Submitted:
24 July 2025
Posted:
30 July 2025
Read the latest preprint version here
Abstract
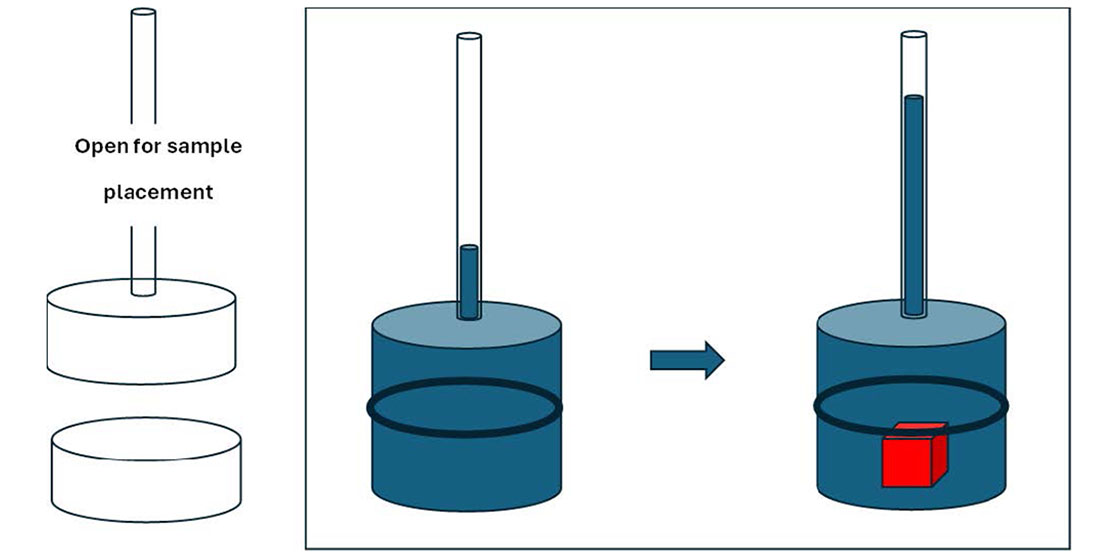
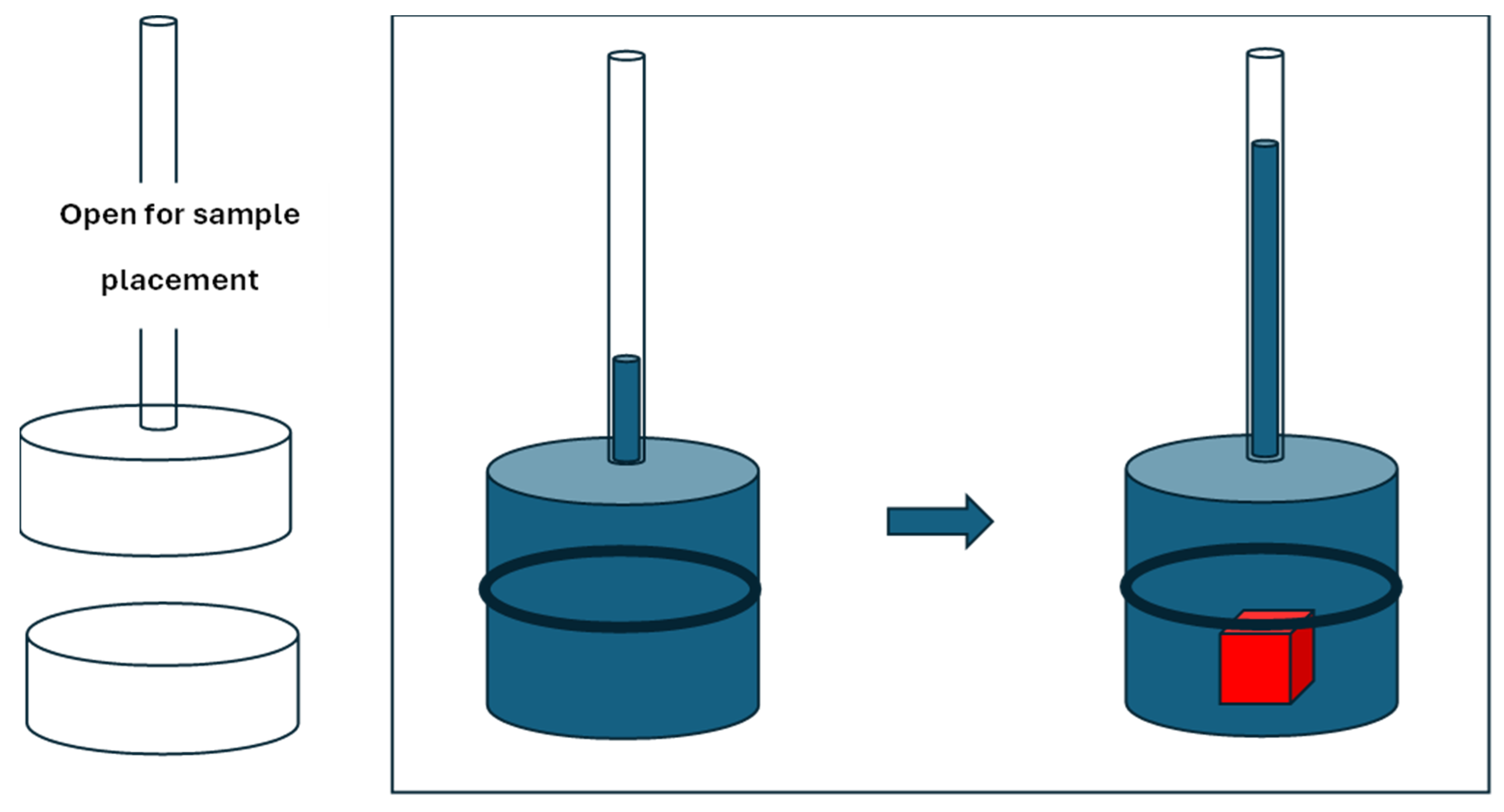
Keywords:
1. Introduction
2. Materials and Methods
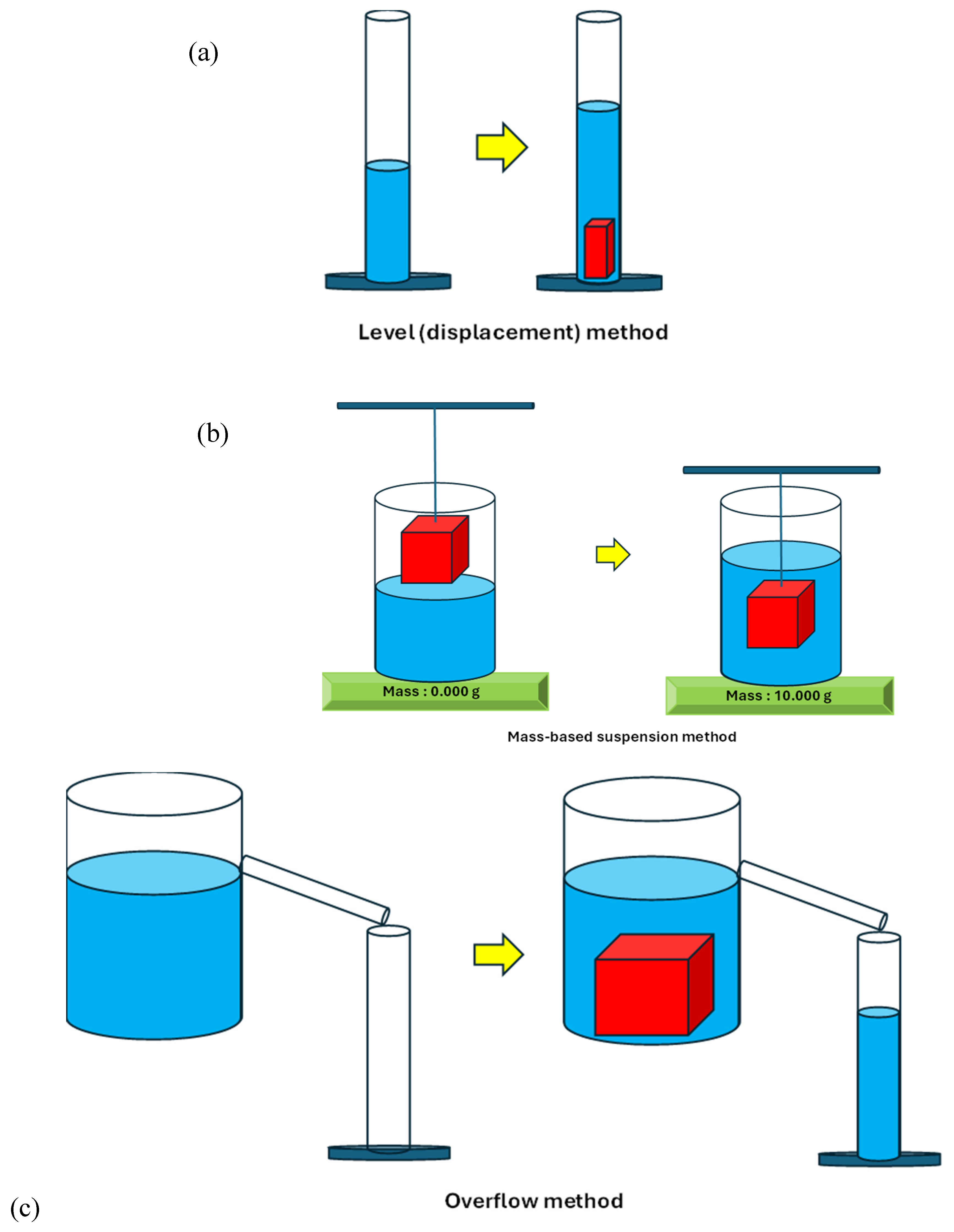
3. Results and Discussion
3.1. Densities of Natural Stones
3.2. Gas Pycnometry – Non-Destructive Density of Solids via Boyle’s Law
3.3. Precision-to-Cost Comparison
- Mass-suspension (this work): ±0.001 g cm⁻³ / $20 ⇒ 5.0 × 10⁻5 precision-units per $.
- AccuPyc II (new): ±0.0001 g cm⁻³ / $18 000 ⇒ 5.6 × 10⁻9 precision-units per $.
3.4. Future Directions in Improving the Measurement of Densities of Natural Stones
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2D | Two dimensional |
| 3D | Three dimensional |
| PP | Polypropylene |
| Micro-CT | Micro computed tomography |
References
- Wahab, G.M.A.; Gouda, M.; Ibrahim, G. Study of physical and mechanical properties for some of Eastern Desert dimension marble and granite utilized in building decoration. Ain Shams Engineering Journal 2019, 10, 907-915. [CrossRef]
- Dong, L.; Zhang, Y.; Wang, L.; Wang, L.; Zhang, S. Temperature dependence of mechanical properties and damage evolution of hot dry rocks under rapid cooling. Journal of Rock Mechanics and Geotechnical Engineering 2024, 16, 645-660. [CrossRef]
- Zhang, W.; Xu, C.; Geng, J. Changes in physical and mechanical properties of limestone and marble after exposure to different high temperatures. Quarterly Journal of Engineering Geology and Hydrogeology 2020, 53, 378-385. [CrossRef]
- Cuccuru, S.; Casini, L.; Oggiano, G.; Cherchi, G.P. Can weathering improve the toughness of a fractured rock? A case study using the San Giacomo granite. Bulletin of Engineering Geology and the Environment 2012, 71, 557-567. [CrossRef]
- Yuan, G.; Cao, Y.; Schulz, H.-M.; Hao, F.; Gluyas, J.; Liu, K.; Yang, T.; Wang, Y.; Xi, K.; Li, F. A review of feldspar alteration and its geological significance in sedimentary basins: From shallow aquifers to deep hydrocarbon reservoirs. Earth-Science Reviews 2019, 191, 114-140. [CrossRef]
- Ernst, W.G. Crystal Chemistry of the Amphiboles. In Amphiboles: Crystal Chemistry Phase Relations and Occurrence, Ernst, W.G., Ed.; Springer Berlin Heidelberg: Berlin, Heidelberg, 1968; pp. 4-18.
- Gardner, J.E.; Llewellin, E.W.; Watkins, J.M.; Befus, K.S. Formation of obsidian pyroclasts by sintering of ash particles in the volcanic conduit. Earth and Planetary Science Letters 2017, 459, 252-263. [CrossRef]
- Sanders, J.V. Colour of Precious Opal. Nature 1964, 204, 1151-1153. [CrossRef]
- Philpotts, J.A.; Pinson, W.H. New data on the chemical composition and origin of moldavites. Geochimica et Cosmochimica Acta 1966, 30, 253-266. [CrossRef]
- Bragg, W.H.; Bragg, W.L. The reflection of X-rays by crystals. Proceedings of the Royal Society of London. Series A, Containing Papers of a Mathematical and Physical Character 1997, 88, 428-438. [CrossRef]
- Bragg, W.L.; Bragg, W.H. The analysis of crystals by the X-ray spectrometer. Proceedings of the Royal Society of London. Series A, Containing Papers of a Mathematical and Physical Character 1997, 89, 468-489. [CrossRef]
- Perutz, M.F. Crystals and X-Rays. Kathleen Lonsdale D.Sc., F.R.S., London: G. Bell & Sons, 1948, 190, pages. 21s. Journal of Glaciology 1949, 1, 345-345. [CrossRef]
- Sanjuan-Szklarz, W.F.; Woinska, M.; Domagala, S.; Dominiak, P.M.; Grabowsky, S.; Jayatilaka, D.; Gutmann, M.; Wozniak, K. On the accuracy and precision of X-ray and neutron diffraction results as a function of resolution and the electron density model. IUCrJ 2020, 7, 920-933, doi:doi:10.1107/S2052252520010441.
- Nebel, S.; Beege, M.; Schneider, S.; Rey, G.D. A Review of Photogrammetry and Photorealistic 3D Models in Education From a Psychological Perspective. Frontiers in Education 2020, Volume 5 - 2020. [CrossRef]
- Wang, Y.; Mingjing, D.; Qingping, Z.; Xiaodong, Z.; and Qu, Z. Volume calculation methods of irregular stone artifacts based on 3D laser scanning technology. Journal of Asian Architecture and Building Engineering 2023, 22, 3386-3402. [CrossRef]
- Reedy, C.L.; Reedy, C.L. High-resolution micro-CT with 3D image analysis for porosity characterization of historic bricks. Heritage Science 2022, 10, 83. [CrossRef]
- Robie, R.A.; Bethke, P.M.; Beardsley, K.M. Quartz (entry). Selected X ray Crystallographic Data, Molar Volumes and Densities of Minerals and Related Substances. U.S. Geological Survey Bulletin 1967, 1248, 10-12.
- Mineralogical Society of America. Handbook of Gemstones, 5th ed.; Mineralogical Society of America: Chantilly, VA. Available online: Available from: https://msaweb.org (accessed on 21/7/2025).
- Deer, W.A.; Howie, R.A.; Zussman, J. Rock Forming Minerals, Volume 2B: Single Chain Silicates, 2nd ed.; The Geological Society: London; 1997; Volume 2B, p. 828.
- Gardner, J.E.; Llewellin, E.W.; Watkins, J.M.; Befus, K.S. Formation of obsidian pyroclasts by sintering of ash particles in the volcanic conduit. Earth and Planetary Science Letters 2017, 459, 252-263. [CrossRef]
- Robie, R.A.; Bethke, P.M.; Beardsley, K.M. Malachite (entry). Selected X ray Crystallographic Data, Molar Volumes and Densities of Minerals and Related Substances,. U.S. Geological Survey Bulletin 1967, 1248, 54-55.
- Ernst, W.G. Crystal Chemistry of the Amphiboles. In Amphiboles: Crystal Chemistry, Phase Relations and Occurrence. 1968, 4-18. [CrossRef]
- Hurlbut, C.S.; Kammerling, R.C. Gemology, 3 rd ed; John Wiley & Sons: New York, 1991; p. 370.
- Sanders, J.V. Colour of Precious Opal. Nature 1964, 204, 1151-1153. [CrossRef]
- Robie, R.A.; Bethke, P.M.; Beardsley, K.M. Beryl (entry). Selected X ray Crystallographic Data, Molar Volumes and Densities of Minerals and Related Substances. U.S. Geological Survey Bulletin 1967, 1248, 88-89.
- Robie, R.A.; Bethke, P.M.; Beardsley, K.M. Fuchsite (entry). Selected X ray Crystallographic Data, Molar Volumes and Densities of Minerals and Related Substances. U.S. Geological Survey Bulletin 1967, 1248, 137.
- Colour Glass Co., Ltd. Opalite Glass – Technical Data Sheet; Manufacturer’s datasheet Available online: https://www.colourglass.com/resources (accessed on 21/7/2025).
- Wang, Y.; Du, M.; Zhang, Q.; Zhao, X.; Qu, Z. Volume calculation methods of irregular stone artifacts based on 3D laser scanning technology. Journal of Asian Architecture and Building Engineering 2023, 22, 3386-3402.
- Reedy, C.L.; Reedy, C.L. High resolution micro CT with 3D image analysis for porosity characterization of historic bricks. Heritage Science 2022, 10, 83. [CrossRef]
- Anderson, K.E. Handbook of Amber Inclusions; Baltic Amber Institute: Gdańsk; 2019; p. 382.
- Robie, R.A.; Bethke, P.M.; Beardsley, K.M. Fluorite (entry). Selected X ray Crystallographic Data, Molar Volumes and Densities of Minerals and Related Substances. U.S. Geological Survey Bulletin 1967, 1248, 274-276.
- Rush, S.A.; Maddox, T.; Fisk, A.T.; Woodrey, M.S.; Cooper, R.J. A precise water displacement method for estimating egg volume. Journal of Field Ornithology 2009, 80, 193-197. [CrossRef]
- Stephen, W.H. Archimedes revisited: a faster, better, cheaper method of accurately measuring the volume of small objects. Physics Education 2005, 40, 468. [CrossRef]
- Gardner, J.E.; Llewellin, E.W.; Watkins, J.M.; Befus, K.S. Formation of obsidian pyroclasts by sintering of ash particles in the volcanic conduit. Earth and Planetary Science Letters. 2017, 459, 252-263. [CrossRef]
- https://www.gemsociety.org/article/select-gems-ordered-density/. Available online: (accessed on 21/4/2025).
- Rescience Inc. Micromeritics AccuPyc II 1340 Gas Pycnometer – Used Listing. Rescience Marketplace 2024. Available online: https://www.rescienceinc.com/product-page/micrometrics-accupyc-ii-1340-gas-pycnometer (accessed on 20/7/2025).
- EquipX. Micromeritics AccuPyc II 1340 Gas Pycnometer – Refurbished. EquipX Equipment Listing 2024. https://www.equipx.net/micromeritics-accupyc-ii-1340-gas-pycnometer.html. Available online: (accessed on 20/7/2025).
- Alibaba.com. Bench Top Helium Gas Pycnometer (aggregated supplier listings). Alibaba Marketplace 2024. Available online: https://www.alibaba.com/showroom/gas-pycnometer.html (accessed on 20/7/2025).
- Martin Graf-Utzmann, Róisín Murtagh (Editors), Gravimetric Analysis: Determining the Density of a Sample for Various Applications, Wiley Analytical Science, 1-16.
- Franzini, M.; Lezzerini, M.; A mercury-displacement method for stone bulk-density determinations. European Journal of Mineralogy. 2003, 15,225–229.

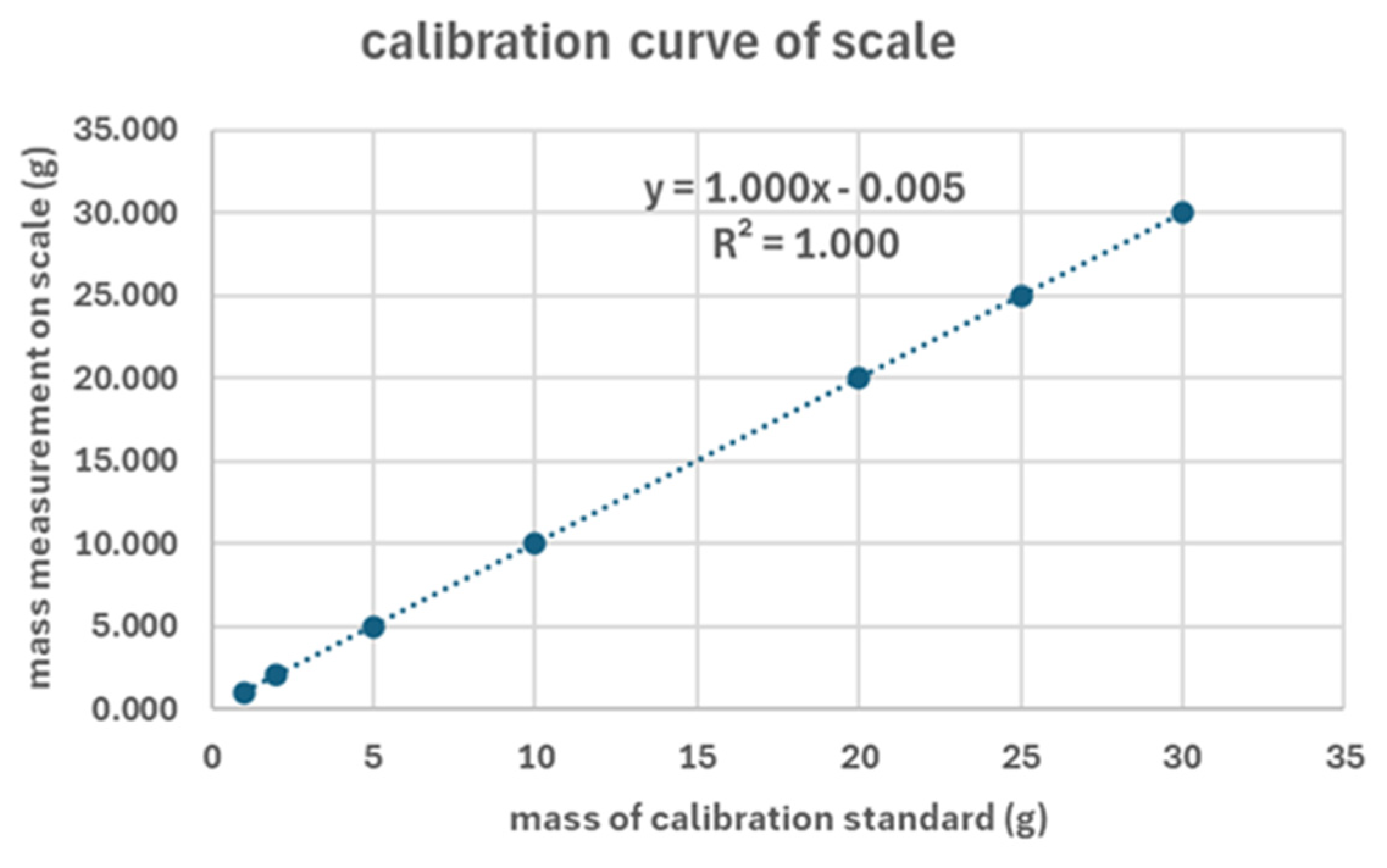




| mass of calib. stand. | Mass measurement on scale | ||||
|---|---|---|---|---|---|
| measur. 1 | measur. 2 | measur. 3 | Mean Value | Stand. Dev. | |
| (g) | (g) | (g) | (g) | (g) | (g) |
| 1 | 0.996 | 0.995 | 0.996 | 0.996 | 0.001 |
| 2 | 1.996 | 1.995 | 1.993 | 1.995 | 0.002 |
| 5 | 5.000 | 5.000 | 5.000 | 5.000 | 0.000 |
| 10 | 9.995 | 9.994 | 9.993 | 9.994 | 0.001 |
| 20 | 20.000 | 20.000 | 20.000 | 20.000 | 0.000 |
| 25 | 25.006 | 25.006 | 25.006 | 25.006 | 0.000 |
| 30 | 30.000 | 30.000 | 30.000 | 30.000 | 0.000 |
| * the mass of 25g was 20g standard plus 5g standard and the mass of 30g was the 20g standard plus the 10g standard | |||||
| Label | Name (common) | Correct mineral name | Dominant formula | Key citation |
|---|---|---|---|---|
| a | Carnelian | Chalcedony (var. of Quartz) | SiO₂ (with Fe³⁺ inclusions) | USGS Quartz entry [17] |
| b | Clear quartz | α-Quartz | SiO₂ | [17] |
| c | Blue banded agate | Blue lace agate (Chalcedony) | SiO₂ + Al-Fe-Mn oxides | [18] |
| d | Nummite | Amphibole mix (Anthophyllite-Gedrite) | (Mg,Fe)₇Si₈O₂₂(OH)₂ | [19] |
| e | Snowflake obsidian | Volcanic glass with cristobalite spherulites | Amorphous SiO₂ + Al₂O₃ + FeO | [20] |
| f | Malachite | Malachite | Cu₂CO₃(OH)₂ | USGS Malachite [21] |
| g | Rose quartz | Rose Quartz | SiO₂ (Ti-Fe-Mn impurities) | [17] |
| h | Nephrite jade | Actinolite-Tremolite solid solution | Ca₂(Mg,Fe)₅Si₈O₂₂(OH)₂ | [22] |
| i | Petrified wood | Silicified wood (Quartz + minor calcite) | SiO₂ ± CaCO₃ | [7] |
| j | Brecciated jasper | Jasper (opaque Quartz) | SiO₂ with Fe-oxide cement | [18] |
| k | Dalmatian jasper | Igneous rhyolite (spotty quartz-feldspar) | (Na,K)AlSi₃O₈ + SiO₂ | [23] |
| l | Turritella agate | Fossiliferous Chalcedony | SiO₂ + CaCO₃ shells | [24] |
| m | Aquamarine | Beryl | Be₃Al₂Si₆O₁₈ | USGS Beryl [25] |
| n | Green aventurine | Quartz with fuchsite mica | SiO₂ + K(Al,Cr)₂(Si₃AlO₁₀)(OH)₂ | [26] |
| o | Pink agate | Dyed Chalcedony | SiO₂ | [18] |
| p | Brecciated jasper | seej | SiO₂ | [18] |
| q | Opalite (man-made) | Opalite glass | SiO₂·nH₂O (amorphous) | [27] |
| r | Yellow ocean jasper | Orbicular Chalcedony | SiO₂ + Fe-oxides | [18] |
| s | Fuchsite | Fuchsite | K(Al,Cr)₂(Si₃AlO₁₀)(OH)₂ | [26] |
| t | Orange moonstone | Orthoclase feldspar | (Na,K)AlSi₃O₈ | [28] |
| u | Shiva lingam | Crypto-crystalline Quartz/Hematite | SiO₂ + Fe₂O₃ | [29] |
| v | Amber | Fossilised tree resin | (C₁₀H₁₆O)ₙ | [30] |
| w | White quartz | α-Quartz | SiO₂ | [17] |
| x | Yellow fluorite | Fluorite | CaF₂ | USGS Fluorite [31] |
| y | Green fluorite | Fluorite | CaF₂ | [31] |
| z | Blue fluorite | Fluorite | CaF₂ | [31] |
| aa | Red jasper | Hematitic Jasper | SiO₂ + Fe₂O₃ | [18] |
| Label | m1 | m2 | m3 | av. m | σ | Vstone | mstone | ρ1 | ρ2 | ρ3 | % err. | ρlit | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (g) | (g) | (g) | (g) | (g) | (cm3)4 | (g) | (g/cm3)1 | (g/cm3)2 | (g/cm3)5 | (g/cm3) | ||||
| a | 5.227 | 5.225 | 5.224 | 5.220 | 0.002 | 5.230 | 13.684 | 2.27 | 2.61 | 2.6123 | 0.0 | 2.64 [17] | ||
| b | 7.033 | 7.036 | 7.032 | 7.029 | 0.002 | 7.041 | 15.519 | 2.38 | 2.20 | 2.1952 | 0.3 | 2.648 [17] | ||
| c | 5.619 | 5.618 | 5.620 | 5.614 | 0.001 | 5.624 | 14.603 | 2.42 | 2.59 | 2.5889 | 0.1 | 2.60 [18] | ||
| d | 5.793 | 5.794 | 5.795 | 5.789 | 0.001 | 5.799 | 13.768 | 2.75 | 2.37 | 2.3658 | 0.2 | 3.01(anthophyllite) [19] | ||
| e | 2.820 | 2.821 | 2.821 | 2.816 | 0.001 | 2.821 | 6.617 | 2.20 | 2.34 | 2.3384 | 0.0 | 2.38 ± 0.05 (obsidian) [7] | ||
| f | 7.894 | 7.895 | 7.893 | 7.889 | 0.001 | 7.903 | 30.618 | 3.82 | 3.87 | 3.8641 | 0.1 | 4.03 [21] | ||
| g | 2.486 | 2.484 | 2.485 | 2.480 | 0.001 | 2.484 | 6.591 | 2.63 | 2.64 | 2.6411 | 0.0 | 2.65 [17] | ||
| h | 2.645 | 2.643 | 2.644 | 2.639 | 0.001 | 2.644 | 7.062 | 2.82 | 2.66 | 2.6554 | 0.2 | 2.95 ± 0.05 [22] | ||
| i | 2.462 | 2.463 | 2.462 | 2.457 | 0.001 | 2.462 | 6.557 | 2.62 | 2.65 | 2.6469 | 0.2 | 2.60 [34] | ||
| j | 3.504 | 3.505 | 3.507 | 3.500 | 0.002 | 3.507 | 10.175 | 2.54 | 2.89 | 2.8874 | 0.2 | 2.60 [18] | ||
| k | 3.880 | 3.879 | 3.878 | 3.874 | 0.001 | 3.881 | 10.13 | 2.53 | 2.60 | 2.6043 | 0.0 | 2.58 [23] | ||
| l | 3.537 | 3.536 | 3.535 | 3.531 | 0.001 | 3.537 | 9.206 | 3.06 | 2.60 | 2.5883 | 0.3 | 2.60 [24] | ||
| m | 3.295 | 3.299 | 3.297 | 3.292 | 0.002 | 3.298 | 8.237 | 2.35 | 2.49 | 2.4857 | 0.2 | 2.641 [25] | ||
| n | 4.000 | 4.000 | 4.000 | 3.995 | 0.000 | 4.002 | 12.718 | 3.17 | 3.17 | 3.1667 | 0.1 | 2.65 [26] | ||
| o | 9.076 | 9.075 | 9.074 | 9.070 | 0.001 | 9.086 | 23.537 | 2.93 | 2.59 | 2.5838 | 0.1 | 2.60 [18] | ||
| p | 2.569 | 2.565 | 2.567 | 2.562 | 0.002 | 2.567 | 8.217 | 3.28 | 3.19 | 3.1923 | 0.1 | 2.60 [18] | ||
| q | 4.000 | 3.995 | 3.996 | 3.992 | 0.003 | 3.999 | 9.632 | 2.40 | 2.40 | 2.4006 | 0.1 | 2.20 (glass) [27] | ||
| r | 3.340 | 3.342 | 3.340 | 3.336 | 0.001 | 3.342 | 8.858 | 2.52 | 2.64 | 2.6372 | 0.2 | 2.60 [18] | ||
| s | 4.633 | 4.632 | 4.631 | 4.627 | 0.001 | 4.635 | 12.551 | 2.66 | 2.70 | 2.6962 | 0.2 | 2.88 [26] | ||
| t | 2.208 | 2.207 | 2.208 | 2.203 | 0.001 | 2.207 | 10.483 | 2.75 | 4.73 | 2.6543 | 0.0 | 2.56 [35] | ||
| u | 2.387 | 2.386 | 2.386 | 2.381 | 0.001 | 2.386 | 8.028 | 2.67 | 3.35 | 2.7891 | 0.2 | 2.70 [29] | ||
| v | 1.658 | 1.657 | 1.656 | 1.652 | 0.001 | 1.655 | 1.784 | 0.67 | 1.07 | 1.0799 | 0.8 | 1.05 [30] | ||
| w3 | - | - | - | - | - | 16.1 | 49.406 | 2.90 | 3.07 | 2.6279 | 4.4 | 2.648 [17] | ||
| x | 9.250 | 9.252 | 9.249 | 9.245 | 0.002 | 9.262 | 25.231 | 3.14 | 2.72 | 2.7204 | 0.0 | 3.18 [31] | ||
| y | 8.009 | 8.008 | 8.005 | 8.002 | 0.002 | 8.017 | 21.819 | 2.72 | 2.72 | 2.7142 | 0.2 | 3.18 [31] | ||
| z | 9.743 | 9.741 | 9.740 | 9.736 | 0.002 | 9.754 | 26.413 | 2.63 | 2.71 | 2.7041 | 0.0 | 3.18 [31] | ||
| aa | 1.888 | 1.889 | 1.888 | 1.883 | 0.001 | 1.887 | 5.225 | 2.61 | 2.76 | 2.7625 | 0.3 | 2.60 [18] | ||
| Cu ref. | 16.370 | 16.363 | 16.361 | 16.370 | 0.005 | 16.426 | 146.67 | 0.17 | 8.929 | |||||
| d6 (cm) | 2.542 | 2.540 | 2.541 | 0.001 | 16.406 | 8.940 | ||||||||
| 1 Displacement of water was used to measure the volume of the natural stone using eqn.4. | ||||||||||||||
| 2 Mass-based suspension method employing Archimedes principle. | ||||||||||||||
|
3 This natural stone specimen mass exceeded the measuring range of the jewellery scale so the level method was used instead 4 density of water used to calculate Vstone 0.9982 g⋅cm-3 at 20 °C (UKAS ISO/IEC17025 and ISO Guide 34 certified). 5 density of water at 21.9 °C was 0.9977956 g⋅cm-3 (CRC handbook of physics and chemistry) 18.2 MΩ·cm deionised water by ELGA (Pure lab chorus). 6 the dimensions of the copper reference cube were measured with a 3 d.p. digital caliper (Draper Expert) and the density of deionised water was taken as 0.99656 g cm-3 (300K, 1 bar). The density of 99.95% w/w copper at 300K is 8.930 g cm-3 (https://www.engineeringtoolbox.com/copper-density-specific-heat-thermal-conductivity-vs-temperature-d_2223.html) The mass of the Cu ref. cube was measured with a 2 d.p. accuracy professional digital scale (146.67 g) and was taken as the average of three measurements (146.66 g, 146.68 g and 146.67 g). 7 Percent error of the mass-based suspension method with a 3 d.p. jewellery scale calculated from Eqn 7 |
||||||||||||||
| Model | Sample volume range | Stated precision (g cm⁻³) | Typical price (USD)* |
|---|---|---|---|
| Micromeritics AccuPyc II 1340 | 0.1–135 cm³ | ±0.0001 | 5 k (refurbished)–18 k (new) [36] |
| Anton Paar Ultrapyc 5000 | 0.5–100 cm³ | ±0.0002 | 18–25 k [37] |
| OEM bench-top units (China) | 1–50 cm³ | ±0.001–0.002 | 1–3 k [38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




