Molecules 2023, 28(12), 4839. [Google Scholar] [CrossRef] [PubMed]
- Duperray, J.; Sergheraert, R.; Chalothorn, K.; Tachalerdmanee, P.; Perin, F. The Effects of the Oral Supplementation of L-Cystine Associated with Reduced L-Glutathione-GSH on Human Skin Pigmentation: A Randomized, Double-blinded, Benchmark- and Placebo-controlled Clinical Trial. J of Cosmetic Dermatology 2022, 21(2), 802–813. [Google Scholar] [CrossRef]
- Kollias, N.; Seo, I.; Bargo, P. R. Interpreting Diffuse Reflectance for in Vivo Skin Reactions in Terms of Chromophores. J Biophotonics 2010, 3(1–2), 15–24. [Google Scholar] [CrossRef]
- Tseng, S.-H.; Bargo, P.; Durkin, A.; Kollias, N. Chromophore Concentrations, Absorption and Scattering Properties of Human Skin in-Vivo. Opt Express 2009, 17(17), 14599–14617. [Google Scholar] [CrossRef]
- Liu, W.; Chen, Q.; Xia, Y. New Mechanistic Insights of Melasma. Clin Cosmet Investig Dermatol 2023, 16, 429–442. [Google Scholar] [CrossRef]
- Hushcha, Y.; Blo, I.; Oton-Gonzalez, L.; Mauro, G. D.; Martini, F.; Tognon, M.; Mattei, M. D. microRNAs in the Regulation of Melanogenesis. Int J Mol Sci 2021, 22(11), 6104. [Google Scholar] [CrossRef]
- Silpa-Archa, N.; Kohli, I.; Chaowattanapanit, S.; Lim, H. W.; Hamzavi, I. Postinflammatory Hyperpigmentation: A Comprehensive Overview: Epidemiology, Pathogenesis, Clinical Presentation, and Noninvasive Assessment Technique. J Am Acad Dermatol 2017, 77(4), 591–605. [Google Scholar] [CrossRef]
- Flament, F.; Mercurio, D. G.; Catalan, E.; Bouhadanna, E.; Delaunay, C.; Miranda, D. F.; Passeron, T. Impact on Facial Skin Aging Signs of a 1-Year Standardized Photoprotection over a Classical Skin Care Routine in Skin Phototypes II–VI Individuals: A Prospective Randomized Trial. Journal of the European Academy of Dermatology and Venereology 2023, 37(10), 2090–2097. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Garg, V. K.; Jain, A.; Agarwal, D.; Wagle, A.; Flament, F.; Verschoore, M. A Randomized Study to Evaluate the Efficacy and Effectiveness of Two Sunscreen Formulations on Indian Skin Types IV and V with Pigmentation Irregularities. Indian J Dermatol Venereol Leprol 2019, 85(2), 160–168. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, M.; Wang, S.; Leyden, J. J.; Cula, G. O.; Pagnoni, A.; Southall, M. D. Daily Use of a Facial Broad Spectrum Sunscreen Over One-Year Significantly Improves Clinical Evaluation of Photoaging. Dermatol Surg 2016, 42(12), 1354–1361. [Google Scholar] [CrossRef]
- Skoczyńska, A.; Budzisz, E.; Trznadel-Grodzka, E.; Rotsztejn, H. Melanin and Lipofuscin as Hallmarks of Skin Aging. Postepy Dermatol Alergol 2017, 34(2), 97–103. [Google Scholar] [CrossRef]
- Poondru, S.; Gaurav, A.; Yang, L. J.; Kundu, R. V. Perceptions of Sun Protection, Skin Tone, Colorism, and Dermatologic Care Among South Asians in the USA. J Racial Ethn Health Disparities 2025, 12(2), 1021–1026. [Google Scholar] [CrossRef] [PubMed]
- Platsidaki, E.; Efstathiou, V.; Markantoni, V.; Kouris, A.; Kontochristopoulos, G.; Nikolaidou, E.; Rigopoulos, D.; Stratigos, A.; Gregoriou, S. Self-Esteem, Depression, Anxiety and Quality of Life in Patients with Melasma Living in a Sunny Mediterranean Area: Results from a Prospective Cross-Sectional Study. Dermatol Ther (Heidelb) 2023, 13(5), 1127–1136. [Google Scholar] [CrossRef]
- Simons, R. E.; Zevy, D. L.; Jafferany, M. Psychodermatology of Vitiligo: Psychological Impact and Consequences. Dermatologic Therapy 2020, 33(3), e13418. [Google Scholar] [CrossRef]
- Darji, K.; Varade, R.; West, D.; Armbrecht, E. S.; Guo, M. A. Psychosocial Impact of Postinflammatory Hyperpigmentation in Patients with Acne Vulgaris. J Clin Aesthet Dermatol 2017, 10(5), 18–23. [Google Scholar] [PubMed]
- Malathi, M.; Thappa, D. M. Systemic Skin Whitening/Lightening Agents: What Is the Evidence? Indian J Dermatol Venereol Leprol 2013, 79, 842. [Google Scholar] [CrossRef] [PubMed]
- Tursi, F.; Pourtau, L.; Roveda, G.; De Ponti, I.; Gaudout, D.; Moras, B.; Pouchieu, C.; Nobile, V. Clinical Efficacy of Belight3TM on Dark Spot Pigmentation in Caucasian Subjects. Cosmetics 2025, 12(1), 27. [Google Scholar] [CrossRef]
- Hanif, N.; Al-Shami, A. M. A.; Khalid, K. A.; Hadi, H. A. Plant-Based Skin Lightening Agents: A Review. J Phytopharmacol 2020, 9(1), 54–60. [Google Scholar] [CrossRef]
- Dilokthornsakul, W.; Dhippayom, T.; Dilokthornsakul, P. The Clinical Effect of Glutathione on Skin Color and Other Related Skin Conditions: A Systematic Review. J Cosmet Dermatol 2019, 18(3), 728–737. [Google Scholar] [CrossRef]
- Zaid, A. N.; Al Ramahi, R. Depigmentation and Anti-Aging Treatment by Natural Molecules. Curr Pharm Des 2019, 25(20), 2292–2312. [Google Scholar] [CrossRef]
- A Ali, S.; Choudhary, R. K.; Naaz, I. Melanogenesis: Key Role of Bioactive Compounds in the Treatment of Hyperpigmentory Disorders. Pigmentary Disorders 2015, 2(11). [Google Scholar] [CrossRef]
- Ribeiro, A. S.; Estanqueiro, M.; Oliveira, M. B.; Sousa Lobo, J. M. Main Benefits and Applicability of Plant Extracts in Skin Care Products. Cosmetics 2015, 2(2), 48–65. [Google Scholar] [CrossRef]
- Hanif, N.; Al-Shami, A. M. A.; Khalid, K. A.; Hadi, H. A. Plant-Based Skin Lightening Agents: A Review. J Phytopharmacol 2020, 9(1), 54–60. [Google Scholar] [CrossRef]
- Guo, Y.; Cariola, A.; Matera, R.; Gabbanini, S.; Valgimigli, L. Real-Time Oxygen Sensing as a Powerful Tool to Investigate Tyrosinase Kinetics Allows Revising Mechanism and Activity of Inhibition by Glabridin. Food Chem 2022, 393, 133423. [Google Scholar] [CrossRef]
- Nerya, O.; Vaya, J.; Musa, R.; Izrael, S.; Ben-Arie, R.; Tamir, S. Glabrene and Isoliquiritigenin as Tyrosinase Inhibitors from Licorice Roots. J. Agric. Food Chem. 2003, 51(5), 1201–1207. [Google Scholar] [CrossRef]
- de Toledo Bagatin J, Bagatin E, Campos PMBGM. A pilot clinical study to evaluate the effectiveness of olive extract containing hydroxytyrosol for oral and topical treatment of melasma. Biomed Biopharm Res. 2020, 17, 48–62. [CrossRef]
- D’Angelo Costa, G. M.; Maia Campos, P. M. B. G. Efficacy of Topical Antioxidants in the Skin Hyperpigmentation Control: A Clinical Study by Reflectance Confocal Microscopy. J of Cosmetic Dermatology 2021, 20(2), 538–545. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, M.-M.; Sun, Y.; Wang, L.-F.; Wang, H.; Zhang, Y.-J.; Li, H.-Y.; Zhuang, P.-W.; Yang, Z. Synergistic Promotion on Tyrosinase Inhibition by Antioxidants. Molecules 2018, 23(1), 106. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A.; Gonçalves, L.; Marto, J.; Martins, A. M.; Silva, A. N.; Pinto, P.; Martins, M.; Fraga, C.; Ribeiro, H. M. Investigations of Olive Oil Industry By-Products Extracts with Potential Skin Benefits in Topical Formulations. Pharmaceutics 2021, 13(4), 465. [Google Scholar] [CrossRef]
- Centrone, M.; D’Agostino, M.; Difonzo, G.; De Bruno, A.; Di Mise, A.; Ranieri, M.; Montemurro, C.; Valenti, G.; Poiana, M.; Caponio, F.; Tamma, G. Antioxidant Efficacy of Olive By-Product Extracts in Human Colon HCT8 Cells. Foods 2020, 10(1), 11. [Google Scholar] [CrossRef]
- El-Saber Batiha, G.; Magdy Beshbishy, A.; El-Mleeh, A.; Abdel-Daim, M. M.; Prasad Devkota, H. Traditional Uses, Bioactive Chemical Constituents, and Pharmacological and Toxicological Activities of Glycyrrhiza Glabra L. (Fabaceae). Biomolecules 2020, 10(3), 352. [Google Scholar] [CrossRef]
- Kwon, Y.-J.; Son, D.-H.; Chung, T.-H.; Lee, Y.-J. A Review of the Pharmacological Efficacy and Safety of Licorice Root from Corroborative Clinical Trial Findings. J Med Food 2020, 23(1), 12–20. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J. J.; Mangos, G.; Williamson, P. M.; Whitworth, J. A. Cortisol and Hypertension. Clin Exp Pharmacol Physiol Suppl 1998, 25, S51–56. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal Biochem 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Whitening efficacy of a food supplement. ISRCTN Registry. https://doi.org/10.1186/ISRCTN12882379 (accessed on 21 March 2025).
- SCCS Notes of Guidance for the Testing of Cosmetic Ingredients and Their Safety Evaluation—12th Revision—European Commission. Available online: https://health.ec.europa.eu/publications/sccs-notes-guidance-testing-cosmetic-ingredients-and-their-safety-evaluation-12th-revision_en (accessed on 21 March 2025).
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products (recast) (Text with EEA Relevance)—EU Law in Force—Publications Office of the EU. EU Law in Force. Available online: https://op.europa.eu/en/web/eu-law-in-force/bibliographic-details/-/elif-publication/4100396c-575c-4150-8d92-e7c23ab1ce97 (accessed on 21 March 2025).
- Demirli, R.; Otto, O.; Viswanathan, R.; Patwardhan, S.; Larkey, J. RBX® Technology Overview https://www.canfieldsci.com/FileLibrary/RBX%20tech%20overview-LoRz1.pdf (accessed on 21 March 2025).
- Meerwaldt, R.; Graaff, R.; Oomen, P. H. N.; Links, T. P.; Jager, J. J.; Alderson, N. L.; Thorpe, S. R.; Baynes, J. W.; Gans, R. O. B.; Smit, A. J. Simple Non-Invasive Assessment of Advanced Glycation Endproduct Accumulation. Diabetologia 2004, 47(7), 1324–1330. [Google Scholar] [CrossRef]
- Chen, C.; Zhang, J.-Q.; Li, L.; Guo, M.; He, Y.; Dong, Y.; Meng, H.; Yi, F. Advanced Glycation End Products in the Skin: Molecular Mechanisms, Methods of Measurement, and Inhibitory Pathways. Front Med (Lausanne) 2022, 9, 837222. [Google Scholar] [CrossRef] [PubMed]
- Marcílio Cândido, T.; Bueno Ariede, M.; Vieira Lima, F.; de Souza Guedes, L.; Robles Velasco, M. V.; Rolim Baby, A.; Rosado, C. Dietary Supplements and the Skin: Focus on Photoprotection and Antioxidant Activity—A Review. Nutrients 2022, 14(6), 1248. [Google Scholar] [CrossRef]
- Januszewski, J.; Forma, A.; Zembala, J.; Flieger, M.; Tyczyńska, M.; Dring, J. C.; Dudek, I.; Świątek, K.; Baj, J. Nutritional Supplements for Skin Health-A Review of What Should Be Chosen and Why. Medicina (Kaunas) 2023, 60(1), 68. [Google Scholar] [CrossRef]
- Yokota, T.; Nishio, H.; Kubota, Y.; Mizoguchi, M. The Inhibitory Effect of Glabridin from Licorice Extracts on Melanogenesis and Inflammation. Pigment Cell Res 1998, 11(6), 355–361. [Google Scholar] [CrossRef]
- Chen, J.; Yu, X.; Huang, Y. Inhibitory Mechanisms of Glabridin on Tyrosinase. Spectrochim Acta A Mol Biomol Spectrosc 2016, 168, 111–117. [Google Scholar] [CrossRef]
| Placebo | Active | Units | pvalue | |
| Age | 42.5 ± 2.4 | 49.2 ± 2.1 | Years | 0.0671 |
| Sex | ||||
| Female | 90.9% (30) | 87.9% (29) | % (no.) | n.a. |
| Male | 9.1% (3) | 12.1% (4) | % (no.) | n.a. |
| Blood pressure | ||||
| Systolic | 124.7 ± 2.41 | 126.9 ± 2.32 | mmHg | 0.5134 |
| Diastolic | 82.5 ± 1.18 | 80.3 ± 1.25 | mmHg | 0.2106 |
| Skin radiance | 10.61 ± 0.52 | 10.12 ± 0.52 | a.u. | 0.2923 |
| Individual typology Angle (ITA°) | 16.33 ± 1.21 | 16.21 ± 1.13 | ° | 0.9567 |
| Dark spots surface | 40.98 ± 2.82 | 42.42 ± 2.53 | a.u. | 0.7083 |
| AGEs 1 | 2.27 ± 0.18 | 1.89 ± 0.10 | a.u. | 0.0545 |
| W0 | W8 | ΔW8 | pvalue | |
| PLA (n = 17) | 2.27 ± 0.18 | 2.16 ± 0.11 | −2.1% | 0.218 |
| SSG (n =17) | 1.89 ± 0.10 | 1.69 ± 0.10 | −10.6% # | 0.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




