1. Introduction
Inherited cardiac channelopathies represent a heterogeneous group of rare disorders characterized by variable clinical expression, age of onset, and risk of life-threatening arrhythmias. In the pediatric population, the diagnostic and prognostic role of genetic testing is increasingly recognized, especially in the context of diseases such as long QT syndrome (LQTS), catecholaminergic polymorphic ventricular tachycardia (CPVT), and Brugada syndrome (BrS). These conditions may manifest in early life or remain silent until adolescence or adulthood, making timely diagnosis essential to prevent sudden cardiac death [
1,
2,
3].
Pediatric-onset cardiac channelopathies are responsible for arrhythmias that can, in some cases, lead to sudden death. Although the genetic basis of these diseases is well established, the complete underlying genetic architecture remains only partially understood [
4]. Genetic testing plays a pivotal role in defining etiology, establishing prognosis, guiding therapeutic choices, and identifying at-risk family members. Early diagnosis and proper risk stratification are particularly relevant in asymptomatic individuals with a known familial variants [
4].
In minors, genetic testing is generally performed in the presence of clinical signs suggesting an inherited arrhythmic syndrome, or when a pathogenic variant has been identified in a family member. According to current guidelines, predictive testing in asymptomatic children should be carefully considered in light of the potential psychological implications, the penetrance and expressivity of the disease, and the availability of preventive measures or effective treatments [
3,
5]. For some channelopathies, such as CPVT or LQTS, early identification of genetically at-risk individuals allows for the implementation of life-saving therapies [
3].
In the past two decades, numerous gene variants have been associated with inherited cardiac arrhythmias. However, genotype–phenotype correlation remains challenging in many cases. Overlapping genetic substrates may be found across distinct arrhythmias, and conversely, the same clinical phenotype may arise from variants in genes with very different molecular functions [
4]. Due to the rarity of these disorders and their concentration in a limited number of referral centers, studies on pediatric cohorts remain limited, as does high-quality data on genotype–phenotype correlations.
Despite advances in next-generation sequencing (NGS) technologies, the integration of genetic findings into clinical practice still requires careful interpretation and multidisciplinary collaboration. There is a need for more comprehensive and clinically integrated data to guide decision-making in this vulnerable population.
The aim of this study was to genetically characterize a cohort of pediatric patients diagnosed with cardiac channelopathies, using a disease-specific NGS panel. We also investigated the correlation between identified genetic variants and clinical phenotypes. In selected cases, cascade testing in first-degree relatives was performed to assess familial segregation and the potential presence of de novo variants.
2. Materials and Methods
2.1. Genetic Counselling and Sample Collection
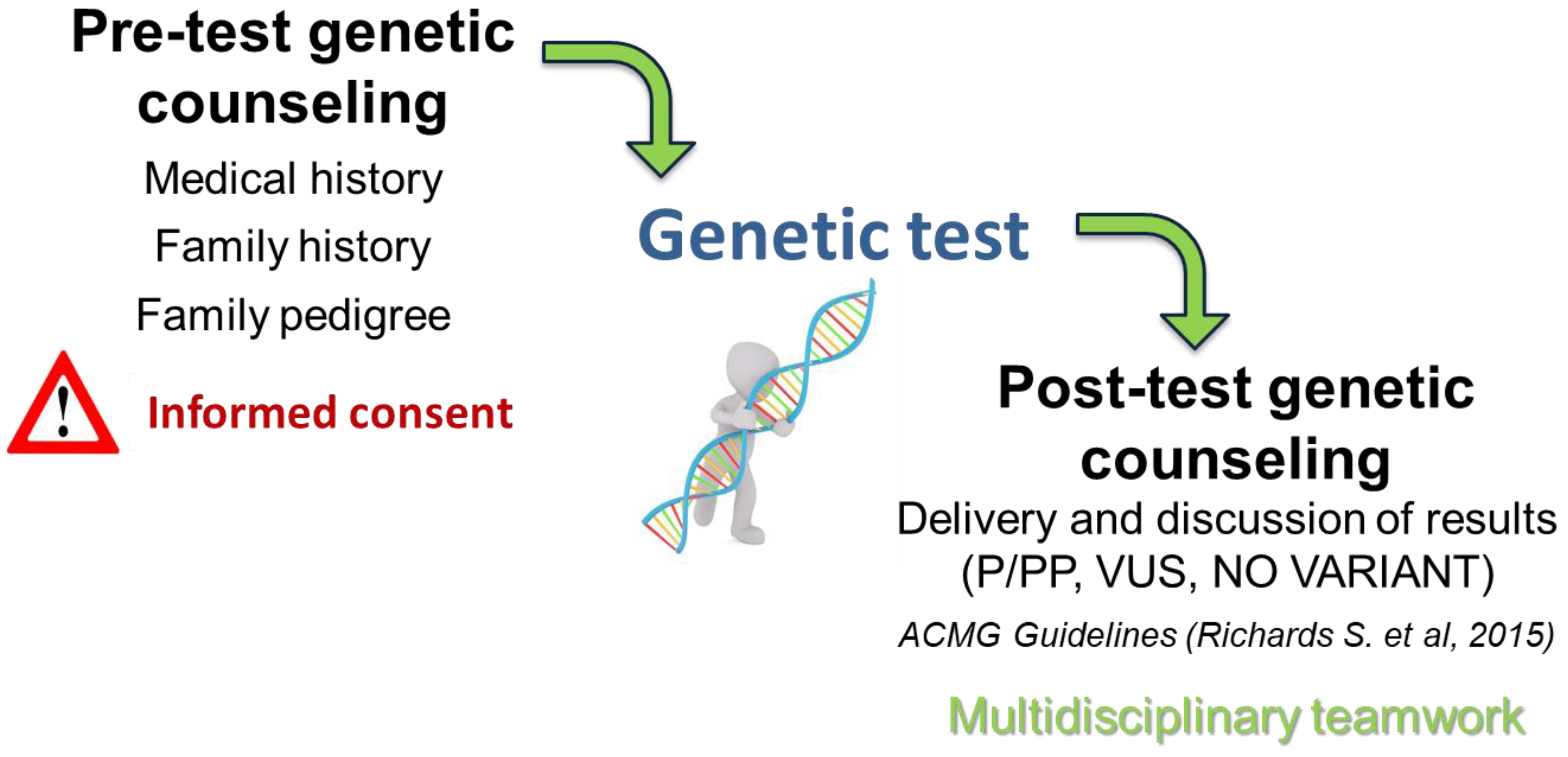
Genetic counselling and molecular analyses were performed as part of a diagnostic workflow approved by the local ethics committee. Written informed consent was obtained from both parents for each underage proband, authorizing genetic testing, sample storage, possible re-analysis in light of emerging gene-disease associations, and anonymized inclusion in national and international research projects (
Figure 1).
A three-generation family pedigree was obtained for each proband during the genetic counselling session to assess family history and identify possible inherited conditions or events such as sudden cardiac death. Peripheral blood samples were collected from the probands and from parents (NGS trio analysis) or other family members.
2.2. DNA Extraction and Quantification
Genomic DNA was extracted from peripheral blood using the EZ1 DNA extraction kit (QIAGEN, Hilden, Germany), based on magnetic bead technology for automated DNA isolation and purification. DNA quantity and purity were assessed via spectrophotometry.
2.3. Genetic Testing
Genetic analysis was performed using a dual approach:
Next-Generation Sequencing (NGS) was carried out using a targeted gene sequencing approach (Paired-End 150 bp) on an Illumina NextSeq550 platform. Library preparation and enrichment were performed using the Illumina DNA Prep with Enrichment protocol and Twist Custom Panel Cardio_V2 probes. The panel comprised 195 genes, of which 29 were specifically analyzed for their association with cardiac channelopathies.
Quality thresholds metrics were defined as >98% of target bases covered at >20X depth and a mean target coverage >200X. Sequencing data were processed using the DRAGEN Enrichment App (BaseSpace Professional, Illumina) and interpreted through BaseSpace Variant Interpreter.
2.4. Variant Filtering and Classification
The analysis has been focused on the coding regions and intron-exon junctions (±15 bp). Synonymous variants and those with a minor allele frequency (MAF) >1% in population databases (GnomAD v4.1.0) were excluded.
Variants were classified according to the American College of Medical Genetics and Genomics (ACMG) and Association for Molecular Pathology (AMP) [
6] guidelines into five classes: (1) pathogenic (P), (2) probably pathogenic (PP), (3) variant of uncertain significance (VUS), or (4) probably benign (PB), (5) benign (B).
2.5. Copy Number Variant Detection (CNVs)
CNVs detection was performed by using the VarSeq tool (Golden Helix) and the EVai (Engenome) tools and confirmed by Multiplex Ligation-dependent Probe Amplification (MLPA-MRC-Holland).
2.6. Parental Testing and De novo Variant Assessment
A variant was considered de novo if absent in both parents, confirmed by Sanger sequencing, and if paternity/maternity was assumed or validated.
3. Results
3.1. NGS Analysis
The analysis of NGS data obtained from the panel of 29 genes associated with inherited cardiac arrhythmias identified, on average, approximately 900 single nucleotide variants (SNVs) or small insertions/deletions per patient.
After an initial functional filter—retaining only exonic or splice-site variants, including missense, stopgain, stoploss, and SNVs—around 820 variants were excluded per sample, resulting in ~70-100 variants per patient selected for further evaluation.
This step reduced the variant count to ~20-30 per patient.
Each variant was individually interpreted through databases and tools previously described.
After reads mapping, 99.4% of target bases were covered at >20X depth and with a mean target coverage of 450X.
3.2. Identified Variants
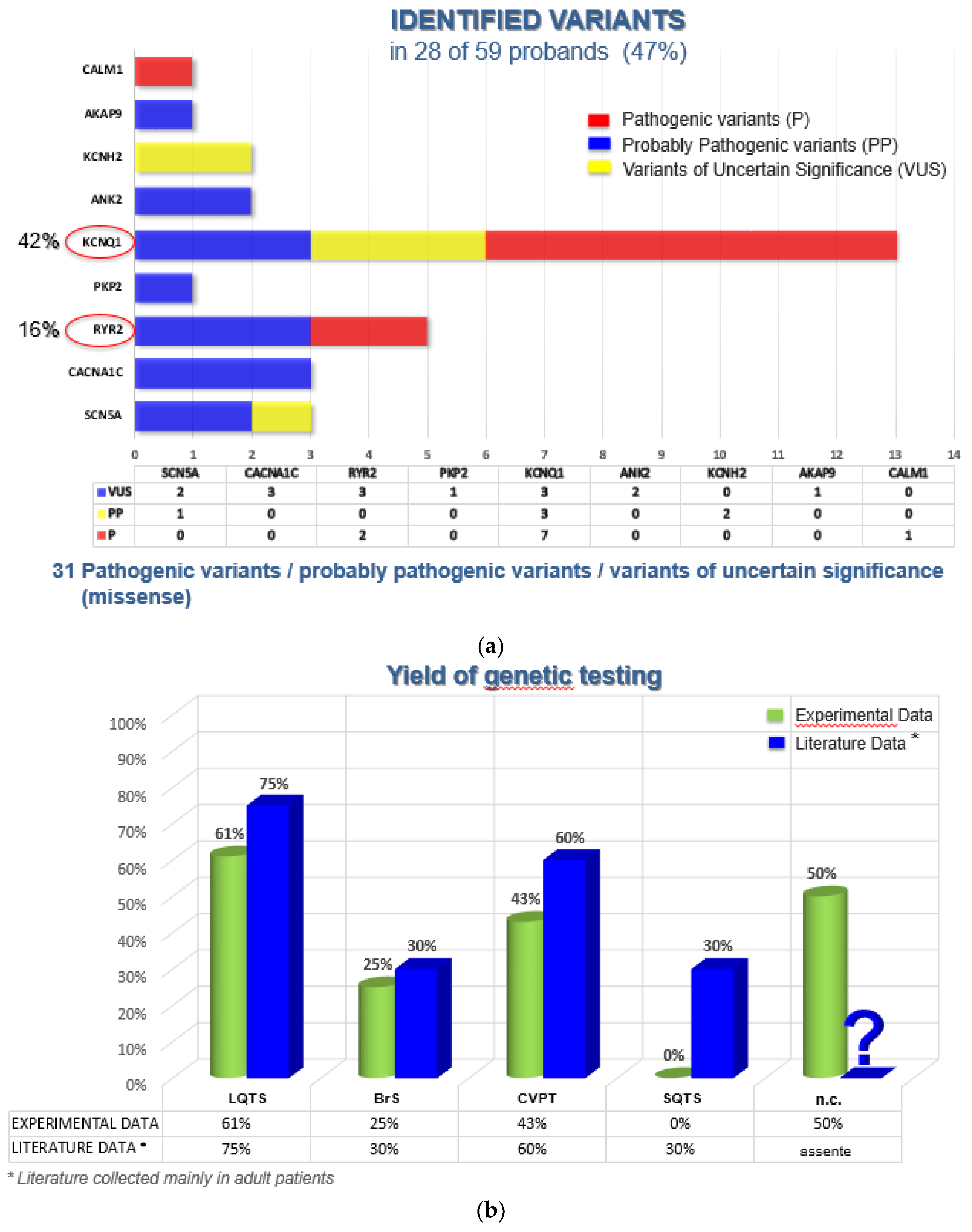
A causative genetic variant was found in 47% of cases, mainly in KCNQ1 (n=13; 42%) associated with Long QT Syndrome (LQTS), RYR2 (n=5; 16%) with Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT), CACNA1C (n=3; 10%) with LQTS and SCN5A (n=3; 10%) with Brugada syndrome (BrS).
3.2.1. Brugada Syndrome (BrS)
A variant (PP or VUS) was identified in 3 out of 12
BrS probands (25%) (
Table 1). Two probands carried three VUSs (n=1 in
CACNA1C and n= 2 in
SCN5A), and one carried a probably pathogenic
SCN5A variant.
Consistent with the literature,
SCN5A emerged as the most involved gene in BrS, accounting for three out of four identified variants [
7]. Two were missense variants (resulting in amino acid substitution), one affected a splice site, and one was a two-base deletion causing a frameshift and premature stop codon.
3.2.2. Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT)
Variants (P or VUS) were identified in 3 out of 7
CPVT probands (43%), all affecting the
RYR2 gene (
Table 1). In two cases, the same known pathogenic missense variant (arginine to glutamine substitution at a conserved position) was detected [
8]; the third variant was a VUS classified as "high VUS". These results are in line with previous reports highlighting the central role of
RYR2 in CPVT [
9].
3.2.3. Long QT Syndrome (LQTS)
A variant (P, LP, or VUS) was identified in 19 out of 31
LQTS probands (61%). Among them, 7 carried VUSs (
AKAP9,
CACNA1C,
ANK2,
KCNQ1), while 12 probands carried 13 P/PP variants (
CALM1,
KCNH2,
KCNQ1).
KCNQ1 was the most frequently affected gene (13/20 variants), as previously described [
10] (
Table 1).
Variants included 11 missense changes, 3 deletions, 5 intronic variants, and 1 frameshift duplication. Notably, a known missense pathogenic variant in
KCNQ1 was supported by functional studies [
11].
An in-frame deletion in
CACNA1C (VUS), a two-nucleotide frameshift deletion in
KCNQ1 (pathogenic) [
12], and a three-exon heterozygous deletion in
KCNH2 (probably pathogenic) were also identified.
Intronic variants included one probably pathogenic splice variant in
KCNQ1 with
in silico support from multiple splicing prediction tools [
13].
3.2.4. Short QT Syndrome (SQTS)
No variants were identified in the 3 SQTS probands.
3.2.5. Conduction Disorders / Unclassified Arrhythmias
A VUS was identified in 3 out of 6 probands with unclassified arrhythmias (50%). Variants affected the
RYR2 (n=2),
CACNA1C (n=1), and
PKP2 (n=1) genes. Among these variants, two were missense, one was an in-frame deletion (
RYR2), and one was a
delins (
PKP2). The
CACNA1C variant was considered a “high VUS” with a Gly-to-Arg substitution (
Table 1).
3.3. Family Analysis
Cascade testing was performed in 22 out of 59 probands (CPVT, LQTS, BrS, and unclassified arrhythmias), extending the analysis to 40 first-degree relatives. Approximately two-thirds of index cases had a positive family history of inherited arrhythmias.
Determining whether a variant is inherited, or de novo is critical for risk assessment and contributes to pathogenicity classification.
Among relatives, variants were identified in 3/40 for CPVT, 14/40 for LQTS, 3/40 for BrS, and 2/40 for conduction disorders/unclassified arrhythmias. The associated genes were SCN5A (n=2), RYR2 (n=3), PKP2 (n=1), KCNQ1 (n=11), KCNH2 (n=1), and ANK2 (n=1).
3.4. Relevant Pediatric Clinical Cases
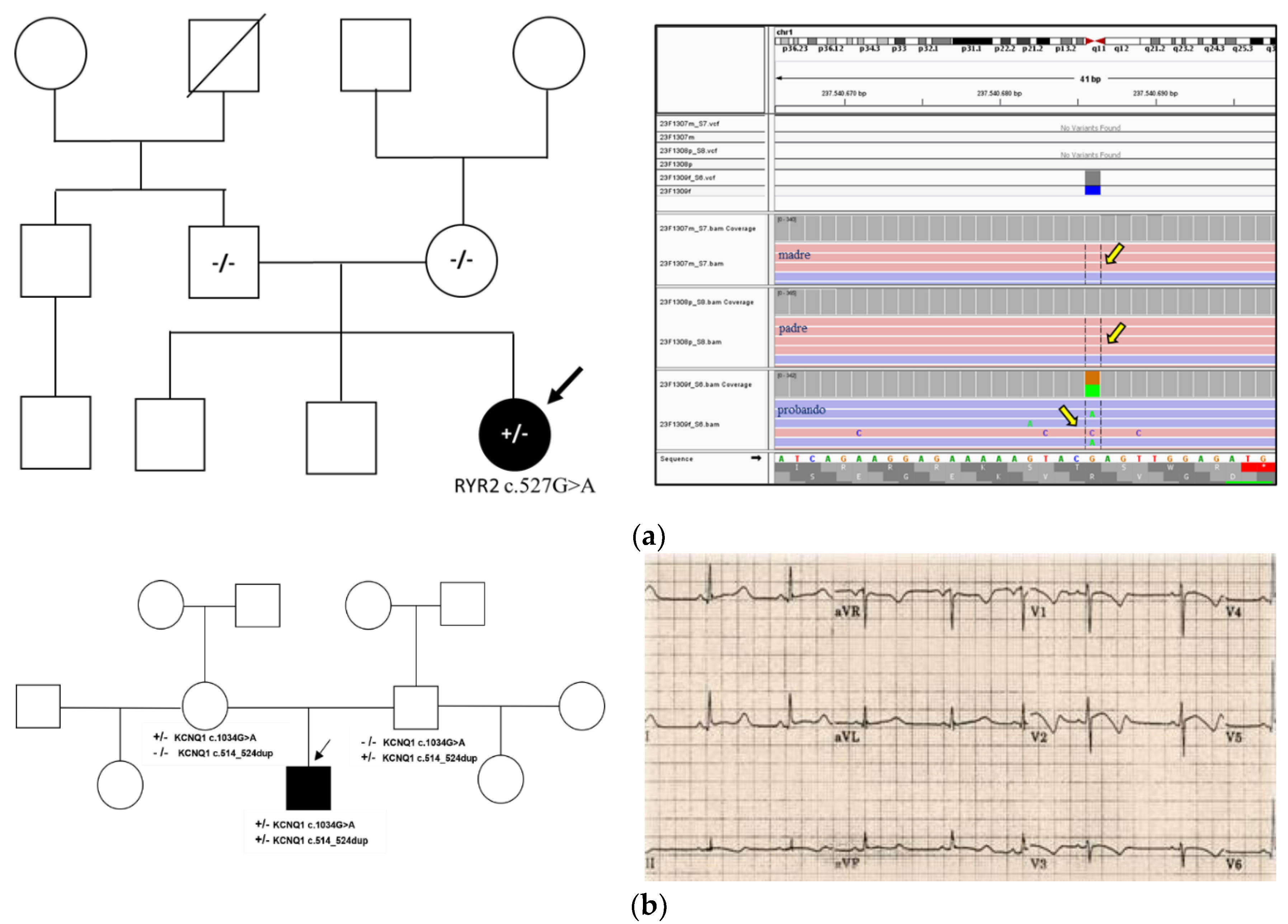
3.4.1. Case 1 – Catecholaminergic Polymorphic Ventricular Tachycardia (RYR2)
A 9-year-old Italian child was referred to our center by the Sports Medicine Department for evaluation of suspected catecholaminergic polymorphic ventricular tachycardia (CPVT) (
Figure 2a). Her family history was negative for cardiac channelopathies. NGS analysis identified a heterozygous variant in the RYR2 gene: c.527G>A p.(Arg176Gln) (
Table 1). This variant has been described as pathogenic and associated with CPVT [
14]. It is absent from GnomAD (v4.1.0) and reported as pathogenic in HGMD Professional (CM010421) and ClinVar (ID: 201194). In silico tools predicted a damaging effect. A different pathogenic variant at the same amino acid residue (p.Arg176Leu) has been previously reported. Parental testing confirmed the variant was
de novo, meeting a strong criterion for pathogenicity. Her siblings are not carriers and do not require further follow-up.
3.4.2. Case 2 – Jervell and Lange-Nielsen Syndrome (KCNQ1)
An 8-year-old child of Brazilian origin, born to non-consanguineous parents, was referred for evaluation of bilateral sensorineural hearing loss, mild intellectual disability, and QT interval prolongation (
Figure 2b). There was no reported family history of inherited channelopathies.
NGS-based genetic testing revealed two heterozygous pathogenic variants in the KCNQ1 gene: c.1034G>A p.(Gly345Glu) and c.514_524dup p.(Gly179Serfs*62) (
Table 1).
Parental testing showed that the variants were inherited from the mother and father, respectively, confirming compound heterozygosity and a diagnosis of Jervell and Lange-Nielsen syndrome (JLNS). Both variants have been previously associated with JLNS and isolated long QT syndrome (LQTS) [
12,
13,
14,
15].
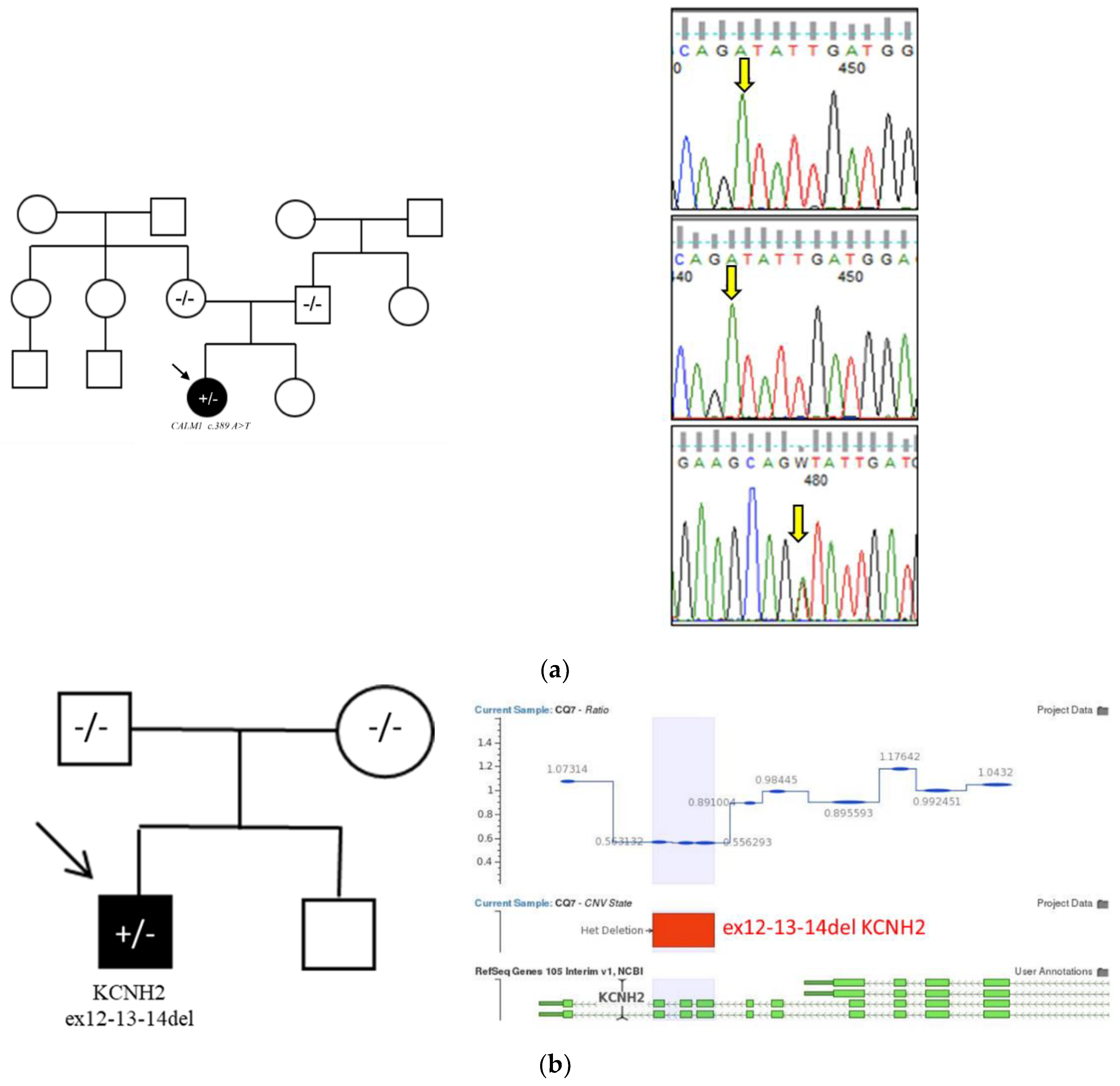
3.4.3. Case 3 – Long QT Syndrome and Calmodulinopathy (CALM1)
A 15-year-old girl, born to non-consanguineous Italian parents, experienced a cardiac arrest at 9 months of age following an intense auditory stimulus (pneumatic drill). She was resuscitated and admitted to hospital, where prolonged QT interval was documented. Initial genetic testing was negative, and beta-blocker therapy was started.
At age 10, an ICD was implanted. Two years later, an appropriate shock was delivered following torsades de pointes. In 2020, with the inclusion of
CALM1 in LQTS panels [
16], repeat genetic testing identified a heterozygous variant in
CALM1 (c.389 A>T p.(Asp130Val) (
Table 1,
Figure 3a).
The variant was not present in either parent, supporting a
de novo origin. The patient was included in the International Calmodulinopathy Registry [
17]. At her latest cardiology visit, the patient was asymptomatic, with a QTc of 592 ms on ECG and normal biventricular function on echocardiography. She remains on beta-blockers with biannual follow-up.
3.4.4. Case 4 – Sudden Cardiac Death (KCNH2 CNV)
A 12-year-old boy died suddenly during sleep, likely triggered by an auditory stressor. A post-mortem cardiogenetic evaluation was initiated. Initial analysis of single nucleotide variants (SNVs) using an arrhythmia gene panel was negative (
Figure 3b).
Subsequent CNV analysis using the VarSeq bioinformatics pipeline (Golden Helix) identified a heterozygous deletion encompassing exons 12–14 of the
KCNH2 gene (ex12-13-14del) (
Table 1).
The deletion was confirmed by MLPA. Parental testing did not detect the variant, indicating a de novo origin and supporting its classification as a pathogenic variant with strong ACMG evidence.
4. Discussion
Inherited arrhythmia syndromes represent a heterogeneous group of disorders in terms of diagnostic approach, genotyping, clinical manifestations, and prognosis. Although most of these syndromes follow an autosomal dominant inheritance pattern, they may exhibit incomplete penetrance, variable expressivity, and overlapping phenotypes [
18]. Given this complexity, molecular genetic testing using Next Generation Sequencing (NGS) technologies has become an essential diagnostic tool.
Currently, most clinical genetic tests are phenotype-driven, yet for accurate diagnosis, molecular investigation is crucial. In Long QT Syndrome (LQTS), for instance, genetic testing can be part of the diagnostic criteria, and understanding the underlying genetic etiology is critical for guiding both prognosis and treatment. Patients with syndromes such as Jervell and Lange-Nielsen or Timothy Syndrome (LQTS-8) tend to have a more severe clinical course, whereas in LQTS-1, the arrhythmic risk partly depends on the specific channel region affected by the mutation. In Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT), the diagnostic yield of genetic testing in suspected cases is moderately high. The identification of affected first-degree relatives is particularly important due to the high penetrance and lethality of the condition. Different pathogenic variants in the RYR2 gene have been associated with variable levels of clinical risk.
Brugada Syndrome (BrS), on the other hand, is clinically challenging to diagnose, and the diagnostic utility of genetic testing remains limited. In clinically diagnosed BrS patients, the genetic diagnostic yield is approximately 20–30%, with the majority of pathogenic variants found in
SCN5A, a gene associated with multiple arrhythmia syndromes due to its pleiotropic effects [
1].
Genetic testing is therefore progressively transforming the management of inherited cardiac diseases, owing to the unique potential of NGS technologies to identify causative variants in potentially lethal conditions and enable cascade screening of at-risk family members [
19]. Furthermore, NGS allows for the simultaneous analysis of multiple genes, facilitating a more comprehensive characterization of patients with polygenic cardiac disorders.
In our cohort, the use of a custom-designed 29-gene panel covering genes associated with LQTS, Short QT Syndrome (SQTS), CPVT, BrS, and conduction or unclassified arrhythmias provided enhanced diagnostic resolution compared to traditional Sanger sequencing, which was nonetheless employed for variant confirmation and familial testing. Among 59 index patients with channelopathies or conduction disorders, the overall diagnostic yield was 47% (28/59), considering both pathogenic/ probably pathogenic variants and variants of uncertain significance (VUS) (
Figure 4a).
We included VUS in the yield calculation because some VUS—particularly high-VUS—may contribute to the disease phenotype. Moreover, segregation analysis in families can help establish or refute a pathogenic correlation [
20]. It is also important to emphasize that a negative genetic test alone cannot exclude the diagnosis in an index case [
1].
Among BrS patients, our findings were consistent with those reported in the literature: 25% tested positive by genetic screening (including 75% VUS), compared to ~30% in recent studies [
7]. As expected,
SCN5A was the most frequently mutated gene in our BrS patients. Only one of the four variants identified in
SCN5A was classified as probably pathogenic, while the others (two in
SCN5A and one in
CACNA1C) were VUS. This aligns with current literature, which reports that approximately 75% of BrS-associated variants involve
SCN5A, and 25% involve other genes.
For CPVT, our data showed a genetic diagnostic yield of 43% (including 33% VUS), slightly below the 50–60% reported in the literature [
9]. This discrepancy may reflect the small sample size. In our series,
RYR2 variants were identified in 100% of genetically positive CPVT cases, in line with previous findings that report gain-of-function variant in
RYR2 in up to 95% of genetically confirmed cases [
21,
22].
Among LQTS patients, genetic testing was positive in 61% of cases (including 35% VUS), compared to the ~75% positivity rate in published data [
23]. The
KCNQ1 gene accounted for the majority of variants in our cohort (65% vs. 35% reported in the literature [
10]), with 13 out of 20 variants affecting this gene. Other identified variants were in
ANK2 (10%, all VUS),
KCNH2 (10%, all probably pathogenic), and
CACNA1C,
AKAP9,
CALM1 (5% each, with variants classified as pathogenic or VUS). These frequencies were notably higher than the ~1% typically attributed to these minor genes in other studies.
In contrast, no pathogenic or probably pathogenic variants were identified in patients diagnosed with SQTS. This finding is consistent with the literature, where the causative variant can only be identified in 20–30% of cases, reflecting the rarity and limited genetic characterization of this condition [
24].
In patients with conduction disorders or unclassified arrhythmias, the diagnostic yield was 50% (3/6), all involving VUS in
RYR2,
PKP2, or
CACNA1C. Without extended family studies, these variants alone are insufficient for a definitive diagnosis of inherited arrhythmia. However, in two cases, family testing confirmed the segregation of the VUS with the disease phenotype. Notably, the
CACNA1C c.1468G>A p.(Gly490Arg) variant was classified as a high-VUS. This variant affects a conserved nucleotide in exon 10 and has previously been reported in a male Brugada patient [
25]. Functional studies demonstrated significantly reduced calcium current (~10% of wild type), although the causal link between this current reduction and arrhythmogenesis remains to be fully elucidated.
Overall, our data partly diverge from published reports, likely due to two main factors: (1) the limited sample size, and (2) the inclusion of patients with high clinical suspicion but without a confirmed diagnosis. Moreover, we included VUS in our statistical analysis, acknowledging that their clinical relevance may change as further data become available through future research and family-based segregation analyses (
Figure 4b).
4.1. Relevant Pediatric Clinical Cases
4.1.1. Case 1 – Catecholaminergic Polymorphic Ventricular Tachycardia (RYR2)
Within this family, the proband is the only individual with a heterozygous variant: the patient presents a de novo variant, classified as pathogenic according to the ACMG guidelines. Given that the variant is considered de novo, other family members are exempt from follow-up, as they are unlikely to carry the variant.
According to the literature,
de novo variants in the
RYR2 gene are commonly found in pediatric patients with Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT). This gene encodes for the cardiac ryanodine receptor (RyR2) and is responsible for calcium release from the sarcoplasmic reticulum into the cytosol. Pathogenic gain-of-function variants in RYR2 lead to an altered RyR2 protein, with increased diastolic cytosolic calcium levels, causing arrhythmic consequences, particularly under adrenergic conditions [
20].
4.1.2. Case 2 – Jervell and Lange-Nielsen Syndrome (KCNQ1)
Within this family, the proband was diagnosed with Long QT Syndrome (LQTS). Genetic testing identified two pathogenic variants in the
KCNQ1 gene, p.(Gly345Glu) and p.(Gly179Serfs*62), both described in the literature as associated with Jervell and Lange-Nielsen Syndrome (JLNS), an autosomal recessive form of LQTS. Based on the genetic test and clinical presentation (the patient exhibits congenital sensorineural hearing loss and mild intellectual disability), the syndromic form of the disease was confirmed and clarified [
20]. Genetic testing was then extended to the parents, revealing the presence of a variant in each, despite normal phenotypes. Specifically, the mother was found to carry the p.(Gly345Glu) variant, and the father carried the p.(Gly179Serfs*62) variant. This result clarified the family’s genetic background: the parents are carriers of different variants in the same gene, both transmitted to the child, who developed the disease. This finding established a recurrence risk of 25% for the couple to have another affected child and a 50% chance of having a child who is a healthy carrier of the condition.
Pathogenic variants in potassium channel genes are responsible for the vast majority of LQTS cases, with
KCNQ1 and
KCNH2 accounting for 80% of all genetically explained cases of LQTS. Pathogenic homozygous or compound heterozygous variants in
KCNQ1 and
KCNE1 cause the recessive form of Jervell and Lange-Nielsen Syndrome (JLNS), where the cardiac phenotype is associated with congenital deafness.
KCNE1 is strongly linked to acquired LQTS (aLQTS), as is
KCNE2, which causes a less common subtype typically associated with low penetrance and a mild phenotype [
20].
4.1.3. Case 3 – Long QT Syndrome and Calmodulinopathy (CALM1)
In this family, the only individual carrying the heterozygous variant is the proband: thus, this variant can be classified as de novo and causative of the disease due to its autosomal dominant inheritance. Her 13-year-old sister is not considered at risk and was excluded from follow-up.
The clinical characteristics of this pathology include its rarity and severity, with the first cardiac event typically occurring around 4 years of age. Adrenergic stimulation serves as a trigger for arrhythmic events in 81% of patients, and major arrhythmic events are seen in 68%. Sudden death is reported in 27% of patients. Of the
de novo variants, 93% are
CALM-LQTS related with a characteristic phenotype: a QTc of 594 + 73 ms, T waves with a late peak onset, perinatal manifestation in 58% of cases, an average age of onset of 1.5 years, and life-threatening arrhythmias in 78% of cases [
17].
4.1.4. Case 4 – Sudden Cardiac Death (KCNH2 CNV)
The genetic analysis of the proband, conducted post-mortem and also including CNV analysis, revealed a deletion affecting exons 12-13-14 of the KCNH2 gene. The variant was absent in the parents, which led to the classification of the CNV as de novo and causative of the fatal pathology. Additionally, other family members, including the proband's brother and any potential future children of the couple, are exempt from follow-up. This case highlights the importance of performing NGS analysis not only for SNVs but also for large deletions-duplications (CNVs) in molecular autopsy.
4.2. Family Studies
Genetic testing proved to be of considerable value in familial contexts, enabling early identification of (likely)pathogenic variants among both symptomatic and asymptomatic relatives. Testing in parents also allowed for the identification of
de novo variants. Phenotypic variability among carriers of the same variant—even within the same family—may be influenced by environmental factors, genetic modifiers, or polymorphisms that affect gene expression [
18]. In our cohort, we analyzed 40 family members from 22 index cases. Of these, 22 individuals (55%) carried the same variant as the proband. Family testing was particularly useful in three cases in which a
de novo variant was identified in the proband and considered causative of the disease.
5. Conclusions
A genetic cause of cardiac channelopathy was identified in 47% of the probands analyzed. The diagnostic yield of genetic testing was highest in cases of Long QT Syndrome (LQTS)—particularly associated with variants in the KCNQ1 gene, responsible for LQTS type 1—and in Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT), with a predominance of RYR2 variants underlying the autosomal dominant form.
Genetic testing has supported clinicians in establishing the etiology and prognosis of channelopathies, particularly in syndromic forms, and has in some cases contributed to therapeutic decision-making (e.g., implantable cardioverter-defibrillator placement) and lifestyle recommendations (e.g., restrictions on competitive sports). Moreover, it has allowed for recurrence risk assessment in the context of future pregnancies, thereby enabling informed reproductive choices through genetic counselling. Cascade screening of family members has also been instrumental in identifying individuals at risk, offering the opportunity for early preventive interventions (e.g., in LQTS cases).
Currently, the causative variant can only be identified in a limited percentage of patients, ranging from 75–80% in LQTS to as low as 20–30% in Brugada Syndrome (BrS). Thus, a negative genetic test does not rule out a clinical diagnosis or the possibility of an underlying genetic cause in genomic regions not yet characterized. Furthermore, genotype–phenotype correlations remain incompletely understood.
In the future, advances in NGS technologies and expanded knowledge of genetic mechanisms are expected to enable the discovery of new disease-causing genes and pathogenic pathways, paving the way for the development of increasingly personalized therapeutic strategies—including the potential for gene-based therapies.
Author Contributions
Conceptualization, S.G., A.B., A.G., I.O. and F.G.; Methodology, S.G., A.B., A.G., A.T., D.M. and F.G.; Software, S.G., A.B., A.G., D.M. and F.G.; Validation, S.G.; Formal analysis, S.G., M.D.L.; Investigation, A.B., A.G., S.P., M.Z., A.M., G.B.C., G.S., G.P., E.B., S.F., I.O. and F.G.; Resources, A.B., A.G., S.P., M.Z., A.M., A.T., G.B.C., G.S., G.P., E.B., S.F., I.O. and F.G.; Data curation, S.G., A.B., A.G., M.D.L., D.M. and F.G.; Writing – original draft, S.G. and M.D.L.; Writing – review and editing, S.G., D.M. and F.G.; Visualization, S.G.; Supervision, S.G. and F.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| NGS |
Next Generation Sequencing |
| LQTS |
Long QT Syndrome |
| CPVT |
Catecholaminergic Polymorphic Ventricular Tachycardia |
| BrS |
Brugada Syndrome |
| DNA |
Deoxyribonucleic Acid |
| DRAGEN |
Dynamic Read Analysis for GENomics (Illumina pipeline) |
| MAF |
Minor Allele Frequency |
| GnomAD |
Genome Aggregation Database |
| HGMD |
Human Gene Mutation Database |
| ACMG |
American College of Medical Genetics |
| AMP |
Association for Molecular Pathology |
| P |
Pathogenic |
| PP |
Probably Pathogenic |
| VUS |
Variant of Uncertain Significance |
| PB |
Probably Benign |
| B |
Benign |
| MLPA |
Multiplex Ligation-dependent Probe Amplification |
| PCR |
Polymerase Chain Reaction |
| SNV |
Single Nucleotide Variant |
| SQTS |
Short QT Syndrome |
| JLNS |
Jervell and Lange-Nielsen syndrome |
| ICD |
Implantable Cardioverter Defibrillator |
| ECG |
electrocardiogram |
| IGV |
Integrative Genomics Viewer |
| CNV |
Copy Number Variants |
References
- Ackerman, M.J.; Priori, S.G.; Willems, S.; Berul, C.; Brugada, R.; Calkins, H.; et al. HRS/EHRA Expert Consensus Statement on the State of Genetic Testing for the Channelopathies and Cardiomyopathies: This document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Europace. 2011 Aug 1;13(8):1077–109. [CrossRef]
- Landstrom, A.P.; Kim, J.J.; Gelb, B.D.; Helm, B.M.; Kannankeril, P.J.; Semsarian, C.; et al. Genetic Testing for Heritable Cardiovascular Diseases in Pediatric Patients: A Scientific Statement From the American Heart Association. Circ: Genomic and Precision Medicine. 2021 Oct;14(5):e000086. [CrossRef]
- Girolami, F.; Vergaro, G.; Pieroni, M.; Passantino, S.; Giannotti, G.; Grippo, G.; Canale, M.L.; Favilli, S.; Cappelli, F.; Olivotto, I.; Casolo, G. Percorso clinico proposto dall’ANMCO Toscana per la diagnosi genetica delle cardiomiopatie in un sistema assistenziale in rete. G Ital Cardiol 2020;21(12):926-934. [CrossRef]
- Baltogiannis, G.; Conte, G.; Sieira, J.; De Ferrari, G.M.; Brugada, P. Editorial: Sudden Cardiac Death and Channelopathies. Front Cardiovasc Med. 2020 Nov 25;7:605834. [CrossRef]
- Committee on Bioethics; Committee on Genetics; the American College of Medical Genetics; Genomics social, ethical, and legal issues committee; et al. Ethical and Policy Issues in Genetic Testing and Screening of Children. Pediatrics. 2013 Mar 1;131(3):620–2.
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genetics in Medicine. 2015 May;17(5):405–24. [CrossRef]
- Cerrone, M.; Costa, S.; Delmar, M. The Genetics of Brugada Syndrome. Annu Rev Genom Hum Genet. 2022 Aug 31;23(1):255–74.
- Haugaa, K.H.; Leren, I.S.; Berge, K.E.; Bathen, J.; Loennechen, J.P.; Anfinsen, O.G.; et al. High prevalence of exercise-induced arrhythmias in catecholaminergic polymorphic ventricular tachycardia mutation-positive family members diagnosed by cascade genetic screening. Europace. 2010 Mar 1;12(3):417–23. [CrossRef]
- Wleklinski, M.J.; Kannankeril, P.J.; Knollmann, B.C. Molecular and tissue mechanisms of catecholaminergic polymorphic ventricular tachycardia. The Journal of Physiology. 2020 Jul;598(14):2817–34. [CrossRef]
- Wallace, E.; Howard, L.; Liu, M.; O’Brien, T.; Ward, D.; Shen, S.; et al. Long QT Syndrome: Genetics and Future Perspective. Pediatr Cardiol. 2019 Oct;40(7):1419–30. [CrossRef]
- Wang, Z.; Tristani-Firouzi, M.; Xu, Q.; Lin, M.; Keating, M.T.; Sanguinetti, M.C. Functional Effects of Mutations in KvLQT1 that Cause Long QT Syndrome. Cardiovasc electrophysiol. 1999 Jun;10(6):817–26. [CrossRef]
- Al-Aama, J.Y.; Al-Ghamdi, S.; Bdier, A.Y.; AlQarawi, A.; Jiman, O.A.; Al-Aama, N.; et al. Genotype–phenotype analysis of Jervell and Lange-Nielsen syndrome in six families from Saudi Arabia. Clinical Genetics. 2015 Jan;87(1):74–9. [CrossRef]
- Vyas, B.; Puri, R.D.; Namboodiri, N.; Nair, M.; Sharma, D.; Movva, S.; et al. KCNQ1 mutations associated with Jervell and Lange–Nielsen syndrome and autosomal recessive Romano–Ward syndrome in India—expanding the spectrum of long QT syndrome type 1. American J of Med Genetics Pt A. 2016 Jun;170(6):1510–9. [CrossRef]
- Walsh, R.; Peters, N.S.; Cook, S.A.; Ware, J.S. Paralogue annotation identifies novel pathogenic variants in patients with Brugada syndrome and catecholaminergic polymorphic ventricular tachycardia. J Med Genet. 2014 Jan;51(1):35–44. [CrossRef]
- Schwartz, P.J.; Moreno, C.; Kotta, M.C.; Pedrazzini, M.; Crotti, L.; Dagradi, F.; et al. Mutation location and I Ks regulation in the arrhythmic risk of long QT syndrome type 1: the importance of the KCNQ1 S6 region. European Heart Journal. 2021 Dec 7;42(46):4743–55. [CrossRef]
- Adler, A.; Novelli, V.; Amin, A.S.; Abiusi, E.; Care, M.; Nannenberg, E.A.; et al. An International, Multicentered, Evidence-Based Reappraisal of Genes Reported to Cause Congenital Long QT Syndrome. Circulation. 2020 Feb 11;141(6):418–28. [CrossRef]
- Crotti, L.; Spazzolini, C.; Tester, D.J.; Ghidoni, A.; Baruteau, A.E.; Beckmann, B.M.; et al. Calmodulin mutations and life-threatening cardiac arrhythmias: insights from the International Calmodulinopathy Registry. European Heart Journal. 2019 Sep 14;40(35):2964–75. [CrossRef]
- Coll, M.; Pérez-Serra, A.; Mates, J.; Del Olmo, B.; Puigmulé, M.; Fernandez-Falgueras, A.; et al. Incomplete Penetrance and Variable Expressivity: Hallmarks in Channelopathies Associated with Sudden Cardiac Death. Biology. 2017 Dec 26;7(1):3. [CrossRef]
- Kalayinia, S.; Goodarzynejad, H.; Maleki, M.; Mahdieh, N. Next generation sequencing applications for cardiovascular disease. Annals of Medicine. 2018 Feb 17;50(2):91–109. [CrossRef]
- Wilde, A.A.M.; Marquez, M.F.; Co-Chair, L.; Shamloo, A.S.; Ackerman, M.J.; Ashley, E.A.; et al. European Heart Rhythm Association (EHRA)/ Heart Rhythm Society (HRS)/Asia Pacific Heart Rhythm Society (APHRS)/Latin American Heart Rhythm Society (LAHRS) Expert Consensus Statement on the state of genetic testing for cardiac diseases.
- Stutzman, M.J.; Kim, C.S.J.; Tester, D.J.; Hamrick, S.K.; Dotzler, S.M.; Giudicessi, J.R.; et al. Characterization of N-terminal RYR2 variants outside CPVT1 hotspot regions using patient iPSCs reveal pathogenesis and therapeutic potential. Stem Cell Reports. 2022 Sep;17(9):2023–36. [CrossRef]
- Pérez-Riera, A.R.; Barbosa-Barros, R.; De Rezende Barbosa, M.P.C.; Daminello-Raimundo, R.; De Lucca, A.A.; De Abreu, L.C. Catecholaminergic polymorphic ventricular tachycardia, an update. Noninvasive Electrocardiol. 2018 Jul;23(4):e12512.
- Giudicessi, J.R.; Wilde, A.A.M.; Ackerman, M.J. The genetic architecture of long QT syndrome: A critical reappraisal. Trends in Cardiovascular Medicine. 2018 Oct;28(7):453–64. [CrossRef]
- Campuzano, O.; Sarquella-Brugada, G.; Cesar, S.; Arbelo, E.; Brugada, J.; Brugada, R. Recent Advances in Short QT Syndrome. Front Cardiovasc Med. 2018 Oct 29;5:149. [CrossRef]
- Antzelevitch, C.; Pollevick, G.D.; Cordeiro, J.M.; Casis, O.; Sanguinetti, M.C.; Aizawa, Y.; et al. Loss-of-Function Mutations in the Cardiac Calcium Channel Underlie a New Clinical Entity Characterized by ST-Segment Elevation, Short QT Intervals, and Sudden Cardiac Death. Circulation. 2007 Jan 30;115(4):442–9.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).