Submitted:
24 July 2025
Posted:
25 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
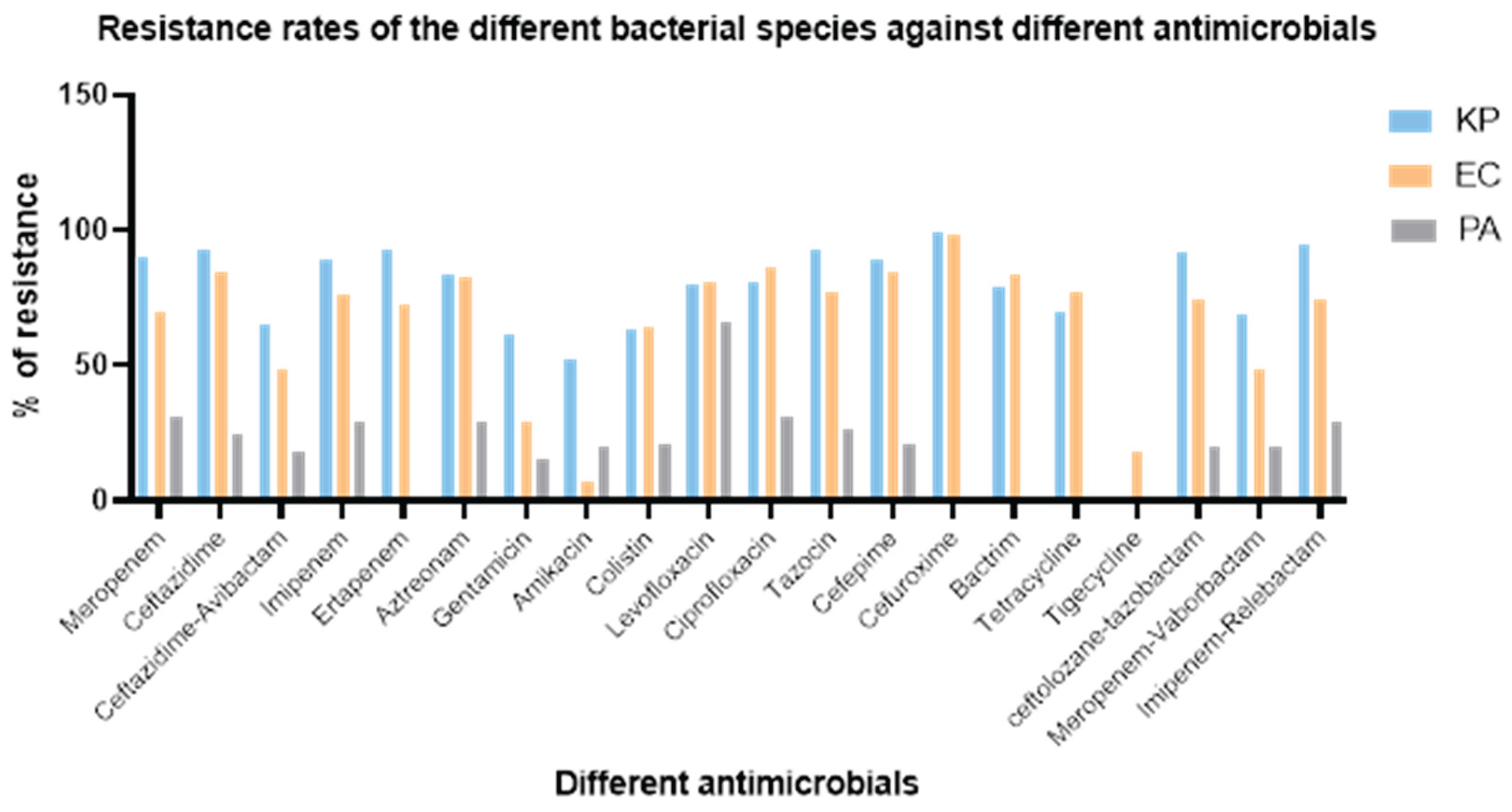
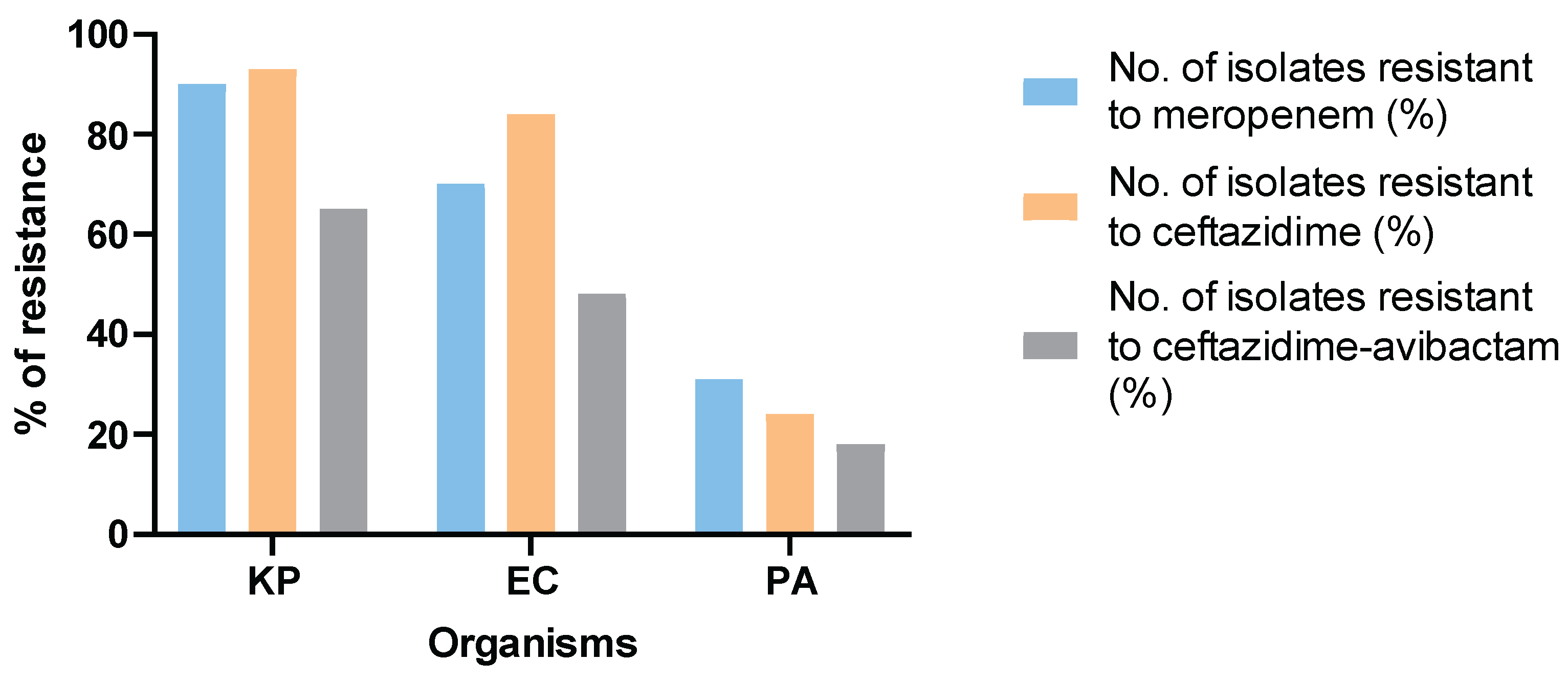
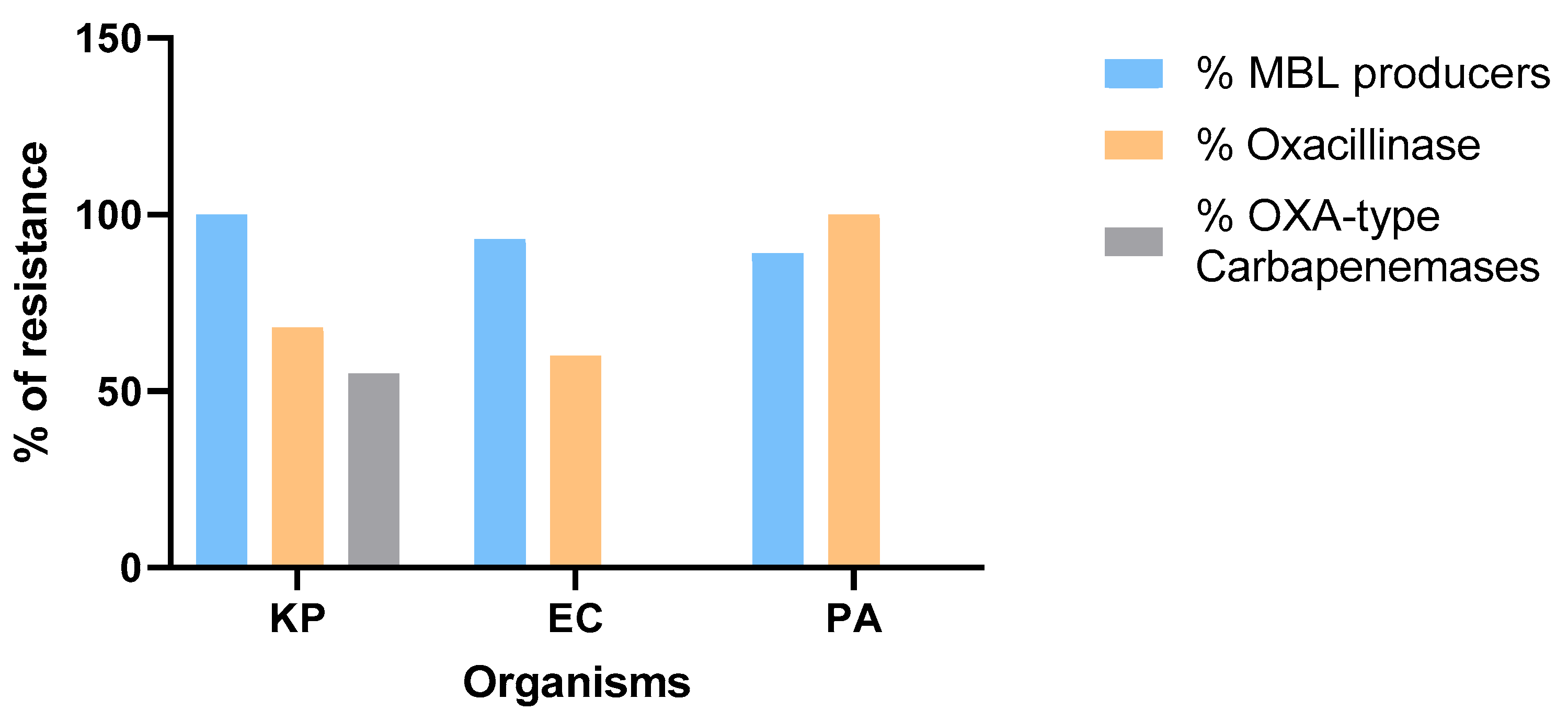
3.1. Antimicrobial Susceptibility Testing Results
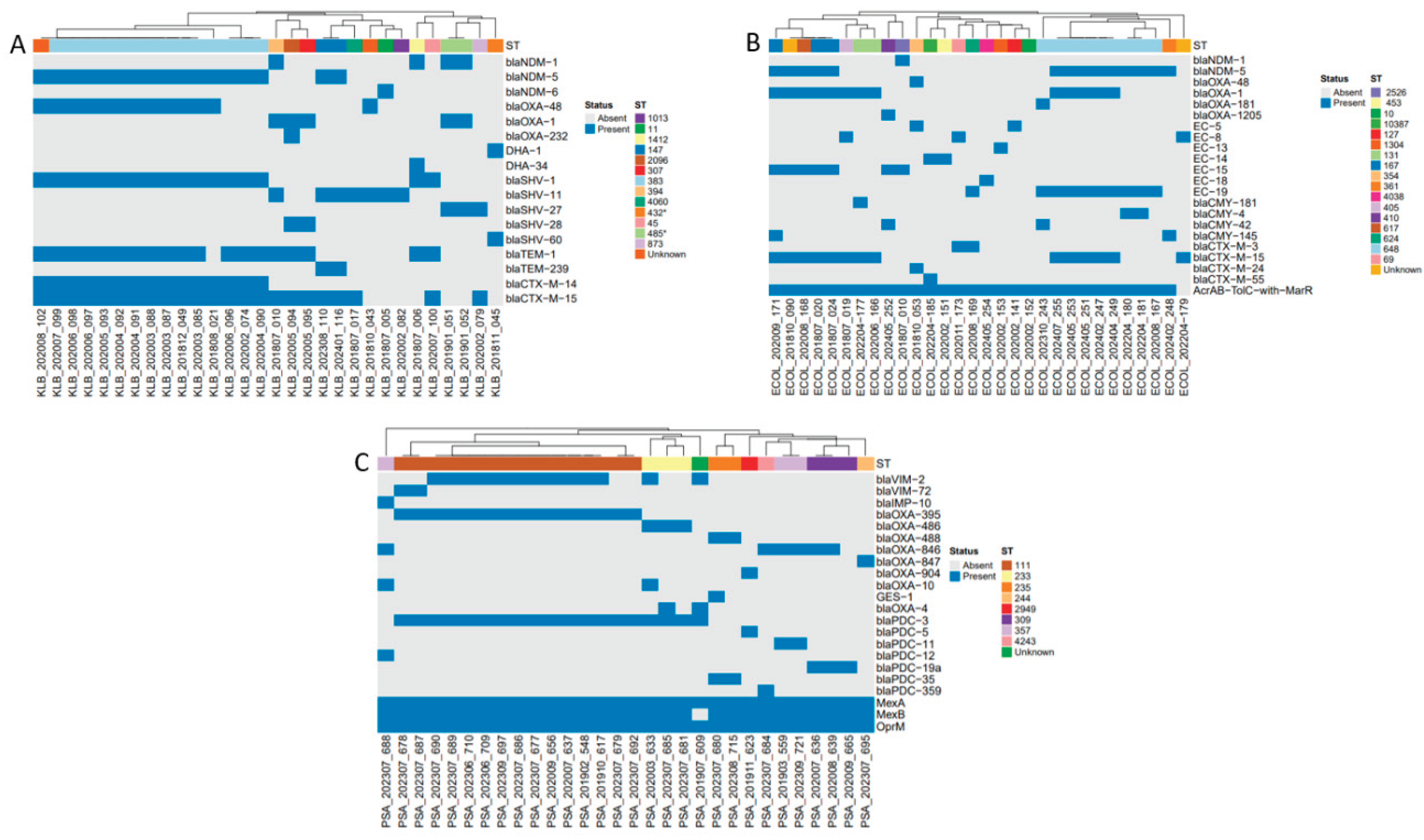
3.2. Whole-Genome Sequencing (WGS) Analysis
3.2.1. Sequence Types (STs)
3.2.2. Plasmids
3.2.3. AMR Genes
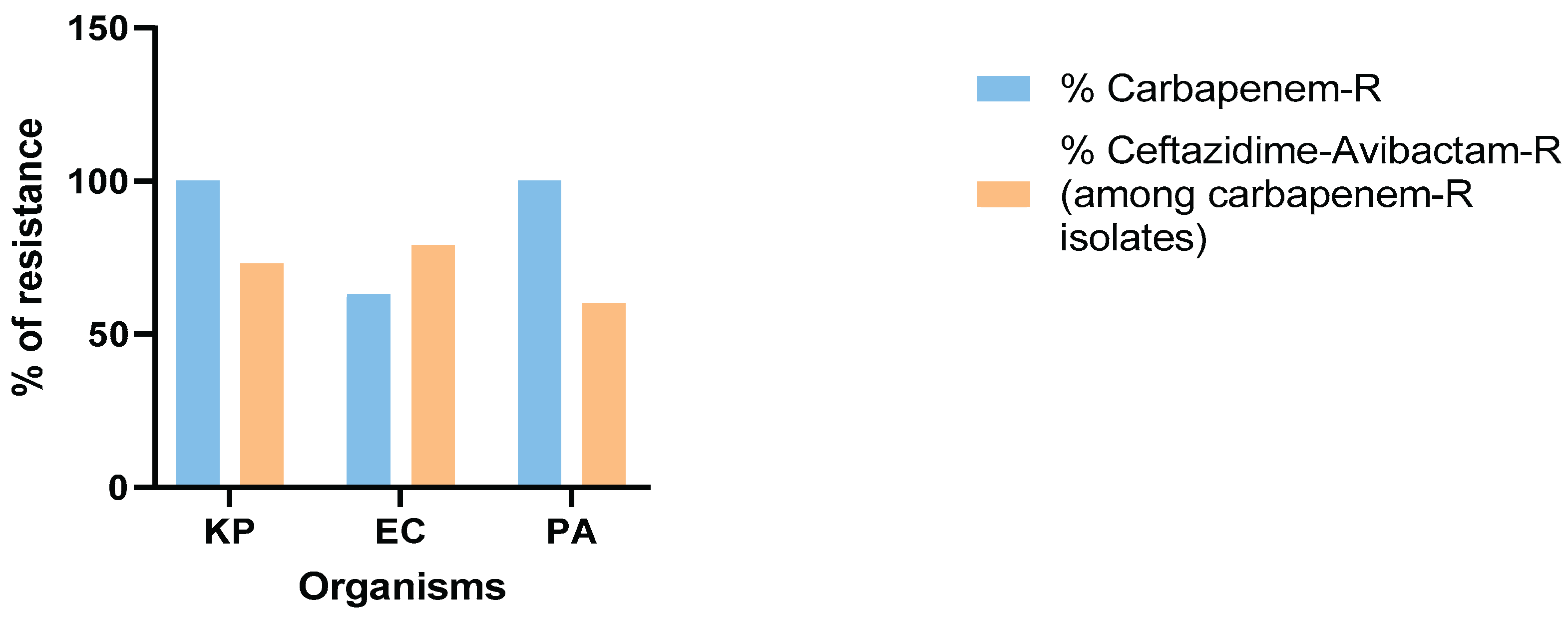
3.3. Antimicrobial Susceptibility and Molecular Characterization of CAZ-AVI-Resistant P. aeruginosa and Enterobaterales
3.4. WGS Analysis of Non-MBL CAZ-AVI-Resistance Isolates and Associated Resistance Genes.
4. Discussion
Supplementary Materials
Author Contributions
Abbreviations
| WHO | World Health Organization |
| CRPA | Carbapenem-resistant Pseudomonas aeruginosa |
| CRE | Carbapenem-resistant Enterobacterales |
| CAZ-AVI | Ceftazidime-Avibactam |
| PA | Pseudomonas aeruginosa |
| EC | Escherichia coli |
| KP | Klebsiella pneumoniae |
| AST | Antimicrobial Susceptibility Testing |
| WGS | Whole-Genome Sequencing |
| MBLs | Metallo-β-lactamases |
| CDC | Centers for Disease Control and Prevention |
| ESBL | Extended-Spectrum Beta-lactamase |
| NDM-1 | New Delhi Metallo-β-lactamase-1 |
| OXA-48 | Oxacillinase-48 |
| KPC | Klebsiella pneumoniae carbapenemase |
| IMP-1 | Imipenemase-1 |
| VIM-1 | Verona integron-encoded metallo-β-lactamase-1 |
| OmpC | Outer membrane protein C |
| MEV | Meropenem-Vaborbactam |
| IMR | Imipenem-Relebactam |
| CARD | Comprehensive Antibiotic Resistance Database |
| CGE | Center of Genomic Epidemiology |
| MLST | Multilocus Sequence Typing |
| ST | Sequence Type |
| SNPs | Single Nucleotide Polymorphisms |
| INDELs | Short Insertions and Deletions |
| PBP4 | Penicillin-Binding Protein 4 |
| AMR | Antimicrobial Resistance |
References
- C. J. L. Murray et al., “Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis,” The Lancet, vol. 399, no. 10325, pp. 629–655, Feb. 2022. [CrossRef]
- D. Chinemerem Nwobodo et al., “Antibiotic resistance: The challenges and some emerging strategies for tackling a global menace,” Clinical Laboratory Analysis, vol. 36, no. 9, Sept. 2022. [CrossRef]
- C. Llor and L. Bjerrum, “Antimicrobial resistance: risk associated with antibiotic overuse and initiatives to reduce the problem,” Therapeutic Advances in Drug Safety, vol. 5, no. 6, pp. 229–241, Dec. 2014. [CrossRef]
- J. A. Al-Tawfiq et al., “Antibiotics in the pipeline: a literature review (2017–2020),” Infection, vol. 50, no. 3, pp. 553–564, June 2022. [CrossRef]
- K. M. Papp-Wallace, A. Endimiani, M. A. Taracila, and R. A. Bonomo, “Carbapenems: Past, Present, and Future,” Antimicrob Agents Chemother, vol. 55, no. 11, pp. 4943–4960, Nov. 2011. [CrossRef]
- X. Qin et al., “Prevalence and Mechanisms of Broad-Spectrum β-Lactam Resistance inEnterobacteriaceae: a Children’s Hospital Experience,” Antimicrob Agents Chemother, vol. 52, no. 11, pp. 3909–3914, Nov. 2008. [CrossRef]
- R. I. El-Herte, S. S. Kanj, G. M. Matar, and G. F. Araj, “The threat of carbapenem-resistant Enterobacteriaceae in Lebanon: An update on the regional and local epidemiology,” Journal of Infection and Public Health, vol. 5, no. 3, pp. 233–243, June 2012. [CrossRef]
- J. G. Moloughney, J. D. Thomas, and J. H. Toney, “Novel IMP-1 metallo-Î2-lactamase inhibitors can reverse meropenem resistance inEscherichia coliexpressing IMP-1,” FEMS Microbiology Letters, vol. 243, no. 1, pp. 65–71, Feb. 2005. [CrossRef]
- C. Urban et al., “Carbapenem-ResistantEscherichia coliHarboringKlebsiella pneumoniaeCarbapenemase β-Lactamases Associated with Long-Term Care Facilities,” CLIN INFECT DIS, vol. 46, no. 11, pp. e127–e130, June 2008. [CrossRef]
- P. Nordmann, L. Dortet, and L. Poirel, “Carbapenem resistance in Enterobacteriaceae: here is the storm!,” Trends in Molecular Medicine, vol. 18, no. 5, pp. 263–272, May 2012. [CrossRef]
- L. Poirel, C. Héritier, V. Tolün, and P. Nordmann, “Emergence of Oxacillinase-Mediated Resistance to Imipenem inKlebsiella pneumoniae,” Antimicrob Agents Chemother, vol. 48, no. 1, pp. 15–22, Jan. 2004. [CrossRef]
- E. Cendejas, R. Gómez-Gil, P. Gómez-Sánchez, and J. Mingorance, “Detection and characterization of Enterobacteriaceae producing metallo-β-lactamases in a tertiary-care hospital in Spain,” Clinical Microbiology and Infection, vol. 16, no. 2, pp. 181–183, Feb. 2010. [CrossRef]
- A. Torres et al., “Ceftazidime-avibactam versus meropenem in nosocomial pneumonia, including ventilator-associated pneumonia (REPROVE): a randomised, double-blind, phase 3 non-inferiority trial,” The Lancet Infectious Diseases, vol. 18, no. 3, pp. 285–295, Mar. 2018. [CrossRef]
- M. Bassetti, D. R. Giacobbe, C. Robba, P. Pelosi, and A. Vena, “Treatment of extended-spectrum β-lactamases infections: what is the current role of new β-lactams/β-lactamase inhibitors?,” Current Opinion in Infectious Diseases, vol. 33, no. 6, pp. 474–481, Dec. 2020. [CrossRef]
- M. Falcone and D. Paterson, “Spotlight on ceftazidime/avibactam: a new option for MDR Gram-negative infections,” J. Antimicrob. Chemother., vol. 71, no. 10, pp. 2713–2722, Oct. 2016. [CrossRef]
- W. W. Nichols, P. A. Bradford, S. D. Lahiri, and G. G. Stone, “The primary pharmacology of ceftazidime/avibactam: in vitro translational biology,” Journal of Antimicrobial Chemotherapy, vol. 77, no. 9, pp. 2321–2340, Aug. 2022. [CrossRef]
- A. Mehwish and I. Iftikhar, “EMERGENCE OF CEFTAZIDIME-AVIBACTAM RESISTANCE IN ENTERO-BACTERALES AND PSEUDOMONAS AERUGINOSA,” Pak J Pathol, vol. 34, no. 4, pp. 113–117, Dec. 2023. [CrossRef]
- S. Zhen, H. Wang, and S. Feng, “Update of clinical application in ceftazidime–avibactam for multidrug-resistant Gram-negative bacteria infections,” Infection, vol. 50, no. 6, pp. 1409–1423, Dec. 2022. [CrossRef]
- G. Sobh et al., “Molecular characterization of carbapenem and ceftazidime-avibactam-resistant Enterobacterales and horizontal spread of blaNDM-5 gene at a Lebanese medical center,” Front. Cell. Infect. Microbiol., vol. 14, June 2024. [CrossRef]
- F. T. Al-Alaq, S. S. Mahmood, N. S. K. AL-Khafaji, H. O. M. Al-Dahmoshi, and M. Memariani, “Investigation of blaIMP-1, blaVIM-1, blaOXA-48 and blaNDM-1 carbapenemase encoding genes among MBL-producing Pseudomonas aeruginosa,” JANS, vol. 14, no. 3, pp. 740–745, Sept. 2022. [CrossRef]
- O. Caille et al., “Structural and Functional Characterization of Pseudomonas aeruginosa Global Regulator AmpR,” J Bacteriol, vol. 196, no. 22, pp. 3890–3902, Nov. 2014. [CrossRef]
- G. Torrens et al., “Activity of Ceftazidime-Avibactam against Clinical and Isogenic Laboratory Pseudomonas aeruginosa Isolates Expressing Combinations of Most Relevant β-Lactam Resistance Mechanisms,” Antimicrob Agents Chemother, vol. 60, no. 10, pp. 6407–6410, Oct. 2016. [CrossRef]
- B. Babouee Flury et al., “Multifactorial resistance mechanisms associated with resistance to ceftazidime-avibactam in clinical Pseudomonas aeruginosa isolates from Switzerland,” Front. Cell. Infect. Microbiol., vol. 13, Apr. 2023. [CrossRef]
- M. Castanheira, T. B. Doyle, C. J. Smith, R. E. Mendes, and H. S. Sader, “Combination of MexAB-OprM overexpression and mutations in efflux regulators, PBPs and chaperone proteins is responsible for ceftazidime/avibactam resistance in Pseudomonas aeruginosa clinical isolates from US hospitals,” Journal of Antimicrobial Chemotherapy, vol. 74, no. 9, pp. 2588–2595, Sept. 2019. [CrossRef]
- L. Xiong et al., “Molecular mechanisms underlying bacterial resistance to ceftazidime/avibactam,” WIREs Mechanisms of Disease, vol. 14, no. 6, Nov. 2022. [CrossRef]
- B. L. Smith, S. Fernando, and M. D. King, “Escherichia coli resistance mechanism AcrAB-TolC efflux pump interactions with commonly used antibiotics: a molecular dynamics study,” Sci Rep, vol. 14, no. 1, Feb. 2024. [CrossRef]
- S. Palwe, Y. D. Bakthavatchalam, K. Khobragadea, A. S. Kharat, K. Walia, and B. Veeraraghavan, “In-Vitro Selection of Ceftazidime/Avibactam Resistance in OXA-48-Like-Expressing Klebsiella pneumoniae: In-Vitro and In-Vivo Fitness, Genetic Basis and Activities of β-Lactam Plus Novel β-Lactamase Inhibitor or β-Lactam Enhancer Combinations,” Antibiotics, vol. 10, no. 11, p. 1318, Oct. 2021. [CrossRef]
- L. Guo et al., “Nosocomial Outbreak of OXA-48-Producing Klebsiella pneumoniae in a Chinese Hospital: Clonal Transmission of ST147 and ST383,” PLoS ONE, vol. 11, no. 8, p. e0160754, Aug. 2016. [CrossRef]
- H. Zhang, C. H. Seward, Z. Wu, H. Ye, and Y. Feng, “Genomic insights into the ESBL and MCR-1-producing ST648 Escherichia coli with multi-drug resistance,” Science Bulletin, vol. 61, no. 11, pp. 875–878, June 2016. [CrossRef]
- A. Oliver, X. Mulet, C. López-Causapé, and C. Juan, “The increasing threat of Pseudomonas aeruginosa high-risk clones,” Drug Resistance Updates, vol. 21–22, pp. 41–59, July 2015. [CrossRef]
- S. Schjorring, C. Struve, and K. A. Krogfelt, “Transfer of antimicrobial resistance plasmids from Klebsiella pneumoniae to Escherichia coli in the mouse intestine,” Journal of Antimicrobial Chemotherapy, vol. 62, no. 5, pp. 1086–1093, July 2008. [CrossRef]
- F. Uddin et al., “NDM Production as a Dominant Feature in Carbapenem-Resistant Enterobacteriaceae Isolates from a Tertiary Care Hospital,” Antibiotics, vol. 11, no. 1, p. 48, Dec. 2021. [CrossRef]
- M. F. Mojica et al., “Molecular Mechanisms of Resistance to Ceftazidime/Avibactam in Clinical Isolates of Enterobacterales and Pseudomonas aeruginosa in Latin American Hospitals”.

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



